Influence of the Accumulation of Unhealthy Eating Habits on Obesity in a General Japanese Population: The Hisayama Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population
2.2. Definition of Obesity and Central Obesity
2.3. Definition of Unhealthy Eating Habits
2.4. Measurement of Other Risk Factors
2.5. Statistical Analysis
2.6. Ethical Considerations
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- NCD Risk Factor Collaboration. Trends in adult body-mass index in 200 countries from 1975 to 2014: A pooled analysis of 1698 population-based measurement studies with 19.2 million participants. Lancet 2016, 387, 1377–1396. [Google Scholar] [CrossRef]
- WHO; FEC. Diet, Nutrition and the Prevention of Chronic Diseases, WHO Technical Report Series 916; WHO: Geneva, Switzerland, 2003.
- Saito, I.; Kokubo, Y.; Kiyohara, Y.; Doi, Y.; Saitoh, S.; Ohnishi, H.; Miyamoto, Y. Prospective study on waist circumference and risk of all-cause and cardiovascular mortality. Circ. J. 2012, 76, 2867–2874. [Google Scholar] [CrossRef]
- Schneider, H.J.; Friedrich, N.; Klotsche, J.; Pieper, L.; Nauck, M.; John, U.; Dörr, M.; Felix, S.; Lehnert, H.; Pittrow, D.; et al. The predictive value of different measures of obesity for incident cardiovascular events and mortality. J. Clin. Endocrinol. Metab. 2010, 95, 1777–1785. [Google Scholar] [CrossRef]
- Liu, X.; Zheng, C.; Xu, C.; Liu, Q.; Wang, J.; Hong, Y.; Zhao, P. Nighttime snacking is associated with risk of obesity and hyperglycemia in adults: A cross-sectional survey from Chinese adult teachers. J. Biomed. Res. 2017, 31, 541–547. [Google Scholar]
- Murakami, K.; Livingstone, M.B.E. Eating Frequency is positively associated with overweight and central obesity in US adults. J. Nutr. 2015, 145, 2715–2724. [Google Scholar] [CrossRef]
- Mesas, A.E.; Muñoz-Pareja, M.; López-García, E.; Rodríguez-Artalejo, F. Selected eating behaviours and excess body weight: A systematic review. Obes. Rev. 2012, 13, 106–135. [Google Scholar] [CrossRef]
- Bes-Rastrollo, M.; Sanchez-Villegas, A.; Basterra-Gortari, F.J.; Nunez-Cordoba, J.M.; Toledo, E.; Serrano-Martinez, M. Prospective study of self-reported usual snacking and weight gain in a Mediterranean cohort: The SUN project. Clin. Nutr. 2010, 29, 323–330. [Google Scholar] [CrossRef]
- Ohkuma, T.; Hirakawa, Y.; Nakamura, U.; Kiyohara, Y.; Kitazono, T.; Ninomiya, T. Association between eating rate and obesity: A systematic review and meta-analysis. Int. J. Obes. 2015, 39, 1589–1596. [Google Scholar] [CrossRef]
- Ohkuma, T.; Fujii, H.; Iwase, M.; Kikuchi, Y.; Ogata, S.; Idewaki, Y.; Ide, H.; Doi, Y.; Hirakawa, Y.; Mukai, N.; et al. Impact of eating rate on obesity and cardiovascular risk factors according to glucose tolerance status: The Fukuoka Diabetes Registry and the Hisayama Study. Diabetologia 2013, 56, 70–77. [Google Scholar] [CrossRef]
- Yoshida, J.; Eguchi, E.; Nagaoka, K.; Ito, T.; Ogino, K. Association of night eating habits with metabolic syndrome and its components: A longitudinal study. BMC Public Health 2018, 18, 1366. [Google Scholar] [CrossRef]
- Kutsuma, A.; Nakajima, K.; Suwa, K. Potential association between breakfast skipping and concomitant late-night-dinner eating with metabolic syndrome and proteinuria in the Japanese population. Scientifica 2014, 2014, 253581. [Google Scholar] [CrossRef] [PubMed]
- Hata, J.; Ninomiya, T.; Hirakawa, Y.; Nagata, M.; Mukai, N.; Gotoh, S.; Fukuhara, M.; Ikeda, F.; Shikata, K.; Yoshida, D.; et al. Secular trends in cardiovascular disease and its risk factors in Japanese: Half-century data from the Hisayama Study (1961–2009). Circulation 2013, 128, 1198–1205. [Google Scholar] [CrossRef] [PubMed]
- Ninomiya, T. Japanese legacy cohort studies: The Hisayama Study. J. Epidemiol. 2018, 28, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Alberti, K.G.M.M.; Eckel, R.H.; Grundy, S.M.; Zimmet, P.Z.; Cleeman, J.I.; Donato, K.A.; Fruchart, J.; James, W.P.T.; Loria, C.M.; Smith, S.C., Jr. Harmonizing the metabolic syndrome: A joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 2009, 120, 1640–1645. [Google Scholar] [PubMed]
- Ministry of Health, Labour and Welfare, Japan. Standardized Health Check-Up and Intervention Program. 2007. Available online: http://tokutei-kensyu.tsushitahan.jp/manage/wp-content/uploads/2014/05/36ec0bcdf91b61a94a1223627abffe8d.pdf (accessed on 3 September 2020).
- Kimura, Y.; Nanri, A.; Matsushita, Y.; Sasaki, S.; Mizoue, T. Eating behavior in relation to prevalence of overweight among Japanese men. Asia Pac. J. Clin. Nutr. 2011, 20, 29–34. [Google Scholar]
- Lee, J.S.; Mishra, G.; Hayashi, K.; Watanabe, E.; Mori, K.; Kawakubo, K. Combined eating behaviors and overweight: Eating quickly, late evening meals, and skipping breakfast. Eat. Behav. 2016, 21, 84–88. [Google Scholar] [CrossRef] [PubMed]
- Larson, N.I.; Miller, J.M.; Watts, A.W.; Story, M.T.; Neumark-Sztainer, D.R. Adolescent snacking behaviors are associated with dietary intake and weight status. J. Nutr. 2016, 146, 1348–1355. [Google Scholar] [CrossRef] [PubMed]
- de Graaf, C. Effects of snacks on energy intake: An evolutionary perspective. Appetite 2006, 47, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Education, Culture, Sports, Science and Technology-JAPAN. Standard Tables of Food Composition in Japan. 2015. Available online: https://www.mext.go.jp/en/policy/science_technology/policy/title01/detail01/1374030.htm (accessed on 3 September 2020).
- Duffey, K.J.; Popkin, B.M. Energy density, portion size, and eating occasions: Contributions to increased energy intake in the United States, 1977–2006. PLoS Med. 2011, 8, e1001050. [Google Scholar] [CrossRef]
- Nielsen, S.J.; Siega-Riz, A.M.; Popkin, B.M. Trends in energy intake in U.S. between 1977 and 1996: Similar shifts seen across age groups. Obes Res. 2002, 10, 370–378. [Google Scholar] [CrossRef]
- Karl, J.P.; Young, A.J.; Rood, J.C.; Montain, S.J. Independent and combined effects of eating rate and energy density on energy intake, appetite, and gut hormones. Obesity 2013, 21, E244–E252. [Google Scholar] [CrossRef]
- Robinson, E.; Almiron-Roig, E.; Rutters, F.; de Graaf, C.; Forde, C.G.; Smith, C.T.; Nolan, S.J.; Jebb, S.A. A systematic review and meta-analysis examining the effect of eating rate on energy intake and hunger. Am. J. Clin. Nutr. 2014, 100, 123–151. [Google Scholar] [CrossRef]
- Bo, S.; Fadda, M.; Castiglione, A.; Ciccone, G.; de Francesco, A.; Fedele, D.; Guggino, A.; Caprino, M.P.; Ferrara, S.; Boggio, M.V.; et al. Is the timing of caloric intake associated with variation in diet-induced thermogenesis and in the metabolic pattern? a randomized cross-over study. Int. J. Obes. 2015, 39, 1689–1695. [Google Scholar] [CrossRef] [PubMed]
- Romon, M.; Edme, J.L.; Boulenguez, C.; Lescroart, J.L.; Frimat, P. Circadian variation of diet-induced thermogenesis. Am. J. Clin. Nutr. 1993, 57, 476–480. [Google Scholar] [CrossRef] [PubMed]
- Imayama, I.; Prasad, B. Role of leptin in obstructive sleep apnea. Ann. Am. Thorac. Soc. 2017, 14, 1607–1621. [Google Scholar] [CrossRef]
- Yasumoto, Y.; Hashimoto, C.; Nakao, R.; Yamazaki, H.; Hiroyama, H.; Nemoto, T.; Yamamoto, S.; Sakurai, M.; Oike, H.; Wada, N.; et al. Short-term feeding at the wrong time is sufficient to desynchronize peripheral clocks and induce obesity with hyperphagia, physical inactivity and metabolic disorders in mice. Metabolism 2016, 65, 714–727. [Google Scholar] [CrossRef]
- Taheri, S.; Lin, L.; Austin, D.; Young, T.; Mignot, E. Short sleep duration is associated with reduced leptin, elevated ghrelin, and increased body mass index. PLoS Med. 2004, 1, e62. [Google Scholar] [CrossRef]
- Lee, K.W.; Song, W.O.; Cho, M.S. Dietary quality differs by consumption of meals prepared at home vs. outside in Korean adults. Nutr. Res. Pract. 2016, 10, 294–304. [Google Scholar] [CrossRef]
- Research Group on Health and Nutrition Information. The National Health and Nutrition Survey in Japan. 2014. Available online: https://www.mhlw.go.jp/file/04-Houdouhappyou-10904750-Kenkoukyoku-Gantaisakukenkouzoushinka/0000117311.pdf (accessed on 3 September 2020). (In Japanese)
- Sasaki, S.; Katagiri, A.; Tsuji, T.; Shimoda, T.; Amano, K. Self-reported rate of eating correlates with body mass index in 18-y-old Japanese women. Int. J. Obes. Relat. Metab. Disord. 2003, 27, 1405–1410. [Google Scholar] [CrossRef]
| Snacking | Eating Quickly | Eating Late-Evening Meals | |||||||
|---|---|---|---|---|---|---|---|---|---|
| No | Yes | p Value | No | Yes | p Value | No | Yes | p Value | |
| (n = 832) | (n = 1074) | (n = 1037) | (n = 869) | (n = 1399) | (n = 507) | ||||
| Age, year | 60.8 (0.3) | 60.0 (0.3) | 0.11 | 61.3 (0.3) | 59.1 (0.3) | <0.001 | 61.8 (0.3) | 56.4 (0.4) | <0.001 |
| Women, % | 40.5 | 68.3 | <0.001 | 57.8 | 54.3 | 0.13 | 63.1 | 37.3 | <0.001 |
| Current smoking, % | 15.9 | 11.5 | 0.005 | 14.3 | 12.2 | 0.16 | 12.2 | 16.9 | 0.006 |
| Current drinking, % | 64.6 | 50.0 | <0.001 | 57.6 | 54.8 | 0.27 | 53.2 | 65.1 | <0.001 |
| Regular exercise, % | 16.0 | 15.5 | 0.77 | 15.1 | 16.4 | 0.42 | 16.3 | 14.1 | 0.27 |
| Married, % | 80.2 | 83.7 | 0.06 | 80.9 | 83.7 | 0.11 | 82.7 | 80.8 | 0.38 |
| Living alone, % | 7.1 | 4.9 | 0.04 | 6.2 | 5.4 | 0.44 | 5.0 | 8.2 | 0.02 |
| Current employment, % | 52.2 | 45.9 | 0.03 | 47.3 | 50.2 | 0.29 | 44.8 | 60.0 | <0.001 |
| Outcomes Unhealthy Eating Habits | Age- and Sex-Adjusted Mean (95% CI) of BMI or WC | No. of Obese or Central Obese Subjects/Total Subjects | Model 1 a) | Model 2 b) | ||
|---|---|---|---|---|---|---|
| OR (95% CI) | p Value | OR (95% CI) | p Value | |||
| Obesity | BMI (kg/m2) | |||||
| Snacking | ||||||
| No | 22.8 (22.5–23.0) c) | 200/832 | 1.00 (reference) | 1.00 (reference) | ||
| Yes | 23.5 (23.2–23.7) c),** | 304/1074 | 1.50 (1.20–1.86) | <0.001 | 1.49 (1.19–1.86) | <0.001 |
| Eating quickly | ||||||
| No | 22.6 (22.3–22.8) c) | 207/1037 | 1.00 (reference) | 1.00 (reference) | ||
| Yes | 23.9 (23.7–24.1) c),** | 297/869 | 2.12 (1.72–2.61) | <0.001 | 2.11 (1.71–2.61) | <0.001 |
| Eating late-evening meals | ||||||
| No | 23.1 (22.9–23.3) c) | 342/1399 | 1.00 (reference) | 1.00 (reference) | ||
| Yes | 23.4 (23.1–23.7) c) | 162/507 | 1.38 (1.09–1.74) | 0.008 | 1.39 (1.09–1.77) | 0.007 |
| Central obesity | WC (cm) | |||||
| Snacking | ||||||
| No | 82.9 (82.2–83.5) d) | 315/832 | 1.00 (reference) | 1.00 (reference) | ||
| Yes | 84.7 (84.1–85.3) d),** | 545/1074 | 1.30 (1.06–1.58) | 0.01 | 1.29 (1.05–1.58) | 0.01 |
| Eating quickly | ||||||
| No | 82.4 (81.9–83.0) d) | 413/1037 | 1.00 (reference) | 1.00 (reference) | ||
| Yes | 85.7 (85.1–86.3) d),** | 447/869 | 1.88 (1.55–2.29) | <0.001 | 1.89 (1.55–2.30) | <0.001 |
| Eating late-evening meals | ||||||
| No | 83.6 (83.1–84.1) d) | 645/1399 | 1.00 (reference) | 1.00 (reference) | ||
| Yes | 84.7 (83.9–85.6) d),* | 215/507 | 1.35 (1.07–1.70) | 0.01 | 1.36 (1.08–1.72) | 0.009 |
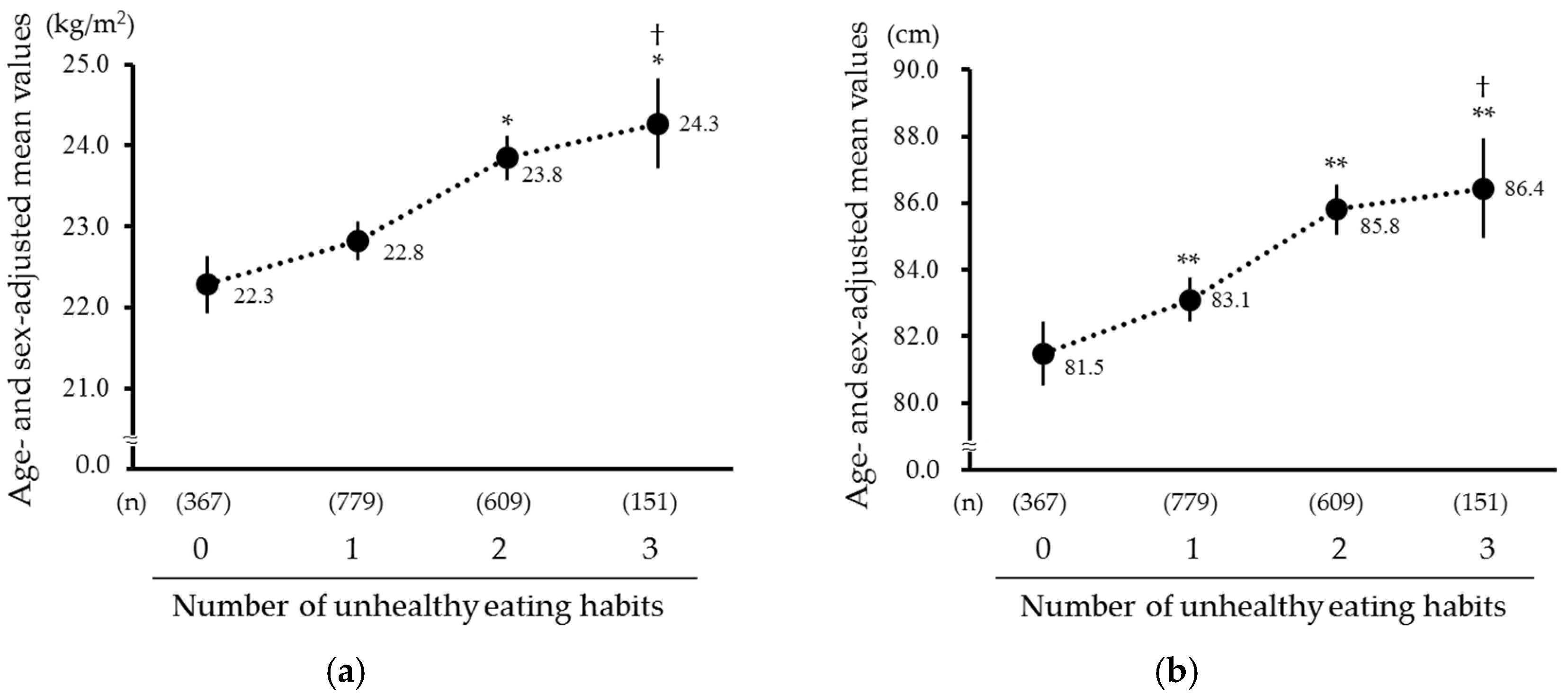
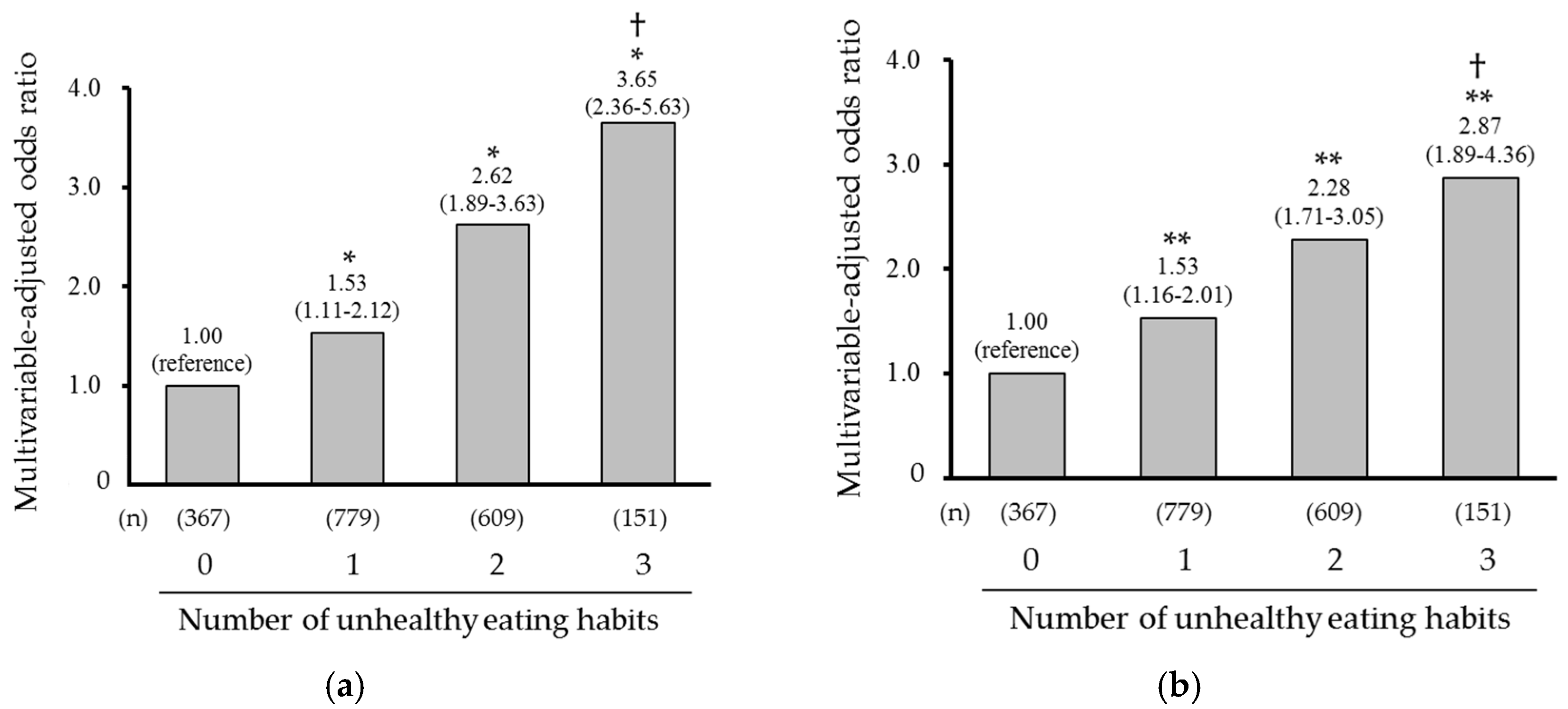
| Number of Unhealthy Eating Habits | |||||
|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | p for Trend | |
| (n = 367) | (n = 779) | (n = 609) | (n = 151) | ||
| Age, year | 63.3 (0.5) | 61.0 (0.3) | 58.7 (0.4) | 56.2 (0.8) | <0.001 |
| Women, % | 45.6 | 61.7 | 57.2 | 49.5 | 0.20 |
| Current smoking, % | 14.1 | 14.3 | 11.5 | 13.9 | 0.36 |
| Current drinking, % | 60.6 | 57.2 | 52.6 | 56.0 | 0.06 |
| Regular exercise, % | 15.5 | 16.4 | 14.8 | 16.2 | 0.81 |
| Married, % | 76.9 | 83.0 | 85.6 | 76.9 | 0.12 |
| Living alone, % | 6.2 | 6.3 | 4.8 | 7.2 | 0.64 |
| Current employment, % | 46.4 | 46.7 | 51.1 | 54.4 | 0.08 |
| Subgroups | Obesity | Central Obesity | ||||||
|---|---|---|---|---|---|---|---|---|
| No. of Events | No. of Subjects | OR (95% CI) per 1 Increment in the Number of Unhealthy Eating Habits | p for Hetero. | No. of Events | No. of Subjects | OR (95% CI) per 1 Increment in the Number of Unhealthy Eating Habits | p for Hetero. | |
| Age | ||||||||
| 40–59 years | 186 | 757 | 1.98 (1.60–2.45) | 298 | 757 | 1.71 (1.42–2.05) | ||
| 60–74 years | 318 | 1149 | 1.40 (1.20–1.63) | 0.01 | 562 | 1149 | 1.27 (1.10–1.47) | 0.01 |
| Sex | ||||||||
| Men | 267 | 835 | 1.75 (1.48–2.08) | 243 | 835 | 1.63 (1.37–1.93) | ||
| Women | 237 | 1071 | 1.41 (1.17–1.69) | 0.02 | 617 | 1071 | 1.32 (1.13–1.54) | 0.007 |
| Current smoking | ||||||||
| No | 413 | 1576 | 1.55 (1.35–1.78) | 760 | 1576 | 1.44 (1.27–1.64) | ||
| Yes | 91 | 330 | 1.77 (1.33–2.35) | 0.38 | 100 | 330 | 1.53 (1.16–2.01) | 0.47 |
| Current drinking | ||||||||
| No | 214 | 855 | 1.55 (1.27–1.88) | 439 | 855 | 1.31 (1.11–1.56) | ||
| Yes | 290 | 1051 | 1.62 (1.38–1.91) | 0.66 | 421 | 1051 | 1.58 (1.36–1.85) | 0.09 |
| Regular exercise | ||||||||
| No | 422 | 1595 | 1.69 (1.47–1.93) | 741 | 1595 | 1.51 (1.33–1.72) | ||
| Yes | 82 | 309 | 1.20 (0.90–1.62) | 0.046 | 119 | 309 | 1.21 (0.90–1.61) | 0.22 |
| Marital status | ||||||||
| Unmarried, divorced, and widowed | 77 | 346 | 1.45 (1.94–1.92) | 150 | 346 | 1.35 (1.05–1.74) | ||
| Married | 427 | 1560 | 1.63 (1.42–1.87) | 0.49 | 710 | 1560 | 1.48 (1.30–1.69) | 0.47 |
| Living status | ||||||||
| Living with others | 477 | 1790 | 1.62 (1.43–1.84) | 807 | 1790 | 1.47 (1.30–1.65) | ||
| Living alone | 27 | 116 | 1.23 (0.75–2.03) | 0.34 | 53 | 116 | 1.35 (0.85–2.14) | 0.66 |
| Employment status | ||||||||
| Unemployed | 239 | 983 | 1.39 (1.16–1.66) | 513 | 983 | 1.25 (1.07–1.47) | ||
| Employed | 265 | 923 | 1.82 (1.53–2.17) | 0.048 | 347 | 923 | 1.71 (1.45–2.02) | 0.006 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ishida, Y.; Yoshida, D.; Honda, T.; Hirakawa, Y.; Shibata, M.; Sakata, S.; Furuta, Y.; Oishi, E.; Hata, J.; Kitazono, T.; et al. Influence of the Accumulation of Unhealthy Eating Habits on Obesity in a General Japanese Population: The Hisayama Study. Nutrients 2020, 12, 3160. https://doi.org/10.3390/nu12103160
Ishida Y, Yoshida D, Honda T, Hirakawa Y, Shibata M, Sakata S, Furuta Y, Oishi E, Hata J, Kitazono T, et al. Influence of the Accumulation of Unhealthy Eating Habits on Obesity in a General Japanese Population: The Hisayama Study. Nutrients. 2020; 12(10):3160. https://doi.org/10.3390/nu12103160
Chicago/Turabian StyleIshida, Yuki, Daigo Yoshida, Takanori Honda, Yoichiro Hirakawa, Mao Shibata, Satoko Sakata, Yoshihiko Furuta, Emi Oishi, Jun Hata, Takanari Kitazono, and et al. 2020. "Influence of the Accumulation of Unhealthy Eating Habits on Obesity in a General Japanese Population: The Hisayama Study" Nutrients 12, no. 10: 3160. https://doi.org/10.3390/nu12103160
APA StyleIshida, Y., Yoshida, D., Honda, T., Hirakawa, Y., Shibata, M., Sakata, S., Furuta, Y., Oishi, E., Hata, J., Kitazono, T., & Ninomiya, T. (2020). Influence of the Accumulation of Unhealthy Eating Habits on Obesity in a General Japanese Population: The Hisayama Study. Nutrients, 12(10), 3160. https://doi.org/10.3390/nu12103160




