Low Serum Magnesium is Associated with Incident Dementia in the ARIC-NCS Cohort
Abstract
1. Introduction
2. Materials and Methods
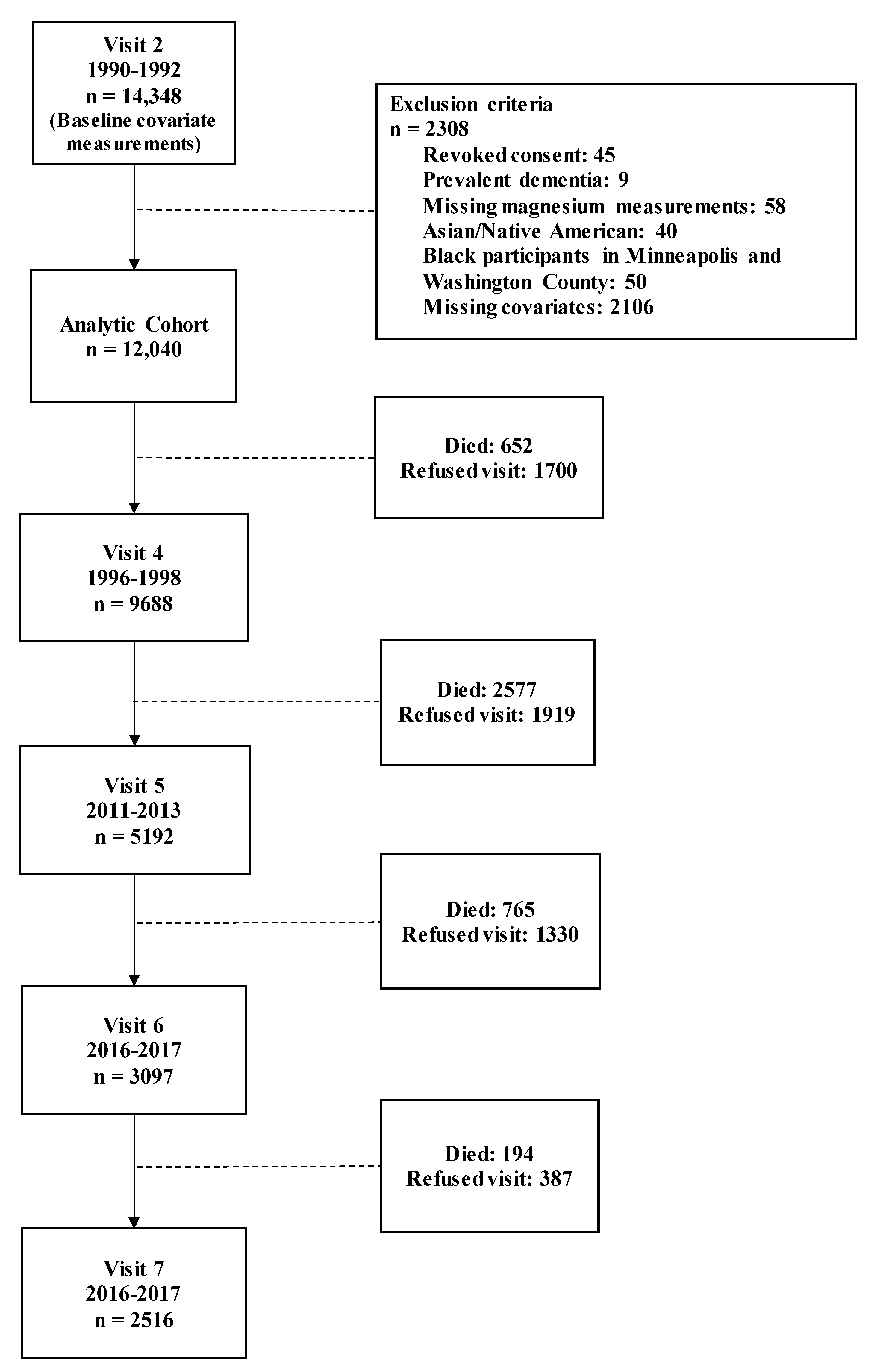
2.1. Study Population
2.2. Incident Dementia
2.3. Cognitive Function
2.4. Serum Magnesium
2.5. Covariates
2.6. Statistical Analysis
3. Results
3.1. Incident Dementia
3.2. Cognitive Decline
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zhao, B.; Hu, L.; Dong, Y.; Xu, J.; Wei, Y.; Yu, D.; Xu, J.; Zhang, W. The Effect of Magnesium Intake on Stroke Incidence: A Systematic Review and Meta-Analysis With Trial Sequential Analysis. Front. Neurol. 2019, 10, 852. [Google Scholar] [CrossRef] [PubMed]
- Barbagallo, M.; Dominguez, L.J. Magnesium and type 2 diabetes. World J. Diabetes 2015, 6, 1152–1157. [Google Scholar] [CrossRef] [PubMed]
- Misialek, J.R.; Lopez, F.L.; Lutsey, P.L.; Huxley, R.R.; Peacock, J.M.; Chen, L.Y.; Soliman, E.Z.; Agarwal, S.K.; Alonso, A. Serum and dietary magnesium and incidence of atrial fibrillation in whites and in African Americans--Atherosclerosis Risk in Communities (ARIC) study. Circ. J. 2013, 77, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Hu, M.; Yang, L.; Xu, H.; Song, W.; Qian, Y.; Zhao, M. Quantitative Association Between Serum/Dietary Magnesium and Cardiovascular Disease/Coronary Heart Disease Risk: A Dose-Response Meta-analysis of Prospective Cohort Studies. J. Cardiovasc. Pharm. 2019, 74, 516–527. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Huang, X.; Zhang, L.; Han, F.; Pang, K.L.; Li, X.; Shen, J.Y. Magnesium boosts the memory restorative effect of environmental enrichment in Alzheimer’s disease mice. CNS Neurosci. 2018, 24, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Yu, X.; Guan, P.P.; Guo, J.W.; Wang, Y.; Zhang, Y.; Zhao, H.; Wang, Z.Y. Magnesium ion influx reduces neuroinflammation in Abeta precursor protein/Presenilin 1 transgenic mice by suppressing the expression of interleukin-1beta. Cell Mol. Immunol. 2017, 14, 451–464. [Google Scholar] [CrossRef]
- Li, W.; Yu, J.; Liu, Y.; Huang, X.; Abumaria, N.; Zhu, Y.; Huang, X.; Xiong, W.; Ren, C.; Liu, X.G.; et al. Elevation of brain magnesium prevents synaptic loss and reverses cognitive deficits in Alzheimer’s disease mouse model. Mol. Brain 2014, 7, 65. [Google Scholar] [CrossRef]
- Lo, K.; Liu, Q.; Madsen, T.; Rapp, S.; Chen, J.C.; Neuhouser, M.; Shadyab, A.; Pal, L.; Lin, X.; Shumaker, S.; et al. Relations of magnesium intake to cognitive impairment and dementia among participants in the Women‘s Health Initiative Memory Study: A prospective cohort study. BMJ Open 2019, 9, e030052. [Google Scholar] [CrossRef]
- Tzeng, N.S.; Chung, C.H.; Lin, F.H.; Huang, C.F.; Yeh, C.B.; Huang, S.Y.; Lu, R.B.; Chang, H.A.; Kao, Y.C.; Yeh, H.W.; et al. Magnesium oxide use and reduced risk of dementia: A retrospective, nationwide cohort study in Taiwan. Curr. Med. Res. Opin. 2018, 34, 163–169. [Google Scholar] [CrossRef]
- Kieboom, B.C.T.; Licher, S.; Wolters, F.J.; Ikram, M.K.; Hoorn, E.J.; Zietse, R.; Stricker, B.H.; Ikram, M.A. Serum magnesium is associated with the risk of dementia. Neurology 2017, 89, 1716–1722. [Google Scholar] [CrossRef]
- The ARIC Investigators. The Atherosclerosis Risk in Communities (ARIC) Study: Design and objectives. Am. J. Epidemiol. 1989, 129, 687–702. [Google Scholar]
- Gindler, E.; Heth, D. Colorimetric determination with bound calmagite of magnesium in human blood serum. In Clinical Chemistry; American Association Clinical Chemistry: Washington, DC, USA, 1971; p. 20037-1526. [Google Scholar]
- Lutsey, P.L.; Steffen, L.M.; Stevens, J. Dietary intake and the development of the metabolic syndrome: The Atherosclerosis Risk in Communities study. Circulation 2008, 117, 754–761. [Google Scholar] [CrossRef] [PubMed]
- Siegler, I.C.; Botwinick, J. A long-term longitudinal study of intellectual ability of older adults: The matter of selective subject attrition. J. Gerontol. 1979, 34, 242–245. [Google Scholar] [CrossRef] [PubMed]
- White, I.R.; Royston, P.; Wood, A.M. Multiple imputation using chained equations: Issues and guidance for practice. Stat. Med. 2011, 30, 377–399. [Google Scholar] [CrossRef] [PubMed]
- Gottesman, R.F.; Rawlings, A.M.; Sharrett, A.R.; Albert, M.; Alonso, A.; Bandeen-Roche, K.; Coker, L.H.; Coresh, J.; Couper, D.J.; Griswold, M.E.; et al. Impact of differential attrition on the association of education with cognitive change over 20 years of follow-up: The ARIC neurocognitive study. Am. J. Epidemiol. 2014, 179, 956–966. [Google Scholar] [CrossRef]
- Olivares, D.; Deshpande, V.K.; Shi, Y.; Lahiri, D.K.; Greig, N.H.; Rogers, J.T.; Huang, X. N-methyl D-aspartate (NMDA) receptor antagonists and memantine treatment for Alzheimer’s disease, vascular dementia and Parkinson’s disease. Curr. Alzheimer Res. 2012, 9, 746–758. [Google Scholar] [CrossRef] [PubMed]
- Lynch, M.A. Long-term potentiation and memory. Physiol. Rev. 2004, 84, 87–136. [Google Scholar] [CrossRef]
- Zipfel, G.J.; Babcock, D.J.; Lee, J.M.; Choi, D.W. Neuronal apoptosis after CNS injury: The roles of glutamate and calcium. J. Neurotrauma 2000, 17, 857–869. [Google Scholar] [CrossRef]
- Hynd, M.R.; Scott, H.L.; Dodd, P.R. Glutamate-mediated excitotoxicity and neurodegeneration in Alzheimer’s disease. Neurochem. Int. 2004, 45, 583–595. [Google Scholar] [CrossRef]
- Greene, J.G.; Greenamyre, J.T. Bioenergetics and glutamate excitotoxicity. Prog. Neurobiol. 1996, 48, 613–634. [Google Scholar] [CrossRef]
- Wenzel, T.J.; Klegeris, A. Novel multi-target directed ligand-based strategies for reducing neuroinflammation in Alzheimer’s disease. Life Sci. 2018, 207, 314–322. [Google Scholar] [CrossRef] [PubMed]
- Rakic, S.; Hung, Y.M.A.; Smith, M.; So, D.; Tayler, H.M.; Varney, W.; Wild, J.; Harris, S.; Holmes, C.; Love, S.; et al. Systemic infection modifies the neuroinflammatory response in late stage Alzheimer’s disease. Acta Neuropathol. Commun. 2018, 6, 88. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Guan, P.P.; Zhu, D.; Liang, Y.Y.; Wang, T.; Wang, Z.Y.; Wang, P. Magnesium Ions Inhibit the Expression of Tumor Necrosis Factor alpha and the Activity of gamma-Secretase in a beta-Amyloid Protein-Dependent Mechanism in APP/PS1 Transgenic Mice. Front. Mol. Neurosci. 2018, 11, 172. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Sun, M.; Chen, Z.; Lu, J.; Liu, Y.; Zhou, L.; Xu, X.; Fan, D.; Chui, D. Magnesium modulates amyloid-beta protein precursor trafficking and processing. J. Alzheimers Dis. 2010, 20, 1091–1106. [Google Scholar] [CrossRef]
- Veronese, N.; Zurlo, A.; Solmi, M.; Luchini, C.; Trevisan, C.; Bano, G.; Manzato, E.; Sergi, G.; Rylander, R. Magnesium Status in Alzheimer’s Disease: A Systematic Review. Am. J. Alzheimers Dis. Other Demen 2016, 31, 208–213. [Google Scholar] [CrossRef]
- Ozawa, M.; Ninomiya, T.; Ohara, T.; Hirakawa, Y.; Doi, Y.; Hata, J.; Uchida, K.; Shirota, T.; Kitazono, T.; Kiyohara, Y. Self-reported dietary intake of potassium, calcium, and magnesium and risk of dementia in the Japanese: The Hisayama Study. J. Am. Geriatr. Soc. 2012, 60, 1515–1520. [Google Scholar] [CrossRef]
- Horibata, K.; Tanoue, A.; Ito, M.; Takemura, Y. Relationship between renal function and serum magnesium concentration in elderly outpatients treated with magnesium oxide. Geriatr. Gerontol. Int. 2016, 16, 600–605. [Google Scholar] [CrossRef]
- Blaine, J.; Chonchol, M.; Levi, M. Renal control of calcium, phosphate, and magnesium homeostasis. Clin. J. Am. Soc. Nephrol. 2015, 10, 1257–1272. [Google Scholar] [CrossRef]
- Razzaque, M.S. Magnesium: Are We Consuming Enough? Nutrients 2018, 10, 1863. [Google Scholar] [CrossRef]
- Andrasi, E.; Pali, N.; Molnar, Z.; Kosel, S. Brain aluminum, magnesium and phosphorus contents of control and Alzheimer-diseased patients. J. Alzheimers Dis. 2005, 7, 273–284. [Google Scholar] [CrossRef]
- Balmus, I.M.; Strungaru, S.A.; Ciobica, A.; Nicoara, M.N.; Dobrin, R.; Plavan, G.; Stefanescu, C. Preliminary Data on the Interaction between Some Biometals and Oxidative Stress Status in Mild Cognitive Impairment and Alzheimer’s Disease Patients. Oxid. Med. Cell Longev. 2017, 2017, 7156928. [Google Scholar] [CrossRef] [PubMed]
- Rawlings, A.M.; Sang, Y.; Sharrett, A.R.; Coresh, J.; Griswold, M.; Kucharska-Newton, A.M.; Palta, P.; Wruck, L.M.; Gross, A.L.; Deal, J.A.; et al. Multiple imputation of cognitive performance as a repeatedly measured outcome. Eur. J. Epidemiol. 2017, 32, 55–66. [Google Scholar] [CrossRef] [PubMed]
- Johnson, D.R.; Young, R. Toward best practices in analyzing datasets with missing data: Comparisons and recommendations. J. Marriage Fam. 2011, 73, 926–945. [Google Scholar] [CrossRef]
| Characteristics | Magnesium Quintiles | ||||
|---|---|---|---|---|---|
| 1st (≤1.4 mg/dL) | 2nd (1.5 mg/dL) | 3rd (1.6 mg/dL) | 4th (1.7 mg/dL) | 5th (≥1.8 mg/dL) | |
| N = 1650 | N = 2370 | N = 3255 | N = 2599 | N = 2166 | |
| Age, years | 57.1 (5.8) | 56.7 (5.8) | 56.8 (5.6) | 57.0 (5.7) | 57.2 (5.7) |
| Female, % | 63.0 | 56.6 | 56.7 | 54.0 | 53.0 |
| African American, % | 43.8 | 29.2 | 22.0 | 18.5 | 16.3 |
| Education, % | |||||
| Did not complete high school | 29.8 | 22.4 | 20.0 | 18.5 | 18.5 |
| High school graduate and/or vocational school | 39.1 | 41.1 | 42.3 | 42.2 | 42.6 |
| At least some college | 31.1 | 36.5 | 37.7 | 39.4 | 39.0 |
| Sodium, mmol/L | 140.3 (2.6) | 140.6 (2.3) | 140.8 (2.3) | 141.0 (2.2) | 141.2 (2.2) |
| Potassium, mmol/L | 4.06 (0.42) | 4.15 (0.38) | 4.18 (0.38) | 4.22 (0.40) | 4.24 (0.40) |
| Calcium, mmol/L | 0.521 (0.027) | 0.518 (0.023) | 0.518 (0.023) | 0.519 (0.023) | 0.520 (0.023) |
| Waist-to-Hip Ratio | 0.93 (0.08) | 0.93 (0.08) | 0.92 (0.08) | 0.92 (0.08) | 0.93 (0.08) |
| Ever smoked, % | 59.0 | 60.5 | 59.1 | 59.2 | 61.6 |
| Current drinker, % | 48.1 | 54.8 | 57.8 | 59.0 | 60.5 |
| Prevalent coronary heart disease, % | 6.8 | 6.4 | 5.9 | 5.2 | 5.2 |
| Previous stroke, % | 2.9 | 1.9 | 1.5 | 1.6 | 1.6 |
| Diabetes, % | 33.4 | 19.4 | 13.1 | 11.0 | 9.0 |
| Systolic BP, mmHg | 125.4 (20.4) | 121.7 (19.0) | 121.0 (18.2) | 119.5 (17.9) | 120.6 (17.8) |
| Diastolic BP, mmHg | 73.1 (10.6) | 72.2 (10.2) | 72.1 (10.3) | 71.6 (10.1) | 71.9 (10.0) |
| Total Cholesterol-to-HDL cholesterol ratio | 4.76 (2.06) | 4.73 (2.00) | 4.62 (1.81) | 4.65 (1.74) | 4.76 (1.93) |
| Antihypertensive medication, % | |||||
| Diuretic | 25.5 | 15.7 | 12.3 | 10.7 | 9.4 |
| Non-diuretic, antihypertensive | 5.0 | 3.0 | 3.1 | 3.3 | 2.6 |
| No antihypertensive medication | 69.6 | 81.3 | 84.7 | 86.0 | 88.0 |
| eGFR, mL/min/1.73m2 | 97.2 (20.1) | 96.1 (17.2) | 96.1 (16.1) | 94.5 (15.7) | 92.3 (16.3) |
| APOE ε4 allele, % | 31.4 | 31.2 | 30.8 | 29.6 | 30.9 |
| C-reactive Protein, mg/L | 5.9 (8.8) | 4.6 (6.9) | 4.2 (7.1) | 3.9 (7.1) | 3.8 (6.3) |
| Western Diet Score | −0.018 (0.981) | −0.012 (1.000) | −0.031 (0.979) | −0.046 (0.967) | −0.009 (0.996) |
| Prudent Diet Score | −0.025 (0.988) | 0.021 (1.027) | 0.011 (0.950) | 0.032 (0.987) | 0.019 (1.00) |
| Quintile of Magnesium | Person Years of Follow-Up | Dementia Cases | IR ‡ | Model 1 * HR (95%CI) | Model 2 ** HR (95%CI) |
|---|---|---|---|---|---|
| Quintile 1 | 32,306 | 367 | 11.36 | 1.34 (1.17, 1.54) | 1.24 (1.07, 1.44) |
| Quintile 2 | 49,507 | 475 | 9.59 | 1.11 (0.97, 1.26) | 1.08 (0.95, 1.24) |
| Quintile 3 | 70,753 | 681 | 9.63 | 1.03 (0.91, 1.16) | 1.03 (0.91, 1.16) |
| Quintile 4 | 56,470 | 559 | 9.90 | 1.07 (0.95, 1.21) | 1.08 (0.95, 1.22) |
| Quintile 5 | 47,188 | 437 | 9.26 | 1 (Ref) | 1 (Ref) |
| Per 1 standard deviation (~0.009 mmol/L) decrease in Mg | 1.09 (1.05, 1.14) | 1.07 (1.02, 1.11) | |||
| Test | Quintiles | Model 1 * | Model 2 ** |
|---|---|---|---|
| Global | |||
| 1 | −0.031 (−0.092, 0.029) | 0.003 (−0.060, 0.067) | |
| 2 | 0.052 (−0.0004, 0.104) | 0.063 (0.011, 0.115) | |
| 3 | 0.027 (−0.022, 0.075) | 0.030 (−0.017, 0.077) | |
| 4 | −0.021 (−0.074, 0.031) | −0.022 (−0.073, 0.028) | |
| 5 | 0 (Referent) | 0 (Referent) | |
| Per 1-SD *** decrease in Mg | 0.004 (-0.013, 0.020) | −0.003 (−0.022, 0.016) | |
| DWR | |||
| 1 | −0.023 (−0.126, 0.080) | 0.001 (−0.106, 0.107) | |
| 2 | 0.058 (−0.033, 0.149) | 0.066 (−0.027, 0.158) | |
| 3 | 0.052 (−0.035, 0.139) | 0.053 (−0.034, 0.141) | |
| 4 | −0.051 (−0.145, 0.043) | −0.054 (−0.146, 0.038) | |
| 5 | 0 (Referent) | 0 (Referent) | |
| Per 1-SD decrease in Mg | 0.011 (−0.017, 0.039) | 0.007 (−0.028, 0.042) | |
| DSS | |||
| 1 | −0.039 (−0.096, 0.017) | −0.0004 (−0.056, 0.055) | |
| 2 | 0.048 (0.006, 0.090) | 0.063 (0.021, 0.105) | |
| 3 | 0.020 (−0.018, 0.059) | 0.027 (−0.011, 0.065) | |
| 4 | −0.023 (−0.069, 0.022) | −0.023 (−0.068, 0.022) | |
| 5 | 0 (Referent) | 0 (Referent) | |
| Per 1-SD decrease in Mg | 0.002 (−0.014, 0.017) | −0.010 (−0.027, 0.008) | |
| WF | |||
| 1 | −0.017 (−0.082, 0.047) | 0.018 (−0.052, 0.089) | |
| 2 | 0.034 (−0.017, 0.085) | 0.047 (−0.003, 0.097) | |
| 3 | 0.009 (−0.041, 0.059) | 0.011 (−0.038, 0.061) | |
| 4 | 0.013 (−0.039, 0.064) | 0.012 (−0.039, 0.063) | |
| 5 | 0 (Referent) | 0 (Referent) | |
| Per 1-SD decrease in Mg | −0.001 (−0.018, 0.017) | −0.007 (−0.030, 0.015) | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alam, A.B.; Lutsey, P.L.; Gottesman, R.F.; Tin, A.; Alonso, A. Low Serum Magnesium is Associated with Incident Dementia in the ARIC-NCS Cohort. Nutrients 2020, 12, 3074. https://doi.org/10.3390/nu12103074
Alam AB, Lutsey PL, Gottesman RF, Tin A, Alonso A. Low Serum Magnesium is Associated with Incident Dementia in the ARIC-NCS Cohort. Nutrients. 2020; 12(10):3074. https://doi.org/10.3390/nu12103074
Chicago/Turabian StyleAlam, Aniqa B., Pamela L. Lutsey, Rebecca F. Gottesman, Adrienne Tin, and Alvaro Alonso. 2020. "Low Serum Magnesium is Associated with Incident Dementia in the ARIC-NCS Cohort" Nutrients 12, no. 10: 3074. https://doi.org/10.3390/nu12103074
APA StyleAlam, A. B., Lutsey, P. L., Gottesman, R. F., Tin, A., & Alonso, A. (2020). Low Serum Magnesium is Associated with Incident Dementia in the ARIC-NCS Cohort. Nutrients, 12(10), 3074. https://doi.org/10.3390/nu12103074





