Dietary Intakes and Cardiovascular Health of Healthy Adults in Short-, Medium-, and Long-Term Whole-Food Plant-Based Lifestyle Program
Abstract
1. Introduction
2. Materials and Methods
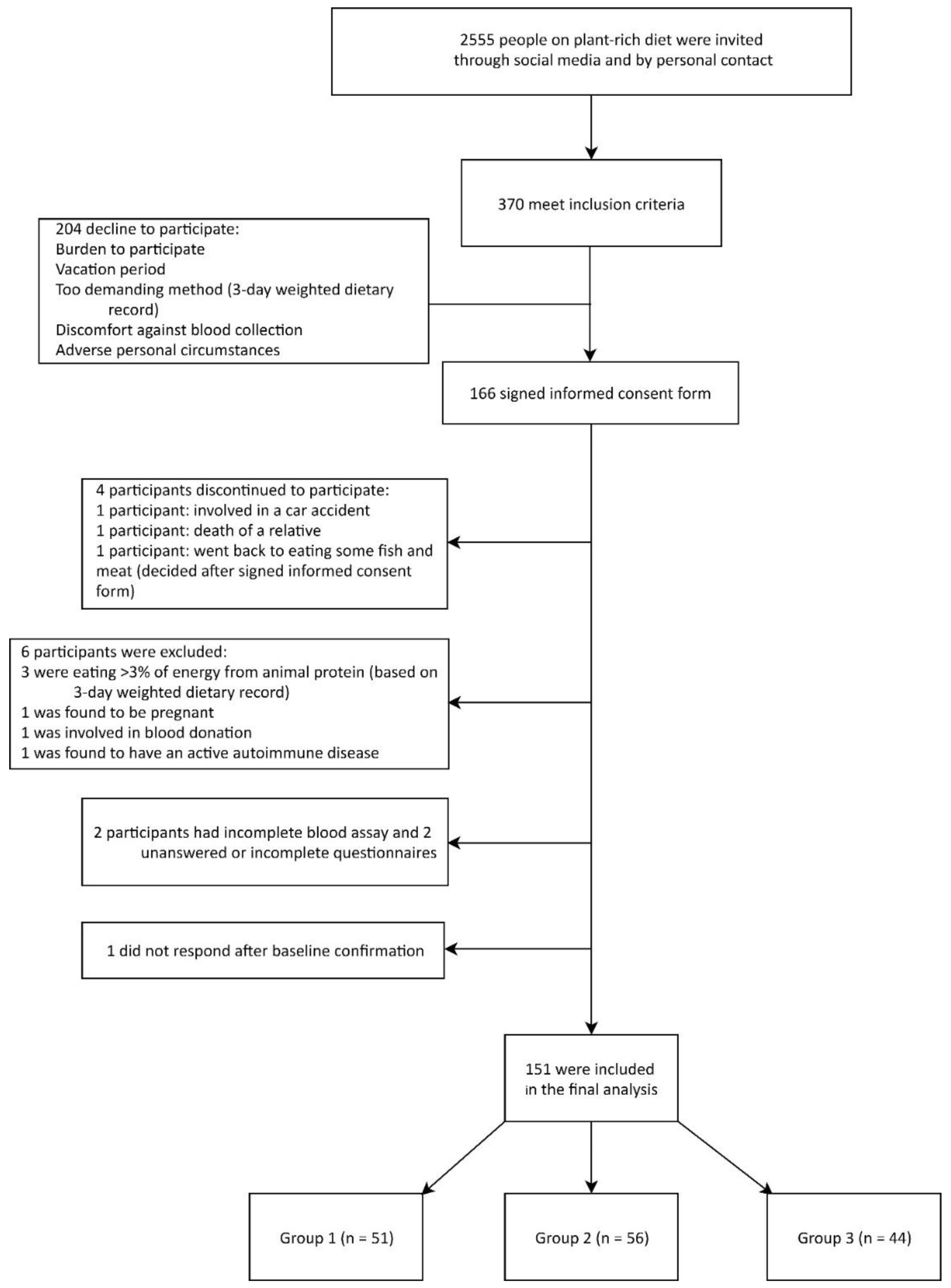
2.1. Study Design and Eligibility
2.2. Subject
2.3. Intervention: WFPB Lifestyle Program
2.3.1. Nutritional Part
2.3.2. Physical Activity
2.3.3. Support System
2.4. Outcomes
2.4.1. Dietary and Food Intakes
2.4.2. Cardiovascular Risk and Safety Factors
2.5. Statistical Analysis
3. Results
3.1. Characteristics of the Participants
3.2. Dietary and Food Intakes of Participants in Our WFPB Lifestyle Program
3.3. Cardiovascular Health and Safety Factors Status of Participants in Our WFPB Lifestyle Program
3.3.1. Main Findings
3.3.2. Safety Markers
4. Discussion
4.1. Main Findings
4.2. Dietary Intake and Diet Quality
| Country | Belgium [8] | Switzerland [14] | Finland [16] | France [66] | U.S. [71] | U.S. [72] | Slovenia (Our Study) |
|---|---|---|---|---|---|---|---|
| Year of publication | 2014 | 2015 | 2016 | 2017 | 2018 | 2019 | 2019 |
| Participants (n) | n = 104 | n = 53 | n = 22 | n = 789 | Theoretical † | n = 200/Theoretical | n = 151 |
| Age (years old) | 20–69 | 18–50 | 24–50 | >18 | - | - | 18–78 |
| Diet name | Vegan diet | Vegan diet | Vegan diet | Vegan diet | Vegan diet | WFPB diet | supplemented WFPB diet |
| Animal food limit | Excluded | - | Self-defined | Excluded | Excluded | Excluded | ≤3% of energy as animal protein |
| Diet assessment | FFQ * | 3-DR | 3-DR | 3 × 24–h DR | 7 × single day | 30-day meal plan | 3-DR |
| Duration on PBD (y, min–max) | - | 3.0 (1.0–18) | 8.6 (2–16) | Currently | - | - | 4.1 (0.8–10.3) |
| Dietary intake/day | |||||||
| Energy (kcal) | 2383 | 2469 | 2150 | Adjusted | 1302 | Adjusted | 2058 |
| Total fat (g) | 68 | 96 | 88 | 72.7 | 35 | 38 | 44 |
| Total fat (% E) | 25 | 33 | 36.5 | 35.2 | 24 | 17 | 20 |
| SFA (g) | 21 | 20 | 21 | 19.4 | 6 | 6.5 | 7 |
| SFA (% E) | 8 | 7 ‡ | 8.6 | 9 ‡ | 4 ‡ | 3 | 3 |
| Cholesterol (mg) | 149 | 12 | 44 | 55.4 | 0 | 0 | 7 |
| Carbohydrates (g) | 336 | 324 | 252 | 235.7 | 225 | 365 | 287 |
| Carbohydrates (% E) | 57 | 54 | 49 | 51.2 | 69 | 73 | 57 (64% E) |
| Sugar (g) | 156 | 180 | - | 105.3 | 109 | - | 85 |
| Dietary fiber (g) | 41 | 52 | 41 | 34.1 | 49 | 70 | 70 (7% E) |
| Proteins (g) | 82 | 65 | 74 | 66.6 | 51 | 81 | 77 |
| Proteins (% E) | 14 | 11 | 13.7 | 13.2 | 15.7 | 16 | 15 |
| B12 (µg) | - | 0.2 | 0.9 | 2.7 | 0.7 | 904 | 280 |
| Sodium (mg) | 1316 | 2994 | - | 2589.6 | - | 2807 | 2075 |
| Calcium (mg) | 738 | 817 | 1004 | 760 | 782 | 959 | 1099 |
| Iron (mg) | 23 | 22.9 | 21 | 18.6 | 17.2 | 26 | 36 |
| Zn (mg) | - | 11.5 | 12 | 10 | 8.3 | - | 20 |
4.3. Cardiovascular Risk Factors
4.4. Support System
5. Strengths and Limitation
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| 3-DR | 3-day weighted dietary record |
| BM | body mass |
| BMI | body mass index |
| BP | blood pressure |
| CVD | cardiovascular disease |
| DHA | docosahexaenoic acid (C22:6n-3) |
| EPA | eicosapentaenoic acid (C20:5n-3) |
| EAS | European Atherosclerosis Society |
| ESC | European Society of Cardiology |
| FFQ | food frequency questionnaire |
| FU | follow up |
| H1 | hypothesis one |
| H2 | hypothesis two |
| H3 | hypothesis three |
| MR | plant-based meal replacement |
| OPEN | Open Platform for Clinical Nutrition |
| PA | physical activity |
| PBD | plant-based diet |
| UA | uric acids |
| WFPB | whole-food plant-based |
References
- Melina, V.; Craig, W.; Levin, S. Position of the Academy of Nutrition and Dietetics: Vegetarian Diets. J. Acad. Nutr. Diet. 2016, 116, 1970–1980. [Google Scholar] [CrossRef] [PubMed]
- Agnoli, C.; Baroni, L.; Bertini, I.; Ciappellano, S.; Fabbri, A.; Papa, M.; Pellegrini, N.; Sbarbati, R.; Scarino, M.L.; Siani, V.; et al. Position paper on vegetarian diets from the working group of the Italian Society of Human Nutrition. Nutr. Metab. Cardiovasc. Dis. 2017, 27, 1037–1052. [Google Scholar] [CrossRef] [PubMed]
- BDA. British Dietetic Association Confirms Well-Planned Vegan Diets Can Support Healthy Living in People of All Ages. Available online: https://www.bda.uk.com/news/view?id=179 (accessed on 3 June 2019).
- Dietitians of Canada What You Need to Know About Following a Vegan Eating Plan—Unlock Food. Available online: https://www.unlockfood.ca/en/Articles/Vegetarian-and-Vegan-Diets/What-You-Need-to-Know-About-Following-a-Vegan-Eati.aspx (accessed on 4 October 2019).
- ADG. Australian Dietary Guidelines|NHMRC. Available online: https://www.nhmrc.gov.au/about-us/publications/australian-dietary-guidelines (accessed on 22 August 2019).
- Gomes, S.C.; João, S.; Pinho, P.; Borges, C.; Santos, C.T.; Santos, A.; Design, P.G.; Mendes De Sousa Editor, S. National Programme for the Promotion of Healthy Eating Guidelines for a Healthy Vegetarian Diet; Direção-Geral da Saúde: Lisbon, Portugal, 2015; ISBN 978-972-675-228-8. [Google Scholar]
- Anderson, L.U.S. Doctors Blast Belgian Misinformation on Vegan Diets. Available online: https://www.pcrm.org/news/news-releases/us-doctors-blast-belgian-misinformation-vegan-diets (accessed on 2 September 2019).
- Clarys, P.; Deliens, T.; Huybrechts, I.; Deriemaeker, P.; Vanaelst, B.; De Keyzer, W.; Hebbelinck, M.; Mullie, P. Comparison of nutritional quality of the vegan, vegetarian, semi-vegetarian, pesco-vegetarian and omnivorous diet. Nutrients 2014, 6, 1318–1332. [Google Scholar] [CrossRef] [PubMed]
- Turner-McGrievy, G.M.; Barnard, N.D.; Cohen, J.; Jenkins, D.J.A.; Gloede, L.; Green, A.A. Changes in Nutrient Intake and Dietary Quality among Participants with Type 2 Diabetes Following a Low-Fat Vegan Diet or a Conventional Diabetes Diet for 22 Weeks. J. Am. Diet. Assoc. 2008, 108, 1636–1645. [Google Scholar] [CrossRef] [PubMed]
- Satija, A.; Bhupathiraju, S.N.; Spiegelman, D.; Chiuve, S.E.; Manson, J.E.; Willett, W.; Rexrode, K.M.; Rimm, E.B.; Hu, F.B. Healthful and Unhealthful Plant-Based Diets and the Risk of Coronary Heart Disease in U.S. Adults. J. Am. Coll. Cardiol. 2017, 70, 411–422. [Google Scholar] [CrossRef] [PubMed]
- Jakše, B.; Jakše, B.; Pajek, M.; Pajek, J. Uric Acid and Plant-Based Nutrition. Nutrients 2019, 11, 1736. [Google Scholar] [CrossRef]
- Davey, G.K.; Spencer, E.A.; Appleby, P.N.; Allen, N.E.; Knox, K.H.; Key, T.J. EPIC-Oxford: Lifestyle characteristics and nutrient intakes in a cohort of 33 883 meat-eaters and 31 546 non meat-eaters in the UK. Public Health Nutr. 2003, 6, 259–269. [Google Scholar] [CrossRef]
- Kristensen, N.B.; Madsen, M.L.; Hansen, T.H.; Allin, K.H.; Hoppe, C.; Fagt, S.; Lausten, M.S.; Gøbel, R.J.; Vestergaard, H.; Hansen, T.; et al. Intake of macro- and micronutrients in Danish vegans. Nutr. J. 2015, 14, 115. [Google Scholar] [CrossRef]
- Schüpbach, R.; Wegmüller, R.; Berguerand, C.; Bui, M.; Herter-Aeberli, I. Micronutrient status and intake in omnivores, vegetarians and vegans in Switzerland. Eur. J. Nutr. 2017, 56, 283–293. [Google Scholar] [CrossRef]
- Orlich, M.J.; Fraser, G.E. Vegetarian diets in the Adventist Health Study 2: A review of initial published findings. Am. J. Clin. Nutr. 2014, 100, 353S–358S. [Google Scholar] [CrossRef]
- Elorinne, A.L.; Alfthan, G.; Erlund, I.; Kivimäki, H.; Paju, A.; Salminen, I.; Turpeinen, U.; Voutilainen, S.; Laakso, J. Food and nutrient intake and nutritional status of Finnish vegans and non-vegetarians. PLoS ONE 2016, 11, e0148235. [Google Scholar] [CrossRef] [PubMed]
- Esselstyn, C.B.; Gendy, G.; Doyle, J.; Golubic, M.; Roizen, M.F. A way to reverse CAD? J. Fam. Pract. 2014, 63, 356b–364b. [Google Scholar]
- Ley, S.H.; Hamdy, O.; Mohan, V.; Hu, F.B. Prevention and management of type 2 diabetes: Dietary components and nutritional strategies. Lancet 2014, 383, 1999–2007. [Google Scholar] [CrossRef]
- Orlich, M.J.; Singh, P.N.; Sabaté, J.; Jaceldo-Siegl, K.; Fan, J.; Knutsen, S.; Beeson, W.L.; Fraser, G.E. Vegetarian Dietary Patterns and Mortality in Adventist Health Study 2. JAMA Intern. Med. 2013, 173, 1230. [Google Scholar] [CrossRef]
- Dinu, M.; Abbate, R.; Gensini, G.F.; Casini, A.; Sofi, F. Vegetarian, vegan diets and multiple health outcomes: A systematic review with meta-analysis of observational studies. Crit. Rev. Food Sci. Nutr. 2017, 57, 3640–3649. [Google Scholar] [CrossRef]
- Crowe, F.L.; Appleby, P.N.; Travis, R.C.; Key, T.J. Risk of hospitalization or death from ischemic heart disease among British vegetarians and nonvegetarians: Results from the EPIC-Oxford cohort study. Am. J. Clin. Nutr. 2013, 97, 597–603. [Google Scholar] [CrossRef]
- Satija, A.; Hu, F.B. Plant-based diets and cardiovascular health. Trends Cardiovasc. Med. 2018, 28, 437–441. [Google Scholar] [CrossRef]
- Kahleova, H.; Levin, S.; Barnard, N. Cardio-Metabolic Benefits of Plant-Based Diets. Nutrients 2017, 9, 848. [Google Scholar] [CrossRef]
- McMacken, M.; Shah, S. A plant-based diet for the prevention and treatment of type 2 diabetes. J. Geriatr. Cardiol. 2017, 14, 342–354. [Google Scholar]
- Viguiliouk, E.; Kendall, C.W.; Kahleová, H.; Rahelić, D.; Salas-Salvadó, J.; Choo, V.L.; Mejia, S.B.; Stewart, S.E.; Leiter, L.A.; Jenkins, D.J.; et al. Effect of vegetarian dietary patterns on cardiometabolic risk factors in diabetes: A systematic review and meta-analysis of randomized controlled trials. Clin. Nutr. 2019, 38, 1133–1145. [Google Scholar] [CrossRef]
- Satija, A.; Bhupathiraju, S.N.; Rimm, E.B.; Spiegelman, D.; Chiuve, S.E.; Borgi, L.; Willett, W.C.; Manson, J.E.; Sun, Q.; Hu, F.B. Plant-Based Dietary Patterns and Incidence of Type 2 Diabetes in US Men and Women: Results from Three Prospective Cohort Studies. PLOS Med. 2016, 13, e1002039. [Google Scholar] [CrossRef]
- Qian, F.; Liu, G.; Hu, F.B.; Bhupathiraju, S.N.; Sun, Q. Association Between Plant-Based Dietary Patterns and Risk of Type 2 Diabetes. JAMA Intern. Med. 2019. [Google Scholar] [CrossRef]
- Barnard, N.D.; Levin, S.M.; Yokoyama, Y. A Systematic Review and Meta-Analysis of Changes in Body Weight in Clinical Trials of Vegetarian Diets. J. Acad. Nutr. Diet. 2015, 115, 954–969. [Google Scholar] [CrossRef]
- Turner-McGrievy, G.; Mandes, T.; Crimarco, A. A plant-based diet for overweight and obesity prevention and treatment. J. Geriatr. Cardiol. 2017, 14, 369–374. [Google Scholar]
- Naghavi, M.; Abajobir, A.A.; Abbafati, C.; Abbas, K.M.; Abd-Allah, F.; Abera, S.F.; Aboyans, V.; Adetokunboh, O.; Afshin, A.; Agrawal, A.; et al. Global, regional, and national age-sex specific mortality for 264 causes of death, 1980–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1151–1210. [Google Scholar] [CrossRef]
- Roth, G.A.; Johnson, C.; Abajobir, A.; Abd-Allah, F.; Abera, S.F.; Abyu, G.; Ahmed, M.; Aksut, B.; Alam, T.; Alam, K.; et al. Global, Regional, and National Burden of Cardiovascular Diseases for 10 Causes, 1990 to 2015. J. Am. Coll. Cardiol. 2017, 70, 1–25. [Google Scholar] [CrossRef]
- Ferdowsian, H.R.; Barnard, N.D. Effects of plant-based diets on plasma lipids. Am. J. Cardiol. 2009, 104, 947–956. [Google Scholar] [CrossRef]
- GBD 2015 Obesity Collaborators; Afshin, A.; Forouzanfar, M.H.; Reitsma, M.B.; Sur, P.; Estep, K.; Lee, A.; Marczak, L.; Mokdad, A.H.; Moradi-Lakeh, M.; et al. Health Effects of Overweight and Obesity in 195 Countries over 25 Years. N. Engl. J. Med. 2017, 377, 13–27. [Google Scholar] [CrossRef]
- Piepoli, M.F.; Hoes, A.W.; Agewall, S.; Albus, C.; Brotons, C.; Catapano, A.L.; Cooney, M.-T.; Corrà, U.; Cosyns, B.; Deaton, C.; et al. 2016 European Guidelines on cardiovascular disease prevention in clinical practice. Eur. Heart J. 2016, 37, 2315–2381. [Google Scholar] [CrossRef]
- Anand, S.S.; Hawkes, C.; de Souza, R.J.; Mente, A.; Dehghan, M.; Nugent, R.; Zulyniak, M.A.; Weis, T.; Bernstein, A.M.; Krauss, R.M.; et al. Food Consumption and its Impact on Cardiovascular Disease: Importance of Solutions Focused on the Globalized Food System: A Report from the Workshop Convened by the World Heart Federation. J. Am. Coll. Cardiol. 2015, 66, 1590–1614. [Google Scholar] [CrossRef]
- Appel, L.J. Lifestyle modification as a means to prevent and treat high blood pressure. J. Am. Soc. Nephrol. 2003, 14, S99–S102. [Google Scholar] [CrossRef]
- Yokoyama, Y.; Levin, S.M.; Barnard, N.D. Association between plant-based diets and plasma lipids: A systematic review and meta-analysis. Nutr. Rev. 2017, 75, 683–698. [Google Scholar] [CrossRef]
- Yokoyama, Y.; Nishimura, K.; Barnard, N.D.; Takegami, M.; Watanabe, M.; Sekikawa, A.; Okamura, T.; Miyamoto, Y. Vegetarian Diets and Blood Pressure. JAMA Intern. Med. 2014, 174, 577. [Google Scholar] [CrossRef] [PubMed]
- Lopez, P.D.; Cativo, E.H.; Atlas, S.A.; Rosendorff, C. The Effect of Vegan Diets on Blood Pressure in Adults: A Meta-Analysis of Randomized Controlled Trials. Am. J. Med. 2019, 132, 875–883. [Google Scholar] [CrossRef] [PubMed]
- Jakše, B.; Pinter, S.; Jakše, B.; Bučar Pajek, M.; Pajek, J. Effects of an Ad Libitum Consumed Low-Fat Plant-Based Diet Supplemented with Plant-Based Meal Replacements on Body Composition Indices. BioMed Res. Int. 2017, 2017, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Jakše, B.; Jakše, B.; Pajek, J.; Pajek, M. Effects of ad libitum consumed, low-fat, high-fiber plant-basediet supplemented with plant-based meal replacements on cardiovascular risk factors. Food Nutr. Res. 2019, 63. [Google Scholar] [CrossRef]
- Wright, N.; Wilson, L.; Smith, M.; Duncan, B.; McHugh, P. The BROAD study: A randomised controlled trial using a whole food plant-based diet in the community for obesity, ischaemic heart disease or diabetes. Nutr. Diabetes 2017, 7, e256. [Google Scholar] [CrossRef]
- Barnard, N.D.; Cohen, J.; Jenkins, D.J.A.; Turner-McGrievy, G.; Gloede, L.; Green, A.; Ferdowsian, H. A low-fat vegan diet and a conventional diabetes diet in the treatment of type 2 diabetes: A randomized, controlled, 74-wk clinical trial. Am. J. Clin. Nutr. 2009, 89, 1588S–1596S. [Google Scholar] [CrossRef]
- Hall, K.D.; Ayuketah, A.; Brychta, R.; Cai, H.; Cassimatis, T.; Chen, K.Y.; Chung, S.T.; Costa, E.; Courville, A.; Darcey, V.; et al. Ultra-Processed Diets Cause Excess Calorie Intake and Weight Gain: An Inpatient Randomized Controlled Trial of Ad Libitum Food Intake. Cell Metab. 2019, 30, 67–77. [Google Scholar] [CrossRef]
- Campbell, T.C.; Campbell, T.M. The China Study: The Most Comprehensive Study of Nutrition Ever Conducted and the Startling Implications for Diet, Weight Loss and Long-Term Health; BenBella Books: Dallas, TX, USA, 2005; ISBN 1932100660. [Google Scholar]
- Ashley, J.M.; Herzog, H.; Clodfelter, S.; Bovee, V.; Schrage, J.; Pritsos, C. Nutrient adequacy during weight loss interventions: A randomized study in women comparing the dietary intake in a meal replacement group with a traditional food group. Nutr. J. 2007, 6, 12. [Google Scholar] [CrossRef]
- Johansson, K.; Neovius, M.; Hemmingsson, E. Effects of anti-obesity drugs, diet, and exercise on weight-loss maintenance after a very-low-calorie diet or low-calorie diet: A systematic review and meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2014, 99, 14–23. [Google Scholar] [CrossRef]
- EFSA. Scientific Opinion on the substantiation of health claims related to meal replacements for weight control (as defined in Directive 96/8/EC on energy restricted diets for weight loss) and reduction in body weight (ID 1417), and maintenance of body weight a. EFSA J. 2010, 8, 1466. [Google Scholar]
- Raynor, H.A.; Champagne, C.M. Position of the Academy of Nutrition and Dietetics: Interventions for the Treatment of Overweight and Obesity in Adults. J. Acad. Nutr. Diet. 2016, 116, 129–147. [Google Scholar] [CrossRef]
- Heymsfield, S.B.; van Mierlo, C.A.J.; van der Knaap, H.C.M.; Heo, M.; Frier, H.I. Weight management using a meal replacement strategy: Meta and pooling analysis from six studies. Int. J. Obes. 2003, 27, 537–549. [Google Scholar] [CrossRef]
- Ross, L.J.; Wallin, S.; Osland, E.J.; Memon, M.A. Commercial Very Low Energy Meal Replacements for Preoperative Weight Loss in Obese Patients: A Systematic Review. Obes. Surg. 2016, 26, 1343–1351. [Google Scholar] [CrossRef]
- Shao, A. Role of Meal Replacements on Weight Management, Health and Nutrition. In Superfood and Functional Food—An Overview of Their Processing and Utilization; InTech: Vienna, Austria, 2017. [Google Scholar]
- NIJZ. Picture Material for Determining Food Intake. Available online: https://www.nijz.si/sl/publikacije/slikovno-gradivo-za-dolocanje-vnosa-zivil (accessed on 28 July 2019).
- OPKP. Computer Web-Based Software: The Open Platform for Clinical Nutrition (OPEN). Available online: http://www.opkp.si/sl_SI/fooddiary/diary (accessed on 28 July 2019).
- IJS JSI—Institut “Jozef Stefan”. Available online: https://www.ijs.si/ijsw/JSI (accessed on 11 November 2019).
- AISBL, E. EuroFIR European Food Information Resource. Available online: http://www.eurofir.org/ (accessed on 28 July 2019).
- Bognár, A. Tables on Weight Yield of Food and Retention Factors of Food Constituents for the Calculation of Nutrient Composition of Cooked Foods (Dishes); BFE Karlsruhe: Karlsruhe, Germany, 2002. [Google Scholar]
- ResPons Pretehtajte Prehranska Dopolnila. Available online: https://pretehtajte.si/ (accessed on 28 July 2019).
- German Nutrition Society; Austrian Nutrition Society; Society for Nutrition Research, S.N. 3. Ergaenzlieferung D-A-CH Referenzwerte für die Nährstoffzufuhr [Reference Values for Nutrient Intake]; 2. Auflage, 4. Aktualisierte Ausgabe, 4th, 3rd ed.; German Nutrition Society: Frankfurt am Main, Germany, 2018. [Google Scholar]
- EFSA. Dietary Reference Values for Nutrients Summary Report; Wiley Online Library: New York, NY, USA, 2017; Volume 14. [Google Scholar]
- Williams, B.; Mancia, G.; Spiering, W.; Agabiti Rosei, E.; Azizi, M.; Burnier, M.; Clement, D.; Coca, A.; De Simone, G.; Dominiczak, A.; et al. 2018 Practice Guidelines for the management of arterial hypertension of the European Society of Cardiology and the European Society of Hypertension. J. Hypertens. 2018, 36, 2284–2309. [Google Scholar] [CrossRef]
- Mach, F.; Baigent, C.; Catapano, A.L.; Koskinas, K.C.; Casula, M.; Badimon, L.; Chapman, M.J.; De Backer, G.G.; Delgado, V.; Ference, B.A.; et al. 2019 ESC/EAS Guidelines for the management of dyslipidaemias: Lipid modification to reduce cardiovascular risk. Eur. Heart J. 2019, 290, 140–205. [Google Scholar]
- Wickham, H.; François, R.; Henry, L.; Müller, K. A Grammar of Data Manipulation. R Package Version 0.8.1. Available online: https://cran.r-project.org/web/packages/dplyr/index.html (accessed on 30 September 2019).
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis, 2nd ed.; Springer: Berlin/Heidelberg, Germany, 2016; ISBN 9783319242774. [Google Scholar]
- Heinzen, E.; Sinnwell, J.; Atkinson, E.; Gunderson, T.; Votruba, P.; Dougherty, G.; Lennon, R.; Hanson, A.; Goergen, K.; Lundt, E.; et al. An Arsenal of “R” Functions for Large-Scale Statistical Summaries. [R Package Arsenal Version 3.3.0]. Available online: https://cran.r-project.org/web/packages/arsenal/index.html (accessed on 30 September 2019).
- Allès, B.; Baudry, J.; Méjean, C.; Touvier, M.; Péneau, S.; Hercberg, S.; Kesse-Guyot, E. Comparison of Sociodemographic and Nutritional Characteristics between Self-Reported Vegetarians, Vegans, and Meat-Eaters from the NutriNet-Santé Study. Nutrients 2017, 9, 1023. [Google Scholar] [CrossRef]
- Desideri, G.; Castaldo, G.; Lombardi, A.; Mussap, M.; Testa, A.; Pontremoli, R.; Punzi, L.; Borghi, C. Is it time to revise the normal range of serum uric acid levels? Eur. Rev. Med. Pharmacol. Sci. 2014, 18, 1295–1306. [Google Scholar]
- WHO. Haemoglobin Concentrations for the Diagnosis of Anaemia and Assessment of Severity. Vitamin and Mineral Nutrition Information System; (WHO/NMH/NHD/MNM/11.1); World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- Paul, C.; Brady, D.M. Comparative Bioavailability and Utilization of Particular Forms of B12 Supplements with Potential to Mitigate B12-related Genetic Polymorphisms. Integr. Med. 2017, 16, 42–49. [Google Scholar]
- Reynolds, A.; Mann, J.; Cummings, J.; Winter, N.; Mete, E.; Te Morenga, L. Carbohydrate quality and human health: A series of systematic reviews and meta-analyses. Lancet 2019, 393, 434–445. [Google Scholar] [CrossRef]
- Engel, M.G.; Kern, H.J.; Brenna, J.T.; Mitmesser, S.H. Micronutrient Gaps in Three Commercial Weight-Loss Diet Plans. Nutrients 2018, 10, 108. [Google Scholar] [CrossRef] [PubMed]
- Karlsen, M.; Rogers, G.; Miki, A.; Lichtenstein, A.; Folta, S.; Economos, C.; Jacques, P.; Livingston, K.; McKeown, N.; Karlsen, M.C.; et al. Theoretical Food and Nutrient Composition of Whole-Food Plant-Based and Vegan Diets Compared to Current Dietary Recommendations. Nutrients 2019, 11, 625. [Google Scholar] [CrossRef] [PubMed]
- Cho, S.K.; Chang, Y.; Kim, I.; Ryu, S. U-Shaped Association Between Serum Uric Acid Level and Risk of Mortality: A Cohort Study. Arthritis Rheumatol. 2018, 70, 1122–1132. [Google Scholar] [CrossRef] [PubMed]
- Wang, F.; Zheng, J.; Yang, B.; Jiang, J.; Fu, Y.; Li, D. Effects of Vegetarian Diets on Blood Lipids: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J. Am. Heart Assoc. 2015, 4, e002408. [Google Scholar] [CrossRef] [PubMed]
- Benatar, J.R.; Stewart, R.A.H. Cardiometabolic risk factors in vegans; A meta-analysis of observational studies. PLoS ONE 2018, 13, e0209086. [Google Scholar] [CrossRef]
- Fontana, L.; Meyer, T.E.; Klein, S.; Holloszy, J.O. Long-Term Low-Calorie Low-Protein Vegan Diet and Endurance Exercise are Associated with Low Cardiometabolic Risk. Rejuvenation Res. 2007, 10, 225–234. [Google Scholar] [CrossRef]
- De Biase, S.G.; Fernandes, S.F.C.; Gianini, R.J.; Duarte, J.L.G. Vegetarian diet and cholesterol and triglycerides levels. Arq. Bras. Cardiol. 2007, 88, 32–36. [Google Scholar] [CrossRef]
- Campbell, T.C.; Parpia, B.; Chen, J. Diet, lifestyle, and the etiology of coronary artery disease: The Cornell China Study. In American Journal of Cardiology; Elsevier Inc.: Amsterdam, The Netherland, 1998; Volume 82. [Google Scholar]
- Lee, C.J.; Kim, J.Y.; Shim, E.; Hong, S.H.; Lee, M.; Jeon, J.Y.; Park, S. The Effects of diet alone or in combination with exercise in patients with prehypertension and hypertension: A randomized controlled trial. Korean Circ. J. 2018, 48, 637. [Google Scholar] [CrossRef]
- Alexander, S.; Ostfeld, R.J.; Allen, K.; Williams, K.A. A plant-based diet and hypertension. J. Geriatr. Cardiol. 2017, 14, 327–330. [Google Scholar]
- Deutschman, A. Change or Die: The Three Keys to Change at Work and in Life; Regan: Jefferson City, MS, USA, 2007; ISBN 0061373672. [Google Scholar]
- Campbell, E.K.; Fidahusain, M.; Campbell, T.M., II. Evaluation of an Eight-Week Whole-Food Plant-Based Lifestyle Modification Program. Nutrients 2019, 11, 2068. [Google Scholar] [CrossRef] [PubMed]
- Barnard, N.D.; Levin, S.M.; Gloede, L.; Flores, R. Turning the Waiting Room into a Classroom: Weekly Classes Using a Vegan or a Portion-Controlled Eating Plan Improve Diabetes Control in a Randomized Translational Study. J. Acad. Nutr. Diet. 2018, 118, 1072–1079. [Google Scholar] [CrossRef] [PubMed]
- Ornish, D.; Lin, J.; Chan, J.M.; Epel, E.; Kemp, C.; Weidner, G.; Marlin, R.; Frenda, S.J.; Magbanua, M.J.M.; Daubenmier, J.; et al. Effect of comprehensive lifestyle changes on telomerase activity and telomere length in men with biopsy-proven low-risk prostate cancer: 5-year follow-up of a descriptive pilot study. Lancet Oncol. 2013, 14, 1112–1120. [Google Scholar] [CrossRef]
- Karlsen, M.C.; Pollard, K.J. Strategies for practitioners to support patients in plant-based eating. J. Geriatr. Cardiol. 2017, 14, 338–341. [Google Scholar] [PubMed]
- Bradbury, K.E.; Guo, W.; Cairns, B.J.; Armstrong, M.E.G.; Key, T.J. Association between physical activity and body fat percentage, with adjustment for BMI: A large cross-sectional analysis of UK Biobank. BMJ Open 2017, 7, e011843. [Google Scholar] [CrossRef] [PubMed]
- Johns, D.J.; Hartmann-Boyce, J.; Jebb, S.A.; Aveyard, P. Diet or Exercise Interventions vs Combined Behavioral Weight Management Programs: A Systematic Review and Meta-Analysis of Direct Comparisons. J. Acad. Nutr. Diet. 2014, 114, 1557–1568. [Google Scholar] [CrossRef] [PubMed]
- Albarrati, A.M.; Alghamdi, M.S.M.; Nazer, R.I.; Alkorashy, M.M.; Alshowier, N.; Gale, N. Effectiveness of Low to Moderate Physical Exercise Training on the Level of Low-Density Lipoproteins: A Systematic Review. BioMed Res. Int. 2018, 2018, 5982980. [Google Scholar] [CrossRef]
- Hollis, J.F.; Gullion, C.M.; Stevens, V.J.; Brantley, P.J.; Appel, L.J.; Ard, J.D.; Champagne, C.M.; Dalcin, A.; Erlinger, T.P.; Funk, K.; et al. Weight loss during the intensive intervention phase of the weight-loss maintenance trial. Am. J. Prev. Med. 2008, 35, 118–126. [Google Scholar] [CrossRef]
- Hoffman, S.R.; Stallings, S.F.; Bessinger, R.C.; Brooks, G.T. Differences between health and ethical vegetarians. Strength of conviction, nutrition knowledge, dietary restriction, and duration of adherence. Appetite 2013, 65, 139–144. [Google Scholar] [CrossRef]
| Nutrition | Physical Activity (PA) | Support System |
|---|---|---|
| Supplemented WFPB diet | Healthy and active lifestyle | Included a social-media component |
| WFPB diet ≥90% of the energy intake | Habitual PA (part of daily life) Organized (guided or prescribed, free) Other PA | Facebook Messenger e- mails |
| PB MR ≤10% of the energy intake | Resistance-exercise activity (own body mass or fitness): 45 min: 2–3 times/week Brisk walking: 30 min/day Brisk walking or hiking: 1–2 h/weekend | Meal recipes (process, pictures, video) Health and nutrition topics Motivation Discussion board |
| Dietary Supplements † Vitamins: B12, D3, EPA and DHA n-3 | Grouped Individual Fit challenges | Practical advice Meal plan evaluation (daily or weekly) Grocery shopping tour (three supermarkets) Cooking class (three hours) |
| Individually optimised meal plan Adjusted after 4–6 weeks: Details on food preparation, combinations, raw/cooked ratio | Health General fitnessBody composition | Follow-up Body-composition measurements Personal consultations Regular medical monitoring (at baseline, 3rd month, yearly) |
| 60-min lecture/week for 10 weeks | No unhealthy or extreme PA | Constant and dynamic improvement |
| Healthful, ad libitum, tasteful, affordable | Independence of participants | No meditation or systematic stress techniques introduced |
| Group 1 (n = 51) | Group 2 (n = 56) | Group 3 (n = 44) | p-Value | ||||
|---|---|---|---|---|---|---|---|
| Macronutrients (Per Day) | Female | Male | Female | Male | Female | Male | F/M |
| Energy intake (kcal) | 1963 (501) | 2683 (2069, 2968) | 1734 (529) | 2502 (2195, 2971) | 1851 (577) | 2832 (2123, 3056) | 0.174/0.900 |
| Carbohydrates (g) | 284 (80) | 366 (280, 412) | 250 (85) | 389 (312, 448) | 270 (96) | 364 (310, 460) | 0.222/0.517 |
| (% E) | 58 (6) | 53 (50, 59) | 57 (4) | 58 (55, 62) | 58 (6) | 58 (53, 65) | 0.685/0.115 |
| Total sugars (g) | 86 (32) | 96 (76, 119) | 74 (23) | 119 (83, 130) | 84 (48) | 90 (65, 98) | 0.233/0.305 |
| (% E) | 18 (5) | 16 (13, 18) | 17 (4) | 17 (15, 19) | 18 (5) | 13 (10, 17) | 0.963/0.095 |
| Free sugars (g) | 19 (18) | 21 (15, 27) | 21 (11) | 29 (19, 36) | 26 (24) | 20 (17, 28) | 0.304/0.197 |
| (% E) | 4 (3) | 3 (2, 4) | 5 (2) | 4 (3, 5) | 5 (3) | 3 (3, 4) | 0.042/0.172 |
| Starches (g) | 90 (45) | 129 (78, 164) | 78 (47) | 119 (86, 154) | 79 (42) | 145 (108, 165) | 0.439/0.680 |
| (% E) | 18 (7) | 19 (15, 22) | 17 (7) | 19 (15, 20) | 17 (6) | 19 (18, 28) | 0.698/0.611 |
| Dietary fibre (g) | 68 (16) | 90 (69, 102) | 61 (16) | 80 (71, 90) | 63 (17) | 80 (72, 96) | 0.144/0.665 |
| (% E) | 7 (1) | 7 (6, 7) | 7 (1) | 6 (5, 7) | 7 (1) | 7 (6, 7) | 0.237/0.254 |
| Fat (g) | 44 (18) | 67 (47, 86) | 38 (13) | 49 (37, 64) | 40 (17) | 57 (44, 81) | 0.305/0.401 |
| (% E) | 20 (5) | 24 (19, 28) | 20 (4) | 19 (13, 21) | 19 (5) | 20 (15, 25) | 0.695/0.155 |
| SFA (g) | 7 (2) | 11 (8, 14) | 6 (3) | 8 (6, 11) | 6 (2) | 10 (7, 14) | 0.849/0.356 |
| (% E) | 3 (1) | 3 (3, 4) | 3 (1) | 3 (3, 3) | 3 (1) | 3 (3, 4) | 0.530/0.340 |
| MUFA (g) | 12 (6) | 17 (14, 22) | 11 (4) | 13 (10, 21) | 10 (5) | 18 (15, 18) | 0.317/0.559 |
| (% E) | 5 (2) | 7 (5, 8) | 6 (2) | 4 (4, 7) | 5 (2) | 5 (5, 7) | 0.094/0.404 |
| PUFA (g) | 20 (8) | 29 (21, 45) | 16 (6) | 24 (15, 28) | 17 (7) | 23 (16, 37) | 0.017/0.358 |
| (% E) | 9 (3) | 11 (8, 13) | 8 (2) | 7 (6, 9) | 8 (2) | 8 (6, 11) | 0.067/0.047 a,b |
| Linoleic acid (g) | 13 (7) | 21 (11, 31) | 10 (5) | 10 (10, 20) | 11 (5) | 16 (11, 29) | 0.050/0.430 |
| Alpha-linolenic acid (g) | 6 (3) | 7 (5, 13) | 4 (2) | 5 (4, 6) | 4 (3) | 6 (3, 8) | 0.026/0.084 |
| EPA (mg) | 333 (284) | 375 (104, 377) | 350 (176) | 378 (250, 379) | 307 (242) | 375 (253, 377) | 0.739/0.436 |
| DHA (mg) | 235 (209) | 250 (70, 250) | 235 (119) | 250 (250, 459) | 205 (162) | 250 (172, 250) | 0.696/0.531 |
| ARA (mg) | 2 (7) | 0.1 (0, 2) | 2 (6) | 0 (0, 0.4) | 2 (5) | 0.1 (0, 7) | 0.980/0.156 |
| Cholesterol (mg) | 8 (24) | 0.01 (0, 5) | 4 (8) | 0 (0, 20) | 7 (20) | 0 (0, 0.3) | 0.657/0.971 |
| Protein (g) | 73 (20) | 96 (82, 114) | 67 (18) | 98 (91, 115) | 72 (18) | 111 (82, 118) | 0.330/0.903 |
| (% E) | 15 (2) | 14 (14, 16) | 16 (2) | 16 (15, 17) | 16 (2) | 15 (14, 16) | 0.087/0.308 |
| (g/kg body mass) | 1.1 (0.4) | 1.3 (1.1, 1.5) | 1.1 (0.3) | 1.3 (1, 1.3) | 1.2 (0.3) | 1.2 (0.9, 1.5) | 0.1300.916 |
| Alcohol (mL/day) | 0.3 (1.0) | 0 | 0 | 0 | 0.2 (1.2) | 0 | 0.323/0.973 |
| Water w (L) | 3.8 (1.1) | 3.6 (3.2, 4) | 4.0 (1.2) | 4.5 (3.8, 5.7) | 4.0 (1.3) | 4.4 (4.1, 5.7) | 0.652/0.024 a |
| Group 1 (n = 51) | Group 2 (n = 56) | Group 3 (n = 44) | p-Value | ||||
|---|---|---|---|---|---|---|---|
| Micronutrients (Per Day) | Female | Male | Female | Male | Female | Male | F/M |
| Vitamins | |||||||
| Thiamine (mg) | 2.7 (1.1) | 3.1 (2.7, 3.7) | 2.8 (0.8) | 3.5 (2.9, 3.7) | 3.2 (2.1) | 3.5 (3.3, 3.9) | 0.199/0.460 |
| Riboflavin (mg) | 2.5 (1.6) | 2.1 (1.8, 3.3) | 3.1 (1.6) | 2.4 (2.1, 3.7) | 3.5 (2.2) | 2.2 (2.0, 4.6) | 0.083/0.676 |
| Niacin (mg) | 28 (8) | 34 (26, 36) | 30 (7) | 33 (28, 39) | 31 (10) | 34 (31, 45) | 0.286/0.787 |
| Pantothenic acid (mg) | 10 (3) | 12 (10, 12) | 11 (3) | 12 (10, 15) | 11 (4) | 13 (11, 15) | 0.381/0.676 |
| Vitamin B6 (pyridoxine) (mg) | 3.7 (1.2) | 4.7 (3.4, 5.0) | 3.7 (1.0) | 4.5 (3.4, 5.4) | 4.2 (1.8) | 4.9 (3.9, 5.5) | 0.141/0.857 |
| Biotin (µg) | 93 (33) | 106 (84, 127) | 98 (37) | 96 (74, 126) | 111 (48) | 116 (86, 134) | 0.169/0.897 |
| Folate (µg folate equivalent) | 757 (215) | 925 (705, 1033) | 761 (210) | 887 (810, 1199) | 772 (219) | 798 (729, 1016) | 0.959/0.570 |
| Vitamin B12 (µg) | 218 (270) | 287 (4, 361) | 230 (319) | 288 (144, 430) | 304 (375) | 288 (287, 289) | 0.501/0.370 |
| Vitamin C (mg) | 346 (230) | 331 (247, 431) | 322 (97) | 279 (261, 410) | 336 (154) | 319 (248, 413) | 0.822/0.891 |
| Retinol equivalents re (mg) | 3.7 (1.3) | 3.9 (3.1, 4.8) | 3.4 (1.6) | 3.6 (3.1, 4.4) | 3.8 (2.9) | 4.6 (2.8, 5.4) | 0.726/0.690 |
| Vitamin D (µg) | 8.4 (5.9) | 5.9 (3.8, 10.2) | 13.1 (14.4) | 5.4 (4.1, 8.0) | 12.9 (8.8) | 7.7 (4.8, 12.3) | 0.118/0.496 |
| Vitamin E (mg) | 26 (12) | 29 (20, 33) | 28 (10) | 22 (21, 25) | 30 (14) | 29 (24, 36) | 0.462/0.582 |
| Minerals | |||||||
| Calcium (mg) | 1012 (313) | 1155 (887, 1401) | 991 (213) | 1173 (1021, 1475) | 1054 (361) | 1235 (981, 1531) | 0.659/0.754 |
| Magnesium (mg) | 902 (311) | 987 (835, 1220) | 801 (223) | 883 (804, 1287) | 819 (351) | 1086 (833, 1145) | 0.294/0.914 |
| Phosphorus (mg) | 1714 (434) | 2189 (1720, 2489) | 1623 (402) | 2201 (1889, 2542) | 1662 (404) | 2382 (1792, 2556) | 0.632/0.762 |
| Potassium (mg) | 4759 (1232) | 6177 (4074, 6605) | 4329 (1220) | 6235 (5229, 6835) | 4635 (1394) | 5968 (5120, 6845) | 0.309/0.761 |
| Sodium (mg) | 1956 (921) | 2160 (1563, 3260) | 2013 (1054) | 1729 (1492, 2366) | 1875 (779) | 1945 (1806, 2684) | 0.824/0.558 |
| Chloride (mg) | 3181 (1423) | 3580 (2703, 4890) | 3208 (1664) | 2781 (2597, 4024) | 3070 (1193) | 3179 (2876, 4748) | 0.918/0.500 |
| Trace Elements | |||||||
| Iron (mg) | 38 (31) | 40 (32, 46) | 33 (7) | 38 (36, 46) | 34 (10) | 40 (36, 42) | 0.528/0.984 |
| Copper (mg) | 3.8 (1.0) | 5.2 (4.1, 5.9) | 3.7 (1.1) | 4.3 (3.1, 5.4) | 3.8 (1.1) | 4.9 (4.1, 5.8) | 0.831/0.679 |
| Iodine (µg) | 215 (75) | 238 (165, 290) | 234 (69) | 238 (198, 282) | 243 (82) | 253 (196, 312) | 0.295/0.717 |
| Zinc (mg) | 19 (6) | 24 (18, 26) | 19 (4) | 21 (19, 24) | 19 (6) | 24 (21, 27) | 0.897/0.819 |
| Chrome (µg) | 82 (27) | 77 (53, 104) | 77 (28) | 77 (50, 110) | 82 (38) | 74 (49, 81) | 0.743/0.711 |
| Molybdenum (µg) | 106 (80) | 68 (48, 115) | 86 (51) | 86 (39, 125) | 90 (56) | 58 (42, 100) | 0.363/0.780 |
| Selenium (µg) | 110 (49) | 127 (119, 150) | 114 (43) | 117 (85, 147) | 111 (41) | 120 (107, 136) | 0.931/0.538 |
| Country | Slovenia (Present Study) | Finland [16] | France [66] | |||
|---|---|---|---|---|---|---|
| Year of Publication | 2019 | 2016 | 2017 | |||
| Food Groups (g Per Day) | Whole Sample (n = 151) | Females (n = 109) | Males (n = 42) | p-Values | Whole Sample (n = 22) | Whole Sample (n = 789) |
| Foods of Vegetable Origin | F vs. M | |||||
| Vegetables (unprocessed) | 455 (190) | 433 (175) | 510 (219) | 0.040 | 277 | 366.2 |
| Fruits (unprocessed) | 363 (187) | 358 (179) | 377 (209) | 0.618 | 254 | 364 |
| Grain and products | 178 (114) | 147 (90) | 257 (131) | <0.001 | 248 a | 232 c |
| Legumes | 166 (115) | 143 (94) | 226 (140) | <0.001 | 156 b | 73.2 |
| Potatoes | 140 (123) | 130 (115) | 165 (139) | 0.217 | 151 | 58 |
| Nuts and seeds | 52 (46) | 44 (29) | 74 (69) | 0.042 | 11 | 52.6 d |
| Bread and bakery products | 43 (50) | 40 (43) | 50 (64) | 0.896 | - | - |
| PB Meal replacement | 43 (72) | 48 (79) | 28 (50) | 0.145 | - | - |
| Spices and herbs | 32 (40) | 35 (44) | 24 (24) | 0.054 | - | - |
| Fruits (processed) | 20 (33) | 18 (27) | 25 (44) | 0.687 | 137 f | - |
| Pasta | 17 (35) | 12 (25) | 29 (50) | 0.163 | 25 | - |
| Fast food and ready meals | 6 (34) | 4 (18) | 12 (58) | 0.505 | - | - |
| Vegetables (processed) | 5 (23) | 5 (24) | 4 (20) | 0.860 | - | - |
| Sweet products | 3 (11) | 3 (13) | 1 (4) | 0.577 | 20 | 108.3 e |
| Alcohol drinks | 1 (12) | 2 (14) | 0.0 | 0.279 | - | 88.8 |
| Vegetable oil and fat | 1 (4) | 1 (4) | 2 (5) | 0.409 | 51 h | 14.5 |
| Sweeteners | 0.2 (0.8) | 0.1 (0.7) | 0.2 (1.1) | 0.524 | - | - |
| Foods of Animal Origin | ||||||
| Sea fish and products | 3 (18) | 2 (13) | 6 (26) | 0.359 | 0 | 12.8 |
| Red meat | 0.6 (0.6) | 0.3 (2.3) | 1.4 (9.2) | 0.817 | 0 | 18.3 |
| White meat | 0.5 (5.3) | 0.7 (5.5) | 0.0 | 0.378 | 0 | - |
| Meat product | 0.2 (2.3) | 0.3 (2.7) | 0.0 | 0.378 | 0 | 5.8 |
| Dairy products | 0.1 (3) | 0.5 (3.4) | 0.3 (2.1) | 0.703 | 7 g | 45 |
| Milk | 0.1 (1.6) | 0.2 (1.9) | 0.0 | 0.535 | 59 g | - |
| Eggs and products | 0.0 | 0.0 | 0.0 | NaN | - | 5.4 |
| Animal fats | 0.0 | 0.0 | 0.0 | NaN | - | 9.5 |
| Parameter | Group 1 (n = 51) | Group 2 (n = 56) | Group 3 (n = 44) | p-Value | |||
|---|---|---|---|---|---|---|---|
| Gender (F/M) | Female | Male | Female | Male | Female | Male | F/M |
| Laboratory Variables | |||||||
| S-cholesterol (mmol/L) | 3.7 (0.8) | 3.8 (0.8) | 3.5 (0.8) | 0.231 * | |||
| 3.9 (0.8) | 3.3 (2.7, 3.9) | 3.9 (0.8) | 3.7 (3.1, 4.2) | 3.7 (0.7) | 2.7 (2.6, 3.4) | 0.495/0.283 | |
| LDL-cholesterol (mmol/L) | 2.1 (0.6) | 2.2 (0.7) | 1.8 (0.6) | 0.010 | |||
| 2.2 (0.6) | 1.9 (1.5, 2.4) | 2.1 (0.7) | 2.2 (1.9, 2.6) | 1.8 (0.5) | 1.6 (1.4, 1.9) | 0.025/0.233 | |
| HDL-cholesterol (mmol/L) | 1.4 (0.4) | 1.4 (0.3) | 1.4 (0.4) | 0.391 * | |||
| 1.5 (0.4) | 1.1 (1.0, 1.2) | 1.5 (0.3) | 1.1 (1.0, 1.2) | 1.6 (0.3) | 1.1 (1.0, 1.2) | 0.233/0.988 | |
| Triglycerides (mmol/L) | 0.9 (0.4) | 0.8 (0.3) | 0.8 (0.4) | 0.648 * | |||
| 0.8 (0.3) | 0.9 (0.7, 1.4) | 0.8 (0.3) | 0.9 (0.7, 1.1) | 0.8 (0.3) | 0.8 (0.6, 0.9) | 0.933/0.681 | |
| S-Uric Acid (μmol/L) | 288 (70) | 266 (67) | 264 (65) | 0.124 * | |||
| 257 (51) | 352 (312, 385) | 238 (41) | 371 (331, 387) | 240 (59) | 304 (294, 362) | 0.194/0.065 | |
| Haemoglobin (g/L) | 141 (12) | 142 (13) | 142 (11) | 0.850 * | |||
| 135 (9) | 154 (150, 157) | 137 (11) | 156 (150, 162) | 139 (10) | 146 (143, 157) | 0.191/0.160 | |
| Blood Pressure (mmHg) | |||||||
| Systolic | 116 (11) | 114 (11) | 115 (10) | 0.832 * | |||
| 114 (12) | 119 (111, 123) | 112 (11) | 121 (117, 124) | 113 (10) | 117 (115, 127) | 0.697/0.674 | |
| Diastolic | 70 (8) | 72 (10) | 72 (7) | 0.384 * | |||
| 70 (8) | 71 (64, 76) | 71 (11) | 76 (72, 79) | 72 (6) | 75 (69, 76) | 0.615/0.220 | |
| Parameter | Whole Sample (n = 151) | ||
|---|---|---|---|
| Gender (F/M) | Female (n = 109) | Male (n = 42) | p-Value |
| Hypothesis (Normal/High) | F vs. M | ||
| LDL-Cholesterol | <3 mmol/L [62] | ||
| Normal | 140 (93%) | ||
| High | 11 (7%) | ||
| Normal | 102 (94%) | 38 (91%) | 0.500 |
| High | 7 (6%) | 4 (9%) | 0.500 |
| Triglycerides | <1.7 mmol/L [62] | ||
| Normal | 146 (97%) | ||
| High | 5 (3%) | ||
| Normal | 108 (99%) | 38 (91%) | 0.021 |
| High | 1 (1%) | 4 (9%) | 0.021 |
| Blood Pressure | ≤129/84 mmHg [61] | ||
| Systolic | ≤129 mmHg | ||
| Normal | 133 (88%) | ||
| High | 18 (12%) | ||
| Normal | 100 (92%) | 33 (79%) | 0.046 |
| High | 9 (8%) | 9 (21%) | 0.046 |
| Diastolic | ≤84 mmHg | ||
| Normal | 143 (95%) | ||
| High | 8 (5%) | ||
| Normal | 101 (93%) | 42 (100%) | 0.107 |
| High | 8 (7%) | 0 (0.0%) | 0.107 |
| S-Uric Acid | Female: 154–357 mmol/L; Male: 308–428 mmol/L [67] | ||
| Normal | 132 (87%) | ||
| High | 5 (3%) | ||
| Low | 14 (9.3%) | ||
| Normal | 106 (97%) | 26 (62%) | <0.001 |
| High | 1 (1%) | 4 (9%) | <0.001 |
| Low | 2 (2%) | 12 (29%) | <0.001 |
| Haemoglobin | Non pregnant female: >120 g/L; Male: >130 g/L [68] | ||
| Normal | 146 (97%) | ||
| High | 5 (3%) | ||
| Normal | 104 (95%) | 42 (100.0%) | 0.323 |
| Low | 5 (5%) | 0 (0.0%) | 0.323 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jakše, B.; Jakše, B.; Pinter, S.; Jug, B.; Godnov, U.; Pajek, J.; Fidler Mis, N. Dietary Intakes and Cardiovascular Health of Healthy Adults in Short-, Medium-, and Long-Term Whole-Food Plant-Based Lifestyle Program. Nutrients 2020, 12, 55. https://doi.org/10.3390/nu12010055
Jakše B, Jakše B, Pinter S, Jug B, Godnov U, Pajek J, Fidler Mis N. Dietary Intakes and Cardiovascular Health of Healthy Adults in Short-, Medium-, and Long-Term Whole-Food Plant-Based Lifestyle Program. Nutrients. 2020; 12(1):55. https://doi.org/10.3390/nu12010055
Chicago/Turabian StyleJakše, Boštjan, Barbara Jakše, Stanislav Pinter, Borut Jug, Uroš Godnov, Jernej Pajek, and Nataša Fidler Mis. 2020. "Dietary Intakes and Cardiovascular Health of Healthy Adults in Short-, Medium-, and Long-Term Whole-Food Plant-Based Lifestyle Program" Nutrients 12, no. 1: 55. https://doi.org/10.3390/nu12010055
APA StyleJakše, B., Jakše, B., Pinter, S., Jug, B., Godnov, U., Pajek, J., & Fidler Mis, N. (2020). Dietary Intakes and Cardiovascular Health of Healthy Adults in Short-, Medium-, and Long-Term Whole-Food Plant-Based Lifestyle Program. Nutrients, 12(1), 55. https://doi.org/10.3390/nu12010055








