Carotenoids and Periodontal Infection
Abstract
1. Introduction
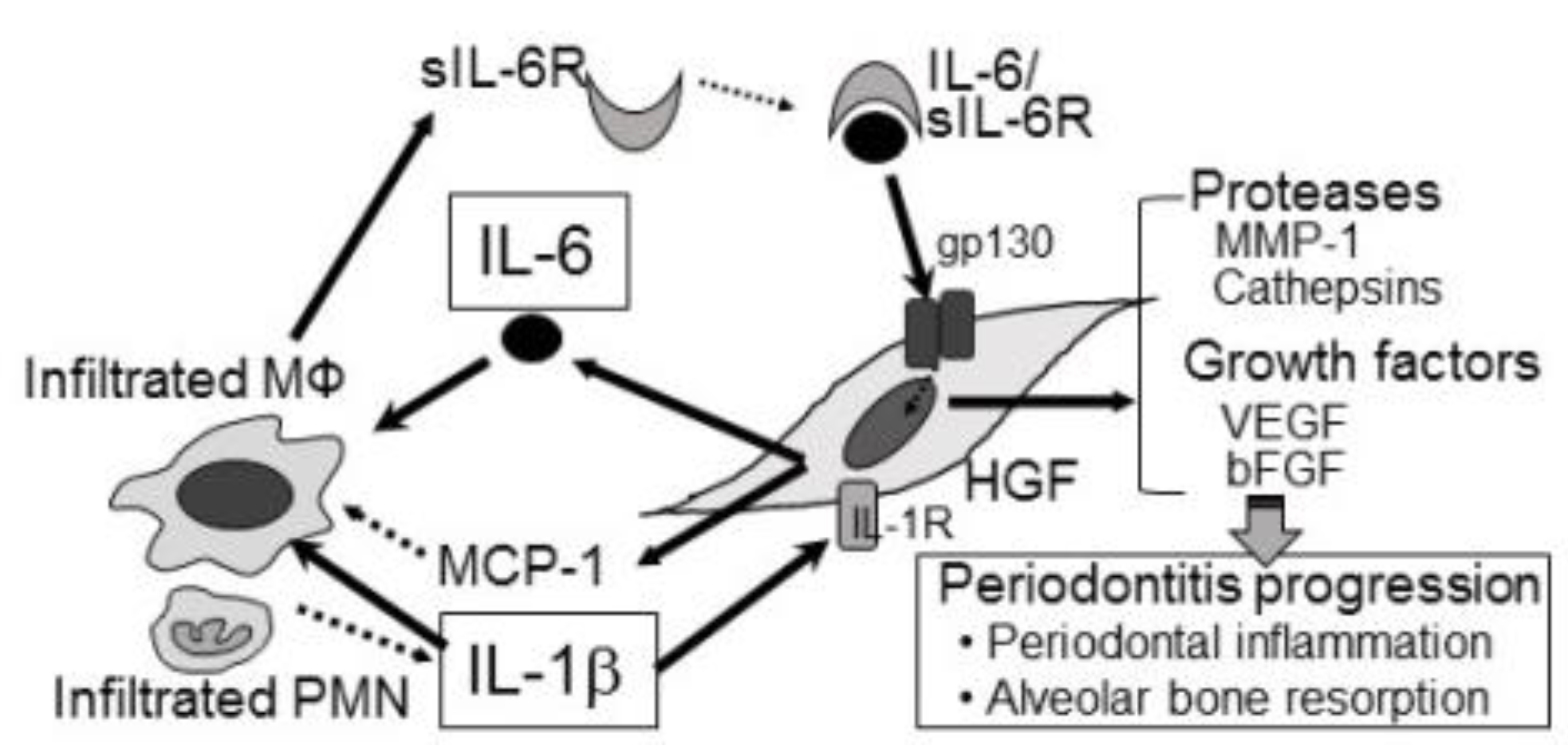
2. Pathogenesis of Periodontitis
2.1. Proteases and Periodontitis
2.2. Chemokines and Periodontitis
2.3. Growth Factors and Periodontitis
3. Carotenoids and Periodontitis
3.1. β-Carotene
3.2. β-Cryptoxanthin
3.3. Astaxanthin
3.4. Fucoxanthin
3.5. All-trans Retinoic Acid
3.6. Lycopene
3.7. Luteolin
4. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Darveau, R.P.; Tanner, A.; Page, R.C. The microbial challenge in periodontitis. Periodontol. 2000 2004, 14, 12–32. [Google Scholar] [CrossRef] [PubMed]
- Paster, B.J.; Olsen, I.; Aas, J.A.; Dewhirst, F.E. The breadth of bacterial diversity in the human periodontal pocket and other oral sites. Periodontol. 2000 2006, 42, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Aukhil, I.; Lopatin, D.E.; Syed, S.A.; Morrison, E.C.; Kowalski, C.J. The effects of periodontal therapy on serum antibody (IgG) levels to plaque microorganisms. J. Clin. Periodontol. 1988, 15, 544–550. [Google Scholar] [CrossRef] [PubMed]
- Zelkha, S.A.; Freilich, R.W.; Amar, S. Periodontal innate immune mechanisms relevant to atherosclerosis and obesity. Periodontol. 2000 2010, 54, 207–221. [Google Scholar] [CrossRef]
- Moutsopoulos, N.M.; Madianos, P.N. Low-grade inflammation in chronic infectious diseases: Paradigm of periodontal infections. Ann. N. Y. Acad. Sci. 2006, 1088, 251–264. [Google Scholar] [CrossRef]
- Rao, A.V.; Rao, L.G. Carotenoids and human health. Pharmacol. Res. 2007, 55, 207–216. [Google Scholar] [CrossRef]
- Birkedal-Hansen, H. Role of matrix metalloproteinases in human periodontal diseases. J. Periodontol. 1993, 64, 474–484. [Google Scholar]
- Hassell, T.M. Tissues and cells of the periodontium. Periodontol. 2000 1993, 3, 9–38. [Google Scholar] [CrossRef]
- Okada, H.; Murakami, S. Cytokine expression in periodontal health and disease. Crit. Rev. Oral Biol. Med. 1998, 9, 248–266. [Google Scholar] [CrossRef]
- Takashiba, S.; Naruishi, K.; Murayama, Y. Perspective of cytokine regulation for periodontal treatment: Fibroblast biology. J. Periodontol. 2003, 74, 103–110. [Google Scholar] [CrossRef]
- Sawada, S.; Chosa, N.; Ishisaki, A.; Naruishi, K. Enhancement of gingival inflammation induced by synergism of IL-1β and IL-6. Biomed. Res. 2013, 34, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Kida, Y.; Kobayashi, M.; Suzuki, T.; Takeshita, A.; Okamatsu, Y.; Hanazawa, S.; Yasui, T.; Hasegawa, K. Interleukin-1 stimulates cytokines, prostaglandin E2 and matrix metalloproteinase-1 production via activation of MAPK/AP-1 and NF-kappaB in human gingival fibroblasts. Cytokine 2005, 29, 159–168. [Google Scholar] [CrossRef]
- Emingil, G.; Han, B.; Gürkan, A.; Berdeli, A.; Tervahartiala, T.; Salo, T.; Pussinen, P.J.; Köse, T.; Atilla, G.; Sorsa, T. Matrix metalloproteinase (MMP)-8 and tissue inhibitor of MMP-1 (TIMP-1) gene polymorphisms in generalized aggressive periodontitis: Gingival crevicular fluid MMP-8 and TIMP-1 levels and outcome of periodontal therapy. J. Periodontol. 2014, 85, 1070–1080. [Google Scholar] [CrossRef]
- Naruishi, K.; Takashiba, S.; Chou, H.H.; Arai, H.; Nishimura, F.; Murayama, Y. Role of soluble interleukin-6 receptor in inflamed gingiva for binding of interleukin-6 to gingival fibroblasts. J. Periodontal. Res. 1999, 34, 296–300. [Google Scholar] [CrossRef]
- Naruishi, K.; Takashiba, S.; Nishimura, F.; Chou, H.H.; Arai, H.; Yamada, H.; Murayama, Y. Impairment of gingival fibroblast adherence by IL-6/sIL-6R. J. Dent. Res. 2001, 80, 1421–1424. [Google Scholar] [CrossRef] [PubMed]
- Naruishi, K.; Nishimura, F.; Yamada-Naruishi, H.; Omori, K.; Yamaguchi, M.; Takashiba, S. C-jun N-terminal kinase (JNK) inhibitor, SP600125, blocks interleukin (IL)-6-induced vascular endothelial growth factor (VEGF) production: Cyclosporine A partially mimics this inhibitory effect. Transplantation 2003, 76, 1380–1382. [Google Scholar] [CrossRef] [PubMed]
- Holden, J.A.; Attard, T.J.; Laughton, K.M.; Mansell, A.; O’Brien-Simpson, N.M.; Reynolds, E.C. Porphyromonas gingivalis lipopolysaccharide weakly activates M1 and M2 polarized mouse macrophages but induces inflammatory cytokines. Infect. Immun. 2014, 82, 4190–4203. [Google Scholar] [CrossRef] [PubMed]
- Sima, C.; Glogauerm, M. Macrophage subsets and osteoimmunology: Tuning of the immunological recognition and effector systems that maintain alveolar bone. Periodontol. 2000 2013, 63, 80–101. [Google Scholar] [CrossRef]
- Reynolds, J.J. Collagenases and tissue inhibitors of metalloproteinases: A functional balance in tissue degradation. Oral Dis. 1996, 2, 70–76. [Google Scholar] [CrossRef]
- Cox, S.W.; Rodriguez-Gonzalez, E.M.; Booth, V.; Eley, B.M. Secretory leukocyte protease inhibitor and its potential interactions with elastase and cathepsin B in gingival crevicular fluid and saliva from patients with chronic periodontitis. J. Periodontal. Res. 2006, 41, 477–485. [Google Scholar] [CrossRef]
- Trabandt, A.; Müller-Ladner, U.; Kriegsmann, J.; Gay, R.E.; Gay, S. Expression of proteolytic cathepsins B, D, and L in periodontal gingival fibroblasts and tissues. Lab. Investig. 1995, 73, 205–212. [Google Scholar]
- Yamaguchi, T.; Naruishi, K.; Arai, H.; Nishimura, F.; Takashiba, S. IL-6/sIL-6R enhances cathepsin B and L production via caveolin-1-mediated JNK-AP-1 pathway in human gingival fibroblasts. J. Cell Physiol. 2008, 217, 423–432. [Google Scholar] [CrossRef]
- Hashimoto, N.; Kawabe, T.; Hara, T.; Imaizumi, K.; Wakayama, H.; Saito, H.; Shimokata, K.; Hasegawa, Y. Effect of erythromycin on matrix metalloproteinase-9 and cell migration. J. Lab. Clin. Med. 2001, 137, 176–183. [Google Scholar] [CrossRef]
- Gamonal, J.; Acevedo, A.; Bascones, A.; Jorge, O.; Silva, A. Levels of interleukin-1beta, -8, and -10 and RANTES in gingival crevicular fluid and cell populations in adult periodontitis patients and the effect of periodontal treatment. J. Periodontol. 2000, 70, 1535–1545. [Google Scholar] [CrossRef] [PubMed]
- Taylor, J.J. Cytokine regulation of immune responses to Porphyromonas gingivalis. Periodontol. 2000 2010, 54, 160–194. [Google Scholar] [CrossRef]
- Rosenkilde, M.M.; Schwartz, T.W. The chemokine system—A major regulator of angiogenesis in health and disease. APMIS 2001, 112, 481–495. [Google Scholar] [CrossRef]
- Ferrara, N. Vascular endothelial growth factor and the regulation of angiogenesis. Recent Prog. Horm. Res. 2000, 55, 15–35. [Google Scholar]
- Nalla, A.K.; Gorantla, B.; Gondi, C.S.; Lakka, S.S.; Rao, J.S. Targeting MMP-9, uPAR, and cathepsin B inhibits invasion, migration and activates apoptosis in prostate cancer cells. Cancer Gene Ther. 2010, 17, 599–613. [Google Scholar] [CrossRef] [PubMed]
- Yanamandra, N.; Gumidyala, K.V.; Waldron, K.G.; Gujrati, M.; Olivero, W.C.; Dinh, D.H.; Rao, J.S. Blockade of cathepsin B expression in human glioblastoma cells is associated with suppression of angiogenesis. Oncogene 2004, 23, 2224–2230. [Google Scholar] [CrossRef] [PubMed]
- Yuan, K.; Jin, Y.T.; Lin, M.T. The detection and comparison of angiogenesis-associated factors in pyogenic granuloma by immunohistochemistry. J. Periodontol. 2000, 71, 701–709. [Google Scholar] [CrossRef] [PubMed]
- Demyanets, S.; Huber, K.; Wojta, J. Vascular effects of glycoprotein130 ligands—Part I: Pathophysiological role. Vasc. Pharm. 2012, 56, 34–46. [Google Scholar] [CrossRef]
- Hirano, T.; Ishihara, K.; Hibi, M. Roles of STAT3 in mediating the cell growth, differentiation and survival signals relayed through the IL-6 family of cytokine receptors. Oncogene 2000, 19, 2548–2556. [Google Scholar] [CrossRef] [PubMed]
- Kajiura, Y.; Nishikawa, Y.; Lew, J.H.; Kido, J.I.; Nagata, T.; Naruishi, K. β-carotene suppresses Porphyromonas gingivalis lipopolysaccharide-mediated cytokine production in THP-1 monocytes cultured with high glucose condition. Cell Biol. Int. 2018, 42, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, C.; Ashida, N.; Yokoyama, S.; Tominari, T.; Hirata, M.; Ogawa, K.; Ugiura, M.S.; Yano, M.; Inada, M.; Miyaura, C. The protective effects of β-cryptoxanthin on inflammatory bone resorption in a mouse experimental model of periodontitis. Biosci. Biotechnol. Biochem. 2013, 77, 860–862. [Google Scholar] [CrossRef] [PubMed]
- Nishigaki, M.; Yamamoto, T.; Ichioka, H.; Honjo, K.; Yamamoto, K.; Oseko, F.; Kita, M.; Mazda, O.; Kanamura, N. β-cryptoxanthin regulates bone resorption related-cytokine production in human periodontal ligament cells. Arch. Oral Biol. 2013, 58, 880–886. [Google Scholar] [CrossRef] [PubMed]
- Yoshizawa, S.; Meguro, M.; Ohyama, H.; Takeuchi-Hatanaka, K.; Matsushita, S.; Takashiba, S.; Nishimura, F. Focal adhesion kinase mediates human leukocyte histocompatibility antigen class II-induced signaling in gingival fibroblasts. J. Periodontal. Res. 2007, 42, 572–579. [Google Scholar] [CrossRef][Green Version]
- Balci, Y.H.; Lektemur, A.A.; Gevrek, F.; Toker, H. Investigation of the effect of astaxanthin on alveolar bone loss in experimental periodontitis. J. Periodontal. Res. 2018, 53, 131–138. [Google Scholar] [CrossRef]
- Kose, O.; Arabaci, T.; Yemenoglu, H.; Kara, A.; Ozkanlar, S.; Kayis, S.; Duymus, Z.Y. Influences of Fucoxanthin on alveolar bone resorption in induced periodontitis in rat molars. Mar. Drugs. 2016, 14, 70. [Google Scholar] [CrossRef]
- Wang, L.; Wang, J.; Jin, Y.; Gao, H.; Lin, X. Oral administration of all-trans retinoic acid suppresses experimental periodontitis by modulating the Th17/Treg imbalance. J. Periodontol. 2014, 85, 740–750. [Google Scholar] [CrossRef]
- Ebersole, J.L.; Lambert, J.; Bush, H.; Huja, P.E.; Basu, A. Serum nutrient levels and aging effects on periodontitis. Nutrients 2018, 10, 1986. [Google Scholar] [CrossRef]
- Dodington, D.W.; Fritz, P.C.; Sullivan, P.J.; Ward, W.E. Higher intakes of fruits and vegetables, β-carotene, vitamin C, α-tocopherol, EPA, and DHA are positively associated with periodontal healing after nonsurgical periodontal therapy in nonsmokers but not in smokers. J. Nutr. 2015, 145, 2512–2519. [Google Scholar] [CrossRef] [PubMed]
- Arora, N.; Avula, H.; Avula, J.K. The adjunctive use of systemic antioxidant therapy (lycopene) in nonsurgical treatment of chronic periodontitis: A short-term evaluation. Quintessence Int. 2013, 44, 395–405. [Google Scholar] [PubMed]
- Belludi, S.A.; Verma, S.; Banthia, R.; Bhusari, P.; Parwani, S.; Kedia, S.; Saiprasad, S.V. Effect of lycopene in the treatment of periodontal disease: A clinical study. J. Contemp. Dent. Pract. 2013, 14, 1054–1059. [Google Scholar] [CrossRef] [PubMed]
- Chandra, R.V.; Sandhya, Y.P.; Nagarajan, S.; Reddy, B.H.; Naveen, A.; Murthy, K.R. Efficacy of lycopene as a locally delivered gel in the treatment of chronic periodontitis: Smokers vs nonsmokers. Quintessence Int. 2012, 43, 401–411. [Google Scholar]
- Higuchi, K.; Saito, I.; Maruyama, K.; Eguchi, E.; Mori, H.; Tanno, S.; Sakurai, S.; Kishida, T.; Nishida, W.; Osawa, H.; et al. Associations of serum β-carotene and retinol concentrations with insulin resistance: The Toon Health Study. Nutrition 2015, 31, 975–980. [Google Scholar] [CrossRef]
- Amengual, J.; Gouranton, E.; van Helden, Y.G.; Hessel, S.; Ribot, J.; Kramer, E.; Kiec-Wilk, B.; Razny, U.; Lietz, G.; Wyss, A.; et al. Beta-carotene reduces body adiposity of mice via BCMO1. PLoS ONE 2011, 6, e20644. [Google Scholar] [CrossRef]
- Hirata, N.; Ichimaru, R.; Tominari, T.; Matsumoto, C.; Watanabe, K.; Taniguchi, K.; Hiratam, M.; Ma, S.; Suzuki, K.; Grundler, F.M.W.; et al. Beta-Cryptoxanthin inhibits lipopolysaccharide-induced osteoclast differentiation and bone resorption via the suppression of inhibitor of NF-κB kinase activity. Nutrients 2019, 11, 368. [Google Scholar] [CrossRef]
- Hwang, Y.H.; Kim, K.J.; Kim, S.J.; Mun, S.K.; Hong, S.G.; Son, Y.J.; Yee, S.T. Suppression effect of Astaxanthin on osteoclast formation in vitro and bone loss in vivo. Int. J. Mol. Sci. 2018, 19, 912. [Google Scholar] [CrossRef]
- Li, D.; Wang, P.; Wang, P.; Hu, X.; Chen, F. Targeting the gut microbiota by dietary nutrients: A new avenue for human health. Crit. Rev. Food Sci. Nutr. 2019, 59, 181–195. [Google Scholar] [CrossRef]
- Salehi, B.; Sharifi-Rad, R.; Sharopov, F.; Namiesnik, J.; Roointan, A.; Kamle, M.; Kumar, P.; Martins, N.; Sharifi-Rad, J. Beneficial effects and potential risks of tomato consumption for human health: An overview. Nutrition 2019, 62, 201–208. [Google Scholar] [CrossRef]
- Imran, M.; Rauf, A.; Abu-Izneid, T.; Nadeemd, M.; Shariati, M.A.; Khan, I.A.; Imran, A.; ErdoganOrhan, I.; Rizwan, M.; Atif, M. Luteolin, a flavonoid, as an anticancer agent: A review. Biomed. Pharmacother. 2019, 112, 108612. [Google Scholar] [CrossRef] [PubMed]
- Ohyama, H.; Nishimura, F.; Meguro, M.; Takashiba, S.; Murayama, Y.; Matsushita, S. Counter-antigen presentation: Fibroblasts produce cytokines by signalling through HLA class II molecules without inducing T-cell proliferation. Cytokine 2002, 17, 175–181. [Google Scholar] [CrossRef] [PubMed]
| Study Designs | Carotenoids | Target | Inhibitory Effects | Ref |
|---|---|---|---|---|
| In vitro | β-Carotene | THP-1 monocytes | Inhibition of Pg LPS-induced TNF, IL-6 and MCP-1 production | [33] |
| β-Cryptoxanthin | Human gingival fibroblasts | LPS-induced osteoclasts differentiation via inhibition of PGE2 production | [33] | |
| Co-cultures of bone marrow cells and osteoblasts | Inhibition of LPS-induced osteoclast formation | [34] | ||
| Human periodontal ligament cells | Inhibition of Pg-induced IL-6 and IL-8 production | [35] | ||
| Luteolin | Human gingival fibroblasts | Inhibition of HLA-DR-induced IL-6 and MCP-1 production | [36] | |
| In vivo | β-Cryptoxanthin | Experimental periodontitis | Inhibition of alveolar bone loss | [33] |
| Inhibition of alveolar bone loss | [34] | |||
| Astaxanthin | Inhibition of osteoclastic activity | [37] | ||
| Fucoxanthin | Decrease of blood TNF, IL-1β and IL-6 levels | [38] | ||
| No effects of alveolar bone loss | [38] | |||
| All-trans retinoic acid | Decrease of blood IL-17 levels | [39] | ||
| Inhibition of alveolar bone loss | [39] | |||
| Clinical study | β-Carotene | NHANES (1999–2004), total N = 15,844 | Lower blood levels | [40] |
| Periodontitis patients, N = 86 | Adjunctive effects of periodontal therapy | [41] | ||
| β-Cryptoxanthin | NHANES (1999–2004), total N = 15,844 | Lower blood levels | [40] | |
| Lycopene | Periodontitis patients, N = 42 | Inhibition of salivary IL-1 levels | [42] | |
| Periodontitis patients, N = 20 | Improvement of periodontal healing | [43] | ||
| Periodontitis patients, N = 100 | Decrease of periodontal probing depth | [44] |
© 2020 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Naruishi, K. Carotenoids and Periodontal Infection. Nutrients 2020, 12, 269. https://doi.org/10.3390/nu12010269
Naruishi K. Carotenoids and Periodontal Infection. Nutrients. 2020; 12(1):269. https://doi.org/10.3390/nu12010269
Chicago/Turabian StyleNaruishi, Koji. 2020. "Carotenoids and Periodontal Infection" Nutrients 12, no. 1: 269. https://doi.org/10.3390/nu12010269
APA StyleNaruishi, K. (2020). Carotenoids and Periodontal Infection. Nutrients, 12(1), 269. https://doi.org/10.3390/nu12010269





