The Role of Vitamin D in Primary Headache–from Potential Mechanism to Treatment
Abstract
1. Introduction
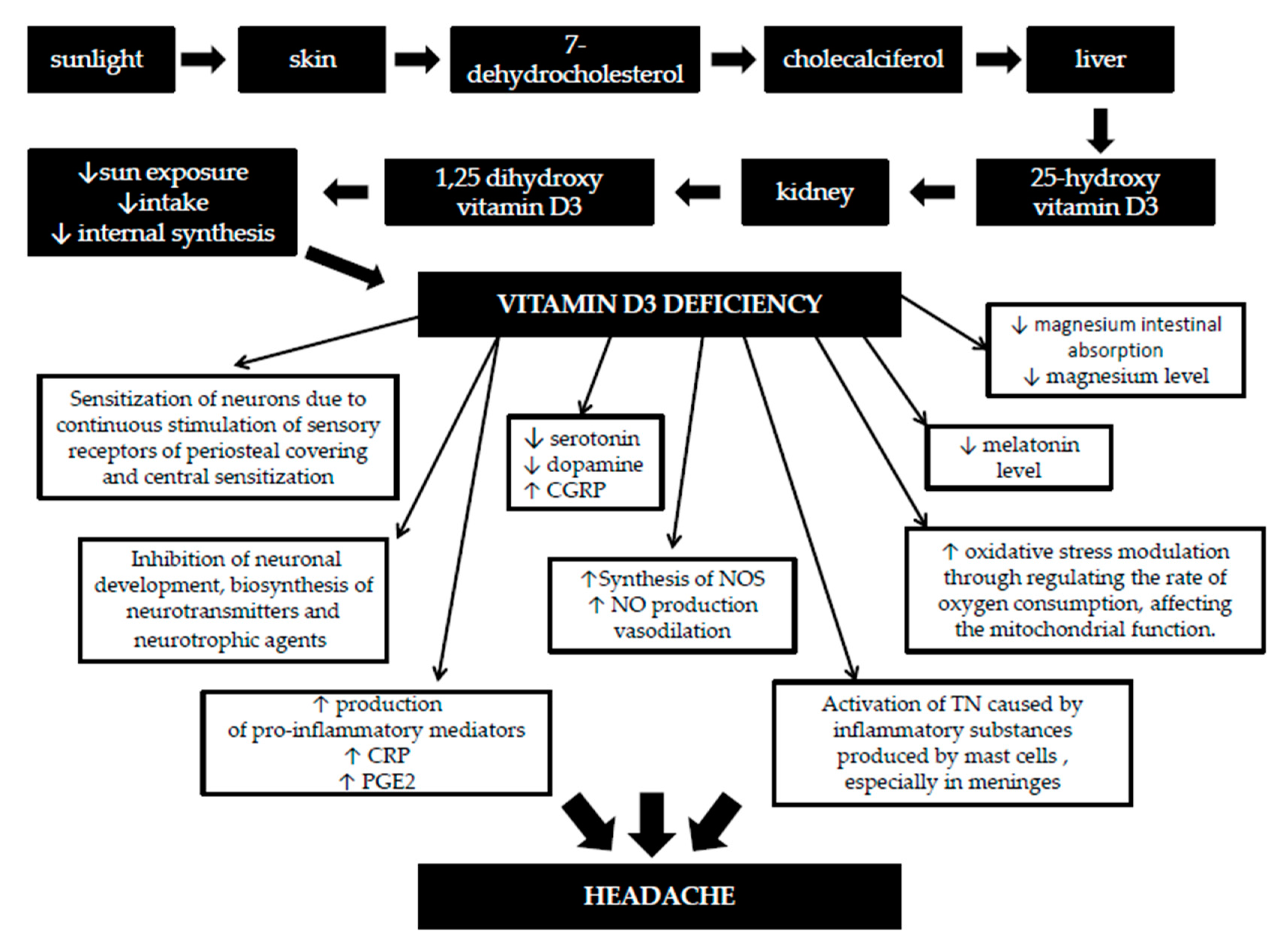
1.1. Metabolism and Functions of Vitamin D
1.2. The Role of Vitamin D in the Brain
1.3. Vitamin D Deficiency and Pain
1.4. The Link between Vitamin D and Headache
2. Methods
3. Results and Discussion
3.1. Vitamin D Deficiency and Tension-Type Headaches
3.2. The Role of Vitamin D in Migraine
3.3. Vitamin D in Cluster Headache
3.4. Association of Vitamin D with Trigeminal Neuralgia and Other Types of Primary Headache
3.5. Benefits of Vitamin D Supplementation in Headache
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd edition. Cephalalgia 2018, 38, 1–211. [Google Scholar] [CrossRef] [PubMed]
- Collaborators, G.H. Global, regional, and national burden of migraine and tension-type headache, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2018, 17, 954–976. [Google Scholar] [CrossRef]
- Wei, D.Y.; Yuan Ong, J.J.; Goadsby, P.J. Cluster Headache: Epidemiology, Pathophysiology, Clinical Features, and Diagnosis. Ann. Indian Acad. Neurol. 2018, 21, S3–S8. [Google Scholar] [CrossRef] [PubMed]
- Zakrzewska, J.M.; Linskey, M.E. Trigeminal neuralgia. BMJ Clin. Evid. 2014, 348, g474. [Google Scholar] [CrossRef] [PubMed]
- Tölle, T.; Dukes, E.; Sadosky, A. Patient burden of trigeminal neuralgia: Results from a cross-sectional survey of health state impairment and treatment patterns in six European countries. Pain Pract. 2006, 6, 153–160. [Google Scholar] [CrossRef]
- Stovner, L.J.; Zwart, J.A.; Hagen, K.; Terwindt, G.M.; Pascual, J. Epidemiology of headache in Europe. Eur. J. Neurol. 2006, 13, 333–345. [Google Scholar] [CrossRef]
- Abu Bakar, N.; Tanprawate, S.; Lambru, G.; Torkamani, M.; Jahanshahi, M.; Matharu, M. Quality of life in primary headache disorders: A review. Cephalalgia 2016, 36, 67–91. [Google Scholar] [CrossRef]
- Nattagh-Eshtivani, E.; Sani, M.A.; Dahri, M.; Ghalichi, F.; Ghavami, A.; Arjang, P.; Tarighat-Esfanjani, A. The role of nutrients in the pathogenesis and treatment of migraine headaches: Review. Biomed. Pharmacother. 2018, 102, 317–325. [Google Scholar] [CrossRef]
- Martin, V.T.; Vij, B. Diet and Headache: Part 2. Headache 2016, 56, 1553–1562. [Google Scholar] [CrossRef]
- Orr, S.L. The Evidence for the Role of Nutraceuticals in the Management of Pediatric Migraine: A Review. Curr. Pain Headache Rep. 2018, 22, 37. [Google Scholar] [CrossRef]
- Holick, M.F. The vitamin D deficiency pandemic: Approaches for diagnosis, treatment and prevention. Rev. Endocr. Metab. Disord. 2017, 18, 153–165. [Google Scholar] [CrossRef]
- Autier, P.; Boniol, M.; Pizot, C.; Mullie, P. Vitamin D status and ill health: A systematic review. Lancet Diabetes Endocrinol. 2014, 2, 76–89. [Google Scholar] [CrossRef]
- Lee, J.H.; O’Keefe, J.H.; Bell, D.; Hensrud, D.D.; Holick, M.F. Vitamin D deficiency an important, common, and easily treatable cardiovascular risk factor? J. Am. Coll. Cardiol. 2008, 52, 1949–1956. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M.; Society, E. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- Eyles, D.W.; Smith, S.; Kinobe, R.; Hewison, M.; McGrath, J.J. Distribution of the vitamin D receptor and 1 alpha-hydroxylase in human brain. J. Chem. Neuroanat. 2005, 29, 21–30. [Google Scholar] [CrossRef]
- Trochoutsou, A.I.; Kloukina, V.; Samitas, K.; Xanthou, G. Vitamin-D in the Immune System: Genomic and Non-Genomic Actions. Mini Rev. Med. Chem. 2015, 15, 953–963. [Google Scholar] [CrossRef]
- Razzaque, M.S. Magnesium: Are We Consuming Enough? Nutrients 2018, 10, 1863. [Google Scholar] [CrossRef]
- Uwitonze, A.M.; Razzaque, M.S. Role of Magnesium in Vitamin D Activation and Function. J. Am. Osteopath. Assoc. 2018, 118, 181–189. [Google Scholar] [CrossRef]
- Martin, K.R.; Reid, D.M. Is there role for vitamin D in the treatment of chronic pain? Ther. Adv. Musculoskelet. Dis. 2017, 9, 131–135. [Google Scholar] [CrossRef]
- Hewison, M. Vitamin D and immune function: An overview. Proc. Nutr. Soc. 2012, 71, 50–61. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D and brain health: The need for vitamin D supplementation and sensible sun exposure. J. Intern. Med. 2015, 277, 90–93. [Google Scholar] [CrossRef]
- Prakash, S.; Kumar, M.; Belani, P.; Susvirkar, A.; Ahuja, S. Interrelationships between chronic tension-type headache, musculoskeletal pain, and vitamin D deficiency: Is osteomalacia responsible for both headache and musculoskeletal pain? Ann. Indian Acad. Neurol. 2013, 16, 650–658. [Google Scholar] [CrossRef]
- Karras, S.; Rapti, E.; Matsoukas, S.; Kotsa, K. Vitamin D in Fibromyalgia: A Causative or Confounding Biological Interplay? Nutrients 2016, 8, 343. [Google Scholar] [CrossRef]
- Knutsen, K.V.; Madar, A.A.; Brekke, M.; Meyer, H.E.; Natvig, B.; Mdala, I.; Lagerløv, P. Effect of vitamin D on musculoskeletal pain and headache: A randomized, double-blind, placebo-controlled trial among adult ethnic minorities in Norway. Pain 2014, 155, 2591–2598. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zhang, H.L.; Wu, J. Is headache related with vitamin D insufficiency? J. Headache Pain 2010, 11, 369, author reply 371. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Carlberg, C.; Haq, A. The concept of the personal vitamin D response index. J. Steroid Biochem. Mol. Biol. 2018, 175, 12–17. [Google Scholar] [CrossRef]
- LeBlanc, E.S.; Perrin, N.; Johnson, J.D.; Ballatore, A.; Hillier, T. Over-the-counter and compounded vitamin D: Is potency what we expect? JAMA Intern. Med. 2013, 173, 585–586. [Google Scholar] [CrossRef]
- Groves, N.J.; McGrath, J.J.; Burne, T.H. Vitamin D as a neurosteroid affecting the developing and adult brain. Annu. Rev. Nutr. 2014, 34, 117–141. [Google Scholar] [CrossRef]
- Wu, Z.; Malihi, Z.; Stewart, A.W.; Lawes, C.M.; Scragg, R. The association between vitamin D concentration and pain: A systematic review and meta-analysis. Public Health Nutr. 2018, 21, 2022–2037. [Google Scholar] [CrossRef]
- Kenis-Coskun, O.; Giray, E.; Gunduz, O.H.; Akyuz, G. The effect of Vitamin D replacement on spinal inhibitory pathways in women with chronic widespread pain. J. Steroid Biochem. Mol. Biol. 2019, 105488. [Google Scholar] [CrossRef]
- Straube, S.; Derry, S.; Straube, C.; Moore, R.A. Vitamin D for the treatment of chronic painful conditions in adults. Cochrane Database Syst. Rev. 2015. [Google Scholar] [CrossRef] [PubMed]
- Gaikwad, M.; Vanlint, S.; Mittinity, M.; Moseley, G.L.; Stocks, N. Does vitamin D supplementation alleviate chronic nonspecific musculoskeletal pain? A systematic review and meta-analysis. Clin. Rheumatol. 2017, 36, 1201–1208. [Google Scholar] [CrossRef] [PubMed]
- Hewison, M. Antibacterial effects of vitamin D. Nat. Rev. Endocrinol. 2011, 7, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Nelson, A.; Wang, X.; Farid, M.; Gunji, Y.; Ikari, J.; Iwasawa, S.; Basma, H.; Feghali-Bostwick, C.; Rennard, S.I. Vitamin D modulates prostaglandin E2 synthesis and degradation in human lung fibroblasts. Am. J. Respir. Cell Mol. Biol. 2014, 50, 40–50. [Google Scholar] [CrossRef] [PubMed]
- Helde-Frankling, M.; Björkhem-Bergman, L. Vitamin D in Pain Management. Int. J. Mol. Sci. 2017, 18, 2170. [Google Scholar] [CrossRef]
- Gendelman, O.; Itzhaki, D.; Makarov, S.; Bennun, M.; Amital, H. A randomized double-blind placebo-controlled study adding high dose vitamin D to analgesic regimens in patients with musculoskeletal pain. Lupus 2015, 24, 483–489. [Google Scholar] [CrossRef]
- Prakash, S.; Mehta, N.C.; Dabhi, A.S.; Lakhani, O.; Khilari, M.; Shah, N.D. The prevalence of headache may be related with the latitude: A possible role of Vitamin D insufficiency? J. Headache Pain 2010, 11, 301–307. [Google Scholar] [CrossRef]
- Goadsby, P.J.; Holland, P.R. An Update: Pathophysiology of Migraine. Neurol. Clin. 2019, 37, 651–671. [Google Scholar] [CrossRef]
- Levy, D.; Burstein, R.; Kainz, V.; Jakubowski, M.; Strassman, A.M. Mast cell degranulation activates a pain pathway underlying migraine headache. Pain 2007, 130, 166–176. [Google Scholar] [CrossRef]
- Burstein, R.; Noseda, R.; Borsook, D. Migraine: Multiple processes, complex pathophysiology. J. Neurosci. 2015, 35, 6619–6629. [Google Scholar] [CrossRef]
- Mottaghi, T.; Askari, G.; Khorvash, F.; Maracy, M.R. Effect of Vitamin D supplementation on symptoms and C-reactive protein in migraine patients. J. Res. Med. Sci. 2015, 20, 477–482. [Google Scholar] [PubMed]
- Mottaghi, T.; Khorvash, F.; Askari, G.; Maracy, M.R.; Ghiasvand, R.; Maghsoudi, Z.; Iraj, B. The relationship between serum levels of vitamin D and migraine. J. Res. Med. Sci. 2013, 18, S66–S70. [Google Scholar] [PubMed]
- Prakash, S.; Shah, N.D. Chronic tension-type headache with vitamin D deficiency: Casual or causal association? Headache 2009, 49, 1214–1222. [Google Scholar] [CrossRef] [PubMed]
- Kirkland, A.E.; Sarlo, G.L.; Holton, K.F. The Role of Magnesium in Neurological Disorders. Nutrients 2018, 10, 730. [Google Scholar] [CrossRef]
- Garcion, E.; Sindji, L.; Nataf, S.; Brachet, P.; Darcy, F.; Montero-Menei, C.N. Treatment of experimental autoimmune encephalomyelitis in rat by 1,25-dihydroxyvitamin D3 leads to early effects within the central nervous system. Acta Neuropathol. 2003, 105, 438–448. [Google Scholar] [CrossRef]
- Messlinger, K.; Lennerz, J.K.; Eberhardt, M.; Fischer, M.J. CGRP and NO in the trigeminal system: Mechanisms and role in headache generation. Headache 2012, 52, 1411–1427. [Google Scholar] [CrossRef]
- Motaghi, M.; Haghjooy Javanmard, S.; Haghdoost, F.; Tajadini, M.; Saadatnia, M.; Rafiee, L.; Zandifar, A. Relationship between vitamin D receptor gene polymorphisms and migraine without aura in an Iranian population. Biomed. Res. Int. 2013, 2013, 351942. [Google Scholar] [CrossRef]
- Fernández-de-Las-Peñas, C.; Cuadrado, M.L.; Arendt-Nielsen, L.; Ge, H.Y.; Pareja, J.A. Increased pericranial tenderness, decreased pressure pain threshold, and headache clinical parameters in chronic tension-type headache patients. Clin. J. Pain 2007, 23, 346–352. [Google Scholar] [CrossRef]
- Sohn, J.H.; Chu, M.K.; Park, K.Y.; Ahn, H.Y.; Cho, S.J. Vitamin D deficiency in patients with cluster headache: A preliminary study. J. Headache Pain 2018, 19, 54. [Google Scholar] [CrossRef]
- Gallelli, L.; Michniewicz, A.; Cione, E.; Squillace, A.; Colosimo, M.; Pelaia, C.; Fazio, A.; Zampogna, S.; Peltrone, F.; Iannacchero, R.; et al. 25-Hydroxy Vitamin D Detection Using Different Analytic Methods in Patients with Migraine. J. Clin. Med. 2019, 8, 895. [Google Scholar] [CrossRef]
- Patel, U.; Kodumuri, N.; Malik, P.; Kapoor, A.; Malhi, P.; Patel, K.; Saiyed, S.; Lavado, L.; Kapoor, V. Hypocalcemia and Vitamin D Deficiency amongst Migraine Patients: A Nationwide Retrospective Study. Medicina (Kaunas) 2019, 55, 407. [Google Scholar] [CrossRef] [PubMed]
- Kılıç, B.; Kılıç, M. Evaluation of Vitamin D Levels and Response to Therapy of Childhood Migraine. Medicina (Kaunas) 2019, 55, 321. [Google Scholar] [CrossRef] [PubMed]
- Hancı, F.; Kabakuş, N.; Türay, S.; Bala, K.A.; Dilek, M. The role of obesity and vitamin D deficiency in primary headaches in childhood. Acta Neurol. Belg. 2019. [Google Scholar] [CrossRef]
- Hussein, M.; Fathy, W.; Elkareem, R.M.A. The potential role of serum vitamin D level in migraine headache: A case–control study. Journal of Pain Research 2019, 12, 2529. [Google Scholar] [CrossRef]
- Togha, M.; Razeghi Jahromi, S.; Ghorbani, Z.; Martami, F.; Seifishahpar, M. Serum Vitamin D Status in a Group of Migraine Patients Compared With Healthy Controls: A Case-Control Study. Headache 2018, 58, 1530–1540. [Google Scholar] [CrossRef]
- Song, T.J.; Chu, M.K.; Sohn, J.H.; Ahn, H.Y.; Lee, S.H.; Cho, S.J. Effect of Vitamin D Deficiency on the Frequency of Headaches in Migraine. J. Clin. Neurol. 2018, 14, 366–373. [Google Scholar] [CrossRef]
- Donmez, A.; Orun, E.; Sonmez, F.M. Vitamin D status in children with headache: A case-control study. Clin. Nutr. ESPEN 2018, 23, 222–227. [Google Scholar] [CrossRef]
- Rapisarda, L.; Mazza, M.R.; Tosto, F.; Gambardella, A.; Bono, F.; Sarica, A. Relationship between severity of migraine and vitamin D deficiency: A case-control study. Neurol. Sci. 2018, 39, 167–168. [Google Scholar] [CrossRef]
- Farajzadeh, A.; Bathaie, S.Z.; Arabkheradmand, J.; Ghodsi, S.M.; Faghihzadeh, S. Different Pain States of Trigeminal Neuralgia Make Significant Changes in the Plasma Proteome and Some Biochemical Parameters: A Preliminary Cohort Study. J. Mol. Neurosci. 2018, 66, 524–534. [Google Scholar] [CrossRef]
- Prakash, S.; Rathore, C.; Makwana, P.; Dave, A.; Joshi, H.; Parekh, H. Vitamin D Deficiency in Patients With Chronic Tension-Type Headache: A Case-Control Study. Headache 2017, 57, 1096–1108. [Google Scholar] [CrossRef]
- Virtanen, J.K.; Giniatullin, R.; Mäntyselkä, P.; Voutilainen, S.; Nurmi, T.; Mursu, J.; Kauhanen, J.; Tuomainen, T.P. Low serum 25-hydroxyvitamin D is associated with higher risk of frequent headache in middle-aged and older men. Sci. Rep. 2017, 7, 39697. [Google Scholar] [CrossRef] [PubMed]
- Tozzi, E.; Boncristiano, A.; Antenucci, A.; Di Loreto, S.; Farello, G. P013. 25(OH)D Level and headache in children sample. J. Headache Pain 2015, 16, A84. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Iannacchero, R.; Costa, A.; Squillace, A.; Gallelli, L.; Cannistrà, U.; De Sarro, G. P060. Vitamin D deficiency in episodic migraine, chronic migraine and medication-overuse headache patients. J. Headache Pain 2015, 16, A184. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Buettner, C.; Burstein, R. Association of statin use and risk for severe headache or migraine by serum vitamin D status: A cross-sectional population-based study. Cephalalgia 2015, 35, 757–766. [Google Scholar] [CrossRef] [PubMed]
- Celikbilek, A.; Gocmen, A.Y.; Zararsiz, G.; Tanik, N.; Ak, H.; Borekci, E.; Delibas, N. Serum levels of vitamin D, vitamin D-binding protein and vitamin D receptor in migraine patients from central Anatolia region. Int. J. Clin. Pract. 2014, 68, 1272–1277. [Google Scholar] [CrossRef]
- Zandifar, A.; Masjedi, S.S.; Banihashemi, M.; Asgari, F.; Manouchehri, N.; Ebrahimi, H.; Haghdoost, F.; Saadatnia, M. Vitamin D status in migraine patients: A case-control study. Biomed. Res. Int. 2014, 2014, 514782. [Google Scholar] [CrossRef]
- Kjaergaard, M.; Eggen, A.E.; Mathiesen, E.B.; Jorde, R. Association between headache and serum 25-hydroxyvitamin D: The Tromsø Study: Tromsø 6. Headache 2012, 52, 1499–1505. [Google Scholar] [CrossRef]
- Gazerani, P.; Fuglsang, R.; Pedersen, J.G.; Sørensen, J.; Kjeldsen, J.L.; Yassin, H.; Nedergaard, B.S. A randomized, double-blinded, placebo-controlled, parallel trial of vitamin D. Curr. Med. Res. Opin. 2019, 35, 715–723. [Google Scholar] [CrossRef]
- Buettner, C.; Nir, R.R.; Bertisch, S.M.; Bernstein, C.; Schain, A.; Mittleman, M.A.; Burstein, R. Simvastatin and vitamin D for migraine prevention: A randomized, controlled trial. Ann. Neurol. 2015, 78, 970–981. [Google Scholar] [CrossRef]
- Yilmaz, R.; Salli, A.; Cingoz, H.T.; Kucuksen, S.; Ugurlu, H. Efficacy of vitamin D replacement therapy on patients with chronic nonspecific widespread musculoskeletal pain with vitamin D deficiency. Int. J. Rheum. Dis. 2016, 19, 1255–1262. [Google Scholar] [CrossRef]
- Batcheller, P. A Survey of Cluster Headache (CH) Sufferers Using Vitamin D3 as a CH Preventative (P1. 256); AAN Enterprises: Bengaluru, India, 2014. [Google Scholar]
- Cayir, A.; Turan, M.I.; Tan, H. Effect of vitamin D therapy in addition to amitriptyline on migraine attacks in pediatric patients. Braz. J. Med. Biol. Res. 2014, 47, 349–354. [Google Scholar] [CrossRef] [PubMed]
| Author (Year) | Study Design | Study Group: Type of Headache (Number of Participants) | Study Population Age (Years) | Mean Serum Vitamin D Levels (ng/mL) | Results | Association |
|---|---|---|---|---|---|---|
| Gallelli (2019) [50] | Prospective, single-blind, single-center, control-group | Migraine (n = 95) Control (n = 120) | Range: 13–54 | 15.4 | Serum vitamin D levels were lower than the normal range in patients with migraines and controls. Mean vitamin D values were significantly higher in the control group compared to the migraine group. | Associated |
| Patel (2019) [51] | Retrospective, cross-sectional | Migraine (n = 446, 446) | Range: 19–80 | No data | Vitamin D deficiency elevated the odds of major/extreme loss of function. There was higher prevalence and higher odds of migraine among vitamin D deficient patients compared to those with hypocalcemia or no-deficiency. | Associated |
| Kılıç (2019) [52] | Retrospective study | Migraine (n = 92) | Mean: 12.6 | No data | There was increased migraine frequency, duration, and PedMIDAS scores in those with vitamin D deficiency and insufficiency. Migraine frequency, duration, and PedMIDAS scores were significantly negatively related to serum vitamin D levels. | Associated |
| Hanci (2019) [53] | Retrospective, observational | Migraine (n = 165) TTH (n = 116) Control (n = 98) | Range: 5–17 | Migraine: 12.4 ± 7.7 TTH: 13.5 ± 9.9 Control: 13.4 ± 8.8 | No significant differences in mean vitamin D levels among the three groups | Not associated |
| Hussain (2019) [54] | Case-control | Migraine (n = 40) Control (n = 40) | 32.18 28.8 | Migraine: 32.11 Control: 41.86 | Vitamin D levels were significantly lower in those with migraines compared to controls The incidence of aura, allodynia, phonophobia/photophobia, autonomic manifestations, and resistance to medications was increased vitamin D deficient patients with migraines than those with normal vitamin D levels. Serum vitamin D levels were significantly negatively related to the frequency, duration, and severity of migraine headache attacks | Associated |
| Togha (2018) [55] | Case-control | Migraine (n = 70) Control (n = 70) | Mean: 37 | Migraine: 30 Control: 43 | There were more subjects with vitamin D deficiency and insufficiency in the migraine group (53.7%) than the control group (26.1%). Serum vitamin D levels were significantly negatively related to migraine headaches Serum vitamin D levels in those with chronic migraine were not different to those in subjects with episodic migraine Serum vitamin D levels showed no correlation with headache parameters. | Associated |
| Song (2018) [56] | Retrospective, observational | Migraine (n = 157) | Mean: 37 | 15.9 ± 7.4 | The majority (94.9%) of subjects with migraine had vitamin D insufficiency. Frequent monthly headache was 1.2 times more common in migraine patients with vitamin D deficiency than those without deficiency. | Associated |
| Donmez (2018) [57] | Retrospective, case-control | Migraine (n = 68) TTH (n = 79) Control (n = 69) | Mean: 12.2 | Migraine: 17.3 TTH: 16.9 Control: 25.8 | Serum vitamin D levels were significantly lower in the migraine and TTH groups compared with the control group. | Associated |
| Sohn (2018) [49] | Case-control | CH (n = 28) Migraine (n = 36) Control (n = 36) | CH: 38.2 Migraine: 35.1 Control: 35.4 | CH: 14.0 ± 3.9 Migraine: 14.7 ± 5.9 Control: 14.6 ± 7.4 | The majority (92.8%) of those with CH had vitamin D deficiency. There was no significant difference in vitamin D levels among patients with CH, migraine, or controls. Patients with a headache periodicity during winter to spring showed a trend of lower serum vitamin D levels than those with periodicity during summer to autumn. | Not associated |
| Rapisarda (2018) [58] | Case-control | CM (n = 100) EM (n = 34) Control (n = 38) | CM/EM: 41.4 Control: 47.6 | CM: 12.7 EM: 17.2 Control: 23.0 | Vitamin D deficiency was severe among headache patients (especially in those with CM) compared to healthy subjects. Vitamin D levels were negatively correlated with the number of days of headache (Pearson’s correlation coefficient: 0.506) | Associated |
| Farajzadeh (2018) [59] | Case-control | TN (n = 13) Control (n = 13) | Mean: 53.3 | TN: 22.61 Control: 39.80 | Vitamin D levels were significantly decreased in patients with TN (before and after microvascular decompression) compared to the control group. | Associated |
| Prakash (2017) [60] | Case-control | Chronic TTH (n = 100) Control (n = 100) | Chronic TTH: 35.63 Control: 36.86 | Chronic TTH: 14.7 Control: 27.4 | Serum vitamin D levels were significantly lower in Chronic TTH patients than in controls. Vitamin D deficiency was more prevalent in patients with Chronic TTH (71%) than controls (25%). Chronic TTH patients with vitamin D deficiency had a higher prevalence of musculoskeletal pain, muscle weakness, muscle and bone tenderness score, associated fatigue, and a more prolonged course. Serum vitamin D levels were positively correlated with the total muscle tenderness score. | Associated |
| Virtanen (2017) [61] | Cross-sectional | Self-reported frequent headache (n = 250) | Range: 42–60 | 38.3 nmol/L | Serum vitamin D levels were lower in subjects with frequent headaches than other participants. Serum vitamin D levels were inversely associated with frequent headaches. | Associated |
| Tozzi (2016) [62] | Cross-sectional | MWoA (n = 91) MWA (n = 32) TTH (n = 36) | Range: 5–18 | No data | Serum vitamin D levels were lower in children with MwoA than those with MWA and THH, albeit not significantly (p = 0.07). | Not associated |
| Iannacchero (2015) [63] | Observational | Migraine (n = 22) | Mean: 45.41 | 13.05 ± 5.70 | Vitamin D levels were similar among those with CM than those with EM Vitamin D levels were not significantly correlated with headache frequency. | Not associated |
| Buettner (2015) [64] | Cross-sectional | 5938 participants from the National Health and Nutrition Examination | No data | No data | People with serum vitamin D levels >57 nmol/l and use a statin had a lower prevalence of severe headache or migraine. | Associated |
| Prakash (2013) [22] | Observational | Chronic TTH (n = 71) | Mean: 38 | No data | Serum vitamin D levels were significantly associated with headache, musculoskeletal pain, and osteomalacia. Mean vitamin D levels were significantly lower in subjects suffering from daily headache compared to those with intermittent headaches. | Associated |
| Celikbilek (2014) [65] | Cross-sectional, prospective | Migraine (n = 52) Control (n = 49) | Migraine: 35.88 Control: 34.24 | Migraine: 38.08 Control: 48.03 | Serum vitamin D and VDR levels were significantly lower in migraine patients than controls. Serum VDBP levels were similar between the two groups. Serum vitamin D, VDBP, and VDR levels showed no correlated with headache characteristics. | Associated |
| Zandifar (2014) [66] | Case-control | Migraine (n = 105) Control (n = 110) | Migraine: 33.59 Control: 32.46 | Migraine: 13.55 ± 0.91 Control: 13.19 ± 1.19 | There was no significant difference in vitamin D levels among between case controls. Severity of headache was not related to vitamin D levels. | Not associated |
| Mottaghi (2013) [42] | Cross-sectional | Migraine (n = 76) | Mean: 33.1 | 23.3 ± 1.8 | Serum vitamin D were weakly positively associated with headache diary result but not related to migraine severity High serum levels of 25-OH-D3 were related to a higher headache diary result. | Associated |
| Kjaergaard (2012) [67] | Cross-sectional | 11,614 participants of the sixth survey of the Tromsø study in 2007–2008 | Range: 55–58 | No data | Serum vitamin D levels were inversely associated with non-migraine headache but there was no significant association between migraine and serum vitamin D. | Associated |
| Knutsen (2014) [24] | Cross-sectional | Headache (n = 63) | No data | No data | Mean serum vitamin D levels in patients with headaches were lower than in those suffering from musculoskeletal pain or fatigue. Headache was inversely associated with hypovitaminosis D. | Associated |
| Author (Year) | Study Design | Study Group: Type of Headache (Number of Participants) | Study Population Age (Years) | Supplementation Period | Vitamin D Dosage | Mean Serum 25(OH)D Levels (ng/mL) and/or 1,25(OH)2D (pg/mL) Before Treatment | Mean Serum 25(OH)D Levels (ng/mL) and/or 1,25(OH)2D (pg/mL) After the Treatment | Results |
|---|---|---|---|---|---|---|---|---|
| Kılıç (2019) [52] | Prospective | Migraine (n = 42) | Mean: 14 | 8 months | 2000 IU/day for 2 months, then 600–1000 IU/day of maintenance therapy for the next 6 months | 25(OH)D 9.4 (4.2–20) | 25(OH)D 34.6 (16.3–45) | Decreased migraine duration, frequency, VAS scores, and PedMIDAS scores compared with baseline values. No effect on headache severity. |
| Gazerani (2019) [68] | Randomized, double-blind, placebo-controlled, parallel | Migraine (n = 48) | Mean: 45.5 | 196 days | 100 μg/day | 25(OH)D 87.43 ± 32.00 1,25(OH)2D 43.55 ± 10.57 | 25(OH)D Increased significantly 1, 25(OH)2D No significant change Values not provided | Decreased migraine frequency, but no effect on severity, pressure pain thresholds, or temporal summation. |
| Buettner (2016) [69] | Randomized, placebo-controlled | Episodic migraine (n = 57) | Mean: 40 | 24 weeks | 1000 IU twice per day (+ simvastatin 20 mg/twice per day) | 25(OH)D 30 (18 to 41) median (IQR) | 25(OH)D 38 (34 to 45 median (IQR | Decreased number of migraine days. |
| Yilmaz (2016) [70] | Pre-post | Headache (n = 29) | Mean: 36.9 | 3 months | 50,000 IU/weekly + calcium of 1000 mg/day | 25(OH)D 10.6 ± 5.1 | 25(OH)D 46.5 ± 24.0 | Decreased headache severity and frequency. |
| Mottaghi (2015) [41] | Randomized, double-blind placebo-controlled | Migraine (n = 65) | Range: 10–65 | 10 weeks | 50,000 IU/week | 25(OH)D 16 ± 5.4 | 25(OH)D No data | Decreased headache frequency and mean headache diary results, but no effect on the severity and duration of headache. |
| Knutsen (2014) [24] | Randomized double-blinded placebo-controlled parallel-group | Headache (n = 157) | Range: 35–40 | 16 weeks | Group 1 25 μg/day Group 2 10 μg/day | 25(OH)D Group 1 27 Group 2 25 | 25(OH)D Group 1 increased by 25 Group 2 increased by 16 | No effect on the occurrence, anatomical localization, and degree of pain parameters or headache frequency. |
| Batcheller (2014) [71] | Prospective | Cluster headache (n = 110) | No data | 30 days | 10,000 IU/day | 25(OH)D 23.4 | 25(OH)D 76 | Decreased frequency, severity, and duration of headache in 80% of patients. |
| Cayir (2014) [72] | Prospective | Migraine (n = 53) | Range: 8–16 | 6 months | Group 1: amitriptyline alone Group 2: 400 IU/day + amitriptyline Group 3: 800 IU/day + amitriptyline Group 4: 5000 IU/day + amitriptyline | Group 1 32.4 ± 2 Group 2 28.1 ± 1.8 Group 3 17.2 ± 0.3 Group 4: 10.9 ± 0.6 | Group 1 33.7 ± 1.8 Group 2 34.1 ± 1.6 Group 3 25.6 ± 1.1 Group 4: 22.3 ± 1.9 | Decreased headache attack frequency in groups 2, 3, and 4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nowaczewska, M.; Wiciński, M.; Osiński, S.; Kaźmierczak, H. The Role of Vitamin D in Primary Headache–from Potential Mechanism to Treatment. Nutrients 2020, 12, 243. https://doi.org/10.3390/nu12010243
Nowaczewska M, Wiciński M, Osiński S, Kaźmierczak H. The Role of Vitamin D in Primary Headache–from Potential Mechanism to Treatment. Nutrients. 2020; 12(1):243. https://doi.org/10.3390/nu12010243
Chicago/Turabian StyleNowaczewska, Magdalena, Michał Wiciński, Stanisław Osiński, and Henryk Kaźmierczak. 2020. "The Role of Vitamin D in Primary Headache–from Potential Mechanism to Treatment" Nutrients 12, no. 1: 243. https://doi.org/10.3390/nu12010243
APA StyleNowaczewska, M., Wiciński, M., Osiński, S., & Kaźmierczak, H. (2020). The Role of Vitamin D in Primary Headache–from Potential Mechanism to Treatment. Nutrients, 12(1), 243. https://doi.org/10.3390/nu12010243






