A Review of Micronutrients and the Immune System–Working in Harmony to Reduce the Risk of Infection
Abstract
1. Introduction
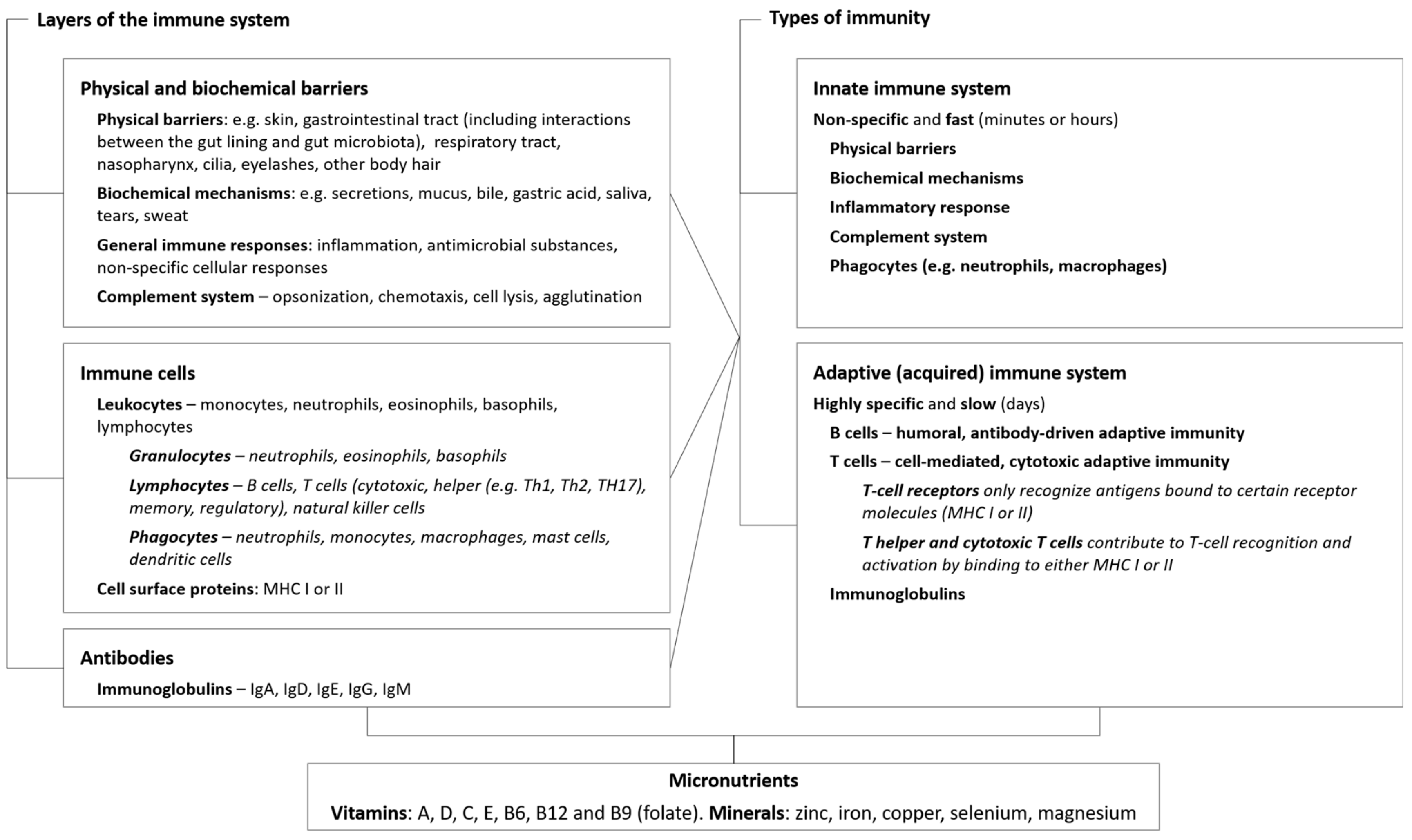
2. Micronutrients Are Integral to Immune Function
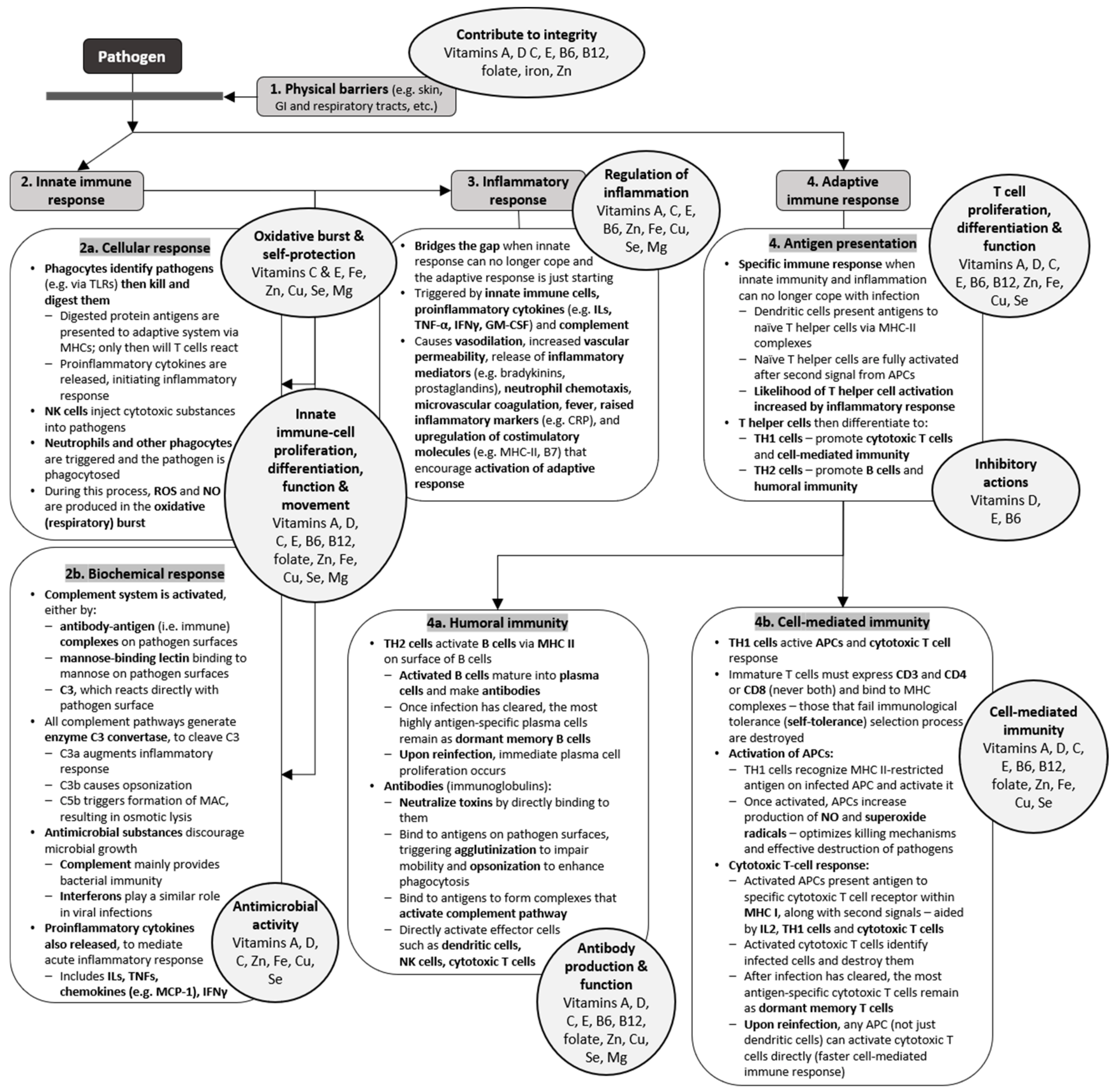
2.1. Physical and Biochemical Barriers
2.2. Innate Immunity
2.2.1. Antimicrobial Substances
2.2.2. Natural Killer Cells and Phagocytes
2.2.3. Inflammatory Response
2.3. Adaptive Immunity
2.3.1. Antigen Recognition
2.3.2. Cell-Mediated Immunity
2.3.3. Antibody-Mediated Immunity
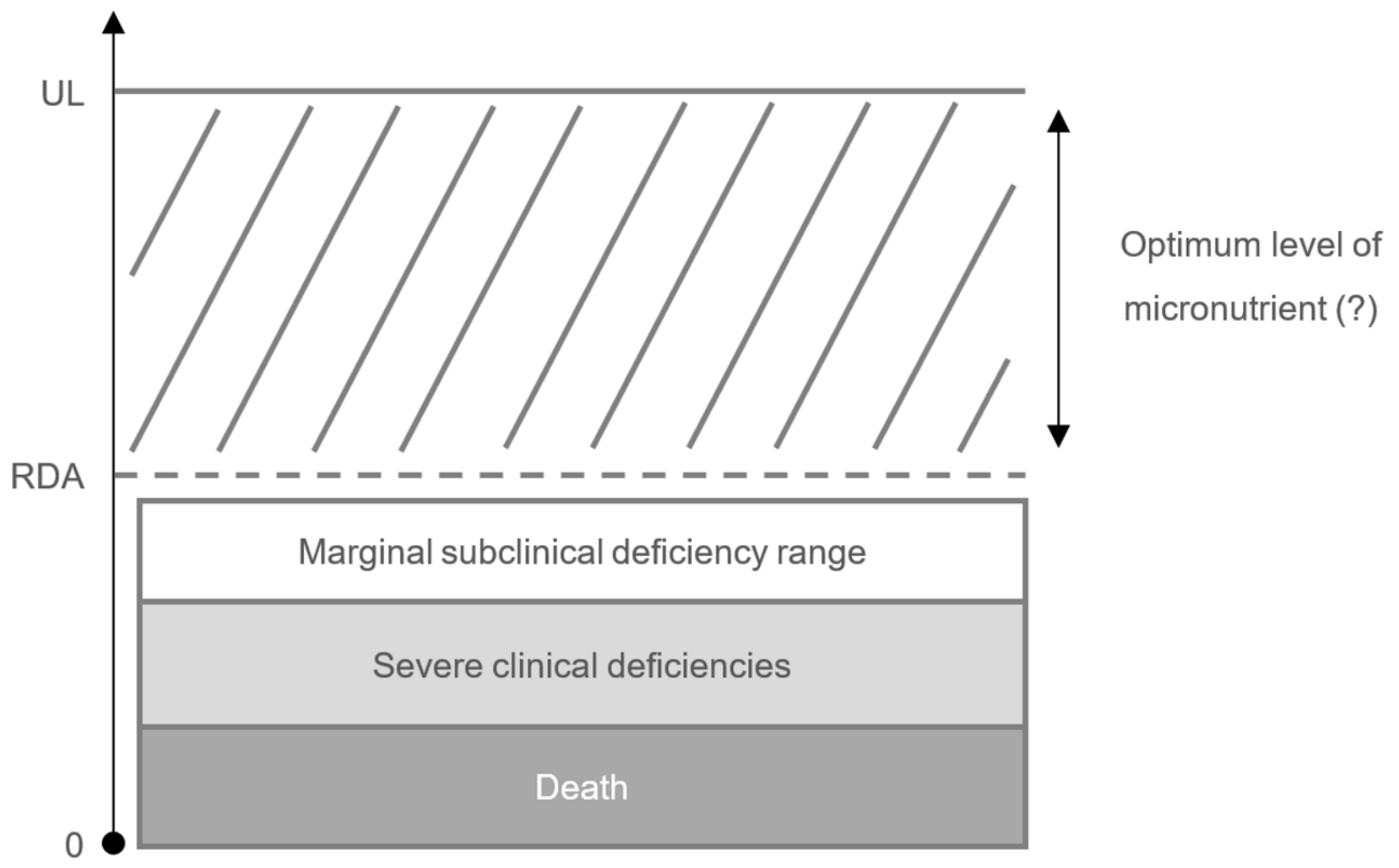
3. Impact of Micronutrient Status on the Immune Response and Risk of Infection
4. Effects of Supplementation on the Risk of Infection
4.1. Micronutrients in Reducing the Risk of Acute Infections
4.2. Micronutrients in the Management of Acute Infections
5. Future Directions
6. Summary
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- McFall-Ngai, M.; Hadfield, M.G.; Bosch, T.C.; Carey, H.V.; Domazet-Loso, T.; Douglas, A.E.; Dubilier, N.; Eberl, G.; Fukami, T.; Gilbert, S.F.; et al. Animals in a bacterial world, a new imperative for the life sciences. Proc. Natl. Acad. Sci. USA 2013, 110, 3229–3236. [Google Scholar] [CrossRef] [PubMed]
- Maggini, S.; Pierre, A.; Calder, P.C. Immune function and micronutrient requirements change over the life course. Nutrients 2018, 10, 1531. [Google Scholar] [CrossRef] [PubMed]
- Haryanto, B.; Suksmasari, T.; Wintergerst, E.; Maggini, S. Multivitamin supplementation supports immune function and ameliorates conditions triggered by reduced air quality. Vitam. Miner. 2015, 4, 1–15. [Google Scholar]
- Dolan, L.C.; Matulka, R.A.; Burdock, G.A. Naturally occurring food toxins. Toxins 2010, 2, 2289–2332. [Google Scholar] [CrossRef]
- Julier, Z.; Park, A.J.; Briquez, P.S.; Martino, M.M. Promoting tissue regeneration by modulating the immune system. Acta Biomater. 2017, 53, 13–28. [Google Scholar] [CrossRef]
- Bartholomew, M. James Lind’s Treatise of the Scurvy (1753). Postgrad. Med. J. 2002, 78, 695–696. [Google Scholar] [CrossRef]
- Micronutrient Information Center. Immunity in Depth. Linus Pauling Institute. 2016. Available online: http://lpi.oregonstate.edu/mic/health-disease/immunity (accessed on 10 May 2019).
- Maggini, S.; Beveridge, S.; Sorbara, J.P.; Senatore, G. Feeding the immune system: The role of micronutrients in restoring resistance to infections. CAB Rev. 2008, 3, 1–21. [Google Scholar] [CrossRef]
- Maggini, S.; Wintergerst, E.; Beveridge, S.; Hornig, D. Selected vitamins and trace elements support immune function by strengthening epithelial barriers and cellular and humoral immune responses. Br. J. Nutr. 2007, 98, S29–S35. [Google Scholar] [CrossRef]
- Biesalski, H.K. Nutrition meets the microbiome: Micronutrients and the microbiota. Ann. N. Y. Acad. Sci. 2016, 1372, 53–64. [Google Scholar] [CrossRef]
- Levy, M.; Thaiss, C.A.; Elinav, E. Metabolites: Messengers between the microbiota and the immune system. Genes Dev. 2016, 30, 1589–1597. [Google Scholar] [CrossRef]
- Sirisinha, S. The pleiotropic role of vitamin A in regulating mucosal immunity. Asian Pac. J. Allergy Immunol. 2015, 33, 71–89. [Google Scholar] [PubMed]
- Chew, B.P.; Park, J.S. Carotenoid action on the immune response. J. Nutr. 2004, 134, 257S–261S. [Google Scholar] [CrossRef] [PubMed]
- Clark, A.; Mach, N. Role of Vitamin D in the Hygiene Hypothesis: The Interplay between Vitamin D, Vitamin D Receptors, Gut Microbiota, and Immune Response. Front. Immunol. 2016, 7, 627. [Google Scholar] [CrossRef] [PubMed]
- Gombart, A.F. The vitamin D–antimicrobial peptide pathway and its role in protection against infection. Future Microbiol. 2009, 4, 1151. [Google Scholar] [CrossRef] [PubMed]
- Clairmont, A.; Tessman, D.; Stock, A.; Nicolai, S.; Stahl, W.; Sies, H. Induction of gap junctional intercellular communication by vitamin D in human skin fibroblasts is dependent on the nuclear Induction of gap junctional intercellular communication by vitamin D in human skin fibroblasts is dependent on the nuclear vitamin D receptor. Carcinogenesis 1996, 17, 1389–1391. [Google Scholar] [PubMed]
- Gniadecki, R.; Gajkowska, B.; Hansen, M. 1,25-dihydroxyvitamin D3 stimulates the assembly of adherens junctions in keratinocytes: Involvement of protein kinase C. Endocrinology 1997, 138, 2241–2248. [Google Scholar] [CrossRef]
- Palmer, H.G.; Gonzalez-Sancho, J.M.; Espada, J.; Berciano, M.T.; Puig, I.; Baulida, J.; Quintanilla, M.; Cano, A.; de Herreros, A.G.; Lafarga, M.; et al. Vitamin D3 promotes the differentiation of colon carcinoma cells by the induction of E-cadherin and the inhibition of beta-catenin signaling. J. Cell Biol. 2001, 154, 369–387. [Google Scholar] [CrossRef]
- Mihajlovic, M.; Fedecostante, M.; Oost, M.J.; Steenhuis, S.K.P.; Lentjes, E.; Maitimu-Smeele, I.; Janssen, M.J.; Hilbrands, L.B.; Masereeuw, R. Role of Vitamin D in Maintaining Renal Epithelial Barrier Function in Uremic Conditions. Int. J. Mol. Sci. 2017, 18, 2531. [Google Scholar] [CrossRef] [PubMed]
- Yin, Z.; Pintea, V.; Lin, Y.; Hammock, B.D.; Watsky, M.A. Vitamin D enhances corneal epithelial barrier function. Investig. Ophthalmol. Vis. Sci. 2011, 52, 7359–7364. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.; Maggini, S. Vitamin C and immune function. Nutrients 2017, 9, 1211. [Google Scholar] [CrossRef] [PubMed]
- Yoshii, K.; Hosomi, K.; Sawane, K.; Kunisawa, J. Metabolism of Dietary and Microbial Vitamin B Family in the Regulation of Host Immunity. Front. Nutr. 2019, 6, 48. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.H.; Sermersheim, M.; Li, H.; Lee, P.H.U.; Steinberg, S.M.; Ma, J. Zinc in Wound Healing Modulation. Nutrients 2017, 10, 16. [Google Scholar] [CrossRef] [PubMed]
- Wishart, K. Increased micronutrient requirements during physiologically demanding situations: Review of the current evidence. Vitamin. Miner. 2017, 6, 1–16. [Google Scholar] [CrossRef]
- Sly, L.M.; Lopez, M.; Nauseef, W.M.; Reiner, N.E. 1alpha,25-Dihydroxyvitamin D3-induced monocyte antimycobacterial activity is regulated by phosphatidylinositol 3-kinase and mediated by the NADPH-dependent phagocyte oxidase. J. Biol. Chem. 2001, 276, 35482–35493. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, H.; Hruska, K.A.; Seino, Y.; Malone, J.D.; Nishii, Y.; Teitelbaum, S.L. Disassociation of the macrophage-maturational effects of vitamin D from respiratory burst priming. J. Biol. Chem. 1991, 266, 10888–10892. [Google Scholar] [PubMed]
- Wu, D.; Lewis, E.D.; Pae, M.; Meydani, S.N. Nutritional modulation of immune function: Analysis of evidence, mechanisms, and clinical relevance. Front. Immunol. 2019, 9, 3160. [Google Scholar] [CrossRef]
- Bozonet, S.M.; Carr, A.C. The Role of Physiological Vitamin C Concentrations on Key Functions of Neutrophils Isolated from Healthy Individuals. Nutrients 2019, 11, 1363. [Google Scholar] [CrossRef]
- Wu, D.; Meydani, S.N. Age-associated changes in immune function: Impact of vitamin E intervention and the underlying mechanisms. Endocr. Metab. Immune. Disord. Drug Targets 2014, 14, 283–289. [Google Scholar] [CrossRef]
- World Health Organization; Food and Agricultural Organization of the United Nations. Part 2. Evaluating the public health significance of micronutrient malnutrition. In Guidelines on Food Fortification with Micronutrients; WHO: Geneva, Switzerland, 2006. [Google Scholar]
- Gao, H.; Dai, W.; Zhao, L.; Min, J.; Wang, F. The Role of Zinc and Zinc Homeostasis in Macrophage Function. J. Immunol. Res. 2018, 2018, 6872621. [Google Scholar] [CrossRef]
- Sheikh, A.; Shamsuzzaman, S.; Ahmad, S.M.; Nasrin, D.; Nahar, S.; Alam, M.M.; Al Tarique, A.; Begum, Y.A.; Qadri, S.S.; Chowdhury, M.I.; et al. Zinc influences innate immune responses in children with enterotoxigenic Escherichia coli-induced diarrhea. J. Nutr. 2010, 140, 1049–1056. [Google Scholar] [CrossRef]
- Agoro, R.; Taleb, M.; Quesniaux, V.F.J.; Mura, C. Cell iron status influences macrophage polarization. PLoS ONE 2018, 13, e0196921. [Google Scholar] [CrossRef] [PubMed]
- Besold, A.N.; Culbertson, E.M.; Culotta, V.C. The Yin and Yang of copper during infection. J. Biol. Inorg. Chem. 2016, 21, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Saeed, F.; Nadeem, M.; Ahmed, R.; Nadeem, M.; Arshad, M.; Ullah, A. Studying the impact of nutritional immunology underlying the modulation of immune responses by nutritional compounds—A review. Food Agric. Immunol. 2016, 27, 205–229. [Google Scholar] [CrossRef]
- Beck, M.A. Trace minerals, immune function, and viral evolution. In Military Strategies for Sustainment of Nutrition and Immune Function in the Field; National Academy Press: Washington, DC, USA, 1999; p. 339. [Google Scholar]
- Petrovic, J.; Stanic, D.; Dmitrasinovic, G.; Plecas-Solarovic, B.; Ignjatovic, S.; Batinic, B.; Popovic, D.; Pesic, V. Magnesium Supplementation Diminishes Peripheral Blood Lymphocyte DNA Oxidative Damage in Athletes and Sedentary Young Man. Oxidat. Med. Cell. Longev. 2016, 2016, 2019643. [Google Scholar] [CrossRef] [PubMed]
- Laires, M.J.; Monteiro, C. Exercise, magnesium and immune function. Magnes. Res. 2008, 21, 92–96. [Google Scholar] [PubMed]
- Wang, T.T.; Nestel, F.P.; Bourdeau, V.; Nagai, Y.; Wang, Q.; Liao, J.; Tavera-Mendoza, L.; Lin, R.; Hanrahan, J.W.; Mader, S.; et al. Cutting edge: 1,25-dihydroxyvitamin D3 is a direct inducer of antimicrobial peptide gene expression. J. Immunol. 2004, 173, 2909–2912. [Google Scholar] [CrossRef]
- Gombart, A.F.; Borregaard, N.; Koeffler, H.P. Human cathelicidin antimicrobial peptide (CAMP) gene is a direct target of the vitamin D receptor and is strongly up-regulated in myeloid cells by 1,25-dihydroxyvitamin D3. FASEB J. 2005, 19, 1067–1077. [Google Scholar] [CrossRef]
- Weber, G.; Heilborn, J.D.; Chamorro Jimenez, C.I.; Hammarsjö, A.; Törmä, H.; Ståhle, M. Vitamin D induces the antimicrobial protein hCAP18 in human skin. J. Investig. Dermatol. 2005, 124, 1080–1082. [Google Scholar] [CrossRef]
- Matsui, T.; Takahashi, R.; Nakao, Y.; Koizumi, T.; Katakami, Y.; Mihara, K.; Sugiyama, T.; Fujita, T. 1,25-Dihydroxyvitamin D3-regulated expression of genes involved in human T-lymphocyte proliferation and differentiation. Cancer Res. 1986, 46, 5827–5831. [Google Scholar]
- Reichel, H.; Koeffler, H.P.; Tobler, A.; Norman, A.W. 1 alpha,25-Dihydroxyvitamin D3 inhibits gamma-interferon synthesis by normal human peripheral blood lymphocytes. Proc. Natl. Acad. Sci. USA 1987, 84, 3385–3389. [Google Scholar] [CrossRef]
- Rigby, W.F.; Denome, S.; Fanger, M.W. Regulation of lymphokine production and human T lymphocyte activation by 1,25-dihydroxyvitamin D3. Specific inhibition at the level of messenger RNA. J. Clin. Investig. 1987, 79, 1659–1664. [Google Scholar] [CrossRef] [PubMed]
- Inoue, M.; Matsui, T.; Nishibu, A.; Nihei, Y.; Iwatsuki, K.; Kaneko, F. Regulatory effects of 1alpha,25-dihydroxyvitamin D3 on inflammatory responses in psoriasis. Eur. J. Dermatol. 1998, 8, 16–20. [Google Scholar] [PubMed]
- Lin, Z.; Li, W. The Roles of Vitamin D and Its Analogs in Inflammatory Diseases. Curr. Top. Med. Chem. 2016, 16, 1242–1261. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Leung, D.Y.M.; Richers, B.N.; Liu, Y.; Remigio, L.K.; Riches, D.W.; Goleva, E. Vitamin D inhibits monocyte/macrophage proinflammatory cytokine production by targeting MAPK phosphatase-1. J. Immunol. 2012, 188, 2127–2135. [Google Scholar] [CrossRef]
- Topilski, I.; Flaishon, L.; Naveh, Y.; Harmelin, A.; Levo, Y.; Shachar, I. The anti-inflammatory effects of 1,25-dihydroxyvitamin D3 on Th2 cells in vivo are due in part to the control of integrin-mediated T lymphocyte homing. Eur. J. Immunol. 2004, 34, 1068–1076. [Google Scholar] [CrossRef] [PubMed]
- Wintergerst, E.; Maggini, S.; Hornig, D. Immune-enhancing role of vitamin C and zinc and effect on clinical conditions. Ann. Nutr. Metab. 2006, 50, 85–94. [Google Scholar] [CrossRef]
- Lee, G.Y.; Han, S.N. The Role of Vitamin E in Immunity. Nutrients 2018, 10, 1614. [Google Scholar] [CrossRef]
- Sakakeeny, L.; Roubenoff, R.; Obin, M.; Fontes, J.D.; Benjamin, E.J.; Bujanover, Y.; Jacques, P.F.; Selhub, J. Plasma Pyridoxal-5-Phosphate Is Inversely Associated with Systemic Markers of Inflammation in a Population of U.S. Adults. J. Nutr. 2012, 142, 1280–1285. [Google Scholar] [CrossRef]
- Ueland, P.M.; McCann, A.; Midttun, O.; Ulvik, A. Inflammation, vitamin B6 and related pathways. Mol. Asp. Med. 2017, 53, 10–27. [Google Scholar] [CrossRef]
- Jarosz, M.; Olbert, M.; Wyszogrodzka, G.; Młyniec, K.; Librowski, T. Antioxidant and anti-inflammatory effects of zinc. Zinc-dependent NF-κB signaling. Inflammopharmacology 2017, 25, 11–24. [Google Scholar] [CrossRef] [PubMed]
- Kitabayashi, C.; Fukada, T.; Kanamoto, M.; Ohashi, W.; Hojyo, S.; Atsumi, T.; Ueda, N.; Azuma, I.; Hirota, H.; Murakami, M.; et al. Zinc suppresses Th17 development via inhibition of STAT3 activation. Int Immunol. 2010, 22, 375–386. [Google Scholar] [CrossRef] [PubMed]
- Maywald, M.; Wang, F.; Rink, L. Zinc supplementation plays a crucial role in T helper 9 differentiation in allogeneic immune reactions and non-activated T cells. J. Trace Elem. Med. Biol. 2018, 50, 482–488. [Google Scholar] [CrossRef] [PubMed]
- Foster, M.; Samman, S. Zinc and regulation of inflammatory cytokines: Implications for cardiometabolic disease. Nutrients 2012, 4, 676–694. [Google Scholar] [CrossRef] [PubMed]
- Wessels, I.; Rink, L. Micronutrients in autoimmune diseases: Possible therapeutic benefits of zinc and vitamin D. J. Nutr. Biochem. 2019, 77, 108240. [Google Scholar] [CrossRef] [PubMed]
- Alpert, P. The role of vitamins and minerals on the immune system. Home Health Care Manag. Pract. 2017, 29, 199–202. [Google Scholar] [CrossRef]
- Bussiere, F.I.; Mazur, A.; Fauquert, J.L.; Labbe, A.; Rayssiguier, Y.; Tridon, A. High magnesium concentration in vitro decreases human leukocyte activation. Magnes. Res. 2002, 15, 43–48. [Google Scholar]
- Ross, A.C. Vitamin A and retinoic acid in T cell-related immunity. Am. J. Clin. Nutr. 2012, 96, 1166s–1172s. [Google Scholar] [CrossRef]
- Sigmundsdottir, H.; Pan, J.; Debes, G.F.; Alt, C.; Habtezion, A.; Soler, D.; Butcher, E.C. DCs metabolize sunlight-induced vitamin D3 to ‘program’ T cell attraction to the epidermal chemokine CCL27. Nat. Immunol. 2007, 8, 285–293. [Google Scholar] [CrossRef]
- Sassi, F.; Tamone, C.; D’Amelio, P. Vitamin D: Nutrient, Hormone, and Immunomodulator. Nutrients 2018, 10, 1656. [Google Scholar] [CrossRef]
- Cantorna, M.T.; Snyder, L.; Lin, Y.D.; Yang, L. Vitamin D and 1,25(OH)2D regulation of T cells. Nutrients 2015, 7, 3011–3021. [Google Scholar] [CrossRef]
- Penna, G.; Adorini, L. 1 Alpha,25-dihydroxyvitamin D3 inhibits differentiation, maturation, activation, and survival of dendritic cells leading to impaired alloreactive T cell activation. J. Immunol. 2000, 164, 2405–2411. [Google Scholar] [CrossRef]
- Piemonti, L.; Monti, P.; Sironi, M.; Fraticelli, P.; Leone, B.E.; Dal Cin, E.; Allavena, P.; Di Carlo, V. Vitamin D3 affects differentiation, maturation, and function of human monocyte-derived dendritic cells. J. Immunol. 2000, 164, 4443–4451. [Google Scholar] [CrossRef] [PubMed]
- Bscheider, M.; Butcher, E.C. Vitamin D immunoregulation through dendritic cells. Immunology 2016, 148, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Wintergerst, E.; Maggini, S.; Hornig, D. Contribution of selected vitamins and trace elements to immune function. Nutr. Metab. 2007, 51, 301–323. [Google Scholar] [CrossRef] [PubMed]
- Hurwitz, B.E.; Klaus, J.R.; Llabre, M.M.; Gonzalez, A.; Lawrence, P.J.; Maher, K.J.; Greeson, J.M.; Baum, M.K.; Shor-Posner, G.; Skyler, J.S.; et al. Suppression of human immunodeficiency virus type 1 viral load with selenium supplementation: A randomized controlled trial. Arch. Intern. Med. 2007, 167, 148–154. [Google Scholar] [CrossRef]
- Shankar, A.H.; Prasad, A.S. Zinc and immune function: The biological basis of altered resistance to infection. Am. J. Clin. Nutr. 1998, 68, 447s–463s. [Google Scholar] [CrossRef]
- Ibs, K.-H.; Rink, L. Zinc-Altered Immune function. J. Nutr. 2003, 133, 1452S–1456S. [Google Scholar] [CrossRef] [PubMed]
- Han, S.N.; Adolfsson, O.; Lee, C.K.; Prolla, T.A.; Ordovas, J.; Meydani, S.N. Vitamin E and gene expression in immune cells. Ann. N. Y. Acad. Sci. 2004, 1031, 96–101. [Google Scholar] [CrossRef]
- Ooi, J.H.; Li, Y.; Rogers, C.J.; Cantorna, M.T. Vitamin D regulates the gut microbiome and protects mice from dextran sodium sulfate-induced colitis. J. Nutr. 2013, 143, 1679–1686. [Google Scholar] [CrossRef] [PubMed]
- Cantorna, M.T.; McDaniel, K.; Bora, S.; Chen, J.; James, J. Vitamin D, immune regulation, the microbiota, and inflammatory bowel disease. Exp. Biol. Med. 2014, 239, 1524–1530. [Google Scholar] [CrossRef]
- Cantorna, M.T.; Snyder, L.; Arora, J. Vitamin A and vitamin D regulate the microbial complexity, barrier function, and the mucosal immune responses to ensure intestinal homeostasis. Crit. Rev. Biochem. Mol. Biol. 2019, 54, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Ko, G. Antiviral effect of vitamin A on norovirus infection via modulation of the gut microbiome. Sci. Rep. 2016, 6, 25835. [Google Scholar] [CrossRef] [PubMed]
- Pullar, J.M.; Carr, A.C.; Vissers, M.C.M. The Roles of Vitamin C in Skin Health. Nutrients 2017, 9, 866. [Google Scholar] [CrossRef]
- Forman, H.J.; Torres, M.; Fukuto, J. Redox signaling. Mol. Cell Biochem. 2002, 234–235, 49–62. [Google Scholar] [CrossRef]
- Rahman, K. Studies on free radicals, antioxidants, and co-factors. Clin. Interv. Aging 2007, 2, 219–236. [Google Scholar]
- Delfino, R.J.; Staimer, N.; Vaziri, N.D. Air pollution and circulating biomarkers of oxidative stress. Air Qual. Atmos. Health 2011, 4, 37–52. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Antioxidants in human health and disease. Annu. Rev. Nutr. 1996, 16, 33–50. [Google Scholar] [CrossRef] [PubMed]
- Takiishi, T.; Fenero, C.I.M.; Câmara, N.O.S. Intestinal barrier and gut microbiota: Shaping our immune responses throughout life. Tissue Barriers 2017, 5, e1373208. [Google Scholar] [CrossRef]
- Broome, C.S.; McArdle, F.; Kyle, J.A.; Andrews, F.; Lowe, N.M.; Hart, C.A.; Arthur, J.R.; Jackson, M.J. An increase in selenium intake improves immune function and poliovirus handling in adults with marginal selenium status. Am. J. Clin. Nutr. 2004, 80, 154–162. [Google Scholar] [CrossRef]
- Nan, R.; Tetchner, S.; Rodriguez, E.; Pao, P.J.; Gor, J.; Lengyel, I.; Perkins, S.J. Zinc-induced self-association of complement C3b and Factor H: Implications for inflammation and age-related macular degeneration. J. Biol. Chem. 2013, 288, 19197–19210. [Google Scholar] [CrossRef]
- Yang, D.; de la Rosa, G.; Tewary, P.; Oppenheim, J.J. Alarmins link neutrophils and dendritic cells. Trends Immunol. 2009, 30, 531–537. [Google Scholar] [CrossRef] [PubMed]
- Davies, L.C.; Jenkins, S.J.; Allen, J.E.; Taylor, P.R. Tissue-resident macrophages. Nat. Immunol. 2013, 14, 986–995. [Google Scholar] [CrossRef]
- Wang, J.; Pantopoulos, K. Regulation of cellular iron metabolism. Biochem. J. 2011, 434, 365–381. [Google Scholar] [CrossRef] [PubMed]
- Hewison, M. Vitamin D and the intracrinology of innate immunity. Mol. Cell. Endocrinol. 2010, 321, 103–111. [Google Scholar] [CrossRef]
- Wu, D.; Meydani, S.N. Age-associated changes in immune and inflammatory responses: Impact of vitamin E intervention. J. Leukoc. Biol. 2008, 84, 900–914. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, G.T.; Green, E.R.; Mecsas, J. Neutrophils to the ROScue: Mechanisms of NADPH Oxidase Activation and Bacterial Resistance. Front. Cell. Infect. Microbiol. 2017, 7, 373. [Google Scholar] [CrossRef]
- Dupont, C.L.; Grass, G.; Rensing, C. Copper toxicity and the origin of bacterial resistance—New insights and applications. Metallomics 2011, 3, 1109–1118. [Google Scholar] [CrossRef] [PubMed]
- Abu-Amer, Y.; Bar-Shavit, Z. Regulation of TNF-alpha release from bone marrow-derived macrophages by vitamin D. J. Cell. Biochem. 1994, 55, 435–444. [Google Scholar] [CrossRef]
- Cañedo-Dorantes, L.; Cañedo-Ayala, M. Skin Acute Wound Healing: A Comprehensive Review. Int. J. Inflamm. 2019, 2019, 3706315. [Google Scholar] [CrossRef]
- Romano, M.; Fanelli, G.; Albany, C.J.; Giganti, G.; Lombardi, G. Past, Present, and Future of Regulatory T Cell Therapy in Transplantation and Autoimmunity. Front. Immunol. 2019, 10. [Google Scholar] [CrossRef]
- Lemire, J.M. Immunomodulatory actions of 1,25-dihydroxyvitamin D3. J. Steroid Biochem. Mol. Biol. 1995, 53, 599–602. [Google Scholar] [CrossRef]
- Jeffery, L.E.; Burke, F.; Mura, M.; Zheng, Y.; Qureshi, O.S.; Hewison, M.; Walker, L.S.; Lammas, D.A.; Raza, K.; Sansom, D.M. 1,25-Dihydroxyvitamin D3 and IL-2 combine to inhibit T cell production of inflammatory cytokines and promote development of regulatory T cells expressing CTLA-4 and FoxP3. J. Immunol. 2009, 183, 5458–5467. [Google Scholar] [CrossRef]
- McCully, M.L.; Ladell, K.; Hakobyan, S.; Mansel, R.E.; Price, D.A.; Moser, B. Epidermis instructs skin homing receptor expression in human T cells. Blood 2012, 120, 4591–4598. [Google Scholar] [CrossRef] [PubMed]
- Rosenkranz, E.; Maywald, M.; Hilgers, R.-D.; Brieger, A.; Clarner, T.; Kipp, M.; Plümäkers, B.; Meyer, S.; Schwerdtle, T.; Rink, L. Induction of regulatory T cells in Th1-/Th17-driven experimental autoimmune encephalomyelitis by zinc administration. J. Nutr. Biochem. 2016, 29, 116–123. [Google Scholar] [CrossRef]
- Rosenkranz, E.; Metz, C.H.D.; Maywald, M.; Hilgers, R.-D.; Weßels, I.; Senff, T.; Haase, H.; Jäger, M.; Ott, M.; Aspinall, R.; et al. Zinc supplementation induces regulatory T cells by inhibition of Sirt-1 deacetylase in mixed lymphocyte cultures. Mol. Nutr. Food Res. 2016, 60, 661–671. [Google Scholar] [CrossRef] [PubMed]
- Rigby, W.F.; Stacy, T.; Fanger, M.W. Inhibition of T lymphocyte mitogenesis by 1,25-dihydroxyvitamin D3 (calcitriol). J. Clin. Investig. 1984, 74, 1451–1455. [Google Scholar] [CrossRef] [PubMed]
- Tsoukas, C.D.; Provvedini, D.M.; Manolagas, S.C. 1,25-dihydroxyvitamin D3: A novel immunoregulatory hormone. Science 1984, 224, 1438–1440. [Google Scholar] [CrossRef] [PubMed]
- Saggese, G.; Federico, G.; Balestri, M.; Toniolo, A. Calcitriol inhibits the PHA-induced production of IL-2 and IFN-gamma and the proliferation of human peripheral blood leukocytes while enhancing the surface expression of HLA class II molecules. J. Endocrinol. Investig. 1989, 12, 329–335. [Google Scholar] [CrossRef] [PubMed]
- Li, T.X.; Li, Y. Synergistic effect of zinc and vitamin A on T cell functions. Biomed. Environ. Sci. 2007, 20, 131–134. [Google Scholar] [PubMed]
- Tamura, J.; Kubota, K.; Murakami, H.; Sawamura, M.; Matsushima, T.; Tamura, T.; Saitoh, T.; Kurabayshi, H.; Naruse, T. Immunomodulation by vitamin B12: Augmentation of CD8+ T lymphocytes and natural killer (NK) cell activity in vitamin B12-deficient patients by methyl-B12 treatment. Clin. Exp. Immunol. 1999, 116, 28–32. [Google Scholar] [CrossRef]
- Manicassamy, S.; Pulendran, B. Retinoic acid-dependent regulation of immune responses by dendritic cells and macrophages. Semin. Immunol. 2009, 21, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Moore, J.B.; Blanchard, R.K.; McCormack, W.T.; Cousins, R.J. cDNA array analysis identifies thymic LCK as upregulated in moderate murine zinc deficiency before T-lymphocyte population changes. J. Nutr. 2001, 131, 3189–3196. [Google Scholar] [CrossRef] [PubMed]
- Calder, P. Conference on ‘Transforming the nutrition landscape in Africa’. Plenary Session 1: Feeding the immune system. Proc. Nutr. Soc. 2013, 72, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Bhaskaram, P. Micronutrient malnutrition, infection, and immunity: An overview. Nutr. Rev. 2002, 60, S40–S45. [Google Scholar] [CrossRef]
- Milner, J.; Beck, M. Micronutrients, immunology and inflammation. The impact of obesity on the immune response to infection. Proc. Nutr. Soc. 2012, 71, 298–306. [Google Scholar] [CrossRef]
- Calder, P.; Prescott, S.; Caplan, M. Scientific Review: The Role of Nutrients in Immune Function of Infants and Young Children Emerging Evidence for Long-Chain Polyunsaturated Fatty Acids; Mead Johnson & Company: Glenview, IL, USA, 2007. [Google Scholar]
- Prentice, S. They are what you eat: Can nutritional factors during gestation and early infancy modulate the neonatal immune response? Front. Immunol. 2017, 8, 1641. [Google Scholar] [CrossRef]
- Hemilä, H. Vitamin C and infections. Nutrients 2017, 9, 339. [Google Scholar] [CrossRef]
- GBD 2017 Causes of Death Collaborators. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1736–1788. [Google Scholar] [CrossRef]
- Semba, R.D. The historical evolution of thought regarding multiple micronutrient nutrition. J. Nutr. 2012, 142, 143s–156s. [Google Scholar] [CrossRef]
- Elmadfa, I.; Meyer, A.; Nowak, V.; Hasenegger, V.; Putz, P.; Verstraeten, R.; Remaut-DeWinter, A.M. European Nutrition and Health Report. Forum. Nutr. 2009, 62, 1–405. [Google Scholar]
- Anderson, R.; Oosthuizen, R.; Maritz, R.; Theron, A.; Van Rensburg, A. The effects of increasing weekly doses of ascorbate on certain cellular and humoral immune functions in normal volunteers. Am. J. Clin. Nutr. 1980, 33, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Hemila, H.; Chalker, E. Vitamin C can shorten the length of stay in the ICU: A meta-analysis. Nutrients 2019, 11, 708. [Google Scholar] [CrossRef]
- Meghil, M.M.; Hutchens, L.; Raed, A.; Multani, N.A.; Rajendran, M.; Zhu, H.; Looney, S.; Elashiry, M.; Arce, R.M.; Peacock, M.E.; et al. The influence of vitamin D supplementation on local and systemic inflammatory markers in periodontitis patients: A pilot study. Oral Dis. 2019, 25, 1403–1413. [Google Scholar] [CrossRef]
- Cannell, J.; Vieth, R.; Umhau, J.; Holick, M.; Grant, W.; Madronich, S.; Garland, C.; Giovannucci, E. Epidemic influenza and vitamin D. Epidemiol. Infect. 2006, 134, 1129–1140. [Google Scholar] [CrossRef]
- Jat, K.R. Vitamin D deficiency and lower respiratory tract infections in children: A systematic review and meta-analysis of observational studies. Trop. Dr. 2017, 47, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Autier, P.; Mullie, P.; Macacu, A.; Dragomir, M.; Boniol, M.; Coppens, K.; Pizot, C.; Boniol, M. Effect of vitamin D supplementation on non-skeletal disorders: A systematic review of meta-analyses and randomised trials. Lancet Diabetes Endocrinol. 2017, 5, 986–1004. [Google Scholar] [CrossRef]
- Villamor, E.; Fawzi, W.W. Effects of vitamin a supplementation on immune responses and correlation with clinical outcomes. Clin. Microbiol. Rev. 2005, 18, 446–464. [Google Scholar] [CrossRef]
- Ross, A.C.; Caballero, B.; Cousins, R.J.; Tucker, K.L.; Ziegler, T.R. Modern Nutrition in Health and Disease, 11th ed.; Wolters Kluwer Health Adis (ESP): Philadelphia, PA, USA, 2012. [Google Scholar]
- Traber, M.G. Vitamin E inadequacy in humans: Causes and consequences. Adv. Nutr. 2014, 5, 503–514. [Google Scholar] [CrossRef] [PubMed]
- De la Fuente, M.; Hernanz, A.; Guayerbas, N.; Victor, V.; Arnalich, F. Vitamin E ingestion improves several immune functions in elderly men and women. Free Radic. Res. 2008, 42, 272–280. [Google Scholar] [CrossRef] [PubMed]
- Qian, B.; Shen, S.; Zhang, J.; Jing, P. Effects of Vitamin B6 Deficiency on the Composition and Functional Potential of T Cell Populations. J. Immunol. Res. 2017, 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.H.; Chang, S.J.; Lee, B.J.; Lin, K.L.; Huang, Y.C. Vitamin B6 supplementation increases immune responses in critically ill patients. Eur. J. Clin. Nutr. 2006, 60, 1207–1213. [Google Scholar] [CrossRef]
- Rowley, C.A.; Kendall, M.M. To B12 or not to B12: Five questions on the role of cobalamin in host-microbial interactions. PLoS Pathog. 2019, 15, e1007479. [Google Scholar] [CrossRef]
- Troen, A.M.; Mitchell, B.; Sorensen, B.; Wener, M.H.; Johnston, A.; Wood, B.; Selhub, J.; McTiernan, A.; Yasui, Y.; Oral, E.; et al. Unmetabolized folic acid in plasma is associated with reduced natural killer cell cytotoxicity among postmenopausal women. J. Nutr. 2006, 136, 189–194. [Google Scholar] [CrossRef]
- Selhub, J. Folate, vitamin B12 and vitamin B6 and one carbon metabolism. J. Nutr. Health Aging 2002, 6, 39–42. [Google Scholar]
- Bunout, D.; Barrera, G.; Hirsch, S.; Gattas, V.; de la Maza, M.P.; Haschke, F.; Steenhout, P.; Klassen, P.; Hager, C.; Avendano, M.; et al. Effects of a nutritional supplement on the immune response and cytokine production in free-living Chilean elderly. J. Parenter. Enteral Nutr. 2004, 28, 348–354. [Google Scholar] [CrossRef]
- Maywald, M.; Wessels, I.; Rink, L. Zinc Signals and Immunity. Int. J. Mol. Sci. 2017, 18, 2222. [Google Scholar] [CrossRef]
- Sandström, B.; Cederblad, A.; Lindblad, B.S.; Lönnerdal, B. Acrodermatitis enteropathica, zinc metabolism, copper status, and immune function. Arch. Pediatr. Adolesc. Med. 1994, 148, 980–985. [Google Scholar] [CrossRef]
- Bonaventura, P.; Benedetti, G.; Albarede, F.; Miossec, P. Zinc and its role in immunity and inflammation. Autoimmun. Rev. 2015, 14, 277–285. [Google Scholar] [CrossRef]
- Savino, W.; Dardenne, M. Nutritional imbalances and infections affect the thymus: Consequences on T-cell-mediated immune responses. Proc. Nutr. Soc. 2010, 69, 636–643. [Google Scholar] [CrossRef]
- Maxfield, L.; Crane, J.S. Zinc Deficiency. In StatPearls; StatPearls Publishing LLC: Treasure Island, FL, USA, 2019. [Google Scholar]
- Kumar, V.; Choudhry, V. Iron deficiency and infection. Indian J. Pediatr. 2010, 77, 789–793. [Google Scholar] [CrossRef]
- Chen, K.; Chen, X.-R.; Zhang, L.; Luo, H.-Y.; Gao, N.; Wang, J.; Fu, G.-Y.; Mao, M. Effect of simultaneous supplementation of vitamin A and iron on diarrheal and respiratory tract infection in preschool children in Chengdu City, China. Nutrition 2013, 29, 1197–1203. [Google Scholar] [CrossRef] [PubMed]
- Percival, S.S. Copper and immunity. Am. J. Clin. Nutr. 1998, 67, 1064s–1068s. [Google Scholar] [CrossRef] [PubMed]
- Bonham, M.; O’Connor, J.M.; Hannigan, B.M.; Strain, J.J. The immune system as a physiological indicator of marginal copper status? Br. J. Nutr. 2002, 87, 393–403. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, F.H.; Lukaski, H.C. Update on the relationship between magnesium and exercise. Magnes. Res. 2006, 19, 180–189. [Google Scholar] [PubMed]
- Johnson, S. The multifaceted and widespread pathology of magnesium deficiency. Med. Hypotheses 2001, 56, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Mooren, F.C.; Golf, S.W.; Volker, K. Effect of magnesium on granulocyte function and on the exercise induced inflammatory response. Magnes. Res. 2003, 16, 49–58. [Google Scholar]
- IOM (Institute of Medicine). Dietary Reference Intakes for Calcium and Vitamin D; The National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
- Padayatty, S.J.; Levine, M. Vitamin C: The known and the unknown and Goldilocks. Oral Dis. 2016, 22, 463–493. [Google Scholar] [CrossRef]
- Elste, V.; Troesch, B.; Eggersdorfer, M.; Weber, P. Emerging Evidence on Neutrophil Motility Supporting Its Usefulness to Define Vitamin C Intake Requirements. Nutrients 2017, 9, 503. [Google Scholar] [CrossRef]
- Hume, R.; Weyers, E. Changes in leucocyte ascorbic acid during the common cold. Scott. Med. J. 1973, 18, 3–7. [Google Scholar] [CrossRef]
- Pallast, E.G.; Schouten, E.G.; de Waart, F.G.; Fonk, H.C.; Doekes, G.; von Blomberg, B.M.; Kok, F.J. Effect of 50- and 100-mg vitamin E supplements on cellular immune function in noninstitutionalized elderly persons. Am. J. Clin. Nutr. 1999, 69, 1273–1281. [Google Scholar] [CrossRef]
- Meydani, S.N.; Meydani, M.; Blumberg, J.B.; Leka, L.S.; Siber, G.; Loszewski, R.; Thompson, C.; Pedrosa, M.C.; Diamond, R.D.; Stollar, B.D. Vitamin E supplementation and in vivo immune response in healthy elderly subjects. A randomized controlled trial. JAMA 1997, 277, 1380–1386. [Google Scholar] [CrossRef]
- Meydani, S.; Leka, L.; Fine, B.; Dallal, G.; Keusch, G.; Singh, M.; Hamer, D. Vitamin E and respiratory tract infections in elderly nursing home residents: A randomized controlled trial. JAMA 2004, 292, 828–836. [Google Scholar] [CrossRef]
- Kwak, H.-K.; Hansen, C.M.; Leklem, J.E.; Hardin, K.; Shultz, T.D. Improved vitamin B-6 status is positively related to lymphocyte proliferation in young women consuming a controlled diet. J. Nutr. 2002, 132, 3308–3313. [Google Scholar] [CrossRef]
- Steinbrenner, H.; Al-Quraishy, S.; Dkhil, M.A.; Wunderlich, F.; Sies, H. Dietary selenium in adjuvant therapy of viral and bacterial infections. Adv. Nutr. 2015, 6, 73–82. [Google Scholar] [CrossRef]
- Hewison, M. An update on vitamin D and human immunity. Clin. Endocrinol. 2012, 76, 315–325. [Google Scholar] [CrossRef] [PubMed]
- Wimalawansa, S.J. Vitamin D in the new millennium. Curr. Osteoporos. Rep. 2012, 10, 4–15. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Nations, Nations, Food and Agricultural Organization of the United Nations. In Guidelines on Food Fortification with Micronutrients; Allen, L., de Benoist, B., Dary, O., Hurrell, R., Eds.; World Health Organization: Geneva, Switzerland, 2006. [Google Scholar]
- World Health Organization. Nutritional Rickets: A Review of Disease Burden, Causes, Diagnosis, Prevention and Treatment; World Health Organization: Geneva, Switzerland, 2019. [Google Scholar]
- WHO. Guideline: Potassium Intake for Adults and Children; World Health Organization (WHO): Geneva, Switzerland, 2012. [Google Scholar]
- Troesch, B.; Hoeft, B.; McBurney, M.; Eggersdorfer, M.; Weber, P. Dietary surveys indicate vitamin intakes below recommendations are common in representative Western countries. Br. J. Nutr. 2012, 108, 692–698. [Google Scholar] [CrossRef] [PubMed]
- United States Department of Agriculture. What We Eat in America, NHANES 2013–2016. Usual Nutrient Intake from Food and Beverages, by Gender and Age. 2019. Available online: https://www.ars.usda.gov/ARSUserFiles/80400530/pdf/usual/Usual_Intake_gender_WWEIA_2013_2016.pdf (accessed on 28 May 2019).
- WHO. Guideline: Use of Multiple Micronutrient Powders for Point-of-Use Fortification of Foods Consumed by Infants and Young Children Aged 6–23 Months and Children Aged 2–12 Years; World Health Organization: Geneva, Switzerland, 2016. Available online: https://www.ncbi.nlm.nih.gov/books/NBK409166/ (accessed on 15 January 2020).
- Bailey, R.; West, K.J.; Black, R. The epidemiology of global micronutrient deficiencies. Ann. Nutr. Metab. 2015, 66, 22–33. [Google Scholar] [CrossRef]
- Platel, K.; Srinivasan, K. Bioavailability of Micronutrients from Plant Foods: An Update. Crit. Rev. Food Sci. Nutr. 2016, 56, 1608–1619. [Google Scholar] [CrossRef]
- Bohn, T.; Davidsson, L.; Walczyk, T.; Hurrell, R.F. Fractional magnesium absorption is significantly lower in human subjects from a meal served with an oxalate-rich vegetable, spinach, as compared with a meal served with kale, a vegetable with a low oxalate content. Br. J. Nutr. 2007, 91, 601–606. [Google Scholar] [CrossRef]
- Gibson, R.S.; Raboy, V.; King, J.C. Implications of phytate in plant-based foods for iron and zinc bioavailability, setting dietary requirements, and formulating programs and policies. Nutr. Rev. 2018, 76, 793–804. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, R.; Spencer, H.; Welsh, J.J. Magnesium absorption in human subjects from leafy vegetables, intrinsically labeled with stable 26 Mg. Am. J. Clin. Nutr. 1984, 39, 571–576. [Google Scholar] [CrossRef] [PubMed]
- Suliburska, J.; Krejpcio, Z. Evaluation of the content and bioaccessibility of iron, zinc, calcium and magnesium from groats, rice, leguminous grains and nuts. J. Food Sci. Technol. 2014, 51, 589–594. [Google Scholar] [CrossRef] [PubMed]
- Hurrell, R.; Egli, I. Iron bioavailability and dietary reference values. Am. J. Clin. Nutr. 2010, 91, 1461S–1467S. [Google Scholar] [CrossRef] [PubMed]
- Kaur, B.; Henry, J. Micronutrient status in type 2 diabetes: A review. Adv. Food Nutr. Res. 2014, 71, 55–100. [Google Scholar] [PubMed]
- Thomas-Valdes, S.; Tostes, M.; Anunciacao, P.C.; da Silva, B.P.; Sant’Ana, H.M.P. Association between vitamin deficiency and metabolic disorders related to obesity. Crit. Rev. Food Sci. Nutr. 2017, 57, 3332–3343. [Google Scholar] [CrossRef] [PubMed]
- Desmarchelier, C.; Borel, P.; Goncalves, A.; Kopec, R.; Nowicki, M.; Morange, S.; Lesavre, N.; Portugal, H.; Reboul, E. A Combination of Single-Nucleotide Polymorphisms Is Associated with Interindividual Variability in Cholecalciferol Bioavailability in Healthy Men. J. Nutr. 2016, 146, 2421–2428. [Google Scholar] [CrossRef]
- Borel, P.; Desmarchelier, C. Bioavailability of Fat-Soluble Vitamins and Phytochemicals in Humans: Effects of Genetic Variation. Ann. Rev. Nutr. 2018, 38, 69–96. [Google Scholar] [CrossRef]
- Péter, S.; Holguin, F.; Wood, L.G.; Clougherty, J.E.; Raederstorff, D.; Antal, M.; Weber, P.; Eggersdorfer, M. Nutritional solutions to reduce risks of negative health impacts of air pollution. Nutrients 2015, 7, 10398–10416. [Google Scholar] [CrossRef]
- Agarwal, K.S.; Mughal, M.Z.; Upadhyay, P.; Berry, J.L.; Mawer, E.B.; Puliyel, J.M. The impact of atmospheric pollution on vitamin D status of infants and toddlers in Delhi, India. Arch. Dis. Child. 2002, 87, 111–113. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Scientific Opinion on Dietary Reference Values for vitamin C. EFSA Panel on Dietetic Products, Nutrition and Allergies (NDA). EFSA J. 2013, 11, 3418. [Google Scholar]
- Carr, A.C.; Shaw, G.M.; Fowler, A.A.; Natarajan, R. Ascorbate-dependent vasopressor synthesis: A rationale for vitamin C administration in severe sepsis and septic shock? Crit. Care 2015, 19, 418. [Google Scholar] [CrossRef] [PubMed]
- Schmoranzer, F.; Fuchs, N.; Markolin, G.; Carlin, E.; Sakr, L.; Sommeregger, U. Influence of a complex micronutrient supplement on the immune status of elderly individuals. Int. J. Vitam. Nutr. Res. 2009, 79, 308–318. [Google Scholar] [CrossRef]
- Scrimgeour, A.G.; Condlin, M.L. Zinc and micronutrient combinations to combat gastrointestinal inflammation. Curr. Opin. Clin. Nutr. Metab. Care 2009, 12, 653–660. [Google Scholar] [CrossRef] [PubMed]
- Girodon, F.; Galan, P.; Monget, A.L.; Boutron-Ruault, M.C.; Brunet-Lecomte, P.; Preziosi, P.; Arnaud, J.; Manuguerra, J.C.; Herchberg, S. Impact of trace elements and vitamin supplementation on immunity and infections in institutionalized elderly patients: A randomized controlled trial. Arch. Intern. Med. 1999, 159, 748–754. [Google Scholar] [CrossRef]
- Penn, N.D.; Purkins, L.; Kelleher, J.; Heatley, R.V.; Mascie-Taylor, B.H.; Belfield, P.W. The effect of dietary supplementation with vitamins A, C, and E on cell-mediated immune function in elderly log-stay patients: A randomized controlled trial. Age Ageing 1991, 20, 169–174. [Google Scholar] [CrossRef]
- Kieliszek, M. Selenium⁻Fascinating Microelement, Properties and Sources in Food. Molecules 2019, 24, 1298. [Google Scholar] [CrossRef]
- EFSA Panel on Food Additives and Nutrient Sources added to Food (ANS). Scientific Opinion on the re-evaluation of iron oxides and hydroxides (E 172) as food additives. EFSA J. 2015, 13, 4317. [Google Scholar]
- Martinez-Estevez, N.S.; Alvarez-Guevara, A.N.; Rodriguez-Martinez, C.E. Effects of zinc supplementation in the prevention of respiratory tract infections and diarrheal disease in Colombian children: A 12-month randomised controlled trial. Allergol. Immunopathol. 2016, 44, 368–375. [Google Scholar] [CrossRef]
- Ginde, A.A.; Blatchford, P.; Breese, K.; Zarrabi, L.; Linnebur, S.A.; Wallace, J.I.; Schwartz, R.S. High-Dose Monthly Vitamin D for Prevention of Acute Respiratory Infection in Older Long-Term Care Residents: A Randomized Clinical Trial. J. Am. Geriatr. Soc. 2017, 65, 496–503. [Google Scholar] [CrossRef]
- Hemila, H. Vitamin E administration may decrease the incidence of pneumonia in elderly males. Clin. Interv. Aging 2016, 11, 1379–1385. [Google Scholar] [CrossRef] [PubMed]
- Yousefichaijan, P.; Naziri, M.; Taherahmadi, H.; Kahbazi, M.; Tabaei, A. Zinc Supplementation in Treatment of Children with Urinary Tract Infection. Iran. J. Kidney Dis 2016, 10, 213–216. [Google Scholar] [PubMed]
- Johnston, C.; Barkyoumb, G.M.; Schumacher, S.S. Vitamin C supplementation slightly improves physical activity levels and reduces cold incidence in men with marginal vitamin C status: A randomized controlled trial. Nutrients 2014, 6, 2572–2583. [Google Scholar] [CrossRef] [PubMed]
- Constantini, N.W.; Dubnov-Raz, G.; Eyal, B.B.; Berry, E.M.; Cohen, A.H.; Hemila, H. The effect of vitamin C on upper respiratory infections in adolescent swimmers: A randomized trial. Eur. J. Pediatr. 2011, 170, 59–63. [Google Scholar] [CrossRef]
- Sezikli, M.; Cetinkaya, Z.A.; Sezikli, H.; Guzelbulut, F.; Tiftikci, A.; Ince, A.T.; Gokden, Y.; Yasar, B.; Atalay, S.; Kurdas, O.O. Oxidative stress in Helicobacter pylori infection: Does supplementation with vitamins C and E increase the eradication rate? Helicobacter 2009, 14, 280–285. [Google Scholar] [CrossRef]
- Meydani, S.N.; Barnett, J.B.; Dallal, G.E.; Fine, B.C.; Jacques, P.F.; Leka, L.S.; Hamer, D.H. Serum zinc and pneumonia in nursing home elderly. Am. J. Clin. Nutr. 2007, 86, 1167–1173. [Google Scholar] [CrossRef]
- Stephen, A.I.; Avenell, A. A systematic review of multivitamin and multimineral supplementation for infection. J. Hum. Nutr Diet. 2006, 19, 179–190. [Google Scholar] [CrossRef]
- Imdad, A.; Mayo-Wilson, E.; Herzer, K.; Bhutta, Z.A. Vitamin A supplementation for preventing morbidity and mortality in children from six months to five years of age. Cochrane Database Syst. Rev. 2017, 3, CD008524. [Google Scholar] [CrossRef]
- Mathew, J.L. Vitamin A supplementation for prophylaxis or therapy in childhood pneumonia: A systematic review of randomized controlled trials. Indian Pediatr. 2010, 47, 255–261. [Google Scholar] [CrossRef]
- Chen, H.; Zhuo, Q.; Yuan, W.; Wang, J.; Wu, T. Vitamin A for preventing acute lower respiratory tract infections in children up to seven years of age. Cochrane Database Syst. Rev. 2008, CD006090. [Google Scholar] [CrossRef]
- Martineau, A.R.; Jolliffe, D.A.; Hooper, R.L.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; et al. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ 2017, 356, i6583. [Google Scholar] [CrossRef] [PubMed]
- Rejnmark, L.; Bislev, L.S.; Cashman, K.D.; Eiriksdottir, G.; Gaksch, M.; Grubler, M.; Grimnes, G.; Gudnason, V.; Lips, P.; Pilz, S.; et al. Non-skeletal health effects of vitamin D supplementation: A systematic review on findings from meta-analyses summarizing trial data. PLoS ONE 2017, 12, e0180512. [Google Scholar] [CrossRef] [PubMed]
- Bergman, P.; Lindh, Å.; Björkhem-Bergman, L.; Lindh, J. Vitamin D and respiratory tract infections: A systematic review and meta-analysis of randomized controlled trials. PLoS ONE 2013, 8, e65835. [Google Scholar] [CrossRef] [PubMed]
- Charan, J.; Goyal, J.; Saxena, D.; Yadav, P. Vitamin D for prevention of respiratory tract infections: A systematic review and meta-analysis. J. Pharmacol. Pharmacother. 2012, 3, 300–303. [Google Scholar] [CrossRef] [PubMed]
- Yamshchikov, A.V.; Desai, N.S.; Blumberg, H.M.; Ziegler, T.R.; Tangpricha, V. Vitamin D for treatment and prevention of infectious diseases: A systematic review of randomized controlled trials. Endocr. Pract. 2009, 15, 438–449. [Google Scholar] [CrossRef]
- Vuichard Gysin, D.; Dao, D.; Gysin, C.M.; Lytvyn, L.; Loeb, M. Effect of Vitamin D3 Supplementation on Respiratory Tract Infections in Healthy Individuals: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. PLoS ONE 2016, 11, e0162996. [Google Scholar] [CrossRef]
- Xiao, L.; Xing, C.; Yang, Z.; Xu, S.; Wang, M.; Du, H.; Liu, K.; Huang, Z. Vitamin D supplementation for the prevention of childhood acute respiratory infections: A systematic review of randomised controlled trials. Br. J. Nutr. 2015, 114, 1026–1034. [Google Scholar] [CrossRef]
- Yakoob, M.Y.; Salam, R.A.; Khan, F.R.; Bhutta, Z.A. Vitamin D supplementation for preventing infections in children under five years of age. Cochrane Database Syst. Rev. 2016, 11, CD008824. [Google Scholar] [CrossRef]
- Hemilä, H.; Chalker, E. Vitamin C for preventing and treating the common cold. Cochrane Database Syst. Rev. 2013, 1, CD000980. [Google Scholar] [CrossRef]
- Moreira, A.; Kekkonen, R.A.; Delgado, L.; Fonseca, J.; Korpela, R.; Haahtela, T. Nutritional modulation of exercise-induced immunodepression in athletes: A systematic review and meta-analysis. Eur. J. Clin. Nutr. 2007, 61, 443–460. [Google Scholar] [CrossRef]
- Hemila, H.; Louhiala, P. Vitamin C for preventing and treating pneumonia. Cochrane Database Syst. Rev. 2013, CD005532. [Google Scholar] [CrossRef]
- Ghouri, F.; Hollywood, A.; Ryan, K. A systematic review of non-antibiotic measures for the prevention of urinary tract infections in pregnancy. BMC Pregnancy Childbirth 2018, 18, 99. [Google Scholar] [CrossRef] [PubMed]
- Gulani, A.; Sachdev, H.S. Zinc supplements for preventing otitis media. Cochrane Database Syst. Rev. 2014, 4, CD006639. [Google Scholar] [CrossRef] [PubMed]
- Roth, D.E.; Richard, S.A.; Black, R.E. Zinc supplementation for the prevention of acute lower respiratory infection in children in developing countries: Meta-analysis and meta-regression of randomized trials. Int. J. Epidemiol. 2010, 39, 795–808. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, R.; Sentz, J.; Miller, M.A. Role of zinc administration in prevention of childhood diarrhea and respiratory illnesses: A meta-analysis. Pediatrics 2007, 119, 1120–1130. [Google Scholar] [CrossRef] [PubMed]
- Mayo-Wilson, E.; Imdad, A.; Junior, J.; Dean, S.; Bhutta, Z.A. Preventive zinc supplementation for children, and the effect of additional iron: A systematic review and meta-analysis. BMJ Open 2014, 4, e004647. [Google Scholar] [CrossRef] [PubMed]
- Mayo-Wilson, E.; Junior, J.A.; Imdad, A.; Dean, S.; Chan, X.H.; Chan, E.S.; Jaswal, A.; Bhutta, Z.A. Zinc supplementation for preventing mortality, morbidity, and growth failure in children aged 6 months to 12 years of age. Cochrane Database Syst. Rev. 2014, 5, CD009384. [Google Scholar] [CrossRef] [PubMed]
- Gera, T.; Sachdev, H.P. Effect of iron supplementation on incidence of infectious illness in children: Systematic review. BMJ 2002, 325, 1142. [Google Scholar] [CrossRef]
- De Gier, B.; Campos Ponce, M.; van de Bor, M.; Doak, C.M.; Polman, K. Helminth infections and micronutrients in school-age children: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2014, 99, 1499–1509. [Google Scholar] [CrossRef] [PubMed]
- El-Kadiki, A.; Sutton, A.J. Role of multivitamins and mineral supplements in preventing infections in elderly people: Systematic review and meta-analysis of randomised controlled trials. BMJ 2005, 330, 871. [Google Scholar] [CrossRef]
- Wu, T.; Ni, J.; Wei, J. Vitamin A for non-measles pneumonia in children. Cochrane Database Syst. Rev. 2005, 3, CD003700. [Google Scholar] [CrossRef] [PubMed]
- Glasziou, P.P.; Mackerras, D.E. Vitamin A supplementation in infectious diseases: A meta-analysis. BMJ 1993, 306, 366–370. [Google Scholar] [CrossRef] [PubMed]
- Das, R.R.; Singh, M.; Naik, S.S. Vitamin D as an adjunct to antibiotics for the treatment of acute childhood pneumonia. Cochrane Database Syst. Rev. 2018, 7, CD011597. [Google Scholar] [CrossRef] [PubMed]
- Ran, L.; Zhao, W.; Wang, J.; Wang, H.; Zhao, Y.; Tseng, Y.; Bu, H. Extra dose of vitamin C based on a daily supplementation shortens the common cold: A meta-analysis of 9 randomized controlled trials. BioMed Res. Int. 2018, 2018, 1837634. [Google Scholar] [CrossRef]
- Li, G.; Li, L.; Yu, C.; Chen, L. Effect of vitamins C and E supplementation on Helicobacter pylori eradication: A meta-analysis. Br. J. Nutr. 2011, 106, 1632–1637. [Google Scholar] [CrossRef]
- Caicedo Ochoa, E.Y.; Quintero Moreno, C.O.; Mendez Fandino, Y.R.; Sanchez Fonseca, S.C.; Cortes Motta, H.F.; Guio Guerra, S.A. Assessment of the use of vitamin C and E supplements concomitantly to antibiotic treatment against Helicobacter pylori: A systematic review and meta-analysis. Med. Clin. 2018, 151, 45–52. [Google Scholar] [CrossRef]
- Hemilä, H. Zinc lozenges may shorten the duration of colds: A systematic review. Open Respir. Med. J. 2011, 5, 51–58. [Google Scholar] [PubMed]
- Tie, H.T.; Tan, Q.; Luo, M.Z.; Li, Q.; Yu, J.L.; Wu, Q.C. Zinc as an adjunct to antibiotics for the treatment of severe pneumonia in children <5 years: A meta-analysis of randomised-controlled trials. Br. J. Nutr. 2016, 115, 807–816. [Google Scholar]
- Das, R.R.; Singh, M.; Shafiq, N. Short-term therapeutic role of zinc in children <5 years of age hospitalised for severe acute lower respiratory tract infection. Paediatr. Respir. Rev. 2012, 13, 184–191. [Google Scholar]
- Haider, B.A.; Lassi, Z.S.; Ahmed, A.; Bhutta, Z.A. Zinc supplementation as an adjunct to antibiotics in the treatment of pneumonia in children 2 to 59 months of age. Cochrane Database Syst. Rev. 2011, 10, CD007368. [Google Scholar] [CrossRef]
- Grobler, L.; Nagpal, S.; Sudarsanam, T.D.; Sinclair, D. Nutritional supplements for people being treated for active tuberculosis. Cochrane Database Syst. Rev. 2016, 6, CD006086. [Google Scholar] [CrossRef] [PubMed]
- Pilz, S.; März, W.; Cashman, K.D.; Kiely, M.E.; Whiting, S.J.; Holick, M.F.; Grant, W.B.; Pludowski, P.; Hiligsmann, M.; Trummer, C.; et al. Rationale and Plan for Vitamin D Food Fortification: A Review and Guidance Paper. Front. Endocrinol. 2018, 9, 373. [Google Scholar] [CrossRef] [PubMed]
- Mason, J.; Lotfi, M.; Dalmiya, N.; Sethuraman, K.; Deitchler, M. The Micronutrient Report: Current Progress and Trends in the Control of Vitamin A, Iodine, and Iron Deficiencies; Micronutrient Initiative and International Development Research Centre: Ottawa, ON, Canada, 2001. [Google Scholar]
- Guallar, E.; Stranges, S.; Mulrow, C.; Appel, L.J.; Miller, E.R. Enough Is Enough: Stop Wasting Money on Vitamin and Mineral Supplements. Ann. Intern. Med. 2013, 159, 850–851. [Google Scholar] [CrossRef]
- Gombart, A.F.; CRONOS Study Group. Effect of multimicronutrient supplementation on the function of the immune system in healthy elderly adults. in preparation.
- World Health Organization. Guideline: Vitamin A Supplementation in Postpartum Women; World Health Organization: Geneva, Switzerland, 2011. Available online: https://www.ncbi.nlm.nih.gov/books/NBK185192/ (accessed on 16 December 2019).
- World Health Organization. Guideline: Vitamin A Supplementation in Pregnant Women; World Health Organization: Geneva, Switzerland, 2011. Available online: https://www.ncbi.nlm.nih.gov/books/NBK304288/ (accessed on 16 December 2019).
- Huang, H.-Y.; Caballero, B.; Chang, S.; Alberg, A.; Semba, R.; Schneyer, C.; Wilson, R.F.; Cheng, T.-Y.; Prokopowicz, G.; Barnes, G.J.; et al. Multivitamin/mineral supplements and prevention of chronic disease. Evid Rep. Technol Assess. Full Rep. 2006, 139, 1–117. [Google Scholar]
- Slatore, C.G.; Littman, A.J.; Au, D.H.; Satia, J.A.; White, E. Long-term use of supplemental multivitamins, vitamin C, vitamin E, and folate does not reduce the risk of lung cancer. Am. J. Respir. Crit. Care Med. 2008, 177, 524–530. [Google Scholar] [CrossRef] [PubMed]
| Immune Function Roles | Micronutrient | Comments |
|---|---|---|
| Physical and biochemical barriers | ||
| Maintenance of structural and functional integrity of mucosal cells in innate barriers (e.g., skin, respiratory tract) | Vitamin A | Normal differentiation of epithelial tissue; retinoic acid essential to imprint T and B cells with gut-homing specificity and array T cells and IgA+ cells into intestinal tissues [8]. important for intestinal immune response, thus supporting the gut barrier [10,11,12]; carotenoids (either provitamin A or nonprovitamin A carotenoids) have immunoregulatory actions including reducing the toxic effects of ROS and regulating membrane fluidity and gap-junctional communication [13] |
| Vitamin D | Calcitriol regulates antimicrobial proteins (cathelicidin and β-defensin), responsible for modifying intestinal microbiota to a healthier composition and supporting the gut barrier [10,14], as well as protecting the lungs against infection [15]; increases tight junction protein expression, E-cadherin, and connexion 43 in the gut [16,17,18]; maintains renal epithelial barrier function [19]; enhances corneal epithelial barrier function [20] | |
| Vitamin C | Promotes collagen synthesis and protects cell membranes from damage caused by free radicals, thus supporting integrity of epithelial barriers [8]; enhances keratinocyte differentiation and lipid synthesis as well as fibroblast proliferation and migration [21] | |
| Vitamin E | Protects cell membranes from damage caused by free radicals and support the integrity of epithelial barriers [7,8] | |
| Vitamins B6, B12 and folate | All involved in intestinal immune regulation (e.g., by mediating lymphocyte migration into the intestine in the case of vitamin B6, while folate is essential for the survival of regulatory T cells in the small intestine, and human gut microbes use vitamin B12 as a cofactor for metabolic pathways), thus supporting the gut barrier [10,22] | |
| Iron | Essential for differentiation and growth of epithelial tissue [3] | |
| Zinc | Helps maintain integrity of skin and mucosal membrane (e.g., cofactor for metalloenzymes required for cell membrane repair [23]) | |
| Immune cells | ||
| Differentiation, proliferation, functioning, and movement of innate immune cells | Vitamin A | For example, regulates number and function of NK cells [2,8], contributes to phagocytic and oxidative burst activity of macrophages [8] |
| Vitamin D | Vitamin D receptor found in, e.g., monocytes, macrophages, and DCs [7]; increases differentiation of monocytes to macrophages [3]; calcitriol promotes movement and phagocytic ability of macrophages [8,24,25,26] | |
| Vitamin C | Involved in proliferation, function, and movement of neutrophils, monocytes, phagocytes [2]; maintains or enhances NK cell activities and chemotaxis [2,3,8,27]; enhances phagocytosis and ROS generation, enhances microbial killing [21]; involved in apoptosis and clearance of spent neutrophils from sites of infection by macrophages [21]; attenuates extracellular trap (NET) formation, thus reducing associated tissue damage [28] | |
| Vitamin E | Maintains or enhances NK cell cytotoxic activity [2,3,8,27]; inhibits PGE2 production by macrophages (thus indirectly protecting T-cell function) [27,29] | |
| Vitamin B6 | Maintains or enhances NK cell cytotoxic activity [2,8,27] | |
| Vitamin B12 | May act as immunomodulator for cellular immunity, effects on cytotoxic cells (e.g., NK cells, cytotoxic T cells) [3] | |
| Folate | Maintains or enhances NK cell cytotoxic activity [2,8,27] | |
| Zinc | Maintains or enhances NK cell cytotoxic activity [2,8,27]; central role in cellular growth and differentiation of immune cells that have a rapid differentiation and turnover [30]; enhances the phagocytic activity of peritoneal macrophages for E. coli and S. aureus [31]; improves phagocytic capacity of monocytes [27,32] | |
| Iron | Forms highly-toxic hydroxyl radicals, thus involved in killing of bacteria by neutrophils; component of enzymes critical for functioning of immune cells (e.g., ribonucleotide reductase involved in DNA synthesis); involved in regulation of cytokine production and action [3]; iron-rich status promotes M2-like macrophage phenotype and negatively regulates M1 pro-inflammatory response [33] | |
| Copper | Role in functions of macrophages (e.g., copper accumulates in phagolysosomes of macrophages to combat certain infectious agents [34]), neutrophils and monocytes [35]; enhances NK cell activity [36] | |
| Selenium | Selenoproteins important for antioxidant host defense system, affecting leukocyte and NK cell function [35] | |
| Magnesium | Cofactor of enzymes of nucleic acid metabolism and stabilizes structure of nucleic acids; involved in DNA replication and repair [37]; roles in antigen binding to macrophages [38]; regulates leukocyte activation [38]; involved in the regulation of apoptosis [37] | |
| Antimicrobial effects | Vitamin A | Downregulates IFNγ production [3,21] |
| Vitamin D | Calcitriol regulates antimicrobial protein expression (cathelicidin and defensin), which directly kill pathogens, especially bacteria [7,39,40,41]; inhibits IFNγ production [27,42,43,44,45] | |
| Vitamin C | High levels can improve antimicrobial effects; increases serum levels of complement proteins [7]; role in IFNγ production [3,21] | |
| Zinc | Involved in complement activity; role in IFNγ production [3,21] | |
| Iron | Role in IFNγ production [3,21] | |
| Copper | Intrinsic antimicrobial properties [7] | |
| Selenium | Increases IFNγ production [3,21] | |
| Roles in inflammation, antioxidant effects, and effects in oxidative burst | Vitamin A | Helps to regulate the production of IL-2 and the proinflammatory TNF-α, which activates the microbial action of macrophages; involved in phagocytic and oxidative burst activity of macrophages activated during inflammation [8] |
| Vitamin D | Calcitriol increases the oxidative burst potential of macrophages [24,25,26]; increases superoxide synthesis [8]; reduces the expression of pro-inflammatory cytokines and increases the expression of anti-inflammatory cytokines by macrophages [24,46,47,48] | |
| Vitamin C | Maintains redox homeostasis within cells and protects against ROS and RNS during oxidative burst [8]; regenerates other important antioxidants, such as glutathione and vitamin E, to their active state [49]; modulates cytokine production and decreases histamine levels [21] | |
| Vitamin E | Important fat-soluble antioxidant that hinders the chain reaction induced by free radicals (chain-breaking effect) and protects cells against them [3,7]; enhances IL-2 production [3]; decreases production of PGE2 (indirectly protecting T-cell function) [50] | |
| Vitamin B6 | Required in endogenous synthesis and metabolism of amino acids, the building blocks of cytokines [7]; helps to regulate inflammation (higher levels of the active form result in lower rates of inflammation) [35,51,52] | |
| Zinc | Anti-inflammatory agent [53]; helps to modulate cytokine release [3,49] by dampening the development pro-inflammatory Th17 and Th9 cells [27,54,55] and influencing the generation of cytokines such as IL-2, IL-6, and TNF-α [56,57]; has antioxidant effects that protect against ROS and reactive nitrogen species [49]; influences activity of antioxidant proteins [8] | |
| Iron | Involved in regulation of cytokine production and action [3]; required for generation of pathogen-killing ROS by neutrophils during oxidative burst [7] | |
| Copper | Accumulates at sites of inflammation [7,35]; part of copper/zinc-superoxide dismutase, a key enzyme in defense against ROS [8]; free-radical scavenger [58]; changes in copper homeostasis a crucial component of respiratory burst [8]; important for IL-2 production and response [7,35]; maintains intracellular antioxidant balance, suggesting important role in inflammatory response [8] | |
| Selenium | Essential for function of selenoproteins that act as redox regulators and cellular antioxidants, potentially counteracting ROS produced during oxidative stress [2] | |
| Magnesium | Can help to protect DNA against oxidative damage [37]; high concentrations reduce superoxide anion production [59] | |
| Differentiation, proliferation and normal functioning of T cells | Vitamin A | Involved in development and differentiation of Th1 and Th2 cells [60]; enhances TGF-β-dependent conversion of naïve T cells into regulatory T cells [8]; plays a role in acquisition of mucosal-homing properties by T and B cells [8] |
| Vitamin D | Homing of T cells to the skin [61]; calcitriol inhibits T-cell proliferation [7]; inhibitory effects mainly in adaptive immunity (e.g., Th1-cell activity) [7]; stimulatory effects in innate immunity [7]; inhibits the effector functions of T helper cells and cytotoxic T cells [27,62], but promotes the production of Tregs [27,62,63]; inhibitory effect on the differentiation and maturation of the antigen-presenting DCs, and helps program DCs for tolerance [27,64,65,66] | |
| Vitamin C | Roles in production, differentiation, and proliferation of T cells, particularly cytotoxic T cells [3,21] | |
| Vitamin E | Enhances lymphocyte proliferation and T-cell-mediated functions [3]; optimizes and enhances Th1 response [3] | |
| Vitamin B6 | Involved in lymphocyte proliferation, differentiation, maturation, and activity [7]; maintains Th1 immune response [3] | |
| Vitamin B12 | Involved in one-carbon metabolism (interactions with folate) [35]; facilitates production of T cells [35], such as cytotoxic T cells [3,8]; helps to regulate ratio between T helper cells and cytotoxic T cells [8] | |
| Folate | Supports Th 1-mediated immune response [35] | |
| Zinc | Induces proliferation of cytotoxic T cells [67]; involved in Th1 cytokine production and thus supports Th1 response [3]; essential for intracellular binding of tyrosine kinase to T cell receptors, required for T cell development, differentiation, and activation [49]; induces development of Treg cells and is thus important in maintaining immune tolerance [27,54,55] | |
| Iron | Important in differentiation and proliferation of T cells [7]; helps to regulate ratio between T helper cells and cytotoxic T cells [3] | |
| Copper | Roles in differentiation and proliferation of T cells [35] | |
| Selenium | Roles in differentiation and proliferation of T cells [35,58]; helps to improve Th cell counts [68] | |
| Antibodies | ||
| Antibody production and development | Vitamin A | Development and differentiation of Th 1 and Th2 cells [8]; maintains normal antibody-mediated Th2 response by suppressing IL-12, TNF-α, and IFN-γ production of Th1 cells [7] |
| Vitamin D | Calcitriol suppresses antibody production by B cells [7] | |
| Vitamin C | Promotes proliferation of lymphocytes, resulting in increased generation of antibodies [21] | |
| Vitamin E | Suppresses Th2 response [3] | |
| Vitamin B6 | Required in endogenous synthesis and metabolism of amino acids, the building blocks of antibodies [7]; inhibits Th2 cytokine-mediated activity [8] | |
| Vitamin B12 | Important for antibody production and metabolism, via folate mechanism [7,8,35]; required for optimal clonal expansion [8] | |
| Folate | Important for antibody production and metabolism [7,8,35] | |
| Zinc | Involved in antibody production, particularly IgG [69,70] | |
| Selenium | Helps to maintain antibody levels [35] | |
| Magnesium | Cofactor in antibody synthesis, role in antibody-dependent cytolysis and IgM lymphocyte binding [38] | |
| Responses to antigen | Vitamin A | Normal functioning of B cells, necessary for generation of antibody responses to antigen [7]; required for B cell-mediated IgA antibody responses to bacterial polysaccharide antigens [8] |
| Vitamin D | Promotes antigen processing [8]; role in the down-regulation of MHC-II [35] | |
| Vitamin E | Helps to form effective immune synapses between and Th cells [27]; increases proportion of antigen-experienced memory T cells [71] | |
| Folate | Important for sufficient antibody response to antigens [35] | |
| Zinc | Involved in antibody response [8]; important in maintaining immune tolerance (i.e., the ability to recognize “self” from “non-self”) [27] | |
| Magnesium | Key role in antigen binding to macrophage RNA [38]; involved in antibody-dependent cytolysis [38] | |
| Micronutrient | Impact of Deficiency | Impact of Supplementation on Immune Functions | |
|---|---|---|---|
| Immune Functions | Decreased Resistance to Infection(s) | ||
| Vitamin C | ✓ |
| |
| [8,21,109] | |||
| Vitamin D | ✓ | ||
| |||
| Vitamin A |
| ✓ | |
| [106,122] | |||
| |||
| Vitamin E | ✓ |
| |
| [35,123] | |||
| Vitamin B6 | ✓ | ||
| |||
| Vitamin B12 * | ✓ |
| |
| (potentially) [127] | |||
| Folate * | ✓ | ||
| [8] | |||
| Zinc |
| ✓ | |
| [122,135] | |||
| Iron |
| ✓ |
|
| [136] | |||
| Copper | ✓ |
| |
| (potentially) [7] | |||
| Selenium | ✓ | ||
| |||
| Magnesium | ✓ | ||
| |||
| Select Micronutrients | Recommended Dietary Allowance [143] | Reported Mean Micronutrient Intakes, Min–Max [114] | ||||
|---|---|---|---|---|---|---|
| Children, M/F a 4–8 years 9–13 years 14–18 years | Adults, M/F 19–50 years b | Older age, M/F 51 to >70 years | Children, M/F | Adults, M/F 19–50 years | Older age, M/F 51 to >70 years | |
| 4–6 years | ||||||
| 7–9 years | ||||||
| 10–14 years | ||||||
| 15–18 years | ||||||
| Vitamin C, mg/day | 25 45 75/65 | 90/75 | 90/75 | 60–157/61–157 | 64–153/62–153 | 59–142/60–160 |
| 63–172/57–172 | ||||||
| 73–197/77–222 | ||||||
| 71–201/67–205 | ||||||
| Vitamin D, μg/day | 15 | 15 | 15–20 | 1.8–5.8/1.5–6.5 | 1.6–10.9/1.2–10.1 | 0.7–15.0/0.7–12.9 |
| 1.5–6.4/1.5–5.1 | ||||||
| 1.5–4.8/1.2–4.5 | ||||||
| 1.8–7.5/1.5–7.1 | ||||||
| Vitamin A, μg/day | 400 600 900/700 | 900/700 | 900/700 | 400–1100/400–1200 | 500–2200/500–2200 | 500–2500/400–2300 |
| 400–1300/400–1100 | ||||||
| 400–2400/300–2300 | ||||||
| 400–1800/300–1600 | ||||||
| Vitamin E, mg/day | 7 11 15 | 15 | 15 | 5.3–9.8/5.1–9.8 | 3.3–17.7/4.2–16.1 | 6.3–13.7/6.7–13.7 |
| 6.3–11.2/5.9–13.3 | ||||||
| 5.9–14.5/5.6–18.1 | ||||||
| 6.8–20.8/6.0–15.5 | ||||||
| Vitamin B6, mg/day | 0.6 1.0 1.3/1.2 | 1.3 | 1.7/1.5 | 1.3–1.8/1.0–1.9 | 1.6–3.5/1.3–2.1 | 1.2–3.0/1.2–2.9 |
| 1.2–2.5/1.1–1.9 | ||||||
| 1.2–2.8/1.1–2.7 | ||||||
| 1.5–3.1/1.2–2.5 | ||||||
| Vitamin B12, μg/day | 1.2 1.8 2.4 | 2.4 | 2.4 | 2.7–5.3/2.6–5.0 | 1.9–9.3/1.0–8.8 | 3.1–8.2/2.5–7.5 |
| 3.6–5.5/2.2–5.3 | ||||||
| 3.2–11.8/2.2–11.1 | ||||||
| 4.9–7.5/3.5–5.2 | ||||||
| Folate, μg/day | 200 300 400 | 300–400 | 400 | 120–256/109–199 | 203–494/131–392 | 139–343/121–335 |
| 144–290/133–264 | ||||||
| 149–428/140–360 | ||||||
| 190–365/154–298 | ||||||
| Zinc, mg/day | 5 8 11/9 | 11/8 | 11/8 | 6.0–9.2/5.3–8.9 | 8.6–14.6/6.7–10.7 | 7.5–12.3/6.7–11.2 |
| 7.0–10.9/6.4–9.4 | ||||||
| 7.0–14.6/6.1–13.9 | ||||||
| 9.3–15.2/6.4–11.0 | ||||||
| Iron, mg/day | 10 8 11/15 | 8/18 | 8 | 7.3–10.6/6.8–10.6 | 10.6–26.9/8.2–22.2 | 10.2–25.2/8.5–20.9 |
| 8.4–11.8/7.7–11.8 | ||||||
| 9.2–19.4/7.7–14.8 | ||||||
| 10.2–19.0/7.8–14.0 | ||||||
| Copper, μg/day | 440 700 890 | 900 | 900 | 700–2200/700–2000 | 1100–2300/1000–2200 | 1100–1900/900–1900 |
| 900–2800/800–2600 | ||||||
| 800–2900/700–2800 | ||||||
| 1200–3400/800–2100 | ||||||
| Selenium, μg/day | 30 40 55 | 55 | 55 | 23–61/24–61 | 36–73/31–54 | 39–62/34–55 |
| 27–41/26–58 | ||||||
| 29–110/28–104 | ||||||
| 39–59/30–38 | ||||||
| Magnesium, mg/day | 130 240 410/360 | 400–420/310–320 | 420/320 | 171–267/166–267 | 256–465/192–372 | 221–403/179–348 |
| 204–303/166–301 | ||||||
| 200–503/181–429 | ||||||
| 260–467/186–369 | ||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gombart, A.F.; Pierre, A.; Maggini, S. A Review of Micronutrients and the Immune System–Working in Harmony to Reduce the Risk of Infection. Nutrients 2020, 12, 236. https://doi.org/10.3390/nu12010236
Gombart AF, Pierre A, Maggini S. A Review of Micronutrients and the Immune System–Working in Harmony to Reduce the Risk of Infection. Nutrients. 2020; 12(1):236. https://doi.org/10.3390/nu12010236
Chicago/Turabian StyleGombart, Adrian F., Adeline Pierre, and Silvia Maggini. 2020. "A Review of Micronutrients and the Immune System–Working in Harmony to Reduce the Risk of Infection" Nutrients 12, no. 1: 236. https://doi.org/10.3390/nu12010236
APA StyleGombart, A. F., Pierre, A., & Maggini, S. (2020). A Review of Micronutrients and the Immune System–Working in Harmony to Reduce the Risk of Infection. Nutrients, 12(1), 236. https://doi.org/10.3390/nu12010236





