Sarcopenia and Heart Failure
Abstract
1. Introduction
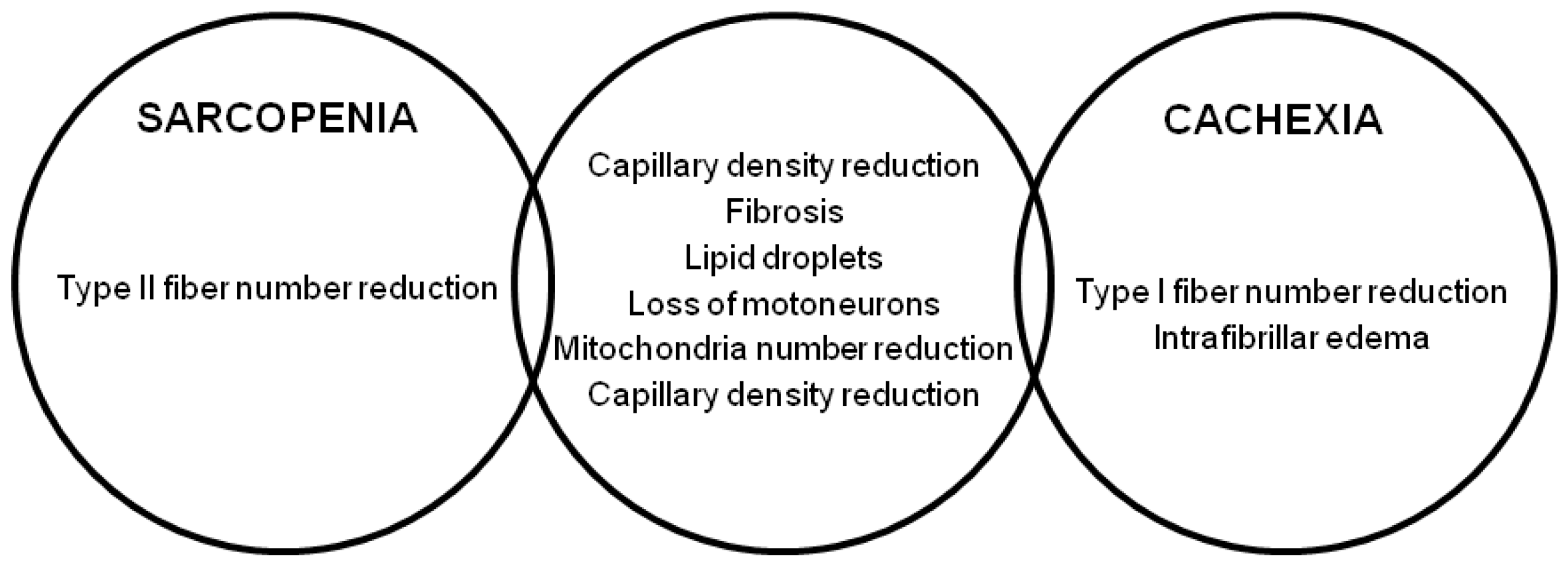
2. Muscle Changes in HF: Cachexia or Sarcopenia?
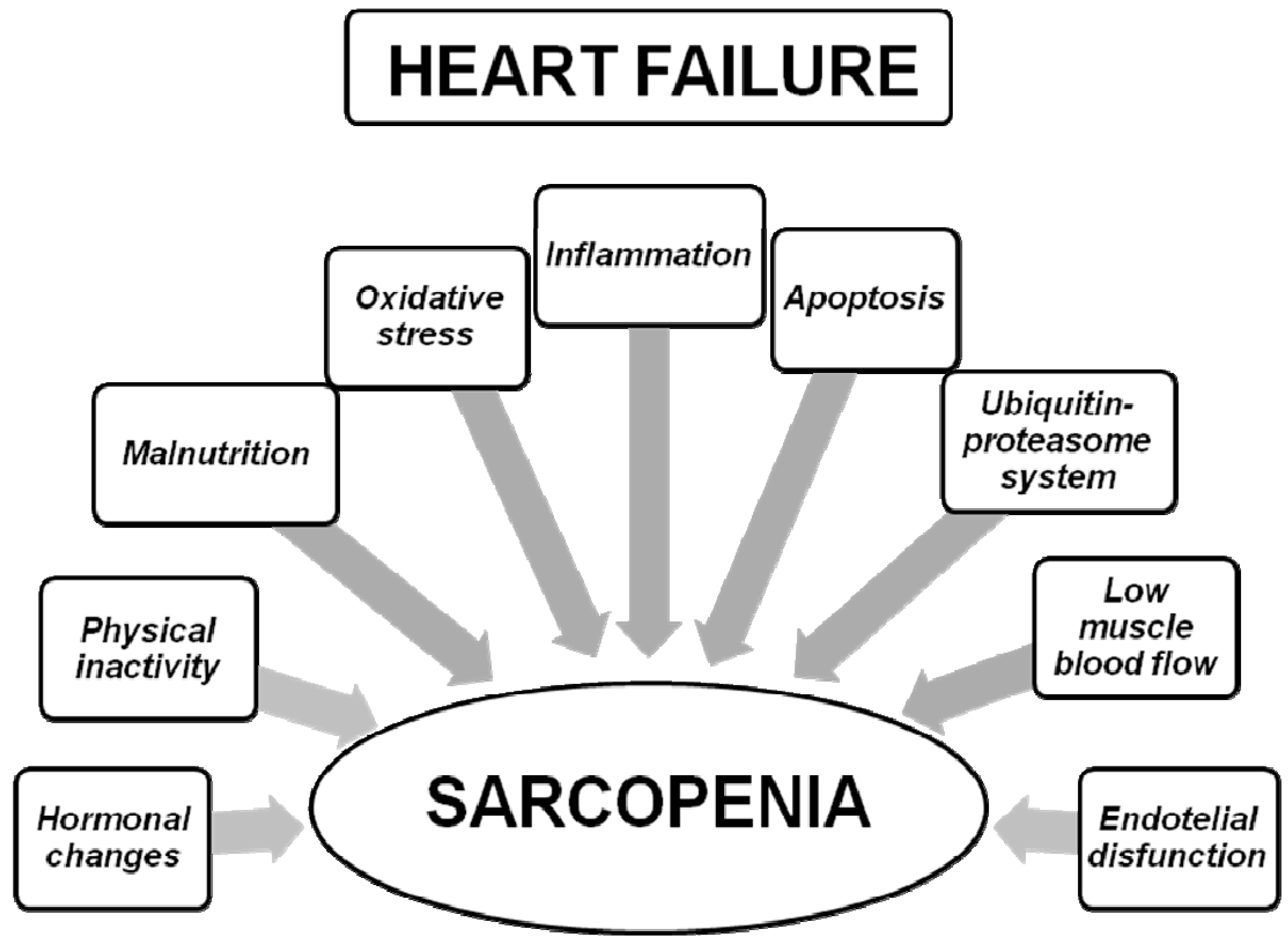
3. From Heart Failure to Sarcopenic Muscle
3.1. Hormonal Changes
3.2. Malnutrition and Malabsorption
3.3. Inflammation and Oxidative Stress
3.4. Apoptosis
3.5. Ubiquitin-Proteasome System (UPS)
3.6. Physical Inactivity
3.7. Low-Muscle Blood Flow and Endotelial Dysfunction
4. From Sarcopenic Muscle to Heart Failure
4.1. Sarcopenia, Ergoreflex, and HF
4.2. Sarcopenia, Muscle Fatigue, and Dyspnea in HF
5. Does a Sarcopenic Heart Exist?
6. Therapeutic Approach in Sarcopenic Patients with Chronic Heart Failure
6.1. Pharmacological Approach
6.2. Physical Activity Approach
6.3. Nutritional Approach
6.4. Single Therapeutic Approach for Targeting HF and Sarcopenia
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ponikowski, P.; Voors Adriaan, A.; Anker, S.D.; Bueno, H.; Cleland, J.G.F.; Coats, A.J.S.; Falk, V.; Gonzlez-Juanatey, J.R.; Harjola, V.-P.; Jankowska, E.A.; et al. ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur. J. Heart Fail. 2016, 18, 891–975. [Google Scholar] [PubMed]
- Go, A.S.; Mozaffarian, D.; Roger, V.L.; Benjamin, E.J.; Berry, J.D.; Blaha, M.J.; Dai, S.; Ford, E.S.; Fox, C.S.; Franco, S.; et al. Heart disease and stroke statistics—2014 update: A report from the American Heart Association. Circulation 2014, 129, e28–e292. [Google Scholar] [CrossRef] [PubMed]
- Carbone, S.; Lavie, C.J.; Arena, R. Obesity and heart failure: Focus on the obesity paradox. Mayo. Clin. Proc. 2017, 92, 266–279. [Google Scholar] [CrossRef] [PubMed]
- Muscaritoli, M.; Anker, S.D.; Argilés, J.; Aversa, Z.; Bauer, J.M.; Biolo, G.; Boirie, Y.; Bosaeus, I.; Cederholm, T.; Costelli, P.; et al. Consensus definition of sarcopenia, cachexia and pre-cachexia: Joint document elaborated by Special Interest Groups (SIG) “cachexia-anorexia in chronic wasting diseases” and “nutrition in geriatrics”. Clin. Nutr. 2010, 29, 154–159. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef]
- Von Haehling, S. The wasting continuum in heart failure: From sarcopenia to cachexia. Proc. Nutr. Soc. 2015, 74, 367–377. [Google Scholar] [CrossRef]
- Emami, A.; Saitoh, M.; Valentova, M.; Sandek, A.; Evertz, R.; Ebner, N.; Loncar, G.; Springer, J.; Doehner, W.; Lainscak, M.; et al. Comparison of sarcopenia and cachexia in men with chronic heart failure: Results from the Studies Investigating Co-Morbidities Aggravating Heart Failure (SICA-HF). Eur. J. Heart Fail. 2018, 20, 1580–1587. [Google Scholar] [CrossRef]
- Lin, J.; Lopez, E.F.; Jin, Y.; Van Remmen, H.; Bauch, T.; Han, H.C.; Lindsey, M.L. Age-related cardiac muscle sarcopenia: Combining experimental and mathematical modelling to identify mechanism. Exp. Gerontol. 2008, 43, 296–306. [Google Scholar] [CrossRef]
- Narici, M.V.; Maffulli, N. Sarcopenia: Characteristics, mechanisms and functional significance. Br. Med. Bull. 2010, 95, 139–159. [Google Scholar] [CrossRef]
- Zamboni, M.; Rossi, A.P.; Corzato, F.; Bambace, C.; Mazzali, G.; Fantin, F. Sarcopenia, cachexia and congestive heart failure in the elderly. Endocr. Metab. Immune Disord. Drug. Targets 2013, 13, 58–67. [Google Scholar] [CrossRef]
- Drexler, H.; Riede, U.; Münzel, T.; König, H.; Funke, E.; Just, H. Alterations of skeletal muscle in chronic heart failure. Circulation 1992, 85, 1751–1759. [Google Scholar] [CrossRef] [PubMed]
- Basile, C.; Della-Morte, D.; Cacciatore, F.; Gargiulo, G.; Galizia, G.; Roselli, M.; Curcio, F.; Bonaduce, D.; Abete, P. Phase angle as bioelectrical marker to identify elderly patients at risk of sarcopenia. Exp. Gerontol. 2014, 58, 43–46. [Google Scholar] [CrossRef]
- Von Haehling, S.; Ebner, N.; Dos Santos, M.R.; Springer, J.; Anker, S.D. Muscle wasting and cachexia in heart failure: Mechanisms and therapies. Nat. Rev. Cardiol. 2017, 14, 323–341. [Google Scholar] [CrossRef]
- Bristow, M.R. The adrenergic nervous system in heart failure. N. Engl. J. Med. 1984, 311, 850–851. [Google Scholar] [CrossRef] [PubMed]
- Brink, M.; Price, S.R.; Chrast, J.; Bailey, J.L.; Anwar, A.; Mitch, W.E.; Delafontaine, P. Angiotensin II induces skeletal muscle wasting through enhanced protein degradation and down-regulates autocrine insulin-like growth factor I. Endocrinology 2001, 142, 1489–1496. [Google Scholar] [CrossRef]
- Delafontaine, P.; Yoshida, T. The renin-angiotensin system and the biology of skeletal muscle: Mechanisms of muscle wasting in chronic disease states. Trans. Am. Clin. Climatol. Assoc. 2016, 127, 245–258. [Google Scholar]
- Marzetti, E.; Calvani, R.; DuPree, J.; Lees, H.A.; Giovannini, S.; Seo, D.O.; Buford, T.W.; Sweet, K.; Morgan, D.; Strehler, K.Y.; et al. Late-life enalapril administration induces nitric oxide-dependent and independent metabolic adaptations in the rat skeletal muscle. Age 2013, 35, 1061–1075. [Google Scholar] [CrossRef] [PubMed]
- Brown, D.A.; Perry, J.B.; Allen, M.E.; Sabbah, H.N.; Stauffer, B.L.; Shaikh, S.R.; Cleland, J.G.; Colucci, W.S.; Butler, J.; Voors, A.A.; et al. Expert consensus document: Mitochondrial function as a therapeutic target in heart failure. Nat. Rev. Cardiol. 2017, 14, 238–250. [Google Scholar] [CrossRef]
- Lee, J.F.; Barrett-O’Keefe, Z.; Nelson, A.D.; Garten, R.S.; Ryan, J.J.; Nativi-Nicolau, J.N.; Richardson, R.S.; Wray, D.W. Impaired skeletal muscle vasodilation during exercise in heart failure with preserved ejection fraction. Int. J. Cardiol. 2016, 211, 14–21. [Google Scholar] [CrossRef]
- Bossone, E.; Arcopinto, M.; Iacoviello, M.; Triggiani, V.; Cacciatore, F.; Maiello, C.; Limongelli, G.; Masarone, D.; Perticone, F.; Sciacqua, A.; et al. TOSCA Investigators. Multiple hormonal and metabolic deficiency syndrome in chronic heart failure: Rationale, design, and demographic characteristics of the TOSCA. Registry. Intern. Emerg. Med. 2018, 13, 661–671. [Google Scholar] [CrossRef]
- Onder, G.; Liperoti, R.; Russo, A.; Soldato, M.; Capoluongo, E.; Volpato, S.; Cesari, M.; Ameglio, F.; Bernabei, R.; Landi, F. Body mass index, free insulin-like growth factor I, and physical function among older adults: Results from the ilSIRENTE study. Am. J. Physiol. Endocrinol. Metab. 2006, 291, E829–E834. [Google Scholar] [CrossRef] [PubMed]
- Josiak, K.; Jankowska, E.A.; Piepoli, M.F.; Banasiak, W.; Ponikowski, P. Skeletal myopathy in patients with chronic heart failure: Significance of anabolic-androgenic hormones. J. Cachexia Sarcopenia Muscle 2014, 5, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Kontoleon, P.E.; Anastasiou-Nana, M.I.; Papapetrou, P.D.; Alexopoulos, G.; Ktenas, V.; Rapti, A.C.; Tsagalou, E.P.; Nanas, J.N. Hormonal profile in patients with congestive heart failure. Int. J. Cardiol. 2003, 87, 179–183. [Google Scholar] [CrossRef]
- Storer, T.W.; Magliano, L.; Woodhouse, L.; Lee, M.L.; Dzekov, C.; Dzekov, J.; Casaburi, R.; Bhasin, S. Testosterone dose-dependently increases maximal voluntary strength and leg power, but does not affect fatigability or specific tension. J. Clin. Endocrinol. Metab. 2003, 88, 1478–1485. [Google Scholar] [CrossRef] [PubMed]
- Müller, T.D.; Nogueiras, R.; Andermann, M.L.; Andrews, Z.B.; Anker, S.D.; Argente, J.; Batterham, R.L.; Benoit, S.C.; Bowers, C.Y.; Broglio, F.; et al. Ghrelin. Mol. Metab. 2015, 4, 437–460. [Google Scholar] [CrossRef]
- Lee, S.J.; McPherron, A.C. Regulation of myostatin activity and muscle growth. Proc. Natl. Acad. Sci. USA 2001, 98, 9306–9311. [Google Scholar] [CrossRef]
- LeBrasseur, N.K.; Schelhorn, T.M.; Bernardo, B.L.; Cosgrove, P.G.; Loria, P.M.; Brown, T.A. Myostatin inhibition enhances the effects of exercise on performance and metabolic outcomes in aged mice. J. Gerontol. A Biol. Sci. Med. Sci. 2009, 64, 940–948. [Google Scholar] [CrossRef]
- Gruson, D.; Ahn, S.A.; Ketelslegers, J.M.; Rousseau, M.F. Increased plasma myostatin in heart failure. Eur. J. Heart Fail. 2011, 13, 734–736. [Google Scholar] [CrossRef]
- Breitbart, A.; Auger-Messier, M.; Molkentin, J.D.; Heineke, J. Myostatin from the heart: Local and systemic actions in cardiac failure and muscle wasting. Am. J. Physiol. Heart Circ. Physiol. 2011, 300, H1973–H1982. [Google Scholar] [CrossRef]
- Heineke, J.; Auger-Messier, M.; Xu, J.; Sargent, M.; York, A.; Welle, S.; Molkentin, J.D. Genetic deletion of myostatin from the heart prevents skeletal muscle atrophy in heart failure. Circulation 2010, 121, 419–425. [Google Scholar] [CrossRef]
- Liguori, I.; Curcio, F.; Russo, G.; Cellurale, M.; Aran, L.; Bulli, G.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; et al. Risk of Malnutrition Evaluated by Mini Nutritional Assessment and Sarcopenia in Noninstitutionalized Elderly People. Nutr. Clin. Pract. 2018, 33, 879–886. [Google Scholar] [CrossRef]
- İlhan, B.; Bahat, G.; Erdoğan, T.; Kılıç, C.; Karan, M.A. Anorexia Is Independently Associated with Decreased Muscle Mass and Strength in Community Dwelling Older Adults. J. Nutr. Health Aging 2019, 23, 202–206. [Google Scholar] [CrossRef] [PubMed]
- Hussain, Z.; Swindle, J.; Hauptman, P.J. Digoxin use and digoxin toxicity in the post-DIG trial era. J. Card. Fail. 2006, 12, 343–346. [Google Scholar] [CrossRef] [PubMed]
- Cacciatore, F.; Della-Morte, D.; Basile, C.; Curcio, F.; Liguori, I.; Roselli, M.; Gargiulo, G.; Galizia, G.; Bonaduce, D.; Abete, P. Butyryl-cholinesterase is related to muscle mass and strength. A new biomarker to identify elderly subjects at risk of sarcopenia. Biomark. Med. 2015, 9, 669–678. [Google Scholar] [CrossRef] [PubMed]
- Schaap, L.A.; Pluijm, S.M.; Deeg, D.J.; Harris, T.B.; Kritchevsky, S.B.; Newman, A.B.; Colbert, L.H.; Pahor, M.; Rubin, S.M.; Tylavsky, F.A.; et al. Higher inflammatory marker levels in older persons: Associations with 5-year change in muscle mass and muscle strength. J. Gerontol. A Biol. Sci. Med. Sci. 2009, 64, 1183–1189. [Google Scholar] [CrossRef]
- Morley, J.E.; Thomas, D.R.; Wilson, M.M. Cachexia: Pathophysiology and clinical relevance. Am. J. Clin. Nutr. 2006, 83, 735–743. [Google Scholar] [CrossRef]
- Curcio, F.; Ferro, G.; Basile, C.; Liguori, I.; Parrella, P.; Pirozzi, F.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Tocchetti, C.G.; et al. Biomarkers in sarcopenia: A multifactorial approach. Exp. Gerontol. 2016, 85, 1–8. [Google Scholar] [CrossRef]
- Liguori, I.; Russo, G.; Curcio, F.; Bulli, G.; Aran, L.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; Bonaduce, D.; et al. Oxidative stress, aging, and diseases. Clin. Interv. Aging 2018, 26, 757–772. [Google Scholar] [CrossRef]
- Nishiyama, Y.; Ikeda, H.; Haramaki, N.; Yoshida, N.; Imaizumi, T. Oxidative stress is related to exercise intolerance in patients with heart failure. Am. Heart J. 1998, 135, 115–120. [Google Scholar] [CrossRef]
- Das, U.N. Free radicals, cytokines and nitric oxide in cardiac failure and myocardial infarction. Mol. Cell. Biochem. 2000, 215, 145–152. [Google Scholar] [CrossRef]
- Bouzid, M.A.; Filaire, E.; McCall, A.; Fabre, C. Radical Oxygen Species, Exercise and Aging: An Update. Sports Med. 2015, 45, 1245–1261. [Google Scholar] [CrossRef] [PubMed]
- Fulle, S.; Protasi, F.; Di Tano, G.; Pietrangelo, T.; Beltramin, A.; Boncompagni, S.; Vecchiet, L.; Fanò, G. The contribution of reactive oxygen species to sarcopenia and muscle ageing. Exp. Gerontol. 2004, 39, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Marzetti, E.; Privitera, G.; Simili, V.; Wohlgemuth, S.E.; Aulisa, L.; Pahor, M.; Leeuwenburgh, C. Multiple pathways to the same end: Mechanisms of myonuclear apoptosis in sarcopenia of aging. Sci. World J. 2010, 10, 340–349. [Google Scholar] [CrossRef] [PubMed]
- Filippatos, G.S.; Kanatselos, C.; Manolatos, D.D.; Vougas, B.; Sideris, A.; Kardara, D.; Anker, S.D.; Kardaras, F.; Uhal, B. Studies on apoptosis and fibrosis in skeletal musculature: A comparison of heart failure patients with and without cardiac cachexia. Int. J. Cardiol. 2003, 90, 107–113. [Google Scholar] [CrossRef]
- Saitoh, M.; Ishida, J.; Doehner, W. Sarcopenia, cachexia, and muscle performance in heart failure Review update 2016. Int. J. Cardiol. 2017, 238, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Gumucio, J.P.; Mendias, C.L. Atrogin-1, MuRF-1, and sarcopenia. Endocrine 2013, 43, 12–21. [Google Scholar] [CrossRef]
- Sakuma, K.; Yamaguchi, A. Sarcopenia and cachexia: The adaptations of negative regulators of skeletal muscle mass. J. Cachexia Sarcopenia Muscle 2012, 3, 77–94. [Google Scholar] [CrossRef] [PubMed]
- Gielen, S.; Sandri, M.; Kozarez, I.; Kratzsch, J.; Teupser, D.; Thiery, J.; Erbs, S.; Mangner, N.; Lenk, K.; Hambrecht, R.; et al. Exercise training attenuates MuRF-1expression in the skeletal muscle of patients with chronic heart failure independent of age: The randomized Leipzig Exercise Intervention in Chronic Heart Failure and Aging catabolism study. Circulation 2012, 125, 2716–2727. [Google Scholar] [CrossRef]
- Forbes, S.C.; Little, J.P.; Candow, D.G. Exercise and nutritional interventions for improving aging muscle health. Endocrine 2012, 42, 29–38. [Google Scholar] [CrossRef]
- Curcio, F.; Liguori, I.; Cellulare, M.; Sasso, G.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; Bonaduce, D.; Abete, P. Physical Activity Scale for the Elderly (PASE) Score Is Related to Sarcopenia in Noninstitutionalized Older Adults. J. Geriatr. Phys. Ther. 2019, 42, 130–135. [Google Scholar] [CrossRef]
- Coker, R.H.; Hays, N.P.; Williams, R.H.; Xu, L.; Wolfe, R.R.; Evans, W.J. Bed rest worsens impairments in fat and glucose metabolism in older, overweight adults. J. Gerontol. A Biol. Sci. Med. Sci. 2014, 69, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Sjöblom, S.; Suuronen, J.; Rikkonen, T.; Honkanen, R.; Kröger, H.; Sirola, J. Relationship between postmenopausal osteoporosis and the components of clinical sarcopenia. Maturitas 2013, 75, 175–180. [Google Scholar] [CrossRef] [PubMed]
- Drummond, M.J.; Timmerman, K.L.; Markofski, M.M.; Walker, D.K.; Dickinson, J.M.; Jamaluddin, M.; Brasier, A.R.; Rasmussen, B.B.; Volpi, E. Short-term bed rest increases TLR4 and IL-6 expression in skeletal muscle of older adults. Am. J. Physiol Regul Integr Comp. Physiol. 2013, 305, R216–R223. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.R.; Fink, L.; Maris, J.; Ferraro, N.; Power-Vanwart, J.; Eleff, S.; Chance, B. Evaluation of energy metabolism in skeletal muscle of patients with heart failure with gated phosphorus-31 nuclear magnetic resonance. Circulation 1985, 71, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Schaufelberger, M.; Eriksson, B.O.; Grimby, G.; Held, P.; Swedberg, K. Skeletal muscle fiber composition and capillarization in patients with chronic heart failure: Relation to exercise capacity and central hemodynamics. J. Card. Fail. 1995, 1, 267–272. [Google Scholar] [CrossRef]
- Dos Santos, M.R.; Saitoh, M.; Ebner, N.; Valentova, M.; Konishi, M.; Ishida, J.; Emami, A.; Springer, J.; Sandek, A.; Doehner, W.; et al. Sarcopenia and Endothelial Function in Patients With Chronic Heart Failure: Results From the Studies Investigating Comorbidities Aggravating HF (SICA-HF). J. Am. Med. Dir. Assoc. 2017, 18, 240–245. [Google Scholar] [CrossRef]
- Khan, H.; Kunutsor, S.; Rauramaa, R.; Savonen, K.; Kalogeropoulos, A.P.; Georgiopoulou, V.V.; Butler, J.; Laukkanen, J.A. Cardiorespiratory fitness and risk of heart failure: A population-based follow-up study. Eur. J. Heart Fail. 2014, 16, 180–188. [Google Scholar] [CrossRef]
- Franciosa, J.A.; Park, M.; Levine, T.B. Lack of correlation between exercise capacity and indexes of resting left ventricular performance in heart failure. Am. J. Cardiol. 1981, 47, 33–39. [Google Scholar] [CrossRef]
- Calvani, R.; Marini, F.; Cesari, M.; Tosato, M.; Anker, S.D.; Von Haehling, S.; Miller, R.R.; Bernabei, R.; Landi, F.; Marzetti, E.; et al. Biomarkers for physical frailty and sarcopenia: State of the science and future developments. J. Cachexia Sarcopenia Muscle 2015, 6, 278–286. [Google Scholar] [CrossRef]
- Liguori, I.; Russo, G.; Aran, L.; Bulli, G.; Curcio, F.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; Bonaduce, D.; et al. Sarcopenia: Assessment of disease burden and strategies to improve outcomes. Clin. Interv. Aging 2018, 13, 913–927. [Google Scholar] [CrossRef]
- Coats, A.J.; Clark, A.L.; Piepoli, M.; Volterrani, M. Poole-Wilson PA Symptoms and quality of life in heart failure: The muscle hypothesis. Br. Heart J. 1994, 72, S36–S39. [Google Scholar] [CrossRef] [PubMed]
- Piepoli, M.; Clark, A.; Volterrani, M.; Adamopoulos, S.; Sleight, P.; Coats, A.J.S. Contribution of muscle afferents to the hemodynamic, autonomic and ventilatory responses to exercise in patients with chronic heart failure. Circulation 1996, 93, 940–952. [Google Scholar] [CrossRef]
- Nichols, S.; O’Doherty, A.F.; Taylor, C.; Clark, A.L.; Carroll, S.; Ingle, L. Low skeletal muscle mass is associated with low aerobic capacity and increased mortality risk in patients with coronary heart disease-a CARE CR study. Clin. Physiol. Funct. Imaging 2019, 39, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Wilson, J.R.; Mancini, D.M. Factors contributing to the exercise limitation of heart failure. J. Am. Coll. Cardiol. 1993, 22, 93A–98A. [Google Scholar] [CrossRef]
- Carbone, S.; Billingsley, H.E.; Rodriguez-Miguelez, P.; Kirkman, D.L.; Garten, R.; Franco, R.L.; Lee, D.C.; Lavie, C.J. Lean Mass Abnormalities in Heart Failure: The Role of Sarcopenia, Sarcopenic Obesity, and Cachexia. Curr. Probl. Cardiol. 2019. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, I.; Minamino, T. Physiological and pathological cardiac hypertrophy. J. Mol. Cell. Cardiol. 2016, 97, 245–262. [Google Scholar] [CrossRef] [PubMed]
- Bekfani, T.; Pellicori, P.; Morris, D.A.; Ebner, N.; Valentova, M.; Steinbeck, L.; Wachter, R.; Elsner, S.; Sliziuk, V.; Schefold, J.C.; et al. Sarcopenia in patients with heart failure with preserved ejection fraction: Impact on muscle strength, exercise capacity and quality of life. Int. J. Cardiol. 2016, 222, 41–46. [Google Scholar] [CrossRef]
- Haykowsky, M.J.; Brubaker, P.H.; John, J.M.; Stewart, K.P.; Morgan, T.M.; Kitzman, D.W. Determinants of exercise intolerance in elderly heart failure patients with preserved ejection fraction. J. Am. Coll. Cardiol. 2011, 58, 265–274. [Google Scholar] [CrossRef]
- Haykowsky, M.J.; Tomczak, C.R.; Scott, J.M.; Paterson, D.I.; Kitzman, D.W. Determinants of exercise intolerance in patients with heart failure and reduced or preserved ejection fraction. J. Appl. Physiol. 2015, 119, 739–744. [Google Scholar] [CrossRef]
- Testa, G.; Cacciatore, F.; Galizia, G.; Della-Morte, D.; Mazzella, F.; Langellotto, A.; Russo, S.; Gargiulo, G.; De Santis, D.; Ferrara, N.; et al. Waist circumference but not body mass index predicts long-term mortality in elderly subjects with chronic heart failure. J. Am. Geriatr. Soc. 2010, 58, 1433–1440. [Google Scholar] [CrossRef]
- Bianchi, L.; Abete, P.; Bellelli, G.; Bo, M.; Cherubini, A.; Corica, F.; Di Bari, M.; Maggio, M.; Manca, G.M.; Rizzo, M.R.; et al. Prevalence and Clinical Correlates of Sarcopenia, Identified According to the EWGSOP Definition and Diagnostic Algorithm, in Hospitalized Older People: The GLISTEN Study. J. Gerontol. A Biol. Sci. Med. Sci. 2017, 72, 1575–1581. [Google Scholar] [CrossRef]
- Beyer, S.E.; Sanghvi, M.M.; Aung, N.; Hosking, A.; Cooper, J.A.; Paiva, J.M.; Lee, A.M.; Fung, K.; Lukaschuk, E.; Carapella, V.; et al. Prospective association between handgrip strength and cardiac structure and function in UK adults. PLoS ONE 2018, 13, e0193124. [Google Scholar] [CrossRef]
- Sartiani, L.; Spinelli, V.; Laurino, A.; Blescia, S.; Raimondi, L.; Cerbai, E.; Mugelli, A. Pharmacological perspectives in sarcopenia: A potential role for renin-angiotensin system blockers? Clin. Cases Miner. Bone Metab. 2015, 12, 135–138. [Google Scholar] [CrossRef]
- Carter, C.S.; Giovannini, S.; Seo, D.O.; DuPree, J.; Morgan, D.; Chung, H.Y.; Lees, H.; Daniels, M.; Hubbard, G.B.; Lee, S.; et al. Differential effects of enalapril and losartan on body composition and indices of muscle quality in aged male Fischer 344 × Brown Norway rats. Age 2011, 33, 167–183. [Google Scholar] [CrossRef] [PubMed]
- Burks, T.N.; Andres-Mateos, E.; Marx, R.; Mejias, R.; Van Erp, C.; Simmers, J.L.; Walston, J.D.; Ward, C.W.; Cohn, R.D. Losartan restores skeletal muscle remodeling and protects against disuse atrophy in sarcopenia. Sci. Transl. Med. 2011, 3, 82ra37. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.S.; Xu, L.J.; Wang, X.Q.; Huang, Y.H.; Xiao, Q. Effect of Angiotensin-Converting Enzyme Inhibitors on Physical Function in Elderly Subjects: A Systematic Review and Meta-Analysis. Drugs Aging 2015, 32, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Lainscak, M.; Keber, I.; Anker, S.D. Body composition changes in patients with systolic heart failure treated with beta blockers: A pilot study. Int. J. Cardiol. 2006, 106, 319–322. [Google Scholar] [CrossRef] [PubMed]
- Hryniewicz, K.; Androne, A.S.; Hudaihed, A.; Katz, S.D. Partial reversal of cachexia by beta-adrenergic receptor blocker therapy in patients with chronic heart failure. J. Card. Fail. 2003, 9, 464–468. [Google Scholar] [CrossRef]
- Burton, L.A.; Mcmurdo, M.E.; Struthers, A.D. Mineralocorticoid antagonism: A novel way to treat sarcopenia and physical impairment in older people? Clin. Endocrinol. 2011, 75, 725–729. [Google Scholar] [CrossRef]
- Cunha, T.F.; Bacurau, A.V.; Moreira, J.B.; Paixão, N.A.; Campos, J.C.; Ferreira, J.C.; Leal, M.L.; Negrão, C.E.; Moriscot, A.S.; Wisløff, U.; et al. Exercise training prevents oxidative stress and ubiquitin–proteasome system overactivity and reverse skeletal muscle atro-phy in heart failure. PLoS ONE 2012, 7, e41701. [Google Scholar] [CrossRef]
- Smart, N.A.; Steele, M. The effect of physical training on systemic proinflammatory cytokine expression in heart failure patients: A systematic review. Congest. Heart Fail. 2011, 17, 110–114. [Google Scholar] [CrossRef] [PubMed]
- Lenk, K.; Erbs, S.; Höllriegel, R.; Beck, E.; Linke, A.; Gielen, S.; Winkler, S.M.; Sandri, M.; Hambrecht, R.; Schuler, G.; et al. Exercise training leads to a reduction of elevated myostatin levels in patients with chronic heart failure. Eur. J. Prev. Cardiol. 2012, 19, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Murphy, C.H.; Oikawa, S.Y.; Phillips, S.M. Dietary protein to maintain muscle mass in aging: A case for per-meal protein recommendations. J. Frailty Aging 2016, 5, 49–58. [Google Scholar] [PubMed]
- Holeček, M. Beta-hydroxy-beta-methylbutyrate supplementation and skeletal muscle in healthy and muscle-wasting conditions. J. Cachexia Sarcopenia Muscle 2017, 8, 529–541. [Google Scholar] [CrossRef] [PubMed]
- Deutz, N.E.; Matheson, E.M.; Matarese, L.E.; Luo, M.; Baggs, G.E.; Nelson, J.L.; Hegazi, R.A.; Tappenden, K.A.; Ziegler, T.R.; NOURISH Study Group. Readmission and mortality in malnourished, older, hospitalized adults treated with a specialized oral nutritional supplement: A randomized clinical trial. Clin. Nutr. 2016, 35, 18–26. [Google Scholar] [CrossRef]
- Boxer, R.S.; Dauser, D.A.; Walsh, S.J.; Hager, W.D.; Kenny, A.M. The association between vitamin D and inflammation with the 6-minute walk and frailty in patients with heart failure. J. Am. Geriatr. Soc. 2008, 56, 454–461. [Google Scholar] [CrossRef]
- Witham, M.D. Vitamin D in chronic heart failure. Curr. Heart Fail. Rep. 2011, 8, 123–130. [Google Scholar] [CrossRef]
- Saitoh, M.; Ebner, N.; von Haehling, S.; Anker, S.D.; Springer, J. Therapeutic considerations of sarcopenia in heart failure patients. Expert Rev. Cardiovasc. Ther. 2018, 16, 133–142. [Google Scholar] [CrossRef] [PubMed]
- Martone, A.M.; Marzetti, E.; Calvani, R.; Picca, A.; Tosato, M.; Santoro, L.; Di Giorgio, A.; Nesci, A.; Sisto, A.; Santoliquido, A.; et al. Exercise and Protein Intake: ASynergistic Approach against Sarcopenia. Biomed Res Int. 2017, 2017, 2672435. [Google Scholar] [CrossRef]
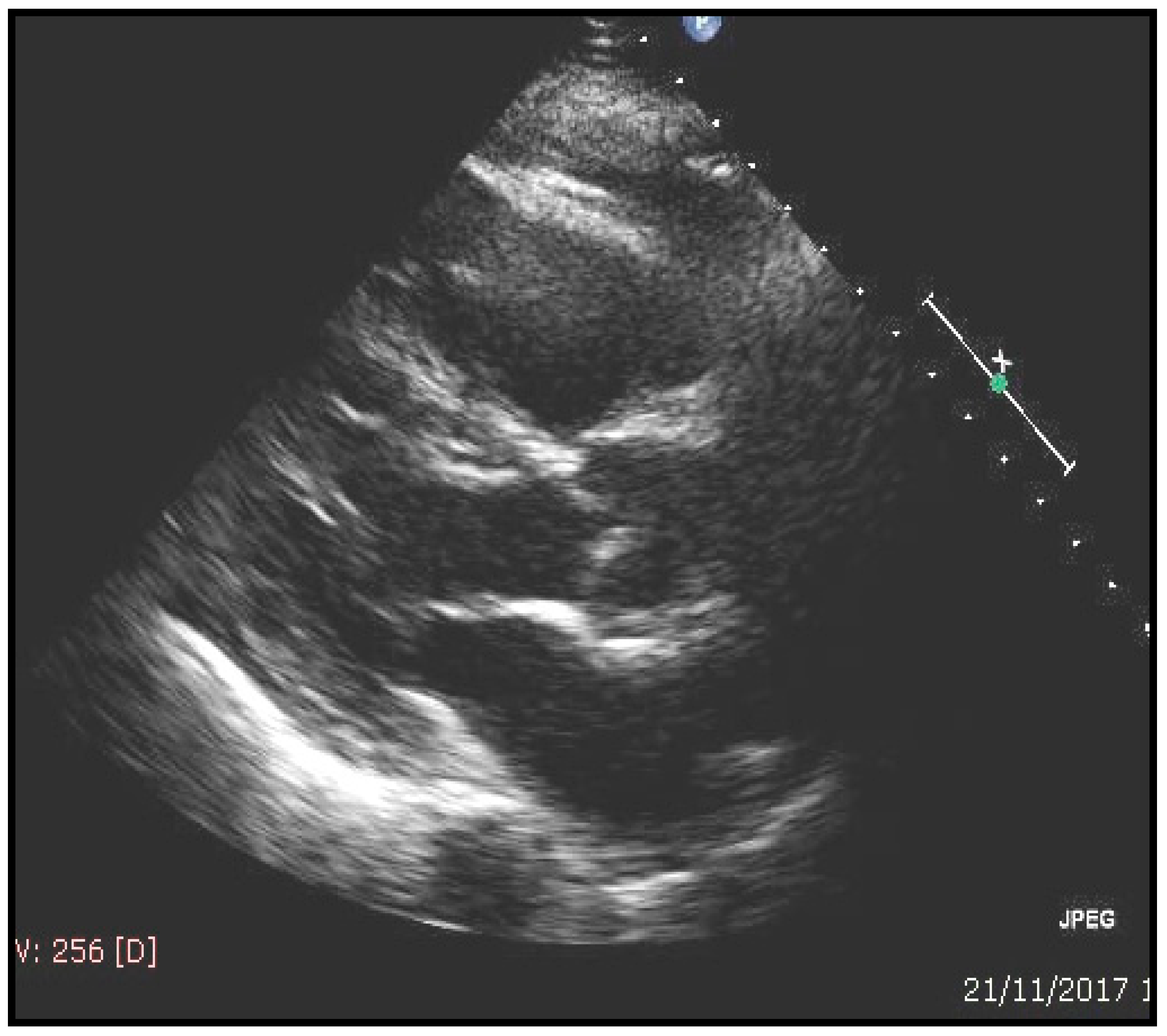
| SARCOPENIA Evaluation | Value | Normal Range |
| Handgrip strength, kg | 20.6 | <27 kg |
| Muscle mass, kg/m2 | 6.2 | <7 |
| Short Performance Physical Battery | 5 | <5 |
| ECHOCARDIOGRAFIC evaluation | Value | normal range |
| Septal thickness, mm | 13.8 | 0.6–0.9 |
| Posterior wall thickness, mm | 12.9 | 0.6–0.9 |
| LV end-diastolic diameter, mm | 45.5 | 39–53 |
| LV mass/Body surface area, g/m2 | 120.4 | 44–88 |
| E/e1 | 13 | <10 |
| Ejection fraction (%) | 49.5 | ≥55 |
| Atrial volume/Body surface area, mL/m2 | 46.4 | 16–28 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Curcio, F.; Testa, G.; Liguori, I.; Papillo, M.; Flocco, V.; Panicara, V.; Galizia, G.; Della-Morte, D.; Gargiulo, G.; Cacciatore, F.; et al. Sarcopenia and Heart Failure. Nutrients 2020, 12, 211. https://doi.org/10.3390/nu12010211
Curcio F, Testa G, Liguori I, Papillo M, Flocco V, Panicara V, Galizia G, Della-Morte D, Gargiulo G, Cacciatore F, et al. Sarcopenia and Heart Failure. Nutrients. 2020; 12(1):211. https://doi.org/10.3390/nu12010211
Chicago/Turabian StyleCurcio, Francesco, Gianluca Testa, Ilaria Liguori, Martina Papillo, Veronica Flocco, Veronica Panicara, Gianluigi Galizia, David Della-Morte, Gaetano Gargiulo, Francesco Cacciatore, and et al. 2020. "Sarcopenia and Heart Failure" Nutrients 12, no. 1: 211. https://doi.org/10.3390/nu12010211
APA StyleCurcio, F., Testa, G., Liguori, I., Papillo, M., Flocco, V., Panicara, V., Galizia, G., Della-Morte, D., Gargiulo, G., Cacciatore, F., Bonaduce, D., Landi, F., & Abete, P. (2020). Sarcopenia and Heart Failure. Nutrients, 12(1), 211. https://doi.org/10.3390/nu12010211




