Estimation of the Minimum Effective Dose of Dietary Supplement Crocetin for Prevention of Myopia Progression in Mice
Abstract
1. Introduction
2. Materials and Methods
2.1. Animals
2.2. In Vivo Analysis with the Dietary Factor in the Murine Model of Lens-Induced Myopia
2.3. Ocular Components Measurement
2.4. Crocetin Concentration in the Eyeballs and Plasma
2.4.1. Sample Preparation
2.4.2. Chemicals and Reagents
2.4.3. Preparation of Calibration Standards and Quality Control (QC) Samples
2.4.4. Sample Preparation Procedure
2.4.5. LC-MS/MS Conditions
2.5. Statistical Analyses
3. Results
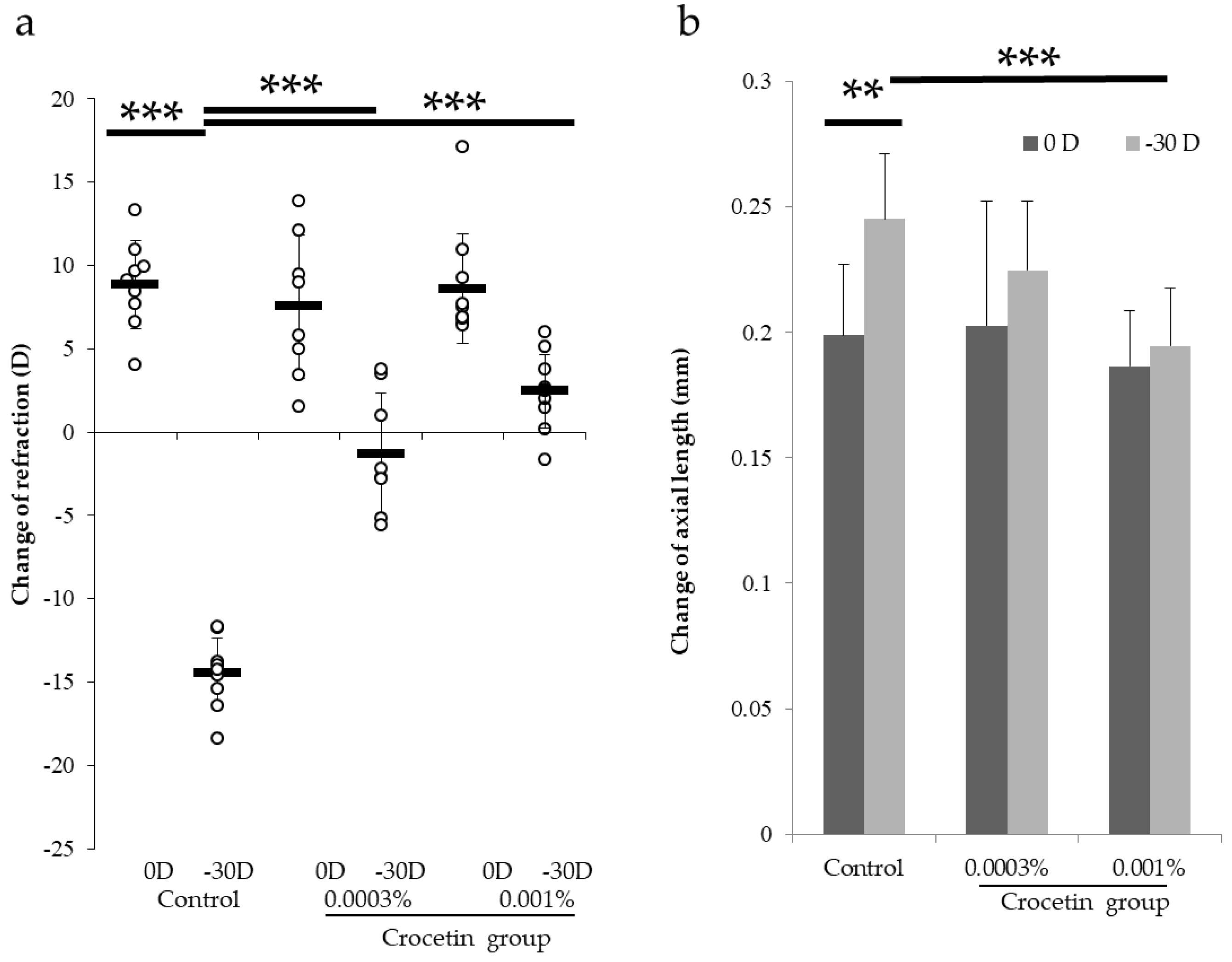
3.1. Determination of Minimum Effective Dosage with a Murine Model of Lens-Induced Myopia
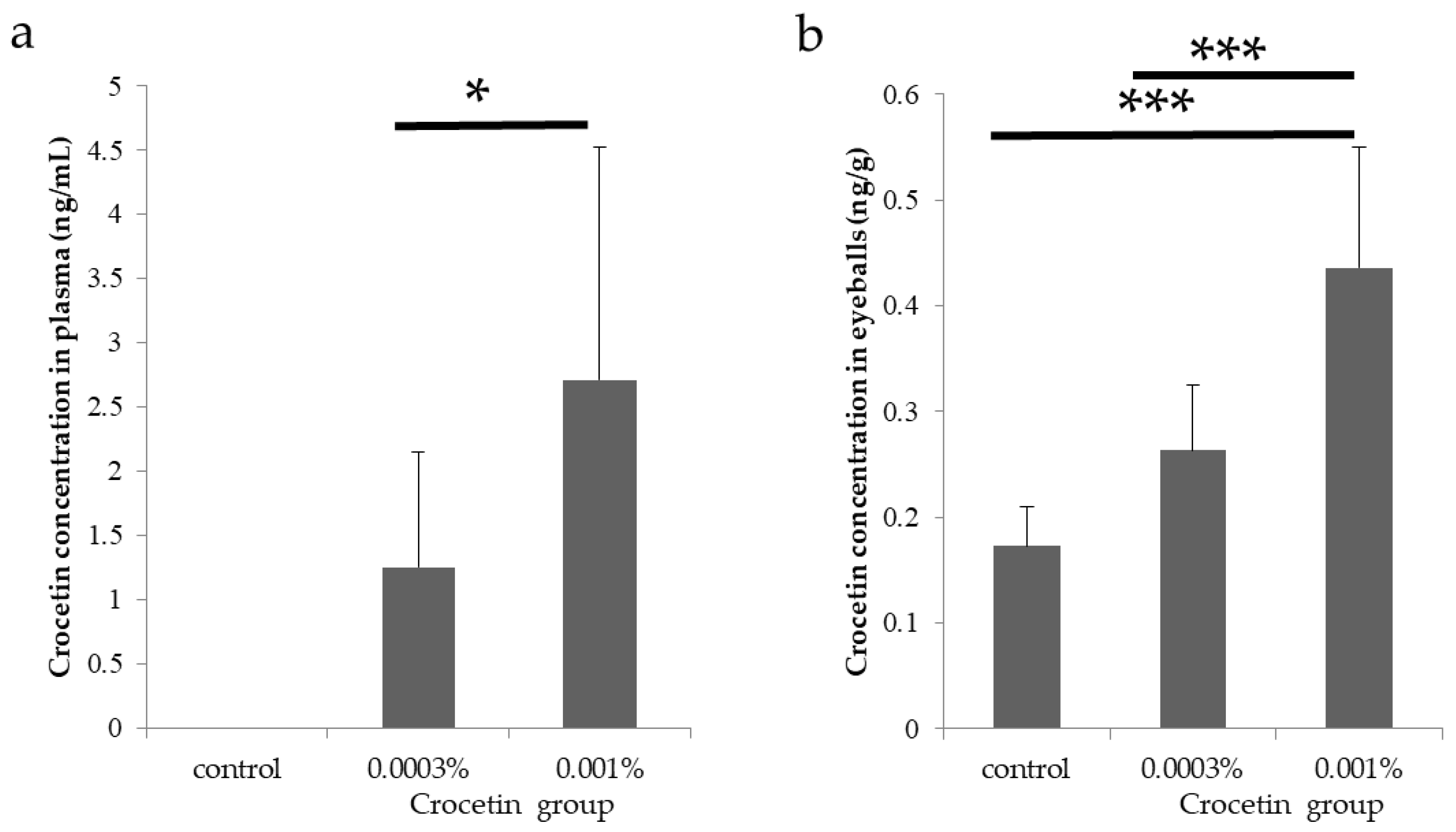
3.2. Crocetin Concentrations in the Plasma and Eyeballs in Mice
4. Discussion
5. Patents
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Holden, B.A.; Fricke, T.R.; Wilson, D.A.; Jong, M.; Naidoo, K.S.; Sankaridurg, P.; Wong, T.Y.; Naduvilath, T.J.; Resnikoff, S. Global Prevalence of Myopia and High Myopia and Temporal Trends from 2000 through 2050. Ophthalmology 2016, 123, 1036–1042. [Google Scholar] [CrossRef] [PubMed]
- Smith, T.S.; Frick, K.D.; Holden, B.A.; Fricke, T.R.; Naidoo, K.S. Potential lost productivity resulting from the global burden of uncorrected refractive error. Bull. World Health Organ. 2009, 87, 431–437. [Google Scholar] [CrossRef] [PubMed]
- Gwiazda, J.; Hyman, L.; Dong, L.M.; Everett, D.; Norton, T.; Kurtz, D.; Manny, R.; Marsh-Tootle, W.; Scheiman, M. Factors associated with high myopia after 7 years of follow-up in the Correction of Myopia Evaluation Trial (COMET) Cohort. Ophthalmic Epidemiol. 2007, 14, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.C.; Chuang, M.N.; Choi, J.; Chen, H.; Wu, G.; Ohno-Matsui, K.; Jonas, J.B.; Cheung, C.M.G. Update in myopia and treatment strategy of atropine use in myopia control. Eye 2019, 33, 3–13. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Wen, D.; Wang, Q.; McAlinden, C.; Flitcroft, I.; Chen, H.; Saw, S.M.; Chen, H.; Bao, F.; Zhao, Y.; et al. Efficacy Comparison of 16 Interventions for Myopia Control in Children: A Network Meta-analysis. Ophthalmology 2016, 123, 697–708. [Google Scholar] [CrossRef] [PubMed]
- Read, S.A.; Collins, M.J.; Vincent, S.J. Light exposure and physical activity in myopic and emmetropic children. Optom. Vsi. Sci. Off. Publ. Am. Acad. Optom. 2014, 91, 330–341. [Google Scholar] [CrossRef] [PubMed]
- Rose, K.A.; Morgan, I.G.; Smith, W.; Burlutsky, G.; Mitchell, P.; Saw, S.M. Myopia, lifestyle, and schooling in students of Chinese ethnicity in Singapore and Sydney. Arch. Ophthalmol. 2008, 126, 527–530. [Google Scholar] [CrossRef] [PubMed]
- Wu, P.C.; Chen, C.T.; Lin, K.K.; Sun, C.C.; Kuo, C.N.; Huang, H.M.; Poon, Y.C.; Yang, M.L.; Chen, C.Y.; Huang, J.C.; et al. Myopia Prevention and Outdoor Light Intensity in a School-Based Cluster Randomized Trial. Ophthalmology 2018, 125, 1239–1250. [Google Scholar] [CrossRef] [PubMed]
- Torii, H.; Kurihara, T.; Seko, Y.; Negishi, K.; Ohnuma, K.; Inaba, T.; Kawashima, M.; Jiang, X.; Kondo, S.; Miyauchi, M.; et al. Violet Light Exposure Can Be a Preventive Strategy Against Myopia Progression. EBioMedicine 2017, 15, 210–219. [Google Scholar] [CrossRef] [PubMed]
- Torii, H.; Ohnuma, K.; Kurihara, T.; Tsubota, K.; Negishi, K. Violet Light Transmission is Related to Myopia Progression in Adult High Myopia. Sci. Rep. 2017, 7, 14523. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Schaeffel, F.; Jiang, B.; Feldkaemper, M. Effects of Light of Different Spectral Composition on Refractive Development and Retinal Dopamine in Chicks. Investig. Ophthalmol. Vis. Sci. 2018, 59, 4413–4424. [Google Scholar] [CrossRef] [PubMed]
- Mori, K.; Kurihara, T.; Miyauchi, M.; Ishida, A.; Jiang, X.; Ikeda, S.I.; Torii, H.; Tsubota, K. Oral crocetin administration suppressed refractive shift and axial elongation in a murine model of lens-induced myopia. Sci. Rep. 2019, 9, 295. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Kurihara, T.; Kunimi, H.; Miyauchi, M.; Ikeda, S.I.; Mori, K.; Tsubota, K.; Torii, H.; Tsubota, K. A highly efficient murine model of experimental myopia. Sci. Rep. 2018, 8, 2026. [Google Scholar] [CrossRef] [PubMed]
- Umigai, N.; Murakami, K.; Ulit, M.V.; Antonio, L.S.; Shirotori, M.; Morikawa, H.; Nakano, T. The pharmacokinetic profile of crocetin in healthy adult human volunteers after a single oral administration. Phytomed. Int. J. Phytother. Phytopharmacol. 2011, 18, 575–578. [Google Scholar] [CrossRef] [PubMed]
- Park, H.; Qazi, Y.; Tan, C.; Jabbar, S.B.; Cao, Y.; Schmid, G.; Pardue, M.T. Assessment of axial length measurements in mouse eyes. Optom. Vsi. Sci. Off. Publ. Am. Acad. Optom. 2012, 89, 296–303. [Google Scholar] [CrossRef] [PubMed]
- Bhardwaj, V.; Rajeshbhai, G.P. Axial length, anterior chamber depth-a study in different age groups and refractive errors. J. Clin. Diagn. Res. JCDR 2013, 7, 2211–2212. [Google Scholar] [CrossRef] [PubMed]
- Bowmaker, J.K.; Astell, S.; Hunt, D.M.; Mollon, J.D. Photosensitive and photostable pigments in the retinae of Old World monkeys. J. Exp. Biol. 1991, 156, 1–19. [Google Scholar] [PubMed]
| Type of Food | Concentration of Crocetin | |||||||
|---|---|---|---|---|---|---|---|---|
| Plasma (ng/mL) | p Value | Eyeballs (ng/g) | p Value | |||||
| Control | NC | 0.17 | ± | 0.04 | ||||
| 0.0003% crocetin | 1.25 | ± | 0.89 | 0.26 | ± | 0.06 | ||
| 0.001% crocetin | 2.71 | ± | 1.81 | 0.44 | ± | 0.11 | ||
| 0.043 † | <0.001 †† | |||||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mori, K.; Kurihara, T.; Jiang, X.; Ikeda, S.-i.; Yotsukura, E.; Torii, H.; Tsubota, K. Estimation of the Minimum Effective Dose of Dietary Supplement Crocetin for Prevention of Myopia Progression in Mice. Nutrients 2020, 12, 180. https://doi.org/10.3390/nu12010180
Mori K, Kurihara T, Jiang X, Ikeda S-i, Yotsukura E, Torii H, Tsubota K. Estimation of the Minimum Effective Dose of Dietary Supplement Crocetin for Prevention of Myopia Progression in Mice. Nutrients. 2020; 12(1):180. https://doi.org/10.3390/nu12010180
Chicago/Turabian StyleMori, Kiwako, Toshihide Kurihara, Xiaoyan Jiang, Shin-ichi Ikeda, Erisa Yotsukura, Hidemasa Torii, and Kazuo Tsubota. 2020. "Estimation of the Minimum Effective Dose of Dietary Supplement Crocetin for Prevention of Myopia Progression in Mice" Nutrients 12, no. 1: 180. https://doi.org/10.3390/nu12010180
APA StyleMori, K., Kurihara, T., Jiang, X., Ikeda, S.-i., Yotsukura, E., Torii, H., & Tsubota, K. (2020). Estimation of the Minimum Effective Dose of Dietary Supplement Crocetin for Prevention of Myopia Progression in Mice. Nutrients, 12(1), 180. https://doi.org/10.3390/nu12010180







