Gastroesophageal Reflux Disease and Probiotics: A Systematic Review
Abstract
1. Introduction
1.1. Definition
1.2. Epidemiology
1.3. Pathophysiology
1.4. Management and Treatment
1.5. Probiotics and GERD
1.6. Aim of the Study
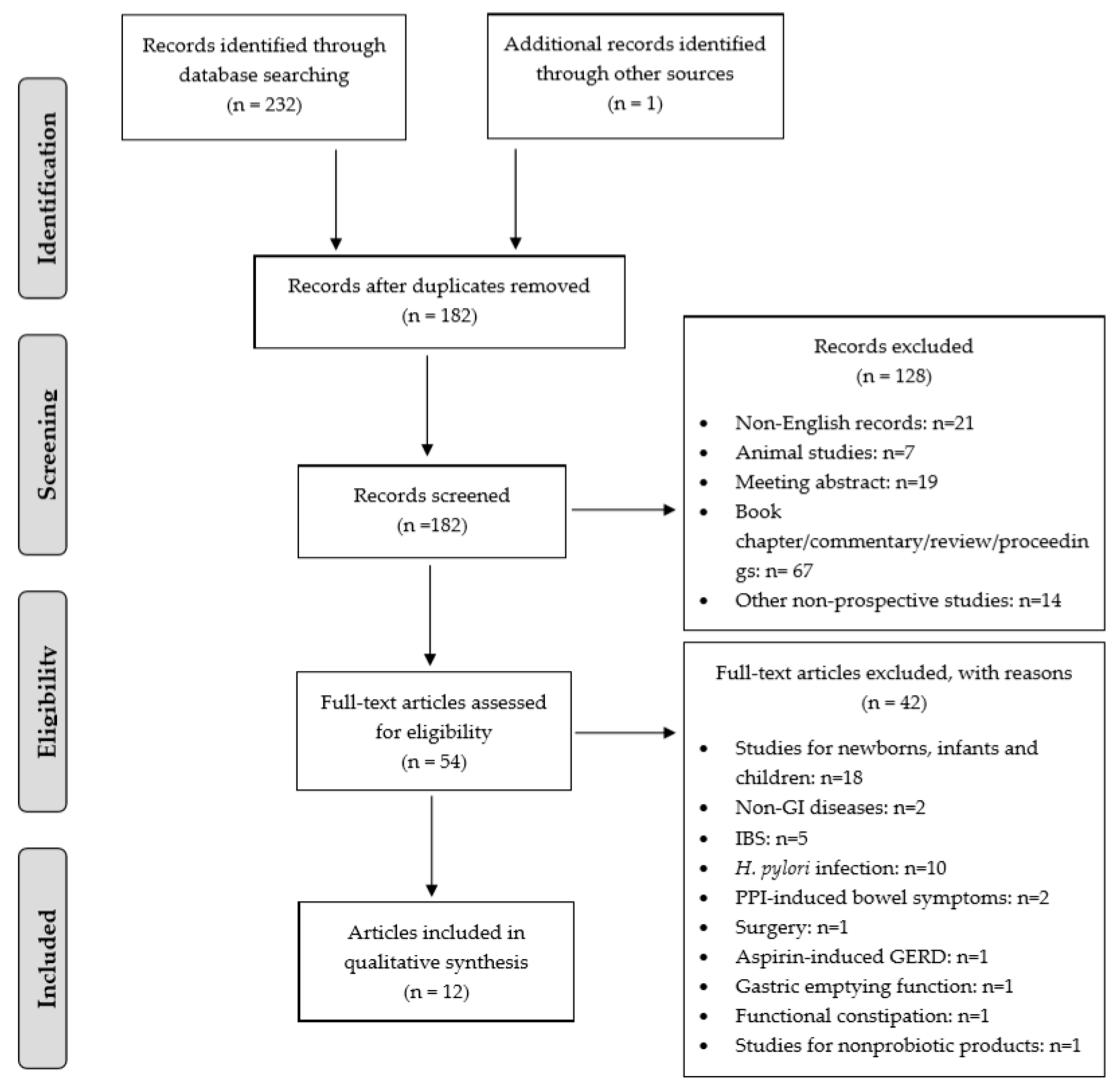
2. Method
2.1. Literature Search
2.2. Study Selection and Data Extraction
3. Results
4. Discussion
4.1. Clinical Efficacy and Potential Mechanisms
4.2. Effects of Product Format on Efficacy
4.3. Safety
4.4. Study Quality
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hunt, R.; Armstrong, D.; Katelaris, P.H.; Afihene, M.; Bane, A.; Bhatia, S.; Chen, M.H.; Choi, M.G.; Melo, A.C.; Fock, K.M.; et al. Global perspective on gastroesophageal reflux disease. World Gastroenterology Organisation Global Guidelines, 2015; pp. 1–37. Available online: https://www.spg.pt/wp-content/uploads/2015/07/2015-Gastroesophageal-Reflux-Disease-GERD.pdf (accessed on 2 January 2020).
- Schmulson, M.J.; Drossman, D.A. What Is New in Rome IV. J. Neurogastroenterol. Motil. 2017, 23, 151–163. [Google Scholar] [CrossRef]
- Benninga, M.A.; Faure, C.; Hyman, P.E.; St James Roberts, I.; Schechter, N.L.; Nurko, S. Childhood Functional Gastrointestinal Disorders: Neonate/Toddler. Gastroenterology 2016, 150, 1443–1455. [Google Scholar] [CrossRef] [PubMed]
- Zeevenhooven, J.; Koppen, I.J.; Benninga, M.A. The New Rome IV Criteria for Functional Gastrointestinal Disorders in Infants and Toddlers. Pediatr. Gastroenterol. Hepatol. Nutr. 2017, 20, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Gyawali, C.P.; Kahrilas, P.J.; Savarino, E.; Zerbib, F.; Mion, F.; Smout, A.; Vaezi, M.; Sifrim, D.; Fox, M.R.; Vela, M.F.; et al. Modern diagnosis of GERD: The Lyon Consensus. Gut 2018, 67, 1351–1362. [Google Scholar] [CrossRef]
- Revicki, D.A.; Wood, M.; Wiklund, I.; Crawley, J. Reliability and validity of the Gastrointestinal Symptom Rating Scale in patients with gastroesophageal reflux disease. Qual. Life Res. 1998, 7, 75–83. [Google Scholar] [CrossRef]
- Kusano, M.; Shimoyama, Y.; Sugimoto, S.; Kawamura, O.; Maeda, M.; Minashi, K.; Kuribayashi, S.; Higuchi, T.; Zai, H.; Ino, K.; et al. Development and evaluation of FSSG: Frequency scale for the symptoms of GERD. J. Gastroenterol. 2004, 39, 888–891. [Google Scholar] [CrossRef]
- El-Serag, H.B.; Sweet, S.; Winchester, C.C.; Dent, J. Update on the epidemiology of gastro-oesophageal reflux disease: A systematic review. Gut 2014, 63, 871–880. [Google Scholar] [CrossRef]
- Chatila, A.T.; Nguyen, M.T.T.; Krill, T.; Roark, R.; Bilal, M.; Reep, G. Natural history, pathophysiology and evaluation of gastroesophageal reflux disease. Dis. Mon. 2019, 22, 100848. [Google Scholar] [CrossRef]
- Savarino, E.; Bredenoord, A.J.; Fox, M.; Pandolfino, J.E.; Roman, S.; Gyawali, C.P. International Working Group for Disorders of Gastrointestinal Motility and Function. Advances in the physiological assessment and diagnosis of GERD. Nat. Rev. Gastroenterol. Hepatol. 2018, 15, 323. [Google Scholar] [CrossRef]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Sanders, M.E.; Heimbach, J.T.; Pot, B.; Tancredi, D.J.; Lenoir-Wijnkoop, I.; Lähteenmäki-Uutela, A.; Gueimonde, M.; Bañares, S. Health claims substantiation for probiotic and prebiotic products. Gut Microbes 2011, 2, 127–133. [Google Scholar] [CrossRef] [PubMed]
- Misra, S.; Mohanty, D.; Mohapatra, S. Applications of Probiotics as a Functional Ingredient in Food and Gut Health. J. Food Nutr. Res. 2019, 7, 213–223. [Google Scholar]
- Hungin, A.P.S.; Mitchell, C.R.; Whorwell, P.; Mulligan, C.; Cole, O.; Agreus, L.; Fracasso, P.; Lionis, C.; Mendive, J.; de Philippart Foy, J.M.; et al. Systematic review: Probiotics in the management of lower gastrointestinal symptoms—An updated evidence-based international consensus. Aliment Pharmacol. Ther. 2018, 47, 1054–1070. [Google Scholar] [CrossRef]
- Kleerebezem, M.; Binda, S.; Bron, P.A.; Gross, G.; Hill, C.; van Hylckama Vlieg, J.E.; Lebeer, S.; Satokari, R.; Ouwehand, A.C. Understanding mode of action can drive the translational pipeline towards more reliable health benefits for probiotics. Curr. Opin. Biotechnol. 2019, 56, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Kristensen, N.B.; Bryrup, T.; Allin, K.H.; Nielsen, T.; Hansen, T.H.; Pedersen, O. Alterations in fecal microbiota composition by probiotic supplementation in healthy adults: A systematic review of randomized controlled trials. Genome Med. 2016, 8, 52. [Google Scholar] [CrossRef]
- Grimm, V.; Riedel, C.U. Manipulation of the Microbiota Using Probiotics. Adv. Exp. Med. Biol. 2016, 902, 109–117. [Google Scholar]
- Azad, M.A.K.; Sarker, M.; Li, T.; Yin, J. Probiotic Species in the Modulation of Gut Microbiota: An Overview. BioMed Res. Int. 2018. [Google Scholar] [CrossRef]
- Keita, A.V.; Soderholm, J.D. Mucosal permeability and mast cells as targets for functional gastrointestinal disorders. Curr. Opin. Pharm. 2018, 43, 66–71. [Google Scholar] [CrossRef] [PubMed]
- Aiba, Y.; Nakano, Y.; Koga, Y.; Takahashi, K.; Komatsu, Y. A highly acid-resistant novel strain of Lactobacillus johnsonii No. 1088 has antibacterial activity, including that against Helicobacter pylori, and inhibits gastrin-mediated acid production in mice. Microbiologyopen 2015, 4, 465–474. [Google Scholar] [CrossRef]
- Urita, Y.; Goto, M.; Watanabe, T.; Matsuzaki, M.; Gomi, A.; Kano, M.; Miyazaki, K.; Kaneko, H. Continuous consumption of fermented milk containing Bifidobacterium bifidum YIT 10347 improves gastrointestinal and psychological symptoms in patients with functional gastrointestinal disorders. Biosci. Microb. Food H 2015, 34, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Igarashi, M.; Nagano, J.; Tsuda, A.; Suzuki, T.; Koike, J.; Uchida, T.; Matsushima, M.; Mine, T.; Koga, Y. Correlation between the Serum Pepsinogen I Level and the Symptom Degree in Proton Pump Inhibitor-Users Administered with a Probiotic. Pharmaceuticals 2014, 7, 754–764. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.H.; Wang, H.Y.; Sun, S.D.; Zhang, X.; Zhang, H. Beneficial effect of probiotics supplements in reflux esophagitis treated with esomeprazole: A randomized controlled trial. World J. Gastroenterol. 2019, 25, 2110–2121. [Google Scholar] [CrossRef]
- Jadad, A.R.; Moore, R.A.; Carroll, D.; Jenkinson, C.; Reynolds, D.J.M.; Gavaghan, D.J.; McQuay, H.J. Assessing the quality of reports of randomized clinical trials: Is blinding necessary? Control. Clin. Trials 1996, 17, 1–12. [Google Scholar] [CrossRef]
- Gomi, A.; Iino, T.; Nonaka, C.; Miyazaki, K.; Ishikawa, F. Health benefits of fermented milk containing Bifidobacterium bifidum YIT 10347 on gastric symptoms in adults. J. Dairy Sci. 2015, 98, 2277–2283. [Google Scholar] [CrossRef] [PubMed]
- Waller, P.A.; Gopal, P.K.; Leyer, G.J.; Ouwehand, A.C.; Reifer, C.; Stewart, M.E.; Miller, L.E. Dose-response effect of Bifidobacterium lactis HN019 on whole gut transit time and functional gastrointestinal symptoms in adults. Scand. J. Gastroenterol. 2011, 46, 1057–1064. [Google Scholar] [CrossRef] [PubMed]
- De Milliano, I.; Tabbers, M.M.; van der Post, J.A.; Benninga, M.A. Is a multispecies probiotic mixture effective in constipation during pregnancy? ‘A pilot study’. Nutr. J. 2012, 11, 80. [Google Scholar] [CrossRef]
- Ianiro, G.; Pizzoferrato, M.; Franceschi, F.; Tarullo, A.; Luisi, T.; Gasbarrini, G. Effect of an extra-virgin olive oil enriched with probiotics or antioxidants on functional dyspepsia: A pilot study. Eur. Rev. Med. Pharm. Sci. 2013, 17, 2085–2090. [Google Scholar]
- Nakae, H.; Tsuda, A.; Matsuoka, T.; Mine, T.; Koga, Y. Gastric microbiota in the functional dyspepsia patients treated with probiotic yogurt. BMJ. Open Gastroenterol. 2016, 3, e000109. [Google Scholar] [CrossRef]
- Ohtsu, T.; Takagi, A.; Uemura, N.; Inoue, K.; Sekino, H.; Kawashima, A.; Uchida, M.; Koga, Y. The Ameliorating Effect of Lactobacillus gasseri OLL2716 on Functional Dyspepsia in Helicobacter pylori-Uninfected Individuals: A Randomized Controlled Study. Digestion 2017, 96, 92–102. [Google Scholar] [CrossRef]
- Gomi, A.; Yamaji, K.; Watanabe, O.; Yoshioka, M.; Miyazaki, K.; Iwama, Y.; Urita, Y. Bifidobacterium bifidum YIT 10347 fermented milk exerts beneficial effects on gastrointestinal discomfort and symptoms in healthy adults: A double-blind, randomized, placebo-controlled study. J. Dairy Sci. 2018, 101, 4830–4841. [Google Scholar] [CrossRef] [PubMed]
- Östlund-Lagerström, L.; Kihlgren, A.; Repsilber, D.; Bjorksten, B.; Brummer, R.J.; Schoultz, I. Probiotic administration among free-living older adults: A double blinded, randomized, placebo-controlled clinical trial. Nutr. J. 2016, 15, 80. [Google Scholar] [CrossRef] [PubMed]
- Nova, E.; Viadel, B.; Waernberg, J.; Carreres, J.E.; Marcos, A. Beneficial Effects of a Synbiotic Supplement on Self-Perceived Gastrointestinal Well-Being and Immunoinflammatory Status of Healthy Adults. J. Med. Food 2011, 14, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.L.; Hilli, L.; Orlowski, J.; Kupperman, J.L.; Baral, M.F.; Waters, R. Efficacy of probiotics and nutrients in functional gastrointestinal disorders: A preliminary clinical trial. Dig. Dis. Sci. 2006, 51, 2134–2144. [Google Scholar] [CrossRef] [PubMed]
- Indrio, F.; Riezzo, G.; Raimondi, F.; Filannino, A.; Bisceglia, M.; Cavallo, L.; Francavilla, R. Lactobacillus Reuterii accelerates gastric emptying and improves regurgitation in infants. Pediatr. Res. 2010, 68, 42. [Google Scholar] [CrossRef][Green Version]
- Quigley, E.M.; Quera, R. Small intestinal bacterial overgrowth: Roles of antibiotics, prebiotics, and probiotics. Gastroenterology 2006, 130, S78–S90. [Google Scholar] [CrossRef]
- Du, L.J.; Chen, B.R.; Kim, J.J.; Kim, S.; Shen, J.H.; Dai, N. Helicobacter pylori eradication therapy for functional dyspepsia: Systematic review and meta-analysis. World J. Gastroenterol. 2016, 22, 3486–3495. [Google Scholar] [CrossRef]
- Qureshi, N.; Li, P.; Gu, Q. Probiotic therapy in Helicobacter pylori infection: A potential strategy against a serious pathogen? Appl. Microbiol. Biotechnol. 2019, 103, 1573–1588. [Google Scholar] [CrossRef]
- Zeilstra, D.; Younes, J.A.; Brummer, R.J.; Kleerebezem, M. Perspective: Fundamental Limitations of the Randomized Controlled Trial Method in Nutritional Research: The Example of Probiotics. Adv. Nutr. 2018, 9, 561–571. [Google Scholar] [CrossRef]
- Matsubara, V.H.; Bandara, H.M.; Ishikawa, K.H.; Mayer, M.P.; Samaranayake, L.P. The role of probiotic bacteria in managing periodontal disease: A systematic review. Expert Rev. Anti-Infect. Ther. 2016, 14, 643–655. [Google Scholar] [CrossRef]
- Paludan-Muller, A.; Teindl Laursen, D.R.; Hrobjartsson, A. Mechanisms and direction of allocation bias in randomised clinical trials. BMC Med. Res. Methodol. 2016, 16, 133. [Google Scholar] [CrossRef] [PubMed]
| Study | Number of Subjects (Probiotic: Control) | Female (%) | Age (mean ± SD, Range) [I] | Delivery Vehicle | Probiotic Strain(s) [II] | Dose (109 CFU/day) | Intervention Duration (wk) |
|---|---|---|---|---|---|---|---|
| [27] | 20 (20:0) | 100 | 29.5 ± 5.3 # | NR (sachet/stick) | B. bifidum W23, B. lactis W52, B. longum W108, L. casei W79, L. plantarum W62, L. rhamnosus W71 and o FOS, inulin | 4 | 4 |
| [28] | 8 (8:8) | NR | NR but suspected to be adults | Olive oil | L. reuteri *, L. rhamnosus GG *, Saccharomyces boulardii *, and vitamin B6 hydrochloride Q10 coenzyme | 46 | 1 |
| [29] | 44 (44:0) | 50% | 42.5 (34.5–50.3) ## | Yogurt | L. gasseri LG21 | 1 | 12 |
| [30] | 106 (54:52) | 75% | 42.8 ± 9.0 | Yogurt | L. gasseri LG21 | >1 | 12 |
| [22] | 24 (24:0) | 33% | 68.6 ± 9.7 | Yogurt | L. gasseri LG21 | 1 | 12 |
| [25] A [III] | 149 (149:149) | 48% | 50.6 ± 7.4 (33–84) | Fermented milk | B. bifidum YIT 10347 | 1 | 2 |
| [25] B [III] | 27 (27:27) | 13% | 35.3 ± 11.3 (21–58) | Fermented milk | B. bifidum YIT 10347 | 1 | 2 |
| [31] | 79 (39:40) | 52% | Probiotic: 41.1 ± 10.1 Placebo: 41.6 ± 9.9 | Fermented milk | B. bifidum YIT 10347 | >3 | 4 |
| [21] | 37 (37:0) | 51% | 52.6 ± 17.5 (12–78) | Fermented milk | B. bifidum YIT 10347 | >1 | 2 |
| [26] A [IV] | 54 (26:28) | 62% | Probiotic: 44 ± 11 Placebo: 45 ± 11 | Capsule | B. lactis HN019 | 1.8 | 2 |
| [26] B [IV] | 61 (33:28) | 61% | Probiotic: 43 ± 12 Placebo: 45 ± 11 | Capsule | B. lactis HN019 | 17.2 | 2 |
| [32] | 249 (125:124) | 57% | Probiotic: 72.6 ± 5.8 Placebo: 72 ± 5.6 | Stick | L. reuteri DSM 1793 and GOS | 0.1 | 12 |
| [33] | 36 (18:18) | 56% | NR (24–45) | Tablet | L. acidophilus La5, B. lactis Bb-12, Lactobacillus bulgaricus *, Lactobacilus paracasei *, Streptococcus thermophilus *, and FOS | 2.4 | 6 |
| [34] | 24 (12:12) | 75% | Probiotic: 41.1 ± 12:5 Placebo: 41.5 ± 15.8 | Caplet | L. acidophilus *, B. bifidum *, Bacillus subtilis *, L. bulgaricus *, L. lactis *, Bacillus licheniformis * | 0.05 | 12 |
| Study | Randomization | Blinding | Design | Jadad Score [I] | Qualitative Rating [II] | IP Compliance | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| (1) | (2) | (3) | (4) | (5) | Total | ||||||
| [27] | No | No | Before-after | 0 | 0 | 0 | 0 | 1 | 1 | Low | 100% |
| [28] | No | No | Crossover | 0 | 0 | 0 | 0 | 1 | 1 | Low | NR |
| [29] | No | No | Before-after | 0 | 0 | 0 | 0 | 0 | 0 | Low | NR |
| [30] | Yes | Yes | Parallel Group | 1 | 0 | 1 | 1 | 1 | 4 | High | NR |
| [22] | No | No | Before-after | 0 | 0 | 0 | 0 | 1 | 1 | Low | >90% |
| [25] A [IV] | No | No | Before-after | 0 | 0 | 0 | 0 | 1 | 1 | Low | >95% |
| [25] B [IV] | NR | Yes | Crossover | 0 | 0 | 1 | 1 | 1 | 3 | Medium | >95% |
| [31] | Yes | Yes | Parallel Group | 1 | 0 | 1 | 1 | 1 | 4 | High | >95% |
| [21] | No | No | Before-after | 0 | 0 | 0 | 0 | 1 | 1 | Low | NR |
| [26] A [V] | Yes | Yes | Parallel Group | 1 | 1 | 1 | 1 | 0 | 4 | High | 100% |
| [26] B [V] | Yes | Yes | Parallel Group | 1 | 1 | 1 | 1 | 0 | 4 | High | 100% |
| [32] | Yes | Yes | Parallel Group | 1 | 1 | 1 | 1 | 1 | 5 | High | NR |
| [33] | Yes | Yes | Parallel Group | 1 | 0 | 1 | 1 | 0 | 3 | Medium | NR |
| [34] | Yes | Yes | Parallel Group | 1 | 1 | 1 | 1 | 1 | 5 | High | >75% |
| Study | Population | Inclusion Criteria | Side Effects/Adverse Events | Clinical Outcomes | ||
|---|---|---|---|---|---|---|
| Reflux Symptoms (Regurgitation/Acid Reflux/Heartburn) | Dyspepsia-Related Symptoms | Other upper-GI Symptoms | ||||
| [27] | Pregnant Woman | Rome III for Functional Constipation | None | Reflux episode presence reduced significantly by 40% | NA | Episodes of abdominal pain reduced significantly by 40% |
| [28] | Adult | Rome III for Functional Dyspepsia | NR | NA | Significantly reduced postprandial gastric distension and postprandial fullness compared with placebo | Compared with placebo, significantly reduced nausea and pain/discomfort in abdominal upper quadrants and relief of belching |
| [29] | Adult | Rome III for Functional Dyspepsia | NR | NR | Significantly reduced postprandial distress by 7.7 points in FSSG | Significantly reduced epigastric pain by 8 points in FSSG |
| [30] | Adult | Rome III for Functional Dyspepsia | No difference in adverse events (AEs) between probiotic (n = 2) and placebo (n = 5) | No significant reduction in regurgitation or heartburn at endpoint (week 12), but at Week 8, a significant decrease in both symptoms was observed | Significantly reduced overall FD symptom score compared with placebo (35.2 vs. 17.3%). Postprandial distress syndrome score was significantly lower versus placebo (37.5 vs. 17.8%). | A trend for the improvement in epigastric burning (p = 0.086). |
| [22] | Adult + Elderly | Patient’s medical history, upper-GI endoscopy and FSSG | NR | Frequency score of reflux reduced significantly, from 6.2 to 4.8 | Significantly increased dysmotility-like dyspepsia, from 3.5 to 4.0 on the FSSG | Overall FSSG score reduced significantly from 10.8 to 8.4. |
| [25] A [I] | Adult | Modified GSRS for gastric symptoms | None | Significantly reduced acid regurgitation, no effect on reflux | Significantly lower individual symptom scores for stomach heaviness | Compared with baseline, significantly fewer gastric symptoms by 0.8 and 1.1 and reduced overall gastric symptom score by 0.9 and 1.2 after 1 and 2 weeks, respectively. Significantly reduced individual symptom scores in burp, no appetite, and repeated abdominal pain or discomfort. |
| [25] B [I] | Adult | Modified FSSG for gastric symptoms | None | No effect on regurgitation or reflux | NR | The modified F-scale score was significantly reduced by 1.0 and 1.1 after 1 and 2 weeks compared with baseline, no comparison with placebo. |
| [31] | Adult | Modified FSSG but not Rome IV for Functional Dyspepsia | No difference between probiotic (n = 7) and placebo (n = 12). | Compared with placebo, no difference in modified FSSG for reflux syndrome. No difference in acid regurgitation but a trend for improved heartburn (−0.90 vs. −0.38) in GSRS. | On the modified FSSG, no difference in acid-related dyspepsia but trend toward postprandial discomfort (−0.56 vs. −0.33) | On the modified FSSG, a trend for improvement in burping (−0.62 vs. −0.38) and postprandial epigastric pain (−0.38 vs. −0.08). On the GSRS, trend for improved upper-GI symptoms (−0.72 vs. −0.45). No difference in severity but significantly lower prevalence of gas-related symptoms (flatus). |
| [21] | Adult | Functional gastrointestinal disorder by physician | AEs (n = 2) for intestinal gas and bloating | No difference for reflux symptoms on the FSSG, but a trend in GSRS (p = 0.06). | Significantly decreased indigestion syndrome scores on the GSRS and acid-related dyspepsia on the FSSG. | Significantly decreased overall GSRS and FSSG scores. |
| [26] A [II] | Adult | Self-reported constipation | None | Significantly lower frequency score for regurgitation by 11.3 vs. 2.3 (placebo) | NA | Significantly lower frequency score for nausea, abdominal pain, gurgling. |
| [26] B [II] | Adult | Self-reported constipation | None | Significantly lower frequency score for regurgitation by 14.9 vs. 2.3 (placebo) | NA | Significantly lower frequency score for nausea, abdominal pain, gurgling, and vomiting. |
| [32] | Elderly | GI discomfort defined by a score of >2 in any domain on the GSRS | No difference of AEs between probiotic and placebo. Serious adverse events: none | No effect on regurgitation or reflux | No effects on indigestion/dyspepsia (−0.14 vs. −0.13). | No effects on abdominal pain (−0.08 vs. −0.09). |
| [33] | Adult | Rome II for dyspepsia, postprandial bloating, constipation, flatulence | None | NA | No difference in dyspepsia | NA |
| [34] | Adult | Rome II for functional bowel disorder | None | NR | NR | A trend toward improved general GI symptoms (lower and upper GI), reduced by 18.9% vs. 8.8% with placebo. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, J.; Ouwehand, A.C. Gastroesophageal Reflux Disease and Probiotics: A Systematic Review. Nutrients 2020, 12, 132. https://doi.org/10.3390/nu12010132
Cheng J, Ouwehand AC. Gastroesophageal Reflux Disease and Probiotics: A Systematic Review. Nutrients. 2020; 12(1):132. https://doi.org/10.3390/nu12010132
Chicago/Turabian StyleCheng, Jing, and Arthur C. Ouwehand. 2020. "Gastroesophageal Reflux Disease and Probiotics: A Systematic Review" Nutrients 12, no. 1: 132. https://doi.org/10.3390/nu12010132
APA StyleCheng, J., & Ouwehand, A. C. (2020). Gastroesophageal Reflux Disease and Probiotics: A Systematic Review. Nutrients, 12(1), 132. https://doi.org/10.3390/nu12010132





