Dietary Relationship with 24 h Urinary Iodine Concentrations of Young Adults in the Mountain West Region of the United States
Abstract
1. Introduction
2. Materials and Methods
2.1. Overview of Testing
2.2. Measurement of Urinary Iodine Concentration and Iodine Status
2.3. Iodine Intake, Frequency of Iodine Containing Foods and Dietary Habits
2.4. Assessment of Thyroid Function
2.5. Statistical Analysis
3. Results
3.1. Subject Characteristics
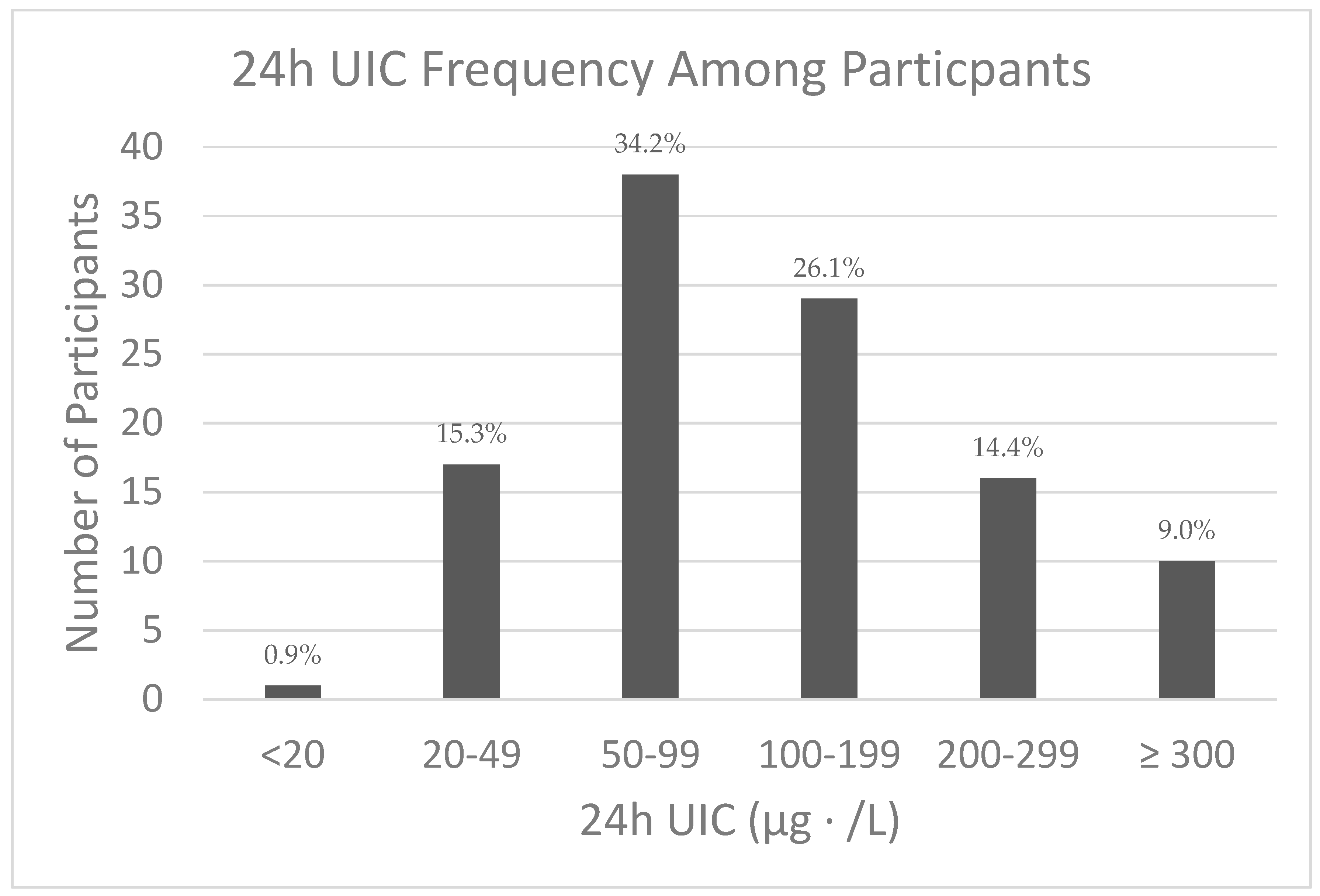
3.2. 24 h UIC and Iodine Status
3.3. Estimated Daily Iodine Intake and Frequency of Intake of Iodine-Containing Foods
3.4. Relationship between Iodine Intake and Iodine Status
3.5. Dietary Habits, Iodine Intakes and Intake Status
3.6. Relationship between Iodine Status and Thyroid Function
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
| Foods Consumed | Never or <1 per mo. | 1–3 per mo | 1 Per wk | 2–4 per wk | 5–6 per wk | 1 per day | 2–3 per day | 4–5 per day | 6+ per day |
| 1.Milk, 1 cup | |||||||||
| 2.Soy Milk, 1 cup | |||||||||
| 3.Soy Protein bars, 1 bar | |||||||||
| 4.Soy Protein powders, serving | |||||||||
| 5.Soy Sauce, 1 Tablespoon | |||||||||
| 6.Other non-dairy milks, Vit D fortified, 1 cup, | |||||||||
| 7.Orange Juice, Vitamin D fortified (Tropicana), 1cup | |||||||||
| 8.Cereal, Vitamin D fortified (Total Corn Flakes, Kelloggs Raisin Bran, Oat Bran, Cheerios), 3/4 cup | |||||||||
| 9.Bread, commercial, not homemade | |||||||||
| 10.Subway Sandwich, 6 inch | |||||||||
| 11.Bagels, 1 each | |||||||||
| 12.Yogurt, 1 cup | |||||||||
| 13.Cheese, 2 ounce | |||||||||
| 14.Egg, 1 whole | |||||||||
| 15.Margarine, Vitamin D fortified (Promise, etc.), 1 teaspoon | |||||||||
| 16.Liver, cooked, 3 ½ oz. | |||||||||
| 17.Cod, cooked, 3 ½ oz | |||||||||
| 18.Grouper cooked, 3 ½ oz | |||||||||
| 19.Haddock cooked, 3 ½ oz | |||||||||
| 20.Halibut, cooked, 3 ½ oz | |||||||||
| 21.Herring, 3 1/2 oz. | |||||||||
| 22.Mackerel, cooked, 3 ½ oz | |||||||||
| 23. Perch, cooked, 3 ½ oz | |||||||||
| 24.Salmon, cooked, 3 ½ oz | |||||||||
| 25.Sardines, canned in oil, 3 ½ oz | |||||||||
| 26.Sea Bass, cooked, 3 ½ oz | |||||||||
| 27.Swordfish, cooked, 3 ½ oz | |||||||||
| 28.Tukaoua, cooked, 3 ½ oz | |||||||||
| 29.Tuna, Albacore, canned3 ½ oz | |||||||||
| 30.Tuna, light, canned, 3 ½ oz | |||||||||
| 31.Walleye, cooked, 3 ½ oz | |||||||||
| 32.Other fish, 3 ½ oz | |||||||||
| 33.Clams, 4 oz | |||||||||
| 34.Crabmeat, 4 oz | |||||||||
| 35.Lobster, 4 oz | |||||||||
| 36.Mussels, 4 oz | |||||||||
| 37.Oysters, 4 oz | |||||||||
| 38.Scallops, 4 oz | |||||||||
| 39.Shrimp, 4 oz | |||||||||
| 40.Sea weed (Nori, Komgu, Dulse, Wakame, Kelp) | |||||||||
| 41.Iodized Table Salt | |||||||||
| 42.Other Salt |
| Supplements Consumed | Never or <1 per mo. | 1–3 per mo. | 1 per wk | 2–4 per wk | 5–6 per wk | 1 per day | 2–3 per day | 4–5 per day | 6+ per day |
| 1.Multiple Vitamin, 1 Tablet | |||||||||
| 2.Calcium Vitamin, 1 Tablet | |||||||||
| 3.Vitamin D, 1 Tablet (list amount __________) | |||||||||
| 4.Calcium + Vitamin D (Viactiv), 1 Tablet | |||||||||
| 5.Kelp (or other iodine supplement), 1 Tablet | |||||||||
| If you take any of the above, please list which you use, brand name, etc. | |||||||||
Appendix B
| How frequently do you use the salt shaker to salt your foods? | Never | Sometimes | Usually | |||
| What type of salt do you use? | ||||||
| How frequently do you use salt in your cooking? | Never | Sometimes | Usually | |||
| What type of salt do you use? | ||||||
References
- Mariotti, S.; Beck-Peccoz, P. Physiology of the Hypothalamic-Pituitary-Thyroid Axis. In Endotext; Feingold, K.R., Anawalt, B., Boyce, A., Chrousos, G., Dungan, K., Grossman, A., Hershman, J.M., Kaltsas, G., Koch, C., Kopp, P., et al., Eds.; South Dartmouth, MA, USA, 2000; Available online: https://www.endotext.org/section/thyroiddiseasemanager/ (accessed on 15 July 2019).
- Fuge, R.; Johnson, C.C. The geochemistry of iodine—A review. Environ. Geochem. Health 1986, 8, 31–54. [Google Scholar] [CrossRef]
- Zimmermann, M.B. Iodine Deficiency. Endocr. Rev. 2009, 30, 376–408. [Google Scholar] [CrossRef]
- Swanson, E.W.; Miller, J.K.; Mueller, F.J.; Patton, C.S.; Bacon, J.A.; Ramsey, N. Iodine in Milk and Meat of Dairy Cows Fed Different Amounts of Potassium Iodide or Ethylenediamine Dihydroiodide1. J. Dairy Sci. 1990, 73, 398–405. [Google Scholar] [CrossRef]
- Galton, D.M.; Petersson, L.G.; Erb, H.N. Milk iodine residues in herds practicing iodophor premilking teat disinfection. J. Dairy Sci. 1986, 69, 267–271. [Google Scholar] [CrossRef]
- van der Reijden, O.L.; Zimmermann, M.B.; Galetti, V. Iodine in dairy milk: Sources, concentrations and importance to human health. Best Pract. Res. Clin. Endocrinol. Metab. 2017, 31, 385–395. [Google Scholar] [CrossRef] [PubMed]
- Niwattisaiwong, S. Iodine deficiency: Clinical implications. Cleve Clin. J. Med. 2017, 84, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Finer, L.B.; Henshaw, S.K. Disparities in rates of unintended pregnancy in the United States, 1994 and 2001. Perspect. Sex. Reprod. Health 2006, 38, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, M.B.; Andersson, M. Update on iodine status worldwide. Curr. Opin. Endocrinol. Diabetes Obes. 2012, 19, 382–387. [Google Scholar] [CrossRef]
- Markel, H. “When it rains it pours”: Endemic goiter, iodized salt, and David Murray Cowie, MD. Am. J. Public Health 1987, 77, 219–229. [Google Scholar] [CrossRef]
- Gibson, R.S. Principles of Nutritional Assessment, 2nd ed.; Oxford University Press: New York, NY, USA, 2005; p. 908. [Google Scholar]
- Caldwell, K.L.; Makhmudov, A.; Ely, E.; Jones, R.L.; Wang, R.Y. Iodine status of the US population, National Health and Nutrition Examination Survey, 2005–2006 and 2007–2008. Thyroid 2011, 21, 419–427. [Google Scholar] [CrossRef]
- Krajčovičová-Kudláčková, M.; Bučková, K.; Klimeš, I.; Šeboková, E. Iodine Deficiency in Vegetarians and Vegans. Ann. Nutr. Metab. 2003, 47, 183–185. [Google Scholar] [CrossRef] [PubMed]
- Pearce, E.N.; Pino, S.; He, X.; Bazrafshan, H.R.; Lee, S.L.; Braverman, L.E. Sources of Dietary Iodine: Bread, Cows’ Milk, and Infant Formula in the Boston Area. J. Clin. Endocrinol. Metab. 2004, 89, 3421–3424. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization; The United Nations Children’s Fund; International Council for Control of Iodine Deficiency Disorders. Assessment of iodine deficiency disorders and monitoring their elimination. In A Guide for Programme Managers; World Health Organization: Geneva, Switzerland, 2007. [Google Scholar]
- Alderman, M.H.; Lamport, B. Moderate Sodium RestrictionDo the Benefits Justify the Hazards? Am. J. Hypertens. 1990, 3, 499–504. [Google Scholar] [CrossRef] [PubMed]
- Tayie, F.A.K.; Jourdan, K. Hypertension, dietary salt restriction, and iodine deficiency among adults. Am. J. Hypertens. 2010, 23, 1095–1102. [Google Scholar] [CrossRef] [PubMed]
- Boon, C.S.; Taylor, C.L.; Henney, J.E. Strategies to Reduce Sodium Intake in the United States; National Academies Press: Washington, DC, USA, 2010. [Google Scholar]
- Appel, L.J.; Frohlich, E.D.; Hall, J.E.; Pearson, T.A.; Sacco, R.L.; Seals, D.R.; Sacks, F.M.; Smith, S.C., Jr.; Vafiadis, D.K.; Van Horn, L.V. The importance of population-wide sodium reduction as a means to prevent cardiovascular disease and stroke: A call to action from the American Heart Association. Circulation 2011, 123, 1138–1143. [Google Scholar] [CrossRef] [PubMed]
- Dean, S. Medical Nutrition Therapy for Thyroid, Adrenal, and Other Endocrine Disorders. In Krause’s Food and the Nutrition Care Process, 14th ed.; Mahn, L.K., Raymond, J.L., Eds.; Elsevier: Saint Louis, MO, USA, 2017; pp. 619–630. [Google Scholar]
- National Instituesd of Health Office of Dietary Supplements. Iodine Fact. Sheet for Health Professionals; U.S. Department of Health & Human Services: Bethesda, MD, USA, 2019. Available online: https://ods.od.nih.gov/factsheets/Iodine-HealthProfessional/90/history/ (accessed on 2 April 2019).
- König, F.; Andersson, M.; Hotz, K.; Aeberli, I.; Zimmermann, M.B. Ten repeat collections for urinary iodine from spot samples or 24-hour samples are needed to reliably estimate individual iodine status in women. J. Nutr. 2011, 141, 2049–2054. [Google Scholar] [CrossRef] [PubMed]
- Skeaff, S.; Thomson, C.; Eastman, C. Iodine deficiency does exist but is difficult to assess in individuals. N. Z. Med. J. 2009, 122, 101–102. [Google Scholar]
- Johnson, E.C.; Huffman, A.E.; Yoder, H.; Dolci, A.; Perrier, E.T.; Larson-Meyer, D.E.; Armstrong, L.E. Urinary Markers of Hydration during 3-day Water Restriction and Graded Rehydration. Eur. J. Nutr. 2019, 1–11. [Google Scholar] [CrossRef]
- Forde, C. Exercise Prescription for the Prevention and Treatment of Disease: Scoring the International Physical Activity Questionnaire (IPAQ). Available online: https://ugc.futurelearn.com/uploads/files/bc/c5/bcc53b14-ec1e-4d90-88e3-1568682f32ae/IPAQ_PDF.pdf (accessed on 2 April 2019).
- Zimmermann, M.B. Iodine requirements and the risks and benefits of correcting iodine deficiency in populations. J. Trace Elem. Med. Biol. 2008, 22, 81–92. [Google Scholar] [CrossRef]
- Trumbo, P.; Yates, A.A.; Schlicker, S.; Poos, M. Dietary reference intakes: Vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium, and zinc. J. Acad. Nutr. Diet. 2001, 101, 294. [Google Scholar]
- Halliday, T.M.; Peterson, N.J.; Thomas, J.J.; Kleppinger, K.; Hollis, B.W.; Larson-Meyer, D.E. Vitamin D status relative to diet, lifestyle, injury, and illness in college athletes. Med. Sci. Sports Exerc. 2011, 43, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Total Diet Study Elements Results Summary Statistics—Market Baskets 2006 through 2013. Available online: https://www.fda.gov/downloads/food...totaldietstudy/ucm184301.pdf (accessed on 4 March 2019).
- Bath, S.C.; Hill, S.; Infante, H.G.; Elghul, S.; Nezianya, C.J.; Rayman, M.P. Iodine concentration of milk-alternative drinks available in the UK in comparison with cows’ milk. Br. J. Nutr. 2017, 118, 525–532. [Google Scholar] [CrossRef] [PubMed]
- Low Iodine Diet. Available online: https://www.thyroid.org/wp-content/uploads/patients/brochures/LowIodineDietFAQ.pdf (accessed on 4 March 2019).
- Pehrsson, P.R.; Patterson, K.Y.; Spungen, J.H.; Wirtz, M.S.; Andrews, K.W.; Dwyer, J.T.; Swanson, C.A. Iodine in food-and dietary supplement–composition databases. Am. J. Clin. Nutr. 2016, 104, 868S–876S. [Google Scholar] [CrossRef] [PubMed]
- Teas, J.; Pino, S.; Critchley, A.; Braverman, L.E. Variability of iodine content in common commercially available edible seaweeds. Thyroid 2004, 14, 836–841. [Google Scholar] [CrossRef]
- Pennington, J.A.; Schoen, S.A. Total diet study: Estimated dietary intakes of nutritional elements, 1982–1991. Int. J. Vitam. Nutr. Res. 1996, 66, 350–362. [Google Scholar]
- Dasgupta, P.K.; Liu, Y.; Dyke, J.V. Iodine Nutrition: Iodine Content of Iodized Salt in the United States. Environ. Sci. Technol. 2008, 42, 1315–1323. [Google Scholar] [CrossRef]
- Baloch, Z.; Carayon, P.; Conte-Devolx, B.; Demers, L.M.; Feldt-Rasmussen, U.; Henry, J.F.; LiVosli, V.A.; Niccoli-Sire, P.; John, R.; Ruf, J. Laboratory medicine practice guidelines. Laboratory support for the diagnosis and monitoring of thyroid disease. Thyroid Off. J. Am. Thyroid Assoc. 2003, 13, 3. [Google Scholar]
- Hagströmer, M.; Oja, P.; Sjöström, M. The International Physical Activity Questionnaire (IPAQ): A study of concurrent and construct validity. Public Health Nutr. 2006, 9, 755–762. [Google Scholar] [CrossRef]
- Stanbury, J.B.; Ermans, A.E.; Bourdoux, P.; Todd, C.; Oken, E.; Tonglet, R.; Vidor, G.; Braverman, L.E.; Medeiros-Neto, G. Iodine-induced hyperthyroidism: Occurrence and epidemiology. Thyroid 1998, 8, 83–100. [Google Scholar] [CrossRef]
- Hemken, R.W. Milk and meat iodine content: Relation to human health. J. Am. Vet. Med Assoc. 1980, 176, 1119–1121. [Google Scholar]
- Kaufmann, S.; Wolfram, G.; Delange, F.; Rambeck, W.A. Iodine supplementation of laying hen feed: A supplementary measure to eliminate iodine deficiency in humans? Z. Ernährungswiss. 1998, 37, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Spitzweg, C.; Joba, W.; Eisenmenger, W.; Heufelder, A.E. Analysis of human sodium iodide symporter gene expression in extrathyroidal tissues and cloning of its complementary deoxyribonucleic acids from salivary gland, mammary gland, and gastric mucosa. J. Clin. Endocrinol. Metab. 1998, 83, 1746–1751. [Google Scholar] [CrossRef] [PubMed]
- Hubble, I.B.; Mein, G.A. Effect of pre-milking udder preparation of dairy cows on milk quality. Aust. J. Dairy Technol. (Aust.) 1986, 41, 66–70. [Google Scholar]
- Chandler, W.L. Iodine on the Poultry farm. Poult. Sci. 1926, 6, 31–35. [Google Scholar] [CrossRef]
- Travnicek, J.; Kroupova, V.; Herzig, I.; Kursa, J. Iodine content in consumer hen eggs. Vet. Med. Praha 2006, 51, 93. [Google Scholar] [CrossRef]
- Bourre, J.M.; Galea, F. An important source of omega-3 fatty acids, vitamins D and E, carotenoids, iodine and selenium: A new natural multi-enriched egg. J. Nutr. Health Aging 2006, 10, 371. [Google Scholar]
- Michella, S.M.; Slaugh, B.T. Producing and marketing a specialty egg. Poult. Sci. 2000, 79, 975–976. [Google Scholar] [CrossRef]
- Liu, L.; Li, X.; Wang, H.; Cao, X.; Ma, W. Reduction of iodate in iodated salt to iodide during cooking with iodine as measured by an improved HPLC/ICP-MS method. J. Nutr. Biochem. 2017, 42, 95–100. [Google Scholar] [CrossRef]
- Meinhardt, A.K.; Muller, A.; Burcza, A.; Greiner, R. Influence of cooking on the iodine content in potatoes, pasta and rice using iodized salt. Food Chem. 2019, 301. [Google Scholar] [CrossRef]
- Andersson, M.; de Benoist, B.; Rogers, L. Epidemiology of iodine deficiency: Salt iodisation and iodine status. Best Pract. Res. Clin. Endocrinol. Metab. 2010, 24, 1–11. [Google Scholar] [CrossRef]
- Kim, J.Y.; Moon, S.J.; Kim, K.R.; Sohn, C.Y.; Oh, J.J. Dietary iodine intake and urinary iodine excretion in normal Korean adults. Yonsei Med. J. 1998, 39, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Ballew, C. Final Report on the Alaska Traditional Diet Survey. Available online: http://anthctoday.org/epicenter/publications/Reports_Pubs/traditional_diet.pdf (accessed on 4 March 2019).
- Gershengorn, M.C.; Wolff, J.; Larsen, P.R. Thyroid-pituitary feedback during iodine repletion. J. Clin. Endocrinol. Metab. 1976, 43, 601–605. [Google Scholar] [CrossRef] [PubMed]
- Chung, H.R. Iodine and thyroid function. Ann. Pediatric Endocrinol. Metab. 2014, 19, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Morton, M.E.; Chaikoff, I.L.; Rosenfeld, S. Inhibiting effect of inorganic iodide on the formation in vitro of thyroxine and diiodotyrosine by surviving thyroid tissue. J. Biol. Chem. 1944, 154, 381–387. [Google Scholar]
- Wolff, J.; Chaikoff, I.L. Plasma inorganic iodide as a homeostatic regulator of thyroid function. J. Biol. Chem. 1948, 174, 555–564. [Google Scholar] [PubMed]
- Ershow, A.; Skeaff, S.; Merkel, J.; Pehrsson, P. Development of databases on iodine in foods and dietary supplements. Nutrients 2018, 10, 100. [Google Scholar] [CrossRef] [PubMed]
- Hetzel, B.S.; Dunn, J.T. The iodine deficiency disorders: Their nature and prevention. Annu. Rev. Nutr. 1989, 9, 21–38. [Google Scholar] [CrossRef]
- Rasmussen, L.B.; Ovesen, L.; Christiansen, E. Day-to-day and within-day variation in urinary iodine excretion. Eur. J. Clin. Nutr. 1999, 53, 401. [Google Scholar] [CrossRef]
- Als, C.; Helbling, A.; Peter, K.; Haldimann, M.; Zimmerli, B.; Gerber, H. Urinary iodine concentration follows a circadian rhythm: A study with 3023 spot urine samples in adults and children. J. Clin. Endocrinol. Metab. 2000, 85, 1367–1369. [Google Scholar] [CrossRef]
- Als, C.; Haldimann, M.; Bürgi, E.; Donati, F.; Gerber, H.; Zimmerli, B. Swiss pilot study of individual seasonal fluctuations of urinary iodine concentration over two years: Is age-dependency linked to the major source of dietary iodine? Eur. J. Clin. Nutr. 2003, 57, 636. [Google Scholar] [CrossRef]
- Moreno-Reyes, R.; Carpentier, Y.A.; Macours, P.; Gulbis, B.; Corvilain, B.; Glinoer, D.; Goldman, S. Seasons but not ethnicity influence urinary iodine concentrations in Belgian adults. Eur. J. Nutr. 2011, 50, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Bourdoux, P. Evaluation of the iodine intake: Problems of the iodine/creatinine ratio-comparison with iodine excretion and daily fluctuations of iodine concentration. Exp. Clin. Endocrinol. Diabetes 1998, 106, S17–S20. [Google Scholar] [CrossRef] [PubMed]
- Ma, Z.F.; Skeaff, S.A. Thyroglobulin as a biomarker of iodine deficiency: A review. Thyroid 2014, 24, 1195–1209. [Google Scholar] [CrossRef] [PubMed]
- Spencer, C.A.; Wang, C.-C. Thyroglobulin measurement: Techniques, clinical benefits, and pitfalls. Endocrinol. Metab. Clin. N. Am. 1995, 24, 841–863. [Google Scholar] [CrossRef]

| Food Item | Estimated Iodine Level (µg per Serving) | Serving Size |
|---|---|---|
| Milk (fluid) | 90.86 a | 1 cup |
| Soy Milk | 2.2 b | 1 cup |
| Soy Protein Bar | 20 c | 1 bar |
| Soy Protein Powder | 0 d | 1 scoop |
| Soy Sauce | 0 d | 1 Tbsp |
| Non-dairy Milk | 2.2 b | 1 cup |
| Orange Juice | 0 a | 1 cup |
| Cereal | 1.62 a | ¾ cup |
| Bread | 1.18 a | 1 slice (26 g) |
| Subway Sandwich | 4.14 a | 6-inch sandwich |
| Bagel | 4.312 a | 1 bagel (95 g) |
| Yogurt | 87 a | 1 cup |
| Cheese | 13.33 a | 2 oz |
| Egg | 21.42 a | 1 egg (50 g) |
| Margarine | 0 a | 1 tsp |
| Liver | 11 a | 100 g (3.5 oz) |
| Cod | 93 e | 3.5 oz |
| Grouper | 84 f | 3.5 oz |
| Haddock | 224 e | 3.5 oz |
| Halibut | 9.9 e | 3.5 oz |
| Herring | 84 f | 3.5 oz |
| Mackerel | 84 f | 3.5 oz |
| Perch | 10.89 e | 3.5 oz |
| Salmon | 10.43 e | 3.5 oz |
| Sardines | 6.69 g | 3.5 oz |
| Seabass | 84 f | 3.5 oz |
| Swordfish | 19.8 e | 3.5 oz |
| Tukaoua | 84 f | 3.5 oz |
| Tuna Albacore | 6.69 a | 3.5 oz |
| Tuna Light | 6.69 a | 3.5 oz |
| Walleye | 84 f | 3.5 oz |
| Other Fish | 22 h | 3.5 oz |
| Clams | 74.8 e | 4 oz |
| Crabmeat | 42.56 e | 4 oz |
| Lobster | 209.67 e | 4 oz |
| Mussels | 9.14 i | 4 oz |
| Oysters | 135 e | 4 oz |
| Scallops | 9.14 e | 4 oz |
| Shimp | 8.184 a | 4 oz |
| Seaweed | 34.56 j | 2.6 g (1 sheet) |
| Iodized Table Salt | 68 k | 1.5 g (1/4 tsp) |
| Other Salt | 0 l | 1.5 g |
| Multivitamin-(Iodine-Containing) | 150 m | 1 tablet |
| Kelp Supplement | 225 c | 1 capsule |
| Mean ± SEM | n | ||
|---|---|---|---|
| Age | 31.6 ± 0.8 | 111 | |
| Height (cm) | 173.2 ± 1.0 | 111 | |
| Body Mass (kg) | 74.9 ± 1.7 | 111 | |
| BMI (kg/M2) | 24.8 ± 0.4 | 111 | |
| Hematocrit (%) | 45.1 ± 0.28 | 110 | |
| TSH (mIU/L) | 2.1 ± 0.11 | 107 | |
| Male (n) | Female (n) | ||
| Sex | 59 | 52 | 111 |
| BMI Underweight | 1 | 0 | 1 |
| BMI Normal | 25 | 36 | 61 |
| BMI Overweight | 26 | 13 | 39 |
| BMI Obese | 7 | 3 | 10 |
| Smokers | 4 | 0 | 4 |
| IPAQ Low | 10 | 7 | 17 |
| IPAQ Moderate | 21 | 30 | 51 |
| IPAQ High | 22 | 14 | 36 |
| Food Serving Size | Iodine (µg per Serving) | Frequency |
|---|---|---|
| Milk (1 cup fluid) (n = 110 *) | 90.86 | 0 or <1 times·month−1 = 17 |
| 1–3 times·month−1 = 12 | ||
| 1 time·week−1 = 10 | ||
| 2–4 times·week−1 = 26 | ||
| 5–6 times·week−1 = 15 | ||
| 1 time·day−1 = 17 | ||
| 2–3 times·day−1 = 11 | ||
| 4–5 times·day−1 = 2 | ||
| Yogurt (1 cup) (n = 111 *) | 87 | 0 or <1 month−1 = 26 |
| 1–3 times·month−1 = 17 | ||
| 1 time·week−1 = 15 | ||
| 2–4 times·week−1 = 24 | ||
| 5–6 times·week−1 = 18 | ||
| 1 time·day−1 = 9 | ||
| 2–3 times·day−1 = 2 | ||
| 4–5 times·day−1 = 0 | ||
| Eggs (1 whole) (n = 109 *) | 21.42 | 0 or <1 month−1 = 16 |
| 1–3 times·month−1 = 8 | ||
| 1 time·week−1 = 15 | ||
| 2–4 times·week−1 = 29 | ||
| 5–6 times·week−1 = 18 | ||
| 1 time·day−1 = 6 | ||
| 2–3 times·day−1 = 11 | ||
| 4–5 times·day−1 = 6 | ||
| Total Seafood (3.75 oz) (n = 110 *) | 61 | 0 or <1 month−1 = 28 |
| 1–3 times·month−1 = 48 | ||
| 1 time·week−1 = 22 | ||
| 2–4 times·week−1 = 9 | ||
| 5–6 times·week−1 = 1 | ||
| 1 time·day−1 = 2 | ||
| 2–3 times·day−1 = 0 | ||
| 4–5 times·day−1 = 0 | ||
| Iodized Table Salt (1.5 g) (n = 108 *) | 68 | 0 or <1 month−1 = 28 |
| 1–3 times·month−1 = 18 | ||
| 1 time·week−1 = 12 | ||
| 2–4 times·week−1 = 12 | ||
| 5–6 times·week−1 = 12 | ||
| 1 time·day−1 = 14 | ||
| 2–3 times·day−1 = 10 | ||
| 4–5 times·day−1 = 2 | ||
| Multivitamin (1 tablet) (n = 106 *) | 150 | 0 or <1 month−1 = 67 |
| 1–3 times·month−1 = 8 | ||
| 1 time·week−1 = 3 | ||
| 2–4 times·week−1 = 4 | ||
| 5–6 times·week−1 = 4 | ||
| 1 time·day−1 = 17 | ||
| 2–3 times·day−1 = 2 | ||
| 4–5 times·day−1 = 1 |
| R2 | SEE | Beta | Sig. | |
|---|---|---|---|---|
| Model | 0.991 | 21.81 | 0.005 | |
| Total Dairy | - | - | 0.066 | >0.001 |
| Total Fish | - | - | 0.108 | >0.001 |
| Multivitamin | - | - | 0.034 | >0.001 |
| Iodized Table Salt | - | - | 0.033 | >0.001 |
| Seaweed | - | - | 0.122 | >0.001 |
| Egg | - | - | 0.032 | >0.001 |
| R2 | SEE | Beta | Sig. | |
|---|---|---|---|---|
| Model 1 | 0.229 | 118.1 | 0.001 | |
| Total Dairy | - | - | 0.017 | 0.001 |
| Total Fish | - | - | −0.033 | 0.249 |
| Multivitamin | - | - | −0.006 | 0.128 |
| Iodized Table Salt | - | - | 0.003 | 0.704 |
| Seaweed | - | - | 0.111 | 0.455 |
| Egg | - | - | 0.037 | 0.032 |
| Model 2 | 0.228 | 117.6 | 0.001 | |
| Total Dairy | - | - | 0.017 | 0.001 |
| Total Fish | - | - | −0.033 | 0.234 |
| Multivitamin | - | - | −0.006 | 0.118 |
| Seaweed | - | - | 0.105 | 0.474 |
| Egg | - | - | 0.038 | 0.024 |
| Model 3 | 0.224 | 117.3 | 0.001 | |
| Total Dairy | - | - | 0.016 | 0.001 |
| Total Fish | - | - | −0.033 | 0.234 |
| Multivitamin | - | - | −0.006 | 0.125 |
| Egg | - | - | 0.040 | 0.016 |
| Model 4 | 0.213 | 117.5 | 0.001 | |
| Total Dairy | - | - | 0.016 | 0.001 |
| Multivitamin | - | - | −0.006 | 0.136 |
| Egg | - | - | 0.041 | 0.013 |
| Model 5 | 0.197 | 118.2 | 0.000 | |
| Total Dairy | - | - | 0.016 | 0.000 |
| Egg | - | - | 0.039 | 0.018 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gostas, D.E.; Larson-Meyer, D.E.; Yoder, H.A.; Huffman, A.E.; Johnson, E.C. Dietary Relationship with 24 h Urinary Iodine Concentrations of Young Adults in the Mountain West Region of the United States. Nutrients 2020, 12, 121. https://doi.org/10.3390/nu12010121
Gostas DE, Larson-Meyer DE, Yoder HA, Huffman AE, Johnson EC. Dietary Relationship with 24 h Urinary Iodine Concentrations of Young Adults in the Mountain West Region of the United States. Nutrients. 2020; 12(1):121. https://doi.org/10.3390/nu12010121
Chicago/Turabian StyleGostas, Demetre E., D. Enette Larson-Meyer, Hillary A. Yoder, Ainsley E. Huffman, and Evan C. Johnson. 2020. "Dietary Relationship with 24 h Urinary Iodine Concentrations of Young Adults in the Mountain West Region of the United States" Nutrients 12, no. 1: 121. https://doi.org/10.3390/nu12010121
APA StyleGostas, D. E., Larson-Meyer, D. E., Yoder, H. A., Huffman, A. E., & Johnson, E. C. (2020). Dietary Relationship with 24 h Urinary Iodine Concentrations of Young Adults in the Mountain West Region of the United States. Nutrients, 12(1), 121. https://doi.org/10.3390/nu12010121





