Animal Models of Undernutrition and Enteropathy as Tools for Assessment of Nutritional Intervention
Abstract
1. Introduction
2. Undernutrition Models
2.1. Caloric Restriction
2.2. Protein Undernutrition
2.2.1. Maternal Protein Undernutrition
2.2.2. Protein-Deficient Diet
2.3. Regional Diets
2.4. Zinc-Deficient Diets
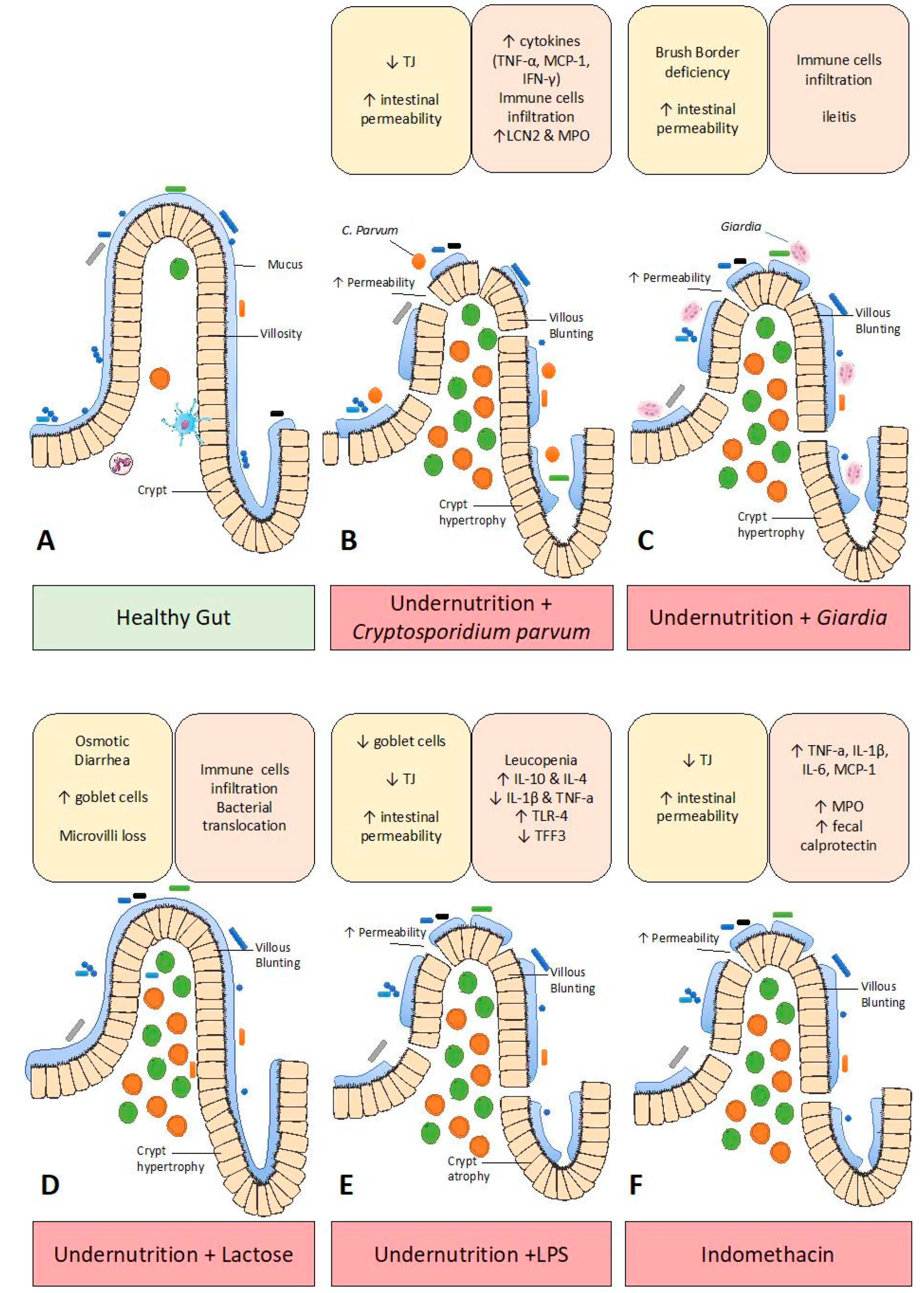
3. Enteropathy Models
3.1. Microbiota Transplantation
3.2. Pathogen-Induced Enteropathy
3.3. Lactose-Induced Enteropathy
3.4. LPS-Induced Enteropathy
3.5. Indomethacin-Induced Enteropathy
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- United Nations Children’s Fund (UNICEF); World Health Organization; International Bank for Reconstruction and Development/The World Bank. Levels and Trends in Child Malnutrition: Key Findings of the 2018 Edition of the Joint Child Malnutrition Estimates; World Health Organization: Geneva, Switzerland, 2018; Licence: CC BY-NC-SA 3.0 IGO. [Google Scholar]
- Black, R.E.; Victora, C.G.; Walker, S.P.; Bhutta, Z.A.; Christian, P.; de Onis, M.; Ezzati, M.; Grantham-McGregor, S.; Katz, J.; Martorell, R.; et al. Maternal and child undernutrition and overweight in low-income and middle-income countries. Lancet 2013, 382, 427–451. [Google Scholar] [CrossRef]
- Victora, C.G.; Adair, L.; Fall, C.; Hallal, P.C.; Martorell, R.; Richter, L.; Sachdev, H.S. Maternal and Child Undernutrition Study Group Maternal and child undernutrition: Consequences for adult health and human capital. Lancet 2008, 371, 340–357. [Google Scholar] [CrossRef]
- Humphrey, J.H. Child undernutrition, tropical enteropathy, toilets, and handwashing. Lancet 2009, 374, 1032–1035. [Google Scholar] [CrossRef]
- Amadi, B.; Besa, E.; Zyambo, K.; Kaonga, P.; Louis-Auguste, J.; Chandwe, K.; Tarr, P.I.; Denno, D.M.; Nataro, J.P.; Faubion, W.; et al. Impaired Barrier Function and Autoantibody Generation in Malnutrition Enteropathy in Zambia. EBioMedicine 2017, 22, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Farràs, M.; Chandwe, K.; Mayneris-Perxachs, J.; Amadi, B.; Louis-Auguste, J.; Besa, E.; Zyambo, K.; Guerrant, R.; Kelly, P.; Swann, J.R. Characterizing the metabolic phenotype of intestinal villus blunting in Zambian children with severe acute malnutrition and persistent diarrhea. PLoS ONE 2018, 13, e0192092. [Google Scholar] [CrossRef]
- Attia, S.; Versloot, C.J.; Voskuijl, W.; van Vliet, S.J.; Di Giovanni, V.; Zhang, L.; Richardson, S.; Bourdon, C.; Netea, M.G.; Berkley, J.A.; et al. Mortality in children with complicated severe acute malnutrition is related to intestinal and systemic inflammation: An observational cohort study. Am. J. Clin. Nutr. 2016, 104, 1441–1449. [Google Scholar] [CrossRef] [PubMed]
- Kosek, M.; Guerrant, R.L.; Kang, G.; Bhutta, Z.; Yori, P.P.; Gratz, J.; Gottlieb, M.; Lang, D.; Lee, G.; Haque, R.; et al. Assessment of environmental enteropathy in the MAL-ED cohort study: Theoretical and analytic framework. Clin. Infect. Dis. 2014, 59 (Suppl. 4), S239–S247. [Google Scholar] [CrossRef]
- Demoinet, E.; Roy, R. Analysis of Transgenerational Phenotypes Following Acute Starvation in AMPK-Deficient C. elegans. Methods Mol. Biol. 2018, 1732, 565–579. [Google Scholar]
- Chalvon-Demersay, T.; Even, P.C.; Chaumontet, C.; Piedcoq, J.; Viollet, B.; Gaudichon, C.; Tomé, D.; Foretz, M.; Azzout-Marniche, D. Modifying the Dietary Carbohydrate-to-Protein Ratio Alters the Postprandial Macronutrient Oxidation Pattern in Liver of AMPK-Deficient Mice. J. Nutr. 2017, 147, 1669–1676. [Google Scholar] [CrossRef]
- Ibrahim, M.K.; Zambruni, M.; Melby, C.L.; Melby, P.C. Impact of Childhood Malnutrition on Host Defense and Infection. Clin. Microbiol. Rev. 2017, 30, 919–971. [Google Scholar] [CrossRef]
- Núñez, I.N.; Galdeano, C.M.; Carmuega, E.; Weill, R.; de Moreno de LeBlanc, A.; Perdigón, G. Effect of a probiotic fermented milk on the thymus in Balb/c mice under non-severe protein-energy malnutrition. Br. J. Nutr. 2013, 110, 500–508. [Google Scholar] [CrossRef]
- Pan, F.; Zhang, L.; Li, M.; Hu, Y.; Zeng, B.; Yuan, H.; Zhao, L.; Zhang, C. Predominant gut Lactobacillus murinus strain mediates anti-inflammaging effects in calorie-restricted mice. Microbiome 2018, 6, 54. [Google Scholar] [CrossRef]
- Park, C.Y.; Park, S.; Kim, M.S.; Kim, H.-K.; Han, S.N. Effects of mild calorie restriction on lipid metabolism and inflammation in liver and adipose tissue. Biochem. Biophys. Res. Commun. 2017, 490, 636–642. [Google Scholar] [CrossRef]
- Ma, T.Y.; Hollander, D.; Dadufalza, V.; Krugliak, P. Effect of aging and caloric restriction on intestinal permeability. Exp. Gerontol. 1992, 27, 321–333. [Google Scholar] [CrossRef]
- Mehus, A.A.; Picklo, M.J. Brain and Hepatic Mt mRNA Is Reduced in Response to Mild Energy Restriction and n-3 Polyunsaturated Fatty Acid Deficiency in Juvenile Rats. Nutrients 2017, 9, 1145. [Google Scholar] [CrossRef]
- Jahng, J.W.; Kim, J.G.; Kim, H.J.; Kim, B.-T.; Kang, D.-W.; Lee, J.-H. Chronic food restriction in young rats results in depression- and anxiety-like behaviors with decreased expression of serotonin reuptake transporter. Brain Res. 2007, 1150, 100–107. [Google Scholar] [CrossRef]
- Hallam, M.C.; Reimer, R.A. Postnatal Prebiotic Fiber Intake in Offspring Exposed to Gestational Protein Restriction Has Sex-Specific Effects on Insulin Resistance and Intestinal Permeability in Rats. J. Nutr. 2014, 144, 1556–1563. [Google Scholar] [CrossRef][Green Version]
- Aubert, P.; Oleynikova, E.; Rizvi, H.; Ndjim, M.; Le Berre-Scoul, C.; Grohard, P.A.; Chevalier, J.; Segain, J.-P.; Le Drean, G.; Neunlist, M.; et al. Maternal protein restriction induces gastrointestinal dysfunction and enteric nervous system remodeling in rat offspring. FASEB J. 2019, 33, 770–781. [Google Scholar] [CrossRef]
- Chen, Y.; Mou, D.; Hu, L.; Zhen, J.; Che, L.; Fang, Z.; Xu, S.; Lin, Y.; Feng, B.; Li, J.; et al. Effects of Maternal Low-Energy Diet during Gestation on Intestinal Morphology, Disaccharidase Activity, and Immune Response to Lipopolysaccharide Challenge in Pig Offspring. Nutrients 2017, 9, 1115. [Google Scholar] [CrossRef]
- D’Inca, R.; Kloareg, M.; Gras-Le Guen, C.; Le Huërou-Luron, I. Intrauterine growth restriction modifies the developmental pattern of intestinal structure, transcriptomic profile, and bacterial colonization in neonatal pigs. J. Nutr. 2010, 140, 925–931. [Google Scholar] [CrossRef]
- Ferenc, K.; Pilżys, T.; Skrzypek, T.; Garbicz, D.; Marcinkowski, M.; Dylewska, M.; Gładysz, P.; Skorobogatov, O.; Gajewski, Z.; Grzesiuk, E.; et al. Structure and Function of Enterocyte in Intrauterine Growth Retarded Pig Neonates. Dis. Markers 2017, 2017, 5238134. [Google Scholar] [CrossRef]
- He, Z.X.; Sun, Z.H.; Yang, W.Z.; Beauchemin, K.A.; Tang, S.X.; Zhou, C.S.; Han, X.F.; Wang, M.; Kang, J.H.; Tan, Z.L. Effects of maternal protein or energy restriction during late gestation on immune status and responses to lipopolysaccharide challenge in postnatal young goats. J. Anim. Sci. 2014, 92, 4856–4864. [Google Scholar] [CrossRef]
- Belmonte, L.; Coëffier, M.; Le Pessot, F.; Miralles-Barrachina, O.; Hiron, M.; Leplingard, A.; Lemeland, J.-F.; Hecketsweiler, B.; Daveau, M.; Ducrotté, P.; et al. Effects of glutamine supplementation on gut barrier, glutathione content and acute phase response in malnourished rats during inflammatory shock. World J. Gastroenterol. 2007, 13, 2833–2840. [Google Scholar] [CrossRef]
- Brown, E.M.; Wlodarska, M.; Willing, B.P.; Vonaesch, P.; Han, J.; Reynolds, L.A.; Arrieta, M.-C.; Uhrig, M.; Scholz, R.; Partida, O.; et al. Diet and specific microbial exposure trigger features of environmental enteropathy in a novel murine model. Nat. Commun. 2015, 6, 7806. [Google Scholar] [CrossRef]
- Eyzaguirre-Velasquez, J.; Olavarria-Ramirez, L.; Gonzalez-Arancibia, C.; Diaz-Merino, C.; Ariz, R.; Lopez, S.; Quiroz, W.; Beltran, C.J.; Bravo, J.A.; Julio-Pieper, M. Protein Malnutrition During Juvenile Age Increases Ileal and Colonic Permeability in the Rat. J. Pediatric Gastroenterol. Nutr. 2017, 64, 707–712. [Google Scholar] [CrossRef]
- Dalvi, P.S.; Yang, S.; Swain, N.; Kim, J.; Saha, S.; Bourdon, C.; Zhang, L.; Chami, R.; Bandsma, R.H.J. Long-term metabolic effects of malnutrition: Liver steatosis and insulin resistance following early-life protein restriction. PLoS ONE 2018, 13, e0199916. [Google Scholar] [CrossRef]
- Fock, R.A.; Vinolo, M.A.R.; Crisma, A.R.; Nakajima, K.; Rogero, M.M.; Borelli, P. Protein-energy malnutrition modifies the production of interleukin-10 in response to lipopolysaccharide (LPS) in a murine model. J. Nutr. Sci. Vitam. 2008, 54, 371–377. [Google Scholar] [CrossRef]
- Mayneris-Perxachs, J.; Bolick, D.T.; Leng, J.; Medlock, G.L.; Kolling, G.L.; Papin, J.A.; Swann, J.R.; Guerrant, R.L. Protein- and zinc-deficient diets modulate the murine microbiome and metabolic phenotype. Am. J. Clin. Nutr. 2016, 104, 1253–1262. [Google Scholar] [CrossRef]
- Santos, A.C.A.; Correia, C.A.; de Oliveira, D.C.; Nogueira-Pedro, A.; Borelli, P.; Fock, R.A. Intravenous Glutamine Administration Modulates TNF-α/IL-10 Ratio and Attenuates NFkB Phosphorylation in a Protein Malnutrition Model. Inflammation 2016, 39, 1883–1891. [Google Scholar] [CrossRef]
- Corware, K.; Yardley, V.; Mack, C.; Schuster, S.; Al-Hassi, H.; Herath, S.; Bergin, P.; Modolell, M.; Munder, M.; Müller, I.; et al. Protein energy malnutrition increases arginase activity in monocytes and macrophages. Nutr. Metab. (Lond.) 2014, 11, 51. [Google Scholar] [CrossRef]
- Ueno, P.M.; Oriá, R.B.; Maier, E.A.; Guedes, M.; Azevedo, O.G.D.; Wu, D.; Willson, T.; Hogan, S.P.; Lima, A.A.M.; Guerrant, R.L.; et al. Alanyl-glutamine promotes intestinal epithelial cell homeostasis in vitro and in a murine model of weanling undernutrition. Am. J. Physiol. Gastrointest. Liver Physiol. 2011, 612–622. [Google Scholar] [CrossRef]
- Sampaio, I.C.; Medeiros, P.H.Q.S.; Rodrigues, F.A.P.; Cavalcante, P.A.; Ribeiro, S.A.; Oliveira, J.S.; Prata, M.M.G.; Costa, D.V.S.; Fonseca, S.G.C.; Guedes, M.M.; et al. Impact of acute undernutrition on growth, ileal morphology and nutrient transport in a murine model. Braz. J. Med. Biol. Res. 2016, 49, e5340. [Google Scholar] [CrossRef]
- Charbonneau, M.R.; O’Donnell, D.; Blanton, L.V.; Totten, S.M.; Davis, J.C.C.; Barratt, M.J.; Cheng, J.; Guruge, J.; Talcott, M.; Bain, J.R.; et al. Sialylated Milk Oligosaccharides Promote Microbiota-Dependent Growth in Models of Infant Undernutrition. Cell 2016, 164, 859–871. [Google Scholar] [CrossRef]
- May, T.; Klatt, K.C.; Smith, J.; Castro, E.; Manary, M.; Caudill, M.A.; Jahoor, F.; Fiorotto, M.L. Choline Supplementation Prevents a Hallmark Disturbance of Kwashiorkor in Weanling Mice Fed a Maize Vegetable Diet: Hepatic Steatosis of Undernutrition. Nutrients 2018, 10, 653. [Google Scholar] [CrossRef]
- Lykke, M.; Hother, A.-L.; Hansen, C.F.; Friis, H.; Mølgaard, C.; Michaelsen, K.F.; Briend, A.; Larsen, T.; Sangild, P.T.; Thymann, T. Malnutrition induces gut atrophy and increases hepatic fat infiltration: Studies in a pig model of childhood malnutrition. Am. J. Transl. Res. 2013, 5, 543–554. [Google Scholar]
- Bolick, D.T.; Kolling, G.L.; Moore, J.H.; de Oliveira, L.A.; Tung, K.; Philipson, C.; Viladomiu, M.; Hontecillas, R.; Bassaganya-Riera, J.; Guerrant, R.L. Zinc deficiency alters host response and pathogen virulence in a mouse model of enteroaggregative Escherichia coli-induced diarrhea. Gut Microbes 2014, 5, 618–627. [Google Scholar] [CrossRef]
- Scott, M.E.; Koski, K.G. Zinc deficiency impairs immune responses against parasitic nematode infections at intestinal and systemic sites. J. Nutr. 2000, 130, 1412S–1420S. [Google Scholar] [CrossRef]
- Kau, A.L.; Planer, J.D.; Liu, J.; Rao, S.; Yatsunenko, T.; Trehan, I.; Manary, M.J.; Liu, T.-C.; Stappenbeck, T.S.; Maleta, K.M.; et al. Functional characterization of IgA-targeted bacterial taxa from undernourished Malawian children that produce diet-dependent enteropathy. Sci. Transl. Med. 2015, 7, 276ra24. [Google Scholar] [CrossRef]
- Islam, M.A.; Rahman, M.M.; Mahalanabis, D. Maternal and socioeconomic factors and the risk of severe malnutrition in a child: A case-control study. Eur. J. Clin. Nutr. 1994, 48, 416–424. [Google Scholar]
- Pinheiro, D.F.; Pacheco, P.D.G.; Alvarenga, P.V.; Buratini, J.; Castilho, A.C.S.; Lima, P.F.; Sartori, D.R.S.; Vicentini-Paulino, M.L.M. Maternal protein restriction affects gene expression and enzyme activity of intestinal disaccharidases in adult rat offspring. Braz. J. Med. Biol. Res. 2013, 46, 287–292. [Google Scholar] [CrossRef]
- Batista, T.H.; Veronesi, V.B.; Ribeiro, A.C.A.F.; Giusti-Paiva, A.; Vilela, F.C. Protein malnutrition during pregnancy alters maternal behavior and anxiety-like behavior in offspring. Nutr. Neurosci. 2017, 20, 437–442. [Google Scholar] [CrossRef]
- Crossland, R.F.; Balasa, A.; Ramakrishnan, R.; Mahadevan, S.K.; Fiorotto, M.L.; Van den Veyver, I.B. Chronic Maternal Low-Protein Diet in Mice Affects Anxiety, Night-Time Energy Expenditure and Sleep Patterns, but Not Circadian Rhythm in Male Offspring. PLoS ONE 2017, 12, e0170127. [Google Scholar] [CrossRef]
- Gressens, P.; Muaku, S.M.; Besse, L.; Nsegbe, E.; Gallego, J.; Delpech, B.; Gaultier, C.; Evrard, P.; Ketelslegers, J.M.; Maiter, D. Maternal protein restriction early in rat pregnancy alters brain development in the progeny. Brain Res. Dev. Brain Res. 1997, 103, 21–35. [Google Scholar] [CrossRef]
- Alamy, M.; Bengelloun, W.A. Malnutrition and brain development: An analysis of the effects of inadequate diet during different stages of life in rat. Neurosci. Biobehav. Rev. 2012, 36, 1463–1480. [Google Scholar] [CrossRef]
- Marwarha, G.; Claycombe-Larson, K.; Schommer, J.; Ghribi, O. Maternal low-protein diet decreases brain-derived neurotrophic factor expression in the brains of the neonatal rat offspring. J. Nutr. Biochem. 2017, 45, 54–66. [Google Scholar] [CrossRef]
- Reyes-Castro, L.A.; Padilla-Gómez, E.; Parga-Martínez, N.J.; Castro-Rodríguez, D.C.; Quirarte, G.L.; Díaz-Cintra, S.; Nathanielsz, P.W.; Zambrano, E. Hippocampal mechanisms in impaired spatial learning and memory in male offspring of rats fed a low-protein isocaloric diet in pregnancy and/or lactation. Hippocampus 2018, 28, 18–30. [Google Scholar] [CrossRef]
- Gould, J.M.; Smith, P.J.; Airey, C.J.; Mort, E.J.; Airey, L.E.; Warricker, F.D.M.; Pearson-Farr, J.E.; Weston, E.C.; Gould, P.J.W.; Semmence, O.G.; et al. Mouse maternal protein restriction during preimplantation alone permanently alters brain neuron proportion and adult short-term memory. Proc. Natl. Acad. Sci. USA 2018, 115, E7398–E7407. [Google Scholar] [CrossRef]
- Laeger, T.; Reed, S.D.; Henagan, T.M.; Fernandez, D.H.; Taghavi, M.; Addington, A.; Münzberg, H.; Martin, R.J.; Hutson, S.M.; Morrison, C.D. Leucine acts in the brain to suppress food intake but does not function as a physiological signal of low dietary protein. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2014, 307, R310–R320. [Google Scholar] [CrossRef]
- Hill, C.M.; Laeger, T.; Albarado, D.C.; McDougal, D.H.; Berthoud, H.-R.; Münzberg, H.; Morrison, C.D. Low protein-induced increases in FGF21 drive UCP1-dependent metabolic but not thermoregulatory endpoints. Sci. Rep. 2017, 7, 8209. [Google Scholar] [CrossRef]
- Pezeshki, A.; Zapata, R.C.; Singh, A.; Yee, N.J.; Chelikani, P.K. Low protein diets produce divergent effects on energy balance. Sci. Rep. 2016, 6, 25145. [Google Scholar] [CrossRef]
- Miyazaki, A.; Kandasamy, S.; Michael, H.; Langel, S.N.; Paim, F.C.; Chepngeno, J.; Alhamo, M.A.; Fischer, D.D.; Huang, H.-C.; Srivastava, V.; et al. Protein deficiency reduces efficacy of oral attenuated human rotavirus vaccine in a human infant fecal microbiota transplanted gnotobiotic pig model. Vaccine 2018, 36, 6270–6281. [Google Scholar] [CrossRef]
- Rho, S.; Kim, H.; Shim, S.H.; Lee, S.Y.; Kim, M.J.; Yang, B.-G.; Jang, M.H.; Han, B.W.; Song, M.K.; Czerkinsky, C.; et al. Protein energy malnutrition alters mucosal IgA responses and reduces mucosal vaccine efficacy in mice. Immunol. Lett. 2017, 190, 247–256. [Google Scholar] [CrossRef]
- Fock, R.A.; Rogero, M.M.; Aurélio, M.; Vinolo, R.; Curi, R.; Borges, M.C.; Borelli, P. Effects of Protein-Energy Malnutrition on NF-KappaB Signalling in Murine Peritoneal Macrophages. Inflammation 2010, 33, 101–109. [Google Scholar] [CrossRef]
- Bhutta, Z.A.; Berkley, J.A.; Bandsma, R.H.J.; Kerac, M.; Trehan, I.; Briend, A. Severe childhood malnutrition. Nat. Rev. Dis. Primers 2017, 3, 17067. [Google Scholar] [CrossRef]
- Teodósio, N.R.; Lago, E.S.; Romani, S.A.M.; Guedes, R.C.A. A regional basic diet from Northeast Brazil as a dietary model of experimental malnutrition. Arch. Latinoam. Nutr. 1990, XL, 533–547. [Google Scholar]
- Brigide, P.; Ataide, T.R.; Baptista, A.S.; Canniatti-Brazaca, S.G.; Abdalla, A.L.; Nascimento Filho, V.F.; Piedade, S.M.S.; Sant’ana, A.E.G. Bioavailability of iron in the regional basic diet (RBD) with dietary supplement in Brazil. Biol. Trace Elem. Res. 2011, 140, 53–65. [Google Scholar] [CrossRef]
- De Queiroz, C.A.; Fonseca, S.G.C.; Frota, P.B.; Figueiredo, I.L.; Aragão, K.S.; Magalhães, C.E.C.; de Carvalho, C.B.; Lima, A.Â.M.; Ribeiro, R.A.; Guerrant, R.L.; et al. Zinc treatment ameliorates diarrhea and intestinal inflammation in undernourished rats. BMC Gastroenterol. 2014, 14, 136. [Google Scholar] [CrossRef]
- Bartelt, L.A.; Bolick, D.T.; Kolling, G.L.; Roche, J.K.; Zaenker, E.I.; Lara, A.M.; Noronha, F.J.; Cowardin, C.A.; Moore, J.H.; Turner, J.R.; et al. Cryptosporidium Priming Is More Effective than Vaccine for Protection against Cryptosporidiosis in a Murine Protein Malnutrition Model. PLoS Negl. Trop. Dis. 2016, 10, 1–29. [Google Scholar] [CrossRef]
- Williams, C.D. A nutritional disease of childhood associated with a maize diet. Arch. Dis Child. 1933, 8, 423–433. [Google Scholar] [CrossRef]
- Furuta, C.; Murakami, H. A Novel Concept of Amino Acid Supplementation to Improve the Growth of Young Malnourished Male Rats. Ann. Nutr. Metab. 2018, 72, 231–240. [Google Scholar] [CrossRef]
- Follis, R.H. A kwashiorkor-like syndrome observed in monkeys fed maize. Proc. Soc. Exp. Biol. Med. 1957, 96, 523–528. [Google Scholar] [CrossRef]
- Krebs, N.F.; Miller, L.V.; Hambidge, K.M. Zinc deficiency in infants and children: A review of its complex and synergistic interactions. Paediatr. Int. Child. Health 2014, 34, 279–288. [Google Scholar] [CrossRef]
- Koo, S.I.; Turk, D.E. Effect of zinc deficiency on the ultrastructure of the pancreatic acinar cell and intestinal epithelium in the rat. J. Nutr. 1977, 107, 896–908. [Google Scholar] [CrossRef]
- Wapnir, R.A.; Garcia-Aranda, J.A.; Mevorach, D.E.; Lifshitz, F. Differential absorption of zinc and low-molecular-weight ligands in the rat gut in protein-energy malnutrition. J. Nutr. 1985, 115, 900–908. [Google Scholar] [CrossRef]
- Van Wouwe, J.P.; Veldhuizen, M. Growth characteristics in laboratory animals fed zinc-deficient, copper-deficient, of histidine-supplemented diets. Biol. Trace Elem. Res. 1996, 55, 71–77. [Google Scholar] [CrossRef]
- Zhong, W.; Zhao, Y.; Sun, X.; Song, Z.; McClain, C.J.; Zhou, Z. Dietary zinc deficiency exaggerates ethanol-induced liver injury in mice: Involvement of intrahepatic and extrahepatic factors. PLoS ONE 2013, 8, e76522. [Google Scholar] [CrossRef]
- QS Medeiros, P.H.; Ledwaba, S.E.; Bolick, D.T.; Giallourou, N.; Yum, L.K.; Costa, D.V.S.; Oriá, R.B.; Barry, E.M.; Swann, J.R.; Lima, A.Â.M.; et al. A murine model of diarrhea, growth impairment and metabolic disturbances with Shigella flexneri infection and the role of zinc deficiency. Gut Microbes 2019, 10, 1–16. [Google Scholar] [CrossRef]
- De Filippo, C.; Cavalieri, D.; Di Paola, M.; Ramazzotti, M.; Poullet, J.B.; Massart, S.; Collini, S.; Pieraccini, G.; Lionetti, P. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Natl. Acad. Sci. USA 2010, 107, 14691–14696. [Google Scholar] [CrossRef]
- Subramanian, S.; Huq, S.; Yatsunenko, T.; Haque, R.; Mahfuz, M.; Alam, M.A.; Benezra, A.; DeStefano, J.; Meier, M.F.; Muegge, B.D.; et al. Persistent gut microbiota immaturity in malnourished Bangladeshi children. Nature 2014, 510, 417–421. [Google Scholar] [CrossRef]
- Vonaesch, P.; Morien, E.; Andrianonimiadana, L.; Sanke, H.; Mbecko, J.-R.; Huus, K.E.; Naharimanananirina, T.; Gondje, B.P.; Nigatoloum, S.N.; Vondo, S.S.; et al. Stunted childhood growth is associated with decompartmentalization of the gastrointestinal tract and overgrowth of oropharyngeal taxa. Proc. Natl. Acad. Sci. USA 2018, 115, E8489–E8498. [Google Scholar] [CrossRef]
- Bartelt, L.A.; Bolick, D.T.; Mayneris-Perxachs, J.; Kolling, G.L.; Medlock, G.L.; Zaenker, E.I.; Donowitz, J.; Thomas-Beckett, R.V.; Rogala, A.; Carroll, I.M.; et al. Cross-modulation of pathogen-specific pathways enhances malnutrition during enteric co-infection with Giardia lamblia and enteroaggregative Escherichia coli. PLoS Pathog. 2017, 13, e1006471. [Google Scholar] [CrossRef]
- Smith, M.I.; Yatsunenko, T.; Manary, M.J.; Trehan, I.; Mkakosya, R.; Cheng, J.; Kau, A.L.; Rich, S.S.; Concannon, P.; Mychaleckyj, J.C.; et al. Gut microbiomes of Malawian twin pairs discordant for kwashiorkor. Science 2013, 339, 548–554. [Google Scholar] [CrossRef]
- Brown, E.M.; Arrieta, M.-C.; Finlay, B.B. A fresh look at the hygiene hypothesis: How intestinal microbial exposure drives immune effector responses in atopic disease. Semin. Immunol. 2013, 25, 378–387. [Google Scholar] [CrossRef]
- Kau, A.L.; Ahern, P.P.; Griffin, N.W.; Goodman, A.L.; Gordon, J.I. Human nutrition, the gut microbiome and the immune system. Nature 2011, 474, 327–336. [Google Scholar] [CrossRef]
- Tzipori, S.; Ward, H. Cryptosporidiosis: Biology, pathogenesis and disease. Microbes Infect. 2002, 4, 1047–1058. [Google Scholar] [CrossRef]
- Dillingham, R.A.; Lima, A.A.; Guerrant, R.L. Cryptosporidiosis: Epidemiology and impact. Microbes Infect. 2002, 4, 1059–1066. [Google Scholar] [CrossRef]
- Huang, D.B.; Chappell, C.; Okhuysen, P.C. Cryptosporidiosis in children. Semin. Pediatr. Infect. Dis. 2004, 15, 253–259. [Google Scholar] [CrossRef]
- Tanriverdi, S.; Grinberg, A.; Chalmers, R.M.; Hunter, P.R.; Petrovic, Z.; Akiyoshi, D.E.; London, E.; Zhang, L.; Tzipori, S.; Tumwine, J.K.; et al. Inferences about the global population structures of Cryptosporidium parvum and Cryptosporidium hominis. Appl. Environ. Microbiol. 2008, 74, 7227–7234. [Google Scholar] [CrossRef]
- Agnew, D.G.; Lima, A.A.; Newman, R.D.; Wuhib, T.; Moore, R.D.; Guerrant, R.L.; Sears, C.L. Cryptosporidiosis in northeastern Brazilian children: Association with increased diarrhea morbidity. J. Infect. Dis. 1998, 177, 754–760. [Google Scholar] [CrossRef]
- Guerrant, D.I.; Moore, S.R.; Lima, A.A.; Patrick, P.D.; Schorling, J.B.; Guerrant, R.L. Association of early childhood diarrhea and cryptosporidiosis with impaired physical fitness and cognitive function four-seven years later in a poor urban community in northeast Brazil. Am. J. Trop. Med. Hyg. 1999, 61, 707–713. [Google Scholar] [CrossRef]
- Gendrel, D.; Treluyer, J.M.; Richard-Lenoble, D. Parasitic diarrhea in normal and malnourished children. Fundam. Clin. Pharm. 2003, 17, 189–197. [Google Scholar] [CrossRef]
- Katona, P.; Katona-Apte, J. The interaction between nutrition and infection. Clin. Infect. Dis. 2008, 46, 1582–1588. [Google Scholar] [CrossRef]
- Coutinho, B.P.; Oriá, R.B.; Vieira, C.M.G.; Sevilleja, J.E.A.D.; Warren, C.A.; Maciel, J.G.; Thompson, M.R.; Pinkerton, R.C.; Aldo, A.M.; Guerrant, R.L. Cryptospordium Infection Causes Undernutrition and Conversly, Weanling Undernutrition Intensifies Infection. J. Parasitol. 2008, 94, 1225–1232. [Google Scholar] [CrossRef]
- Costa, L.B.; JohnBull, E.A.; Reeves, J.T.; Sevilleja, J.E.; Freire, R.S.; Hoffman, P.S.; Lima, A.A.M.; Oriá, R.B.; Roche, J.K.; Guerrant, R.L.; et al. Cryptosporidium-malnutrition interactions: Mucosal disruption, cytokines, and TLR signaling in a weaned murine model. J. Parasitol. 2011, 97, 1113–1120. [Google Scholar] [CrossRef]
- Bolick, D.T.; Mayneris-Perxachs, J.; Medlock, G.L.; Kolling, G.L.; Papin, J.A.; Swann, J.R.; Guerrant, R.L. Increased Urinary Trimethylamine N-Oxide Following Cryptosporidium Infection and Protein Malnutrition Independent of Microbiome Effects. J. Infect. Dis. 2017, 216, 64–71. [Google Scholar] [CrossRef]
- Tinuade, O.; John, O.; Saheed, O.; Oyeku, O.; Fidelis, N.; Olabisi, D. Parasitic etiology of childhood diarrhea. Indian J. Pediatr 2006, 73, 1081–1084. [Google Scholar] [CrossRef]
- Einarsson, E.; Ma’ayeh, S.; Svärd, S.G. An up-date on Giardia and giardiasis. Curr. Opin. Microbiol. 2016, 34, 47–52. [Google Scholar] [CrossRef]
- Squire, S.A.; Ryan, U. Cryptosporidium and Giardia in Africa: Current and future challenges. Parasites Vectors 2017, 10, 195. [Google Scholar] [CrossRef]
- Caron, Y.; Hong, R.; Gauthier, L.; Laillou, A.; Wieringa, F.T.; Berger, J.; Poirot, E. Stunting, Beyond Acute Diarrhoea: Giardia Duodenalis, in Cambodia. Nutrients 2018, 10, 1420. [Google Scholar] [CrossRef]
- Denno, D.M.; VanBuskirk, K.; Nelson, Z.C.; Musser, C.A.; Hay Burgess, D.C.; Tarr, P.I. Use of the lactulose to mannitol ratio to evaluate childhood environmental enteric dysfunction: A systematic review. Clin. Infect. Dis. 2014, 59 (Suppl. 4), S213–S219. [Google Scholar] [CrossRef]
- Hjelt, K.; Paerregaard, A.; Krasilnikoff, P.A. Giardiasis causing chronic diarrhoea in suburban Copenhagen: Incidence, physical growth, clinical symptoms and small intestinal abnormality. Acta Paediatr. 1992, 81, 881–886. [Google Scholar] [CrossRef]
- Goto, R.; Panter-Brick, C.; Northrop-Clewes, C.A.; Manahdhar, R.; Tuladhar, N.R. Poor intestinal permeability in mildly stunted Nepali children: Associations with weaning practices and Giardia lamblia infection. Br. J. Nutr. 2002, 88, 141–149. [Google Scholar] [CrossRef]
- Upadhyay, P.; Ganguly, N.K.; Mahajan, R.C.; Walia, B.N. Intestinal uptake of nutrients in normal and malnourished animals infected with Giardia lamblia. Digestion 1985, 32, 243–248. [Google Scholar] [CrossRef] [PubMed]
- Buret, A.; Gall, D.G.; Olson, M.E. Effects of murine giardiasis on growth, intestinal morphology, and disaccharidase activity. J. Parasitol. 1990, 76, 403–409. [Google Scholar] [CrossRef]
- Astiazarán-García, H.; Espinosa-Cantellano, M.; Castañón, G.; Chávez-Munguía, B.; Martínez-Palomo, A. Giardia lamblia: Effect of infection with symptomatic and asymptomatic isolates on the growth of gerbils (Meriones unguiculatus). Exp. Parasitol. 2000, 95, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Shukla, G.; Bhatia, R.; Sharma, A. Prebiotic inulin supplementation modulates the immune response and restores gut morphology in Giardia duodenalis-infected malnourished mice. Parasitol. Res. 2016, 115, 4189–4198. [Google Scholar] [CrossRef]
- Shukla, G.; Sidhu, R.K. Lactobacillus casei as a probiotic in malnourished Giardia lamblia-infected mice: A biochemical and histopathological study. Can. J. Microbiol. 2011, 57, 127–135. [Google Scholar] [CrossRef] [PubMed]
- Bartelt, L.A.; Roche, J.; Kolling, G.; Bolick, D.; Noronha, F.; Naylor, C.; Hoffman, P.; Warren, C.; Singer, S.; Guerrant, R.; et al. lamblia impairs growth in a murine malnutrition model. J. Clin. Investig. 2013, 123, 2672–2684. [Google Scholar] [CrossRef]
- Platts-Mills, J.A.; Babji, S.; Bodhidatta, L.; Gratz, J.; Haque, R.; Havt, A.; McCormick, B.J.; McGrath, M.; Olortegui, M.P.; Samie, A.; et al. Pathogen-specific burdens of community diarrhoea in developing countries: A multisite birth cohort study (MAL-ED). Lancet Glob. Health 2015, 3, e564–e575. [Google Scholar] [CrossRef]
- Muhsen, K.; Levine, M.M. A systematic review and meta-analysis of the association between Giardia lamblia and endemic pediatric diarrhea in developing countries. Clin. Infect. Dis. 2012, 55 (Suppl. 4), S271–S293. [Google Scholar] [CrossRef]
- Kosek, M.N. MAL-ED Network Investigators Causal Pathways from Enteropathogens to Environmental Enteropathy: Findings from the MAL-ED Birth Cohort Study. EBioMedicine 2017, 18, 109–117. [Google Scholar] [CrossRef] [PubMed]
- Rogawski, E.T.; Liu, J.; Platts-Mills, J.A.; Kabir, F.; Lertsethtakarn, P.; Siguas, M.; Khan, S.S.; Praharaj, I.; Murei, A.; Nshama, R.; et al. Use of quantitative molecular diagnostic methods to investigate the effect of enteropathogen infections on linear growth in children in low-resource settings: Longitudinal analysis of results from the MAL-ED cohort study. Lancet Glob. Health 2018, 6, e1319–e1328. [Google Scholar] [CrossRef]
- Diarrhoeal Disease. Available online: https://www.who.int/news-room/fact-sheets/detail/diarrhoeal-disease (accessed on 20 April 2019).
- Grenov, B.; Briend, A.; Sangild, P.T.; Thymann, T.; Rytter, M.H.; Hother, A.-L.; Mølgaard, C.; Michaelsen, K.F. Undernourished Children and Milk Lactose. Food Nutr. Bull. 2016, 37, 85–99. [Google Scholar] [CrossRef] [PubMed]
- Mitra, S.K.; Sachan, A.; Udupa, V.; Seshadri, S.J.; Jayakumar, K. Histological changes in intestine in semichronic diarrhoea induced by lactose enriched diet in rats: Effect of Diarex-Vet. Indian J. Exp. Biol. 2003, 41, 211–215. [Google Scholar] [PubMed]
- Galvez, J.; Zarzuelo, A.; Crespo, M.E.; Lorente, M.D.; Ocete, M.A.; Jiménez, J. Antidiarrhoeic Activity of Euphorbia hirta Extract and Isolation of an Active Flavonoid Constituent. Planta Med. 1993, 59, 333–336. [Google Scholar] [CrossRef] [PubMed]
- Leforestier, G.; Blais, A.; Blachier, F.; Marsset-Baglieri, A.; Davila-Gay, A.-M.; Perrin, E.; Tomé, D. Effects of galacto-oligosaccharide ingestion on the mucosa-associated mucins and sucrase activity in the small intestine of mice. Eur. J. Nutr. 2009, 48, 457–464. [Google Scholar] [CrossRef] [PubMed]
- Whitfield, C.; Trent, M.S. Biosynthesis and export of bacterial lipopolysaccharides. Annu. Rev. Biochem. 2014, 83, 99–128. [Google Scholar] [CrossRef]
- Park, B.S.; Lee, J.-O. Recognition of lipopolysaccharide pattern by TLR4 complexes. Exp. Mol. Med. 2013, 45, e66. [Google Scholar] [CrossRef]
- Bandsma, R.H.J.; Ackerley, C.; Koulajian, K.; Zhang, L.; Zutphen, T.V.; Dijk, T.H.V.; Xiao, C.; Giacca, A.; Lewis, G.F. A low-protein diet combined with low-dose endotoxin leads to changes in glucose homeostasis in weanling rats. Am. J. Physiol. Endocrinol. Metab. 2015, 309, 466–473. [Google Scholar] [CrossRef]
- Fock, R.A.; Vinolo, M.A.R.; Blatt, S.L.; Borelli, P. Impairment of the hematological response and interleukin-1?? production in protein-energy malnourished mice after endotoxemia with lipopolysaccharide. Braz. J. Med. Biol. Res. 2012, 45, 1163–1171. [Google Scholar] [CrossRef]
- Iwasa, T.; Matsuzaki, T.; Tungalagsuvd, A.; Munkhzaya, M.; Kuwahara, A.; Yasui, T.; Irahara, M. Prenatal undernutrition increases the febrile response to lipopolysaccharides in adulthood in male rats. Int. J. Dev. Neurosci. 2015, 44, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Lyoumi, S.; Tamion, F.; Petit, J.; Dechelotte, P.; Dauguet, C.; Scotte, M.; Hiron, M.; Leplingard, A.; Salier, J.P.; Daveau, M.; et al. Induction and modulation of acute-phase response by protein malnutrition in rats: Comparative effect of systemic and localized inflammation on interleukin-6 and acute-phase protein synthesis. J. Nutr. 1998, 128, 166–174. [Google Scholar] [CrossRef] [PubMed]
- Cui, Y.; Wang, Q.; Wang, M.; Jia, J.; Wu, R. Gardenia Decoction Prevent Intestinal Mucosal Injury by Inhibiting Pro-inflammatory Cytokines and NF-κB Signaling. Front. Pharm. 2019, 10, 180. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.-J.; Qian, J.-X.; Wei, Y.; Guo, Q.; Jin, J.; Sun, X.; Liu, S.-L.; Xu, C.-F.; Zhang, G.-X. Tanshinone IIA Sodium Sulfonate Attenuates LPS-Induced Intestinal Injury in Mice. Gastroenterol. Res. Pract. 2018, 2018, 9867150. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Xu, Y.; Yang, D.; Yu, N.; Bai, Z.; Bian, L. Effect of Polysaccharides from Acanthopanax senticosus on Intestinal Mucosal Barrier of Escherichia coli Lipopolysaccharide Challenged Mice. Asian-Australas. J. Anim. Sci. 2016, 29, 134–141. [Google Scholar] [CrossRef] [PubMed]
- Nozu, T.; Miyagishi, S.; Kumei, S.; Nozu, R.; Takakusaki, K.; Okumura, T. Lovastatin inhibits visceral allodynia and increased colonic permeability induced by lipopolysaccharide or repeated water avoidance stress in rats. Eur. J. Pharm. 2018, 818, 228–234. [Google Scholar] [CrossRef]
- Bein, A.; Zilbershtein, A.; Golosovsky, M.; Davidov, D.; Schwartz, B. LPS Induces Hyper-Permeability of Intestinal Epithelial Cells. J. Cell. Physiol. 2017, 232, 381–390. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Al-sadi, R.; Said, H.M.; Ma, T.Y. Lipopolysaccharide Causes an Increase in Intestinal Tight Junction Permeability in Vitro and in Vivo by Inducing Enterocyte Membrane Expression and Localization of. Am. J. Pathol. 2013, 182, 375–387. [Google Scholar] [CrossRef]
- Guo, S.; Nighot, M.; Al-Sadi, R.; Alhmoud, T.; Nighot, P.; Ma, T.Y. Lipopolysaccharide Regulation of Intestinal Tight Junction Permeability Is Mediated by TLR4 Signal Transduction Pathway Activation of FAK and MyD88. J. Immunol. 2015, 195, 4999–5010. [Google Scholar] [CrossRef]
- Yue, C.; Wang, W.; Tian, W.-L.; Huang, Q.; Zhao, R.-S.; Zhao, Y.-Z.; Li, Q.-R.; Li, J.-S. Lipopolysaccharide-induced failure of the gut barrier is site-specific and inhibitable by growth hormone. Inflamm. Res. 2013, 62, 407–415. [Google Scholar] [CrossRef]
- Chen, S.; Bu, D.; Ma, Y.; Zhu, J.; Sun, L.; Zuo, S.; Ma, J.; Li, T.; Chen, Z.; Zheng, Y.; et al. GYY4137 ameliorates intestinal barrier injury in a mouse model of endotoxemia. Biochem. Pharm. 2016, 118, 59–67. [Google Scholar] [CrossRef]
- Holloway, P.M.; Gillespie, S.; Becker, F.; Vital, S.A.; Nguyen, V.; Alexander, J.S.; Evans, P.C.; Gavins, F.N.E. Sulforaphane induces neurovascular protection against a systemic inflammatory challenge via both Nrf2-dependent and independent pathways. Vasc. Pharm. 2016, 85, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Cai, K.C.; van Mil, S.; Murray, E.; Mallet, J.-F.; Matar, C.; Ismail, N. Age and sex differences in immune response following LPS treatment in mice. Brain Behav. Immun. 2016, 58, 327–337. [Google Scholar] [CrossRef] [PubMed]
- Thun, M.J.; Henley, S.J.; Patrono, C. Nonsteroidal Anti-inflammatory Drugs as Anticancer Agents: Mechanistic, Pharmacologic, and Clinical Issues. J. Natl. Cancer Inst. 2002, 94, 252–266. [Google Scholar] [CrossRef]
- Nash, D.B. The Use of Medicines in the United States: A Detailed Review. Am. Health Drug Benefits 2012, 5, 423. [Google Scholar] [PubMed]
- Syer, S.D.; Wallace, J.L. Environmental and NSAID-enteropathy: Dysbiosis as a common factor. Curr. Gastroenterol. Rep. 2014, 16, 377. [Google Scholar] [CrossRef]
- Graham, D.Y.; Opekun, A.R.; Willingham, F.F.; Qureshi, W.A. Visible small-intestinal mucosal injury in chronic NSAID users. Clin. Gastroenterol. Hepatol. 2005, 3, 55–59. [Google Scholar] [CrossRef]
- Sugimori, S.; Watanabe, T.; Tabuchi, M.; Kameda, N.; Machida, H.; Okazaki, H.; Tanigawa, T.; Yamagami, H.; Shiba, M.; Watanabe, K.; et al. Evaluation of small bowel injury in patients with rheumatoid arthritis by capsule endoscopy: Effects of anti-rheumatoid arthritis drugs. Digestion 2008, 78, 208–213. [Google Scholar] [CrossRef]
- Tanaka, A.; Hase, S.; Miyazawa, T.; Ohno, R.; Takeuchi, K. Role of cyclooxygenase (COX)-1 and COX-2 inhibition in nonsteroidal anti-inflammatory drug-induced intestinal damage in rats: Relation to various pathogenic events. J. Pharm. Exp. 2002, 303, 1248–1254. [Google Scholar] [CrossRef]
- Fortun, P.J.; Hawkey, C.J. Nonsteroidal antiinflammatory drugs and the small intestine. Curr. Opin. Gastroenterol. 2005, 21, 169–175. [Google Scholar] [CrossRef]
- Bjarnason, I.; Hayllar, J.; MacPherson, A.J.; Russell, A.S. Side effects of nonsteroidal anti-inflammatory drugs on the small and large intestine in humans. Gastroenterology 1993, 104, 1832–1847. [Google Scholar] [CrossRef]
- Horibe, S.; Tanahashi, T.; Kawauchi, S.; Mizuno, S.; Rikitake, Y. Preventative effects of sodium alginate on indomethacin-induced small-intestinal injury in mice. Int. J. Med. Sci. 2016, 13, 653–663. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, K.; Kurihara, C.; Furuhashi, H.; Takajo, T.; Maruta, K.; Yasutake, Y.; Sato, H.; Narimatsu, K.; Okada, Y.; Higashiyama, M.; et al. Psychological stress exacerbates NSAID-induced small bowel injury by inducing changes in intestinal microbiota and permeability via glucocorticoid receptor signaling. J. Gastroenterol. 2017, 52, 61–71. [Google Scholar] [CrossRef] [PubMed]
- Han, Y.-M.; Park, J.-M.; Kang, J.X.; Cha, J.-Y.; Lee, H.-J.; Jeong, M.; Go, E.-J.; Hahm, K.B. Mitigation of indomethacin-induced gastrointestinal damages in fat-1 transgenic mice via gate-keeper action of $ømega$-3-polyunsaturated fatty acids. Sci. Rep. 2016, 6, 33992. [Google Scholar] [CrossRef] [PubMed]
- Nagai, K.; Ueno, Y.; Tanaka, S.; Hayashi, R.; Shinagawa, K.; Chayama, K. Polysaccharides derived from Ganoderma lucidum fungus mycelia ameliorate indomethacin-induced small intestinal injury via induction of GM-CSF from macrophages. Cell. Immunol. 2017, 320, 20–28. [Google Scholar] [CrossRef] [PubMed]
- Kwak, D.S.; Lee, O.Y.; Lee, K.N.; Jun, D.W.; Lee, H.L.; Yoon, B.C.; Choi, H.S. The Effect of DA-6034 on Intestinal Permeability in an Indomethacin- Induced Small Intestinal Injury Model. Gut Liver 2016, 10, 406–411. [Google Scholar] [CrossRef] [PubMed]
- Jacob, M.; Foster, R.; Sigthorsson, G.; Simpson, R.; Bjarnason, I. Role of bile in pathogenesis of indomethacin-induced enteropathy. Arch. Toxicol. 2007, 81, 291–298. [Google Scholar] [CrossRef]
- Whitfield-Cargile, C.M.; Cohen, N.D.; Chapkin, R.S.; Weeks, B.R.; Davidson, L.A.; Goldsby, J.S.; Hunt, C.L.; Steinmeyer, S.H.; Menon, R.; Suchodolski, J.S.; et al. The microbiota-derived metabolite indole decreases mucosal inflammation and injury in a murine model of NSAID enteropathy. Gut Microbes 2016, 7, 246–261. [Google Scholar] [CrossRef]
- Bertrand, V.; Guimbaud, R.; Tulliez, M.; Mauprivez, C.; Sogni, P.; Couturier, D.; Giroud, J.P.; Chaussade, S.; Chauvelot-Moachon, L. Increase in tumor necrosis factor-alpha production linked to the toxicity of indomethacin for the rat small intestine. Br. J. Pharm. 1998, 124, 1385–1394. [Google Scholar] [CrossRef]
- Saud, B.; Nandi, J.; Ong, G.; Finocchiaro, S.; Levine, R.A. Inhibition of TNF-alpha improves indomethacin-induced enteropathy in rats by modulating iNOS expression. Dig. Dis. Sci. 2005, 50, 1677–1683. [Google Scholar] [CrossRef]
- Watanabe, T.; Higuchi, K.; Kobata, A.; Nishio, H.; Tanigawa, T.; Shiba, M.; Tominaga, K.; Fujiwara, Y.; Oshitani, N.; Asahara, T.; et al. Non-steroidal anti-inflammatory drug-induced small intestinal damage is Toll-like receptor 4 dependent. Gut 2008, 57, 181–187. [Google Scholar] [CrossRef]
- Konaka, A.; Kato, S.; Tanaka, A.; Kunikata, T.; Korolkiewicz, R.; Takeuchi, K. Roles of enterobacteria, nitric oxide and neutrophil in pathogenesis of indomethacin-induced small intestinal lesions in rats. Pharm. Res. 1999, 40, 517–524. [Google Scholar] [CrossRef] [PubMed]
| Preclinical Model | Growth | Gut Hyperpermeability | Gut Inflammation | Ref. |
|---|---|---|---|---|
| Caloric Restriction (CR) | ||||
| −15% CR | Weight loss | n/a | Lower MCP-1 mRNA (liver/adipose tissue) | [14] |
| −25% CR | Weight loss | n/a | Thymus atrophy | [12] |
| −30% CR | Weight loss | No gut hyperpermeability | Decreased systemic TNFα levels | [13] |
| Intrauterine Undernutrition | ||||
| 8% of protein in gestational rats/20% in offspring | Low birth weight | Lower colonic ZO-1 mRNA expression | n/a | [18] |
| No hyperpermeability to LPS | [18] | |||
| Ex vivo colonic hyperpermeability to FSA | [19] | |||
| 20% in gestating sows/low birth weight | Low birth weight | Lower villus length ileum/duodenum | [20,21,22] | |
| 7.3% in gestating sows/20% for piglets | Low birth weight | Lower villus:crypt ratio in piglets | Lower IL-6 and TNF-α mRNA in offspring (ileum) | [20] |
| Maternal protein restriction or energy restriction (60% vs. CT) | Low birth weight | n/a | Lower C3, C4, IgG, and IgM concentration in plasma offspring | [23] |
| Decreased jejunal IL-2 and IL-6 mRNA expression in offspring | ||||
| Protein Energy Undernutrition | ||||
| 0% of protein | Weight loss | Lower jejunal villus length | No difference in plasmatic α-1-Acid Glycoprotein | [24] |
| 7% of protein | Weight loss | No villus atrophy/ | Decreased MCP-1 macrophages release in vitro | [25] |
| In vivo hyperpermeability | ||||
| Lower jejunal ZO-1 and higher claudin-2 mRNA | ||||
| 4% protein | Weight loss | No hyperpermeability to large molecules | n/a | [26] |
| Hyperpermeability to small molecules | n/a | |||
| Lower colonic and ileal occludin | n/a | |||
| n/a | Higher TNF-α, MCP-1 and IL1-β production (liver) | [27] | ||
| Leucopenia with higher systemic IL-10 production | [28] | |||
| Lower CD-4 and TLR-4/MD-2 (macrophages) | ||||
| Lower IL-6, TNF-a and IL1-B production by cultured cell from bone marrow, spleen and peritoneum after in vitro LPS treatment | ||||
| 2% protein | Weight loss | n/a | Higher MPO and LCN-2 production | [29] |
| Decreased leucocyte, peripheral lymphocyte, monocyte and polynuclear cells levels | [30] | |||
| Lower TLR-4 expression | [28] | |||
| Lower mRNA expression of TNF-α by macrophage in vitro | ||||
| Lower NF-κB activation in vitro | ||||
| 0.7% of protein | Weight loss | n/a | Increased monocytes and macrophages number in bone marrow and blood | [31] |
| Higher monocyte arginase expression | ||||
| RBD | Weight loss | Jejunal hyperpermeability | n/a | [32] |
| Decreased jejunal claudin-3 tight junction protein expression | ||||
| Jejunal villous, crypt atrophy | ||||
| Lower ileal basal short circuit current | [33] | |||
| Higher ileal claudin-2 and occludin mRNA expression | ||||
| Ileal crypt atrophy No villous length difference | ||||
| M8 | Weight loss | n/a | n/a | [34] |
| MAIZE | Weight loss | Small mucosal atrophy/shorter villi and crypt | n/a | [35,36] |
| Zinc deficiency | ||||
| Zinc deficiency | Weight loss | No impact on villi/crypt ratio | No inflammation | [37] |
| Decreased villi/crypt (EAEC -infected mice) | Decreased TNF-a, IL1-B and IL-6 | |||
| No ileal hyperpermeability | Decreased ileal neutrophil infiltration | [38] | ||
| No higher plasmatic endotoxin | Altered immune response to parasitic nematodes | |||
| Lower production of IL-4, Decreased level of IgE, IgG1 | ||||
| Lower eosinophils and impaired of antigen-presenting cells function | ||||
| Microbiota Transfer | ||||
| Cocktail of bacteria | Weight loss | Increased global intestinal permeability | Increased IL-6, MCP-1 (macrophages) | [25] |
| Villus and crypt atrophy | ||||
| IgA+ bacteria consortium + M8 in gnotobiotic mice | Higher weight loss | Villus and crypt atrophy | Bacterial translocation | [39] |
| Mucosal immune activation with neutrophils infiltration into lamina propria |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salameh, E.; Morel, F.B.; Zeilani, M.; Déchelotte, P.; Marion-Letellier, R. Animal Models of Undernutrition and Enteropathy as Tools for Assessment of Nutritional Intervention. Nutrients 2019, 11, 2233. https://doi.org/10.3390/nu11092233
Salameh E, Morel FB, Zeilani M, Déchelotte P, Marion-Letellier R. Animal Models of Undernutrition and Enteropathy as Tools for Assessment of Nutritional Intervention. Nutrients. 2019; 11(9):2233. https://doi.org/10.3390/nu11092233
Chicago/Turabian StyleSalameh, Emmeline, Fanny B. Morel, Mamane Zeilani, Pierre Déchelotte, and Rachel Marion-Letellier. 2019. "Animal Models of Undernutrition and Enteropathy as Tools for Assessment of Nutritional Intervention" Nutrients 11, no. 9: 2233. https://doi.org/10.3390/nu11092233
APA StyleSalameh, E., Morel, F. B., Zeilani, M., Déchelotte, P., & Marion-Letellier, R. (2019). Animal Models of Undernutrition and Enteropathy as Tools for Assessment of Nutritional Intervention. Nutrients, 11(9), 2233. https://doi.org/10.3390/nu11092233





