A Web-Based 24-H Dietary Recall Could Be a Valid Tool for the Indicative Assessment of Dietary Intake in Older Adults Living in Slovenia
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Study Sample
2.2. Data Collection
2.3. Dietary Assessment
2.4. OPEN Dietary Software
2.5. Statistical Analysis
3. Results
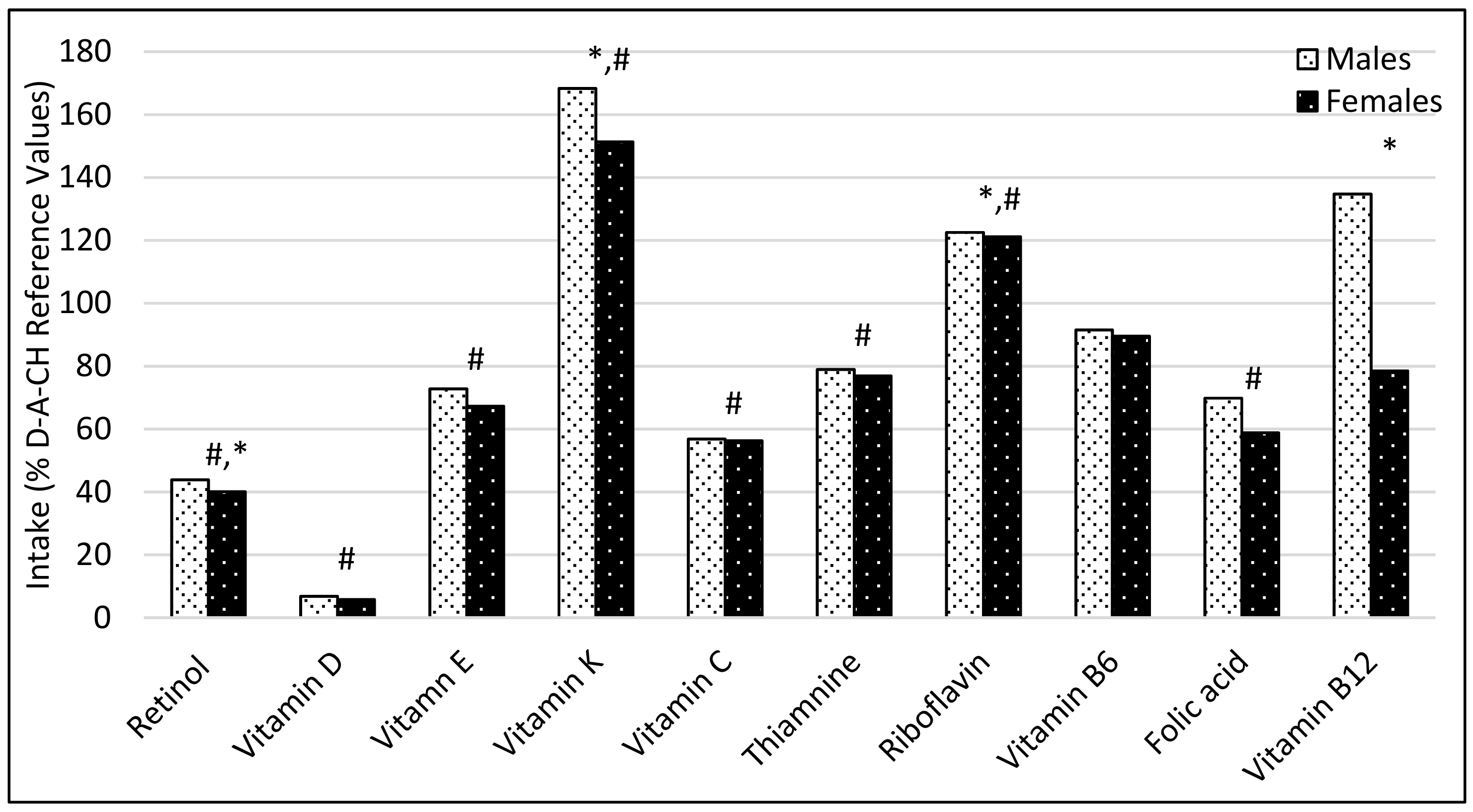
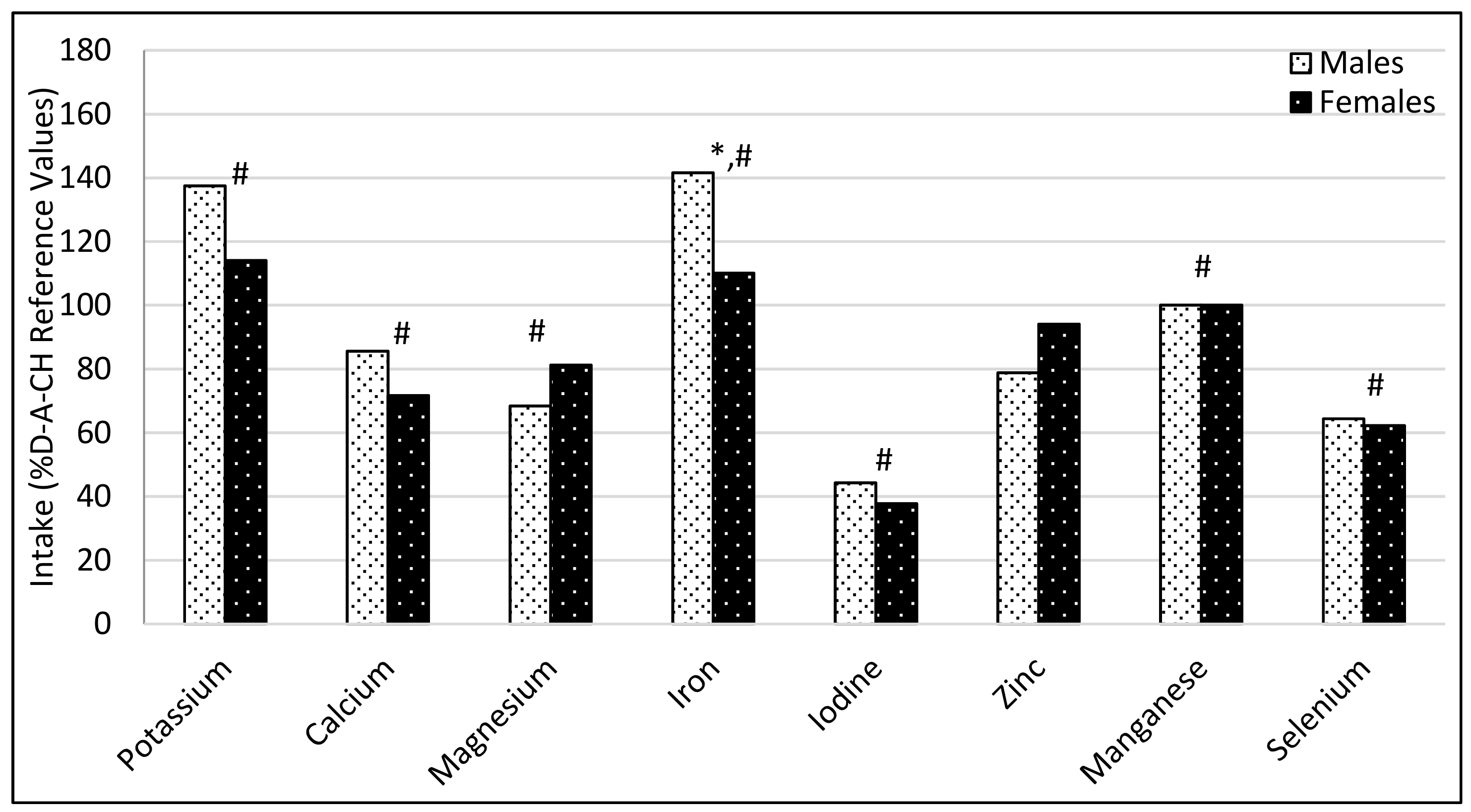
3.1. Comparison of Participant’s Dietary Intakes with the Recommendations
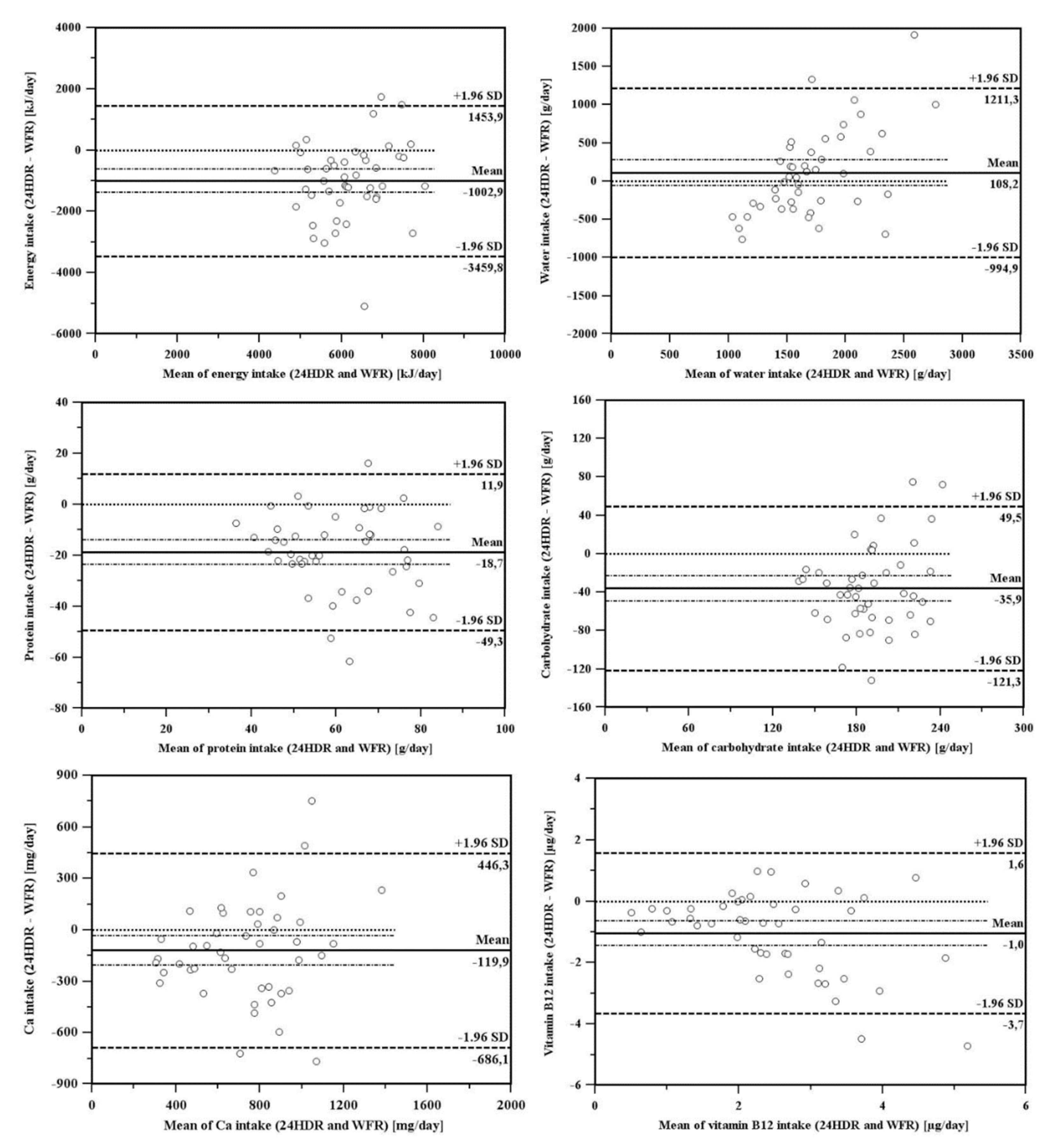
3.2. Comparison of Dietary Assessment Methods
4. Discussion
4.1. Comparison of Participant’s Dietary Intakes with the Recommendations
4.2. Comparison of Dietary Assessment Methods
4.3. Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- McNaughton, S.A.; Crawford, D.; Ball, K.; Salmon, J. Understanding determinants of nutrition, physical activity and quality of life among older adults: The Wellbeing, Eating and Exercise for a Long Life (WELL) study. Health Qual. Life Outcomes 2012, 10, 109. [Google Scholar] [CrossRef] [PubMed]
- Rotovnik Kozjek, N.; Situlin, R.; Gabrijelčič Blenkuš, M.; Mohorko, M.; Šimunič, B.; Pišot, R. Nutrition Recommendations for the Older Adults. The PANGeA Study; Annales: Koper, Slovenia, 2015; pp. 39–45. [Google Scholar]
- Vellas, B.; Lauque, S.; Andrieu, S.; Nourhashemi, F.; Rolland, Y.; Baumgartner, R.; Garry, P. Nutrition assessment in the elderly. Curr. Opin. Clin. Nutr. Metab. Care 2001, 4, 5–8. [Google Scholar] [CrossRef] [PubMed]
- Suominem, M.; Laine, T.; Routasalo, P.; Pitkala, K.H.; Rasanen, L. Nutrient content of served food, nutrient intake and nutritional status of residents with dementia in a Finnish nursing home. J. Nutr. Health Aging 2004, 8, 234–238. [Google Scholar] [PubMed]
- Volkert, D.; Beck, A.M.; Cederholm, T.; Cruz-Jentoft, A.; Goisser, S.; Hooper, L.; Kiesswetter, E.; Maggio, M.; Raynaud-Simon, A.; Sieber, C.C.; et al. ESPEN guideline on clinical nutrition and hydration in geriatrics. Clin. Nutr. 2019, 38, 10–47. [Google Scholar] [CrossRef] [PubMed]
- Mak, T.N.; Caldeira, S. The Role of Nutrition in Active and Healthy Ageing: For Prevention and Treatment of Age-Related Diseases: Evidence so Far; European Commission. Joint Research Centre, Institute for Health and Consumer Protection: Luxembourg, 2014; pp. 25–33. [Google Scholar]
- Volkert, D.; Schrader, E. Dietary assessment methods for older persons: What is the best approach? Curr. Opin. Clin. Nutr. Metab. Care 2013, 16, 534–540. [Google Scholar] [CrossRef] [PubMed]
- De Vries, J.H.M.; de Groot, L.C.P.G.M.; van Staveren, W.A. Dietary assessment in elderly people: Experiences gained from studies in the Netherlands. Eur. J. Clin. Nutr. 2009, 63, 69–74. [Google Scholar] [CrossRef] [PubMed]
- Schröder, H.; Covas, M.I.; Marrugat, J.; Vila, J.; Pena, A.; Alcántara, M.; Masiá, R. Use of a three-day estimated food record, a 72-h recall and a food-frequency questionnaire for dietary assessment in a Mediterranean Spanish population. Clin. Nutr. 2001, 20, 429–437. [Google Scholar] [CrossRef] [PubMed]
- De Keyzer, W.; Bracke, T.; McNaughton, S.A.; Parnell, W.; Moshfegh, A.J.; Pereira, R.A.; Lee, H.-S.; van’t Veer, P.; De Henauw, S.; Huybrechts, I. Cross-Continental Comparison of National Food Consumption Survey Methods—A Narrative Review. Nutrients 2015, 7, 3587–3620. [Google Scholar] [CrossRef]
- Bally, M.R.; Blaser Yildirim, P.Z.; Bounoure, L.; Gloy, V.L.; Mueller, B.; Briel, M.; Schuetz, P. Nutritional Support and Outcomes in Malnourished Medical Inpatients: A Systematic Review and Meta-analysis. JAMA Int. Med. 2016, 176, 43–53. [Google Scholar] [CrossRef]
- De Boer, E.J.; Slimani, N.; van ’t Veer, P.; Boeing, H.; Feinberg, M.; Leclercq, C.; Trolle, E.; Amiano, P.; Andersen, L.F.; Freisling, H.; et al. Rationale and methods of the European Food Consumption Validation (EFCOVAL) Project. Eur. J. Clin. Nutr. 2011, 65, 1–4. [Google Scholar] [CrossRef]
- European Food Safety Authority EFSA. Guidance on the EU Menu methodology. EFSA J. 2014, 12, 12. [Google Scholar] [CrossRef]
- Shim, J.S.; Oh, K.; Kim, H.C. Dietary assessment methods in epidemiologic studies. Epidemiol. Health 2014, 36. [Google Scholar] [CrossRef] [PubMed]
- Willett, W. Nutritional Epidemiology, 3rd ed.; Oxford University Press: New York, NY, USA, 2013; pp. 34–69. [Google Scholar]
- Holst, M.; Ofei, K.T.; Skadhauge, L.B.; Rasmussen, H.H.; Beermann, T. Monitoring of Nutrition Intake in Hospitalized Patients: Can We Rely on the Feasible Monitoring Systems? J. Clin. Nutr. Metab. 2017, 1, 1000102. [Google Scholar] [CrossRef]
- Jayawardena, R. Comparison dietary assessment methods in Sri Lankan adults: Use of 24-h dietary recall and 7-day weighed intake. BMC Nutr. 2016, 2, 18. [Google Scholar] [CrossRef]
- European Food Safety Authority EFSA. General principles for the collection of national food consumption data in the view of a pan-European dietary survey. EFSA J. 2009, 7. [Google Scholar] [CrossRef]
- Carlsen, M.H.; Lillegaard, I.T.; Karlsen, A.; Blomhoff, R.; Drevon, C.A.; Andersen, L.F. Evaluation of energy and dietary intake estimates from a food frequency questionnaire using independent energy expenditure measurement and weighed food records. Nutr. J. 2010, 9, 37. [Google Scholar] [CrossRef]
- Pišot, R.; Pegan, N.; Šimunič, B. Pangea: Physical activity and nutrition for great ageing. Ann. Kinesiol. 2012, 2, 201–205. [Google Scholar]
- Winter, J.; MacInnis, R.J.; Wattanapenpaiboon, N.; Nowson, C.A. BMI and all-cause mortality in older adults: A meta-analysis. Am. J. Clin. Nutr. 2014, 99, 875–890. [Google Scholar] [CrossRef]
- Ishii, Y.; Ishihara, J.; Takachi, R.; Shinozawa, Y.; Imaeda, N.; Goto, C.; Wakai, K.; Takahashi, T.; Iso, H.; Nakamura, K.; et al. Comparison of weighed food record procedures for the reference methods in two validation studies of food frequency questionnaires. J. Epidemiol. 2017, 27, 331–337. [Google Scholar] [CrossRef]
- Ortega, R.M.; Pérez-Rodrigo, C.; López-Sobaler, A.M. Dietary assessment methods: Dietary records. Nutr. Hosp. 2015, 31, 38–45. [Google Scholar] [CrossRef]
- Nightingale, H.; Walsh, K.J.; Olupot-Olupot, P.; Engoru, C.; Ssenyondo, T.; Nteziyaremye, J.; Amorut, D.; Nakuya, M.; Arimi, M.; Frost, G.; et al. Validation of triple pass 24-h dietary recall in Ugandan children by simultaneous weighed food assessment. BMC Nutr. 2016, 2, 56. [Google Scholar] [CrossRef] [PubMed]
- Koroušić Seljak, B. Computer-based dietary menu planning. J. Food Comp. Anal. 2009, 22, 414–420. [Google Scholar] [CrossRef]
- Koroušić Seljak, B.; Stibilj, V.; Pograjc, L.; Mis, N.F.; Benedik, E. Food composition databases for effective quality nutritional care. Food Chem. 2013, 140, 553–561. [Google Scholar] [CrossRef] [PubMed]
- Gregorič, M.; Turk Fajdiga, V.; Gabrijelčič Blenkuš, M. Slikovno Gradivo s Prikazom Velikosti Porcij; Inštitut za varovanje zdravja Republike Slovenije, IVZ Pub.: Ljubljana, Slovenia, 2013; Available online: https://www.nijz.si/sites/www.nijz.si/files/publikacije-datoteke/slikovno_gradivo_s_prikazom_velikosti_porcij.pdf (accessed on 21 November 2018).
- Computer Web-Based Software: The Open Platform for Clinical Nutrition (OPEN). Jozef Stefan Institute Pub. Available online: http://opkp.si/en_GB/cms/introduction/my-food-diary (accessed on 21 November 2018).
- Pandis, N. Comparison of 2 means for matched observations (paired t test) and t test assumptions. Am. J. Orthod. Dentofac. Orthop. 2015, 148, 515–516. [Google Scholar] [CrossRef] [PubMed]
- Hanneman, S.K. Design, Analysis and Interpretation of Method-Comparison Studies. AACN Adv. Crit. Care 2008, 19, 223–234. [Google Scholar] [CrossRef] [PubMed]
- Bland, J.M.; Altman, D.G. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986, 1, 307–310. [Google Scholar] [CrossRef]
- Deutsche Gesellschaft für Ernährung; Österreichische Gesellschaft für Ernährung; Schweizerische Gesellschaft für Ernährung. Referenzwerte Für Die Nährstoffzufuhr, 2nd ed.; DGE, ÖGE, SGE: Bonn, Germany, 2017. [Google Scholar]
- World Health Organization (WHO). Guideline: Sugars Intake for Adults and Children; World Health Organization: Geneva, Switzerland, 2015; pp. 12–17. [Google Scholar]
- World Health Organization (WHO). Guideline: Sodium Intake for Adults and Children; WHO: Geneva, Switzerland, 2012; pp. 1–3. [Google Scholar]
- Carlsson, M. Nutritional status, body composition and physical activity among older people living in residential care facilities. Ph.D. Thesis, Department of Community Medicine and Rehabilitation, Geriatric Medicine, Umeå Univerity, Umeå, Sweden, 2011; pp. 48–52. [Google Scholar]
- Nouri Saeidlou, S.; Merdol, T.; Mikaili, P.; Bektaş, Y. Assessment of the nutritional status of elderly people living at nursing homes in Northwest Iran. Int. J. Acad. Res. 2011, 3, 173–181. [Google Scholar]
- Gabrijelčič Blenkuš, M.; Gregorič, M.; Tivadar, B.; Koch, V.; Kostanjevec, S.; Fajdiga Turk, V.; Žalar, A.; Lavtar, D.; Kuhar, D.; Rozman, U. Prehrambene Navade Odraslih Prebivalcev Slovenije Z Vidika Varovanja Zdravja; Univerza v Ljubljani. Pedagoška fakulteta: Ljubljana, Slovenia, 2009; pp. 86–97. [Google Scholar]
- Baum, J.I.; Kim, I.Y.; Wolfe, R.R. Protein Consumption and the Elderly: What Is the Optimal Level of Intake? Nutrients 2016, 8, 259. [Google Scholar] [CrossRef]
- Volkert, D.; Kreuel, K.; Heseker, H.; Stehle, P. Energy and nutrient intake of young-old, old-old and very-old elderly in Germany. Eur. J. Clin. Nutr. 2004, 58, 1190–1200. [Google Scholar] [CrossRef]
- King, D.E.; Mainous, A.G.; Lambourne, C.A. Trends in dietary fiber intake in the United States, 1999–2008. J. Acad. Nutr. Diet. 2012, 112, 642–648. [Google Scholar] [CrossRef]
- Thomas, D.R.; Tariq, S.H.; Makhdomm, S.; Haddad, R.; Moinuddin, A. Physician misdiagnosis of dehydration in older adults. J. Am. Med. Dir. Assoc. 2003, 4, 31–34. [Google Scholar] [CrossRef]
- Ter Borg, S.; Verlaan, S.; Hemsworth, J.; Mijnarends, D.M.; Schols, J.M.; Luiking, Y.C.; de Groot, L.C. Micronutrient intakes and potential inadequacies of community-dwelling older adults: A systematic review. Br. J. Nutr. 2015, 113, 1195–1206. [Google Scholar] [CrossRef] [PubMed]
- Strasburg, A.; Eisinger-Watzl, M.; Krems, C.; Roth, A.; Hoffmann, I. Comparison of food consumption and nutrient intake assessed with three dietary assessment methods: Results of the German National Nutrition Survey II. Eur. J. Nutr. 2019, 58, 193–210. [Google Scholar] [CrossRef] [PubMed]
- De Keyzer, W.; Huybrechts, I.; De Vriendt, V.; Vandevijvere, S.; Slimani, N.; Van Oyen, H.; De Henauw, S. Repeated 24-h recalls versus dietary records for estimating nutrient intakes in a national food consumption survey. Food. Nutr. Res. 2011, 55. [Google Scholar] [CrossRef] [PubMed]
- Gibson, R.S.; Charrondiere, U.R.; Bell, W. Measurement Errors in Dietary Assessment Using Self-Reported 24-Hour Recalls in Low-Income Countries and Strategies for Their Prevention. Adv. Nutr. 2017, 8, 980–991. [Google Scholar] [CrossRef] [PubMed]
- Salvador Castell, G.; Serra-Majem, L.; Ribas-Barba, L. What and how much do we eat? 24-h dietary recall method. Nutr. Hosp. 2015, 31, 46–48. [Google Scholar] [PubMed]
- Ahmed, T.; Haboubi, N. Assessment and management of nutrition in older people and its importance to health. Clin. Interv. Aging 2010, 5, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Han, T.S.; Tajar, A.; Lean, M.E.J. Obesity and weight management in the elderly. Br. Med. Bull. 2011, 97, 169–196. [Google Scholar] [CrossRef] [PubMed]
- Peralta, M.; Ramos, M.; Lipert, A.; Martins, J.; Marques, A. Prevalence and trends of overweight and obesity in older adults from 10 European countries from 2005 to 2013. Scand. J. Public Health 2018, 46, 522–529. [Google Scholar] [CrossRef] [PubMed]
- Lainscak, M.; von Haehling, S.; Doehner, W.; Anker, S.D. The obesity paradox in chronic disease: Facts and numbers. J. Cachexia Sarcopenia Muscle 2012, 3, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Batsis, J.A.; Zagaria, A.B. Addressing Obesity in Aging Patients. Med. Clin. N. Am. 2018, 102, 65–85. [Google Scholar] [CrossRef] [PubMed]
- Probst, Y.C.; Tapsell, L.C. Overview of computerized dietary assessment programs for research and practice in nutrition education. J. Nutr. Educ. Behav. 2005, 37, 20–26. [Google Scholar] [CrossRef]
- Turck, D.; Braegger, C.P.; Colombo, C.; Declercq, D.; Morton, A.; Pancheva, R.; Robberecht, E.; Stern, M.; Strandvik, B.; Wolfe, S.; et al. ESPEN-ESPGHAN-ECFS guidelines on nutrition care for infants, children, and adults with cystic fibrosis. Clin. Nutr. 2016, 35, 557–577. [Google Scholar] [CrossRef] [PubMed]
- Ward, H.A.; McLellan, H.; Udeh-Momoh, C.; Giannakopoulou, P.; Robb, C.; Wark, P.A.; Middleton, L. Use of Online Dietary Recalls among Older UK Adults: A Feasibility Study of an Online Dietary Assessment Tool. Nutrients 2019, 11, 1451. [Google Scholar] [CrossRef] [PubMed]
| Gender, Age and BMI † | |
|---|---|
| Male (n) | 15 |
| Female (n) | 34 |
| Age (Mean, SD *) (years) | 75.2 (5.7) |
| Age range | 65–91 years |
| BMI a (kg/m2) (Mean, SD *) | 30.9 (6.7) |
| Unit | Male | Female | D-A-CH Reference Values [32] | ||
|---|---|---|---|---|---|
| Male | Female | ||||
| Energy ** | kJ | 6995 ± 844 | 6519 ± 1077 | 8790 | 7120 |
| Energy | kJ/kg body weight | 85 ± 12 | 86 ± 23 | / | / |
| Water **,† | mL | 1783 ± 315 | 1611 ± 337 | 2250 | 2250 |
| Proteins ** | g | 76 ± 14 * | 65 ± 15 * | 67 | 57 |
| Proteins ** | g/kg body weight | 0.9 ± 0.3 | 0.8 ± 0.3 | 1.0 | 1.0 |
| Proteins | % kJ | 18 ± 2 | 17 ± 2 | / | / |
| Carbohydrates | g | 210 ± 27 | 205 ± 35 | / | / |
| Carbohydrates ** | % kJ | 51 ± 5 | 54 ± 5 | >50 | >50 |
| Total Sugars ‡ | g | 65 ± 11 | 66 ± 23 | / | / |
| Total Sugars ‡ | % kJ | 16 ± 2 | 17 ± 6 | / | / |
| Dietary Fibre ** | g | 15 ± 3 | 16 ± 4 | >30 | >30 |
| Dietary Fibre ** | g/MJ | 2.2 ± 0.3 | 2.5 ± 0.4 | >3.1 | >3.9 |
| Total Fats | g | 57 ± 11 | 52 ± 13 | / | / |
| Total Fats | % kJ | 30 ± 4 | 29 ± 5 | <30 | <30 |
| Saturated Fatty Acids | g | 21 ± 5 | 18 ± 5 | / | / |
| Saturated Fatty Acids | % kJ | 11 ± 2 | 10 ± 2 | <10 | <10 |
| Monounsaturated Fatty Acids | g | 14 ± 3 | 12 ± 3 | / | / |
| Monounsaturated Fatty Acids ** | % kJ | 7 ± 2 | 7 ± 1 | >13 | >13 |
| n-3 Fatty Acids | g | 1 ± 0.4 | 0.8 ± 0.4 | / | / |
| n-3 Fatty Acids | % kJ | 0.5 ± 0.2 | 0.5 ± 0.2 | 0.5 | 0.5 |
| n-6 Fatty Acids | g | 5.2 ± 1.4 | 4.8 ± 2.1 | / | / |
| n-6 Fatty Acids | % kJ | 2.7 ± 0.7 | 2.7 ± 1.0 | 2.5 | 2.5 |
| Polyunsaturated Fatty Acids | g | 7 ± 2 | 7 ± 2 | / | / |
| Polyunsaturated Fatty Acids ** | % kJ | 4 ± 1 | 4 ± 1 | >7 | >7 |
| Cholesterol ** | mg | 264 ± 80 * | 206 ± 69 * | <300 | <300 |
| Unit | 3-Day WFR | 2 × 24HDR | Ratio 24HDR/WFR | Paired Samples t-Test | Pearson Correlation | |||
|---|---|---|---|---|---|---|---|---|
| t | p * | r | p ** | |||||
| Energy | kJ | 6665.2 ± 1026.9 | 5739.8 ± 1193.8 | 0.86 | 5.367 | 0.000 | 0.39 | 0.005 |
| Water † | ml | 1664.1 ± 336.6 | 1763.8 ± 585.3 | 1.06 | −1.290 | 0.204 | 0.42 | 0.003 |
| Proteins | g | 68.9 ± 15.5 | 51.3 ± 13.2 | 0.74 | 8.051 | 0.000 | 0.39 | 0.006 |
| Carbohydrates | g | 206.7 ± 32.4 | 173.8 ± 39.6 | 0.84 | 5.526 | 0.000 | 0.31 | 0.033 |
| Total Sugars ‡ | g | 65.9 ± 20.1 | 59.7 ± 27.6 | 0.91 | 1.869 | 0.068 | 0.56 | 0.000 |
| Dietary Fibre | g | 15.9 ± 3.8 | 14.2 ± 5.6 | 0.89 | 2.228 | 0.031 | 0.40 | 0.011 |
| Fats (Total) | g | 53.6 ± 12.6 | 50.9 ± 13.6 | 0.95 | 1.472 | 0.148 | 0.50 | 0.000 |
| Saturated Fatty Acids | g | 19.1 ± 5.4 | 18.5 ± 6.7 | 0.97 | 0.780 | 0.440 | 0.33 | 0.020 |
| Monounsaturated Fatty Acids | g | 12.6 ± 3.4 | 9.9 ± 4.1 | 0.79 | 4.528 | 0.000 | 0.36 | 0.012 |
| n-3 Fatty Acids | g | 0.9 ± 0.4 | 0.7 ± 0.4 | 0.78 | 3.381 | 0.002 | 0.39 | 0.006 |
| n-6 Fatty Acids | g | 4.9 ± 1.9 | 3.4 ± 1.8 | 0.69 | 5.688 | 0.000 | 0.47 | 0.002 |
| Polyunsaturated Fatty Acids | g | 6.9 ± 2.1 | 6.2 ± 3.8 | 0.90 | 1.228 | 0.226 | 0.09 | 0.544 |
| Cholesterol | mg | 224.4 ± 76.8 | 203.8 ± 80.6 | 0.91 | 1.593 | 0.118 | 0.34 | 0.016 |
| Vitamins | ||||||||
| Retinol | mg | 0.4 ± 0.1 | 0.4 ± 0.3 | 1.00 | −0.713 | 0.480 | 0.61 | 0.000 |
| Vitamin D | µg | 1.3 ± 0.7 | 1.1 ± 0.6 | 0.85 | 2.073 | 0.046 | 0.36 | 0.120 |
| Vitamin E | mg | 8.2 ± 3.4 | 6.4 ± 3.2 | 0.78 | 3.725 | 0.001 | 0.45 | 0.001 |
| Vitamin K | µg | 118.9 ± 57.2 | 176.4 ± 127.5 | 1.48 | −3.892 | 0.000 | 0.60 | 0.000 |
| Vitamin C | mg | 59.4 ± 22.1 | 61.0 ± 31.9 | 1.03 | -0.332 | 0.742 | 0.25 | 0.090 |
| Thiamine | mg | 1.0 ± 0.1 | 0.8 ± 0.2 | 0.80 | 4.380 | 0.000 | 0.38 | 0.008 |
| Riboflavin | mg | 1.4 ± 0.4 | 1.2 ± 0.5 | 0.86 | 4.147 | 0.000 | 0.58 | 0.000 |
| Vitamin B6 | mg | 1.1 ± 0.3 | 0.9 ± 0.3 | 0.82 | 4.114 | 0.000 | 0.15 | 0.338 |
| Folic acid | µg | 197.7 ± 54.6 | 194.4 ± 83.0 | 0.98 | 0.341 | 0.735 | 0.59 | 0.000 |
| Vitamin B12 | µg | 3.0 ± 1.4 | 2.0 ± 1.0 | 0.67 | 5.276 | 0.000 | 0.47 | 0.001 |
| Macro Elements | ||||||||
| Sodium | mg | 2134.9 ± 611.4 | 1745.8 ± 673.2 | 0.82 | 3.356 | 0.002 | 0.19 | 0.193 |
| Potassium | mg | 2526.8 ± 460.5 | 2154.5 ± 662.8 | 0.85 | 3.779 | 0.000 | 0.27 | 0.060 |
| Calcium | mg | 796.3 ± 265.5 | 686.6 ± 306.1 | 0.86 | 2.785 | 0.008 | 0.53 | 0.000 |
| Magnesium | mg | 251.1 ± 100.2 | 212.1 ± 80.4 | 0.84 | 2.820 | 0.007 | 0.36 | 0.012 |
| Micro and Trace Elements | ||||||||
| Iron | mg | 12.5 ± 3.1 | 8.8 ± 2.6 | 0.70 | 7.313 | 0.000 | 0.30 | 0.036 |
| Iodine | µg | 73.9 ± 22.4 | 81.8 ± 50.2 | 1.11 | −1.112 | 0.272 | 0.27 | 0.060 |
| Zinc | mg | 7.3 ± 1.9 | 5.5 ± 1.9 | 0.75 | 6.818 | 0.000 | 0.51 | 0.000 |
| Manganese | mg | 4.2 ± 1.6 | 3.1 ± 1.2 | 0.74 | 4.042 | 0.000 | 0.07 | 0.648 |
| Selenium | µg | 41.6 ± 12.6 | 36.6 ± 16 | 0.88 | 2.904 | 0.024 | 0.50 | 0.000 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gregorič, M.; Zdešar Kotnik, K.; Pigac, I.; Gabrijelčič Blenkuš, M. A Web-Based 24-H Dietary Recall Could Be a Valid Tool for the Indicative Assessment of Dietary Intake in Older Adults Living in Slovenia. Nutrients 2019, 11, 2234. https://doi.org/10.3390/nu11092234
Gregorič M, Zdešar Kotnik K, Pigac I, Gabrijelčič Blenkuš M. A Web-Based 24-H Dietary Recall Could Be a Valid Tool for the Indicative Assessment of Dietary Intake in Older Adults Living in Slovenia. Nutrients. 2019; 11(9):2234. https://doi.org/10.3390/nu11092234
Chicago/Turabian StyleGregorič, Matej, Katja Zdešar Kotnik, Igor Pigac, and Mojca Gabrijelčič Blenkuš. 2019. "A Web-Based 24-H Dietary Recall Could Be a Valid Tool for the Indicative Assessment of Dietary Intake in Older Adults Living in Slovenia" Nutrients 11, no. 9: 2234. https://doi.org/10.3390/nu11092234
APA StyleGregorič, M., Zdešar Kotnik, K., Pigac, I., & Gabrijelčič Blenkuš, M. (2019). A Web-Based 24-H Dietary Recall Could Be a Valid Tool for the Indicative Assessment of Dietary Intake in Older Adults Living in Slovenia. Nutrients, 11(9), 2234. https://doi.org/10.3390/nu11092234




