Dairy Fat Consumption and the Risk of Metabolic Syndrome: An Examination of the Saturated Fatty Acids in Dairy
Abstract
1. Introduction
2. Background of SFA Found in Dairy
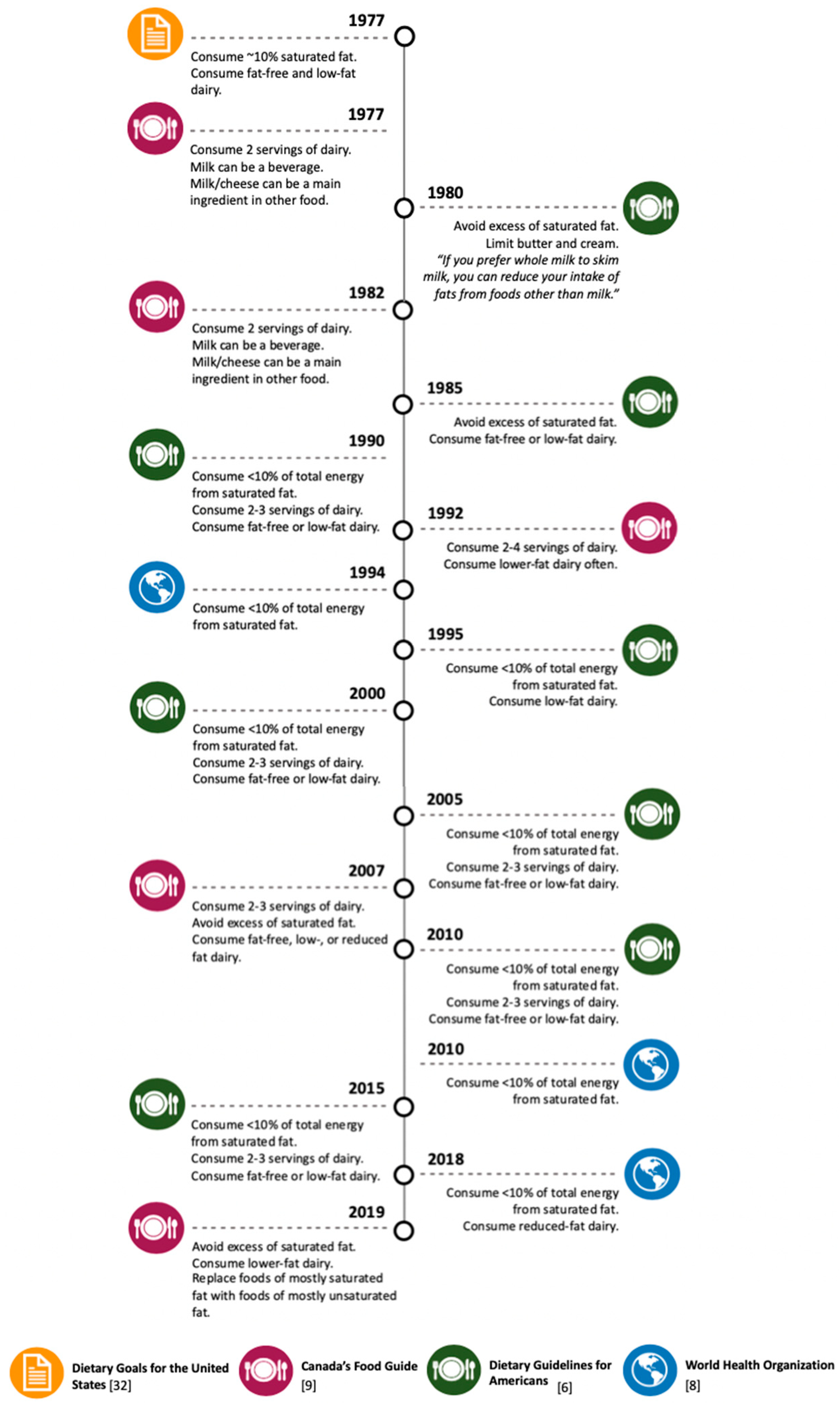
3. Dairy-Derived SFA Intake Recommendations in Dietary Guidelines
4. Epidemiological and Clinical Studies: The Effects of Dairy-Derived SFA on MetS or One of Its Four Risk Factors
4.1. Methods
4.2. MetS
4.3. Hyperglycemia
4.4. Obesity
4.5. Atherogenic Dyslipidemia
4.6. Hypertension
4.7. Limitations of Epidemiological and Clinical Studies Examining Dairy-Derived SFA Consumption and MetS
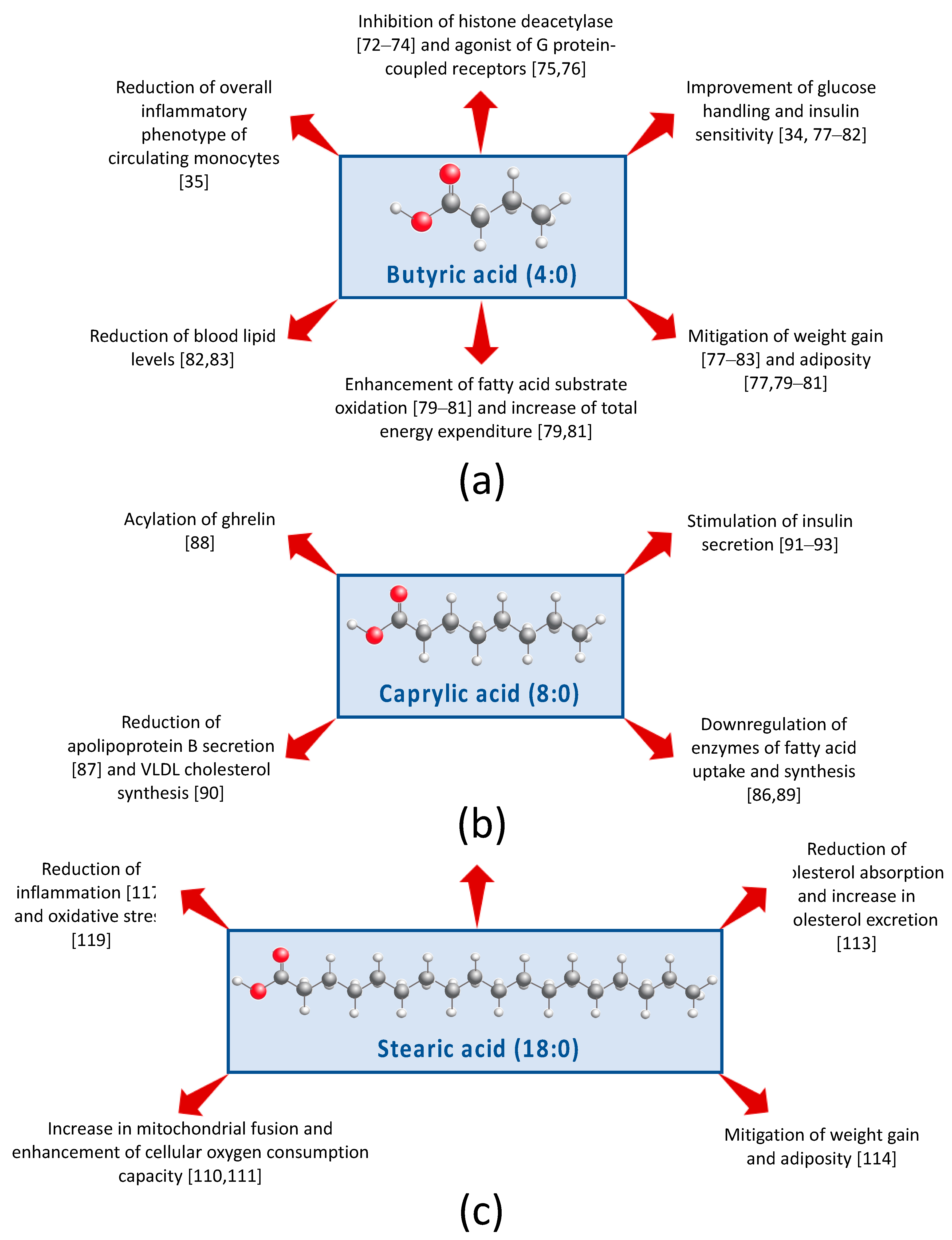
5. Potential Mechanisms Driving Differential Effects of SFA Found in Dairy on MetS Components
5.1. Short-Chain FA
5.2. Medium-Chain Fatty Acids
5.3. Long-Chain FA
5.4. Odd- and Branched-Chain FA
5.5. Limitations of Mechanistic Studies Examining Dairy-Derived SFA Consumption on MetS Components
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Metabolic Syndrome—Diagnosis and Treatment. Available online: https://www.mayoclinic.org/diseases-conditions/metabolic-syndrome/diagnosis-treatment/drc-20351921 (accessed on 3 April 2019).
- Metabolic Syndrome—National Heart, Lung, and Blood Institute (NHLBI). Available online: https://www.nhlbi.nih.gov/health-topics/metabolic-syndrome (accessed on 3 April 2019).
- IDF Consensus Worldwide Definition of the Metabolic Syndrome. Available online: https://www.idf.org/e-library/consensus-statements/60-idfconsensus-worldwide-definitionof-the-metabolic-syndrome (accessed on 3 April 2019).
- Devers, M.C.; Campbell, S.; Simmons, D. Influence of age on the prevalence and components of the metabolic syndrome and the association with cardiovascular disease. BMJ Open Diabetes Res. Care 2016, 4, e000195. [Google Scholar] [CrossRef] [PubMed]
- Moore, J.X.; Chaudhary, N.; Akinyemiju, T. Metabolic syndrome prevalence by race/ethnicity and sex in the United States, National Health and Nutrition Examination Survey, 1988–2012. Prev. Chronic Dis. 2017, 14, 160287. [Google Scholar] [CrossRef] [PubMed]
- Dietary Guidelines for Americans 2015–2020. Available online: https://health.gov/dietaryguidelines/2015/ (accessed on 20 May 2019).
- The American Heart Association’s Diet and Lifestyle Recommendations. Available online: http://www.heart.org/HEARTORG/HealthyLiving/HealthyEating/Nutrition/The-American-Heart-Associations-Diet-and-Lifestyle-eecommendations_UCM_305855_Article.jsp#.XKItpetKjOQ (accessed on 20 May 2019).
- Healthy Diet—World Health Organization. Available online: https://www.who.int/news-room/fact-sheets/detail/healthy-diet (accessed on 20 May 2019).
- Health Canada Canada’s Dietary Guidelines for Health Professionals and Policy Makers. Available online: https://food-guide.canada.ca/en/guidelines/ (accessed on 20 May 2019).
- Keys, A.; Anderson, J.T.; Grande, F. Serum cholesterol response to changes in the diet: IV. Particular saturated fatty acids in the diet. Metabolism 1965, 14, 776–787. [Google Scholar] [CrossRef]
- Houston, M. The relationship of saturated fats and coronary heart disease: Fa(c)t or fiction? A commentary. Ther. Adv. Cardiovasc. Dis. 2018, 12, 33–37. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, P.; Cheng, L.; Wang, J.; Zhang, Y.; Jiao, J. Saturated fatty acid intake is associated with total mortality in a nationwide cohort study. J. Nutr. 2019, 149, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Bainbridge, M.L.; Cersosimo, L.M.; Wright, A.-D. G.; Kraft, J. Content and composition of branched-chain fatty acids in bovine milk are affected by lactation stage and breed of dairy cow. PLoS ONE 2016, 11, e0150386. [Google Scholar] [CrossRef] [PubMed]
- Dubois, V.; Breton, S.; Linder, M.; Fanni, J.; Parmentier, M. Fatty acid profiles of 80 vegetable oils with regard to their nutritional potential. Eur. J. Lipid Sci. Technol. 2007, 109, 710–732. [Google Scholar] [CrossRef]
- Guo, Q.; Ye, A.; Bellissimo, N.; Singh, H.; Rousseau, D. Modulating fat digestion through food structure design. Prog. Lipid Res. 2017, 68, 109–118. [Google Scholar] [CrossRef]
- Aguilera, J.M. The food matrix: Implications in processing, nutrition and health. Crit. Rev. Food Sci. Nutr. 2018, 1–18. [Google Scholar] [CrossRef]
- Månsson, H.L. Fatty acids in bovine milk fat. Food Nutr. Res. 2008, 52. [Google Scholar] [CrossRef]
- Kim, Y.; Je, Y. Dairy consumption and risk of metabolic syndrome: A meta-analysis. Diabet. Med. 2016, 33, 428–440. [Google Scholar] [CrossRef] [PubMed]
- Rice, B.H. Dairy and cardiovascular disease: A review of recent observational research. Curr. Nutr. Rep. 2014, 3, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Babio, N.; Becerra-Tomás, N.; Martínez-González, M.Á.; Corella, D.; Estruch, R.; Ros, E.; Sayón-Orea, C.; Fitó, M.; Serra-Majem, L.; Arós, F.; et al. PREDIMED Investigators Consumption of yogurt, low-fat milk, and other low-fat dairy products is associated with lower risk of metabolic syndrome incidence in an elderly mediterranean population. J. Nutr. 2015, 145, 2308–2316. [Google Scholar] [CrossRef] [PubMed]
- Drehmer, M.; Pereira, M.A.; Schmidt, M.I.; Alvim, S.; Lotufo, P.A.; Luft, V.C.; Duncan, B.B. Total and full-fat, but not low-fat, dairy product intakes are inversely associated with metabolic syndrome in adults. J. Nutr. 2016, 146, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Parodi, P. Milk fat in human nutrition. Aust. J. Dairy Technol. 2004, 59, 3–8. [Google Scholar]
- Stergiadis, S.; Leifert, C.; Seal, C.J.; Eyre Larsen, M.K.; Nielsen, J.H.; Slots, T.; Butler, G. A 2-Year study on milk quality of three pasture-based dairy systems of contrasting production intensities in Wales. J. Agric. Sci. 2015, 153, 708–731. [Google Scholar] [CrossRef]
- O’Donnell-Megaro, A.M.; Barbano, D.M.; Bauman, D.E. Survey of the fatty acid composition of retail milk in the United States including regional and seasonal variations. J. Dairy Sci. 2011, 94, 59–65. [Google Scholar] [CrossRef]
- Food Composition Databases Show Foods—Whole Milk, UPC: 070852993669. Available online: https://ndb.nal.usda.gov/ndb/foods/show/45339816?fgcd=&manu=none&format=&count=&max=25&offset=&sort=default&order=asc&qlookup=whole+milk&ds=&qt=&qp=&qa=&qn=&q=&ing= (accessed on 23 May 2019).
- Bainbridge, M.L.; Egolf, E.; Barlow, J.W.; Alvez, J.P.; Roman, J.; Kraft, J. Milk from cows grazing on cool-season pastures provides an enhanced profile of bioactive fatty acids compared to those grazed on a monoculture of pearl millet. Food Chem. 2017, 217, 750–755. [Google Scholar] [CrossRef]
- Harfoot, C.; Hazelwood, G. Lipid metabolism in the rumen. In The Rumen Microbial Ecosystem; Hobson, P., Stewart, C., Eds.; Blackie Academic & Professional: London, UK, 1997; pp. 382–426. [Google Scholar]
- Jensen, R.G. The composition of bovine milk lipids: January 1995 to December 2000. J. Dairy Sci. 2002, 85, 295–350. [Google Scholar] [CrossRef]
- Wolk, A.; Vessby, B.; Ljung, H.; Barrefors, P. Evaluation of a biological marker of dairy fat intake. Am. J. Clin. Nutr. 1998, 68, 291–295. [Google Scholar] [CrossRef]
- Risérus, U.; Marklund, M. Milk fat biomarkers and cardiometabolic disease. Curr. Opin. Lipidol. 2016, 28, 1. [Google Scholar] [CrossRef] [PubMed]
- Page, I.H.; Allen, E.V.; Chamberlain, F.L.; Keys, A.; Stamler, J.; Stare, F.J. Dietary fat and its relation to heart attacks and strokes. Circulation 1961, 23, 133–136. [Google Scholar] [CrossRef]
- Dietary Goals for the United States. Available online: https://thescienceofnutrition.files.wordpress.com/2014/03/dietary-goals-for-the-united-states.pdf (accessed on 20 May 2019).
- Thorning, T.K.; Bertram, H.C.; Bonjour, J.-P.; de Groot, L.; Dupont, D.; Feeney, E.; Ipsen, R.; Lecerf, J.M.; Mackie, A.; McKinley, M.C.; et al. Whole dairy matrix or single nutrients in assessment of health effects: Current evidence and knowledge gaps. Am. J. Clin. Nutr. 2017, 105, 1033–1045. [Google Scholar] [CrossRef] [PubMed]
- Bouter, K.; Bakker, G.J.; Levin, E.; Hartstra, A.V.; Kootte, R.S.; Udayappan, S.D.; Katiraei, S.; Bahler, L.; Gilijamse, P.W.; Tremaroli, V.; et al. Differential metabolic effects of oral butyrate treatment in lean versus metabolic syndrome subjects. Clin. Transl. Gastroenterol. 2018, 9, 155. [Google Scholar] [CrossRef] [PubMed]
- Cleophas, M.C.P.; Ratter, J.M.; Bekkering, S.; Quintin, J.; Schraa, K.; Stroes, E.S.; Netea, M.G.; Joosten, L.A.B. Effects of oral butyrate supplementation on inflammatory potential of circulating peripheral blood mononuclear cells in healthy and obese males. Sci. Rep. 2019, 9, 775. [Google Scholar] [CrossRef] [PubMed]
- Mayneris-Perxachs, J.; Guerendiain, M.; Castellote, A.I.; Estruch, R.; Covas, M.I.; Fitó, M.; Salas-Salvadó, J.; Martínez-González, M.A.; Aros, F.; Lamuela-Raventós, R.M.; et al. Plasma fatty acid composition, estimated desaturase activities, and their relation with the metabolic syndrome in a population at high risk of cardiovascular disease. Clin. Nutr. 2014, 33, 90–97. [Google Scholar] [CrossRef] [PubMed]
- Kratz, M.; Baars, T.; Guyenet, S. The relationship between high-fat dairy consumption and obesity, cardiovascular, and metabolic disease. Eur. J. Nutr. 2013, 52, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Rautiainen, S.; Wang, L.; Lee, I.-M.; Manson, J.E.; Buring, J.E.; Sesso, H.D. Dairy consumption in association with weight change and risk of becoming overweight or obese in middle-aged and older women: A prospective cohort study. Am. J. Clin. Nutr. 2016, 103, 979–988. [Google Scholar] [CrossRef] [PubMed]
- Reaven, G.M. Banting lecture 1988: Role of insulin resistance in human disease. Diabetes 1988, 37, 1595–1607. [Google Scholar] [CrossRef] [PubMed]
- Laakso, M.; Kuusisto, J. Insulin resistance and hyperglycaemia in cardiovascular disease development. Nat. Rev. Endocrinol. 2014, 10, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Santaren, I.D.; Watkins, S.M.; Liese, A.D.; Wagenknecht, L.E.; Rewers, M.J.; Haffner, S.M.; Lorenzo, C.; Hanley, A.J. Serum pentadecanoic acid (15:0), a short-term marker of dairy food intake, is inversely associated with incident type 2 diabetes and its underlying disorders. Am. J. Clin. Nutr. 2014, 100, 1532–1540. [Google Scholar] [CrossRef] [PubMed]
- Iggman, D.; Ärnlöv, J.; Vessby, B.; Cederholm, T.; Sjögren, P.; Risérus, U. Adipose tissue fatty acids and insulin sensitivity in elderly men. Diabetologia 2010, 53, 850–857. [Google Scholar] [CrossRef] [PubMed]
- Wanders, A.J.; Alssema, M.; de Koning, E.J.P.; le Cessie, S.; de Vries, J.H.; Zock, P.L.; Rosendaal, F.R.; den Heijer, M.; de Mutsert, R. Fatty acid intake and its dietary sources in relation with markers of type 2 diabetes risk: The NEO study. Eur. J. Clin. Nutr. 2017, 71, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Drehmer, M.; Pereira, M.A.; Schmidt, M.I.; Molina, M.D.C.B.; Alvim, S.; Lotufo, P.A.; Duncan, B.B. Associations of dairy intake with glycemia and insulinemia, independent of obesity, in Brazilian adults: The Brazilian Longitudinal Study of Adult Health (ELSA-Brasil). Am. J. Clin. Nutr. 2015, 101, 775–782. [Google Scholar] [CrossRef] [PubMed]
- Kratz, M.; Marcovina, S.; Nelson, J.E.; Yeh, M.M.; Kowdley, K.V.; Callahan, H.S.; Song, X.; Di, C.; Utzschneider, K.M. Dairy fat intake is associated with glucose tolerance, hepatic and systemic insulin sensitivity, and liver fat but not β-cell function in humans. Am. J. Clin. Nutr. 2014, 99, 1385–1396. [Google Scholar] [CrossRef]
- Bohl, M.; Bjørnshave, A.; Larsen, M.K.; Gregersen, S.; Hermansen, K. The effects of proteins and medium-chain fatty acids from milk on body composition, insulin sensitivity and blood pressure in abdominally obese adults. Eur. J. Clin. Nutr. 2017, 71, 76–82. [Google Scholar] [CrossRef]
- Bohl, M.; Bjørnshave, A.; Rasmussen, K.V.; Schioldan, A.G.; Amer, B.; Larsen, M.K.; Dalsgaard, T.K.; Holst, J.J.; Herrmann, A.; O’Neill, S.; et al. Dairy proteins, dairy lipids, and postprandial lipemia in persons with abdominal obesity (DairyHealth): A 12-wk, randomized, parallel-controlled, double-blinded, diet intervention study. Am. J. Clin. Nutr. 2015, 101, 870–878. [Google Scholar] [CrossRef]
- Werner, L.B.; Hellgren, L.I.; Raff, M.; Jensen, S.K.; Petersen, R.A.; Drachmann, T.; Tholstrup, T. Effects of butter from mountain-pasture grazing cows on risk markers of the metabolic syndrome compared with conventional Danish butter: A randomized controlled study. Lipids Health Dis. 2013, 12, 99. [Google Scholar] [CrossRef]
- Wennersberg, M.H.; Smedman, A.; Turpeinen, A.M.; Retterstøl, K.; Tengblad, S.; Lipre, E.; Aro, A.; Mutanen, P.; Seljeflot, I.; Basu, S.; et al. Dairy products and metabolic effects in overweight men and women: Results from a 6-mo intervention study. Am. J. Clin. Nutr. 2009, 90, 960–968. [Google Scholar] [CrossRef]
- Bjermo, H.; Iggman, D.; Kullberg, J.; Dahlman, I.; Johansson, L.; Persson, L.; Berglund, J.; Pulkki, K.; Basu, S.; Uusitupa, M.; et al. Effects of n-6 PUFAs compared with SFAs on liver fat, lipoproteins, and inflammation in abdominal obesity: A randomized controlled trial. Am. J. Clin. Nutr. 2012, 95, 1003–1012. [Google Scholar] [CrossRef]
- Iggman, D.; Gustafsson, I.-B.; Berglund, L.; Vessby, B.; Marckmann, P.; Risérus, U. Replacing dairy fat with rapeseed oil causes rapid improvement of hyperlipidaemia: A randomized controlled study. J. Intern. Med. 2011, 270, 356–364. [Google Scholar] [CrossRef] [PubMed]
- Engin, A. The definition and prevalence of obesity and metabolic syndrome. Adv. Exp. Med. Biol. 2017, 960, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Kwon, H.; Kim, D.; Kim, J.S. Body fat distribution and the risk of incident metabolic syndrome: A longitudinal cohort study. Sci. Rep. 2017, 7, 10955. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Fox, C.S.; Hickson, D.A.; May, W.D.; Hairston, K.G.; Carr, J.J.; Taylor, H.A. Impact of abdominal visceral and subcutaneous adipose tissue on cardiometabolic risk factors: The Jackson Heart Study. J. Clin. Endocrinol. Metab. 2010, 95, 5419–5426. [Google Scholar] [CrossRef] [PubMed]
- Kaess, B.M.; Pedley, A.; Massaro, J.M.; Murabito, J.; Hoffmann, U.; Fox, C.S. The ratio of visceral to subcutaneous fat, a metric of body fat distribution, is a unique correlate of cardiometabolic risk. Diabetologia 2012, 55, 2622–2630. [Google Scholar] [CrossRef] [PubMed]
- Venkatramanan, S.; Joseph, S.V.; Chouinard, P.Y.; Jacques, H.; Farnworth, E.R.; Jones, P.J.H. Milk enriched with conjugated linoleic acid fails to alter blood lipids or body composition in moderately overweight, borderline hyperlipidemic individuals. J. Am. Coll. Nutr. 2010, 29, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Pintus, S.; Murru, E.; Carta, G.; Cordeddu, L.; Batetta, B.; Accossu, S.; Pistis, D.; Uda, S.; Elena Ghiani, M.; Mele, M.; et al. Sheep cheese naturally enriched in α-linolenic, conjugated linoleic and vaccenic acids improves the lipid profile and reduces anandamide in the plasma of hypercholesterolaemic subjects. Br. J. Nutr. 2013, 109, 1453–1462. [Google Scholar] [CrossRef] [PubMed]
- Matualatupauw, J.C.; Bohl, M.; Gregersen, S.; Hermansen, K.; Afman, L.A. Dietary medium-chain saturated fatty acids induce gene expression of energy metabolism-related pathways in adipose tissue of abdominally obese subjects. Int. J. Obes. 2017, 41, 1348. [Google Scholar] [CrossRef] [PubMed]
- Arsenault, B.J.; Boekholdt, S.M.; Kastelein, J.J.P. Lipid parameters for measuring risk of cardiovascular disease. Nat. Rev. Cardiol. 2011, 8, 197–206. [Google Scholar] [CrossRef] [PubMed]
- Intorre, F.; Foddai, M.S.; Azzini, E.; Martin, B.; Montel, M.-C.; Catasta, G.; Toti, E.; Finotti, E.; Palomba, L.; Venneria, E.; et al. Differential effect of cheese fatty acid composition on blood lipid profile and redox status in normolipidemic volunteers: A pilot study. Int. J. Food Sci. Nutr. 2011, 62, 660–669. [Google Scholar] [CrossRef] [PubMed]
- Malpuech-Brugère, C.; Mouriot, J.; Boue-Vaysse, C.; Combe, N.; Peyraud, J.-L.; LeRuyet, P.; Chesneau, G.; Morio, B.; Chardigny, J.-M. Differential impact of milk fatty acid profiles on cardiovascular risk biomarkers in healthy men and women. Eur. J. Clin. Nutr. 2010, 64, 752–759. [Google Scholar] [CrossRef] [PubMed]
- Amer, B.; Clausen, M.R.; Bertram, H.C.; Bohl, M.; Nebel, C.; Zheng, H.; Skov, T.; Larsen, M.K.; Gregersen, S.; Hermansen, K.; et al. Consumption of whey in combination with dairy medium-chain fatty acids (MCFAs) may reduce lipid storage due to urinary loss of tricarboxylic acid cycle intermediates and increased rates of MCFAs oxidation. Mol. Nutr. Food Res. 2017, 61, 1601048. [Google Scholar] [CrossRef] [PubMed]
- Hodson, L.; Skeaff, C.M.; Fielding, B.A. Fatty acid composition of adipose tissue and blood in humans and its use as a biomarker of dietary intake. Prog. Lipid Res. 2008, 47, 348–380. [Google Scholar] [CrossRef] [PubMed]
- White, P. Fatty acids in oilseeds (vegetable oils). In Fatty Acids in Foods and Their Health Implications; Chow, C.K., Ed.; CRC Press: Boca Raton, FL, USA, 2008; pp. 227–301. ISBN 9780429127557. [Google Scholar]
- Ackman, R. Fatty acids in fish and shellfish. In Fatty Acids in Foods and Their Health Implications; Chow, C., Ed.; CRC Press: Boca Raton, FL, USA, 2008; pp. 155–185. ISBN 9780429127557. [Google Scholar]
- Bruckner, G.; Peng, A. Vegetables and vegetable products fatty acids. In Fatty Acids in Foods and Their Health Implications; Chow, C.K., Ed.; CRC Press: Boca Raton, FL, USA, 2008; pp. 187–226. ISBN 9780429127557. [Google Scholar]
- Özogul, Y.; Özogul, F.; Çi˙çek, E.; Polat, A.; Kuley, E. Fat content and fatty acid compositions of 34 marine water fish species from the Mediterranean Sea. Int. J. Food Sci. Nutr. 2009, 60, 464–475. [Google Scholar] [CrossRef] [PubMed]
- Sebedio, J.L.; Ackman, R.G. Some minor fatty acids of rapeseed oils. J. Am. Oil Chem. Soc. 1979, 56, 15–21. [Google Scholar] [CrossRef]
- Jenkins, B.; Aoun, M.; Feillet-Coudray, C.; Coudray, C.; Ronis, M.; Koulman, A. The dietary total-fat content affects the in vivo circulating C15:0 and C17:0 fatty acid levels independently. Nutrients 2018, 10, 1646. [Google Scholar] [CrossRef] [PubMed]
- Ratnayake, W.M.N. Concerns about the use of 15:0, 17:0, and trans-16:1n-7 as biomarkers of dairy fat intake in recent observational studies that suggest beneficial effects of dairy food on incidence of diabetes and stroke. Am. J. Clin. Nutr. 2015, 101, 1102–1103. [Google Scholar] [CrossRef] [PubMed]
- Fellows, R.; Denizot, J.; Stellato, C.; Cuomo, A.; Jain, P.; Stoyanova, E.; Balázsi, S.; Hajnády, Z.; Liebert, A.; Kazakevych, J.; et al. Microbiota derived short chain fatty acids promote histone crotonylation in the colon through histone deacetylases. Nat. Commun. 2018, 9, 105. [Google Scholar] [CrossRef] [PubMed]
- Candido, E.P.M.; Reeves, R.; Davie, J.R. Sodium butyrate inhibits histone deacetylation in cultured cells. Cell 1978, 14, 105–113. [Google Scholar] [CrossRef]
- Li, H.; Gao, Z.; Zhang, J.; Ye, X.; Xu, A.; Ye, J.; Jia, W. Sodium butyrate stimulates expression of fibroblast growth factor 21 in liver by inhibition of histone deacetylase 3. Diabetes 2012, 61, 797–806. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.J.; Goldsworthy, S.M.; Barnes, A.A.; Eilert, M.M.; Tcheang, L.; Daniels, D.; Muir, A.I.; Wigglesworth, M.J.; Kinghorn, I.; Fraser, N.J.; et al. The Orphan G protein-coupled receptors GPR41 and GPR43 are activated by propionate and other short chain carboxylic acids. J. Biol. Chem. 2003, 278, 11312–11319. [Google Scholar] [CrossRef] [PubMed]
- Stilling, R.M.; van de Wouw, M.; Clarke, G.; Stanton, C.; Dinan, T.G.; Cryan, J.F. The neuropharmacology of butyrate: The bread and butter of the microbiota-gut-brain axis? Neurochem. Int. 2016, 99, 110–132. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Yi, C.-X.; Katiraei, S.; Kooijman, S.; Zhou, E.; Chung, C.K.; Gao, Y.; van den Heuvel, J.K.; Meijer, O.C.; Berbée, J.F.P.; et al. Butyrate reduces appetite and activates brown adipose tissue via the gut-brain neural circuit. Gut 2018, 67, 1269–1279. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.V.; Frassetto, A.; Kowalik, E.J., Jr.; Nawrocki, A.R.; Lu, M.M.; Kosinski, J.R.; Hubert, J.A.; Szeto, D.; Yao, X.; Forrest, G.; et al. Butyrate and propionate protect against diet-induced obesity and regulate gut hormones via free fatty acid receptor 3-independent mechanisms. PLoS ONE 2012, 7, e35240. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Yin, J.; Zhang, J.; Ward, R.E.; Martin, R.J.; Lefevre, M.; Cefalu, W.T.; Ye, J. Butyrate improves insulin sensitivity and increases energy expenditure in mice. Diabetes 2009, 58, 1509. [Google Scholar] [CrossRef] [PubMed]
- Henagan, T.M.; Stefanska, B.; Fang, Z.; Navard, A.M.; Ye, J.; Lenard, N.R.; Devarshi, P.P. Sodium butyrate epigenetically modulates high-fat diet-induced skeletal muscle mitochondrial adaptation, obesity and insulin resistance through nucleosome positioning. Br. J. Pharmacol. 2015, 172, 2782–2798. [Google Scholar] [CrossRef]
- Den Besten, G.; Bleeker, A.; Gerding, A.; van Eunen, K.; Havinga, R.; van Dijk, T.H.; Oosterveer, M.H.; Jonker, J.W.; Groen, A.K.; Reijngoud, D.-J.; et al. Short-chain fatty acids protect against high-fat diet-induced obesity via a PPARγ-dependent switch from lipogenesis to fat oxidation. Diabetes 2015, 64, 2398–2408. [Google Scholar] [CrossRef]
- Li, H.-P.; Chen, X.; Li, M.-Q. Butyrate alleviates metabolic impairments and protects pancreatic β cell function in pregnant mice with obesity. Int. J. Clin. Exp. Pathol. 2013, 6, 1574–1584. [Google Scholar]
- Lu, Y.; Fan, C.; Li, P.; Lu, Y.; Chang, X.; Qi, K. Short chain fatty acids prevent high-fat-diet-induced obesity in mice by regulating G protein-coupled receptors and gut microbiota. Sci. Rep. 2016, 6, 37589. [Google Scholar] [CrossRef]
- Aguilar, E.C.; Leonel, A.J.; Teixeira, L.G.; Silva, A.R.; Silva, J.F.; Pelaez, J.M.N.; Capettini, L.S.A.; Lemos, V.S.; Santos, R.A.S.; Alvarez-Leite, J.I. Butyrate impairs atherogenesis by reducing plaque inflammation and vulnerability and decreasing NFκB activation. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 606–613. [Google Scholar] [CrossRef]
- Luu, M.; Pautz, S.; Kohl, V.; Singh, R.; Romero, R.; Lucas, S.; Hofmann, J.; Raifer, H.; Vachharajani, N.; Carrascosa, L.C.; et al. The short-chain fatty acid pentanoate suppresses autoimmunity by modulating the metabolic-epigenetic crosstalk in lymphocytes. Nat. Commun. 2019, 10, 760. [Google Scholar] [CrossRef] [PubMed]
- Akpa, M.M.; Point, F.; Sawadogo, S.; Radenne, A.; Mounier, C. Inhibition of insulin and T3-induced fatty acid synthase by hexanoate. Lipids 2010, 45, 997–1009. [Google Scholar] [CrossRef] [PubMed]
- Sato, K.; Cho, Y.; Tachibana, S.; Chiba, T.; Schneider, W.J.; Akiba, Y. Impairment of VLDL secretion by medium-chain fatty acids in chicken primary hepatocytes is affected by the chain length. J. Nutr. 2005, 135, 1636–1641. [Google Scholar] [CrossRef] [PubMed]
- Kojima, M.; Hosoda, H.; Date, Y.; Nakazato, M.; Matsuo, H.; Kangawa, K. Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature 1999, 402, 656–660. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Xie, W.; Han, J. Modulation of adipocyte lipogenesis by octanoate: Involvement of reactive oxygen species. Nutr. Metab. (Lond) 2006, 3, 30. [Google Scholar] [CrossRef] [PubMed]
- Tachibana, S.; Sato, K.; Cho, Y.; Chiba, T.; Schneider, W.J.; Akiba, Y. Octanoate reduces very low-density lipoprotein secretion by decreasing the synthesis of apolipoprotein B in primary cultures of chicken hepatocytes. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2005, 1737, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Stein, D.T.; Stevenson, B.E.; Chester, M.W.; Basit, M.; Daniels, M.B.; Turley, S.D.; McGarry, J.D. The insulinotropic potency of fatty acids is influenced profoundly by their chain length and degree of saturation. J. Clin. Investig. 1997, 100, 398–403. [Google Scholar] [CrossRef] [PubMed]
- Leem, J.; Shim, H.; Cho, H.; Park, J.-H. Octanoic acid potentiates glucose-stimulated insulin secretion and expression of glucokinase through the olfactory receptor in pancreatic β-cells. Biochem. Biophys. Res. Commun. 2018, 503, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Munakata, Y.; Yamada, T.; Imai, J.; Takahashi, K.; Tsukita, S.; Shirai, Y.; Kodama, S.; Asai, Y.; Sugisawa, T.; Chiba, Y.; et al. Olfactory receptors are expressed in pancreatic β-cells and promote glucose-stimulated insulin secretion. Sci. Rep. 2018, 8, 1499. [Google Scholar] [CrossRef] [PubMed]
- Malapaka, R.R. V.; Khoo, S.; Zhang, J.; Choi, J.H.; Zhou, X.E.; Xu, Y.; Gong, Y.; Li, J.; Yong, E.-L.; Chalmers, M.J.; et al. Identification and mechanism of 10-carbon fatty acid as modulating ligand of peroxisome proliferator-activated receptors. J. Biol. Chem. 2012, 287, 183–195. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, A.; Ghosh, M. Comparison of native and capric acid-enriched mustard oil effects on oxidative stress and antioxidant protection in rats. Br. J. Nutr. 2012, 107, 845–849. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Fu, J.; Li, L.; Gong, D.; Wen, X.; Yu, P.; Zeng, Z. Medium-chain fatty acid reduces lipid accumulation by regulating expression of lipid-sensing genes in human liver cells with steatosis. Int. J. Food Sci. Nutr. 2016, 67, 288–297. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.I.; Kang, K.S. Function of capric acid in cyclophosphamide-induced intestinal inflammation, oxidative stress, and barrier function in pigs. Sci. Rep. 2017, 7, 16530. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Checklin, H.L.; Wishart, J.M.; Stevens, J.E.; Jones, K.L.; Horowitz, M.; Meyer, J.H.; Rayner, C.K. A randomised trial of enteric-coated nutrient pellets to stimulate gastrointestinal peptide release and lower glycaemia in type 2 diabetes. Diabetologia 2013, 56, 1236–1242. [Google Scholar] [CrossRef] [PubMed]
- Alves, N.F.B.; de Queiroz, T.M.; de Almeida Travassos, R.; Magnani, M.; de Andrade Braga, V. Acute treatment with lauric acid reduces blood pressure and oxidative stress in spontaneously hypertensive rats. Basic Clin. Pharmacol. Toxicol. 2017, 120, 348–353. [Google Scholar] [CrossRef] [PubMed]
- Ong, M.-H.-L.; Wong, H.-K.; Tengku-Muhammad, T.-S.; Choo, Q.-C.; Chew, C.-H. Pro-atherogenic proteoglycanase ADAMTS-1 is down-regulated by lauric acid through PI3K and JNK signaling pathways in THP-1 derived macrophages. Mol. Biol. Rep. 2019, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Maurer-Stroh, S.; Gouda, M.; Novatchkova, M.; Schleiffer, A.; Schneider, G.; Sirota, F.L.; Wildpaner, M.; Hayashi, N.; Eisenhaber, F. MYRbase: Analysis of genome-wide glycine myristoylation enlarges the functional spectrum of eukaryotic myristoylated proteins. Genome Biol. 2004, 5, R21. [Google Scholar] [CrossRef]
- Ren, X.-M.; Cao, L.-Y.; Zhang, J.; Qin, W.-P.; Yang, Y.; Wan, B.; Guo, L.-H. Investigation of the binding interaction of fatty acids with human G protein-coupled receptor 40 using a site-specific fluorescence probe by flow cytometry. Biochemistry 2016, 55, 1989–1996. [Google Scholar] [CrossRef]
- Snook, J.T.; Park, S.; Williams, G.; Tsai, Y.-H.; Lee, N. Effect of synthetic triglycerides of myristic, palmitic, and stearic acid on serum lipoprotein metabolism. Eur. J. Clin. Nutr. 1999, 53, 597–605. [Google Scholar] [CrossRef]
- Temme, E.H.; Mensink, R.P.; Hornstra, G. Effects of medium chain fatty acids (MCFA), myristic acid, and oleic acid on serum lipoproteins in healthy subjects. J. Lipid Res. 1997, 38, 1746–1754. [Google Scholar]
- Takato, T.; Iwata, K.; Murakami, C.; Wada, Y.; Sakane, F. Chronic administration of myristic acid improves hyperglycaemia in the Nagoya–Shibata–Yasuda mouse model of congenital type 2 diabetes. Diabetologia 2017, 60, 2076–2083. [Google Scholar] [CrossRef] [PubMed]
- Wada, Y.; Sakiyama, S.; Sakai, H.; Sakane, F. Myristic acid enhances diacylglycerol kinase δ-dependent glucose uptake in myotubes. Lipids 2016, 51, 897–903. [Google Scholar] [CrossRef] [PubMed]
- Iwata, K.; Sakai, H.; Takahashi, D.; Sakane, F. Myristic acid specifically stabilizes diacylglycerol kinase δ protein in C2C12 skeletal muscle cells. Biochim. Biophys. Acta Mol. Cell Biol. Lipids 2019, 1864, 1031–1038. [Google Scholar] [CrossRef]
- Rioux, V.; Catheline, D.; Beauchamp, E.; Le Bloc’h, J.; Pédrono, F.; Legrand, P. Substitution of dietary oleic acid for myristic acid increases the tissue storage of α-linolenic acid and the concentration of docosahexaenoic acid in the brain, red blood cells and plasma in the rat. Animal 2008, 2, 636–644. [Google Scholar] [CrossRef] [PubMed]
- Rioux, V.; Catheline, D.; Bouriel, M.; Legrand, P. Dietary myristic acid at physiologically relevant levels increases the tissue content of C20:5 n-3 and C20:3 n-6 in the rat. Reprod. Nutr. Dev. 2005, 45, 599–612. [Google Scholar] [CrossRef] [PubMed]
- Senyilmaz, D.; Virtue, S.; Xu, X.; Tan, C.Y.; Griffin, J.L.; Miller, A.K.; Vidal-Puig, A.; Teleman, A.A. Regulation of mitochondrial morphology and function by stearoylation of TFR1. Nature 2015, 525, 124–128. [Google Scholar] [CrossRef] [PubMed]
- Senyilmaz-Tiebe, D.; Pfaff, D.H.; Virtue, S.; Schwarz, K.V.; Fleming, T.; Altamura, S.; Muckenthaler, M.U.; Okun, J.G.; Vidal-Puig, A.; Nawroth, P.; et al. Dietary stearic acid regulates mitochondria in vivo in humans. Nat. Commun. 2018, 9, 3129. [Google Scholar] [CrossRef] [PubMed]
- Hassel, C.A.; Mensing, E.A.; Gallaher, D.D. Dietary stearic acid reduces plasma and hepatic cholesterol concentrations without increasing bile acid excretion in cholesterol-fed hamsters. J. Nutr. 1997, 127, 1148–1155. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Schneider, C.L.; Cowles, R.L.; Stuefer-Powell, C.L.; Carr, T.P. Dietary stearic acid reduces cholesterol absorption and increases endogenous cholesterol excretion in hamsters fed cereal-based diets. J. Nutr. 2000, 130, 1232–1238. [Google Scholar] [CrossRef]
- Gouk, S.-W.; Cheng, S.-F.; Soon-Hock Ong, A.; Chuah, C.-H. Stearic acids at sn-1, 3 positions of TAG are more efficient at limiting fat deposition than palmitic and oleic acids in C57BL/6 mice. Br. J. Nutr. 2013, 111, 1174–1180. [Google Scholar] [CrossRef]
- Hunter, J.E.; Zhang, J.; Kris-Etherton, P.M. Cardiovascular disease risk of dietary stearic acid compared with trans, other saturated, and unsaturated fatty acids: A systematic review. Am. J. Clin. Nutr. 2010, 91, 46–63. [Google Scholar] [CrossRef] [PubMed]
- Cowles, R.L.; Lee, J.-Y.; Gallaher, D.D.; Stuefer-Powell, C.L.; Carr, T.P. Dietary stearic acid alters gallbladder bile acid composition in hamsters fed cereal-based diets. J. Nutr. 2002, 132, 3119–3122. [Google Scholar] [CrossRef] [PubMed]
- Nishitani, Y.; Okazaki, S.; Imabayashi, K.; Katada, R.; Umetani, K.; Yajima, H.; Matsumoto, H. Saturated and monounsaturated fatty acids increase interleukin-10 production in rat hepatocytes. Nihon Arukoru Yakubutsu Igakkai Zasshi 2007, 42, 32–35. [Google Scholar] [PubMed]
- Pan, P.-H.; Lin, S.-Y.; Ou, Y.-C.; Chen, W.-Y.; Chuang, Y.-H.; Yen, Y.-J.; Liao, S.-L.; Raung, S.-L.; Chen, C.-J. Stearic acid attenuates cholestasis-induced liver injury. Biochem. Biophys. Res. Commun. 2010, 391, 1537–1542. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Liang, C.; Li, G.; Yu, C.; Yin, M. Stearic acid protects primary cultured cortical neurons against oxidative stress. Acta Pharmacol. Sin. 2007, 28, 315–326. [Google Scholar] [CrossRef] [PubMed]
- Holman, R.T.; Adams, C.E.; Nelson, R.A.; Grater, S.J.; Jaskiewicz, J.A.; Johnson, S.B.; Erdman, J.W. Patients with anorexia nervosa demonstrate deficiencies of selected essential fatty acids, compensatory changes in nonessential fatty acids and decreased fluidity of plasma lipids. J. Nutr. 1995, 125, 901–917. [Google Scholar] [CrossRef]
- Kaneda, T. Iso-and anteiso-fatty acids in bacteria: Biosynthesis, function, and taxonomic significance. Microbiol. Rev. 1991, 55, 288–302. [Google Scholar]
- Poger, D.; Caron, B.; Mark, A.E. Effect of methyl-branched fatty acids on the structure of lipid bilayers. J. Phys. Chem. B 2014, 118, 13838–13848. [Google Scholar] [CrossRef]
- Cai, Q.; Huang, H.; Qian, D.; Chen, K.; Luo, J.; Tian, Y.; Lin, T.; Lin, T. 13-Methyltetradecanoic acid exhibits anti-tumor activity on T-cell lymphomas in vitro and in vivo by down-regulating p-AKT and activating caspase-3. PLoS ONE 2013, 8, e65308. [Google Scholar] [CrossRef]
- Yang, Z.; Liu, S.; Chen, X.; Chen, H.; Huang, M.; Zheng, J. Induction of apoptotic cell death and in vivo growth inhibition of human cancer cells by a saturated branched-chain fatty acid, 13-methyltetradecanoic acid. Cancer Res. 2000, 60, 505–509. [Google Scholar]
- Kraft, J.; Jetton, T.; Satish, B.; Gupta, D. Dairy-derived bioactive fatty acids improve pancreatic ß-cell function. FASEB J. 2015, 29, 608–625. [Google Scholar]
- Ran-Ressler, R.R.; Khailova, L.; Arganbright, K.M.; Adkins-Rieck, C.K.; Jouni, Z.E.; Koren, O.; Ley, R.E.; Brenna, J.T.; Dvorak, B. Branched chain fatty acids reduce the incidence of necrotizing enterocolitis and alter gastrointestinal microbial ecology in a neonatal rat model. PLoS ONE 2011, 6, e29032. [Google Scholar] [CrossRef]
- Yan, Y.; Wang, Z.; Greenwald, J.; Kothapalli, K.S.D.; Park, H.G.; Liu, R.; Mendralla, E.; Lawrence, P.; Wang, X.; Brenna, J.T. BCFA suppresses LPS induced IL-8 mRNA expression in human intestinal epithelial cells. Prostaglandins Leukot. Essent. Fat. Acids 2017, 116, 27–31. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Wang, Z.; Wang, D.; Lawrence, P.; Wang, X.; Kothapalli, K.S.D.; Greenwald, J.; Liu, R.; Park, H.G.; Brenna, J.T. BCFA-enriched vernix-monoacylglycerol reduces LPS-induced inflammatory markers in human enterocytes in vitro. Pediatr. Res. 2018, 83, 874–879. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; Hao, T.; Rimm, E.B.; Willett, W.C.; Hu, F.B. Changes in diet and lifestyle and long-term weight gain in women and men. N. Engl. J. Med. 2011, 364, 2392. [Google Scholar] [CrossRef] [PubMed]
| Fatty acid | Median (g/100 g) | Range (g/100 g) | Median (mg/Three Daily Servings) 8 |
|---|---|---|---|
| Total SCFA 1 | 5.18 | (4.83–7.44) | 1150 |
| 4:0 | 3.05 | (2.90–5.37) | 677 |
| 6:0 | 2.01 | (1.86–2.25) | 445 |
| Total MCFA 2 | 8.34 | (4.03–9.77) | 1850 |
| 8:0 | 1.18 | (0.82–1.37) | 261 |
| 10:0 | 2.86 | (1.48–3.38) | 635 |
| 12:0 | 3.60 | (1.53–4.20) | 798 |
| Total LCFA 3 | 58.13 | (43.02–67.66) | 12,903 |
| 14:0 | 12.14 | (8.56–12.57) | 2695 |
| 16:0 | 34.83 | (25.01–36.60) | 7730 |
| 18:0 | 9.05 | (7.91–15.94) | 2008 |
| Total VLCFA 4 | 0.13 | (0.08–0.14) | 29 |
| Total OCFA 5 | 2.8 | (1.83–2.91) | 622 |
| 15:0 | 1.28 | (0.72–1.39) | 284 |
| 17:0 | 0.71 | (0.70–0.97) | 158 |
| Total BCFA 6 | 1.65 | (1.49–1.8) | 366 |
| Total SFA 7 | 72.81 | (62.88–76.73) | 16,162 |
| Reference | Population | Study Design | Adjustments | Risk Factors | |
|---|---|---|---|---|---|
| Hyperglycemia | Metabolic Syndrome (Risk or Prevalence) | ||||
| Drehmer et al. (2015) [44] | Brazilian adults (10,010; 35–74 year old) | Cross-sectional | 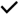 | NA 1 | |
| Drehmer et al. (2016) [21] | Brazilian adults (n = 9835; 35–74 year old); | Cross-sectional | 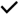 | NA | |
| Iggman et al. (2010) [42] | 795 Swedish men (~71 year old) | Cross-sectional | 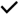 | 2 | NA |
| Kratz et al. (2014) [45] | American adults with non-alcoholic fatty liver disease (n = 17) and controls (n = 15) | Cross-sectional | 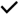 | NA | |
| Mayneris-Perxachs et al. (2014) [36] | Spanish adults, asymptomatic with cardiovascular disease risk (n = 427; 55–80 year old) | Cross-sectional | 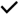 | NA | |
| Santaren et al. (2014) [41] | Hispanic, African American, and non-Hispanic white American adults, free of type 2 diabetes at baseline (n = 659; 40–60 year old) | Cross-sectional | 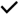 | NA | |
| Wanders et al. (2017) [43] | Dutch adults, overweight (n = 5675; 45–65 year old) | Cross-sectional | 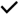 | NA | |
| Reference | Population | Study Design | Study Duration | Adjustments | Risk Factors | |||
|---|---|---|---|---|---|---|---|---|
| Hyperglycemia | Obesity | Dyslipidemia | Hypertension | |||||
| Amer et al. (2017) [62], Bohl et al. (2015) [47], Bohl et al. (2017) [46], Matualatupauw et al. (2017) [58] | Danish adults, abdominally overweight (n = 52; ≥18 year old) | Parallel | 12 weeks | - 1 | 2 | - | - | |
| Bjermo et al. (2012) [50] | Swedish adults, abdominally overweight (n = 61; 30–65 year old) | Parallel | 10 weeks | - | NA 3 | |||
| Iggman et al. (2011) [51] | Swedish adults, hyperlipidemic (n = 20; 25–68 year old) | Crossover | 9 weeks | 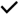 | - | - | NA | |
| Intorre et al. (2011) [60] | Italian adults (n = 60; 20–40 year old) | Crossover | 16 weeks | NA | NA | - | NA | |
| Malpuech-Brugere et al. (2010) [61] | French adults (n = 111; 18–50 year old) | Parallel | 4 weeks | NA | NA | NA | ||
| Pintus et al. (2013) [57] | Italian adults, hypercholesterolemic (n = 42; 30–60 year old) | Crossover | 9 weeks | NA | - | NA | ||
| Venkatramanan et al. (2010) [56] | Canadian adults, overweight and borderline hyperlipidemic (n = 15; 30–60 year old) | Crossover | 32 weeks | NA | - | - | NA | |
| Wennersberg et al. (2009) [49] | Finnish, Norwegian, and Swedish adult men and postmenopausal women, overweight with traits of MetS (n = 121; 30–65 year old) | Parallel | 6 months | - | - | |||
| Werner et al. (2013) [48] | Danish adult and elderly subjects (n = 38; 50–70 year old) | Parallel | 12 weeks | - | - | - | NA | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Unger, A.L.; Torres-Gonzalez, M.; Kraft, J. Dairy Fat Consumption and the Risk of Metabolic Syndrome: An Examination of the Saturated Fatty Acids in Dairy. Nutrients 2019, 11, 2200. https://doi.org/10.3390/nu11092200
Unger AL, Torres-Gonzalez M, Kraft J. Dairy Fat Consumption and the Risk of Metabolic Syndrome: An Examination of the Saturated Fatty Acids in Dairy. Nutrients. 2019; 11(9):2200. https://doi.org/10.3390/nu11092200
Chicago/Turabian StyleUnger, Allison L., Moises Torres-Gonzalez, and Jana Kraft. 2019. "Dairy Fat Consumption and the Risk of Metabolic Syndrome: An Examination of the Saturated Fatty Acids in Dairy" Nutrients 11, no. 9: 2200. https://doi.org/10.3390/nu11092200
APA StyleUnger, A. L., Torres-Gonzalez, M., & Kraft, J. (2019). Dairy Fat Consumption and the Risk of Metabolic Syndrome: An Examination of the Saturated Fatty Acids in Dairy. Nutrients, 11(9), 2200. https://doi.org/10.3390/nu11092200





