Consumption of Raw Orange, 100% Fresh Orange Juice, and Nectar- Sweetened Orange Juice—Effects on Blood Glucose and Insulin Levels on Healthy Subjects
Abstract
1. Introduction
2. Methods
2.1. Intervention
2.2. Anthropometric Data
2.3. Blood Collection
2.4. Statistics
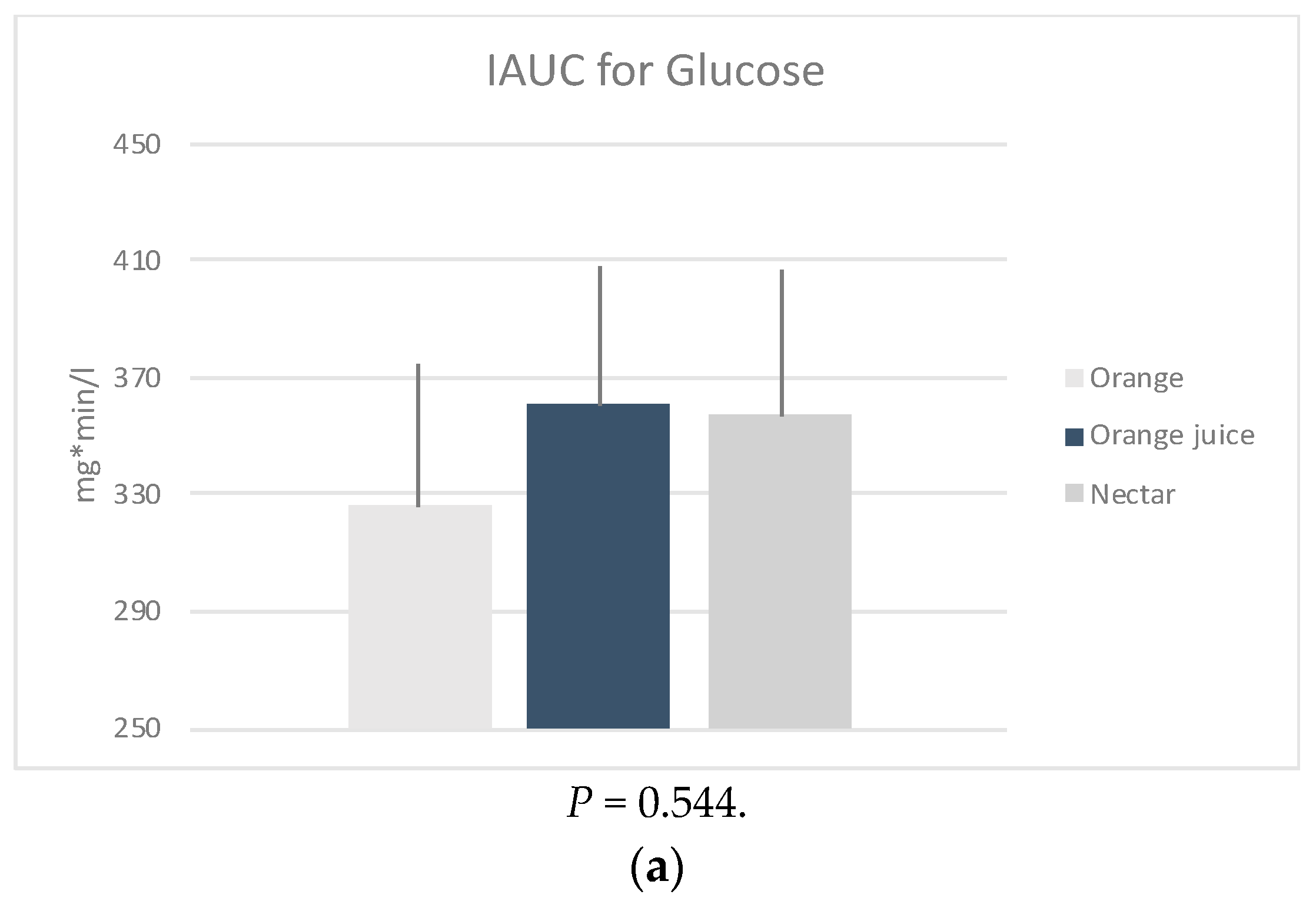
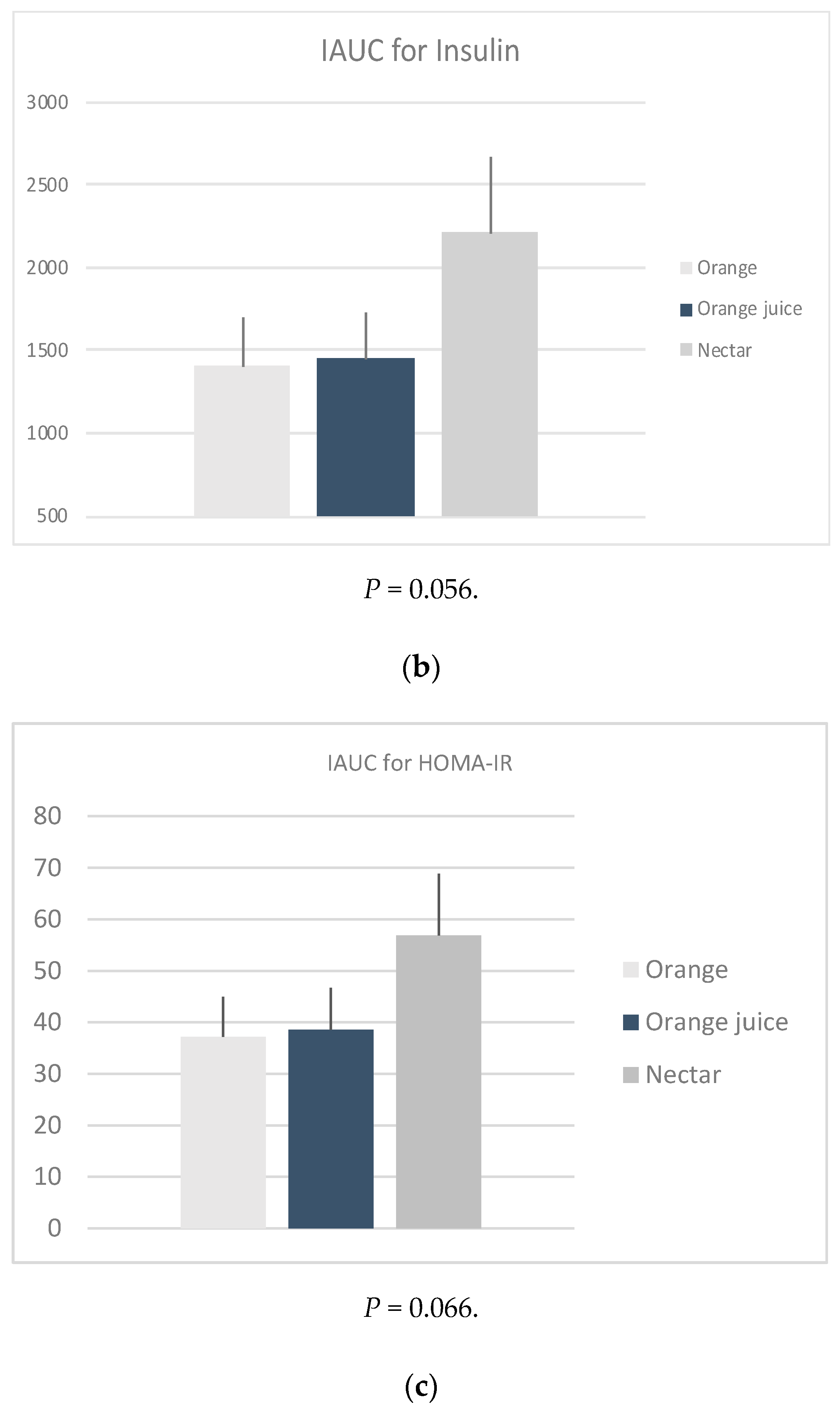
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Data Availability
References
- Whiting, D.R.; Guariguata, L.; Weil, C.; Shaw, J. IDF diabetes atlas: Global estimates of the prevalence of diabetes for 2011 and 2030. Diabetes Res. Clin. Pract. 2011, 94, 311–321. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Status Report on Noncommunicable Diseases 2010; World Health Organization: Geneva, Switzerland, 2011. [Google Scholar]
- International Diabetes Federation. IDF Diabetes Atlas, 6th ed.; International Diabetes Federation: Brussels, Belgium, 2013. [Google Scholar]
- Badran, M.; Laher, I. Type II Diabetes Mellitus in Arabic-Speaking Countries. Int. J. Endocrinol. 2012, 902873. [Google Scholar] [CrossRef] [PubMed]
- Lathief, S.; Inzucchi, S.E. Approach to diabetes management in patients with CVD. Trends Cardiovasc. Med. 2016, 26, 165–179. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.N.; Zhao, D.D.; Gao, B.; Zhong, K.; Zhu, R.X.; Zhang, Y.; Xie, W.J.; Jia, L.R.; Gao, H. Anti-hyperglycemic effect of chebulagic acid from the fruits of Terminalia chebula Retz. Int. J. Mol. 2012, 13, 6320–6333. [Google Scholar] [CrossRef] [PubMed]
- Lindstrom, J.; Peltonen, M.; Eriksson, J.G.; Aunola, S.; Hämäläinen, H.; Ilanne-Parikka, P.; Keinänen-Kiukaanniemi, S.; Uusitupa, M.; Tuomilehto, J.; Finnish Diabetes Prevention Study (DPS) Group. Determinants for the effectiveness of lifestyle intervention in the Finnish Diabetes Prevention Study. Diabetes Care 2008, 31, 857–862. [Google Scholar] [CrossRef] [PubMed]
- Reis, J.P.; Loria, C.M.; Sorlie, P.D.; Park, Y.; Hollenbeck, A.; Schatzkin, A. Lifestyle factors and risk for new-onset diabetes: A population-based cohort study. Ann. Int. Med. 2011, 155, 292–299. [Google Scholar] [CrossRef]
- Xi, B.; Li, S.; Liu, Z.; Tian, H.; Yin, X.; Huai, P.; Tang, W.; Zhou, D.; Steffen, L.M. Intake of Fruit Juice and Incidence of Type 2 Diabetes: A Systematic Review and Meta-Analysis. PLoS ONE 2014, 9, e93471. [Google Scholar] [CrossRef]
- Pereira, M.A.; Fulgoni, V.L., III. Consumption of 100% fruit juice and risk of obesity and metabolic syndrome: Findings from the national health and nutrition examination survey 1999–2004. J. Am. Coll. Nutr. 2010, 29, 625–629. [Google Scholar] [CrossRef]
- Eshak, E.S.; Iso, H.; Mizoue, T.; Inoue, M.; Noda, M.; Tsugane, S. Soft drink, 100% fruit juice, and vegetable juice intakes and risk of diabetes mellitus. Clin. Nutr. 2013, 3, 300–308. [Google Scholar] [CrossRef]
- Wang, Y.C.; Bleich, S.N.; Gortmaker, S.L. Increasing caloric contribution from sugar-sweetened beverages and 100% fruit juices among US children and adolescents, 1988–2004. Pediatrics 2008, 121, e1604–e1614. [Google Scholar] [CrossRef]
- Schulze, M.B.; Liu, S.; Rimm, E.B.; Manson, J.E.; Willett, W.C.; Hu, F.B. Glycemic index, glycemic load, and dietary fiber intake and incidence of type 2 diabetes in younger and middle-aged women. Am. J. Clin. Nutr. 2004, 80, 348–356. [Google Scholar] [CrossRef] [PubMed]
- Hodge, A.M.; English, D.R.; O’Dea, K.; Giles, G.G. Glycemic index and dietary fiber and the risk of type 2 diabetes. Diabetes Care 2004, 27, 2701–2706. [Google Scholar] [CrossRef] [PubMed]
- Cole, T.J.; Bellizzi, C.M.; Flegal, M.K. Establishing a standard definition for child overweight and obesity worldwide: International survey. BMJ 2000, 320, 1240–1243. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Liu, K.; Mi, M.; Wang, J. Effect of fruit juice on glucose control and insulin sensitivity in adults: A meta-analysis of 12 randomized controlled trials. PLoS ONE 2014, 9, 95323. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.M.; Barrett, E.C.; Bresnahan, K.A.; Barraj, L.M. 100 % Fruit juice and measures of glucose control and insulin sensitivity: A systematic review and meta-analysis of randomized controlled trials. J. Nutr. Sci. 2017, 6, e59. [Google Scholar] [CrossRef] [PubMed]
- Wolever, T.M.S.; Jenkins, D.J.A.; Jenkins, A.L.; Josse, R.G. The glycemic index: Methodology and clinical implications. Am. J. Clin. Nutr. 1991, 54, 846–854. [Google Scholar] [CrossRef] [PubMed]
- Odegaard, A.O.; Koh, W.P.; Arakawa, K.; Yu, M.C.; Pereira, M.A. Soft drink and juice consumption and risk of physician-diagnosed incident type 2 diabetes: The Singapore Chinese Health Study. Am. J. Epidemiol. 2010, 171, 701–708. [Google Scholar] [CrossRef] [PubMed]
- Bazzano, L.A.; Li, T.Y.; Joshipura, K.J.; Hu, F.B. Intake of fruit, vegetables, and fruit juices and risk of diabetes in women. Diabetes Care 2008, 31, 1311–1317. [Google Scholar] [CrossRef] [PubMed]
- Tsilas, C.S.; de Souza, R.J.; Mejia, S.B.; Mirrahimi, A.; Cozma, A.I.; Jayalath, V.H.; Ha, V.; Tawfik, R.; Di Buono, M.; Jenkins, A.L.; et al. Relation of total sugars, fructose and sucrose with incident type 2 diabetes: A systematic review and meta-analysis of prospective cohort studies. CMAJ 2017, 189, E711–E720. [Google Scholar] [CrossRef]
- InterAct Consortium; Romaguera, D.; Norat, T.; Wark, P.A.; Vergnaud, A.C.; Schulze, M.B.; van Woudenbergh, G.J.; Drogan, D.; Amiano, P.; Molina-Montes, E.; et al. Consumption of sweet beverages and type 2 diabetes incidence in European adults: Results from EPIC-InterAct. Diabetologia 2013, 56, 1520–1530. [Google Scholar] [CrossRef]
- Ruxton, C.H.; Gardner, E.J.; Walker, D. Can pure fruit and vegetable juices protect against cancer and cardiovascular disease too? A review of the evidence. Int. J. Food Sci. Nutr. 2006, 57, 249–272. [Google Scholar] [CrossRef] [PubMed]
- Imamura, F.; O’Connor, L.; Ye, Z.; Mursu, J.; Hayashino, Y.; Bhupathiraju, S.N.; Forouhi, N.G. Consumption of sugar sweetened beverages, artificially sweetened beverages, and fruit juice and incidence of type 2 diabetes: Systematic review, meta-analysis, and estimation of population attributable fraction. Br. J. Sports Med. 2016, 50, 496–504. [Google Scholar] [CrossRef] [PubMed]
- Malik, V.S.; Popkin, B.M.; Bray, G.A.; Després, J.P.; Willett, W.C.; Hu, F.B. Sugar-sweetened beverages and risk of metabolic syndrome and type 2 diabetes: A meta-analysis. Diabetes Care 2010, 33, 2477–2483. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, M.; McKeown, N.M.; Rogers, G.; Meigs, J.B.; Saltzman, E.; D’Agostino, R.; Jacques, P.F. Surrogate markers of insulin resistance are associated with consumption of sugar-sweetened drinks and fruit juice in middle and older-aged adults. J. Nutr. 2007, 137, 2121–2127. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Jacques, P.F.; Meigs, J.B.; Fox, C.S.; Rogers, G.T.; Smith, C.E.; Hruby, A.; Saltzman, E.; McKeown, N.M. Sugar-Sweetened Beverage but Not Diet Soda Consumption Is Positively Associated with Progression of Insulin Resistance and Prediabetes. J. Nutr. 2016, 146, 2544–2550. [Google Scholar] [CrossRef] [PubMed]
- Paneni, F.; Costantino, S.; Cosentino, F. Insulin resistance, diabetes, and cardiovascular risk. Curr. Atheroscler. Rep. 2014, 16, 419. [Google Scholar] [CrossRef] [PubMed]
- Manios, Y.; Moschonis, G.; Mavrogianni, C.; Tsoutsoulopoulou, K.; Kogkas, S.; Lambrinou, C.P.; Efstathopoulou, E. Postprandial glucose and insulin levels in type 2 diabetes mellitus patients after consumption of ready-to-eat mixed meals. Eur. J. Nutr. 2017, 56, 1359–1367. [Google Scholar] [CrossRef] [PubMed]
| Fruit Samples | Glucose (g) | Fructose (g) | Sucrose (g) | Maltose (g) | Total (g) |
|---|---|---|---|---|---|
| 2 Oranges (260 mL) | 5.9 | 6 | 14 g | 0.5 | 26.4 |
| 100% Fruit Juice (265 mL) | 7.5 | 8 | 11 g | 0.5 | 27 |
| Sweetened Orange Juice (225 mL) | 6.5 | 7 | 13 g | 0.5 | 27 |
| Variables1 | Total (N = 20) | Normal Weight (N = 13) | Overweight & Obese (N = 7) | P-Value by Weight Status * |
|---|---|---|---|---|
| Age, years | 21.1 (0.9) | 21.0 (0.9) | 21.3 (0.7) | 0.271 |
| Weight (kg) | 61.5 (15.3) | 53.0 (8.8) | 77.2 (11.9) | <0.001 |
| BMI (kg/m2) | 23.6 (5.4) | 20.7 (2.8) | 29.1 (2.5) | <0.001 |
| Fasting Glucose 2 | 0.025 | |||
| RO | 85.3 (7.6) | 83.5 (6.5) | 88.7 (8.8) | |
| FOJ | 86.8 (6.9) | 85.6 (7.1) | 88.9 (6.2) | |
| NSOJ | 85.6 (6.1) | 84.3 (5.5) | 88.0 (6.8) | |
| Fasting Insulin 2 | 0.163 | |||
| RO | 55.5 (20.5) | 56.7 (22.6) | 53.3 (17.6) | |
| FOJ | 55.7 (27.8) | 49.4 (27.2) | 67.2 (27.2) | |
| NSOJ | 56.3 (35.2) | 50.1 (37.3) | 67.7 (30.1) | |
| Energy (Kcal) | 2452.3 (1067.9) | 1834.2 (474.5) | 3600.3 (832.9) | <0.001 |
| Fiber (g) | 14.3 (10.1, 25.4) | 11.8 (9.7, 14.5) | 28.8 (14.2, 42.6) | <0.001 |
| T0 | T30 | T60 | T90 | T120 | Mean Change | P-Value * (Time × Treatment) | |
|---|---|---|---|---|---|---|---|
| Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | Mean (SD) | (T120-T0) Mean (95% CI) | ||
| Serum Glucose | 0.462 | ||||||
| RO | 85.3 (7.6) | 98.1 (14.9) | 86.6 (11.0) | 84.6 (6.7) | 83.3 (6.7) | −2.0 (−4.94, 0.87) | |
| OJ | 86.8 (6.9) | 104.0 (14.2) | 86.1 (9.3) | 84.9 (7.9) | 84.7 (7.2) | −2.1 (−4.05, −0.11) | |
| SOJ | 85.6 (6.1) | 102.3 (17.8) | 85.5 (12.4) | 83.6 (8.4) | 83.3 (7.4) | −2.3 (−4.93, 0.38) | |
| P-value + (treatment effect) | 0.836 | 0.204 | 0.649 | 0.631 | 0.962 | ||
| Serum Insulin ** | <0.001 | ||||||
| RO | 55.5 (20.5) | 111.3 (75.4) | 60.8 (40.9) | 51.4 (31.1) | 41.5 (18.4) | −14.0 (−27.05, −0.90) | |
| FOJ | 55.7 (27.8) | 107.5 (77.2) | 60.3 (43.9) | 49.2 (26.1) | 42.0 (18.8) | −13.7 (−28.80, 1.44) | |
| NSOJ | 56.3 (35.2) | 136.9 (71.3) | 73.8 (56.9) | 62.8 (38.6) | 47.1 (21.4) | −9.2 (−28.75, 10.30) | |
| P-value + (treatment effect) | 0.925 | 0.233 | 0.282 | 0.123 | 0.290 | ||
| HOMA-IR | <0.001 | ||||||
| RO | 1.2 (0.5) | 2.7 (1.9) | 1.4 (1.1) | 1.1 (0.8) | 0.9 (0.4) | −0.3 (−0.61, −0.03) | |
| FOJ | 1.2 (0.7) | 2.8 (2.3) | 1.4 (1.1) | 1.1 (0.6) | 0.9 (0.4) | −0.3 (−0.67, 0.01) | |
| NSOJ | 1.2 (0.8) | 3.6 (2.3) | 1.7 (1.6) | 1.3 (0.9) | 1.0 (0.5) | −0.2 (−0.66, 0.22) | |
| P-value+ (treatment effect) | 0.894 | 0.163 | 0.346 | 0.188 | 0.283 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Papandreou, D.; Magriplis, E.; Abboud, M.; Taha, Z.; Karavolia, E.; Karavolias, C.; Zampelas, A. Consumption of Raw Orange, 100% Fresh Orange Juice, and Nectar- Sweetened Orange Juice—Effects on Blood Glucose and Insulin Levels on Healthy Subjects. Nutrients 2019, 11, 2171. https://doi.org/10.3390/nu11092171
Papandreou D, Magriplis E, Abboud M, Taha Z, Karavolia E, Karavolias C, Zampelas A. Consumption of Raw Orange, 100% Fresh Orange Juice, and Nectar- Sweetened Orange Juice—Effects on Blood Glucose and Insulin Levels on Healthy Subjects. Nutrients. 2019; 11(9):2171. https://doi.org/10.3390/nu11092171
Chicago/Turabian StylePapandreou, Dimitrios, Emmanouella Magriplis, Myriam Abboud, Zainab Taha, Eleftheria Karavolia, Christos Karavolias, and Antonis Zampelas. 2019. "Consumption of Raw Orange, 100% Fresh Orange Juice, and Nectar- Sweetened Orange Juice—Effects on Blood Glucose and Insulin Levels on Healthy Subjects" Nutrients 11, no. 9: 2171. https://doi.org/10.3390/nu11092171
APA StylePapandreou, D., Magriplis, E., Abboud, M., Taha, Z., Karavolia, E., Karavolias, C., & Zampelas, A. (2019). Consumption of Raw Orange, 100% Fresh Orange Juice, and Nectar- Sweetened Orange Juice—Effects on Blood Glucose and Insulin Levels on Healthy Subjects. Nutrients, 11(9), 2171. https://doi.org/10.3390/nu11092171








