Comparative Meta-Analysis of the Effect of Concentrated, Hydrolyzed, and Isolated Whey Protein Supplementation on Body Composition of Physical Activity Practitioners
Abstract
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria and Characterization of Intervention
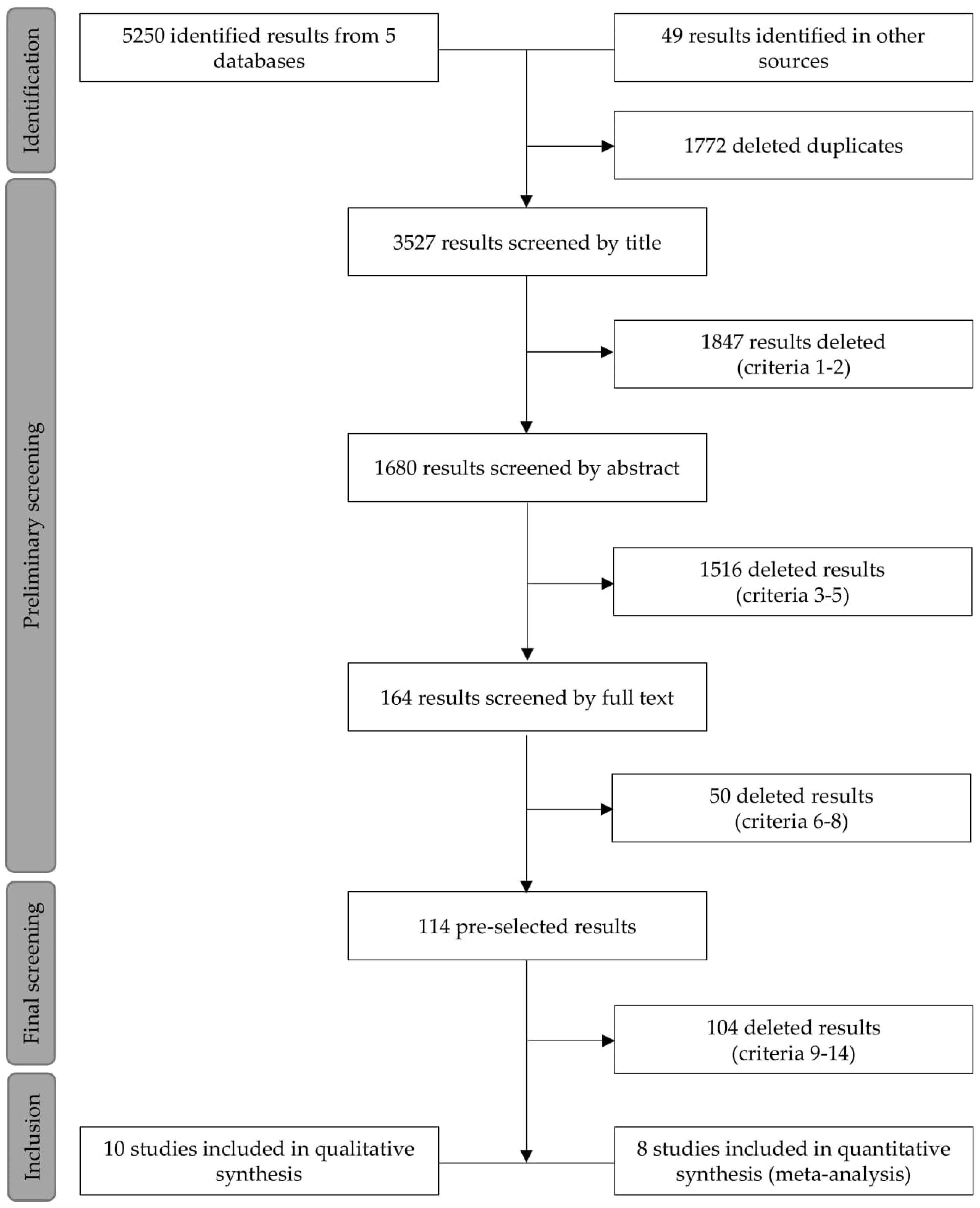
2.2. Search Strategy and Preliminary Screening
2.3. Data Extraction and Final Screening
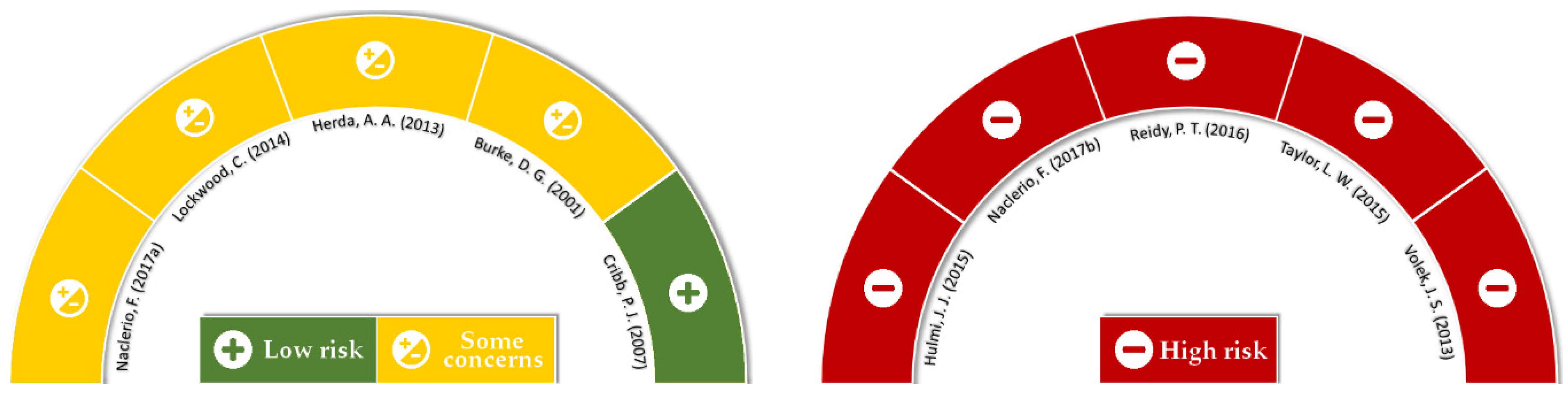
2.4. Risk of Bias and Quality Assessment
2.5. Statistical Analysis
3. Results
3.1. Searches, Screening, and Quality Analysis
3.2. Methodological Characterization
3.3. Nutritional Intervention
3.4. Physical Intervention
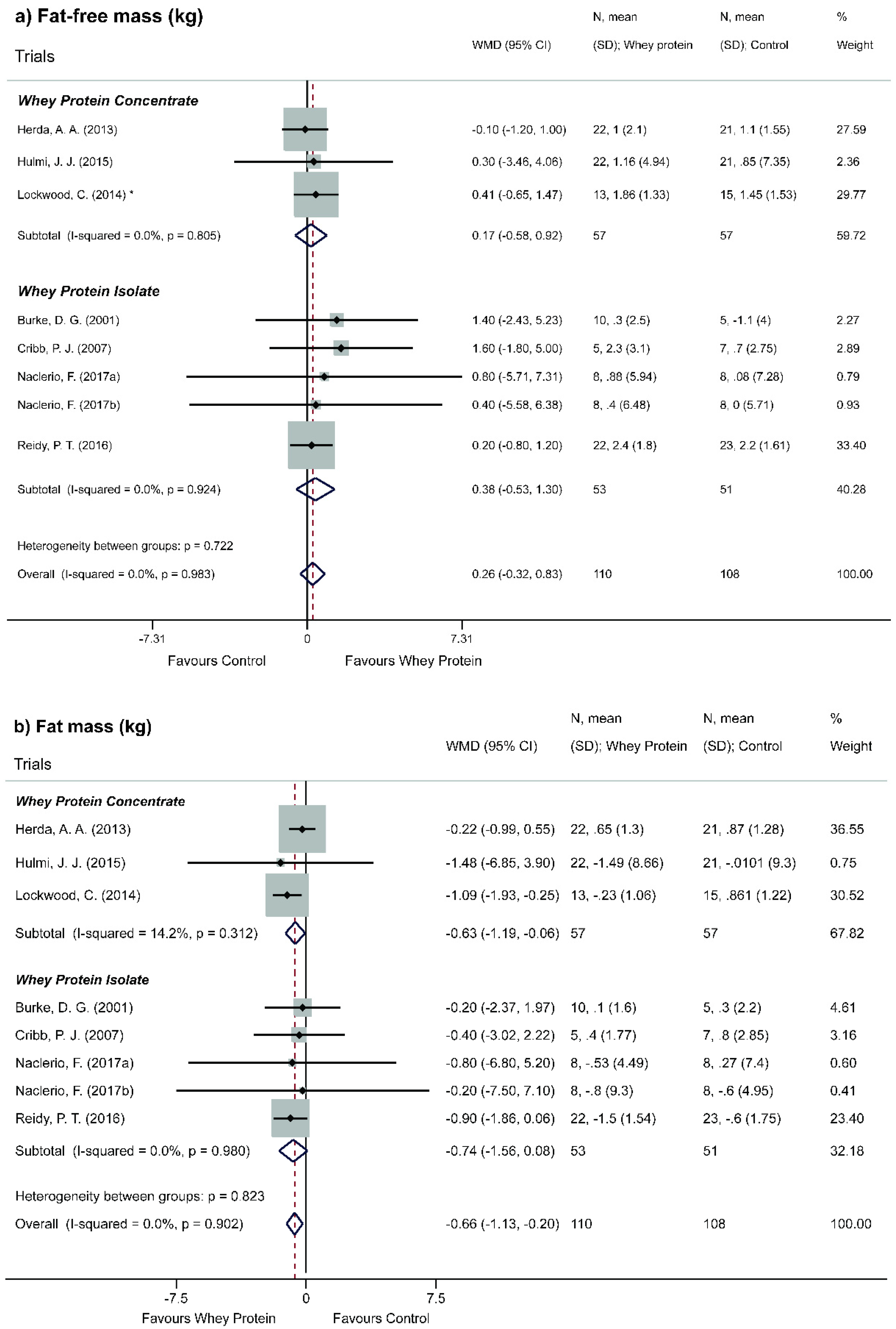
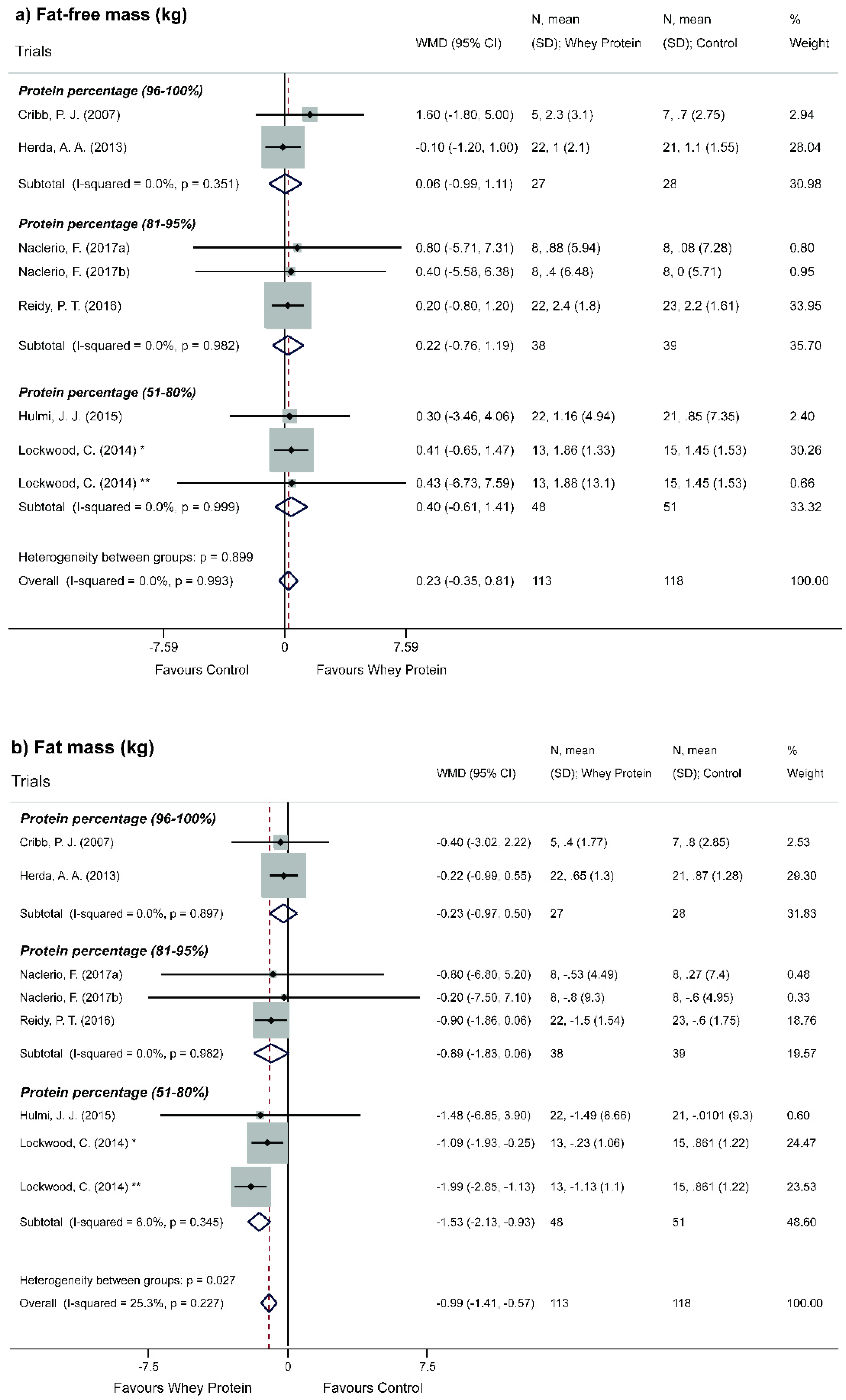
3.5. Effect of Whey Protein on Body Composition
3.6. Adverse Effects
4. Discussion
4.1. Methodological Approach of the Problem
4.2. Effect of Whey Protein on Body Composition
4.3. Limitations, Strengths, and Quality of Evidence
- Low power of evidence, in which WP does not significantly change FFM when compared to other non-protein isocaloric interventions and that concentrated and isolated WP, even with different protein levels, are virtually identical regarding the amplitude of this effect;
- Moderate power of evidence, which after adapted to resistance exercise, the non-occurrence of FFM gain regarding WP supplementation, both in its concentrated and isolated form, does not depend on the level of physical activity;
- High power of evidence, in which WP supplementation is favorable to the loss of FM and this reduction is observed only for WPC;
- Moderate power of evidence, in which the depletive effect of FM occurs only in WPs of low protein content (51%–80%); and this result is verified only in regular practitioners of resisting and/or strength activity;
- Very low power of evidence, in which there is an inversely proportional trend between protein WP content and FM reduction.
4.4. Future Recommendations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Andoyo, R.; Dianti Lestari, V.; Mardawati, E.; Nurhadi, B. Fractal Dimension Analysis of Texture Formation of Whey Protein-Based Foods. Int. J. Food Sci. 2018, 2018, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Patel, S. Emerging trends in nutraceutical applications of whey protein and its derivatives. J. Food Sci. Technol. 2015, 52, 6847–6858. [Google Scholar] [CrossRef] [PubMed]
- Cheison, S.C.; Kulozik, U. Impact of the environmental conditions and substrate pre-treatment on whey protein hydrolysis: A review. Crit. Rev. Food Sci. Nutr. 2017, 57, 418–453. [Google Scholar] [CrossRef]
- Larsen, A.; Bibby, B.; Hansen, M. Effect of a Whey Protein Supplement on Preservation of Fat Free Mass in Overweight and Obese Individuals on an Energy Restricted Very Low Caloric Diet. Nutrients 2018, 10, 1918. [Google Scholar] [CrossRef] [PubMed]
- Carter, B.G.; Drake, M.A. Invited review: The effects of processing parameters on the flavor of whey protein ingredients. J. Dairy Sci. 2018, 101, 6691–6702. [Google Scholar] [CrossRef]
- Lagrange, V.; Whitsett, D.; Burris, C. Global Market for Dairy Proteins: Global market for dairy proteins…. J. Food Sci. 2015, 80, A16–A22. [Google Scholar] [CrossRef]
- Meena, G.S.; Singh, A.K.; Panjagari, N.R.; Arora, S. Milk protein concentrates: Opportunities and challenges. J. Food Sci. Technol. 2017, 54, 3010–3024. [Google Scholar] [CrossRef]
- Jeewanthi, R.K.C.; Lee, N.-K.; Paik, H.-D. Improved Functional Characteristics of Whey Protein Hydrolysates in Food Industry. Korean J. Food Sci. Anim. Resour. 2015, 35, 350–359. [Google Scholar] [CrossRef] [PubMed]
- Davies, R.; Carson, B.; Jakeman, P. The Effect of Whey Protein Supplementation on the Temporal Recovery of Muscle Function Following Resistance Training: A Systematic Review and Meta-Analysis. Nutrients 2018, 10, 221. [Google Scholar] [CrossRef]
- Draganidis, D.; Karagounis, L.G.; Athanailidis, I.; Chatzinikolaou, A.; Jamurtas, A.Z.; Fatouros, I.G. Inflammaging and Skeletal Muscle: Can Protein Intake Make a Difference? J. Nutr. 2016, 146, 1940–1952. [Google Scholar] [CrossRef]
- Westerterp, K.R. Exercise, energy balance and body composition. Eur. J. Clin. Nutr. 2018, 72, 1246–1250. [Google Scholar] [CrossRef] [PubMed]
- Sonksen, P. Determination and regulation of body composition in elite athletes. Br. J. Sports Med. 2018, 52, 219–229. [Google Scholar] [CrossRef] [PubMed]
- Müller, M.J.; Braun, W.; Pourhassan, M.; Geisler, C.; Bosy-Westphal, A. Application of standards and models in body composition analysis. Proc. Nutr. Soc. 2016, 75, 181–187. [Google Scholar] [CrossRef] [PubMed]
- Trumbo, P.; Schlicker, S.; Yates, A.A.; Poos, M. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein and Amino Acids. J. Am. Diet. Assoc. 2002, 102, 1621–1630. Available online: https://www.nal.usda.gov/sites/default/files/fnic_uploads/energy_full_report.pdf (accessed on 14 July 2019). [CrossRef]
- Egan, B. Protein intake for athletes and active adults: Current concepts and controversies. Nutr. Bull. 2016, 41, 202–213. [Google Scholar] [CrossRef]
- Samal, J.R.K.; Samal, I.R. Protein Supplements: Pros and Cons. J. Diet. Suppl. 2018, 15, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S.M.; Chevalier, S.; Leidy, H.J. Protein “requirements” beyond the RDA: Implications for optimizing health. Appl. Physiol. Nutr. Metab. 2016, 41, 565–572. [Google Scholar] [CrossRef]
- Morr, C.V.; Ha, E.Y.W. Whey protein concentrates and isolates: Processing and functional properties. Crit. Rev. Food Sci. Nutr. 1993, 33, 431–476. [Google Scholar] [CrossRef]
- Moher, D. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. Ann. Intern. Med. 2009, 151, 264. [Google Scholar] [CrossRef]
- Hind, K.; Slater, G.; Oldroyd, B.; Lees, M.; Thurlow, S.; Barlow, M.; Shepherd, J. Interpretation of Dual-Energy X-Ray Absorptiometry-Derived Body Composition Change in Athletes: A Review and Recommendations for Best Practice. J. Clin. Densitom. 2018, 21, 429–443. [Google Scholar] [CrossRef]
- Kuriyan, R. Body composition techniques. Indian J. Med. Res. 2018, 148, 648. [Google Scholar] [CrossRef] [PubMed]
- Scafoglieri, A.; Clarys, J.P. Dual energy X-ray absorptiometry: Gold standard for muscle mass?: Letter to the Editor. J. Cachexia Sarcopenia Muscle 2018, 9, 786–787. [Google Scholar] [CrossRef]
- Kim, J.; Wang, Z.; Heymsfield, S.B.; Baumgartner, R.N.; Gallagher, D. Total-body skeletal muscle mass: Estimation by a new dual-energy X-ray absorptiometry method. Am. J. Clin. Nutr. 2002, 76, 378–383. [Google Scholar] [CrossRef] [PubMed]
- Speakman, J.; Booles, D.; Butterwick, R. Validation of dual energy X-ray absorptiometry (DXA) by comparison with chemical analysis of dogs and cats. Int. J. Obes. 2001, 25, 439–447. [Google Scholar] [CrossRef] [PubMed]
- Clarys, J.P.; Scafoglieri, A.; Provyn, S.; Louis, O.; Wallace, J.A.; De Mey, J. A Macro-quality Evaluation of DXA Variables Using Whole Dissection, Ashing, and Computer Tomography in Pigs. Obesity 2010, 18, 1477–1485. [Google Scholar] [CrossRef]
- Pietrobelli, A.; Formica, C.; Wang, Z.; Heymsfield, S.B. Dual-energy X-ray absorptiometry body composition model: Review of physical concepts. Am. J. Physiol. Endocrinol. Metab. 1996, 271, E941–E951. [Google Scholar] [CrossRef]
- Scafoglieri, A.; Deklerck, R.; Tresignie, J.; De Mey, J.; Clarys, J.P.; Bautmans, I. Assessment of regional adipose tissue depots: A DXA and CT comparison in cadavers of elderly persons. Exp. Gerontol. 2013, 48, 985–991. [Google Scholar] [CrossRef]
- Higgins, J.P. Revised Cochrane Risk of Bias Tool for Randomized Trials (RoB 2.0). 2016. Available online: https://www.riskofbias.info/welcome/rob-2-0-tool (accessed on 14 July 2019).
- Guyatt, G.H.; Oxman, A.D.; Schünemann, H.J.; Tugwell, P.; Knottnerus, A. GRADE guidelines: A new series of articles in the Journal of Clinical Epidemiology. J. Clin. Epidemiol. 2011, 64, 380–382. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Altman, D.G.; Gotzsche, P.C.; Juni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A.C.; et al. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef]
- Burke, D.G.; Chilibeck, P.D.; Davison, K.S.; Candow, D.C.; Farthing, J.; Smith-Palmer, T. The Effect of Whey Protein Supplementation with and Without Creatine Monohydrate Combined with Resistance Training on Lean Tissue Mass and Muscle Strength. Int. J. Sport Nutr. Exerc. Metab. 2001, 11, 349–364. [Google Scholar] [CrossRef]
- Cribb, P.J.; Williams, A.D.; Stathis, C.G.; Carey, M.F.; Hayes, A. Effects of Whey Isolate, Creatine, and Resistance Training on Muscle Hypertrophy. Med. Sci. Sports Exerc. 2007, 39, 298–307. [Google Scholar] [CrossRef] [PubMed]
- Herda, A.A.; Herda, T.J.; Costa, P.B.; Ryan, E.D.; Stout, J.R.; Cramer, J.T. Muscle Performance, Size, and Safety Responses After Eight Weeks of Resistance Training and Protein Supplementation: A Randomized, Double-Blinded, Placebo-Controlled Clinical Trial. J. Strength Cond. Res. 2013, 27, 3091–3100. [Google Scholar] [CrossRef] [PubMed]
- Hulmi, J.J.; Laakso, M.; Mero, A.A.; Häkkinen, K.; Ahtiainen, J.P.; Peltonen, H. The effects of whey protein with or without carbohydrates on resistance training adaptations. J. Int. Soc. Sports Nutr. 2015, 12, 48. [Google Scholar] [CrossRef] [PubMed]
- Lockwood, C.M. Effect of Whey Protein Quality on Physiological Response to Chronic Resistance Exercise in Trained Men: A Double-Blind, Placebo-Controlled, Randomized Trial, The University of Oklahoma: Oklahoma. 2010. Available online: https://shareok.org/handle/11244/318696 (accessed on 14 July 2019).
- Naclerio, F.; Seijo-Bujia, M.; Larumbe-Zabala, E.; Earnest, C.P. Carbohydrates Alone or Mixing with Beef or Whey Protein Promote Similar Training Outcomes in Resistance Training Males: A Double-Blind, Randomized Controlled Clinical Trial. Int. J. Sport Nutr. Exerc. Metab. 2017, 27, 408–420. [Google Scholar] [CrossRef] [PubMed]
- Naclerio, F.; Seijo, M.; Larumbe-Zabala, E.; Ashrafi, N.; Christides, T.; Karsten, B.; Nielsen, B.V. Effects of Supplementation with Beef or Whey Protein Versus Carbohydrate in Master Triathletes. J. Am. Coll. Nutr. 2017, 36, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Reidy, P.T.; Borack, M.S.; Markofski, M.M.; Dickinson, J.M.; Deer, R.R.; Husaini, S.H.; Walker, D.K.; Igbinigie, S.; Robertson, S.M.; Cope, M.B.; et al. Protein Supplementation Has Minimal Effects on Muscle Adaptations during Resistance Exercise Training in Young Men: A Double-Blind Randomized Clinical Trial. J. Nutr. 2016, 146, 1660–1669. [Google Scholar] [CrossRef] [PubMed]
- Taylor, L.W.; Wilborn, C.; Roberts, M.D.; White, A.; Dugan, K. Eight weeks of pre- and postexercise whey protein supplementation increases lean body mass and improves performance in Division III collegiate female basketball players. Appl. Physiol. Nutr. Metab. 2016, 41, 249–254. [Google Scholar] [CrossRef]
- Volek, J.S.; Volk, B.M.; Gómez, A.L.; Kunces, L.J.; Kupchak, B.R.; Freidenreich, D.J.; Aristizabal, J.C.; Saenz, C.; Dunn-Lewis, C.; Ballard, K.D.; et al. Whey Protein Supplementation During Resistance Training Augments Lean Body Mass. J. Am. Coll. Nutr. 2013, 32, 122–135. [Google Scholar] [CrossRef]
- Haraguchi, F.K.; de Abreu, W.C.; de Paula, H. Whey protein: Composition, nutritional properties, appications in sports and benefits for human health. Rev. Nutr. 2006, 19, 479–488. [Google Scholar] [CrossRef]
- Hayes, A.; Cribb, P.J. Effect of whey protein isolate on strength, body composition and muscle hypertrophy during resistance training. Curr. Opin. Clin. Nutr. Metab. Care 2008, 11, 40–44. [Google Scholar] [CrossRef]
- Schoenfeld, B.J.; Aragon, A.A. How much protein can the body use in a single meal for muscle-building? Implications for daily protein distribution. J. Int. Soc. Sports Nutr. 2018, 15, 10. [Google Scholar] [CrossRef] [PubMed]
- Morton, R.W.; Murphy, K.T.; McKellar, S.R.; Schoenfeld, B.J.; Henselmans, M.; Helms, E.; Aragon, A.A.; Devries, M.C.; Banfield, L.; Krieger, J.W.; et al. A systematic review, meta-analysis and meta-regression of the effect of protein supplementation on resistance training-induced gains in muscle mass and strength in healthy adults. Br. J. Sports Med. 2018, 52, 376–384. [Google Scholar] [CrossRef] [PubMed]
- Murphy, C.H.; Hector, A.J.; Phillips, S.M. Considerations for protein intake in managing weight loss in athletes. Eur. J. Sport Sci. 2015, 15, 21–28. [Google Scholar] [CrossRef] [PubMed]
- Mollahosseini, M.; Shab-Bidar, S.; Rahimi, M.H.; Djafarian, K. Effect of whey protein supplementation on long and short term appetite: A meta-analysis of randomized controlled trials. Clin. Nutr. ESPEN 2017, 20, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Hector, A.J.; Phillips, S.M. Protein Recommendations for Weight Loss in Elite Athletes: A Focus on Body Composition and Performance. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 170–177. [Google Scholar] [CrossRef] [PubMed]
- Tinsley, G.M.; La Bounty, P.M. Effects of intermittent fasting on body composition and clinical health markers in humans. Nutr. Rev. 2015, 73, 661–674. [Google Scholar] [CrossRef] [PubMed]
- Dwyer, J.; Coates, P.; Smith, M. Dietary Supplements: Regulatory Challenges and Research Resources. Nutrients 2018, 10, 41. [Google Scholar] [CrossRef] [PubMed]
- Pereira, T.T.; Pereira, M.; da G. de, L.; de Oliveira, C.M.V.; de Morais, C.S.; Ota, G.E.; Castro, L.H.A.; de Araújo, F.H.S.; Oesterreich, S.A.; de Alencar, G.P. Strength training variables: A brief bibliographic review. In The Production of Knowledge in Health Sciences; Atena Editora: Ponta Grossa, Paraná, Brazil, 2019; Volume IV, pp. 297–305. ISBN 9788572473019. [Google Scholar] [CrossRef]
- Hopkins, M.; Blundell, J.E. Energy balance, body composition, sedentariness and appetite regulation: Pathways to obesity. Clin. Sci. 2016, 130, 1615–1628. [Google Scholar] [CrossRef] [PubMed]
- Nose, H.; Kamijo, Y.; Masuki, S. Interactions between body fluid homeostasis and thermoregulation in humans. In Handbook of Clinical Neurology; Elsevier: Amsterdam, The Netherlands, 2018; Volume 156, pp. 417–429. ISBN 9780444639127. [Google Scholar] [CrossRef]
- Dulloo, A.G.; Jacquet, J.; Miles-Chan, J.L.; Schutz, Y. Passive and active roles of fat-free mass in the control of energy intake and body composition regulation. Eur. J. Clin. Nutr. 2017, 71, 353–357. [Google Scholar] [CrossRef] [PubMed]
- Nilaweera, K.N.; Speakman, J.R. Regulation of intestinal growth in response to variations in energy supply and demand: Adipose-hypothalamic-intestinal signalling. Obes. Rev. 2018, 19, 61–72. [Google Scholar] [CrossRef]
- Heydenreich, J.; Kayser, B.; Schutz, Y.; Melzer, K. Total Energy Expenditure, Energy Intake, and Body Composition in Endurance Athletes Across the Training Season: A Systematic Review. Sports Med. Open 2017, 3, 8. [Google Scholar] [CrossRef] [PubMed]
- Obert, J.; Pearlman, M.; Obert, L.; Chapin, S. Popular Weight Loss Strategies: A Review of Four Weight Loss Techniques. Curr. Gastroenterol. Rep. 2017, 19, 61. [Google Scholar] [CrossRef] [PubMed]
- Ralston, G.W.; Kilgore, L.; Wyatt, F.B.; Baker, J.S. The Effect of Weekly Set Volume on Strength Gain: A Meta-Analysis. Sports Med. 2017, 47, 2585–2601. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, H.; Wirth, K.; Keiner, M.; Mickel, C.; Sander, A.; Szilvas, E. Short-term Periodization Models: Effects on Strength and Speed-strength Performance. Sports Med. 2015, 45, 1373–1386. [Google Scholar] [CrossRef] [PubMed]
- Colonetti, T.; Grande, A.J.; Milton, K.; Foster, C.; Alexandre, M.C.M.; Uggioni, M.L.R.; Rosa, M.I. da Effects of whey protein supplement in the elderly submitted to resistance training: Systematic review and meta-analysis. Int. J. Food Sci. Nutr. 2017, 68, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Devries, M.C.; Phillips, S.M. Supplemental Protein in Support of Muscle Mass and Health: Advantage Whey: Whey, body composition, and muscle mass…. J. Food Sci. 2015, 80, A8–A15. [Google Scholar] [CrossRef] [PubMed]
- Schoenfeld, B.J.; Aragon, A.A.; Krieger, J.W. The effect of protein timing on muscle strength and hypertrophy: A meta-analysis. J. Int. Soc. Sports Nutr. 2013, 10, 53. [Google Scholar] [CrossRef]
- Naclerio, F.; Larumbe-Zabala, E. Effects of Whey Protein Alone or as Part of a Multi-ingredient Formulation on Strength, Fat-Free Mass, or Lean Body Mass in Resistance-Trained Individuals: A Meta-analysis. Sports Med. 2016, 46, 125–137. [Google Scholar] [CrossRef]
- Nissen, S.L.; Sharp, R.L. Effect of dietary supplements on lean mass and strength gains with resistance exercise: A meta-analysis. J. Appl. Physiol. 2003, 94, 651–659. [Google Scholar] [CrossRef]
- Miller, P.E.; Alexander, D.D.; Perez, V. Effects of Whey Protein and Resistance Exercise on Body Composition: A Meta-Analysis of Randomized Controlled Trials. J. Am. Coll. Nutr. 2014, 33, 163–175. [Google Scholar] [CrossRef]
- Li, M.; Liu, F. Effect of whey protein supplementation during resistance training sessions on body mass and muscular strength: A meta-analysis. Food Funct. 2019, 10, 2766–2773, in preparation. [Google Scholar] [CrossRef] [PubMed]
- Human Development Indices and Indicators: 2018 Statistical Update; UNDP, Ed.; United Nations Development Programme: New York, NY, USA, 2018; ISBN 9789211264371. Available online: http://hdr.undp.org/sites/default/files/2018_human_development_statistical_update.pdf (accessed on 14 July 2019).
- Jones, A.D. Critical review of the emerging research evidence on agricultural biodiversity, diet diversity, and nutritional status in low- and middle-income countries. Nutr. Rev. 2017, 75, 769–782. [Google Scholar] [CrossRef] [PubMed]
- Lindsay Smith, G.; Banting, L.; Eime, R.; O’Sullivan, G.; van Uffelen, J.G.Z. The association between social support and physical activity in older adults: A systematic review. Int. J. Behav. Nutr. Phys. Act. 2017, 14, 56. [Google Scholar] [CrossRef] [PubMed]
- Guthold, R.; Louazani, S.A.; Riley, L.M.; Cowan, M.J.; Bovet, P.; Damasceno, A.; Sambo, B.H.; Tesfaye, F.; Armstrong, T.P. Physical Activity in 22 African Countries. Am. J. Prev. Med. 2011, 41, 52–60. [Google Scholar] [CrossRef] [PubMed]

| Target | Description | Operator | Uniterms |
|---|---|---|---|
| Population | Healthy adults practicing physical activity | -- | physical activity; physical exercise; training; exercise; coaching; resistance training; resistance exercise; gym; fitness; crossfit; weight lifting; non-sedentary; athlete. |
| AND + NOT/AND NOT | child; pediatric; elder; aged; rat; mice. | ||
| Intervention | Whey protein supplementation (concentrated, hydrolyzed, and isolated) | AND | whey protein; whey; protein supplementation; whey supplementation; casein; casein supplementation; whey intake; casein intake; protein intake; whey concentrate; casein concentrate; concentrate protein; whey isolated; casein isolated; isolated protein; hydrolyzed whey; hydrolyzed casein; hydrolyzed protein; milk; milk protein; soy; soy protein. |
| Comparator | Randomized clinical trials | AND | controlled clinical trial; clinical trial; randomized clinical trial; group control; placebo; comparative study; cross-over; crossover; cross over; double-blind; factorial. |
| Outcome | Body composition related to hypertrophy | AND | muscle; mass gain; muscle gain; muscular gain; muscle strength; muscular strength; hypertrophy; lean mass; body composition; lean body mass; lean body tissue; fat-free mass; fat free mass; body weight; body mass; skeletal muscle. |
| RCT | Country | Age (Years) (Minimum-Maximum) | Physical Activity Level * | Trainings per Week (Mean) ** | Body Composition Measurement Method | Dose of WP (g·kg−1) | Nutritional Intervention | n (M:F) |
|---|---|---|---|---|---|---|---|---|
| Burke et al. [31] | Canada | 18–31 | Regular | 4 | DXA | 1.2 | WPI | 10:0 |
| Maltodextrin | 5:0 | |||||||
| Cribb et al. [32] | Australia | 18–31 | Regular | 4 | DXA | 1.28 | WPI | 5:0 |
| Non-specific carbohydrate | 7:0 | |||||||
| Herda et al. [33] | USA | 18.5–23.9 | Regular | 3 | Hydrostatic Weighing | 0.25 | WPC | 22:0 |
| Maltodextrin | 21:0 | |||||||
| Hulmi et al. [34] | Finland | 33.1–35.7 | Sporadic | 2.5 | DXA | 0.35 | WPC | 22:0 |
| Maltodextrin | 21:0 | |||||||
| Lockwood et al. [35] | USA | 18–35 | Regular | 2 | DXA | 0.37 | WPC | 13:0 |
| Dextrose | 15:0 | |||||||
| WPH | 13:0 | |||||||
| Dextrose | 15:0 | |||||||
| Naclerio et al. [36] | USA | 22–34 | Regular | 5 | Plethysmography | 0.26 | WPI | 8:0 |
| Non-specific carbohydrate | 8:0 | |||||||
| Naclerio et al. [37] | USA | 36.4–54.2 | Athlete | 3 | Plethysmography | 0.24 | WPI | 8:0 |
| Non-specific carbohydrate | 8:0 | |||||||
| Reidy et al. [38] | USA | 18–30 | Sporadic | 3 | DXA | 0.27 | WPI | 22:0 |
| Maltodextrin | 23:0 | |||||||
| Taylor et al. [39] *** | USA | 18–24 | Athlete | 4 | DXA | 0.36 | WPC | 0:8 |
| Maltodextrin | 0:6 | |||||||
| Volek et al. [40] *** | USA | 19.1–26.9 | Sporadic | 2.5 | DXA | 0.24 | WPC | 13:6 |
| Maltodextrin | 13:9 |
| RCT | FFM | FM | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| WP | Placebo | WP | Placebo | ||||||||||
| PRE | POST | Δ | PRE | POST | Δ | PRE | POST | Δ | PRE | POST | Δ | ||
| Burke et al. [31] | 62.30 ± 2.50 | 62.60 ± 2.50 | +0.30 | 61.80 ± 4.00 | 60.70 ± 4.00 | −1.10 | 13.90 ± 1.60 | 14.00 ± 1.60 | +0.10 | 9.50 ± 2.20 | 9.80 ± 2.20 | +0.30 | |
| Cribb et al. [32] | 59.00 ± 3.20 | 61.30 ± 3.00 | +2.30 | 62.30 ± 2.80 | 63.00 ± 2.70 | +0.70 | 10.60 ± 1.90 | 11.00 ± 1.60 | +0.40 | 13.2 ± 2.80 | 14.00 ± 2.90 | +0.80 | |
| Herda et al. [33] | 64.10 ± 2.10 | 65.10 ± 2.10 | +1.00 | 64.60 ± 1.60 | 65.70 ± 1.50 | +1.10 | 12.07 ± 0.80 | 12.72 ± 1.50 | +0.65 | 12.06 ± 1.10 | 12.93 ± 1.40 | +0.87 | |
| Hulmi et al. [34] | 59.81 ± 4.72 | 60.96 ± 5.13 | +1.15 | 60.02 ± 7.38 | 60.87 ± 7.30 | +0.85 | 20.79 ± 8.87 | 19.31 ± 8.43 | −1.48 | 18.40 ± 9.18 | 18.39 ± 9.41 | −0.01 | |
| Lockwood et al. [35] | WPC | 63.63 ± 1.34 | 65.50 ± 1.32 | +1.86 | 59.10 ± 1.54 | 60.55 ± 1.51 | +1.45 | 17.06 ± 1.08 | 16.83 ± 1.04 | −0.22 | 9.46 ± 1.23 | 10.32 ± 1.19 | +0.86 |
| WPH | 60.51 ± 1.40 | 62.39 ± 13.74 | +1.88 | 59.10 ± 1.54 | 60.55 ± 1.51 | +1.45 | 18.94 ± 1.12 | 17.82 ± 1.08 | −1.12 | 9.46 ± 1.23 | 10.32 ± 1.19 | +0.86 | |
| Naclerio et al. [36] | 66.10 ± 5.75 | 66.98 ± 6.12 | +0.88 | 64.15 ± 7.28 | 64.23 ± 7.27 | +0.08 | 11.94 ± 4.08 | 11.41 ± 4.81 | −0.53 | 13.91 ± 7.48 | 14.18 ± 7.32 | +0.27 | |
| Naclerio et al. [37] | 65.40 ± 6.10 | 65.80 ± 6.80 | +0.40 | 64.70 ± 5.90 | 64.70 ± 5.50 | 0.00 | 19.70 ± 8.40 | 18.9 ± 10.00 | −0.80 | 15.40 ± 4.90 | 14.80 ± 5.00 | −0.60 | |
| Reidy et al. [38] | 57.60 ± 1.50 | 60.00 ± 2.00 | +2.40 | 55.20 ± 1.50 | 57.40 ± 1.70 | +2.20 | 20.50 ± 1.30 | 19.00 ± 1.70 | −1.50 | 18.40 ± 1.70 | 17.80 ± 1.80 | −0.60 | |
| RCT | Intensity | Volume | Total Log Duration (days) | Body Work | Outside Parallel Training | ||
|---|---|---|---|---|---|---|---|
| Blocks | Duration (Days) | N° of Sets | |||||
| Burke et al. [31] | Adaptable to each participant’s progress based on RM | 5 | 8 | 2 | 40 | Whole body | Not mentioned |
| Cribb et al. [32] | 70–95% RM | 3 | 14–35 | 2–3 | 70 | Whole body | Allowed, recorded, and analyzed |
| Herda et al. [33] | 80% RM | 2 | 7 | 2–3 | 56 | Lower limbs | Allowed, but not registered |
| Hulmi et al. [34] | Adaptable to each participant’s progress based on the strength test (2–6 RM) | 3 | 28 | 2–3 | 84 | Whole body | Allowed for recreational activities, but not registered |
| Lockwood et al. [35] | 5–10 RM | 2 × 4 | 7 | 3 | 56 | Whole body | Not mentioned |
| Naclerio et al. [36] | Adaptable to each participant’s progress based on RM | 8 | 7 | 1 | 56 | Whole body | Allowed in the protocol intervals, but only for recreational activities and not recorded |
| Naclerio et al. [37] | Adaptable to each participant’s progress based on the VT2 and HRmax | 4–6 | 14 | 3 | 70 | Whole body | The usual training routine was used as an intervention protocol |
| Reidy et al. [38] | 60–80% do RM | 2 | 28–56 | 3–4 | 84 | Whole body | Prohibited for force activity and allowed for recreational activities, but not recorded |
| Outcome | Participants (n) (WP: Placebo) | RCT (n) | WMD (95% CI) | Statistical Significance * | Quality of Evidence (GRADE) | Due to | Comments | ||
|---|---|---|---|---|---|---|---|---|---|
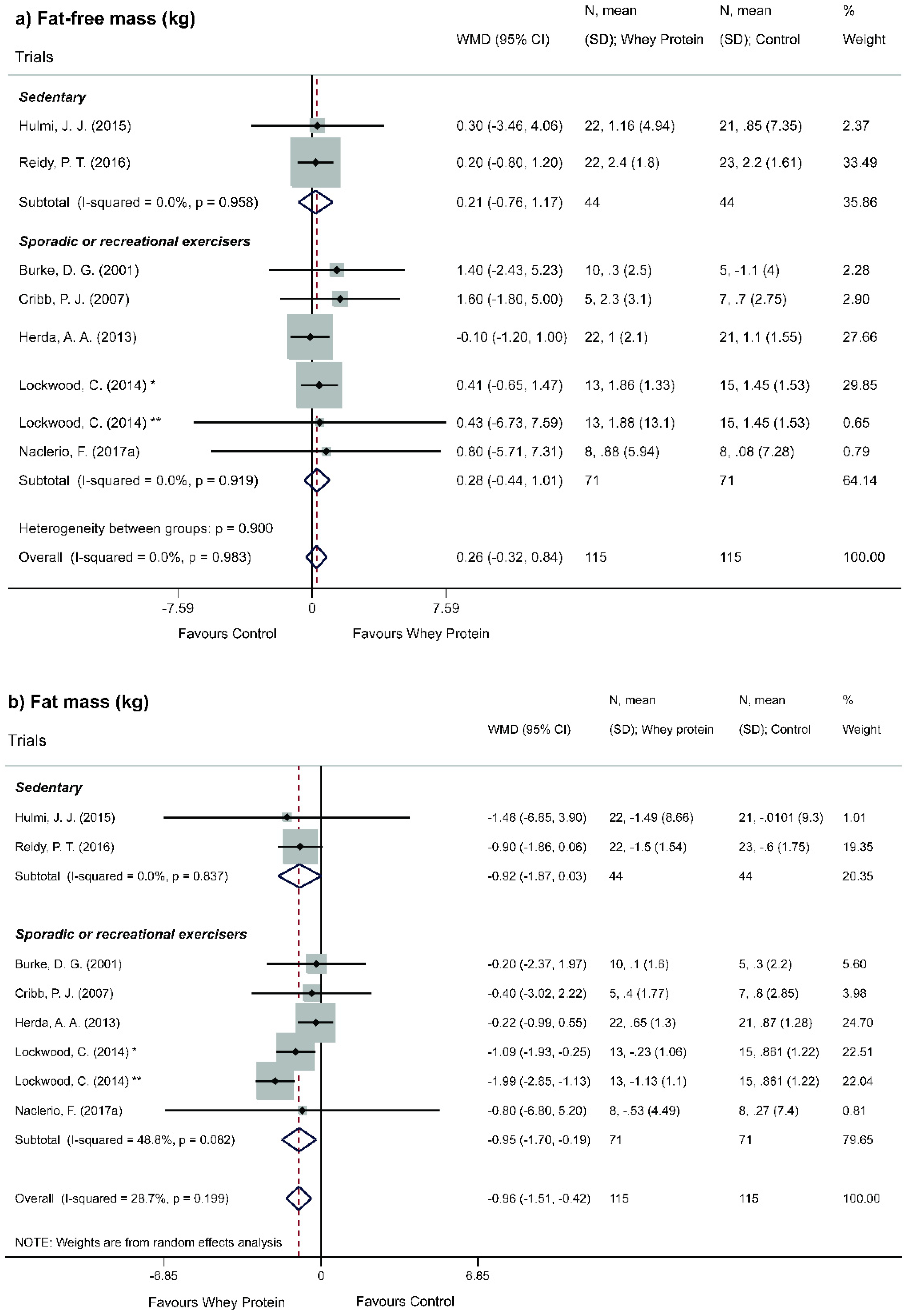
| FFM | Global | 123:123 | 8 | 0.26 (−0.32, −0.83) | None | ⊕⊕◯◯◯ Low | Imprecision | Range of effect possibly underestimated by random statistics and low number of events. | |
| WP type | WPC | 57:57 | 3 | 0.17 (−0.58, 0.92) | None | ⊕⊕◯◯◯ Low | Imprecision | Low sample size. | |
| WPI | 53:51 | 6 | 0.38 (−0.53, 1.30) | ||||||
| Protein percentage (%) | 96–100 | 27:28 | 2 | 0.06 (−0.99, 1.11) | None | ⊕⊕◯◯◯ Low | Risk of bias | Only one study evaluated at low risk, others had consistent failures in randomization and in the selection of the reported result. | |
| 81–95 | 38:39 | 3 | 0.22 (−0.76, 1.19) | ||||||
| 51–80 | 48:51 | 2 | 0.40 (−0.61, 1.41) | ||||||
| Physical activity level | Sporadic or recreational | 44:44 | 2 | 0.21 (−0.76, 1.17) | None | ⊕⊕⊕◯◯ Moderate | Risk of bias | Presence of deviations from the intended interventions and inconsistencies in randomization. | |
| Regular | 71:71 | 5 | 0.26 (−0.32, 0.84) | ||||||
| FM | Global | 123:123 | 8 | −0.96 (−1.37, −0.55) | p < 0.001 | ⊕⊕⊕⊕◯ High | Risk of bias | Only one low-risk study. | |
| WP type | WPC | 57:57 | 3 | −0.63 (−1.19, −0.06) | p = 0.030 | ⊕⊕⊕⊕◯ High | Risk of bias | Only one low-risk study. | |
| WPI | 110:108 | 5 | −0.66 (−1.13, −0.20) | None | |||||
| Protein percentage (%) | 96–100 | 27:28 | 2 | −0.23 (−0.97, 0.50) | None | ⊕⊕⊕◯◯ Moderate | Imprecision | Range of effect possibly underestimated by random statistics and low number of events. | |
| 81–95 | 38:39 | 3 | −0.89 (−1.83, 0.06) | None | |||||
| 51–80 | 48:51 | 2 | −1.53 (−2.13, −0.93) | p < 0.001 | |||||
| Global | 113:118 | 7 | −0.99 (−1.41, −0.57) | - | ⊕◯◯◯◯ Very low | Indirect evidence | Metaregression not performed by the low number of studies (Figure 5b). | ||
| Physical activity level | Sporadic or recreational | 44:44 | 2 | −0.92 (−1.87, 0.03) | None | ⊕⊕⊕◯◯ Moderate | Risk of bias | Measure flaw (participants were not under the same restrictions to practice physical activity outside the study). | |
| Regular | 71:71 | 5 | −0.95 (−1.70, −0.19) | p = 0.014 | |||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
A. Castro, L.H.; S. de Araújo, F.H.; M. Olimpio, M.Y.; B. de B. Primo, R.; T. Pereira, T.; F. Lopes, L.A.; B. S. de M. Trindade, E.; Fernandes, R.; A. Oesterreich, S. Comparative Meta-Analysis of the Effect of Concentrated, Hydrolyzed, and Isolated Whey Protein Supplementation on Body Composition of Physical Activity Practitioners. Nutrients 2019, 11, 2047. https://doi.org/10.3390/nu11092047
A. Castro LH, S. de Araújo FH, M. Olimpio MY, B. de B. Primo R, T. Pereira T, F. Lopes LA, B. S. de M. Trindade E, Fernandes R, A. Oesterreich S. Comparative Meta-Analysis of the Effect of Concentrated, Hydrolyzed, and Isolated Whey Protein Supplementation on Body Composition of Physical Activity Practitioners. Nutrients. 2019; 11(9):2047. https://doi.org/10.3390/nu11092047
Chicago/Turabian StyleA. Castro, Luis Henrique, Flávio Henrique S. de Araújo, Mi Ye M. Olimpio, Raquel B. de B. Primo, Thiago T. Pereira, Luiz Augusto F. Lopes, Erasmo B. S. de M. Trindade, Ricardo Fernandes, and Silvia A. Oesterreich. 2019. "Comparative Meta-Analysis of the Effect of Concentrated, Hydrolyzed, and Isolated Whey Protein Supplementation on Body Composition of Physical Activity Practitioners" Nutrients 11, no. 9: 2047. https://doi.org/10.3390/nu11092047
APA StyleA. Castro, L. H., S. de Araújo, F. H., M. Olimpio, M. Y., B. de B. Primo, R., T. Pereira, T., F. Lopes, L. A., B. S. de M. Trindade, E., Fernandes, R., & A. Oesterreich, S. (2019). Comparative Meta-Analysis of the Effect of Concentrated, Hydrolyzed, and Isolated Whey Protein Supplementation on Body Composition of Physical Activity Practitioners. Nutrients, 11(9), 2047. https://doi.org/10.3390/nu11092047





