Effectiveness of Non-Animal Chondroitin Sulfate Supplementation in the Treatment of Moderate Knee Osteoarthritis in a Group of Overweight Subjects: A Randomized, Double-Blind, Placebo-Controlled Pilot Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Population
2.2. Inclusion/Exclusion Criteria
2.3. Description of the Intervention
2.4. Assessment of Experimental Product Tolerance
2.5. Assessment of the Effectiveness of the Experimental Product
2.6. Pain and Knee Function Assessment
2.6.1. Assessment of knee function by WOMAC Index
2.6.2. Assessment of knee symptoms and function by the TLKS Scale
2.6.3. Visual Analogue Scale of Pain
2.7. Assessment of Quality of Life
2.8. Inflammation Biochemical Parameters
2.9. Assessment of Body Composition
2.10. Run-In, Randomization and Masking
2.11. Sample Size
2.12. Statistical Analysis
3. Results
3.1. Characteristics at Baseline
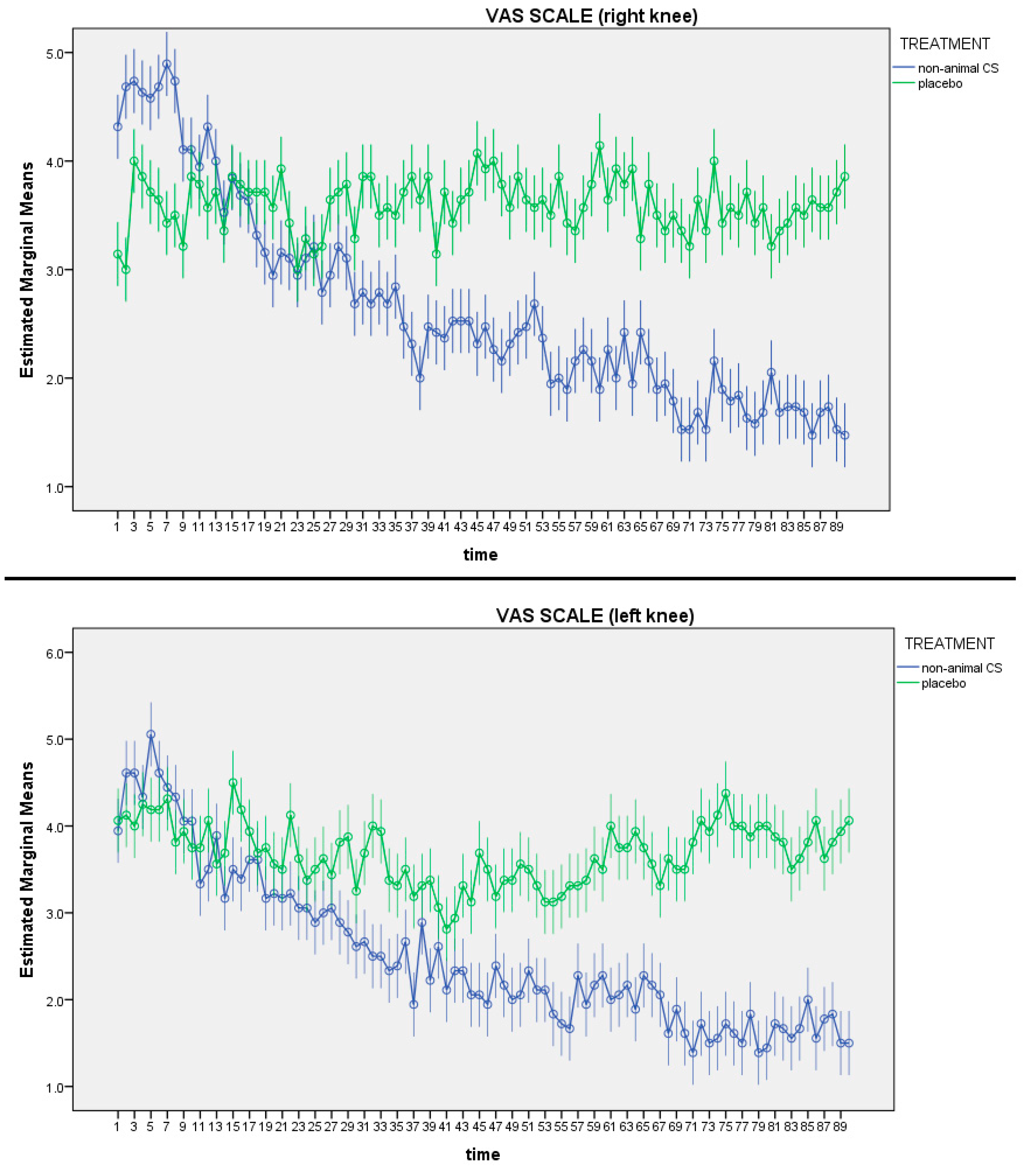
3.2. Pain and Motility
3.3. Inflammation
3.4. Body Composition
3.5. Quality of Life
3.6. Side Effects
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cross, M.; Smith, E.; Hoy, D.; Nolte, S.; Ackerman, I.; Fransen, M.; Bridgett, L.; Williams, S.; Guillemin, F.; Hill, C.L.; et al. The global burden of hip and knee osteoarthritis: Estimates from the Global Burden of Disease 2010 study. Ann. Rheum. Dis. 2014, 73, 1323–1330. [Google Scholar] [CrossRef] [PubMed]
- Paoloni, M.; Bernetti, A.; Belelli, A.; Brignoli, O.; Buoso, S.; Caputi, A.P.; Catani, F.; Coclite, D.; Fini, M.; Mantovani, L.; et al. Appropriateness of clinical and organizational criteria for intra-articular injectiontherapies in osteoarthritis. A Delphi method consensus initiative among experts in Italy. Ann. Ist. Super. Sanita 2015, 51, 131–138. [Google Scholar] [PubMed]
- De Filippis, L.; Gulli, S.; Caliri, A.; Romano, C.; Munaò, F.; Trimarchi, G.; La Torre, D.; Fichera, C.; Pappalardo, A.; Triolo, G.; et al. Epidemiology and risk factors in osteoarthritis: Literature review data from “OASIS” study. Reumatismo 2011, 56, 169–184. [Google Scholar] [CrossRef]
- Silverwood, V.; Blagojevic-Bucknall, M.; Jinks, C.; Jordan, J.L.; Protheroe, J.; Jordan, K.P. Current evidence on risk factors for knee osteoarthritis in older adults: A systematic review and meta-analysis. Osteoarthr. Cartil. 2015, 23, 507–515. [Google Scholar] [CrossRef] [PubMed]
- Grotle, M.; Hagen, K.B.; Natvig, B.; Dahl, F.A.; Kvien, T.K. Obesity and osteoarthritis in knee, hip and/or hand: An epidemiological study in the general population with 10 years follow-up. BMC Musculoskelet. Disord. 2008, 9, 132. [Google Scholar] [CrossRef] [PubMed]
- Blagojevic, M.; Jinks, C.; Jeffery, A.; Jordan, K.P. Risk factors for onset of osteoarthritis of the knee in older adults: A systematic review and meta-analysis. Osteoarthr. Cartil. 2010, 18, 24–33. [Google Scholar] [CrossRef] [PubMed]
- Kulkarni, K.; Karssiens, T.; Kumar, V.; Pandit, H. Obesity and osteoarthritis. Maturitas 2016, 89, 22–28. [Google Scholar] [CrossRef]
- Eaton, C.B. Obesity As a Risk Factor for Osteoarthritis: Mechanical Versus Metabolic. Med. Health R. I. 2004, 87, 201–204. [Google Scholar] [PubMed]
- Iannone, F.; Lapadula, G. Obesity and inflammation—Targets for OA therapy. Curr. Drug Targets 2010, 11, 586–598. [Google Scholar] [CrossRef]
- Bruyère, O.; Cooper, C.; Cutolo, M.; Reginster, J.Y. International endorsement of the ESCEO algorithm for management of knee osteoarthritis in clinical practice. Semin. Arthritis Rheum. 2017, 47, e10. [Google Scholar] [CrossRef]
- Jordan, K.M.; Arden, N.K.; Doherty, M.; Bannwarth, B.; Bijlsma, J.W.J.; Dieppe, P.; Günther, K.; Hauselmann, H.; Herrero-Beaumont, G.; Kaklamanis, P.; et al. EULAR Recommendations 2003: An evidence based approach to the management of knee osteoarthritis: Report of a Task Force of the Standing Committee for International Clinical Studies Including Therapeutic Trials (ESCISIT). Ann. Rheum. Dis. 2003, 62, 1145–1155. [Google Scholar] [CrossRef] [PubMed]
- McAlindon, T.; Bannuru, R.; Sullivan, M.; Arden, N.; Berenbaum, F.; Bierma-Zeinstra, S.; Hawker, G.; Henrotin, Y.; Hunter, D.; Kawaguchi, H.; et al. OARSI guidelines for the non-surgical management of knee osteoarthritis. Osteoarthr. Cartil. 2014, 22, 363–388. [Google Scholar] [CrossRef] [PubMed]
- Henrotin, Y.; Marty, M.; Mobasheri, A. What is the current status of chondroitin sulfate and glucosamine for the treatment of knee osteoarthritis? Maturitas 2014, 78, 184–187. [Google Scholar] [CrossRef] [PubMed]
- Vallières, M.; du Souich, P. Modulation of inflammation by chondroitin sulfate. Osteoarthr. Cartil. 2010, 18, S1–S6. [Google Scholar] [CrossRef] [PubMed]
- Du Souich, P.; García, A.G.; Vergés, J.; Montell, E. Immunomodulatory and anti-inflammatory effects of chondroitin sulphate. J. Cell. Mol. Med. 2009, 13, 1451–1463. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.H.; Woo, J.H.; Choi, S.J.; Ji, J.D.; Song, G.G. Effect of glucosamine or chondroitin sulfate on the osteoarthritis progression: A meta-analysis. Rheumatol. Int. 2010, 30, 357–363. [Google Scholar] [CrossRef] [PubMed]
- Melgar-Lesmes, P.; Garcia-Polite, F.; Del-Rey-Puech, P.; Rosas, E.; Dreyfuss, J.L.; Montell, E.; Vergés, J.; Edelman, E.R.; Balcells, M. Treatment with chondroitin sulfate to modulate inflammation and atherogenesis in obesity. Atherosclerosis 2016, 245, 82–87. [Google Scholar] [CrossRef]
- Stabler, T.V.; Montell, E.; Vergés, J.; Huebner, J.L.; Kraus, V.B. Chondroitin Sulfate Inhibits Monocyte Chemoattractant Protein-1 Release From 3T3-L1 Adipocytes: A New Treatment Opportunity for Obesity-Related Inflammation? Biomark. Insights 2017, 12. [Google Scholar] [CrossRef]
- Yusuf, E.; Nelissen, R.G.; Ioan-Facsinay, A.; Stojanovic-Susulic, V.; DeGroot, J.; van Osch, G.; Middeldorp, S.; Huizinga, T.W.; Kloppenburg, M. Association between weight or body mass index and hand osteoarthritis: A systematic review. Ann. Rheum. Dis. 2010, 69, 761–765. [Google Scholar] [CrossRef]
- Bauerová, K.; Ponist, S.; Kuncirova, V.; Dráfi, F.; Mihalova, D.; Paulovicova, E.; Volpi, N. Effect of nonanimal high- and low-molecular-mass chondroitin sulfates produced by a biotechnological process in an animal model of polyarthritis. Pharmacology 2014, 94, 109–114. [Google Scholar] [CrossRef]
- Singh, J.A.; Noorbaloochi, S.; Macdonald, R.; Maxwell, L.J. Chondroitin for osteoarthritis. Cochrane Database Syst. Rev. 2015, 1, CD005614. [Google Scholar] [PubMed]
- Miraglia, N.; Bianch, D.; Trentin, A.; Volpi, N.; Soni, M.G. Safety assessment of non-animal chondroitin sulfate sodium: Subchronic study in rats, genotoxicity tests and human bioavailability. Food Chem. Toxicol. 2016, 93, 89–101. [Google Scholar] [CrossRef] [PubMed]
- Pomin, V.H.; Piquet, A.A.; Pereira, M.S.; Mourão, P.A.S. Residual keratan sulfate in chondroitin sulfate formulations for oral administration. Carbohydr. Polym. 2012, 90, 839–846. [Google Scholar] [CrossRef] [PubMed]
- Galeotti, F.; Maccari, F.; Volpi, N. Selective removal of keratan sulfate in chondroitin sulfate samples by sequential precipitation with ethanol. Anal. Biochem. 2014, 448, 113–115. [Google Scholar] [CrossRef] [PubMed]
- Schiraldi, C.; Cimini, D.; De Rosa, M. Production of chondroitin sulfate and chondroitin. Appl. Microbiol. Biotechnol. 2010, 87, 1209–1220. [Google Scholar] [CrossRef] [PubMed]
- Volpi, N.; Mantovani, V.; Galeotti, F.; Bianchi, D.; Straniero, V.; Valoti, E.; Miraglia, N. Oral Bioavailability and Pharmacokinetics of Nonanimal Chondroitin Sulfate and Its Constituents in Healthy Male Volunteers. Clin. Pharmacol. Drug Dev. 2018, 8, 336–345. [Google Scholar] [CrossRef] [PubMed]
- Bellamy, N.; Buchanan, W.W.; Goldsmith, C.H.; Campbell, J.; Stitt, L.W. Validation study of WOMAC: A health status instrument for measuring clinically important patient relevant outcomes to antirheumatic drug therapy in patients with osteoarthritis of the hip or knee. J. Rheumatol. 1988, 15, 1833–1840. [Google Scholar] [PubMed]
- Lysholm, J.; Gillquist, J. Evaluation of knee ligament surgery results with special emphasis on use of a scoring scale. Am. J. Sports Med. 1982, 10, 150–154. [Google Scholar] [CrossRef] [PubMed]
- Wewers, M.E.; Lowe, N.K. A critical review of visual analogue scales in the measurement of clinical phenomena. Res. Nurs. Health 1990, 13, 227–236. [Google Scholar] [CrossRef]
- McHorney, C.A.; Ware, J.E.; Raczek, A.E. The MOS 36-item short-form health survey (Sf-36): II. Psychometric and clinical tests of validity in measuring physical and mental health constructs. Med. Care 1993, 31, 247–263. [Google Scholar] [CrossRef]
- Baumgartner, R.N.; Koehler, K.M.; Gallagher, D.; Romero, L.; Heymsfield, S.B.; Ross, R.R.; Garry, P.J.; Lindeman, R.D. Epidemiology of Sarcopenia among the Elderly in New Mexico. Am. J. Epidemiol. 1998, 147, 755–763. [Google Scholar] [CrossRef]
- Mohammad, A.; Rolfe, E.D.L.; Sleigh, A.; Kivisild, T.; Behbehani, K.; Wareham, N.J.; Brage, S.; Mohammad, T. Validity of visceral adiposity estimates from DXA against MRI in Kuwaiti men and women. Nutr. Diabetes 2017, 7, e238. [Google Scholar] [CrossRef] [PubMed]
- Cohen, M.; Wolfe, R.; Mai, T.; Lewis, D. A randomized, double blind, placebo controlled trial of a topical cream containing glucosamine sulfate, chondroitin sulfate, and camphor for osteoarthritis of the knee. J. Rheumatol. 2003, 30, 523–528. [Google Scholar] [PubMed]
- Honvo, G.; Bruyère, O.; Geerinck, A.; Veronese, N.; Reginster, J.-Y. Efficacy of Chondroitin Sulfate in Patients with Knee Osteoarthritis: A Comprehensive Meta-Analysis Exploring Inconsistencies in Randomized, Placebo-Controlled Trials. Adv. Ther. 2019, 36, 1085–1099. [Google Scholar] [CrossRef] [PubMed]
- Volpi, N. Quality of different chondroitin sulfate preparations in relation to their therapeutic activity. J. Pharm. Pharmacol. 2009, 61, 1271–1280. [Google Scholar] [CrossRef] [PubMed]
- Bruyère, O.; Cooper, C.; Pelletier, J.-P.; Maheu, E.; Rannou, F.; Branco, J.; Brandi, M.L.; Kanis, J.A.; Altman, R.D.; Hochberg, M.C.; et al. A consensus statement on the European Society for Clinical and Economic Aspects of Osteoporosis and Osteoarthritis (ESCEO) algorithm for the management of knee osteoarthritis-From evidence-based medicine to the real-life setting. Semin. Arthritis Rheum. 2016, 45, S3–S11. [Google Scholar] [CrossRef]
- Hanada, M.; Takahashi, M.; Furuhashi, H.; Koyama, H.; Matsuyama, Y. Elevated erythrocyte sedimentation rate and high-sensitivity C-reactive protein in osteoarthritis of the knee: Relationship with clinical findings and radiographic severity. Ann. Clin. Biochem. 2016, 53, 548–553. [Google Scholar] [CrossRef] [PubMed]
- Jin, X.; Beguerie, J.R.; Zhang, W.; Blizzard, L.; Otahal, P.; Jones, G.; Ding, C. Circulating C reactive protein in osteoarthritis: A systematic review and meta-analysis. Ann. Rheum. Dis. 2015, 74, 703–710. [Google Scholar] [CrossRef] [PubMed]
- Iovu, M.; Dumais, G.; Du Souich, P. Anti-inflammatory activity of chondroitin sulfate. Osteoarthr. Cartil. 2008, 16, S14–S18. [Google Scholar] [CrossRef] [PubMed]
- Park, H.S.; Park, J.Y.; Yu, R. Relationship of obesity and visceral adiposity with serum concentrations of CRP, TNF-α and IL-6. Diabetes Res. Clin. Pract. 2005, 69, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Hochberg, M.C.; Martel-Pelletier, J.; Monfort, J.; Möller, I.; Castillo, J.R.; Arden, N.; Berenbaum, F.; Blanco, F.J.; Conaghan, P.G.; Doménech, G.; et al. Combined chondroitin sulfate and glucosamine for painful knee osteoarthritis: A multicentre, randomised, double-blind, non-inferiority trial versus celecoxib. Ann. Rheum. Dis. 2016, 75, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Clegg, D.O.; Reda, D.J.; Harris, C.L.; Klein, M.A.; O’Dell, J.R.; Hooper, M.M.; Bradley, J.D.; Bingham, C.O.; Weisman, M.H.; Jackson, C.G.; et al. Glucosamine, Chondroitin Sulfate, and the Two in Combination for Painful Knee Osteoarthritis. N. Engl. J. Med. 2006, 354, 795–808. [Google Scholar] [CrossRef] [PubMed]
| Treatment | Duration | OA Assessment Times |
|---|---|---|
| Placebo (30 subjects) | 12 weeks | Pretreatment (T0), 4 week (T1), 12 week (T2) |
| Non animal CS (30 subjects) | 12 weeks | Pretreatment (T0), 4 week (T1), 12 week (T2) |
| Variables | PLACEBO (Mean; SD) | Non Animal CS (Mean; SD) | TOTAL SAMPLE (Mean; SD) | P Value between Groups at Baseline |
|---|---|---|---|---|
| Demographic Characteristics | ||||
| Gender (F: Female/M: Male) | 30 (0F/10M) | 30 (18F/12M) | 60 (38F/22M) | 0.568 |
| Age (years) | 62.77 (8.23) | 62.53 (9.25) | 62.64 (8.73) | 0.924 |
| Height (m) | 1.62 (0.12) | 1.59 (0.10) | 1.60 (0.11) | 0.321 |
| Weight (kg) | 74.52 (20.34) | 71.76 (16.62) | 72.98 (18.20) | 0.609 |
| Body composition | ||||
| BMI (kg/m2) | 27.56 (3.38) | 27.88 (3.60) | 27.74 (3.47) | 0.743 |
| FFM (g) | 41,977.77 (9000.14) | 42,777.42 (8123.84) | 42,425.58 (8440.48) | 0.746 |
| FM (g) | 30,377.13 (12,528.26) | 26,641.00 (12,845.20) | 28,284.90 (12,715.93) | 0.300 |
| VAT | 1185.50 (997.38) | 1087.11 (725.64) | 1131.28 (850.22) | 0.701 |
| Knee pain and function | ||||
| TLKS (0–100) pts | 68.31 (12.00) | 64.17 (14.64) | 66.00 (13.57) | 0.278 |
| WOMAC (0–96) pts | 42.93 (14.35) | 46.22 (15.73) | 44.77 (15.07) | 0.445 |
| Quality of life | ||||
| SF-36: Physical Activity | 67.04 (25.66) | 58.75 (16.97) | 62.40 (21.41) | 0.201 |
| SF-36: Physical Role Limitations | 59.77 (39.26) | 54.46 (44.12) | 56.80 (41.72) | 0.655 |
| SF-36: Physical Pain | 49.00 (12.99) | 46.32 (16.34) | 47.50 (14.88) | 0.552 |
| SF-36: General Health | 55.59 (15.17) | 55.17 (16.22) | 55.36 (15.60) | 0.927 |
| SF-36: Vitality | 50.22 (13.75) | 53.75 (16.47) | 52.20 (15.29) | 0.404 |
| SF-36: Social Activities | 71.90 (19.24) | 70.28 (18.99) | 71.00 (18.92) | 0.767 |
| SF-36: Emotional Role Limitations | 77.18 (37.69) | 71.32 (39.31) | 73.90 (38.32) | 0.595 |
| SF-36: Mental Health | 58.00 (17.94) | 64.14 (16.24) | 60.97 (17.27) | 0.235 |
| Inflammation | ||||
| ESR (mm/h) | 22.31 (20.01) | 16.42 (6.80) | 19.02 (14.35) | 0.198 |
| CRP (mg/dL) | 0.22 (0.26) | 0.18 (0.24) | 0.20 (0.25) | 0.603 |
| Variables | PLACEBO Intra-Group Δ Change and CI 95% | Non animal CS Intra-Group Δ Change and CI 95% | Treatment Effect between Groups (Non-Animal CS Minus Placebo) Δ Change and CI 95% | p-Value between Groups |
|---|---|---|---|---|
| Body composition outcomes | ||||
| Weight (kg) | −2.29 (−8.61; 4.02) | −3.75 (−9.37; 1.88) | −1.45 (−9.91; 7.01) | 0.507 |
| BMI (kg/m2) | 0.07 (−0.22; 0.36) | −0.18 (−0.45; 0.08) | −0.253 (−0.65; 0.14) | 0.140 |
| FFM (g) | 81.92 (−466.79; 630.62) | 538.53 * (52.16; 1024.90) | 456.61 (−276.66; 1189.89) | 0.150 |
| FM (g) | −68.70 (−810.54; 673.14) | −719.38 (−1376.94; −61.81) | −650.67 (−1642.05; 340.70) | 0.134 |
| VAT (g) | −223.41(−391.89; −54.94) | −164.16 (−335.52; +7.21) | 59.26 (−181.39; 299.91) | 0.240 |
| Knee pain and function | ||||
| TLKS (score) | −1.04 (−4.83; 7,75) | 9.60 * (6.24; 12.96) | 10.64 (5.57; 15.70) | p < 0.001 |
| WOMAC (score) | 3.54 (0.65; 6.42) | −8.70 * (−11.25; −6.14) | −12.24 (−16.01; −8.38) | p < 0.001 |
| Quality of life | ||||
| SF-36: Physical Activity (score) | 0.05 (−4.54; 5.93) | 5.99 * (1.43; 10.55) | 5.294 (−1.65; 12.23) | 0.091 |
| SF-36: Physical Role Limitations (score) | −4.19 (−19.47; 11.09) | 6.14 (−7.16; 19.45) | −10.33 (−30.54; 9.92) | 0.215 |
| SF-36: Physical Pain (score) | −0.58 (−7.75; +6.60) | 8.81 * (2.57; 15.06) | 9.39 (−0.12; 18.91) | 0.053 |
| SF-36: General Health (score) | 0.06 (−6.11; 6.24) | −0.32 (−5.70; 5.05) | −0.39 (−8.58; +7.80) | 0.641 |
| SF-36: Vitality (score) | 0,10 (−5.25; 8.84) | 0.53 (−5.60; +6.67) | −1.26 (−10.60; 8.08) | 0.546 |
| SF-36: Social Activities (score) | −8.09 (−17.14; 0.94) | −0.37 (−8.24; 7.50) | 7.72 (−4.26; 19.71) | 0.139 |
| SF-36: Emotional Role limitations (score) | −6.25 (−19.97; 7.46) | 1.30 (−13.23; 10.75) | 4.96 (−13.23; 23.51) | 0.406 |
| SF-36: Mental Health (score) | −2.00 (−8.90; 4.90) | −4.07 (−9.84; 1.71) | −2.06 (−11.70; 6.94) | 0.449 |
| Inflammation markers | ||||
| ESR (mm/h) | 0.04 (−2.56; 3.75) | −4.41 * (−7.14; −1.69) | −5.01 (−9.18; −0.84) | p < 0.01 |
| CRP (mg/dL) | 0.21 (−2.56; 3.75) | −0.08 * (−0.16; −0.01) | −0.14 (−0.26; −0.04) | p < 0.01 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rondanelli, M.; Braschi, V.; Gasparri, C.; Nichetti, M.; Faliva, M.A.; Peroni, G.; Naso, M.; Iannello, G.; Spadaccini, D.; Miraglia, N.; et al. Effectiveness of Non-Animal Chondroitin Sulfate Supplementation in the Treatment of Moderate Knee Osteoarthritis in a Group of Overweight Subjects: A Randomized, Double-Blind, Placebo-Controlled Pilot Study. Nutrients 2019, 11, 2027. https://doi.org/10.3390/nu11092027
Rondanelli M, Braschi V, Gasparri C, Nichetti M, Faliva MA, Peroni G, Naso M, Iannello G, Spadaccini D, Miraglia N, et al. Effectiveness of Non-Animal Chondroitin Sulfate Supplementation in the Treatment of Moderate Knee Osteoarthritis in a Group of Overweight Subjects: A Randomized, Double-Blind, Placebo-Controlled Pilot Study. Nutrients. 2019; 11(9):2027. https://doi.org/10.3390/nu11092027
Chicago/Turabian StyleRondanelli, Mariangela, Valentina Braschi, Clara Gasparri, Mara Nichetti, Milena Anna Faliva, Gabriella Peroni, Maurizio Naso, Giancarlo Iannello, Daniele Spadaccini, Niccolò Miraglia, and et al. 2019. "Effectiveness of Non-Animal Chondroitin Sulfate Supplementation in the Treatment of Moderate Knee Osteoarthritis in a Group of Overweight Subjects: A Randomized, Double-Blind, Placebo-Controlled Pilot Study" Nutrients 11, no. 9: 2027. https://doi.org/10.3390/nu11092027
APA StyleRondanelli, M., Braschi, V., Gasparri, C., Nichetti, M., Faliva, M. A., Peroni, G., Naso, M., Iannello, G., Spadaccini, D., Miraglia, N., Putignano, P., Alalwan, T. A., & Perna, S. (2019). Effectiveness of Non-Animal Chondroitin Sulfate Supplementation in the Treatment of Moderate Knee Osteoarthritis in a Group of Overweight Subjects: A Randomized, Double-Blind, Placebo-Controlled Pilot Study. Nutrients, 11(9), 2027. https://doi.org/10.3390/nu11092027






