Native Hypovitaminosis D in CKD Patients: From Experimental Evidence to Clinical Practice
Abstract
1. Introduction
2. Native Hypovitaminosis D during Chronic Kidney Disease (CKD): The Size of the Problem
2.1. Vitamin D Metabolism and Status Categorization in CKD Patients
2.2. Native Hypovitaminosis D Epidemiology in CKD Patients
2.3. Native Vitamin D Supplementation: Dose, Interval,Toxicity
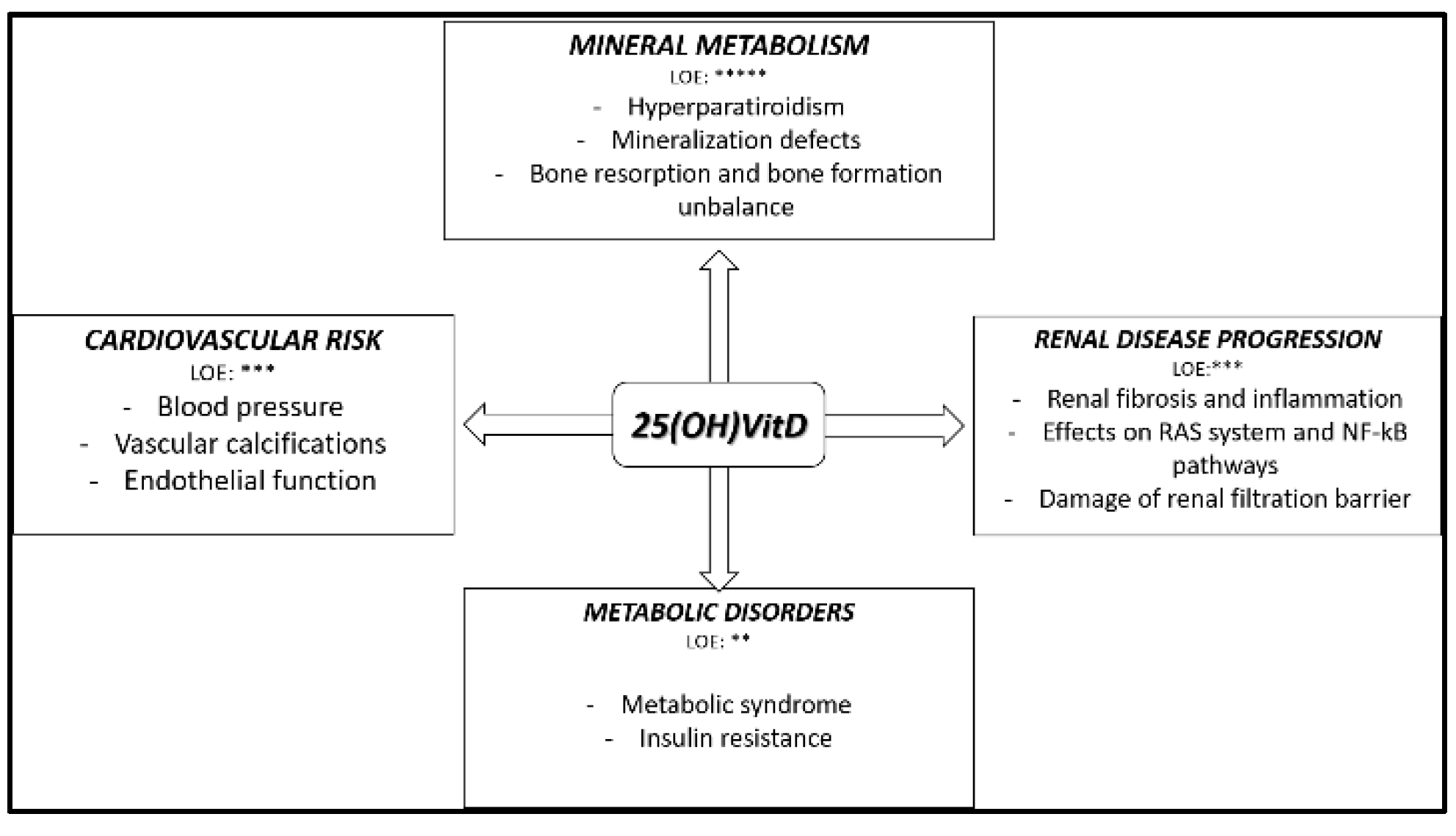
3. Native Hypovitaminosis D in CKD: Experimental and Clinical Evidence
3.1. Native Hypovitaminosis D and Mineral Metabolism
3.2. Native Hypovitaminosis D and Metabolic Disorders
3.3. Native Hypovitaminosis D and Cardiovascular Risk
3.4. Native Hypovitaminosis D in Animal Model with Kidney Disease
3.5. Native Hypovitaminosis D and Immune System in CKD
3.6. Native Hypovitaminosis D and Mortality in the Presence of CKD
4. Native Vitamin D Supplementation in CKD Patients: Evidence from Randomized Interventional Studies
5. Conclusions and Future Perspectives
- -
- Concerning the control of CKDMBD parameters, and only on this issue, both experimental, association and interventional data agree on the fact that 25(OH)VitD has a crucial role. Consequently, in our opinion, 25(OH)VitD should be used as primary choice, when indicated, in the treatment of CKDMBD, and, in cases of severe hypovitaminosis, a supplementation, when possible and safe, might be suggested independently to the other CMKMBD parameters.
- -
- The results from interventional studies are discordant concerning beneficial effects, beyond CKDMBD, of 25(OH)VitD supplementation. It is important to underscore, however, that at the moment we are not able to define the optimal level of 25(OH)VitD that should be maintained in CKD patients, so we still refer to recommendations for the general population. Hopefully, future more focused and more interventional studies might add some knowledge indicating and “personalizing” the target 25(OH)VitD levels.
- -
- Native Vitamin D supplementation does not exclude the use of the active form of vitamin D. So, in agreement with what the guidelines support, the two forms should be considered not in contrast but potentially together in the treatment of secondary hyperparathyroidism and, probably, in bone health. More trials in the future might provide information about the lack of classical effectiveness of 25(OH)VitD supplementation in CKD patients in clinical practice.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- González, E.A.; Sachdeva, A.; Oliver, D.A.; Martin, K.J. Vitamin D insufficiency and deficiency in chronic kidney disease. A single center observational study. Am. J. Nephrol. 2004, 24, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Cozzolino, M.; Ciceri, P.; Volpi, E.M.; Olivi, L.; Messa, P.G. Pathophysiology of calcium and phosphate metabolism impairment in chronic kidney disease. Blood Purif. 2009, 27, 338–344. [Google Scholar] [CrossRef]
- Messa, P.; Regalia, A.; Alfieri, C. Nutritional vitamin D in renal transplant patients: Speculations and reality. Nutrients 2017, 9, 550. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; MacLaughlin, J.A.; Clark, M.B.; Holick, S.A.; Potts, J.T.; Anderson, R.R.; Elias, P. Photosynthesis of previtamin D3 in human skin and the physiologic consequences. Science 1980, 210, 203–205. [Google Scholar] [CrossRef] [PubMed]
- Nykjaer, A.; Dragun, D.; Walther, D.; Vorum, H.; Jacobsen, C.; Herz, J.; Willnow, T.E. An endocytic pathway essential for renal uptake and activation of the steroid 25-(OH) vitamin D3. Cell 1999, 96, 507–515. [Google Scholar] [CrossRef]
- Jones, G.; Prosser, D.E.; Kaufmann, M. 25-Hydroxyvitamin D-24-hydroxylase (CYP24A1): Its important role in the degradation of vitamin D. Arch. Biochem. Biophys. 2012, 523, 9–18. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef]
- Ginde, A.A.; Liu, M.C.; Camargo, C.A. Demographic differences and trends of vitamin D insufficiency in the US population, 1988–2004. Arch. Intern. Med. 2009, 169, 626. [Google Scholar] [CrossRef]
- Kuro-o, M.; Moe, O.W. FGF23-αKlotho as a paradigm for a kidney-bone network. Bone 2017, 100, 4–18. [Google Scholar] [CrossRef]
- Takemoto, F.; Shinki, T.; Yokoyama, K.; Inokami, T.; Hara, S.; Yamada, A.; Uchida, S. Gene expression of vitamin D hydroxylase and megalin in the remnant kidney of nephrectomized rats. Kidney Int. 2003, 64, 414–420. [Google Scholar] [CrossRef]
- Matsui, I.; Hamano, T.; Tomida, K.; Inoue, K.; Takabatake, Y.; Nagasawa, Y.; Isaka, Y. Active vitamin D and its analogue, 22-oxacalcitriol, ameliorate puromycin aminonucleoside-induced nephrosis in rats. Nephrol. Dial. Transpl. 2009, 24, 2354–2361. [Google Scholar] [CrossRef] [PubMed]
- Leheste, J.R.; Rolinski, B.; Vorum, H.; Hilpert, J.; Nykjaer, A.; Jacobsen, C.; Willnow, T.E. Megalin knockout mice as an animal model of low molecular weight proteinuria. Am. J. Pathol. 1999, 155, 1361–1370. [Google Scholar] [CrossRef]
- Bohnert, B.N.; Daniel, C.; Amann, K.; Voelkl, J.; Alesutan, I.; Lang, F.; Artunc, F. Impact of phosphorus restriction and vitamin D-substitution on secondary hyperparathyroidism in a proteinuric mouse Model. Kidney Blood Press. Res. 2015, 40, 153–165. [Google Scholar] [CrossRef] [PubMed]
- Messa, P.; Alfieri, C.; Rastaldi, M.P. Recent insights into vitamin D and its receptor. J. Nephrol. 2011, 24 (Suppl. 18), 30–37. [Google Scholar] [CrossRef] [PubMed]
- Cupisti, A.; Vigo, V.; Baronti, M.E.; D’Alessandro, C.; Ghiadoni, L.; Egidi, M.F. Vitamin D status and cholecalciferol supplementation in chronic kidney disease patients: An Italian cohort report. Int. J. Nephrol. Renov. Dis. 2015, 8, 151–157. [Google Scholar] [CrossRef]
- Jhee, J.H.; Kim, H.; Park, S.; Yun, H.R.; Jung, S.Y.; Kee, Y.K.; Yoo, T.H. Vitamin D deficiency is significantly associated with depression in patients with chronic kidney disease. PLoS ONE 2017, 12, e0171009. [Google Scholar] [CrossRef]
- Kidney Disease: Improving global outcomes (KDIGO) CKD-MBD update work group. KDIGO 2017 clinical practice guideline update for the diagnosis, evaluation, prevention, and treatment of chronic kidney disease–mineral and bone disorder (CKD-MBD). Kidney Int. 2017, 7 (Suppl. 1), 1–59. [Google Scholar] [CrossRef]
- Tripkovic, L.; Lambert, H.; Hart, K.; Smith, C.P.; Bucca, G.; Penson, S.; Lanham-New, S. Comparison of vitamin D2 and vitamin D3 supplementation in raising serum 25-hydroxyvitamin D status: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2012, 95, 1357–1364. [Google Scholar] [CrossRef]
- Vieth, R. Vitamin D toxicity, policy, and science. J. Bone Miner. Res. 2007, 22 (Suppl. S2), V64–V68. [Google Scholar] [CrossRef]
- Sprague, S.M.; Crawford, P.W.; Melnick, J.Z.; Strugnell, S.A.; Ali, S.; Mangoo-Karim, R.; Bishop, C.W. Use of extended-release calcifediol to treat secondary hyperparathyroidism in stages 3 and 4 chronic kidney disease. Am. J. Nephrol. 2016, 44, 316–325. [Google Scholar] [CrossRef]
- Alfieri, C.; Regalia, A.; Zanoni, F.; Vettoretti, S.; Cozzolino, M.; Messa, P. The importance of adherence in the treatment of secondary hyperparathyroidism. Blood Purif. 2019, 47, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Morrone, L.F.; Bolasco, P.; Camerini, C.; Cianciolo, G.; Cupisti, A.; Galassi, A.; Cozzolino, M. Vitamin D in patients with chronic kidney disease: A position statement of the Working Group; Trace elements and mineral metabolism. J. Nephrol. 2016, 29, 305–328. [Google Scholar] [CrossRef]
- Townsend, K.; Evans, K.N.; Campbell, M.J.; Colston, K.W.; Adams, J.S.; Hewison, M. Biological actions of extra-renal 25-hydroxyvitamin D-1α-hydroxylase and implications for chemoprevention and treatment. J. Steroid. Biochem. Mol. Biol. 2005, 97, 103–109. [Google Scholar] [CrossRef] [PubMed]
- Zehnder, D.; Bland, R.; Williams, M.C.; McNinch, R.W.; Howie, A.J.; Stewart, P.M.; Hewison, M. Extrarenal expression of 25-hydroxyvitamin D3 -1α-Hydroxylase 1. J. Clin. Endocrinol. Metab. 2001, 86, 888–894. [Google Scholar] [PubMed]
- Holick, M.F. Vitamin D in health and disease: Vitamin D for health and in chronic kidney disease. Semin. Dial. 2005, 18, 266–275. [Google Scholar] [CrossRef] [PubMed]
- Jean, G.; Souberbielle, J.; Chazot, C. Vitamin D in chronic kidney disease and dialysis patients. Nutrients 2017, 9, 328. [Google Scholar] [CrossRef] [PubMed]
- Lips, P.; Goldsmith, D.; de Jongh, R. Vitamin D and osteoporosis in chronic kidney disease. J. Nephrol. 2005, 30, 671–675. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.C.; Lin, Y.C.; Hsu, C.Y.; Kao, C.C.; Chang, F.C.; Chen, T.W.; Wu, M.S. Effect modifying role of serum calcium on mortality-predictability of PTH and alkaline phosphatase in hemodialysis patients: An investigation using data from the Taiwan renal registry data system from 2005 to 2012. PLoS ONE 2015, 10, e0129737. [Google Scholar] [CrossRef]
- Yadav, A.K.; Kumar, V.; Kumar, V.; Banerjee, D.; Gupta, K.L.; Jha, V. The effect of vitamin D supplementation on bone metabolic markers in chronic kidney disease. J. Bone Miner. Res. 2018, 33, 404–409. [Google Scholar] [CrossRef]
- Seiki, S.; Chonchol, M.; Cheung, A.K.; Kaufman, J.S.; Greene, T.; Roberts, W.L.; Smits, G.; Kendrick, J.; the HOST Investigators. 25-hydroxyvitamin D deficiency is associated with an increased risk of metabolic syndrome in patients with non-diabetic chronic kidney disease. Clin. Nephrol. 2012, 78, 432–441. [Google Scholar] [CrossRef]
- Chonchol, M.; Scragg, R. 25-Hydroxyvitamin D, insulin resistance, and kidney function in the third national health and nutrition examination survey. Kidney Int. 2007, 71, 134–139. [Google Scholar] [CrossRef] [PubMed]
- DeFronzo, R.A. Lilly lecture 1987. The triumvirate: Beta-cell, muscle, liver. A collusion responsible for NIDDM. Diabetes 1988, 37, 667–687. [Google Scholar] [CrossRef] [PubMed]
- Lim, L.L.; Ng, Y.M.; Kang, P.S.; Lim, S.K. Association between serum 25-hydroxyvitamin D and glycated hemoglobin levels in type 2 diabetes patients with chronic kidney disease. J. Diabetes Investig. 2018, 9, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Weishaar, R.E.; Kim, S.N.; Saunders, D.E.; Simpson, R.U. Involvement of vitamin D3 with cardiovascular function. III. Effects on physical and morphological properties. Am. J. Physiol. 1990, 258, E134–E142. [Google Scholar] [CrossRef] [PubMed]
- Weng, S.; Sprague, J.E.; Oh, J.; Riek, A.E.; Chin, K.; Garcia, M.; Bernal-Mizrachi, C. Vitamin D deficiency induces high blood pressure and accelerates atherosclerosis in mice. PLoS ONE 2013, 8, e54625. [Google Scholar] [CrossRef] [PubMed]
- Merke, J.; Milde, P.; Lewicka, S.; Hügel, U.; Klaus, G.; Mangelsdorf, D.J.; Ritz, E. Identification and regulation of 1,25-dihydroxyvitamin D3 receptor activity and biosynthesis of 1,25-dihydroxyvitamin D3. Studies in cultured bovine aortic endothelial cells and human dermal capillaries. J. Clin. Investig. 1989, 83, 1903–1915. [Google Scholar] [CrossRef] [PubMed]
- Panizo, S.; Barrio-Vazquez, S.; Naves-Diaz, M.; Carrillo-Lopez, N.; Rodriguez, I.; Fernandez-Vazquez, A.; Cannata-Andia, J.B. Vitamin D receptor activation, left ventricular hypertrophy and myocardial fibrosis. Nephrol. Dial. Transpl. 2013, 28, 2735–2744. [Google Scholar] [CrossRef]
- Pilz, S.; Tomaschitz, A.; Drechsler, C.; Dekker, J.M.; März, W. Vitamin D deficiency and myocardial diseases. Mol. Nutr. Food Res. 2010, 54, 1103–1113. [Google Scholar] [CrossRef]
- Hewison, M.; Burke, F.; Evans, K.N.; Lammas, D.A.; Sansom, D.M.; Liu, P.; Adams, J.S. Extra-renal 25-hydroxyvitamin D3-1α-hydroxylase in human health and disease. J. Steroid Biochem. Mol. Biol. 2007, 103, 316–321. [Google Scholar] [CrossRef]
- Forman, J.P.; Giovannucci, E.; Holmes, M.D.; Bischoff-Ferrari, H.A.; Tworoger, S.S.; Willett, W.C.; Curhan, G.C. Plasma 25-Hydroxyvitamin D levels and risk of incident hypertension. Hypertension 2007, 49, 1063–1069. [Google Scholar] [CrossRef]
- Murni, I.K.; Sulistyoningrum, D.C.; Oktaria, V. Association of vitamin D deficiency with cardiovascular disease risk in children: Implications for the Asia Pacific region. Asia Pac. J. Clin. Nutr. 2016, 25 (Suppl. 1), S8–S19. [Google Scholar] [PubMed]
- Garcia-Canton, C.; Bosch, E.; Ramirez, A.; Gonzalez, Y.; Auyanet, I.; Guerra, R.; Checa, M.D. Vascular calcification and 25-hydroxyvitamin D levels in non-dialysis patients with chronic kidney disease stages 4 and 5. Nephrol. Dial. Transpl. 2011, 26, 2250–2256. [Google Scholar] [CrossRef] [PubMed]
- Luo, Q.; Wang, L.L.; Gao, Y.H. Association between serum 25-hydroxyvitamin D and arterial stiffness in non-dialysis-dependent CKD. Eur. J. Clin. Nutr. 2016, 70, 274–276. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Jiang, C.; Sun, C.; Tang, T.; Jin, B.; Cao, D.; Zhang, M. Hypovitaminosis D is associated with endothelial dysfunction in patients with non-dialysis chronic kidney disease. J. Nephrol. 2015, 28, 471–476. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.C.; Lu, C.L.; Zheng, C.M.; Chen, R.M.; Lin, Y.F.; Liu, W.C.; Lu, K.C. Emerging role of vitamins D and K in modulating uremic vascular calcification: The aspect of passive calcification. Nutrients 2019, 11, 152. [Google Scholar] [CrossRef] [PubMed]
- Ureña Torres, O.; Cozzolino, M.; Vervloet, M. Vitamin D in Chronic Kindey Disease, 1st ed.; Springer: Berlin/Heidelberg, Germany, 2016; pp. 537–562. [Google Scholar]
- Mary, A.; Hénaut, L.; Boudot, C.; Six, I.; Brazier, M.; Massy, Z.A.; Mentaverri, R. Calcitriol prevents in vitro vascular smooth muscle cell mineralization by regulating calcium-sensing receptor expression. Endocrinology 2015, 156, 1965–1974. [Google Scholar] [CrossRef] [PubMed]
- Lai, S.; Coppola, B.; Dimko, M.; Galani, A.; Innico, G.; Frassetti, N.; Mariotti, A. Vitamin D deficiency, insulin resistance, and ventricular hypertrophy in the early stages of chronic kidney disease. Ren. Fail. 2014, 36, 58–64. [Google Scholar] [CrossRef]
- Bi, J.; Watanabe, H.; Fujimura, R.; Nishida, K.; Nakamura, R.; Oshiro, S.; Maruyama, T. A downstream molecule of 1,25-dihydroxyvitamin D3, alpha-1-acid glycoprotein, protects against mouse model of renal fibrosis. Sci. Rep. 2018, 8, 17329. [Google Scholar] [CrossRef]
- De Bragança, A.C.; Canale, D.; Gonçalves, J.G.; Shimizu, M.H.M.; Seguro, A.C.; Volpini, R.A. Vitamin D deficiency aggravates the renal features of moderate chronic kidney disease in 5/6 nephrectomized rats. Front. Med. 2018, 5, 282. [Google Scholar] [CrossRef]
- Carey, R.M.; Siragy, H.M. The intrarenal renin-angiotensin system and diabetic nephropathy. Trends Endocrinol. Metab. 2003, 14, 274–281. [Google Scholar] [CrossRef]
- Zhang, Y.; Kong, J.; Deb, D.K.; Chang, A.; Li, Y.C. Vitamin D receptor attenuates renal fibrosis by suppressing the renin-angiotensin system. J. Am. Soc. Nephrol. 2010, 21, 966–973. [Google Scholar] [CrossRef] [PubMed]
- Trohatou, O.; Tsilibary, E.F.; Charonis, A.; Iatrou, C.; Drossopoulou, G. Vitamin D3 ameliorates podocyte injury through the nephrin signalling pathway. J. Cell. Mol. Med. 2017, 21, 2599–2609. [Google Scholar] [CrossRef] [PubMed]
- Prietl, B.; Treiber, G.; Pieber, T.R.; Amrein, K. Vitamin D and immune function. Nutrients 2013, 5, 2502–2521. [Google Scholar] [CrossRef] [PubMed]
- Hewison, M. Antibacterial effects of vitamin D. Nat. Rev. Endocrinol. 2011, 7, 337–345. [Google Scholar] [CrossRef] [PubMed]
- Gois, P.; Ferreira, D.; Olenski, S.; Seguro, A. Vitamin D and infectious diseases: Simple bystander or contributing factor? Nutrients 2017, 9, 651. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.S.; Ren, S.; Liu, P.T.; Chun, R.F.; Lagishetty, V.; Gombart, A.F.; Hewison, M. Vitamin D-directed rheostatic regulation of monocyte antibacterial responses. J. Immunol. 2009, 182, 4289–4295. [Google Scholar] [CrossRef] [PubMed]
- Riek, A.E.; Oh, J.; Darwech, I.; Moynihan, C.E.; Bruchas, R.R.; Bernal-Mizrachi, C. 25(OH) vitamin D suppresses macrophage adhesion and migration by downregulation of ER stress and scavenger receptor A1 in type 2 diabetes. J. Steroid. Biochem. Mol. Biol. 2014, 144, 172–179. [Google Scholar] [CrossRef]
- Sanchez-Niño, M.D.; Bozic, M.; Córdoba-Lanús, E.; Valcheva, P.; Gracia, O.; Ibarz, M.; Valdivielso, J.M. Beyond proteinuria: VDR activation reduces renal inflammation in experimental diabetic nephropathy. Am. J. Physiol. Ren. Physiol. 2012, 302, F647–F657. [Google Scholar] [CrossRef]
- Guijarro, C.; Egido, J. Transcription factor-kB (NF-kB) and renal disease. Kidney Int. 2001, 59, 415–424. [Google Scholar] [CrossRef]
- Xu, S.; Chen, Y.H.; Tan, Z.X.; Xie, D.D.; Zhang, C.; Zhang, Z.H.; Xu, D.X. Vitamin D3 pretreatment regulates renal inflammatory responses during lipopolysaccharide-induced acute kidney injury. Sci. Rep. 2015, 5, 18687. [Google Scholar] [CrossRef]
- Du, J.; Jiang, S.; Hu, Z.; Tang, S.; Sun, Y.; He, J.; Li, Y.C. Vitamin D receptor activation protects against lipopolysaccharide-induced acute kidney injury through suppression of tubular cell apoptosis. Am. J. Physiol. Cell Physiol. 2019, 316, F1068–F1077. [Google Scholar] [CrossRef] [PubMed]
- Rüster, C.; Franke, S.; Reuter, S.; Mrowka, R.; Bondeva, T.; Wolf, G. Vitamin D3 partly antagonizes advanced-glycation endproducts-induced NFkB activation in mouse podocytes. Nephron 2016, 134, 105–116. [Google Scholar] [CrossRef] [PubMed]
- Meireles, M.S.; Kamimura, M.A.; Dalboni, M.A.; Giffoni de Carvalho, J.T.; Aoike, D.T.; Cuppari, L. Effect of cholecalciferol on vitamin D-regulatory proteins in monocytes and on inflammatory markers in dialysis patients: A randomized controlled trial. Clin. Nutri. 2016, 35, 1251–1258. [Google Scholar] [CrossRef] [PubMed]
- Zehnder, D.; Quinkler, M.; Eardley, K.S.; Bland, R.; Lepenies, J.; Hughes, S.V.; Hewison, M. Reduction of the vitamin D hormonal system in kidney disease is associated with increased renal inflammation. Kidney Int. 2008, 74, 1343–1353. [Google Scholar] [CrossRef] [PubMed]
- Pilz, S.; Iodice, S.; Zittermann, A.; Grant, W.B.; Gandini, S. Vitamin D status and mortality risk in CKD: A meta-analysis of prospective studies. Am. J. Kidney Dis. 2011, 58, 374–382. [Google Scholar] [CrossRef] [PubMed]
- Mehrotra, R.; Kermah, D.A.; Salusky, I.B.; Wolf, M.S.; Thadhani, R.I.; Chiu, Y.W.; Norris, K.C. Chronic kidney disease, hypovitaminosis D, and mortality in the United States. Kidney Int. 2009, 76, 977–983. [Google Scholar] [CrossRef] [PubMed]
- Namir, Y.; Cohen, M.; Haviv, Y.; Slotki, I.; Shavit, L. Vitamin D levels, vitamin D supplementation, and prognosis in patients with chronic kidney disease. Clin. Nephrol. 2016, 86, 165–174. [Google Scholar] [CrossRef]
- Shroff, R.; Wan, M.; Gullett, A.; Ledermann, S.; Shute, R.; Knott, C.; Rees, L. Ergocalciferol supplementation in children with CKD delays the onset of secondary hyperparathyroidism: A randomized trial. Clin. J. Am. Soc. Nephrol. 2012, 7, 216–223. [Google Scholar] [CrossRef]
- Marckmann, P.; Agerskov, H.; Thineshkumar, S.; Bladbjerg, E.M.; Sidelmann, J.J.; Jespersen, J.; Scholze, A. Randomized controlled trial of cholecalciferol supplementation in chronic kidney disease patients with hypovitaminosis D. Nephrol. Dial. Transpl. 2012, 27, 3523–3531. [Google Scholar] [CrossRef]
- Levin, A.; Tang, M.; Perry, T.; Zalunardo, N.; Beaulieu, M.; Dubland, J.A.; Djurdjev, O. Randomized controlled trial for the effect of vitamin D supplementation on vascular stiffness in CKD. Clin. J. Am. Soc. Nephrol. 2017, 12, 1447–1460. [Google Scholar] [CrossRef]
- Kumar, V.; Yadav, A.K.; Lal, A.; Kumar, V.; Singhal, M.; Billot, L.; Jha, V. A randomized trial of vitamin D supplementation on vascular function in CKD. J. Am. Soc. Nephrol. 2017, 28, 3100–3108. [Google Scholar] [CrossRef] [PubMed]
- Manson, J.E.; Cook, N.R.; Lee, I.M.; Christen, W.; Bassuk, S.S.; Mora, S.; VITAL research group. Vitamin D supplements and prevention of cancer and cardiovascular disease. New Engl. J. Med. 2019, 380, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Shoji, T.; Inaba, M.; Fukagawa, M.; Ando, R.; Emoto, M.; Fujii, H.; Nishizawa, Y. Effect of oral alfacalcidol on clinical outcomes in patients without secondary hyperparathyroidism receiving maintenance hemodialysis. JAMA 2018, 320, 2325. [Google Scholar] [PubMed]
- Mann, M.C.; Hobbs, A.J.; Hemmelgarn, B.R.; Roberts, D.J.; Ahmed, S.B.; Rabi, D.M. Effect of oral vitamin D analogs on mortality and cardiovascular outcomes among adults with chronic kidney disease: A meta-analysis. Clin. Kidney J. 2015, 8, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Morrone, L.F.; Cozzolino, M. The beneficial impact of vitamin D treatment in CKD patients: What’s next? Clin. Kidney J. 2015, 8, 38–40. [Google Scholar] [CrossRef] [PubMed]
- Molina, P.; Gorriz, J.L.; Molina, M.D.; Peris, A.; Beltran, S.; Kanter, J.; Pallardo, L.M. The effect of cholecalciferol for lowering albuminuria in chronic kidney disease: A prospective controlled study. Nephrol. Dial. Transplant. 2014, 29, 97–109. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Yu, H.; Lu, J.; Guo, K.; Zhang, L.; Bao, Y.; Jia, W. Oral supplementation with cholecalciferol 800 IU ameliorates albuminuria in Chinese type 2 diabetic patients with nephropathy. PLoS ONE 2012, 7, e50510. [Google Scholar] [CrossRef] [PubMed]
| Type of Patients | Number of Patients | Type of Intervention | Results | Reference |
|---|---|---|---|---|
| CKD stages 2–4 | 87 | GrA: Cholecalciferol 5000 IU/wk; GrB: Cholecalciferol 20,000 IU/wk | In GrB vs. GrA: significant increase of 25OHVitD, 1,25OHD2 and reduction PTH | Oksa et al., 2008 |
| CKD stages 1–5 | 25 | GrA: Cholecalciferol 40,000 IU/wk; GrB: placebo | In GrA vs. GrB: significant increase of 25OHVitD, 1,25(OH)D2, FGF23 and reduction PTH | Markmann et al., 2012 |
| CKD 3–4 | 38 | GrA: Ergocalciferol 50,000 IU/wk for one month followed by 50,000 IU/mth for 5 mths; GrB: Placebo | In GrA vs. GrB: significant increase of 25(OH)VitD, endothelium dependent microcirculatory vasodilatation and reduction of pulse pressure. No effects on PWV and LVMI | Dreyer et al., 2014 |
| CKD stages 3–4 | 429 | Study 1 (n = 213): GrA: ER Calcifediol 12 wk at 30 g/daily + 14 wks 30 or 60 g/daily; GrB 26 wks placebo Study 2 (n = 216): GrA: ER Calcifediol 12 wks 30 g/daily + 14 wks 30 or 60 g/daily; GrB: 26 wks placebo | In GrA vs. GrB in both studies: significant increase of 25OHVitD and reduction PTH | Sprague et al., 2016 |
| CKD 3–5 | 44 | GrA:Cholecalciferol 50.000 IU/wk; GrB: Ergocalciferol 50.000 IU/wk | In GrA vs. GrB: significantly higher increase of 25(OH)VitD. No differences in 1,25(OH)D2 and PTH | Weltmore et al., 2016 |
| CKD stages 3–4 | 128 | GrA: Cholecalciferol 2000 IU/day; GrB: Calcitriol 0.5 g/day | In GrA vs. GrB: significant increase of 25OHVitD in GrA vs. GrB. No effects on FMD | Kendrick et al., 2017 |
| CKD stages 3–4 | 120 | GrA: Cholecalciferol 300,000 UI mth; GrB: Placebo | In GrA vs. GrB: significant increase of 25OHVitD, 1,25OHD2 and reduction PTH. Increase of FMD and PWV | Kumar et al., 2017 |
| CKD stages 3–4 | 119 | GrA: Placebo; GrB: Calcitriol 0.5 g 3×/wk; GrC: Calcifediol 5.000 IU 3×/wk | In GrC vs. others: significant decrease of PWV. Patients in the highest 25(OH)VitD tertile at trial end had significant decreases in PWV | Levin et al., 2017 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alfieri, C.; Ruzhytska, O.; Vettoretti, S.; Caldiroli, L.; Cozzolino, M.; Messa, P. Native Hypovitaminosis D in CKD Patients: From Experimental Evidence to Clinical Practice. Nutrients 2019, 11, 1918. https://doi.org/10.3390/nu11081918
Alfieri C, Ruzhytska O, Vettoretti S, Caldiroli L, Cozzolino M, Messa P. Native Hypovitaminosis D in CKD Patients: From Experimental Evidence to Clinical Practice. Nutrients. 2019; 11(8):1918. https://doi.org/10.3390/nu11081918
Chicago/Turabian StyleAlfieri, Carlo, Oksana Ruzhytska, Simone Vettoretti, Lara Caldiroli, Mario Cozzolino, and Piergiorgio Messa. 2019. "Native Hypovitaminosis D in CKD Patients: From Experimental Evidence to Clinical Practice" Nutrients 11, no. 8: 1918. https://doi.org/10.3390/nu11081918
APA StyleAlfieri, C., Ruzhytska, O., Vettoretti, S., Caldiroli, L., Cozzolino, M., & Messa, P. (2019). Native Hypovitaminosis D in CKD Patients: From Experimental Evidence to Clinical Practice. Nutrients, 11(8), 1918. https://doi.org/10.3390/nu11081918







