Of Older Mice and Men: Branched-Chain Amino Acids and Body Composition
Abstract
1. Introduction
2. Materials and Methods
2.1. Human Cohort
2.1.1. Study Design
2.1.2. Dietary Data
2.1.3. Outcome Variables
2.1.4. Covariates
2.2. Mouse Cohort
2.2.1. Animals and Husbandry
2.2.2. Experimental Diets
2.2.3. Outcome Variables
2.2.4. Statistical Analyses
3. Results
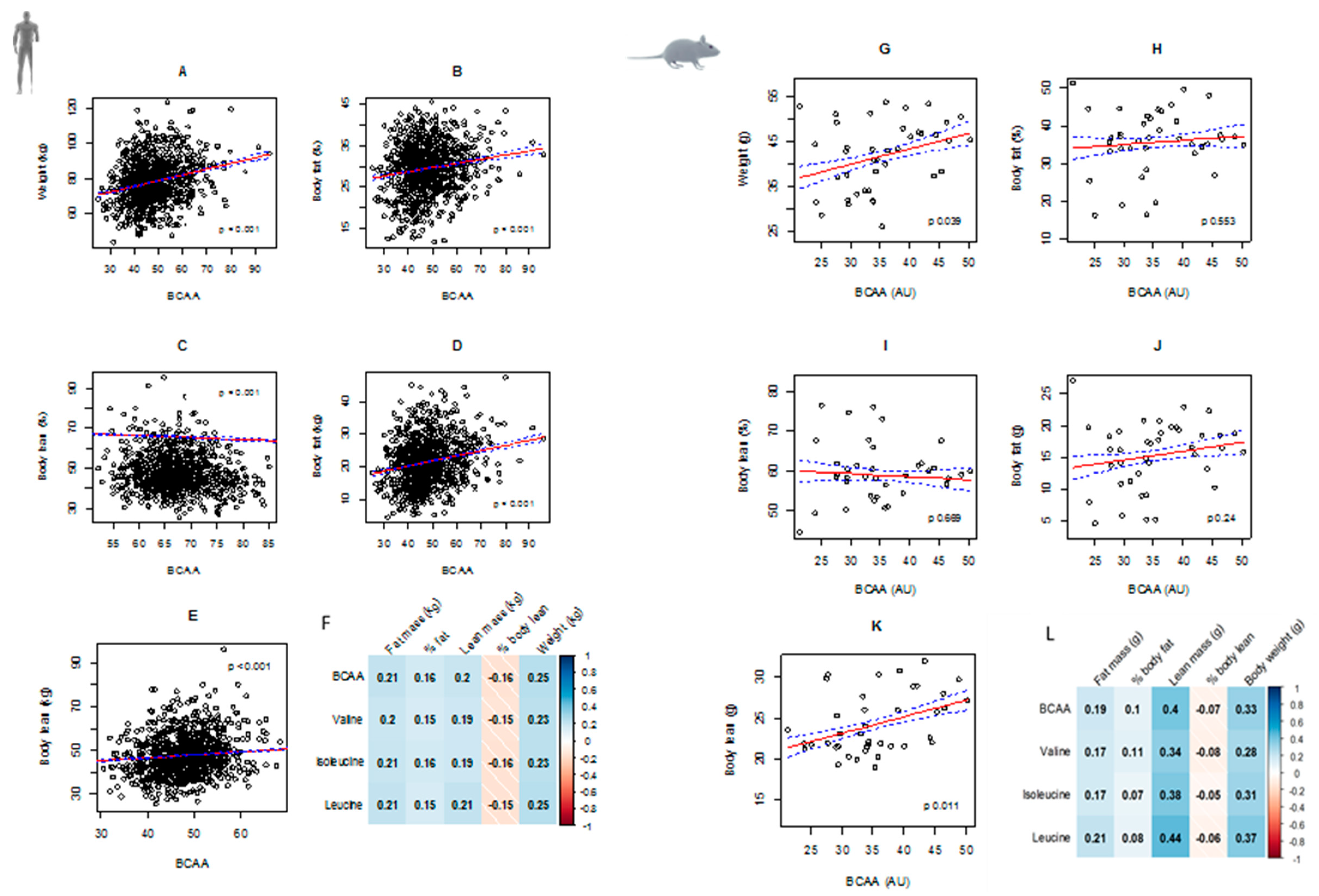
3.1. Circulating BCAAs and Body Composition
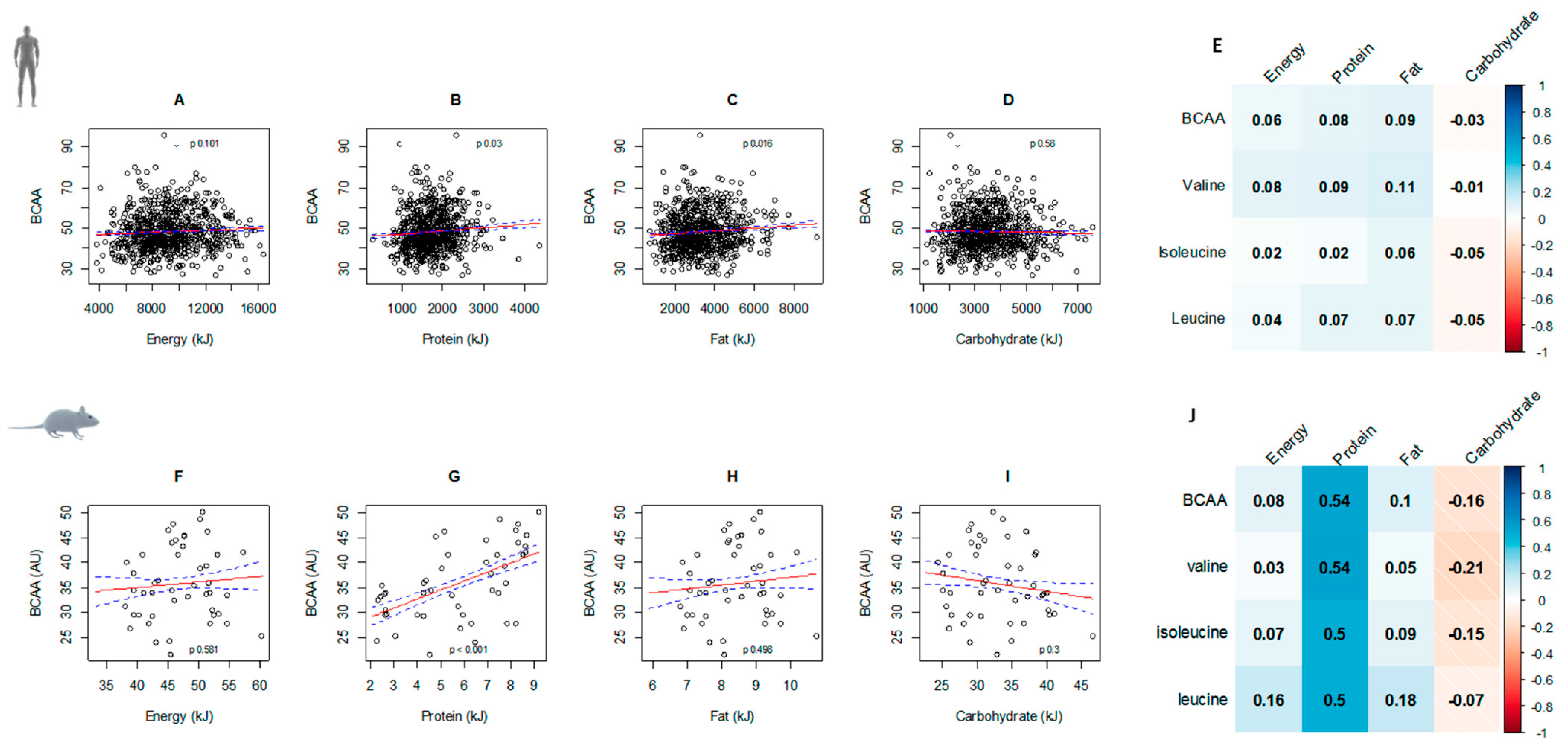
3.2. Circulating BCAAs and Diet
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Fontana, L.; Cummings, N.E.; Apelo, S.I.A.; Neuman, J.C.; Kasza, I.; Schmidt, B.A.; Cava, E.; Spelta, F.; Tosti, V.; Syed, F.A.; et al. Decreased consumption of branched chain amino acids improves metabolic health. Cell Rep. 2016, 16, 520–530. [Google Scholar] [CrossRef] [PubMed]
- Newgard, C.B.; An, J.; Bain, J.R.; Muehlbauer, M.J.; Stevens, R.D.; Lien, L.F.; Haqq, A.M.; Shah, S.H.; Arlotto, M.; Slentz, C.A.; et al. A Branched-Chain Amino Acid-Related Metabolic Signature that Differentiates Obese and Lean Humans and Contributes to Insulin Resistance. Cell Metab. 2009, 9, 565–566. [Google Scholar] [CrossRef]
- Maida, A.; Chan, J.S.; Sjøberg, K.A.; Zota, A.; Schmoll, D.; Kiens, B.; Herzig, S.; Rose, A.J. Repletion of branched chain amino acids reverses mTORC1 signaling but not improved metabolism during dietary protein dilution. Mol. Metab. 2017, 6, 873–881. [Google Scholar] [CrossRef] [PubMed]
- Solon-Biet, S.M.; McMahon, A.C.; Ballard, J.W.O.; Ruohonen, K.; Wu, L.E.; Cogger, V.C.; Warren, A.; Huang, X.; Pichaud, N.; Melvin, R.G.; et al. The Ratio of Macronutrients, Not Caloric Intake, Dictates Cardiometabolic Health, Aging, and Longevity in Ad Libitum-Fed Mice. Cell Metab. 2014, 19, 418–430. [Google Scholar] [CrossRef] [PubMed]
- Rousseau, M.; Guénard, F.; Garneau, V.; Allam-Ndoul, B.; Lemieux, S.; Pérusse, L.; Vohl, M.C. Associations Between Dietary Protein Sources, Plasma BCAA and Short-Chain Acylcarnitine Levels in Adults. Nutrients 2019, 11, 173. [Google Scholar] [CrossRef] [PubMed]
- Solon-Biet, S.M.; Cogger, V.C.; Pulpitel, T.; Wahl, D.; Clark, X.; Bagley, E.E.; Gregoriou, G.C.; Senior, A.M.; Wang, Q.P.; Brandon, A.E.; et al. Branched-chain amino acids impact health and lifespan indirectly via amino acid balance and appetite control. Nat. Metab. 2019, 1, 532–545. [Google Scholar] [CrossRef]
- Lynch, C.J.; Adams, S.H. Branched-chain amino acids in metabolic signalling and insulin resistance. Nat. Rev. Endocrinol. 2014, 10, 723–736. [Google Scholar] [CrossRef]
- Cummings, N.E.; Williams, E.M.; Kasza, I.; Konon, E.N.; Schaid, M.D.; Schmidt, B.A.; Poudel, C.; Sherman, D.S.; Yu, D.; Apelo, S.I.A.; et al. Restoration of metabolic health by decreased consumption of branched-chain amino acids. J. Physiol. 2017, 596, 623–645. [Google Scholar] [CrossRef]
- Wang, T.J.; Larson, M.G.; Vasan, R.S.; Cheng, S.; Rhee, E.P.; McCabe, E.; Lewis, G.D.; Fox, C.S.; Jacques, P.F.; Fernandez, C.; et al. Metabolite Profiles and the Risk of Developing Diabetes. Nat. Med. 2011, 17, 448–453. [Google Scholar] [CrossRef]
- Shah, S.H.; Crosslin, D.R.; Haynes, C.S.; Nelson, S.; Turer, C.B.; Stevens, R.D.; Muehlbauer, M.J.; Wenner, B.R.; Bain, J.R.; Laferrere, B.; et al. Branched-chain amino acid levels are associated with improvement in insulin resistance with weight loss. Diabetologia 2012, 55, 321–330. [Google Scholar] [CrossRef]
- Zheng, Y.; Li, Y.; Qi, Q.; Hruby, A.; Manson, J.E.; Willett, W.C.; Wolpin, B.M.; Hu, F.B.; Qi, L. Cumulative consumption of branched-chain amino acids and incidence of type 2 diabetes. Int. J. Epidemiol. 2016, 45, 1482–1492. [Google Scholar] [CrossRef]
- Bifari, F.; Nisoli, E. Branched-chain amino acids differently modulate catabolic and anabolic states in mammals: A pharmacological point of view. Br. J. Pharmacol. 2017, 174, 1366–1377. [Google Scholar] [CrossRef]
- D’Antona, G.; Ragni, M.; Cardile, A.; Tedesco, L.; Dossena, M.; Bruttini, F.; Caliaro, F.; Corsetti, G.; Bottinelli, R.; Carruba, M.O.; et al. Branched-Chain Amino Acid Supplementation Promotes Survival and Supports Cardiac and Skeletal Muscle Mitochondrial Biogenesis in Middle-Aged Mice. Cell Metab. 2010, 12, 362–372. [Google Scholar] [CrossRef]
- Cumming, R.G.; Handelsman, D.; Seibel, M.J.; Creasey, H.; Sambrook, P.; Waite, L.; Naganathan, V.; Le Couteur, D.; Litchfield, M. Cohort Profile: The Concord Health and Ageing in Men Project (CHAMP). Int. J. Epidemiol. 2009, 38, 374–378. [Google Scholar] [CrossRef]
- Waern, R.V.; Cumming, R.G.; Blyth, F.; Naganathan, V.; Allman-Farinelli, M.; Le Couteur, D.; Simpson, S.J.; Kendig, H.; Hirani, V. Adequacy of nutritional intake among older men living in Sydney, Australia: Findings from the Concord Health and Ageing in Men Project (CHAMP). Br. J. Nutr. 2015, 114, 812–821. [Google Scholar] [CrossRef]
- Burke, B. The dietary history as a tool in research. J. Am. Diet. Assoc. 1947, 23, 1041–1046. [Google Scholar]
- Rosilene, W.; Cumming, R.; Travison, T.; Blyth, F.; Naganathan, V.; Allman-Farinelli, M.; Hirani, V. Relative validity of a diet history questionnaire against a four-day weighed food record among older men in Australia: The Concord Health and Ageing in Men Project (CHAMP). J. Nutr. Health Aging 2015, 19, 603–610. [Google Scholar] [CrossRef]
- Washburn, R.A.; Smith, K.W.; Jette, A.M.; Janney, C.A. The physical activity scale for the elderly (PASE): Development and evaluation. J. Clin. Epidemiol. 1993, 46, 153–162. [Google Scholar] [CrossRef]
- Wahl, D.; Solon-Biet, S.M.; Wang, Q.P.; Wali, J.A.; Pulpitel, T.; Clark, X.; Raubenheimer, D.; Senior, A.M.; Sinclair, D.A.; Cooney, G.J.; et al. Comparing the Effects of Low-Protein and High-Carbohydrate Diets and Caloric Restriction on Brain Aging in Mice. Cell Rep. 2018, 25, 2234–2243. [Google Scholar] [CrossRef]
- Patsch, C.; Challet-Meylan, L.; Thoma, E.C.; Urich, E.; Heckel, T.; O’Sullivan, J.F.; Grainger, S.J.; Kapp, F.G.; Sun, L.; Christensen, K.; et al. Generation of vascular endothelial and smooth muscle cells from human pluripotent stem cells. Nat. Cell Biol. 2015, 17, 994. [Google Scholar] [CrossRef]
- Chambers, J.M. Chapter 4 of Statistical Models in S. In Linear Models; Wadsworth & Brooks/Cole: Pacific Grove, CA, USA, 1992. [Google Scholar]
- Wilkinson GNaR, C.E. Symbolic descriptions of factorial models for analysis of variance. Appl. Stat. 1973, 22, 392–399. [Google Scholar] [CrossRef]
- Wei, T.; Simko, V. R Package “Corrplot”: Visualization of a Correlation Matrix (Version 0.84); R Foundation for Statistical Computing: Vienna, Austria, 2017. [Google Scholar]
- Chaleckis, R.; Murakami, I.; Takada, J.; Kondoh, H.; Yanagida, M. Individual variability in human blood metabolites identifies age-related differences. Proc. Natl. Acad. Sci. USA 2016, 113, 4252–4259. [Google Scholar] [CrossRef]
- Kouchiwa, T.; Wada, K.; Uchiyama, M.; Kasezawa, N.; Niisato, M.; Murakami, H.; Fukuyama, K.; Yokogoshi, H. Age-related changes in serum amino acids concentrations in healthy individuals. Clin. Chem. Lab. Med. 2012, 50, 861–870. [Google Scholar] [CrossRef]
- Oja, S.S.; Kemppainen, K.; Mero, A.A.; Pitkänen, H.T.; Seppä, J.M. Serum amino acid concentrations in aging men and women. Amino Acids 2003, 24, 413–421. [Google Scholar]
- Calvani, R.; Picca, A.; Marini, F.; Biancolillo, A.; Gervasoni, J.; Persichilli, S.; Primiano, A.; Coelho-Junior, H.; Bossola, M.; Urbani, A.; et al. A Distinct Pattern of Circulating Amino Acids Characterizes Older Persons with Physical Frailty and Sarcopenia: Results from the Biosphere Study. Nutrients 2018, 10, 1691. [Google Scholar] [CrossRef]
- Adachi, Y.; Ono, N.; Imaizumi, A.; Muramatsu, T.; Andou, T.; Shimodaira, Y.; Nagao, K.; Kageyama, Y.; Mori, M.; Noguchi, Y.; et al. Plasma amino acid profile in severely frail elderly patients in Japan. Int. J. Gerontol. 2018, 12, 290–293. [Google Scholar] [CrossRef]
- Moaddel, R.; Fabbri, E.; Khadeer, M.A.; Carlson, O.D.; Gonzalez-Freire, M.; Zhang, P.; Semba, R.D.; Ferrucci, L. Plasma Biomarkers of Poor Muscle Quality in Older Men and Women from the Baltimore Longitudinal Study of Aging. J. Gerontol. A Biol. Sci. Med. Sci. 2016, 71, 1266–1272. [Google Scholar] [CrossRef]
- Ter Borg, S.; Luiking, Y.C.; van Helvoort, A.; Boirie, Y.; Schols, J.; de Groot, C. Low Levels of Branched Chain Amino Acids, Eicosapentaenoic Acid and Micronutrients Are Associated with Low Muscle Mass, Strength and Function in Community-Dwelling Older Adults. J. Nutr. Health Aging 2019, 23, 27–34. [Google Scholar] [CrossRef]
- Fujita, S.; Volpi, E. Amino acids and muscle loss with aging. J. Nutr. 2006, 136 (Suppl. 1), 277S–280S. [Google Scholar] [CrossRef]
- Ottestad, I.; Ulven, S.M.; Øyri, L.K.L.; Sandvei, K.S.; Gjevestad, G.O.; Bye, A.; Sheikh, N.A.; Biong, A.S.; Andersen, L.F.; Holven, K.B. Reduced plasma concentration of branched-chain amino acids in sarcopenic older subjects: A cross-sectional study. Br. J. Nutr. 2018, 120, 445–453. [Google Scholar] [CrossRef]
- Würtz, P.; Mäkinen, V.P.; Soininen, P.; Kangas, A.J.; Tukiainen, T.; Kettunen, J.; Savolainen, M.J.; Tammelin, T.; Viikari, J.S.; Rönnemaa, T.; et al. Metabolic Signatures of Insulin Resistance in 7098 Young Adults. Diabetes 2012, 61, 1372–1380. [Google Scholar]
- Layman, D.K.; Baum, J.I. Dietary Protein Impact on Glycemic Control during Weight Loss. J. Nutr. 2004, 134, 968S–973S. [Google Scholar] [CrossRef]
- Rietman, A.; Schwarz, J.; Tomé, D.; Kok, F.J.; Mensink, M. High dietary protein intake, reducing or eliciting insulin resistance? Eur. J. Clin. Nutr. 2014, 68, 973. [Google Scholar] [CrossRef]
- Gannon, N.P.; Schnuck, J.K.; Vaughan, R.A. BCAA Metabolism and Insulin Sensitivity—Dysregulated by Metabolic Status? Mol. Nutr. Food Res. 2018, 62, 1700756. [Google Scholar] [CrossRef]
- Du, X.; You, H.; Li, Y.; Wang, Y.; Hui, P.; Qiao, B.; Lu, J.; Zhang, W.; Zhou, S.; Zheng, Y.; et al. Relationships between circulating branched chain amino acid concentrations and risk of adverse cardiovascular events in patients with STEMI treated with PCI. Sci. Rep. 2018, 8, 15809. [Google Scholar] [CrossRef]
- Wolfe, R.R. Branched-chain amino acids and muscle protein synthesis in humans: Myth or reality? J. Int. Soc. Sports Nutr. 2017, 14, 30. [Google Scholar] [CrossRef]
| HUMANS | ||
| Fat mass (kg) | β (SE) | p |
| Model 1 | 0.16 (0.02) | <0.001 |
| Model 2 | 0.15 (0.03) | <0.001 |
| Model 3 | 0.15 (0.03) | <0.001 |
| Body fat (%) | ||
| Model 1 | 0.10 (0.02) | <0.001 |
| Model 2 | 0.10 (0.02) | <0.001 |
| Model 3 | 0.10 (0.02) | <0.001 |
| Lean mass (kg) | ||
| Model 1 | 0.13 (0.02) | <0.001 |
| Model 2 | 0.10 (0.02) | <0.001 |
| Model 3 | 0.11 (0.02) | <0.001 |
| Body lean (%) | ||
| Model 1 | −0.10 (0.02) | <0.001 |
| Model 2 | −0.10 (0.02) | <0.001 |
| Model 3 | −0.10 (0.02) | <0.001 |
| Weight (kg) | ||
| Model 1 | 0.33 (0.04) | <0.001 |
| Model 2 | 0.28 (0.04) | <0.001 |
| Model 3 | 0.30 (0.04) | <0.001 |
| MICE | ||
| Fat mass (g) | B (SE) | p |
| Model 1 | 0.14 (0.11) | 0.24 |
| Body fat (%) | ||
| Model 1 | 0.11 (0.19) | 0.55 |
| Lean mass (g) | ||
| Model 1 | 0.2 (0.07) | 0.01 |
| Body lean (%) | ||
| Model 1 | −0.07 (0.17) | 0.67 |
| Weight (g) | ||
| Model 1 | 0.34 (0.16) | 0.04 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ribeiro, R.V.; Solon-Biet, S.M.; Pulpitel, T.; Senior, A.M.; Cogger, V.C.; Clark, X.; O’Sullivan, J.; Koay, Y.C.; Hirani, V.; Blyth, F.M.; et al. Of Older Mice and Men: Branched-Chain Amino Acids and Body Composition. Nutrients 2019, 11, 1882. https://doi.org/10.3390/nu11081882
Ribeiro RV, Solon-Biet SM, Pulpitel T, Senior AM, Cogger VC, Clark X, O’Sullivan J, Koay YC, Hirani V, Blyth FM, et al. Of Older Mice and Men: Branched-Chain Amino Acids and Body Composition. Nutrients. 2019; 11(8):1882. https://doi.org/10.3390/nu11081882
Chicago/Turabian StyleRibeiro, Rosilene V., Samantha M. Solon-Biet, Tamara Pulpitel, Alistair M. Senior, Victoria C. Cogger, Ximonie Clark, John O’Sullivan, Yen Chin Koay, Vasant Hirani, Fiona M. Blyth, and et al. 2019. "Of Older Mice and Men: Branched-Chain Amino Acids and Body Composition" Nutrients 11, no. 8: 1882. https://doi.org/10.3390/nu11081882
APA StyleRibeiro, R. V., Solon-Biet, S. M., Pulpitel, T., Senior, A. M., Cogger, V. C., Clark, X., O’Sullivan, J., Koay, Y. C., Hirani, V., Blyth, F. M., Seibel, M. J., Waite, L. M., Naganathan, V., Cumming, R. G., Handelsman, D. J., Simpson, S. J., & Couteur, D. G. L. (2019). Of Older Mice and Men: Branched-Chain Amino Acids and Body Composition. Nutrients, 11(8), 1882. https://doi.org/10.3390/nu11081882






