Role of Peptide Hormones in the Adaptation to Altered Dietary Protein Intake
Abstract
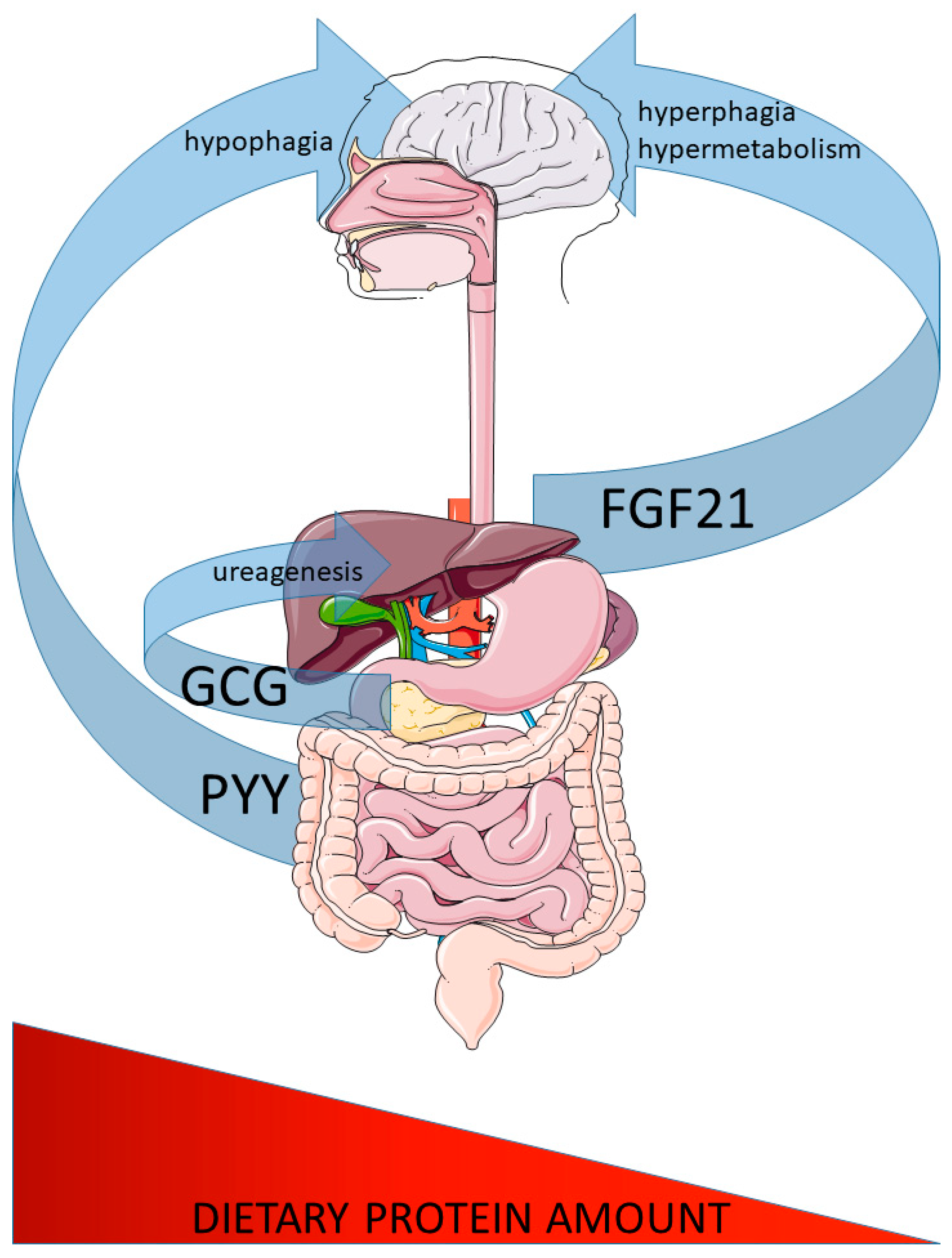
1. Introduction
2. Peptide Hormones from the Alimentary Tract
2.1. Stomach Derived Peptide Hormones
2.1.1. Gastrin
2.1.2. Ghrelin
2.2. Small/Large Intestine Derived Peptide Hormones
2.2.1. Secretin
2.2.2. Cholecystokinin
2.2.3. Glucagon-Like Peptide 1
2.2.4. Glucagon Like Peptide 2
2.2.5. Glucose-Dependent Insulinotropic Polypeptide
2.2.6. Peptide Tyrosine-Tyrosine
2.2.7. Fibroblast Growth Factor 19
3. Pancreatic Peptide Hormones
3.1. Insulin
3.2. Glucagon
3.3. Amylin
3.4. Pancreatic Polypeptide
3.5. Somatostatin
4. Liver-Derived Peptide Hormones
4.1. Fibroblast Growth Factor 21
4.2. Insulin-Like Growth Factor 1
5. Adipose Tissue Derived Peptide Hormones
5.1. Leptin
5.2. Adiponectin
6. Summary and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Simpson, S.J.; Le Couteur, D.G.; Raubenheimer, D.; Solon-Biet, S.M.; Cooney, G.J.; Cogger, V.C.; Fontana, L. Dietary protein, aging and nutritional geometry. Ageing Res. Rev. 2017, 39, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Rose, W.C. II. The sequence of events leading to the establishment of the amino acid needs of man. Am. J. Public Health Nation’s Health 1968, 58, 2020–2027. [Google Scholar] [CrossRef] [PubMed]
- Reeds, P.J. Dispensable and indispensable amino acids for humans. J. Nutr. 2000, 130, 1835S–1840S. [Google Scholar] [CrossRef] [PubMed]
- Efeyan, A.; Zoncu, R.; Sabatini, D.M. Amino acids and mtorc1: From lysosomes to disease. Trends Mol. Med. 2012, 18, 524–533. [Google Scholar] [CrossRef] [PubMed]
- Grallert, B.; Boye, E. Gcn2, an old dog with new tricks. Biochem. Soc. Trans. 2013, 41, 1687–1691. [Google Scholar] [CrossRef] [PubMed]
- Conigrave, A.D.; Hampson, D.R. Broad-spectrum amino acid-sensing class c g-protein coupled receptors: Molecular mechanisms, physiological significance and options for drug development. Pharm. Ther. 2010, 127, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Yabut, J.M.; Crane, J.D.; Green, A.E.; Keating, D.J.; Khan, W.I.; Steinberg, G.R. Emerging roles for serotonin in regulating metabolism: New implications for an ancient molecule. Endocr. Rev. 2019, 40, 1092–1107. [Google Scholar] [CrossRef] [PubMed]
- Prosapio, J.G.; Jialal, I. Physiology, Gastrin. In Statpearls; StatPearls Publishing: Treasure Island, FL, USA, 2019. [Google Scholar]
- Kojima, M.; Hosoda, H.; Date, Y.; Nakazato, M.; Matsuo, H.; Kangawa, K. Ghrelin is a growth-hormone-releasing acylated peptide from stomach. Nature 1999, 402, 656–660. [Google Scholar] [CrossRef] [PubMed]
- Muller, T.D.; Nogueiras, R.; Andermann, M.L.; Andrews, Z.B.; Anker, S.D.; Argente, J.; Batterham, R.L.; Benoit, S.C.; Bowers, C.Y.; Broglio, F.; et al. Ghrelin. Mol. Metab. 2015, 4, 437–460. [Google Scholar] [CrossRef] [PubMed]
- Karhunen, L.J.; Juvonen, K.R.; Huotari, A.; Purhonen, A.K.; Herzig, K.H. Effect of protein, fat, carbohydrate and fibre on gastrointestinal peptide release in humans. Regul. Pept. 2008, 149, 70–78. [Google Scholar] [CrossRef]
- Moran, L.J.; Luscombe-Marsh, N.D.; Noakes, M.; Wittert, G.A.; Keogh, J.B.; Clifton, P.M. The satiating effect of dietary protein is unrelated to postprandial ghrelin secretion. J. Clin. Endocrinol. Metab. 2005, 90, 5205–5211. [Google Scholar] [CrossRef]
- DiGregorio, N.; Sharma, S. Physiology, secretin. In Statpearls; StatPearls Publishing: Treasure Island, FL, USA, 2019. [Google Scholar]
- Mynatt, R.L.; Ravussin, E. Secretin: An old hormone with a burning secret. Cell 2018, 175, 1459–1460. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Schnabl, K.; Gabler, S.M.; Willershauser, M.; Reber, J.; Karlas, A.; Laurila, S.; Lahesmaa, M.; Din, M.U.; Bast-Habersbrunner, A.; et al. Secretin-activated brown fat mediates prandial thermogenesis to induce satiation. Cell 2018, 175, 1561–1574.e12. [Google Scholar] [CrossRef] [PubMed]
- Fahrenkrug, J.; Schaffalitzky de Muckadell, O.B. Plasma secretin concentration in man: Effect of intraduodenal glucose, fat, amino acids, ethanol, hcl, or ingestion of a meal. Eur. J. Clin. Investig. 1977, 7, 201–203. [Google Scholar] [CrossRef]
- Liddle, R.A. Cholecystokinin cells. Annu. Rev. Physiol. 1997, 59, 221–242. [Google Scholar] [CrossRef] [PubMed]
- Lieverse, R.J.; Jansen, J.B.; Masclee, A.A.; Lamers, C.B. Satiety effects of a physiological dose of cholecystokinin in humans. Gut 1995, 36, 176–179. [Google Scholar] [CrossRef]
- Kopin, A.S.; Mathes, W.F.; McBride, E.W.; Nguyen, M.; Al-Haider, W.; Schmitz, F.; Bonner-Weir, S.; Kanarek, R.; Beinborn, M. The cholecystokinin-a receptor mediates inhibition of food intake yet is not essential for the maintenance of body weight. J. Clin. Investig. 1999, 103, 383–391. [Google Scholar] [CrossRef]
- Hopman, W.P.; Jansen, J.B.; Lamers, C.B. Comparative study of the effects of equal amounts of fat, protein, and starch on plasma cholecystokinin in man. Scand. J. Gastroenterol. 1985, 20, 843–847. [Google Scholar] [CrossRef]
- Holst, J.J. Glucagonlike peptide 1: A newly discovered gastrointestinal hormone. Gastroenterology 1994, 107, 1848–1855. [Google Scholar] [CrossRef]
- Baggio, L.L.; Drucker, D.J. Biology of incretins: Glp-1 and gip. Gastroenterology 2007, 132, 2131–2157. [Google Scholar] [CrossRef]
- Drucker, D.J. The biology of incretin hormones. Cell Metab. 2006, 3, 153–165. [Google Scholar] [CrossRef] [PubMed]
- Vilsboll, T.; Krarup, T.; Sonne, J.; Madsbad, S.; Volund, A.; Juul, A.G.; Holst, J.J. Incretin secretion in relation to meal size and body weight in healthy subjects and people with type 1 and type 2 diabetes mellitus. J. Clin. Endocrinol. Metab. 2003, 88, 2706–2713. [Google Scholar] [CrossRef] [PubMed]
- Brubaker, P.L.; Anini, Y. Direct and indirect mechanisms regulating secretion of glucagon-like peptide-1 and glucagon-like peptide-2. Can. J. Physiol. Pharmacol. 2003, 81, 1005–1012. [Google Scholar] [CrossRef] [PubMed]
- Raben, A.; Agerholm-Larsen, L.; Flint, A.; Holst, J.J.; Astrup, A. Meals with similar energy densities but rich in protein, fat, carbohydrate, or alcohol have different effects on energy expenditure and substrate metabolism but not on appetite and energy intake. Am. J. Clin. Nutr. 2003, 77, 91–100. [Google Scholar] [CrossRef] [PubMed]
- Lejeune, M.P.; Westerterp, K.R.; Adam, T.C.; Luscombe-Marsh, N.D.; Westerterp-Plantenga, M.S. Ghrelin and glucagon-like peptide 1 concentrations, 24-h satiety, and energy and substrate metabolism during a high-protein diet and measured in a respiration chamber. Am. J. Clin. Nutr. 2006, 83, 89–94. [Google Scholar] [CrossRef]
- Mansour, A.; Hosseini, S.; Larijani, B.; Pajouhi, M.; Mohajeri-Tehrani, M.R. Nutrients related to glp1 secretory responses. Nutrition 2013, 29, 813–820. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Koehler, J.; Yusta, B.; Bahrami, J.; Matthews, D.; Rafii, M.; Pencharz, P.B.; Drucker, D.J. Enteroendocrine-derived glucagon-like peptide-2 controls intestinal amino acid transport. Mol. Metab. 2017, 6, 245–255. [Google Scholar] [CrossRef]
- Meier, J.J.; Nauck, M.A.; Schmidt, W.E.; Gallwitz, B. Gastric inhibitory polypeptide: The neglected incretin revisited. Regul. Pept. 2002, 107, 1–13. [Google Scholar] [CrossRef]
- Nauck, M.A.; Bartels, E.; Orskov, C.; Ebert, R.; Creutzfeldt, W. Additive insulinotropic effects of exogenous synthetic human gastric inhibitory polypeptide and glucagon-like peptide-1-(7-36) amide infused at near-physiological insulinotropic hormone and glucose concentrations. J. Clin. Endocrinol. Metab. 1993, 76, 912–917. [Google Scholar]
- Eckel, R.H.; Fujimoto, W.Y.; Brunzell, J.D. Gastric inhibitory polypeptide enhanced lipoprotein lipase activity in cultured preadipocytes. Diabetes 1979, 28, 1141–1142. [Google Scholar] [CrossRef]
- Beck, B.; Max, J.P. Gastric inhibitory polypeptide enhancement of the insulin effect on fatty acid incorporation into adipose tissue in the rat. Regul. Pept. 1983, 7, 3–8. [Google Scholar] [CrossRef]
- Meier, J.J.; Goetze, O.; Anstipp, J.; Hagemann, D.; Holst, J.J.; Schmidt, W.E.; Gallwitz, B.; Nauck, M.A. Gastric inhibitory polypeptide does not inhibit gastric emptying in humans. Am. J. Physiol. Endocrinol. Metab. 2004, 286, E621–E625. [Google Scholar] [CrossRef] [PubMed]
- Cataland, S.; Crockett, S.E.; Brown, J.C.; Mazzaferri, E.L. Gastric inhibitory polypeptide (gip) stimulation by oral glucose in man. J. Clin. Endocrinol. Metab. 1974, 39, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Sirinek, K.R.; Crockett, S.E.; Mazzaferri, E.L.; Cataland, S.; Thomford, N.R. Release of gastric inhibitory polypeptide: Comparison of glucose and fat as stimuli. Surg. Forum 1974, 25, 361–363. [Google Scholar] [PubMed]
- Elliott, R.M.; Morgan, L.M.; Tredger, J.A.; Deacon, S.; Wright, J.; Marks, V. Glucagon-like peptide-1 (7-36) amide and glucose-dependent insulinotropic polypeptide secretion in response to nutrient ingestion in man: Acute post-prandial and 24-h secretion patterns. J. Endocrinol. 1993, 138, 159–166. [Google Scholar] [CrossRef] [PubMed]
- Sarson, D.L.; Bryant, M.G.; Bloom, S.R. A radioimmunoassay of gastric inhibitory polypeptide in human plasma. J. Endocrinol. 1980, 85, 487–496. [Google Scholar] [CrossRef] [PubMed]
- Adrian, T.E.; Ferri, G.L.; Bacarese-Hamilton, A.J.; Fuessl, H.S.; Polak, J.M.; Bloom, S.R. Human distribution and release of a putative new gut hormone, peptide yy. Gastroenterology 1985, 89, 1070–1077. [Google Scholar] [CrossRef]
- Chan, J.L.; Stoyneva, V.; Kelesidis, T.; Raciti, P.; Mantzoros, C.S. Peptide yy levels are decreased by fasting and elevated following caloric intake but are not regulated by leptin. Diabetologia 2006, 49, 169–173. [Google Scholar] [CrossRef]
- Pironi, L.; Stanghellini, V.; Miglioli, M.; Corinaldesi, R.; De Giorgio, R.; Ruggeri, E.; Tosetti, C.; Poggioli, G.; Morselli Labate, A.M.; Monetti, N.; et al. Fat-induced ileal brake in humans: A dose-dependent phenomenon correlated to the plasma levels of peptide yy. Gastroenterology 1993, 105, 733–739. [Google Scholar] [CrossRef]
- Boey, D.; Sainsbury, A.; Herzog, H. The role of peptide yy in regulating glucose homeostasis. Peptides 2007, 28, 390–395. [Google Scholar] [CrossRef]
- Batterham, R.L.; Heffron, H.; Kapoor, S.; Chivers, J.E.; Chandarana, K.; Herzog, H.; Le Roux, C.W.; Thomas, E.L.; Bell, J.D.; Withers, D.J. Critical role for peptide yy in protein-mediated satiation and body-weight regulation. Cell Metab. 2006, 4, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Batterham, R.L.; ffytche, D.H.; Rosenthal, J.M.; Zelaya, F.O.; Barker, G.J.; Withers, D.J.; Williams, S.C. Pyy modulation of cortical and hypothalamic brain areas predicts feeding behaviour in humans. Nature 2007, 450, 106–109. [Google Scholar] [CrossRef] [PubMed]
- Batterham, R.L.; Cowley, M.A.; Small, C.J.; Herzog, H.; Cohen, M.A.; Dakin, C.L.; Wren, A.M.; Brynes, A.E.; Low, M.J.; Ghatei, M.A.; et al. Gut hormone pyy(3-36) physiologically inhibits food intake. Nature 2002, 418, 650–654. [Google Scholar] [CrossRef] [PubMed]
- Kir, S.; Kliewer, S.A.; Mangelsdorf, D.J. Roles of fgf19 in liver metabolism. Cold Spring Harb. Symp. Quant. Biol. 2011, 76, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Avila, M.A.; Moschetta, A. The fxr-fgf19 gut-liver axis as a novel “hepatostat”. Gastroenterology 2015, 149, 537–540. [Google Scholar] [CrossRef] [PubMed]
- Rask-Madsen, C.; Kahn, C.R. Tissue-specific insulin signaling, metabolic syndrome, and cardiovascular disease. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 2052–2059. [Google Scholar] [CrossRef]
- Newsholme, P.; Cruzat, V.; Arfuso, F.; Keane, K. Nutrient regulation of insulin secretion and action. J. Endocrinol. 2014, 221, R105–R120. [Google Scholar] [CrossRef] [PubMed]
- Komatsu, M.; Takei, M.; Ishii, H.; Sato, Y. Glucose-stimulated insulin secretion: A newer perspective. J. Diabetes Investig. 2013, 4, 511–516. [Google Scholar] [CrossRef]
- Floyd, J.C., Jr.; Fajans, S.S.; Conn, J.W.; Knopf, R.F.; Rull, J. Stimulation of insulin secretion by amino acids. J. Clin. Investig. 1966, 45, 1487–1502. [Google Scholar] [CrossRef]
- Floyd, J.C., Jr.; Fajans, S.S.; Conn, J.W.; Knopf, R.F.; Rull, J. Insulin secretion in response to protein ingestion. J. Clin. Investig. 1966, 45, 1479–1486. [Google Scholar] [CrossRef]
- Rocha, D.M.; Faloona, G.R.; Unger, R.H. Glucagon-stimulating activity of 20 amino acids in dogs. J. Clin. Investig. 1972, 51, 2346–2351. [Google Scholar] [CrossRef]
- Kuhara, T.; Ikeda, S.; Ohneda, A.; Sasaki, Y. Effects of intravenous infusion of 17 amino acids on the secretion of gh, glucagon, and insulin in sheep. Am. J. Physiol. 1991, 260, E21–E26. [Google Scholar] [CrossRef]
- Holst, J.J.; Holland, W.; Gromada, J.; Lee, Y.; Unger, R.H.; Yan, H.; Sloop, K.W.; Kieffer, T.J.; Damond, N.; Herrera, P.L. Insulin and glucagon: Partners for life. Endocrinology 2017, 158, 696–701. [Google Scholar] [CrossRef]
- James, H.A.; O’Neill, B.T.; Nair, K.S. Insulin regulation of proteostasis and clinical implications. Cell Metab. 2017, 26, 310–323. [Google Scholar] [CrossRef]
- Rossetti, L.; Rothman, D.L.; DeFronzo, R.A.; Shulman, G.I. Effect of dietary protein on in vivo insulin action and liver glycogen repletion. Am. J. Physiol. 1989, 257, E212–E219. [Google Scholar] [CrossRef]
- Solon-Biet, S.M.; McMahon, A.C.; Ballard, J.W.; Ruohonen, K.; Wu, L.E.; Cogger, V.C.; Warren, A.; Huang, X.; Pichaud, N.; Melvin, R.G.; et al. The ratio of macronutrients, not caloric intake, dictates cardiometabolic health, aging, and longevity in ad libitum-fed mice. Cell Metab. 2014, 19, 418–430. [Google Scholar] [CrossRef]
- Tremblay, F.; Lavigne, C.; Jacques, H.; Marette, A. Role of dietary proteins and amino acids in the pathogenesis of insulin resistance. Annu. Rev. Nutr. 2007, 27, 293–310. [Google Scholar] [CrossRef]
- Maida, A.; Zota, A.; Sjoberg, K.A.; Schumacher, J.; Sijmonsma, T.P.; Pfenninger, A.; Christensen, M.M.; Gantert, T.; Fuhrmeister, J.; Rothermel, U.; et al. A liver stress-endocrine nexus promotes metabolic integrity during dietary protein dilution. J. Clin. Investig. 2016, 126, 3263–3278. [Google Scholar] [CrossRef]
- Maida, A.; Zota, A.; Vegiopoulos, A.; Appak-Baskoy, S.; Augustin, H.G.; Heikenwalder, M.; Herzig, S.; Rose, A.J. Dietary protein dilution limits dyslipidemia in obesity through fgf21-driven fatty acid clearance. J. Nutr. Biochem. 2018, 57, 189–196. [Google Scholar] [CrossRef]
- Lin, H.V.; Accili, D. Hormonal regulation of hepatic glucose production in health and disease. Cell Metab. 2011, 14, 9–19. [Google Scholar] [CrossRef]
- Muller, W.A.; Faloona, G.R.; Aguilar-Parada, E.; Unger, R.H. Abnormal alpha-cell function in diabetes. Response to carbohydrate and protein ingestion. N. Engl. J. Med. 1970, 283, 109–115. [Google Scholar] [CrossRef]
- Ang, T.; Bruce, C.R.; Kowalski, G.M. Postprandial aminogenic insulin and glucagon secretion can stimulate glucose flux in humans. Diabetes 2019, 68, 939–946. [Google Scholar] [CrossRef]
- Assan, R.; Attali, J.R.; Ballerio, G.; Boillot, J.; Girard, J.R. Glucagon secretion induced by natural and artificial amino acids in the perfused rat pancreas. Diabetes 1977, 26, 300–307. [Google Scholar] [CrossRef]
- Pagliara, A.S.; Stillings, S.N.; Hover, B.; Martin, D.M.; Matschinsky, F.M. Glucose modulation of amino acid-induced glucagon and insulin release in the isolated perfused rat pancreas. J. Clin. Investig. 1974, 54, 819–832. [Google Scholar] [CrossRef]
- Hayashi, Y.; Seino, Y. Regulation of amino acid metabolism and alpha-cell proliferation by glucagon. J. Diabetes Investig. 2018, 9, 467–472. [Google Scholar] [CrossRef]
- Wahren, J.; Felig, P.; Hagenfeldt, L. Effect of protein ingestion on splanchnic and leg metabolism in normal man and in patients with diabetes mellitus. J. Clin. Investig. 1976, 57, 987–999. [Google Scholar] [CrossRef]
- Cheng, X.; Kim, S.Y.; Okamoto, H.; Xin, Y.; Yancopoulos, G.D.; Murphy, A.J.; Gromada, J. Glucagon contributes to liver zonation. Proc. Natl. Acad. Sci. USA 2018, 115, E4111–E4119. [Google Scholar] [CrossRef]
- Yang, J.; MacDougall, M.L.; McDowell, M.T.; Xi, L.; Wei, R.; Zavadoski, W.J.; Molloy, M.P.; Baker, J.D.; Kuhn, M.; Cabrera, O.; et al. Polyomic profiling reveals significant hepatic metabolic alterations in glucagon-receptor (gcgr) knockout mice: Implications on anti-glucagon therapies for diabetes. BMC Genom. 2011, 12, 281. [Google Scholar] [CrossRef]
- Kim, J.; Okamoto, H.; Huang, Z.; Anguiano, G.; Chen, S.; Liu, Q.; Cavino, K.; Xin, Y.; Na, E.; Hamid, R.; et al. Amino acid transporter slc38a5 controls glucagon receptor inhibition-induced pancreatic alpha cell hyperplasia in mice. Cell Metab. 2017, 25, 1348–1361. [Google Scholar] [CrossRef]
- Longuet, C.; Robledo, A.M.; Dean, E.D.; Dai, C.; Ali, S.; McGuinness, I.; de Chavez, V.; Vuguin, P.M.; Charron, M.J.; Powers, A.C.; et al. Liver-specific disruption of the murine glucagon receptor produces alpha-cell hyperplasia: Evidence for a circulating alpha-cell growth factor. Diabetes 2013, 62, 1196–1205. [Google Scholar] [CrossRef]
- Dean, E.D.; Li, M.; Prasad, N.; Wisniewski, S.N.; Von Deylen, A.; Spaeth, J.; Maddison, L.; Botros, A.; Sedgeman, L.R.; Bozadjieva, N.; et al. Interrupted glucagon signaling reveals hepatic alpha cell axis and role for l-glutamine in alpha cell proliferation. Cell Metab. 2017, 25, 1362–1373.e5. [Google Scholar] [CrossRef]
- Solloway, M.J.; Madjidi, A.; Gu, C.; Eastham-Anderson, J.; Clarke, H.J.; Kljavin, N.; Zavala-Solorio, J.; Kates, L.; Friedman, B.; Brauer, M.; et al. Glucagon couples hepatic amino acid catabolism to mtor-dependent regulation of alpha-cell mass. Cell Rep. 2015, 12, 495–510. [Google Scholar] [CrossRef]
- Galsgaard, K.D.; Winther-Sorensen, M.; Orskov, C.; Kissow, H.; Poulsen, S.S.; Vilstrup, H.; Prehn, C.; Adamski, J.; Jepsen, S.L.; Hartmann, B.; et al. Disruption of glucagon receptor signaling causes hyperaminoacidemia exposing a possible liver-alpha-cell axis. Am. J. Physiol. Endocrinol. Metab. 2018, 314, E93–E103. [Google Scholar] [CrossRef]
- Galsgaard, K.D.; Winther-Sorensen, M.; Pedersen, J.; Kjeldsen, S.A.S.; Rosenkilde, M.M.; Wewer Albrechtsen, N.J.; Holst, J.J. Glucose and amino acid metabolism in mice depend mutually on glucagon and insulin receptor signaling. Am. J. Physiol. Endocrinol. Metab. 2019, 316, E660–E673. [Google Scholar] [CrossRef]
- Wewer Albrechtsen, N.J.; Faerch, K.; Jensen, T.M.; Witte, D.R.; Pedersen, J.; Mahendran, Y.; Jonsson, A.E.; Galsgaard, K.D.; Winther-Sorensen, M.; Torekov, S.S.; et al. Evidence of a liver-alpha cell axis in humans: Hepatic insulin resistance attenuates relationship between fasting plasma glucagon and glucagonotropic amino acids. Diabetologia 2018, 61, 671–680. [Google Scholar] [CrossRef]
- Habegger, K.M.; Stemmer, K.; Cheng, C.; Muller, T.D.; Heppner, K.M.; Ottaway, N.; Holland, J.; Hembree, J.L.; Smiley, D.; Gelfanov, V.; et al. Fibroblast growth factor 21 mediates specific glucagon actions. Diabetes 2013, 62, 1453–1463. [Google Scholar] [CrossRef]
- Omar, B.A.; Andersen, B.; Hald, J.; Raun, K.; Nishimura, E.; Ahren, B. Fibroblast growth factor 21 (fgf21) and glucagon-like peptide 1 contribute to diabetes resistance in glucagon receptor-deficient mice. Diabetes 2014, 63, 101–110. [Google Scholar] [CrossRef]
- Mighiu, P.I.; Yue, J.T.; Filippi, B.M.; Abraham, M.A.; Chari, M.; Lam, C.K.; Yang, C.S.; Christian, N.R.; Charron, M.J.; Lam, T.K. Hypothalamic glucagon signaling inhibits hepatic glucose production. Nat. Med. 2013, 19, 766–772. [Google Scholar] [CrossRef]
- LaPierre, M.P.; Abraham, M.A.; Yue, J.T.; Filippi, B.M.; Lam, T.K. Glucagon signalling in the dorsal vagal complex is sufficient and necessary for high-protein feeding to regulate glucose homeostasis in vivo. EMBO Rep. 2015, 16, 1299–1307. [Google Scholar] [CrossRef]
- Cooper, G.J.; Willis, A.C.; Clark, A.; Turner, R.C.; Sim, R.B.; Reid, K.B. Purification and characterization of a peptide from amyloid-rich pancreases of type 2 diabetic patients. Proc. Natl. Acad. Sci. USA 1987, 84, 8628–8632. [Google Scholar] [CrossRef]
- Lutz, T.A.; Geary, N.; Szabady, M.M.; Del Prete, E.; Scharrer, E. Amylin decreases meal size in rats. Physiol. Behav. 1995, 58, 1197–1202. [Google Scholar] [CrossRef]
- Lutz, T.A. Amylinergic control of food intake. Physiol. Behav. 2006, 89, 465–471. [Google Scholar] [CrossRef]
- Butler, P.C.; Chou, J.; Carter, W.B.; Wang, Y.N.; Bu, B.H.; Chang, D.; Chang, J.K.; Rizza, R.A. Effects of meal ingestion on plasma amylin concentration in niddm and nondiabetic humans. Diabetes 1990, 39, 752–756. [Google Scholar] [CrossRef]
- Nakazato, M.; Miyazato, M.; Asai, J.; Mitsukawa, T.; Kangawa, K.; Matsuo, H.; Matsukura, S. Islet amyloid polypeptide, a novel pancreatic peptide, is a circulating hormone secreted under glucose stimulation. Biochem. Biophys. Res. Commun. 1990, 169, 713–718. [Google Scholar] [CrossRef]
- Van Hulst, K.L.; Nieuwenhuis, M.G.; Hoppener, J.W.; Lips, C.J.; Blankenstein, M.A. Lack of islet amyloid polypeptide/amylin-immunoreactivity in urine collected from healthy volunteers after ingestion of a carbohydrate-rich meal. Exp. Clin. Endocrinol. Diabetes Off. J. Ger. Soc. Endocrinol. Ger. Diabetes Assoc. 1996, 104, 177–179. [Google Scholar] [CrossRef]
- Shahid, Z.; Singh, G. Physiology, islets of langerhans. In Statpearls; StatPearls Publishing: Treasure Island, FL, USA, 2019. [Google Scholar]
- Batterham, R.L.; Le Roux, C.W.; Cohen, M.A.; Park, A.J.; Ellis, S.M.; Patterson, M.; Frost, G.S.; Ghatei, M.A.; Bloom, S.R. Pancreatic polypeptide reduces appetite and food intake in humans. J. Clin. Endocrinol. Metab. 2003, 88, 3989–3992. [Google Scholar] [CrossRef]
- Katsuura, G.; Asakawa, A.; Inui, A. Roles of pancreatic polypeptide in regulation of food intake. Peptides 2002, 23, 323–329. [Google Scholar] [CrossRef]
- O’Toole, T.J.; Sharma, S. Physiology, somatostatin. In Statpearls; StatPearls Publishing: Treasure Island, FL, USA, 2019. [Google Scholar]
- Martinez, V.; Curi, A.P.; Torkian, B.; Schaeffer, J.M.; Wilkinson, H.A.; Walsh, J.H.; Tache, Y. High basal gastric acid secretion in somatostatin receptor subtype 2 knockout mice. Gastroenterology 1998, 114, 1125–1132. [Google Scholar] [CrossRef]
- Huising, M.O.; van der Meulen, T.; Huang, J.L.; Pourhosseinzadeh, M.S.; Noguchi, G.M. The difference delta-cells make in glucose control. Physiology 2018, 33, 403–411. [Google Scholar] [CrossRef]
- BonDurant, L.D.; Potthoff, M.J. Fibroblast growth factor 21: A versatile regulator of metabolic homeostasis. Annu. Rev. Nutr. 2018, 38, 173–196. [Google Scholar] [CrossRef]
- Domouzoglou, E.M.; Maratos-Flier, E. Fibroblast growth factor 21 is a metabolic regulator that plays a role in the adaptation to ketosis. Am. J. Clin. Nutr. 2011, 93, 901S–905S. [Google Scholar] [CrossRef] [PubMed]
- Inagaki, T.; Dutchak, P.; Zhao, G.; Ding, X.; Gautron, L.; Parameswara, V.; Li, Y.; Goetz, R.; Mohammadi, M.; Esser, V.; et al. Endocrine regulation of the fasting response by pparalpha-mediated induction of fibroblast growth factor 21. Cell Metab. 2007, 5, 415–425. [Google Scholar] [CrossRef] [PubMed]
- Dushay, J.R.; Toschi, E.; Mitten, E.K.; Fisher, F.M.; Herman, M.A.; Maratos-Flier, E. Fructose ingestion acutely stimulates circulating fgf21 levels in humans. Mol. Metab. 2015, 4, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Soberg, S.; Sandholt, C.H.; Jespersen, N.Z.; Toft, U.; Madsen, A.L.; Von Holstein-Rathlou, S.; Grevengoed, T.J.; Christensen, K.B.; Bredie, W.L.P.; Potthoff, M.J.; et al. Fgf21 is a sugar-induced hormone associated with sweet intake and preference in humans. Cell Metab. 2017, 25, 1045–1053. [Google Scholar] [CrossRef] [PubMed]
- Lundsgaard, A.M.; Fritzen, A.M.; Sjoberg, K.A.; Myrmel, L.S.; Madsen, L.; Wojtaszewski, J.F.P.; Richter, E.A.; Kiens, B. Circulating fgf21 in humans is potently induced by short term overfeeding of carbohydrates. Mol. Metab. 2017, 6, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Vinales, K.L.; Begaye, B.; Bogardus, C.; Walter, M.; Krakoff, J.; Piaggi, P. Fgf21 is a hormonal mediator of the human “thrifty” metabolic phenotype. Diabetes 2018, 68, 318–323. [Google Scholar] [CrossRef] [PubMed]
- Laeger, T.; Henagan, T.M.; Albarado, D.C.; Redman, L.M.; Bray, G.A.; Noland, R.C.; Munzberg, H.; Hutson, S.M.; Gettys, T.W.; Schwartz, M.W.; et al. Fgf21 is an endocrine signal of protein restriction. J. Clin. Investig. 2014, 124, 3913–3922. [Google Scholar] [CrossRef] [PubMed]
- Fontana, L.; Cummings, N.E.; Arriola Apelo, S.I.; Neuman, J.C.; Kasza, I.; Schmidt, B.A.; Cava, E.; Spelta, F.; Tosti, V.; Syed, F.A.; et al. Decreased consumption of branched-chain amino acids improves metabolic health. Cell Rep. 2016, 16, 520–530. [Google Scholar] [CrossRef] [PubMed]
- Perez-Marti, A.; Garcia-Guasch, M.; Tresserra-Rimbau, A.; Carrilho-Do-Rosario, A.; Estruch, R.; Salas-Salvado, J.; Martinez-Gonzalez, M.A.; Lamuela-Raventos, R.; Marrero, P.F.; Haro, D.; et al. A low-protein diet induces body weight loss and browning of subcutaneous white adipose tissue through enhanced expression of hepatic fibroblast growth factor 21 (fgf21). Mol. Nutr. Food Res. 2017, 61, 1600725. [Google Scholar] [CrossRef] [PubMed]
- Gosby, A.K.; Lau, N.S.; Tam, C.S.; Iglesias, M.A.; Morrison, C.D.; Caterson, I.D.; Brand-Miller, J.; Conigrave, A.D.; Raubenheimer, D.; Simpson, S.J. Raised fgf-21 and triglycerides accompany increased energy intake driven by protein leverage in lean, healthy individuals: A randomised trial. PLoS ONE 2016, 11, e0161003. [Google Scholar] [CrossRef] [PubMed]
- Kennedy, A.R.; Pissios, P.; Otu, H.; Roberson, R.; Xue, B.; Asakura, K.; Furukawa, N.; Marino, F.E.; Liu, F.F.; Kahn, B.B.; et al. A high-fat, ketogenic diet induces a unique metabolic state in mice. Am. J. Physiol. Endocrinol. Metab. 2007, 292, E1724–E1739. [Google Scholar] [CrossRef] [PubMed]
- Badman, M.K.; Pissios, P.; Kennedy, A.R.; Koukos, G.; Flier, J.S.; Maratos-Flier, E. Hepatic fibroblast growth factor 21 is regulated by pparalpha and is a key mediator of hepatic lipid metabolism in ketotic states. Cell Metab. 2007, 5, 426–437. [Google Scholar] [CrossRef] [PubMed]
- Bielohuby, M.; Sisley, S.; Sandoval, D.; Herbach, N.; Zengin, A.; Fischereder, M.; Menhofer, D.; Stoehr, B.J.; Stemmer, K.; Wanke, R.; et al. Impaired glucose tolerance in rats fed low-carbohydrate, high-fat diets. Am. J. Physiol. Endocrinol. Metab. 2013, 305, E1059–E1070. [Google Scholar] [CrossRef] [PubMed]
- Solon-Biet, S.M.; Cogger, V.C.; Pulpitel, T.; Heblinski, M.; Wahl, D.; McMahon, A.C.; Warren, A.; Durrant-Whyte, J.; Walters, K.A.; Krycer, J.R.; et al. Defining the nutritional and metabolic context of fgf21 using the geometric framework. Cell Metab. 2016, 24, 555–565. [Google Scholar] [CrossRef] [PubMed]
- Trevino-Villarreal, J.H.; Reynolds, J.S.; Bartelt, A.; Langston, P.K.; MacArthur, M.R.; Arduini, A.; Tosti, V.; Veronese, N.; Bertozzi, B.; Brace, L.E.; et al. Dietary protein restriction reduces circulating vldl triglyceride levels via crebh-apoa5-dependent and -independent mechanisms. JCI Insight 2018, 3, e99470. [Google Scholar] [CrossRef] [PubMed]
- Zhuo, Y.; Hua, L.; Feng, B.; Jiang, X.; Li, J.; Jiang, D.; Huang, X.; Zhu, Y.; Li, Z.; Yan, L.; et al. Fibroblast growth factor 21 coordinates adiponectin to mediate the beneficial effects of low-protein diet on primordial follicle reserve. EBioMedicine 2019, 41, 623–635. [Google Scholar] [CrossRef]
- Hill, C.M.; Laeger, T.; Dehner, M.; Albarado, D.C.; Clarke, B.; Wanders, D.; Burke, S.J.; Collier, J.J.; Qualls-Creekmore, E.; Solon-Biet, S.M.; et al. Fgf21 signals protein status to the brain and adaptively regulates food choice and metabolism. Cell Rep. 2019, 27, 2934–2947. [Google Scholar] [CrossRef] [PubMed]
- Hill, C.M.; Laeger, T.; Albarado, D.C.; McDougal, D.H.; Berthoud, H.R.; Munzberg, H.; Morrison, C.D. Low protein-induced increases in fgf21 drive ucp1-dependent metabolic but not thermoregulatory endpoints. Sci. Rep. 2017, 7, 8209. [Google Scholar] [CrossRef] [PubMed]
- Maida, A.; Chan, J.S.K.; Sjoberg, K.A.; Zota, A.; Schmoll, D.; Kiens, B.; Herzig, S.; Rose, A.J. Repletion of branched chain amino acids reverses mtorc1 signaling but not improved metabolism during dietary protein dilution. Mol. Metab. 2017, 6, 873–881. [Google Scholar] [CrossRef]
- Piper, M.D.W.; Soultoukis, G.A.; Blanc, E.; Mesaros, A.; Herbert, S.L.; Juricic, P.; He, X.; Atanassov, I.; Salmonowicz, H.; Yang, M.; et al. Matching dietary amino acid balance to the in silico-translated exome optimizes growth and reproduction without cost to lifespan. Cell Metab. 2017, 25, 1206. [Google Scholar] [CrossRef]
- Jiang, Y.; Rose, A.J.; Sijmonsma, T.P.; Broer, A.; Pfenninger, A.; Herzig, S.; Schmoll, D.; Broer, S. Mice lacking neutral amino acid transporter b(0)at1 (slc6a19) have elevated levels of fgf21 and glp-1 and improved glycaemic control. Mol. Metab. 2015, 4, 406–417. [Google Scholar] [CrossRef] [PubMed]
- Solon-Biet, S.M.; Cogger, V.C.; Pulpitel, T.; Wahl, D.; Clark, X.; Bagley, E.E.; Gregoriou, G.C.; Senior, A.M.; Wang, Q.-P.; Brandon, A.E.; et al. Branched-chain amino acids impact health and lifespan indirectly via amino acid balance and appetite control. Nat. Metab. 2019, 1, 532–545. [Google Scholar] [CrossRef]
- Green, C.L.; Lamming, D.W. Regulation of metabolic health by essential dietary amino acids. Mech. Ageing Dev. 2019, 177, 186–200. [Google Scholar] [CrossRef]
- Spring, S.; Singh, A.; Zapata, R.C.; Chelikani, P.K.; Pezeshki, A. Methionine restriction partly recapitulates the sympathetically mediated enhanced energy expenditure induced by total amino acid restriction in rats. Nutrients 2019, 11, 707. [Google Scholar] [CrossRef] [PubMed]
- Zapata, R.C.; Singh, A.; Pezeshki, A.; Chelikani, P.K. Tryptophan restriction partially recapitulates the age-dependent effects of total amino acid restriction on energy balance in diet-induced obese rats. J. Nutr. Biochem. 2019, 65, 115–127. [Google Scholar] [CrossRef]
- Levine, M.E.; Suarez, J.A.; Brandhorst, S.; Balasubramanian, P.; Cheng, C.W.; Madia, F.; Fontana, L.; Mirisola, M.G.; Guevara-Aguirre, J.; Wan, J.; et al. Low protein intake is associated with a major reduction in igf-1, cancer, and overall mortality in the 65 and younger but not older population. Cell Metab. 2014, 19, 407–417. [Google Scholar] [CrossRef]
- Gokarn, R.; Solon-Biet, S.M.; Cogger, V.C.; Cooney, G.J.; Wahl, D.; McMahon, A.C.; Mitchell, J.R.; Mitchell, S.J.; Hine, C.; de Cabo, R.; et al. Long-term dietary macronutrients and hepatic gene expression in aging mice. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2018, 73, 1618–1625. [Google Scholar] [CrossRef]
- Friedman, J. 20 years of leptin: Leptin at 20: An overview. J. Endocrinol. 2014, 223, T1–T8. [Google Scholar] [CrossRef]
- Flier, J.S. Starvation in the midst of plenty: Reflections on the history and biology of insulin and leptin. Endocr. Rev. 2019, 40, 1–16. [Google Scholar] [CrossRef]
- Specter, S.E.; Hamilton, J.S.; Stern, J.S.; Horwitz, B.A. Chronic protein restriction does not alter energetic efficiency or brown adipose tissue thermogenic capacity in genetically obese (fa/fa) zucker rats. J. Nutr. 1995, 125, 2183–2193. [Google Scholar] [CrossRef]
- Robertson, L.T.; Trevino-Villarreal, J.H.; Mejia, P.; Grondin, Y.; Harputlugil, E.; Hine, C.; Vargas, D.; Zheng, H.; Ozaki, C.K.; Kristal, B.S.; et al. Protein and calorie restriction contribute additively to protection from renal ischemia reperfusion injury partly via leptin reduction in male mice. J. Nutr. 2015, 145, 1717–1727. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.V.; Scherer, P.E. Adiponectin, the past two decades. J. Mol. Cell Biol. 2016, 8, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Rose, A.J. Dietary protein and age-dependent female fertility: Fgf21 trumps mtorc1. EBioMedicine 2019, 41, 32–33. [Google Scholar] [CrossRef] [PubMed]
- Simpson, S.J.; Le Couteur, D.G.; Raubenheimer, D. Putting the balance back in diet. Cell 2015, 161, 18–23. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rose, A.J. Role of Peptide Hormones in the Adaptation to Altered Dietary Protein Intake. Nutrients 2019, 11, 1990. https://doi.org/10.3390/nu11091990
Rose AJ. Role of Peptide Hormones in the Adaptation to Altered Dietary Protein Intake. Nutrients. 2019; 11(9):1990. https://doi.org/10.3390/nu11091990
Chicago/Turabian StyleRose, Adam J. 2019. "Role of Peptide Hormones in the Adaptation to Altered Dietary Protein Intake" Nutrients 11, no. 9: 1990. https://doi.org/10.3390/nu11091990
APA StyleRose, A. J. (2019). Role of Peptide Hormones in the Adaptation to Altered Dietary Protein Intake. Nutrients, 11(9), 1990. https://doi.org/10.3390/nu11091990




