Validation of a Food Frequency Questionnaire to Assess Intake of n-3 Polyunsaturated Fatty Acids in Switzerland
Abstract
1. Introduction
2. Methods
2.1. Questionnaire
2.2. Study Population
2.3. Study Design
2.4. Dietary Record Analysis
2.5. Fatty Acids Analysis in RBC
2.6. Data Analysis
3. Results
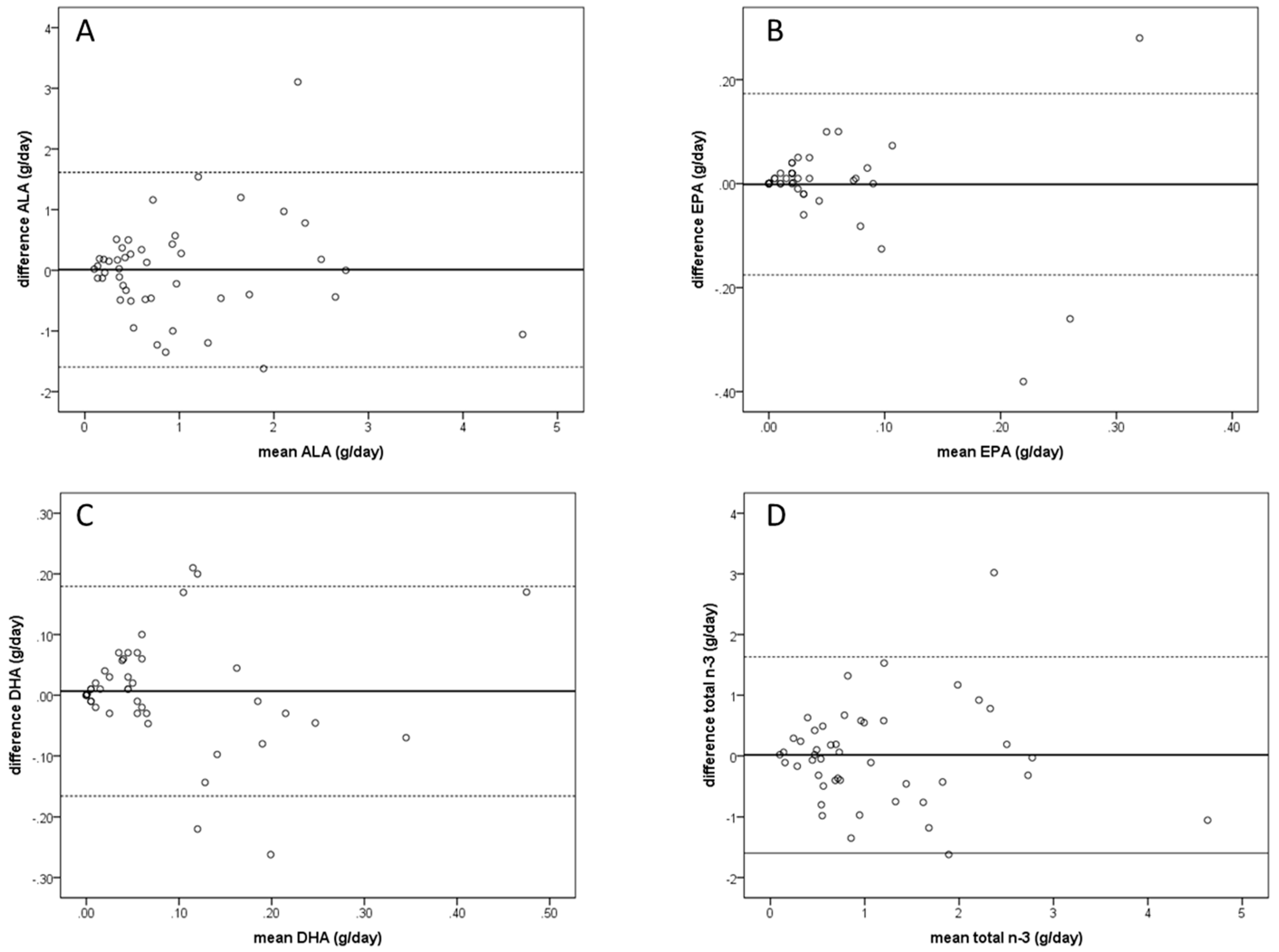
3.1. Questionnaire Validity Based on FR
3.2. Questionnaire Validity Based on RBC PUFA Composition
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kromann, N.; Green, A. Epidemiological-Studies in the Upernavik District, Greenland—Incidence of Some Chronic Diseases 1950–1974. Acta Med. Scand. 1980, 208, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Weylandt, K.H.; Serini, S.; Chen, Y.Q.; Su, H.M.; Lim, K.; Cittadini, A.; Calviello, G. Omega-3 Polyunsaturated Fatty Acids: The Way Forward in Times of Mixed Evidence. Biomed. Res. Int. 2015, 2015, 143109. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. BBA Mol. Cell Biol. Lipids 2015, 1851, 469–484. [Google Scholar] [CrossRef] [PubMed]
- Dyall, S.C. Long-chain omega-3 fatty acids and the brain: A review of the independent and shared effects of EPA, DPA and DHA. Front. Aging Neurosci. 2015, 7, 52. [Google Scholar] [CrossRef] [PubMed]
- McNamara, R.K. Role of Omega-3 Fatty Acids in the Etiology, Treatment, and Prevention of Depression: Current Status and Future Directions. J. Nutr. Intermed. Metab. 2016, 5, 96–106. [Google Scholar] [CrossRef] [PubMed]
- Del Gobbo, L.C.; Imamura, F.; Aslibekyan, S.; Marklund, M.; Virtanen, J.K.; Wennberg, M.; Yakoob, M.Y.; Chiuve, S.E.; dela Cruz, L.; Frazier-Wood, A.C.; et al. omega-3 Polyunsaturated Fatty Acid Biomarkers and Coronary Heart Disease Pooling Project of 19 Cohort Studies. JAMA Intern. Med. 2016, 176, 1155–1166. [Google Scholar] [CrossRef]
- Abdelhamid, A.S.; Brown, T.J.; Brainard, J.S.; Biswas, P.; Thorpe, G.C.; Moore, H.J.; Deane, K.H.; AlAbdulghafoor, F.K.; Summerbell, C.D.; Worthington, H.V.; et al. Omega-3 fatty acids for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2018, 11, CD003177. [Google Scholar]
- Simopoulos, A.P. Evolutionary Aspects of Diet: The Omega-6/Omega-3 Ratio and the Brain. Mol. Neurobiol. 2011, 44, 203–215. [Google Scholar] [CrossRef]
- Simopoulos, A.P. Evolutionary aspects of the dietary omega-6/omega-3 fatty acid Ratio: Medical implications. In Evolutionary Thinking in Medicine; Springer: Berlin/Heidelberg, Germany, 2016; pp. 119–134. [Google Scholar]
- Serra-Majem, L.; Nissensohn, M.; Overby, N.C.; Fekete, K. Dietary methods and biomarkers of omega 3 fatty acids: A systematic review. Br. J. Nutr. 2012, 107, S64–S76. [Google Scholar] [CrossRef]
- Hodge, A.M.; Simpson, J.A.; Gibson, R.A.; Sinclair, A.J.; Makrides, M.; O’Dea, K.; English, D.R.; Giles, G.G. Plasma phospholipid fatty acid composition as a biomarker of habitual dietary fat intake in an ethnically diverse cohort. Nutr. Metab. Cardiovas 2007, 17, 415–426. [Google Scholar] [CrossRef]
- Arterburn, L.M.; Hall, E.B.; Oken, H. Distribution, interconversion, and dose response of n-3 fatty acids in humans. Am. J. Clin. Nutr. 2006, 83, 1467s–1476s. [Google Scholar] [CrossRef] [PubMed]
- Gibson, R.S. Nutritional Assessment: A Laboratory Manual; Oxford University Press: Oxford, UK, 1993; pp. 79–100. [Google Scholar]
- Sublette, M.E.; Isaacson, C.J.S.; Cooper, T.B.; Fekri, S.; Vanegas, N.; Galfalvy, H.C.; Oquendo, M.A.; Mann, J.J. Validation of a Food Frequency Questionnaire to Assess Intake of n-3 Polyunsaturated Fatty Acids in Subjects with and without Major Depressive Disorder. J. Am. Diet. Assoc. 2011, 111, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Camenzind-Frey, E.; Zuberbühler, C. Schweizerisches Fotobuch/Livre Photo Suisse/Manuale Fotografico Svizzero (Swiss Picture Book); Bundesamt für Gesundheit BAG und Bundesamt für Lebensmittelsicherheit und Veterinärwesen BLV: Bern, Switzerland, 2014. [Google Scholar]
- Souci, S.W.; FAchmann, W.; Kraut, H. Food Composition and Nutrition Tables, 8th ed.; MedPharm Scientific Publishers: Stuttgart, Germany, 2016. [Google Scholar]
- USDA. USDA Food Composition Databases; United States Department of Agriculture: Washington, DC, USA, 2017. [Google Scholar]
- Moser, A.B.; Jones, D.S.; Raymond, G.V.; Moser, H.W. Plasma and red blood cell fatty acids in peroxisomal disorders. Neurochem. Res. 1999, 24, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Weiss, G.A.; Troxler, H.; Klinke, G.; Rogler, D.; Braegger, C.; Hersberger, M. High levels of anti-inflammatory and pro-resolving lipid mediators lipoxins and resolvins and declining docosahexaenoic acid levels in human milk during the first month of lactation. Lipids Health Dis. 2013, 12, 89. [Google Scholar] [CrossRef] [PubMed]
- Rahmawaty, S.; Charlton, K.; Lyons-Wall, P.; Meyer, B.J. Development and validation of a food frequency questionnaire to assess omega-3 long chain polyunsaturated fatty acid intake in Australian children aged 9–13 years. J. Hum. Nutr. Diet. 2017, 30, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Takechi, R.; Alfonso, H.; Harrison, A.; Hiramatsu, N.; Ishisaka, A.; Tanaka, A.; Tan, L.; Lee, A.H. Assessing self-reported green tea and coffee consumption by food frequency questionnaire and food record and their association with polyphenol biomarkers in Japanese women. Asia Pac. J. Clin. Nutr. 2018, 27, 460–465. [Google Scholar] [PubMed]
- Pampaloni, B.; Bartolini, E.; Barbieri, M.; Piscitelli, P.; Di Tanna, G.L.; Giolli, L.; Brandi, M.L. Validation of a Food-Frequency Questionnaire for the Assessment of Calcium Intake in Schoolchildren Aged 9–10 Years. Calcif. Tissue Int. 2013, 93, 23–38. [Google Scholar] [CrossRef]
- Da Silva, D.C.G.; Segheto, W.; de Lima, M.F.C.; Pessoa, M.C.; Peluzio, M.C.G.; Marchioni, D.M.L.; Cunha, D.B.; Longo, G.Z. Using the method of triads in the validation of a food frequency questionnaire to assess the consumption of fatty acids in adults. J. Hum. Nutr. Diet. 2018, 31, 85–95. [Google Scholar] [CrossRef]
- Goyens, P.L.; Spilker, M.E.; Zock, P.L.; Katan, M.B.; Mensink, R.P. Conversion of alpha-linolenic acid in humans is influenced by the absolute amounts of alpha-linolenic acid and linoleic acid in the diet and not by their ratio. Am. J. Clin. Nutr. 2006, 84, 44–53. [Google Scholar] [CrossRef]
- Schaeffer, L.; Gohlke, H.; Muller, M.; Heid, I.M.; Palmer, L.J.; Kompauer, I.; Demmelmair, H.; Illig, T.; Koletzko, B.; Heinrich, J. Common genetic variants of the FADS1 FADS2 gene cluster and their reconstructed haplotypes are associated with the fatty acid composition in phospholipids. Hum. Mol. Genet. 2006, 15, 1745–1756. [Google Scholar] [CrossRef]
- Astorg, P.; Bertrais, S.; Laporte, F.; Arnault, N.; Estaquio, C.; Galan, P.; Favier, A.; Hercberg, S. Plasma n-6 and n-3 polyunsaturated fatty acids as biomarkers of their dietary intakes: A cross-sectional study within a cohort of middle-aged French men and women. Eur. J. Clin. Nutr. 2008, 62, 1155–1161. [Google Scholar] [CrossRef]
- Holmes, M.D.; Powell, I.J.; Campos, H.; Stampfer, M.J.; Giovannucci, E.L.; Willett, W.C. Validation of a food frequency questionnaire measurement of selected nutrients using biological markers in African-American men. Eur. J. Clin. Nutr. 2007, 61, 1328–1336. [Google Scholar] [CrossRef][Green Version]
- Parra, M.S.; Schnaas, L.; Meydani, M.; Perroni, E.; Martinez, S.; Romieu, I. Erythrocyte cell membrane phospholipid levels compared against reported dietary intakes of polyunsaturated fatty acids in pregnant Mexican women. Public Health Nutr. 2002, 5, 931–937. [Google Scholar] [CrossRef]
- Burdge, G.C.; Wootton, S.A. Conversion of alpha-linolenic acid to eicosapentaenoic, docosapentaenoic and docosahexaenoic acids in young women. Br. J. Nutr. 2002, 88, 411–420. [Google Scholar] [CrossRef]
- Goyens, P.L.L.; Spilker, M.E.; Zock, P.L.; Katan, M.B.; Mensink, R.P. Compartmental modeling to quantify alpha-linolenic acid conversion after longer term intake of multiple tracer boluses. J. Lipid Res. 2005, 46, 1474–1483. [Google Scholar] [CrossRef]
- Mozaffarian, D.; Wu, J.H.Y. (n-3) Fatty Acids and Cardiovascular Health: Are Effects of EPA and DHA Shared or Complementary? J. Nutr. 2012, 142, 614s–625s. [Google Scholar] [CrossRef]
- ISSFAL. Recommendations for Intake of Polyunsaturated Fatty Acids in Healthy Aduls. Available online: https://www.issfal.org/statement-3 (accessed on 27 June 2019).
- Abedi, E.; Sahari, M.A. Long-chain polyunsaturated fatty acid sources and evaluation of their nutritional and functional properties. Food Sci. Nutr. 2014, 2, 443–463. [Google Scholar] [CrossRef]
- EFSA. Scientific Opinion on the Tolerable Upper Intake Level of Eicosapentaenoic Acid (EPA), Docosahexaenoic Acid (DHA) and Docosapentaenoic acid (DPA); European Food Safety Authority: Parma, Italy, 2012. [Google Scholar]
- Harris, W.S.; von Schacky, C. The Omega-3 Index: A new risk factor for death from coronary heart disease? Prev. Med. 2004, 39, 212–220. [Google Scholar] [CrossRef]
| FFQ vs. FR | FFQ vs. RBC | |
|---|---|---|
| n | 46 | 152 |
| Gender m/f (n (%)) | 10 (22%)/36 (78%) | 61 (40%)/91 (60%) |
| Age (year) | 24 (19–53) | 26 (18–59) |
| Height (m) | - | 1.71 (1.46–1.95) |
| Weight (kg) | - | 67.5 (36.0–109.5) |
| BMI (kg/m2) | - | 22.7 (16.6–35.2) |
| Fish oil supplements n (%) | - | 14 (9.2%) |
| Hormonal contraception n (%) 1 | - | 27 (29.7) |
| FR Intake | FFQ Intake | Correlations | Group Comparison 1 | ||||
|---|---|---|---|---|---|---|---|
| Median (g/day) | Range | Median (g/day) | Range | Sprearman’s r | p-Value | p-Value | |
| ALA | 0.645 | 0.060–5.160 | 0.585 | 0.040–4.103 | 0.526 | <0.001 | 0.915 |
| EPA | 0.010 | <0.00–0.410 | 0.024 | <0.001–0.460 | 0.585 | <0.001 | 0.196 |
| DHA | 0.020 | <0.001–0.390 | 0.050 | <0.001–0.560 | 0.586 | <0.001 | 0.467 |
| Total n-3 | 0.835 | 0.080–5.160 | 0.775 | 0.060–4.106 | 0.523 | <0.001 | 0.874 |
| FFQ Intake | % RBC Membrane Composition | Correlations | ||||
|---|---|---|---|---|---|---|
| Median (g/day) | Range | Median (%) | Range | Spearman’s r | p-Value | |
| ALA | 0.277 | 0.005–6.324 | 0.08 | 0.04–0.3 | 0.314 | <0.001 |
| EPA | 0.021 | <0.001–0.348 | 0.53 | 0.26–2.12 | 0.430 | <0.001 |
| DHA | 0.044 | <0.001–0.732 | 5.74 | 1.53–9.93 | 0.605 | <0.001 |
| Total n-3 | 0.433 | 0.005–6.455 | 7.47 | 3.12–13.75 | 0.211 | 0.009 |
| n-3 index | - | - | 6.13 | 1.95–12.05 | - | - |
| n-3/n-6 ratio | - | - | 4.30 | 2.08–11.57 | - | - |
| Unstandardized B | Model R2 | ||||||
|---|---|---|---|---|---|---|---|
| Constant | FFQ % 1 | Gender | Age | Supplement Intake | Fish Intake | ||
| RBC ALA | 0.102 | 0.013 ** | −0.014 * | <0.001 | −0.004 | 0.004 | 0.212 |
| RBC EPA | 0.368 | 1.497 ** | 0.016 | 0.005 * | 0.280 ** | −0.053 | 0.449 |
| RBC DHA | 5.344 | 5.029 ** | −0.631 * | 0.013 | 0.595 | −1.397 ** | 0.430 |
| RBC total n-3 | 7.158 | 0.246 * | −0.323 | 0.021 | 1.612 ** | −1.874 ** | 0.367 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Herter-Aeberli, I.; Graf, C.; Vollenweider, A.; Häberling, I.; Srikanthan, P.; Hersberger, M.; Berger, G.; Mathis, D. Validation of a Food Frequency Questionnaire to Assess Intake of n-3 Polyunsaturated Fatty Acids in Switzerland. Nutrients 2019, 11, 1863. https://doi.org/10.3390/nu11081863
Herter-Aeberli I, Graf C, Vollenweider A, Häberling I, Srikanthan P, Hersberger M, Berger G, Mathis D. Validation of a Food Frequency Questionnaire to Assess Intake of n-3 Polyunsaturated Fatty Acids in Switzerland. Nutrients. 2019; 11(8):1863. https://doi.org/10.3390/nu11081863
Chicago/Turabian StyleHerter-Aeberli, Isabelle, Celeste Graf, Anna Vollenweider, Isabelle Häberling, Pakeerathan Srikanthan, Martin Hersberger, Gregor Berger, and Déborah Mathis. 2019. "Validation of a Food Frequency Questionnaire to Assess Intake of n-3 Polyunsaturated Fatty Acids in Switzerland" Nutrients 11, no. 8: 1863. https://doi.org/10.3390/nu11081863
APA StyleHerter-Aeberli, I., Graf, C., Vollenweider, A., Häberling, I., Srikanthan, P., Hersberger, M., Berger, G., & Mathis, D. (2019). Validation of a Food Frequency Questionnaire to Assess Intake of n-3 Polyunsaturated Fatty Acids in Switzerland. Nutrients, 11(8), 1863. https://doi.org/10.3390/nu11081863





