Preterm Birth: A Narrative Review of the Current Evidence on Nutritional and Bioactive Solutions for Risk Reduction
Abstract
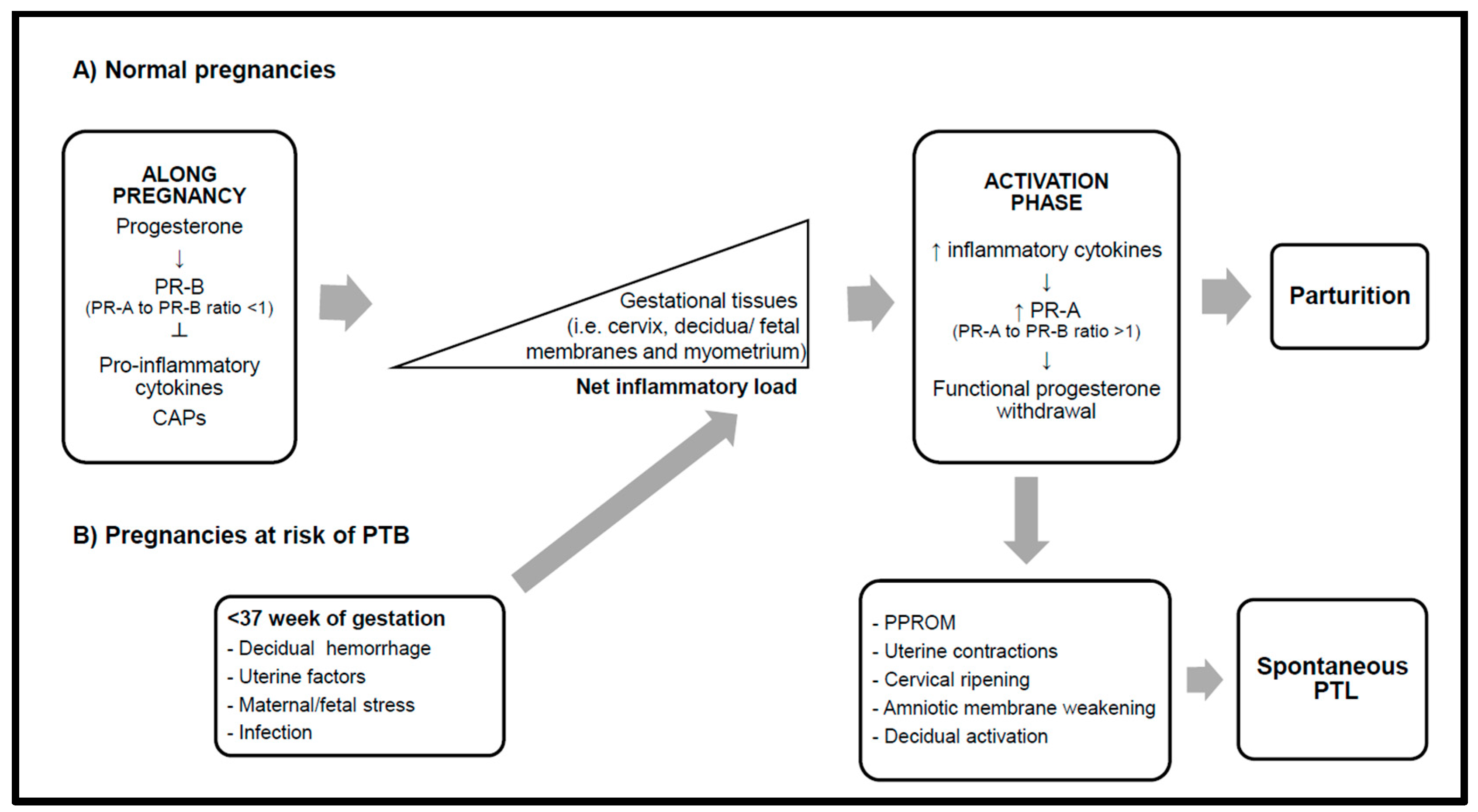
1. Introduction
2. Materials and Methods
3. Results
3.1. The Role of Nutrition in Reducing the Risk of PTB
3.1.1. Evidence from Dietary Pattern Analyses
3.1.2. Nutrient-Based Interventions
Omega 3 Fatty Acids
Other Macro and Micronutrients
3.2. Role of Probiotics in Reducing the Risk for PTB
3.2.1. Probiotic Intervention studies
3.2.2. The effect of Oral Probiotics on PTB Rates
3.2.3. The Effect of Vaginal Probiotics on Outcomes Related to PTB
3.2.4. Ongoing Probiotic Studies
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Blencowe, H.; Cousens, S.; Chou, D.; Oestergaard, M.; Say, L.; Moller, A.B.; Kinney, M.; Lawn, J. Born Too Soon: The global epidemiology of 15 million preterm births. Reprod. Health 2013, 10, S2. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Johnson, H.L.; Cousens, S.; Perin, J.; Scott, S.; Lawn, J.E.; Rudan, I.; Campbell, H.; Cibulskis, R.; Li, M.; et al. Global, regional, and national causes of child mortality: An updated systematic analysis for 2010 with time trends since 2000. Lancet 2012, 379, 2151–2161. [Google Scholar] [CrossRef]
- Raju, T.N.K.; Pemberton, V.L.; Saigal, S.; Blaisdell, C.J.; Moxey-Mims, M.; Buist, S. Long-Term Healthcare Outcomes of Preterm Birth: An Executive Summary of a Conference Sponsored by the National Institutes of Health. J. Pediatr. 2017, 181, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Luu, T.M.; Rehman Mian, M.O.; Nuyt, A.M. Long-Term Impact of Preterm Birth: Neurodevelopmental and Physical Health Outcomes. Clin. Perinatol. 2017, 44, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Jacob, J.; Lehne, M.; Mischker, A.; Klinger, N.; Zickermann, C.; Walker, J. Cost effects of preterm birth: A comparison of health care costs associated with early preterm, late preterm, and full-term birth in the first 3 years after birth. Eur. J. Health 2016, 18, 1041–1046. [Google Scholar] [CrossRef]
- Heinonen, K.; Eriksson, J.G.; Kajantie, E.; Pesonen, A.K.; Barker, D.J.; Osmond, C.; Räikkönen, K. Late-Preterm Birth and Lifetime Socioeconomic Attainments: The Helsinki Birth Cohort Study. Pediatrics 2013, 132, 647–655. [Google Scholar] [CrossRef] [PubMed]
- Chawanpaiboon, S.; Vogel, J.P.; Moller, A.-B.; Lumbiganon, P.; Petzold, M.; Hogan, D.; Landoulsi, S.; Jampathong, N.; Kongwattanakul, K.; Laopaiboon, M.; et al. Global, regional, and national estimates of levels of preterm birth in 2014, a systematic review and modelling analysis. Lancet Glob. Health 2018, 7, e37–e46. [Google Scholar] [CrossRef]
- Blencowe, H.; Cousens, S.; Oestergaard, M.Z.; Chou, D.; Moller, A.B.; Narwal, R.; Adler, A.; Garcia, C.V.; Rohde, S.; Say, L.; et al. National, regional, and worldwide estimates of preterm birth rates in the year 2010 with time trends since 1990 for selected countries: A systematic analysis and implications. Lancet 2012, 379, 2162–2172. [Google Scholar] [CrossRef]
- Van Zijl, M.D.; Koullali, B.; Mol, B.W.; Pajkrt, E.; Oudijk, M.A. Prevention of preterm delivery: Current challenges and future prospects. Int. J. Womens Health 2016, 8, 633–645. [Google Scholar] [CrossRef]
- Leader, J.; Bajwa, A.; Lanes, A.; Hua, X.; White, R.R.; Rybak, N.; Walker, M. The Effect of Very Advanced Maternal Age on Maternal and Neonatal Outcomes: A Systematic Review. J. Obstet. Gynaecol. Can. 2018, 40, 1208–1218. [Google Scholar] [CrossRef]
- Mayo, J.A.; Shachar, B.Z.; Stevenson, D.K.; Shaw, G.M. Nulliparous teenagers and preterm birth in California. J. Périnat. Med. 2017, 45, 959–967. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, F.; Senat, M.V. Multiple gestations and preterm birth. Semin. Fetal Neonatal Med. 2016, 21, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Berghella, V. Universal Cervical Length Screening for Prediction and Prevention of Preterm Birth. Obstet. Gynecol. Surv. 2012, 67, 653–657. [Google Scholar] [CrossRef] [PubMed]
- Kyrgiou, M.; Koliopoulos, G.; Martin-Hirsch, P.; Arbyn, M.; Prendiville, W.; Paraskevaidis, E. Obstetric outcomes after conservative treatment for intraepithelial or early invasive cervical lesions: Systematic review and meta-analysis. Lancet 2006, 367, 489–498. [Google Scholar] [CrossRef]
- Lemmers, M.; Verschoor, M.A.; Hooker, A.B.; Opmeer, B.C.; Limpens, J.; Huirne, J.A.; Ankum, W.M.; Mol, B.W. Dilatation and curettage increases the risk of subsequent preterm birth: A systematic review and meta-analysis. Hum. Reprod. (Oxf. Engl.) 2016, 31, 34–45. [Google Scholar] [CrossRef] [PubMed]
- Smid, M.C.; Lee, J.H.; Grant, J.H.; Miles, G.; Stoddard, G.J.; Chapman, D.A.; Manuck, T.A. Maternal race and intergenerational preterm birth recurrence. Am. J. Obstet. Gynecol. 2017, 217, 480.e1–480.e9. [Google Scholar] [CrossRef] [PubMed]
- Manuck, T.A. The genomics of prematurity in an era of more precise clinical phenotyping: A review. Semin. Fetal Neonatal Med. 2016, 21, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Strauss, J.F., 3rd; Romero, R.; Gomez-Lopez, N.; Haymond-Thornburg, H.; Modi, B.P.; Teves, M.E.; Pearson, L.N.; York, T.P.; Schenkein, H.A. Spontaneous Preterm Birth: Advances Toward the Discovery of Genetic Predisposition. Am. J. Obstet. Gynecol. 2017, 218, 294–314. [Google Scholar] [CrossRef] [PubMed]
- Manuck, T.A. Racial and ethnic differences in preterm birth: A complex, multifactorial problem. Semin. Perinatol. 2017, 41, 511–518. [Google Scholar] [CrossRef]
- Zhang, G.; Feenstra, B.; Bacelis, J.; Liu, X.; Muglia, L.M.; Juodakis, J.; Miller, D.E.; Litterman, N.; Jiang, P.P.; Russell, L.; et al. Genetic Associations with Gestational Duration and Spontaneous Preterm Birth. N. Engl. J. Med. 2017, 377, 1156–1167. [Google Scholar] [CrossRef]
- Jarde, A.; Morais, M.; Kingston, D.; Giallo, R.; MacQueen, G.M.; Giglia, L.; Beyene, J.; Wang, Y.; McDonald, S.D. Neonatal Outcomes in Women With Untreated Antenatal Depression Compared With Women Without Depression. JAMA Psychiatry 2016, 73, 826–837. [Google Scholar] [CrossRef] [PubMed]
- Rubens, C.E.; Sadovsky, Y.; Muglia, L.; Gravett, M.G.; Lackritz, E.; Gravett, C. Prevention of preterm birth: Harnessing science to address the global epidemic. Sci. Transl. Med. 2014, 6, 262sr5. [Google Scholar] [CrossRef] [PubMed]
- Frey, H.A.; Klebanoff, M.A. The epidemiology, etiology, and costs of preterm birth. Semin. Fetal Neonatal Med. 2016, 21, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Nijman, T.A.; van Vliet, E.O.; Benders, M.J.; Mol, B.W.; Franx, A.; Nikkels, P.G.; Oudijk, M.A. Placental histology in spontaneous and indicated preterm birth: A case control study. Placenta 2016, 48, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Norman, S.M.; Odibo, A.O.; Macones, G.A.; Dicke, J.M.; Crane, J.P.; Cahill, A.G. Ultrasound-detected subchorionic hemorrhage and the obstetric implications. Obstet. Gynecol. 2010, 116, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Nagy, S.; Bush, M.; Stone, J.; Lapinski, R.H.; Gardo, S. Clinical significance of subchorionic and retroplacental hematomas detected in the first trimester of pregnancy. Obstet. Gynecol. 2003, 102, 94–100. [Google Scholar] [PubMed]
- Vink, J.; Feltovich, H. Cervical etiology of spontaneous preterm birth. Semin. Fetal Neonatal Med. 2016, 21, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Staneva, A.; Bogossian, F.; Pritchard, M.; Wittkowski, A. The effects of maternal depression, anxiety, and perceived stress during pregnancy on preterm birth: A systematic review. Women Birth 2015, 28, 179–193. [Google Scholar] [CrossRef]
- Robinson, B.G.; Emanuel, R.L.; Frim, D.M.; Majzoub, J.A. Glucocorticoid stimulates expression of corticotropin-releasing hormone gene in human placenta. Proc. Natl. Acad. Sci. USA 1988, 85, 5244–5248. [Google Scholar] [CrossRef]
- Howland, M.A.; Sandman, C.A.; Glynn, L.M.; Crippen, C.; Davis, E.P. Fetal exposure to placental corticotropin-releasing hormone is associated with child self-reported internalizing symptoms. Psychoneuroendocrinology 2016, 67, 10–17. [Google Scholar] [CrossRef]
- Sun, K.; Ma, R.; Cui, X.; Campos, B.; Webster, R.; Brockman, D.; Myatt, L. Glucocorticoids Induce Cytosolic Phospholipase A2 and Prostaglandin H Synthase Type 2 But Not Microsomal Prostaglandin E Synthase (PGES) and Cytosolic PGES Expression in Cultured Primary Human Amnion Cells. J. Clin. Endocrinol. Metab. 2003, 88, 5564–5571. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.O.; Yang, Z.; Guo, C.M.; Ni, X.T.; Li, J.N.; Ge, Y.C.; Myatt, L.; Sun, K. Paradoxical stimulation of cyclooxygenase-2 expression by glucocorticoids via a cyclic AMP response element in human amnion fibroblasts. Mol. Endocrinol. 2009, 23, 1839–1849. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dunn, A.B.; Dunlop, A.L.; Hogue, C.J.; Miller, A.; Corwin, E.J. The Microbiome and Complement Activation: A Mechanistic Model for Preterm Birth. Boil. Res. Nurs. 2017, 19, 295–307. [Google Scholar] [CrossRef] [PubMed]
- Romero, R.; Dey, S.K.; Fisher, S.J. Preterm Labor: One Syndrome, Many Causes. Science 2014, 345, 760–765. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.J.; Romero, R.; Chaemsaithong, P.; Chaiyasit, N.; Yoon, B.H.; Kim, Y.M. Acute chorioamnionitis and funisitis: Definition, pathologic features, and clinical significance. Am. J. Obstet. Gynecol. 2015, 213, S29–S52. [Google Scholar] [CrossRef] [PubMed]
- Cappelletti, M.; Della Bella, S.; Ferrazzi, E.; Mavilio, D.; Divanovic, S. Inflammation and preterm birth. J. Leukoc. Biol. 2016, 99, 67–78. [Google Scholar] [CrossRef] [PubMed]
- Romero, R.; Chaiworapongsa, T.; Alpay Savasan, Z.; Xu, Y.; Hussein, Y.; Dong, Z.; Kusanovic, J.P.; Kim, C.J.; Hassan, S.S. Damage-associated molecular patterns (DAMPs) in preterm labor with intact membranes and preterm PROM: A study of the alarmin HMGB1. J. Matern. Fetal Neonatal Med. 2011, 24, 1444–1455. [Google Scholar] [CrossRef] [PubMed]
- Goldenberg, R.L.; Culhane, J.F.; Iams, J.D.; Romero, R. Epidemiology and causes of preterm birth. Lancet 2008, 371, 75–84. [Google Scholar] [CrossRef]
- Lannon, S.M.R.; Vanderhoeven, J.P.; Eschenbach, D.A.; Gravett, M.G.; Waldorf, K.M.A. Synergy and Interactions Among Biological Pathways Leading to Preterm Premature Rupture of Membranes. Reprod. Sci. 2014, 21, 1215–1227. [Google Scholar] [CrossRef]
- Norwitz, E.R.; Bonney, E.A.; Snegovskikh, V.V.; Williams, M.A.; Phillippe, M.; Park, J.S.; Abrahams, V.M. Molecular Regulation of Parturition: The Role of the Decidual Clock. Cold Spring Harb. Perspect. Med. 2015, 5, a023143. [Google Scholar] [CrossRef]
- Menon, R.; Bonney, E.A.; Condon, J.; Mesiano, S.; Taylor, R.N. Novel concepts on pregnancy clocks and alarms: Redundancy and synergy in human parturition. Hum. Reprod. Updat. 2016, 22, 535–560. [Google Scholar] [CrossRef] [PubMed]
- Navathe, R.; Berghella, V. Tocolysis for Acute Preterm Labor: Where Have We Been, Where Are We Now, and Where are We Going? Am. J. Perinatol. 2016, 33, 229–235. [Google Scholar] [PubMed]
- Son, M.; Miller, E.S. Predicting preterm birth: Cervical length and fetal fibronectin. Semin. Perinatol. 2017, 41, 445–451. [Google Scholar] [CrossRef] [PubMed]
- Koullali, B.; Oudijk, M.A.; Nijman, T.A.; Mol, B.W.; Pajkrt, E. Risk assessment and management to prevent preterm birth. Semin. Fetal Neonatal Med. 2016, 21, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Romero, R.; Conde-Agudelo, A.; El-Refaie, W.; Rode, L.; Brizot, M.L.; Cetingoz, E.; Serra, V.; Da Fonseca, E.; Abdelhafez, M.S.; Tabor, A.; et al. Vaginal progesterone decreases preterm birth and neonatal morbidity and mortality in women with a twin gestation and a short cervix: An updated meta-analysis of individual patient data. Ultrasound Obstet. Gynecol. 2017, 49, 303–314. [Google Scholar] [CrossRef]
- Romero, R.; Nicolaides, K.H.; Conde-Agudelo, A.; O’Brien, J.M.; Cetingoz, E.; Da Fonseca, E.; Creasy, G.W.; Hassan, S.S. Vaginal progesterone decreases preterm birth birth </=34 weeks of gestation in women with a singleton pregnancy and a short cervix: an updated meta-analysis including data from the OPPTIMUM study. Ultrasound Obstet. Gynecol. 2016, 48, 308–317. [Google Scholar] [CrossRef] [PubMed]
- Jarde, A.; Lutsiv, O.; Beyene, J.; McDonald, S.D. Vaginal progesterone, oral progesterone, 17-OHPC, cerclage and pessary for preventing preterm birth in singleton pregnancies: An updated systematic review and network meta-analysis. BJOG Int. J. Obstet. Gynaecol. 2019, 126, 556–567. [Google Scholar] [CrossRef]
- Jarde, A.; Lutsiv, O.; Park, C.K.; Beyene, J.; Dodd, J.M.; Barrett, J.; Shah, P.S.; Cook, J.L.; Saito, S.; Biringer, A.B.; et al. Effectiveness of progesterone, cerclage and pessary for preventing preterm birth in singleton pregnancies: A systematic review and network meta-analysis. BJOG Int. J. Obstet. Gynaecol. 2017, 124, 1176–1189. [Google Scholar] [CrossRef]
- Newnham, J.P.; Kemp, M.W.; White, S.W.; Arrese, C.A.; Hart, R.J.; Keelan, J.A. Applying Precision Public Health to Prevent Preterm Birth. Front. Public Health 2017, 5, 66. [Google Scholar] [CrossRef]
- Bloomfield, F.H. How is maternal nutrition related to preterm birth? Annu. Rev. Nutr. 2011, 31, 235–261. [Google Scholar] [CrossRef]
- Romero, R.; Espinoza, J.; Goncalves, L.F.; Kusanovic, J.P.; Friel, L.; Hassan, S. The role of inflammation and infection in preterm birth. Semin. Reprod. Med. 2007, 25, 21–39. [Google Scholar] [CrossRef] [PubMed]
- Englund-Ögge, L.; Brantsæter, A.L.; Haugen, M.; Sengpiel, V.; Khatibi, A.; Myhre, R.; Myking, S.; Meltzer, H.M.; Kacerovsky, M.; Nilsen, R.M.; et al. Association between intake of artificially sweetened and sugar-sweetened beverages and preterm delivery: A large prospective cohort study123. Am. J. Clin. Nutr. 2012, 96, 552–559. [Google Scholar] [CrossRef] [PubMed]
- Englund-Ögge, L.; Birgisdóttir, B.E.; Sengpiel, V.; Brantsæter, A.L.; Haugen, M.; Myhre, R.; Meltzer, H.M.; Jacobsson, B. Meal frequency patterns and glycemic properties of maternal diet in relation to preterm delivery: Results from a large prospective cohort study. PLoS ONE 2017, 12, e0172896. [Google Scholar] [CrossRef] [PubMed]
- Englund-Ögge, L.; Brantsæter, A.L.; Sengpiel, V.; Haugen, M.; Birgisdottir, B.E.; Myhre, R.; Meltzer, H.M.; Jacobsson, B. Maternal dietary patterns and preterm delivery: Results from large prospective cohort study. BMJ 2014, 348, g1446. [Google Scholar] [CrossRef] [PubMed]
- Mikkelsen, T.B.; Osterdal, M.L.; Knudsen, V.K.; Haugen, M.; Meltzer, H.M.; Bakketeig, L.; Olsen, S.F. Association between a Mediterranean-type diet and risk of preterm birth among Danish women: A prospective cohort study. Acta Obstet. Gynecol. Scand. 2008, 87, 325–330. [Google Scholar] [CrossRef] [PubMed]
- Saunders, L.; Guldner, L.; Costet, N.; Kadhel, P.; Rouget, F.; Monfort, C.; Thomé, J.-P.; Multigner, L.; Cordier, S. Effect of a Mediterranean Diet during Pregnancy on Fetal Growth and Preterm Delivery: Results From a French Caribbean Mother-Child Cohort Study (TIMOUN). Paediatr. Périnat. Epidemiol. 2014, 28, 235–244. [Google Scholar] [CrossRef] [PubMed]
- Khoury, J.; Henriksen, T.; Christophersen, B.; Tonstad, S. Effect of a cholesterol-lowering diet on maternal, cord, and neonatal lipids, and pregnancy outcome: A randomized clinical trial. Am. J. Obstet. Gynecol. 2005, 193, 1292–1301. [Google Scholar] [CrossRef] [PubMed]
- Haugen, M.; Meltzer, H.M.; Brantsaeter, A.L.; Mikkelsen, T.; Osterdal, M.L.; Alexander, J.; Olsen, S.F.; Bakketeig, L. Mediterranean-type diet and risk of preterm birth among women in the Norwegian Mother and Child Cohort Study (MoBa): A prospective cohort study. Acta Obstet. Gynecol. Scand. 2008, 87, 319–324. [Google Scholar] [CrossRef]
- Position of the American Dietetic Association and Dietitians of Canada: Vegetarian diets. J. Am. Diet. Assoc. 2003, 103, 748–765. [CrossRef]
- Burdge, G.C.; Tan, S.Y.; Henry, C.J. Long-chain n-3 PUFA in vegetarian women: A metabolic perspective. J. Nutr. Sci. 2017, 6, e58. [Google Scholar] [CrossRef]
- Rogne, T.; Tielemans, M.J.; Chong, M.F.; Yajnik, C.S.; Krishnaveni, G.V.; Poston, L.; Jaddoe, V.W.; Steegers, E.A.; Joshi, S.; Chong, Y.S.; et al. Associations of Maternal Vitamin B12 Concentration in Pregnancy With the Risks of Preterm Birth and Low Birth Weight: A Systematic Review and Meta-Analysis of Individual Participant Data. Am. J. Epidemiol. 2017, 185, 212–223. [Google Scholar] [CrossRef] [PubMed]
- Raghavan, R.; Dreibelbis, C.; Kingshipp, B.L.; Wong, Y.P.; Abrams, B.; Gernand, A.D.; Rasmussen, K.M.; Siega-Riz, A.M.; Stang, J.O.; Casavale, K.; et al. Dietary patterns before and during pregnancy and birth outcomes: A systematic review. Am. J. Clin. Nutr. 2019, 109, 729S–756S. [Google Scholar] [CrossRef] [PubMed]
- Chia, A.R.; Chen, L.W.; Lai, J.S.; Wong, C.H.; Neelakantan, N.; Van Dam, R.M.; Chong, M.F.F. Maternal Dietary Patterns and Birth Outcomes: A Systematic Review and Meta-Analysis. Adv. Nutr. 2019, 10, 685–695. [Google Scholar] [CrossRef] [PubMed]
- Kibret, K.T.; Chojenta, C.; Gresham, E.; Tegegne, T.K.; Loxton, D. Maternal dietary patterns and risk of adverse pregnancy (hypertensive disorders of pregnancy and gestational diabetes mellitus) and birth (preterm birth and low birth weight) outcomes: A systematic review and meta-analysis. Public Health Nutr. 2018, 22, 506–520. [Google Scholar] [CrossRef] [PubMed]
- Burdge, G.C.; Calder, P.C. Conversion of alpha-linolenic acid to longer-chain polyunsaturated fatty acids in human adults. Reprod. Nutr. Dev. 2005, 45, 581–597. [Google Scholar] [CrossRef] [PubMed]
- Burdge, G.C.; Jones, A.E.; Wootton, S.A. Eicosapentaenoic and docosapentaenoic acids are the principal products of α-linolenic acid metabolism in young men. Br. J. Nutr. 2002, 88, 355–363. [Google Scholar] [CrossRef] [PubMed]
- Baker, E.J.; Miles, E.A.; Burdge, G.C.; Yaqoob, P.; Calder, P.C. Metabolism and functional effects of plant-derived omega-3 fatty acids in humans. Prog. Lipid Res. 2016, 64, 30–56. [Google Scholar] [CrossRef] [PubMed]
- Papanikolaou, Y.; Brooks, J.; Reider, C.; Fulgoni, V.L., 3rd. U.S. adults are not meeting recommended levels for fish and omega-3 fatty acid intake: Results of an analysis using observational data from NHANES 2003–2008. Nutr. J. 2014, 13, 31. [Google Scholar] [CrossRef]
- Kuriki, K.; Nagaya, T.; Imaeda, N.; Tokudome, Y.; Fujiwara, N.; Sato, J.; Ikeda, M.; Maki, S.; Tokudome, S. Discrepancies in dietary intakes and plasma concentrations of fatty acids according to age among Japanese female dietitians. Eur. J. Clin. Nutr. 2002, 56, 524–531. [Google Scholar] [CrossRef]
- Olsen, S.F.; Hansen, H.S.; Sorensen, T.I.; Jensen, B.; Secher, N.J.; Sommer, S.; Knudsen, L.B. Intake of marine fat, rich in (n-3)-polyunsaturated fatty acids, may increase birthweight by prolonging gestation. Lancet (Lond. Engl.) 1986, 2, 367–369. [Google Scholar] [CrossRef]
- Olsen, S.F.; Østerdal, M.L.; Salvig, J.D.; Kesmodel, U.; Henriksen, T.B.; Hedegaard, M.; Secher, N.J. Duration of pregnancy in relation to seafood intake during early and mid pregnancy: Prospective cohort. Eur. J. Epidemiol. 2006, 21, 749–758. [Google Scholar] [CrossRef] [PubMed]
- Brantsæter, A.L.; Englund-Ögge, L.; Haugen, M.; Birgisdottir, B.E.; Knutsen, H.K.; Sengpiel, V.; Myhre, R.; Alexander, J.; Nilsen, R.M.; Jacobsson, B.; et al. Maternal intake of seafood and supplementary long chain n-3 poly-unsaturated fatty acids and preterm delivery. BMC Pregnancy Childbirth 2017, 17, 61. [Google Scholar]
- Olsen, S.F.; Secher, N.J. Low Consumption of Seafood in Early Pregnancy as a Risk Factor for Preterm Delivery: Prospective Cohort Study. Obstet. Gynecol. Surv. 2002, 57, 651–652. [Google Scholar] [CrossRef][Green Version]
- Oken, E.; Kleinman, K.P.; Olsen, S.F.; Rich-Edwards, J.W.; Gillman, M.W. Associations of Seafood and Elongated n-3 Fatty Acid Intake with Fetal Growth and Length of Gestation: Results from a US Pregnancy Cohort. Am. J. Epidemiol. 2004, 160, 774–783. [Google Scholar] [CrossRef] [PubMed]
- Rogers, I.; Emmett, P.; Ness, A.; Golding, J. Maternal fish intake in late pregnancy and the frequency of low birth weight and intrauterine growth retardation in a cohort of British infants. J. Epidemiol. Community Health 2004, 58, 486–492. [Google Scholar] [CrossRef] [PubMed]
- Carlson, S.E.; Colombo, J.; Gajewski, B.J.; Gustafson, K.M.; Mundy, D.; Yeast, J.; Georgieff, M.K.; Markley, L.A.; Kerling, E.H.; Shaddy, D.J. DHA supplementation and pregnancy outcomes. Am. J. Clin. Nutr. 2013, 97, 808–815. [Google Scholar] [CrossRef] [PubMed]
- Makrides, M.; Gibson, R.A.; McPhee, A.J.; Yelland, L.; Quinlivan, J.; Ryan, P. Effect of DHA supplementation during pregnancy on maternal depression and neurodevelopment of young children: A randomized controlled trial. JAMA 2010, 304, 1675–1683. [Google Scholar] [CrossRef] [PubMed]
- Ramakrishnan, U.; Stein, A.D.; Parra-Cabrera, S.; Wang, M.; Imhoff-Kunsch, B.; Juarez-Marquez, S.; Rivera, J.; Martorell, R. Effects of docosahexaenoic acid supplementation during pregnancy on gestational age and size at birth: Randomized, double-blind, placebo-controlled trial in Mexico. Food Nutr. Bull. 2010, 31, S108–S116. [Google Scholar] [CrossRef]
- Helland, I.B.; Saugstad, O.D.; Smith, L.; Saarem, K.; Solvoll, K.; Ganes, T.; Drevon, C.A. Similar Effects on Infants of n-3 and n-6 Fatty Acids Supplementation to Pregnant and Lactating Women. Pediatrics 2001, 108, e82. [Google Scholar] [CrossRef]
- Olsen, S.F.; Secher, N.J.; Tabor, A.; Weber, T.; Walker, J.J.; Gluud, C. Randomised clinical trials of fish oil supplementation in high risk pregnancies. Fish Oil Trials in Pregnancy (FOTIP) Team. BJOG Int. J. Obstet. Gynaecol. 2000, 107, 382–395. [Google Scholar] [CrossRef]
- Onwude, J.L.; Lilford, R.J.; Hjartardottir, H.; Staines, A.; Tuffnell, D. A randomised double blind placebo controlled trial of fish oil in high risk pregnancy. BJOG: Int. J. Obstet. Gynaecol. 1995, 102, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Huisjes, H.J.; Visser, G.H.A.; Bulstra-Ramakers, M.T.E.W.; Bulstra-Ramakers, M.T.E.W. The effects of 3g eicosapentaenoic acid daily on recurrence of intrauterine growth retardation and pregnancy induced hypertension. BJOG: Int. J. Obstet. Gynaecol. 1995, 102, 123–126. [Google Scholar]
- Olsen, S.; Sorensen, J.; Secher, N.; Hedegaard, M.; Henriksen, T.; Hansen, H.; Grant, A. Randomised controlled trial of effect of fish-oil supplementation on pregnancy duration. Int. J. Gynecol. Obstet. 1992, 39, 365–366. [Google Scholar] [CrossRef]
- Mardones, F.; Urrutia, M.T.; Villarroel, L.; Rioseco, A.; Castillo, O.; Rozowski, J.; Tapia, J.L.; Bastias, G.; Bacallao, J.; Rojas, I. Effects of a dairy product fortified with multiple micronutrients and omega-3 fatty acids on birth weight and gestation duration in pregnant Chilean women. Public Health Nutr. 2008, 11, 30–40. [Google Scholar] [CrossRef] [PubMed]
- Smuts, C.M.; Huang, M.; Mundy, D.; Plasse, T.; Major, S.; Carlson, S.E. A randomized trial of docosahexaenoic acid supplementation during the third trimester of pregnancy. Obstet. Gynecol. 2003, 101, 469–479. [Google Scholar] [PubMed]
- Glaser, C.; Heinrich, J.; Koletzko, B. Role of FADS1 and FADS2 polymorphisms in polyunsaturated fatty acid metabolism. Metabolism 2010, 59, 993–999. [Google Scholar] [CrossRef] [PubMed]
- Moltó-Puigmartí, C.; Van Dongen, M.C.J.M.; Dagnelie, P.C.; Plat, J.; Mensink, R.P.; Tan, F.E.S.; Heinrich, J.; Thijs, C. Maternal but Not Fetal FADS Gene Variants Modify the Association between Maternal Long-Chain PUFA Intake in Pregnancy and Birth Weight. J. Nutr. 2014, 144, 1430–1437. [Google Scholar] [CrossRef] [PubMed]
- Carlson, S.E.; Gajewski, B.J.; Valentine, C.J.; Rogers, L.K.; Weiner, C.P.; DeFranco, E.A.; Buhimschi, C.S. Assessment of DHA on reducing early preterm birth: The ADORE randomized controlled trial protocol. BMC Pregnancy Childbirth 2017, 17, 62. [Google Scholar] [CrossRef]
- Zhou, S.J.; Best, K.; Gibson, R.; McPhee, A.; Yelland, L.; Quinlivan, J.; Makrides, M. Study protocol for a randomised controlled trial evaluating the effect of prenatal omega-3 LCPUFA supplementation to reduce the incidence of preterm birth: The ORIP trial. BMJ Open 2017, 7, e018360. [Google Scholar] [CrossRef]
- Briggs, V.; Goldenberg, T.; Ramakrishnan, U.; Imhoff-Kunsch, B.; Imhoff-Kunsch, B. Effect of n-3 Long-chain Polyunsaturated Fatty Acid Intake during Pregnancy on Maternal, Infant, and Child Health Outcomes: A Systematic Review. Paediatr. Périnat. Epidemiol. 2012, 26, 91–107. [Google Scholar]
- Szajewska, H.; Horvath, A.; Koletzko, B. Effect of n-3 long-chain polyunsaturated fatty acid supplementation of women with low-risk pregnancies on pregnancy outcomes and growth measures at birth: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2006, 83, 1337–1344. [Google Scholar] [CrossRef] [PubMed]
- Makrides, M.; Duley, L.; Olsen, S.F. Marine oil, and other prostaglandin precursor, supplementation for pregnancy uncomplicated by pre-eclampsia or intrauterine growth restriction. Cochrane Database Syst. Rev. 2006, 3, CD003402. [Google Scholar] [CrossRef] [PubMed]
- Horvath, A.; Koletzko, B.; Szajewska, H. Effect of supplementation of women in high-risk pregnancies with long-chain polyunsaturated fatty acids on pregnancy outcomes and growth measures at birth: A meta-analysis of randomized controlled trials. Br. J. Nutr. 2007, 98, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Kar, S.; Wong, M.; Rogozinska, E.; Thangaratinam, S. Effects of omega-3 fatty acids in prevention of early preterm delivery: A systematic review and meta-analysis of randomized studies. Eur. J. Obstet. Gynecol. Reprod. Biol. 2016, 198, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Middleton, P.; Gomersall, J.C.; Gould, J.F.; Shepherd, E.; Olsen, S.F.; Makrides, M. Omega-3 fatty acid addition during pregnancy. Cochrane Database Syst. Rev. 2018, 11, CD003402. [Google Scholar] [CrossRef] [PubMed]
- Park, H.G.; Lawrence, P.; Engel, M.G.; Kothapalli, K.; Brenna, J.T. Metabolic fate of docosahexaenoic acid (DHA; 22:6n-3) in human cells: Direct retroconversion of DHA to eicosapentaenoic acid (20:5n-3) dominates over elongation to tetracosahexaenoic acid (24:6n-3). FEBS Lett. 2016, 590, 3188–3194. [Google Scholar] [CrossRef] [PubMed]
- Olsen, S.F.; Secher, N.J.; Bjornsson, S.; Weber, T.; Atke, A. The potential benefits of using fish oil in relation to preterm labor: The case for a randomized controlled trial? Acta Obstet. Gynecol. Scand. 2003, 82, 978–982. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. The role of marine omega-3 (n-3) fatty acids in inflammatory processes, atherosclerosis and plaque stability. Mol. Nutr. Food Res. 2012, 56, 1073–1080. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, R.S. The role of zinc in growth and cell proliferation. J. Nutr. 2000, 130, 1500s–1508s. [Google Scholar] [CrossRef]
- Black, R.E.; Allen, L.H.A.; Bhutta, Z.; Caulfield, L.E.; De Onis, M.; Ezzati, M.; Mathers, C.; Rivera, J. Maternal and child undernutrition: Global and regional exposures and health consequences. Lancet 2008, 371, 243–260. [Google Scholar] [CrossRef]
- Keen, C.L.; Uriu-Adams, J.Y.; Uriu-Adams, J.Y. Zinc and reproduction: Effects of zinc deficiency on prenatal and early postnatal development. Birth Defects Res. Part B Dev. Reprod. Toxicol. 2010, 89, 313–325. [Google Scholar]
- Chaffee, B.W.; King, J.C. Effect of Zinc Supplementation on Pregnancy and Infant Outcomes: A Systematic Review. Paediatr. Périnat. Epidemiol. 2012, 26, 118–137. [Google Scholar] [CrossRef] [PubMed]
- Ota, E.; Mori, R.; Middleton, P.; Tobe-Gai, R.; Mahomed, K.; Miyazaki, C.A.; Bhutta, Z. Zinc supplementation for improving pregnancy and infant outcome. Cochrane Database Syst. Rev. 2015, 2, CD000230. [Google Scholar] [CrossRef] [PubMed]
- Wilson, R.L.; Grieger, J.A.; Bianco-Miotto, T.; Roberts, C.T. Association between Maternal Zinc Status, Dietary Zinc Intake and Pregnancy Complications: A Systematic Review. Nutrients 2016, 8, 641. [Google Scholar] [CrossRef] [PubMed]
- DeLuca, H.F. Overview of general physiologic features and functions of vitamin D. Am. J. Clin. Nutr. 2004, 80, 1689s–1696s. [Google Scholar] [CrossRef] [PubMed]
- Mithal, A.; Wahl, D.A.; Bonjour, J.P.; Burckhardt, P.; Dawson-Hughes, B.; Eisman, J.A.; El-Hajj Fuleihan, G.; Josse, R.G.; Lips, P.; Morales-Torres, J. Global vitamin D status and determinants of hypovitaminosis D. Osteoporos. Int. 2009, 20, 1807–1820. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.Q.; Hewison, M. Vitamin D, the placenta and pregnancy. Arch. Biochem. Biophys. 2012, 523, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Liu, N.Q.; Kaplan, A.T.; Lagishetty, V.; Ouyang, Y.B.; Ouyang, Y.; Simmons, C.F.; Equils, O.; Hewison, M. Vitamin D and the Regulation of Placental Inflammation. J. Immunol. 2011, 186, 5968–5974. [Google Scholar] [CrossRef]
- Zhou, S.; Tao, Y.; Huang, K.; Zhu, B. Vitamin D and risk of preterm birth: Up-to-date meta-analysis of randomized controlled trials and observational studies. J. Obstet. Gynaecol. Res. 2017, 43, 247–256. [Google Scholar] [CrossRef] [PubMed]
- Qin, L.L.; Lu, F.G.; Yang, S.H.; Xu, H.L.; Luo, B.A. Does Maternal Vitamin D Deficiency Increase the Risk of Preterm Birth: A Meta-Analysis of Observational Studies. Nutrients 2016, 8, 301. [Google Scholar] [CrossRef] [PubMed]
- Thorne-Lyman, A.; Fawzi, W.W. Vitamin D during pregnancy and maternal, neonatal and infant health outcomes: A systematic review and meta-analysis. Paediatr. Périnat. Epidemiol. 2012, 26, 75–90. [Google Scholar] [CrossRef] [PubMed]
- De-Regil, L.M.; Palacios, C.; Lombardo, L.K.; Peña-Rosas, J.P. Vitamin D supplementation for women during pregnancy. Cochrane Database Syst. Rev. 2016, 1, CD008873. [Google Scholar]
- Cooper, C.; Harvey, N.C.; Bishop, N.J.; Kennedy, S.; Papageorghiou, A.T.; Schoenmakers, I.; Fraser, R.; Gandhi, S.V.; Carr, A.; D’Angelo, S.; et al. Maternal gestational vitamin D supplementation and offspring bone health (MAVIDOS): A multicentre, double-blind, randomised placebo-controlled trial. Lancet Diabetes Endocrinol. 2016, 4, 393–402. [Google Scholar] [CrossRef]
- Arikan, G.M.; Panzitt, T.; Gücer, F.; Scholz, H.S.; Reinisch, S.; Haas, J.; Weiss, P.A. Course of Maternal Serum Magnesium Levels in Low-Risk Gestations and in Preterm Labor and Delivery. Fetal Diagn. Ther. 1999, 14, 332–336. [Google Scholar] [CrossRef] [PubMed]
- King, D.E.; Mainous, A.G., 3rd; Geesey, M.E.; Woolson, R.F. Dietary magnesium and C-reactive protein levels. J. Am. Coll. Nutr. 2005, 24, 166–171. [Google Scholar] [CrossRef] [PubMed]
- Wynn, A.; Wynn, M. Magnesium and Other Nutrient Deficiencies as Possible Causes of Hypertension and Low Birthweight. Nutr. Health 1988, 6, 69–88. [Google Scholar] [CrossRef] [PubMed]
- Lopez Bernal, A. The regulation of uterine relaxation. Semin. Cell Dev. Biol. 2007, 18, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Makrides, M.A.; Crowther, C. Magnesium supplementation in pregnancy. Cochrane Database Syst. Rev. 2001, CD000937. [Google Scholar] [CrossRef]
- Crosby, D.D.; Shepherd, E.A.; Crowther, C.; Makrides, M. Magnesium supplementation in pregnancy. Cochrane Database Syst. Rev. 2014, 4, CD000937. [Google Scholar]
- Crowther, C.A.; Brown, J.; McKinlay, C.J.D.; Middleton, P. Magnesium sulphate for preventing preterm birth in threatened preterm labour. Cochrane Database Syst. Rev. 2014, 8, CD001060. [Google Scholar] [CrossRef]
- Alves, J.G.B.; De Araújo, C.A.F.L.; Pontes, I.E.; Guimarães, A.C.; Ray, J.G. The BRAzil MAGnesium (BRAMAG) trial: A randomized clinical trial of oral magnesium supplementation in pregnancy for the prevention of preterm birth and perinatal and maternal morbidity. BMC Pregnancy Childbirth 2014, 14, 222. [Google Scholar] [CrossRef] [PubMed]
- Buppasiri, P.; Lumbiganon, P.; Thinkhamrop, J.; Ngamjarus, C.; Laopaiboon, M.; Medley, N. Calcium supplementation (other than for preventing or treating hypertension) for improving pregnancy and infant outcomes. Cochrane Database Syst. Rev. 2015, CD007079. [Google Scholar] [CrossRef] [PubMed]
- Villar, J.; Merialdi, M.; Gulmezoglu, A.M.; Abalos, E.; Carroli, G.; Kulier, R.; de Onis, M. Nutritional interventions during pregnancy for the prevention or treatment of maternal morbidity and preterm delivery: An overview of randomized controlled trials. J. Nutr. 2003, 133, 1606s–1625s. [Google Scholar] [CrossRef] [PubMed]
- Peña-Rosas, J.P.; Dowswell, T.; Peña-Rosas, J.P.; De-Regil, L.M.; Garcia-Casal, M.N.; Pena-Rosas, J.P.; De-Regil, L.M.; Garcia-Casal, M.N. Daily oral iron supplementation during pregnancy. Cochrane Database Syst. Rev. 2015, CD004736. [Google Scholar] [CrossRef] [PubMed]
- De-Regil, L.M.; Peña-Rosas, J.P.; Fernández-Gaxiola, A.C.; Rayco-Solon, P. Effects and safety of periconceptional oral folate supplementation for preventing birth defects. Cochrane Database Syst. Rev. 2015, CD007950. [Google Scholar] [CrossRef] [PubMed]
- Klebanoff, M.; Shiono, P.; Selby, J.; Trachtenberg, A.; Graubard, B. Anemia and spontaneous preterm birth. Int. J. Gynecol. Obstet. 1991, 164, 59–63. [Google Scholar]
- Scholl, T.O.; Hediger, M.L.; Fischer, R.L.; Shearer, J.W. Anemia vs. iron deficiency: Increased risk of preterm delivery in a prospective study. Am. J. Clin. Nutr. 1992, 55, 985–988. [Google Scholar] [CrossRef]
- Lassi, Z.S.; Salam, R.A.; Haider, B.A.; Bhutta, Z. Folic acid supplementation during pregnancy for maternal health and pregnancy outcomes. Cochrane Database Syst. Rev. 2013, 3, CD006896. [Google Scholar] [CrossRef]
- West, K.P., Jr. Extent of vitamin A deficiency among preschool children and women of reproductive age. J. Nutr. 2002, 132, 2857s–2866s. [Google Scholar] [CrossRef]
- Thorne-Lyman, A.L.; Fawzi, W.W. Vitamin A and carotenoids during pregnancy and maternal, neonatal and infant health outcomes: A systematic review and meta-analysis. Paediatr. Périnat. Epidemiol. 2012, 26, 36–54. [Google Scholar] [CrossRef]
- West, K.P., Jr.; Katz, J.; Khatry, S.K.; LeClerq, S.C.; Pradhan, E.K.; Shrestha, S.R.; Connor, P.B.; Dali, S.M.; Christian, P.; Pokhrel, R.P.; et al. Double blind, cluster randomised trial of low dose supplementation with vitamin A or beta carotene on mortality related to pregnancy in Nepal. The NNIPS-2 Study Group. BMJ 1999, 318, 570–575. [Google Scholar] [CrossRef]
- Mistry, H.D.; Williams, P.J. The importance of antioxidant micronutrients in pregnancy. Oxid. Med. Cell. Longev. 2011, 2011, 841749. [Google Scholar] [CrossRef] [PubMed]
- Ray, J.G.; Laskin, C.A. Folic acid and homocyst(e)ine metabolic defects and the risk of placental abruption, pre-eclampsia and spontaneous pregnancy loss: A systematic review. Placenta 1999, 20, 519–529. [Google Scholar] [CrossRef] [PubMed]
- Himes, K.P.; Simhan, H.N. Risk of recurrent preterm birth and placental pathology. Obstet. Gynecol. 2008, 112, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Catov, J.M.; Bodnar, L.M.; Olsen, J.; Olsen, S.A.; Nohr, E. Periconceptional multivitamin use and risk of preterm or small-for-gestational-age births in the Danish National Birth Cohort1234. Am. J. Clin. Nutr. 2011, 94, 906–912. [Google Scholar] [CrossRef] [PubMed]
- Haider, B.A.; Bhutta, Z.A. Multiple-micronutrient supplementation for women during pregnancy. Cochrane Database Syst. Rev. 2015, 11, CD004905. [Google Scholar]
- de Onis, M.; Villar, J.; Gulmezoglu, M. Nutritional interventions to prevent intrauterine growth retardation: Evidence from randomized controlled trials. Eur. J. Clin. Nutr. 1998, 52, S83–S93. [Google Scholar]
- Kramer, M.S.; Kakuma, R. Energy and protein intake in pregnancy. Cochrane Database Syst. Rev. 2003, 4, CD000032. [Google Scholar]
- Hillier, S.L.; Martin, D.H.; Pastorek, J.G.; Rao, A.V.; McNellis, D.; Regan, J.A.; Nugent, R.P.; Eschenbach, D.A.; Krohn, M.A.; Gibbs, R.S.; et al. Association between Bacterial Vaginosis and Preterm Delivery of a Low-Birth-Weight Infant. N. Engl. J. Med. 1995, 333, 1737–1742. [Google Scholar] [CrossRef]
- Brocklehurst, P.; Gordon, A.; Heatley, E.; Milan, S.J. Antibiotics for treating bacterial vaginosis in pregnancy. Cochrane Database Syst. Rev. 2013. [Google Scholar] [CrossRef]
- Al-Ghazzewi, F.H.; Tester, R.F. Biotherapeutic agents and vaginal health. J. Appl. Microbiol. 2016, 121, 18–27. [Google Scholar] [CrossRef] [PubMed]
- Hemarajata, P.; Versalovic, J. Effects of probiotics on gut microbiota: Mechanisms of intestinal immunomodulation and neuromodulation. Ther. Adv. Gastroenterol. 2013, 6, 39–51. [Google Scholar] [CrossRef] [PubMed]
- Krauss-Silva, L.; Moreira, M.E.L.; Alves, M.B.; Rezende, M.R.; Braga, A.; Camacho, K.G.; Batista, M.R.R.; Savastano, C.; Almada-Horta, A.; Guerra, F. Randomized controlled trial of probiotics for the prevention of spontaneous preterm delivery associated with intrauterine infection: Study protocol. Reprod. Health 2010, 7, 14. [Google Scholar] [CrossRef] [PubMed]
- Krauss-Silva, L.; Moreira, M.E.L.; Alves, M.B.; Braga, A.; Camacho, K.G.; Batista, M.R.R.; Almada-Horta, A.; Rebello, M.R.; Guerra, F. A randomised controlled trial of probiotics for the prevention of spontaneous preterm delivery associated with bacterial vaginosis: Preliminary results. Trials 2011, 12, 239. [Google Scholar] [CrossRef] [PubMed]
- Gille, C.; Böer, B.; Marschal, M.; Urschitz, M.S.; Heinecke, V.; Hund, V.; Speidel, S.; Tarnow, I.; Mylonas, I.; Franz, A.; et al. Effect of probiotics on vaginal health in pregnancy. EFFPRO, a randomized controlled trial. Am. J. Obstet. Gynecol. 2016, 215, 608.e1–608.e7. [Google Scholar] [CrossRef] [PubMed]
- Luoto, R.; Laitinen, K.; Nermes, M.; Isolauri, E. Impact of maternal probiotic-supplemented dietary counselling on pregnancy outcome and prenatal and postnatal growth: A double-blind, placebo-controlled study. Br. J. Nutr. 2010, 103, 1792–1799. [Google Scholar] [CrossRef]
- Mantaring, J.; Benyacoub, J.; Destura, R.; Pecquet, S.; Vidal, K.; Volger, S.; Guinto, V. Effect of maternal supplement beverage with and without probiotics during pregnancy and lactation on maternal and infant health: A randomized controlled trial in the Philippines. BMC Pregnancy Childbirth 2018, 18, 193. [Google Scholar] [CrossRef]
- Rautava, S.; Collado, M.C.; Salminen, S.; Isolauri, E. Probiotics modulate host-microbe interaction in the placenta and fetal gut: A randomized, double-blind, placebo-controlled trial. Neonatology 2012, 102, 178–184. [Google Scholar] [CrossRef]
- Nordqvist, M.; Jacobsson, B.; Brantsæter, A.-L.; Myhre, R.; Nilsson, S.; Sengpiel, V. Timing of probiotic milk consumption during pregnancy and effects on the incidence of preeclampsia and preterm delivery: A prospective observational cohort study in Norway. BMJ Open 2018, 8, e018021. [Google Scholar] [CrossRef]
- Myhre, R.; Brantsaeter, A.L.; Myking, S.; Gjessing, H.K.; Sengpiel, V.; Meltzer, H.M.; Haugen, M.; Jacobsson, B. Intake of probiotic food and risk of spontaneous preterm delivery. Am. J. Clin. Nutr. 2011, 93, 151–157. [Google Scholar] [CrossRef]
- Othman, M.; Neilson, J.P.; Alfirevic, Z. Probiotics for preventing preterm labour. Cochrane Database Syst. Rev. 2007, 1, CD005941. [Google Scholar] [CrossRef] [PubMed]
- Jarde, A.; Lewis-Mikhael, A.-M.; Moayyedi, P.; Stearns, J.C.; Collins, S.M.; Beyene, J.; McDonald, S.D. Pregnancy outcomes in women taking probiotics or prebiotics: A systematic review and meta-analysis. BMC Pregnancy Childbirth 2018, 18, 14. [Google Scholar] [CrossRef] [PubMed]
- Grev, J.; Berg, M.; Soll, R. Maternal probiotic supplementation for prevention of morbidity and mortality in preterm infants. Cochrane Database Syst. Rev. 2018, 12, CD012519. [Google Scholar] [CrossRef] [PubMed]
- Stojanovic, N.; Plecas, D.; Plesinac, S. Normal vaginal flora, disorders and application of probiotics in pregnancy. Arch. Gynecol. Obstet. 2012, 286, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Jespers, V.; van de Wijgert, J.; Cools, P.; Verhelst, R.; Verstraelen, H.; Delany-Moretlwe, S.; Mwaura, M.; Ndayisaba, G.F.; Mandaliya, K.; Menten, J.; et al. The significance of Lactobacillus crispatus and L. vaginalis for vaginal health and the negative effect of recent sex: A cross-sectional descriptive study across groups of African women. BMC Infect. Dis. 2015, 15, 115. [Google Scholar] [CrossRef]
- Srinivasan, S.; Morgan, M.T.; Fiedler, T.L.; Djukovic, D.; Hoffman, N.G.; Raftery, D.; Marrazzo, J.M.; Fredricks, D.N. Metabolic signatures of bacterial vaginosis. MBio 2015, 6, e00204–e00215. [Google Scholar] [CrossRef]
- Kim, J.Y.; Kwon, J.H.; Ahn, S.H.; Lee, S.I.; Han, Y.S.; Choi, Y.O.; Lee, S.Y.; Ahn, K.M.; Ji, G.E. Effect of probiotic mix (Bifidobacterium bifidum, Bifidobacterium lactis, Lactobacillus acidophilus) in the primary prevention of eczema: A double-blind, randomized, placebo-controlled trial. Pediatr. Allergy Immunol. 2010, 21, e386–e393. [Google Scholar] [CrossRef]
- Ou, C.Y.; Kuo, H.C.; Wang, L.; Hsu, T.Y.; Chuang, H.; Liu, C.A.; Chang, J.C.; Yu, H.R.; Yang, K.D. Prenatal and postnatal probiotics reduces maternal but not childhood allergic diseases: A randomized, double-blind, placebo-controlled trial. Clin. Exp. Allergy 2012, 42, 1386–1396. [Google Scholar] [CrossRef]
- Vitali, B.; Cruciani, F.; Baldassarre, M.E.; Capursi, T.; Spisni, E.; Valerii, M.C.; Candela, M.; Turroni, S.; Brigidi, P. Dietary supplementation with probiotics during late pregnancy: Outcome on vaginal microbiota and cytokine secretion. BMC Microbiol. 2012, 12, 236. [Google Scholar] [CrossRef]
| Ref. | Study | Ingredient, Daily Dosage | Main Results | Comments | |||
|---|---|---|---|---|---|---|---|
| Objective | Design | Population/Sample Size | Duration | ||||
| Carlson et al. 2013 [76] | To assess if DHA supplementation can increase maternal and newborn DHA status, gestation duration, birth weight, and length | RCT, DB, PC. | Healthy pregnant women between 8 and 20 weeks of gestation from the USA, n = 350 | <20 weeks of gestation until delivery | Intervention: 3 capsules/day of a marine algae-oil source of DHA (600 mg DHA/day) Placebo: 3 capsules containing half soybean and half corn oil | Compared to placebo, DHA supplementation resulted in: (1) Longer gestation duration (2.9 day; p = 0.041). (2) Fewer infants born <34 weeks of gestation (p = 0.025). EPTB reduced by 87.5%. (3) Shorter hospital stays for PT infants (40.8 compared with 8.9 day; p = 0.026). (4) Similar PTB incidence between groups, with more EPTB in the placebo group (4.8% vs. 0.6%, p = 0.025). (5) Greater birth weight (172 g; p = 0.004), length (0.7 cm; p = 0.022), and head circumference (0.5 cm; p = 0.012). (6) Higher maternal and cord Red Blood Cell-phospholipid-DHA (2.6%; p < 0.001). | Women taking supplements <300 mg DHA/day were not excluded. Dietary n−3 LC-PUFA intakes were not assessed Many secondary variables were studied but without adjustment for multiple comparisons. Incidence of PTB and EPTB were secondary outcomes. |
| Makrides et al. 2010 [77] | To assess if DHA supplementation during the last half of pregnancy has a beneficial effect on maternal depressive symptoms and child neurodevelopment | RCT, DB, PC. | Healthy pregnant women <21 weeks gestation from Australia n = 2399 | <21 weeks of gestation until delivery | Intervention: 3 capsules/day of DHA-rich fish oil concentrate (800 mg DHA/day A + 100 mg EPA/day) Placebo: 3 capsules/day of vegetable oil containing a blend of rapeseed, Sunflower, and palm oil | Compared to placebo, DHA+EPA supplementation resulted in (1) No differences in the rate of women with depressive symptoms, as well as the cognitive and language composite scores of their children. (2) A small to modest increase in the duration of gestation (precise estimate of effect size could not be determined due to obstetric interventions). (3) Fewer infants born <34 weeks gestation (1.09 % vs. 2.25% adjusted Relative Risk (RR), 0.49; p = 0.03), and association with fewer low birth weight infants and fewer admissions to neonatal intensive care. EPTB was reduced by 51.6%. | Dietary intake of n-3 LC-PUFAs was not assessed. The study failed to demonstrate an improvement in primary outcomes such as reduction in depressive symptoms among women and improvement in cognitive and language scores of their children. |
| Ramakrishnan et al. 2010 [78] | To assess if prenatal DHA supplementation increases gestational age and birth size | RCT, DB, PC. | Healthy pregnant women from 18 to 22 weeks of gestation from Mexico n = 1094 | From 18 to 22 weeks of gestation until delivery | Intervention: 2 capsules/day of 200 mg of DHA derived from an algal source (400 mg DHA/day) Placebo: 2 capsules/day containing olive oil | Compared to placebo, DHA supplementation resulted in (1) No differences in mean gestational age, PTB, weight, length and head circumference at birth. | |
| Helland et al. 2001 [79] | To evaluate the effect of n-3 or n-6 long-chain PUFAs on birth weight, gestational length, and infant development | RCT, DB, PC. | Healthy, nulli- or primiparous women in weeks 17 to 19 of pregnancy from Norway n = 590 | 17 to 19 weeks of gestation until 3 months after delivery | Intervention: 10 mL/day of cod liver oil, providing around 2 g daily of the long chain omega-3 fatty acids. Placebo: 10 mL/day of corn oil, providing around 5 g daily of omega-6 fatty acid linoleic acid. | In comparison with placebo, cod liver oil supplementation resulted in (1) No differences in gestational length or birth weight, length or head circumference. (2) Higher concentrations of n-3 fatty acids EPA, DHA, and DHA in umbilical plasma phospholipids. (3) Neonates with high concentration of DHA in umbilical plasma phospholipids (upper quartile) had longer gestational length than those with low concentration (lower quartile; 282.5 (8.5) vs. 275.4 (9.3) days). | Substantial numbers of women excluded from the two groups post randomization due to withdrawals. It does not mention gestational lengths to facilitate undertaking of an ITT analyses. In this population, baseline intake of long-chain n-3 fatty acids was estimated to be relatively high (0.5 g/day) and less than one 1% had a PTB. |
| Olsen et al. 2000 [80] | To test the preventive effects of dietary n-3 fatty acids on Pre-term delivery, Intrauterine growth retardation, and pregnancy-induced hypertension | Multicenter RCT, PC (4 prophylactic + 2 therapeutic trials) | High risk pregnancies from 19 hospitals in 7 different countries in Europe Four prophylactic trials: previous pre-term (n = 232), IUGR (n = 280), PIH (n = 386) and twin pregnancies (n = 579) Two therapeutic trials: threatening pre-eclampsia (n = 79) and suspected IUGR (n = 63) | From ~20 weeks (prophylactic trials) or 33 weeks (therapeutic trials) until delivery. | Intervention: prophylactic trials (4 capsules/day of fish oil, 1.3 g EPA and 0.9 g DHA) and therapeutic trials (9 capsules/day of fish oil, 2.9 g EPA and 2.1g DHA) (32% EPA, 23% DHA, 2 mg tocopherol/mL) Placebo: identical looking capsules of olive oil (72% oleic acid, 12% linoleic acid) | Compared to placebo, fish oil supplementation resulted in the following among women with a previous Pre-term delivery in the prophylactic trial: (1) Reduced recurrence risk of PTB from 33% to 21% (Odds Ratio (OR) 0.54 (95% Confidence Interval (CI) 0.30–0.98)) (2) Reduced recurrence risk of EPTB from 13.3% to 4.6% (OR 0.32 (95% CI 0.11–0.89)). (3) Longer mean gestational length by 8.5 day (95% CI 1.9–15.2. (4) No effect on PTB in twin pregnancies. | |
| Onwude et al. 1995 [81] | To determine whether n-3 fatty acid (EPA/DHA) prophylaxis is beneficial in high-risk pregnancies | RCT, DB, PC. | Pregnant women at high risk of developing PIH and asymmetrical IUGR from an antenatal clinic from UK n = 233 | From around 25 weeks of gestation until 38 weeks of gestation | Intervention: 9 capsules/day of fish oil providing 2.7 g omega-3 fatty acids/day (1.62 g of EPA and 1.08 g of DHA) Placebo: matching air-filled capsules | Compared to placebo, fish oil supplementation resulted in (1) No difference in the duration of gestation or other outcomes such as proteinuric PIH, non-proteinuric PIH, or birth weight within the lowest 3% on the growth charts. | This study failed to support the hypothesis that fish oil supplementation improved pregnancy outcome in an at risk population for impaired fetal growth or PIH. |
| Bulstra Ramakers et al. 1995 [82] | To study the effects of adding 3 g/day of EPA to the diet, on recurrence rate of IUGR and PIH in a high-risk population | RCT, DB, PC. | Pregnant women with a history of IUGR with or without PIH in the previous pregnancy from the Netherlands n = 63 | From 12 to 14 weeks of gestation until delivery | Intervention: 4 capsules 3 times daily, which corresponded to a daily dose of 3 g of EPA Placebo: Identical capsules with coconut oil | Compared to placebo, EPA supplementation resulted in (1) No difference in the rates of PTB | No information was provided about content of DHA No estimate of mean gestational length was provided |
| Olsen et al. 1992 [83] | To study the effect of a fish-oil supplement, a control olive-oil supplement, and no supplementation on pregnancy duration, birthweight, and birth length | RCT | Healthy pregnant women from Denmark n = 533 | From gestation week 30 until delivery | Intervention: Four 1 g fish oil capsules/day containing 2.7 g n-3 fatty acids- 32% EPA, 23% DHA, 2 mg tocopherol Placebo: Four 1 g olive oil capsules/day No supplement group | Compared to placebo fish oil supplementation resulted in: (1) The highest mean length of gestation when all 3 groups were compared in a single analysis (fish oil, olive oil and no supplement: 283, 279.4 and 281.7 days respectively, p = 0.006). (2) On an average 4 days longer pregnancies in the fish-oil group compared to the olive oil group (95% CI: 1.5–6.4, p = 0.005). (3) The effect seemed to depend on the baseline intake of fish. -Among those 20% of the women who had the highest intake of fish at randomization, no difference could be detected between the oil groups. -In those 20% who had the lowest intake for fish, a difference of 7.4 days was observed (95% CI 2.2–12.6 days, p = 0.01). -In the middle 60%, the groups differed by 4.8 days (95 CI 1.8–7.8, p = 0.005). | Maternal baseline dietary intake could explain differences in the duration of gestation and higher intakes may have a saturating effect |
| Mardones et al. 2008 [84] | To study the effect of maternal food fortification with omega-3 fatty acids and multiple micronutrients on birth weight and gestation duration | Non-blinded, RCT, PC. | Healthy pregnant women up to 20 weeks Gestation from Chile n = 972 | From up to 20 weeks of gestation until delivery | Intervention: 2 kg/month of powdered milk fortified with multiple micronutrients and both a-linolenic acid and linoleic acid; iron was supplied in an amino-chelated form Placebo: 2 kg/month powdered milk fortified with small amounts of iron sulphate, copper, zinc, and vitamin C. | Based on ITT analyses and in comparison with placebo, the intervention resulted in (1) Lower incidence of EPTB (0.4% vs. 2.1%; crude OR (95% CI): 5.26 (1.08–34.90), p = 0.02). (2) Increase in gestation duration (1.40 days difference, 95% CI: -0.02–2.82 d, p = 0.05). (3) Higher mean birth weight (65.4 g difference, 95% CI: 5–126 g; p = 0.03). (4) Higher infant length (0.37 cm difference, 95% CI: 0.06–0.68 cm, p = 0.019). | Impossibility to perform a blinded design and have strict control of compliance with the prescribed amounts of the products taken to the homes of the study subjects Slight difference in gestational age at recruitment Associations with gestation duration would need a larger sample size for confirmation (the statistical power reached only 0.61 in ITT analyses) |
| Smuts et al. 2003 [85] | To assess whether higher intake of DHA would increase duration of gestation and birth weight in US women | RCT, DB, PC. | Healthy pregnant women between the 24th and 28th week of pregnancy from the US (predominant black population) n = 291 | From 24–28 weeks of gestation until delivery | Intervention: 1 DHA enriched egg/day (133 mg DHA) Placebo: 1 ordinary egg/day (33 mg DHA) | Compared to the placebo group, the supplementation with DHA-enriched egg resulted in (1) Increased duration of gestation (6.0 ± 2.3 days, p = 0.009) (based on analyses adjusted for maternal BMI at enrollment and number of prior pregnancies). | The unadjusted analysis showed a difference of 2.6 days (not statistically significant), while adjustment for maternal BMI at enrollment and number of prior pregnancies resulted in an increased duration of gestation by 6 days. The adjustments may have introduced a post hoc element into the interpretation of the result. |
| Ref. | Study | Ingredient, Daily Dose | Main Results | Comments | |||
|---|---|---|---|---|---|---|---|
| Objective | Design | Population/Sample Size | Duration | ||||
| Gille et al. 2016 [145] | To assess whether probiotic supplementation with Lactobacillus rhamnosus GR-1 and Lactobacillus reuteri RC-14 can improve maternal vaginal microbiota | RCT, DB, PC | Healthy pregnant women (first trimester) from Germany, n = 320, 2010–2012 | 8 weeks to assess Nugent scores; entire pregnancy for PTB (secondary outcome) | Capsules with 109 CFU, once daily | Compared to placebo, DHA supplementation resulted in No effect on vaginal microbiota (improvement in Nugent scores). No effect on PTB rates. | Low rate of preterm of 4% Very low rate of bacterial vaginosis 3%. Trend increase on miscarriages in treated (7.7% vs. 3.1%, p = 0.08). |
| Luoto et al. 2010 [146] | To assess whether dietary counselling and probiotic supplementation with (Lactobacillus rhamnosus GG and Bifidobacterium lactis Bb12) can improve pregnancy outcomes | RCT, PC 3 groups: (1) Probiotics and dietary counselling vs. (2) Placebo and dietary counselling (DB); (3) Placebo without dietary counselling (SB) | Healthy pregnant women in the first trimester from Finland, n = 256, late 1990s | From the first antenatal visit to the end of pregnancy | Capsules with 1010 CFU, once daily | Compared to placebo, probiotic supplementation resulted in 1. No effect on PTB rate.2. No effect on duration of gestation. | Very low rate of PTB: 1.7%. |
| Kraus Silva 2011 et al. [143] | To assess whether probiotic supplementation with (L. rhamnosus GR-1 and L. reuteri RC-14) can reduce BV and PTB | RCT, DB, PC | Pregnant women (8 to 20 weeks gestation), With asymptomatic BV: Vaginal pH >4.5, Nugent >4 from Brazil n = 644 randomized, late 1990s | <20 weeks gestation to 24 or 26 weeks | Capsules with 106 colony-forming units each, twice daily | Compared to placebo, probiotics supplementation resulted in no effect on PTB rate. However, the PTB rates were lower with treatment (ITT: 1.6%, 5 in 304; vs. 3.3% 10 in 301) | Low rate of PTB 2.5% Low probiotics dose Exclusion criteria were very broad: previous history of PTB, hypertension, diabetes, asthma, cervical incompetence, atypical vaginal bleeding, atypical vaginal secretion, HPV, gonorrhea, syphilis, dysuria, pruritus, burning, corticotherapy, recent antibiotic therapy (within 8 weeks prior to screening) |
| Rautava et al. 2012 [148] | The effect of maternal administration of probiotics on atopic disease in infants. | RCT, DB, PC. | Pregnant women with atopic sensitization and either a history of or active allergic disease from Finland n = 241 | Probiotics given to the mother 8 weeks before and 8 weeks after delivery. | (1) Dietary food supplement with Lactobacillus rhamnosus LPR + Bifidobacterium longum NCC 3001 (109 CFU/day) (2) Dietary food supplement with Lactobacillus paracasei ST11 + NCC 3001 (19 CFU/day) (3) Placebo | No information on preterm birth rates. Gestational age in all groups was 39 weeks with a similar range (34–41 weeks). | Not possible to draw firm conclusions about effects on preterm delivery. However, papers seems to suggest lack of effect because gestational ages were similar between groups. |
| Kim et al. 2010 [157] | The effect of maternal and infant administration of probiotics on atopic disease in infants | RCT, DB, PC. | Pregnant women with a family history of allergic diseases day n = 112, and their infants. from Korea | Probiotic was given to mothers from 8 weeks before delivery until 3 months post-delivery, then to infants from 4 months until 6 months | (1) Bifido Inc mix (Bifidobacterium bifidum BGN4, Bifidobacterium lactis AD011, Lactobacillus acidophilus AD030), 1.6 × 109 CFU/day each, in powder (2) Placebo powder (maltodextrin and alpha-corn) | Infants delivered before 36 weeks were excluded. No difference observed in the number of infants removed between the two groups, suggesting no difference in PTB rates. In both groups the gestational ages were around 40 weeks, and birth weights were similar. | Not possible to draw conclusions about effects on PTB. However, papers seems to suggest lack of effect. |
| Ou et al. 2012 [158] | The effect of maternal administration of probiotics on atopic disease in infants | RCT, DB, PC | Pregnant women with atopic diseases history and Total IgE >100 kU/L from Taiwan n = 191 | From 24 weeks gestation until delivery. After delivery, administration was exclusively to breastfeeding mothers | (1) L. rhamnosus GG (Valio, ATCC 53103) 1010 CFU/ day (2) Placebo (microcrystalline cellulose) | PTB rates were not reported. However, gestational age was 39 weeks in both groups (range 31–41 weeks in the L. rhamnosus GG group and 35–41 weeks in placebo group), which suggests lack of efficacy on PTB rates. | The study suggests that L. rhamnosus GG probably has no impact on PTB rates. |
| Vitali et al. 2012 [159] | The effect of probiotic supplementation during late pregnancy on vaginal microbiota and cytokine secretion | Non-randomized, controlled, pilot | Healthy pregnant women with no symptoms of vaginal or urinary tract infection from Italy n = 27 | Probiotic was given during weeks 32–37 of gestation. | (1) Probiotic group: one sachet of VSL #3 (Lactobacillus acidophilus, Lactobacillus plantarum, Lactobacillus casei, Lactobacillus delbrueckii ssp. bulgaricus, Bifidobacterium breve, Bifidobacterium longum, Bifidobacterium infantis, S. salivaris ssp. thermophilus) (n = 12) 9 × 1011 total CFU/day (2) Control group: no supplementation (n = 12) | PTB rates were not reported, but the gestational ages were not different between the two groups. This suggests that the probiotic did had no effect on PTB rates. No significant changes were found in the amounts of the principal vaginal bacterial populations in women administered with VSL#3, but qPCR results suggested a potential role of the probiotic product in counteracting the decrease of Bifidobacterium and the increase of Atopobium, that occurred in control women during late pregnancy. Incidence of vaginal infections was not reported. | The study is too small to draw conclusions, but it did not show any effect of VSL3 on gestational age. |
| Stojanovic et al. 2012 [154] | The effect of probiotics on vaginal microflora, cervical length, cervical consistency, and fetal positioning. | Observational, randomized, prospective | Pregnant women | Probiotic was administered for 12 weeks during pregnancy | (1) untreated arm of the study (n = 30) (2) vaginal application of one tablet containing L. rhamnosus BMX 54 (Normogin™-(n = 30) once a week | No data on PTB rates as women were not followed until delivery. Increase in pathogenic microorganisms in the vaginal and/or cervical swabs of untreated women (p <0.05), also in average pH values (p <0.05), amount (p <0.05) and “whiff test” positivity (p <0.05) of vaginal discharge. Significant trend was also found for decrease in length (p <0.0001) and increase in dilatation (p <0.05) of cervix, as well as for lower position of the fetus (p <0.0001). In the group treated with L. rhamnosus BMX 54, none of these values significantly changed throughout the observation period, with the exception of cervical length that was significantly decreased at T3 (p <0.01). | Cannot conclude on PTB rates. However, it suggests that vaginally administered probiotic had a positive impact on parameters associated with PTB. |
| Nutrient | Evidence for Efficacy | Dose | Duration | Comments |
|---|---|---|---|---|
| n−3 LC-PUFA (combinations of EPA and DHA) | 26–61% reduction in the risk of early PTB | DHA: 133 to 2100 mg DHA/day EPA: 100 to 3000 mg EPA/day | Supplementation started between 12 to 30 weeks of gestation | Eight trials supplementing either DHA or EPA alone or using varying combinations of both (five trials in healthy pregnancies and three in at-risk pregnancies), two food-based interventions and 6 meta-analyses |
| DHA (predominantly DHA) | 51.6% to 87.5% reduction in the risk of early PTB (<34 weeks) | 600 to 800 mg DHA/day | Supplementation started <20 to 21 weeks of gestation | Two large RCTs available where PTB and EPTB were secondary outcomes and not the primary outcome. |
| Zinc | 14% reduction in PTB | 5 mg/day to 44 mg/day | Supplementations started from as early as before conception (one study) to at least starting before 26 weeks | Most studies were conducted in low income countries among women with poor nutritional status and likely to have low zinc concentrations. The reduction in PTB was not accompanied by reduction in LBW or a difference in the gestational age at birth. |
| Vitamin D | 64% reduction in PTB | 400 to 1000 IU/day (two trials), 60000–12000 IU (depending on baseline serum 25 (OH)D (one trial) cholecalciferol D3 | Supplementation started between 20–30 weeks of gestation | The trials available were all of low quality. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Samuel, T.M.; Sakwinska, O.; Makinen, K.; Burdge, G.C.; Godfrey, K.M.; Silva-Zolezzi, I. Preterm Birth: A Narrative Review of the Current Evidence on Nutritional and Bioactive Solutions for Risk Reduction. Nutrients 2019, 11, 1811. https://doi.org/10.3390/nu11081811
Samuel TM, Sakwinska O, Makinen K, Burdge GC, Godfrey KM, Silva-Zolezzi I. Preterm Birth: A Narrative Review of the Current Evidence on Nutritional and Bioactive Solutions for Risk Reduction. Nutrients. 2019; 11(8):1811. https://doi.org/10.3390/nu11081811
Chicago/Turabian StyleSamuel, Tinu M., Olga Sakwinska, Kimmo Makinen, Graham C. Burdge, Keith M. Godfrey, and Irma Silva-Zolezzi. 2019. "Preterm Birth: A Narrative Review of the Current Evidence on Nutritional and Bioactive Solutions for Risk Reduction" Nutrients 11, no. 8: 1811. https://doi.org/10.3390/nu11081811
APA StyleSamuel, T. M., Sakwinska, O., Makinen, K., Burdge, G. C., Godfrey, K. M., & Silva-Zolezzi, I. (2019). Preterm Birth: A Narrative Review of the Current Evidence on Nutritional and Bioactive Solutions for Risk Reduction. Nutrients, 11(8), 1811. https://doi.org/10.3390/nu11081811





