Cardioprotective Effect of a Virgin Olive Oil Enriched with Bioactive Compounds in Spontaneously Hypertensive Rats
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Oils
2.2. Experimental Design
2.3. Blood Pressure Determination
2.4. Plasma Biochemical Analysis
2.5. Urine Biochemical Analysis
2.6. Vascular Reactivity in Aortic Rings
2.7. Statistical Analysis
3. Results
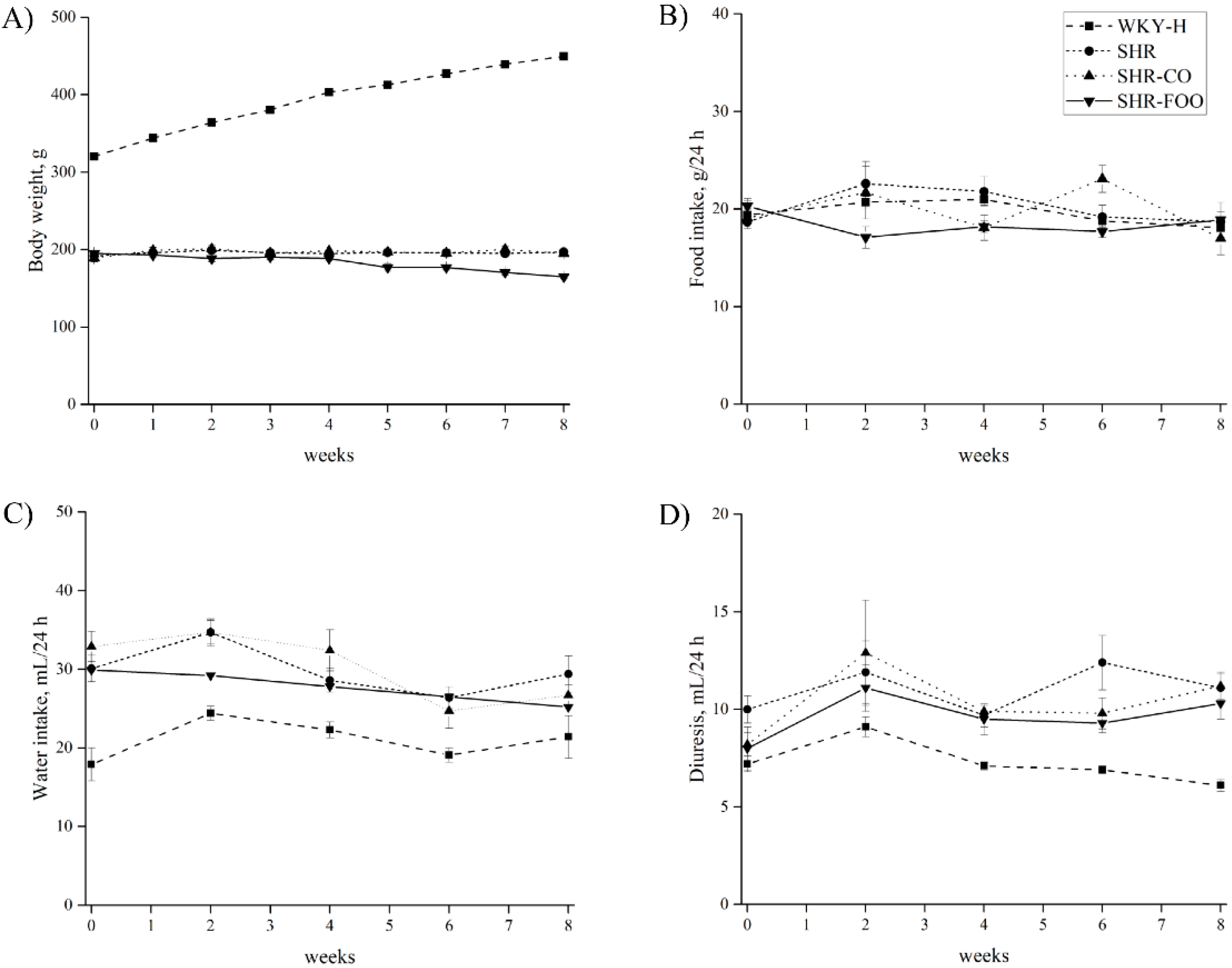
3.1. Time-Course of Body Weight, Food Intake, Water Intake, and Diuresis
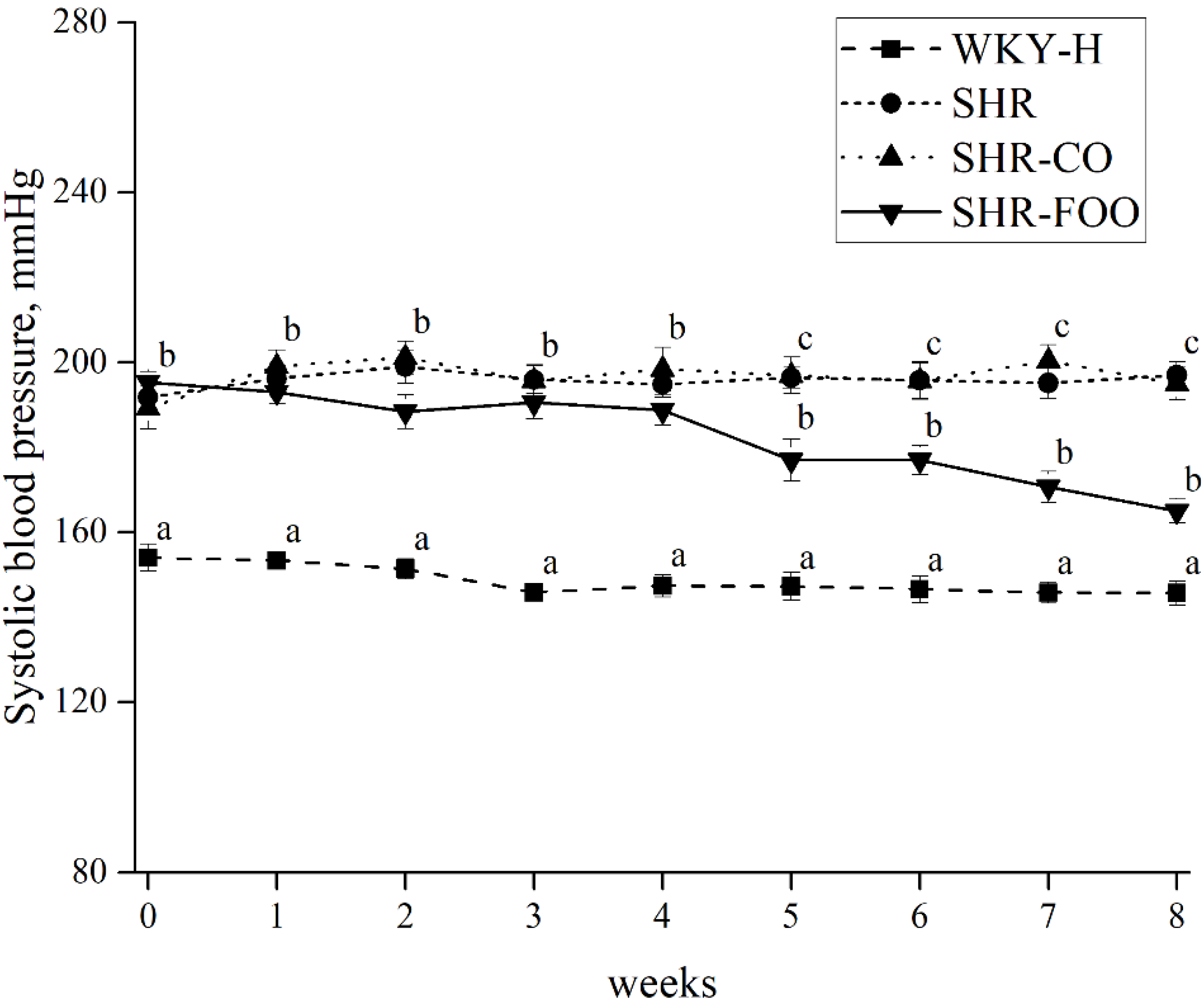
3.2. Effects of Enriched Olive Oil on Blood Pressure in SHR
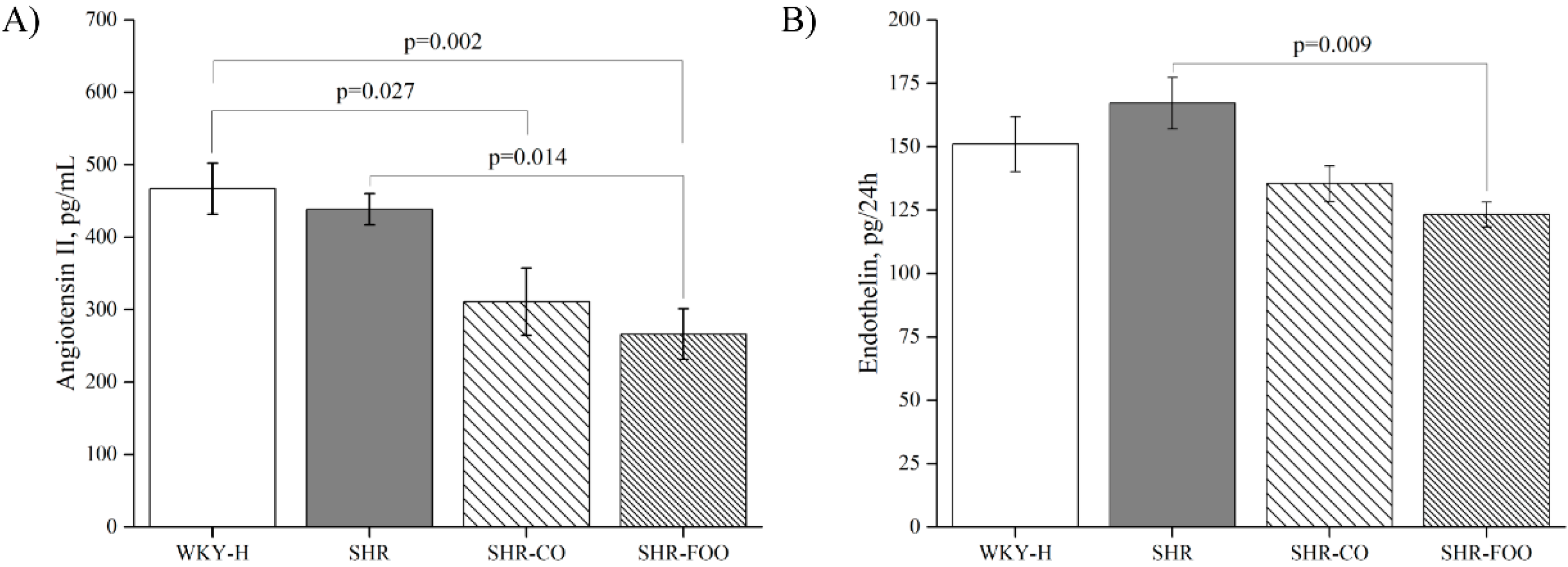
3.3. Vasoactive Peptides: Plasma Angiotensin II and Urinary Endothelin-1
3.4. Morphological Variables
3.5. Plasma Urine Biochemical Variables
3.6. Oxidative and Inflammatory Biomarkers
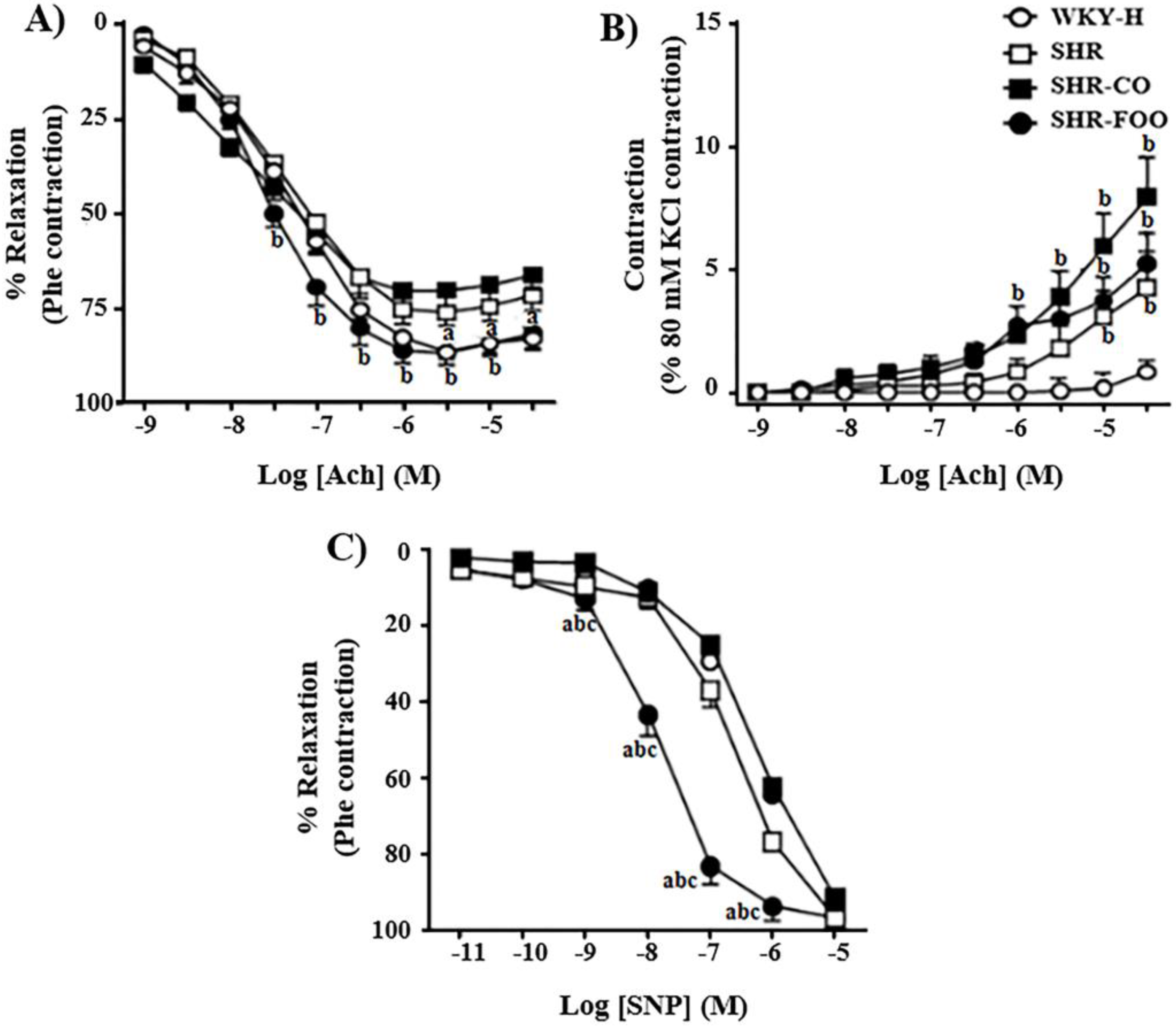
3.7. Ex-Vivo Effects on Vascular Function
4. Discussion
5. Limitations and Strengths
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
References
- Cogolludo, A.; Pérez-Vizcaino, F.; Tamargo, J. New insights in the pharmacological therapy of arterial hypertension. Curr. Opin. Nephrol. Hypertens. 2005, 14, 423–427. [Google Scholar] [CrossRef] [PubMed]
- George, E.S.; Marshall, S.; Mayr, H.L.; Trakman, G.L.; Tatucu-Babet, O.A.; Lassemillante, A.M.; Bramley, A.; Reddy, A.J.; Forsyth, A.; Tierney, A.C.; et al. The effect of high-polyphenol extra virgin olive oil on cardiovascular risk factors: A systematic review and meta-analysis. Crit. Rev. Food Sci. Nutr. 2018, 30, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Yubero-Serrano, E.M.; Lopez-Moreno, J.; Gomez-Delgado, F.; Lopez-Miranda, J. Extra virgin olive oil: More than a healthy fat. Eur. J. Clin. Nutr. 2018, 28. [Google Scholar] [CrossRef] [PubMed]
- Jänicke, C.; Grünwald, J.; Brendler, T. Handbuch Phytotherapie; Wissenschaftliche Verlagsgesellschaft: Stuttgart, Germany, 2003. [Google Scholar]
- Cherif, S.; Rahal, N.; Haouala, M.; Hizaoui, B.; Dargouth, F.; Gueddiche, M.; Kallel, Z.; Balansard, G.; Boukef, K. A clinical trial of a titrated Olea extract in the treatment of essential arterial hypertension. J. Pharm. Belg. 1996, 51, 69–71. [Google Scholar] [PubMed]
- Bennani-Kabchi, N.; Fdhil, H.; Cherrah, Y.; Kehel, L.; Bouayadi, F.; Amarti, A.; Saïdi, M.; Marquié, G. Effects of Olea europea var. oleaster leaves in hypercholesterolemic insulin-resistant sand rats. Thérapie 1999, 54, 717–723. [Google Scholar] [PubMed]
- Susalit, E.; Agus, N.; Effendi, I.; Tjandrawinata, R.R.; Nofiarny, D.; Perrinjaquet-Moccetti, T.; Verbruggen, M. Olive (Olea europaea) leaf extract effective in patients with stage-1 hypertension: Comparison with Captopril. Phytomedicine 2011, 18, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Villarejo, A.B.; Ramírez-Sánchez, M.; Segarra, A.B.; Martínez-Cañamero, M.; Prieto, I. Influence of extra virgin olive oil on blood pressure and kidney angiotensinase activities in spontaneously hypertensive rats. Planta Med. 2015, 81, 664–669. [Google Scholar] [CrossRef]
- Rodriguez-Rodriguez, R.; Herrera, M.D.; de Sotomayor, M.A.; Ruiz-Gutierrez, V. Pomace olive oil improves endothelial function in spontaneously hypertensive rats by increasing endothelial nitric oxide synthase expression. Am. J. Hypertens. 2007, 20, 728–734. [Google Scholar] [CrossRef]
- Rodriguez-Rodriguez, R.; Herrera, M.D.; de Sotomayor, M.A.; Ruiz-Gutierrez, V. Effects of pomace olive oil-enriched diets on endothelial function of small mesenteric arteries from spontaneously hypertensive rats. Br. J. Nutr. 2009, 102, 1435–1444. [Google Scholar] [CrossRef]
- Zarzuelo, A.; Duarte, J.; Jiménez, J.; Gonzalez, M.; Utrilla, M.P. Vasodilator effect of olive leaf. Planta Med. 1991, 57, 417–419. [Google Scholar] [CrossRef]
- Romero, M.; Toral, M.; Gómez-Guzmán, M.; Jimenez, R.; Galindo, P.; Sanchez, M.; Olivares, M.; Gálvez, J.; Duarte, J. Antihypertensive effects of oleuropein-enrichedolive leaf extract in spontaneously hypertensive rats. Food Funct. 2016, 7, 584–593. [Google Scholar] [CrossRef] [PubMed]
- Olmo-García, L.; Polari, J.J.; Li, X.; Bajoub, A.; Fernández-Gutiérrez, A.; Wang, S.C.; Carrasco-Pancorbo, A. Deep insight into the minor fraction of virgin olive oil by using LC-MS and GC-MS multi-class methodologies. Food Chem. 2018, 261, 184–193. [Google Scholar] [CrossRef] [PubMed]
- Vera, R.; Jiménez, R.; Lodi, F.; Sánchez, M.; Galisteo, M.; Zarzuelo, A.; Pérez-Vizcaíno, F.; Duarte, J. Genistein restores caveolin-1 and AT-1 receptor expression and vascular function in large vessels of ovariectomized hypertensive rats. Menopause 2007, 14, 933–940. [Google Scholar] [CrossRef] [PubMed]
- Núñez-Córdoba, J.M.; Valencia-Serrano, F.; Toledo, E.; Alonso, A.; Martínez-González, M.A. The Mediterranean diet and incidence of hypertension: The seguimiento universidad de navarra (SUN) study. Am. J. Epidemiol. 2009, 169, 339–346. [Google Scholar] [CrossRef] [PubMed]
- Toledo, E.; Hu, F.B.; Estruch, R.; Buil-Cosiales, P.; Corella, D.; Salas-Salvadó, J.; Covas, M.I.; Arós, F.; Gómez-Gracia, E.; Fiol, M.; et al. Effect of the Mediterranean diet on blood pressure in the PREDIMED trial: Results from a randomized controlled trial. BMC Med. 2013, 11, 207. [Google Scholar] [CrossRef] [PubMed]
- Lockyer, S.; Rowland, I.; Rowland, I.; Spencer, J.P.E.; Yaqoob, P.; Stonehouse, W. Impact of phenolic-rich olive leaf extract on blood pressure, plasma lipids and inflammatory markers: A randomised controlled trial. Eur. J. Nutr. 2017, 56, 1421–1432. [Google Scholar] [CrossRef] [PubMed]
- Lopez, S.; Bermudez, B.; Montserrat de la Paz, S.; Jaramillo, S.; Abia, R.; Muriana, F.J. Virgin olive oil and hypertension. Curr. Vasc. Pharmacol. 2016, 14, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Peyrol, J.; Riva, C.; Amiot, M.J. Hydroxytyrosol in the prevention of the metabolic syndrome and related disorders. Nutrients 2017, 9, 306. [Google Scholar] [CrossRef] [PubMed]
- Schiffrin, E.L. Vascular endothelin in hypertension. Vasc. Pharmacol. 2005, 43, 19–29. [Google Scholar] [CrossRef]
- Chamorro, V.; Wangensteen, R.; Sainz, J.; Duarte, J.; O’Valle, F.; Osuna, A.; Vargas, F. Protective effects of the angiotensin II type 1(AT1) receptor blockade in low-renin deoxycorticosterone acetate (DOCA)-treated spontaneously hypertensive rats. Clin. Sci. 2004, 106, 251–259. [Google Scholar] [CrossRef]
- Oliveira-Paula, G.H.; Pinheiro, L.C.; Tanus-Santos, J.E. Mechanisms impairing blood pressure responses to nitrite and nitrate. Nitric Oxide 2019, 85, 35–43. [Google Scholar] [CrossRef] [PubMed]
- Ramírez-Expósito, M.J.; Martínez-Martos, J.M.; Prieto, I.; Alba, F.; Ramírez, M. Angiotensinase activity in mice fed an olive oil-supplemented diet. Peptides 2001, 22, 945–952. [Google Scholar] [CrossRef]
- Williams, P.T.; Fortmann, S.P.; Terry, R.B.; Garay, S.C.; Vranizan, K.M.; Ellsworth, N.; Wood, P.D. Associations of dietary fat, regional adiposityand blood pressure in men. JAMA 1987, 257, 3251–3256. [Google Scholar] [CrossRef] [PubMed]
- Hansen, K.; Adsersen, A.; Christensen, S.B.; Jensen, S.R.; Nyman, U.; Smitt, U.W. Isolation of an angiotensin converting enzyme (ACE) inhibitor from Olea europaea and Olea lancea. Phytomedicine 1996, 2, 319–325. [Google Scholar] [CrossRef]
- Storniolo, C.E.; Casillas, R.; Bullo, M.; Castañer, O.; Ros, E.; Saez, G.T.; Toledo, E.; Estruch, R.; Ruiz-Gutiérrez, V.; Fito, M.; et al. A Mediterranean diet supplemented with extra virgin olive oil or nuts improves endothelial markers involved in blood pressure control in hypertensive women. Eur. J. Nutr. 2017, 56, 89–97. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Rodriguez, E.; Lima-Cabello, E.; Biel-Glesson, S.; Fernandez-Navarro, J.R.; Calleja, M.A.; Roca, M.; Espejo-Calvo, J.A.; Gil-Extremera, B.; Soria-Florido, M.; de la Torre, R.; et al. Effects of virgin olive oils differing in their bioactive compound contents on metabolic syndrome and endothelial functional risk biomarkers in healthy adults: A randomized double-blind controlled trial. Nutrients 2018, 10, 626. [Google Scholar] [CrossRef] [PubMed]
- Duarte, J.; Jiménez, R.; O’Valle, F.; Galisteo, M.; Pérez-Palencia, R.; Vargas, F.; Pérez-Vizcaíno, F.; Zarzuelo, A.; Tamargo, J. Protective effects of the flavonoid quercetin in chronic nitric oxide deficient rats. J. Hypertens. 2002, 20, 1843–1854. [Google Scholar] [CrossRef] [PubMed]
- Martín-Peláez, S.; Castañer, O.; Konstantinidou, V.; Subirana, I.; Muñoz-Aguayo, D.; Blanchart, G.; Gaixas, S.; de la Torre, R.; Farré, M.; Sáez, G.T.; et al. Effect of olive oil phenolic compounds on the expression of blood pressure-related genes in healthy individuals. Eur. J. Nutr. 2017, 56, 663–670. [Google Scholar] [CrossRef]
- Trippodo, N.; Frolich, E. Similarities of genetic (spontaneous) hypertension: Man and rat. Circ. Res. 1981, 48, 309–319. [Google Scholar] [CrossRef]
- Karam, H.; Heudes, D.; Bruneval, P.; Gonzales, M.F.; Löffler, B.M.; Clozel, M.; Clozel, J.P. Endothelin antagonism in end-organ damage of spontaneously hypertensive rats. Comparison with angiotensin-converting enzyme inhibition and calcium antagonism. Hypertension 1996, 28, 379–385. [Google Scholar] [CrossRef]
- Simonson, M.S. Endothelins: Multifunctional renal peptides. Physiol. Rev. 1993, 73, 375–411. [Google Scholar] [CrossRef] [PubMed]
- Franco, M.N.; Galeano-Díaz, T.; López, O.; Fernández-Bolaños, J.G.; Sánchez, J.; De Miguel, C.; Gil, M.V.; Martín-Vertedor, D. Phenolic compounds and antioxidant capacity of virgin olive oil. Food Chem. 2014, 163, 289–298. [Google Scholar] [CrossRef] [PubMed]
- Gorzynik-Debicka, M.; Przychodzen, P.; Cappello, F.; Kuban-Jankowska, A.; Marino Gammazza, A.; Knap, N.; Wozniak, M.; Gorska-Ponikowska, M. Potential health benefits of olive oil and plant polyphenols. Int. J. Mol. Sci. 2018, 19, 686. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Rodriguez, E.; Biel-Glesson, S.; Fernandez-Navarro, J.R.; Calleja, M.A.; Espejo-Calvo, J.A.; Gil-Extremera, B.; de la Torre, R.; Fito, M.; Covas, M.I.; Vilchez, P.; et al. Effects of virgin olive oils differing in their bioactive compound contents on biomarkers of oxidative stress and inflammation in healthy adults: A randomized double-blind controlled trial. Nutrients 2019, 11, 561. [Google Scholar] [CrossRef] [PubMed]
- Tsartsou, E.; Proutsos, N.; Castanas, E.; Kampa, M. Network meta-analysis of metabolic effects of olive-oil in humans shows the importance of olive oil consumption with moderate polyphenol levels as part of the mediterranean diet. Front. Nutr. 2019, 6, 6. [Google Scholar] [CrossRef] [PubMed]
- Nekooeian, A.A.; Khalili, A.; Khosravi, M.B. Effects of oleuropein in rats with simultaneous type 2 diabetes and renal hypertension: A study of antihypertensive mechanisms. J. Asian Nat. Prod. Res. 2014, 16, 953–962. [Google Scholar] [CrossRef] [PubMed]
- Poudyal, H.; Campbell, F.; Brown, L. Olive leaf extract attenuates cardiac, hepatic, and metabolic changes in high carbohydrate-, high fat-fed rats. J. Nutr. 2010, 140, 946–953. [Google Scholar] [CrossRef] [PubMed]
- Raederstorff, D. Antioxidant activity of olive polyphenols in humans: A review. Int. J. Vitam. Nutr. Res. 2009, 79, 152–165. [Google Scholar] [CrossRef] [PubMed]
- Oskarsson, H.J.; Heistad, D.D. Oxidative stress produced by Angiotensin II: Implications for hypertension and vascular injury. Circulation 1997, 95, 557–559. [Google Scholar] [CrossRef] [PubMed]
- Wind, S.; Beuerlein, K.; Armitage, M.E.; Taye, A.; Kumar, A.H.; Janowitz, D.; Neff, C.; Shah, A.M.; Wingler, K.; Schmidt, H.H. Oxidative stress and endothelial dysfunction in aortas of aged spontaneously hypertensive rats by NOX1/2 is reversed by NADPH oxidase inhibition. Hypertension 2010, 56, 490–497. [Google Scholar] [CrossRef] [PubMed]
- Casas, R.; Estruch, R.; Sacanella, E. The protective effects of extra virgin olive oil on immune-mediated inflammatory responses. Endocr. Metab. Immune Disord. Drug Targ. 2018, 18, 23–35. [Google Scholar] [CrossRef] [PubMed]
- Yarla, N.S.; Polito, A.; Peluso, I. Effects of Olive Oil on TNF-α and IL-6 in Humans: Implication in obesity and frailty. Endocr. Metab. Immune Disord. Drug Targ. 2018, 18, 63–74. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Saleh, M.A.; Kirabo, A.; Itani, H.A.; Montaniel, K.R.; Xiao, L.; Chen, W.; Mernaugh, R.L.; Cai, H.; Bernstein, K.E.; et al. Immune activation caused by vascular oxidation promotes fibrosis and hypertension. J. Clin. Investig. 2016, 126, 50–67. [Google Scholar] [CrossRef] [PubMed]
- Guasch-Ferré, M.; Hu, F.B.; Martínez-González, M.A.; Fitó, M.; Bulló, M.; Estruch, R.; Fiol, M. Olive oil intake and risk of cardiovascular disease and mortality in the PREDIMED Study. BMC Med. 2014, 12, 78. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | CO | FOO |
|---|---|---|
| Acidity (%) | 0.14 | 0.14 |
| Peroxide value (meq/Kg) | 5.3 | 9.5 |
| K270 | 0.14 | 0.15 |
| K232 | 1.79 | 1.85 |
| DeltaK | <0.01 | <0.01 |
| Main Fatty acids | ||
| Palmithic (C16) (%) | 10.12 | 10.09 |
| Stearic (C18) (%) | 3.81 | 3.79 |
| Oleic (C18:1) (%) | 79.71 | 79.87 |
| Linoleic (C18:2n6) (%) | 3.95 | 3.91 |
| Total ethyl esters (mg/Kg) | 7 | 13 |
| Total sterols (mg/Kg) | 1316 | 1328 |
| Total phenolic compounds (mg/Kg) | 17.6 | 749.9 |
| WKY-H | SHR | SHR-CO | SHR-FOO | p | |
|---|---|---|---|---|---|
| BW(g) | 444.9 ± 4.0 a | 403.4 ± 9.7 b | 394.9 ± 8.0 b | 390.5 ± 10.8 b | 0.001 |
| HW (g) | 1.0 ± 0.01 a | 1.3 ± 0.03 c | 1.3 ± 0.03 bc | 1.2 ± 0.03 b | 0.001 |
| HW/BW (mg/g) | 2.4 ± 0.02 a | 3.3 ± 0.1 c | 3.3 ± 0.08 bc | 3.1 ± 0.07 b | 0.001 |
| LVW (mg) | 0.86 ± 0.01 a | 1.1 ± 0.02 c | 1.2 ± 0.03 bc | 1.0 ± 0.03 b | 0.001 |
| LVW/BW (mg/g) | 1.9 ± 0.02 a | 2.8 ± 0.09 c | 3.0 ± 0.08 bc | 2.6 ± 0.05 b | 0.001 |
| KW (g) | 1.3 ± 0.02 | 1.2 ± 0.04 | 1.2 ± 0.03 | 1.2 ± 0.02 | 0.020 |
| KW/BW (mg/g) | 2.9 ± 0.05 | 3.1 ± 0.09 | 3.0 ± 0.06 | 3.1 ± 0.08 | 0.147 |
| WKY-H | SHR | SHR-CO | SHR-FOO | p | |
|---|---|---|---|---|---|
| Plasma | |||||
| Sodium (mEq/L) | 144.1 ± 0.7 | 143.9 ± 1.2 | 144.7 ± 0.9 | 144.1 ± 0.8 | 0.940 |
| Potassium (mEq/L) | 4.3 ± 0.1 ab | 4.7 ± 0.2 b | 4.2 ± 0.1 a | 4.3 ± 0.1 ab | 0.040 |
| Creatinine (mg/dL) | 0.4 ± 0.03 a | 0.5 ± 0.05 b | 0.4 ± 0.03 a | 0.4 ± 0.03 a | 0.004 |
| Urea (mg/dL) | 37.5 ± 1.4 | 41.2 ± 2.8 | 41.9 ± 1.9 | 40.0 ± 1.4 | 0.400 |
| Total cholesterol (mg/dL) | 79.9 ± 3.3 a | 77.9 ± 5.9 ab | 74.3 ± 2.2 ab | 65.2 ± 1.9 b | 0.020 |
| HDL cholesterol (mg/dL) | 63.2 ± 2.6 | 63.6 ± 5.0 | 59.4 ± 2.0 | 63.2 ± 2.7 | 0.770 |
| LDL cholesterol (mg/dL) | 9.2 ± 0.7 | 10.8 ± 1.1 | 10.0 ± 0.7 | 9.2 ± 0.7 | 0.500 |
| Urine | |||||
| Diuresis (mL/100 g/24 h) | 1.3 ± 0.1 a | 2.7 ± 0.2 b | 2.8 ± 0.1 b | 2.7 ± 0.2 b | 0.001 |
| Natriuresis (µEq/100 g/24 h) | 243.7 ± 31.1 a | 285.8 ± 27.0 ab | 336.7 ± 20.9 b | 248.4 ± 18.1 ab | 0.040 |
| Kaliuresis (µEq/100 g/24 h) | 520.5 ± 50.8 | 609.4 ± 40.9 | 617.7 ± 37.9 | 531.5 ± 50.0 | 0.320 |
| Creatinine (mL/min/kg) | 1.8 ± 0.1 a | 2.7 ± 0.1 b | 2.8 ± 0.1 b | 2.5 ± 0.1 b | 0.001 |
| Clearance/Creatinine (mL/min/kg) | 0.64 ± 0.07 a | 0.63 ± 0.05 a | 0.94 ± 0.06 b | 0.91 ± 0.08 b | 0.001 |
| Proteinuria (g/dL) | 2.8 ± 2.3 | 2.9 ± 2.1 | 2.6 ± 1.0 | 2.3 ± 2.1 | 0.200 |
| Nitrites µM | 208.1 ± 43.9 ab | 144.8 ± 50.1 a | 344.4 ± 41.5 bc | 393.3 ± 48.7 c | 0.002 |
| Water balance (mL/100 g/24 h) | 3.4 ± 0.6 | 4.6 ± 0.7 | 3.9 ± 0.3 | 3.6 ± 0.2 | 0.510 |
| Sodium balance (µEq/100 g/day) | 462.9 ± 38.7 | 528.4 ± 45.7 | 415.4 ± 82.1 | 587.4 ± 61.7 | 0.210 |
| WKY-H | SHR | SHR-CO | SHR-FOO | p | |
|---|---|---|---|---|---|
| Urine | |||||
| 8-OH-dG (ng/mL) | 53.2 ± 5.0 a | 26.7 ± 4.2 b | 24.5 ± 4.4 b | 17.1 ± 4.4 c | 0.001 |
| F2-isoprostanes (ng/mL) | 7.6 ± 0.8 a | 3.8 ± 0.5 b | 2.8 ± 0.3 b | 3.3 ± 0.3 b | 0.001 |
| Plasma | |||||
| IL-6 (pg/mL) | 3.6 ± 0.1 | 3.3 ± 0.1 | 3.4 ± 0.1 | 3.3 ± 0.2 | 0.280 |
| TNF-α (pg/mL) | 45.9 ± 8.9 b | 26.3 ± 2.7 ab | 24.4 ± 3.2 ab | 23.6 ± 3.2 a | 0.021 |
| VEGF (pg/mL) | 12.9 ± 1.3 | 10.6 ± 0.7 | 9.6 ± 0.5 | 11.5 ± 0.7 | 0.060 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vazquez, A.; Sanchez-Rodriguez, E.; Vargas, F.; Montoro-Molina, S.; Romero, M.; Espejo-Calvo, J.A.; Vilchez, P.; Jaramillo, S.; Olmo-García, L.; Carrasco-Pancorbo, A.; et al. Cardioprotective Effect of a Virgin Olive Oil Enriched with Bioactive Compounds in Spontaneously Hypertensive Rats. Nutrients 2019, 11, 1728. https://doi.org/10.3390/nu11081728
Vazquez A, Sanchez-Rodriguez E, Vargas F, Montoro-Molina S, Romero M, Espejo-Calvo JA, Vilchez P, Jaramillo S, Olmo-García L, Carrasco-Pancorbo A, et al. Cardioprotective Effect of a Virgin Olive Oil Enriched with Bioactive Compounds in Spontaneously Hypertensive Rats. Nutrients. 2019; 11(8):1728. https://doi.org/10.3390/nu11081728
Chicago/Turabian StyleVazquez, Alejandra, Estefania Sanchez-Rodriguez, Félix Vargas, Sebastián Montoro-Molina, Miguel Romero, Juan Antonio Espejo-Calvo, Pedro Vilchez, Sara Jaramillo, Lucía Olmo-García, Alegría Carrasco-Pancorbo, and et al. 2019. "Cardioprotective Effect of a Virgin Olive Oil Enriched with Bioactive Compounds in Spontaneously Hypertensive Rats" Nutrients 11, no. 8: 1728. https://doi.org/10.3390/nu11081728
APA StyleVazquez, A., Sanchez-Rodriguez, E., Vargas, F., Montoro-Molina, S., Romero, M., Espejo-Calvo, J. A., Vilchez, P., Jaramillo, S., Olmo-García, L., Carrasco-Pancorbo, A., de la Torre, R., Fito, M., Covas, M.-I., Martínez de Victoria, E., & Mesa, M. D. (2019). Cardioprotective Effect of a Virgin Olive Oil Enriched with Bioactive Compounds in Spontaneously Hypertensive Rats. Nutrients, 11(8), 1728. https://doi.org/10.3390/nu11081728









