Preconception and Prenatal Nutrition and Neurodevelopmental Disorders: A Systematic Review and Meta-Analysis
Abstract
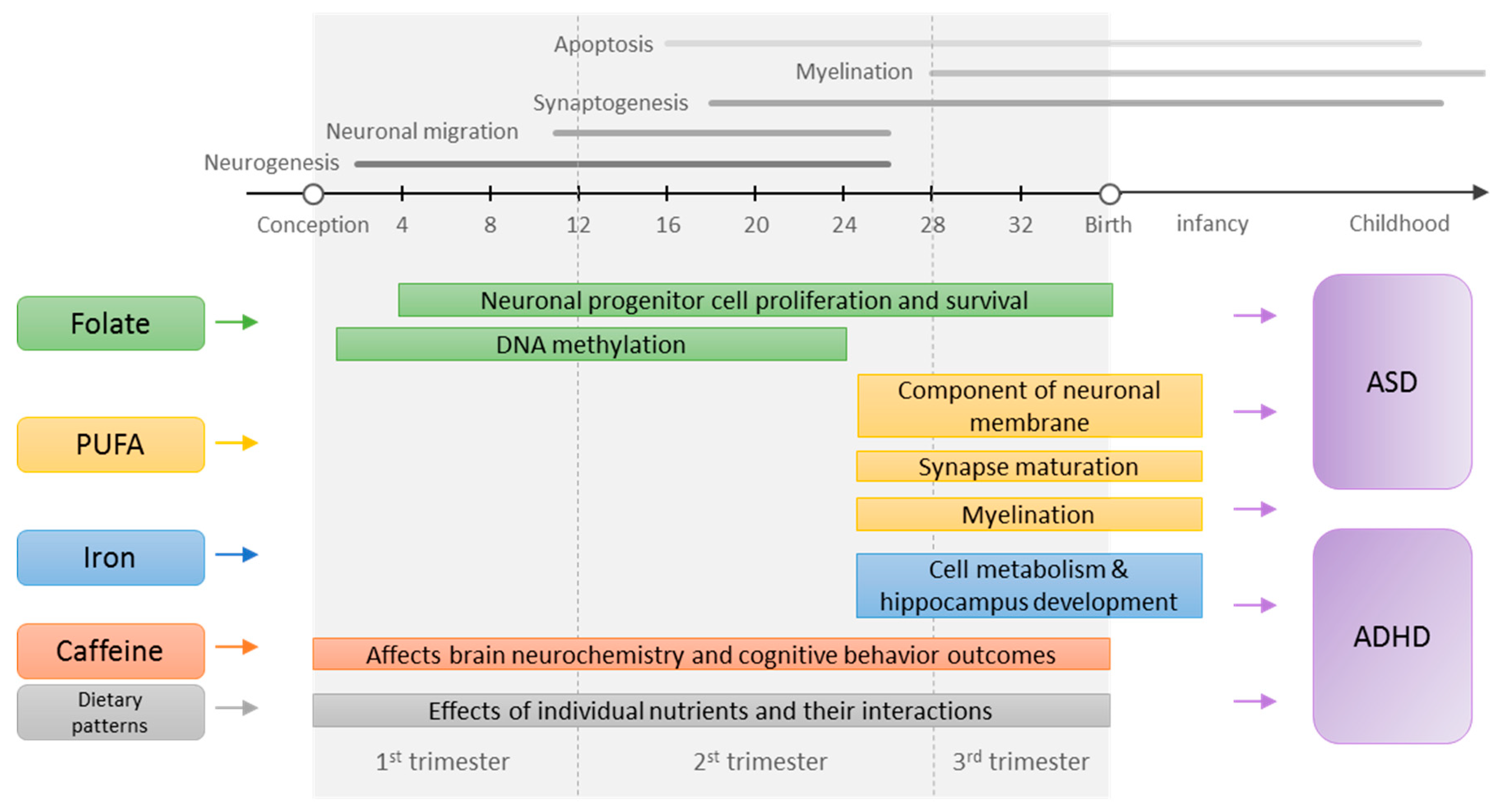
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
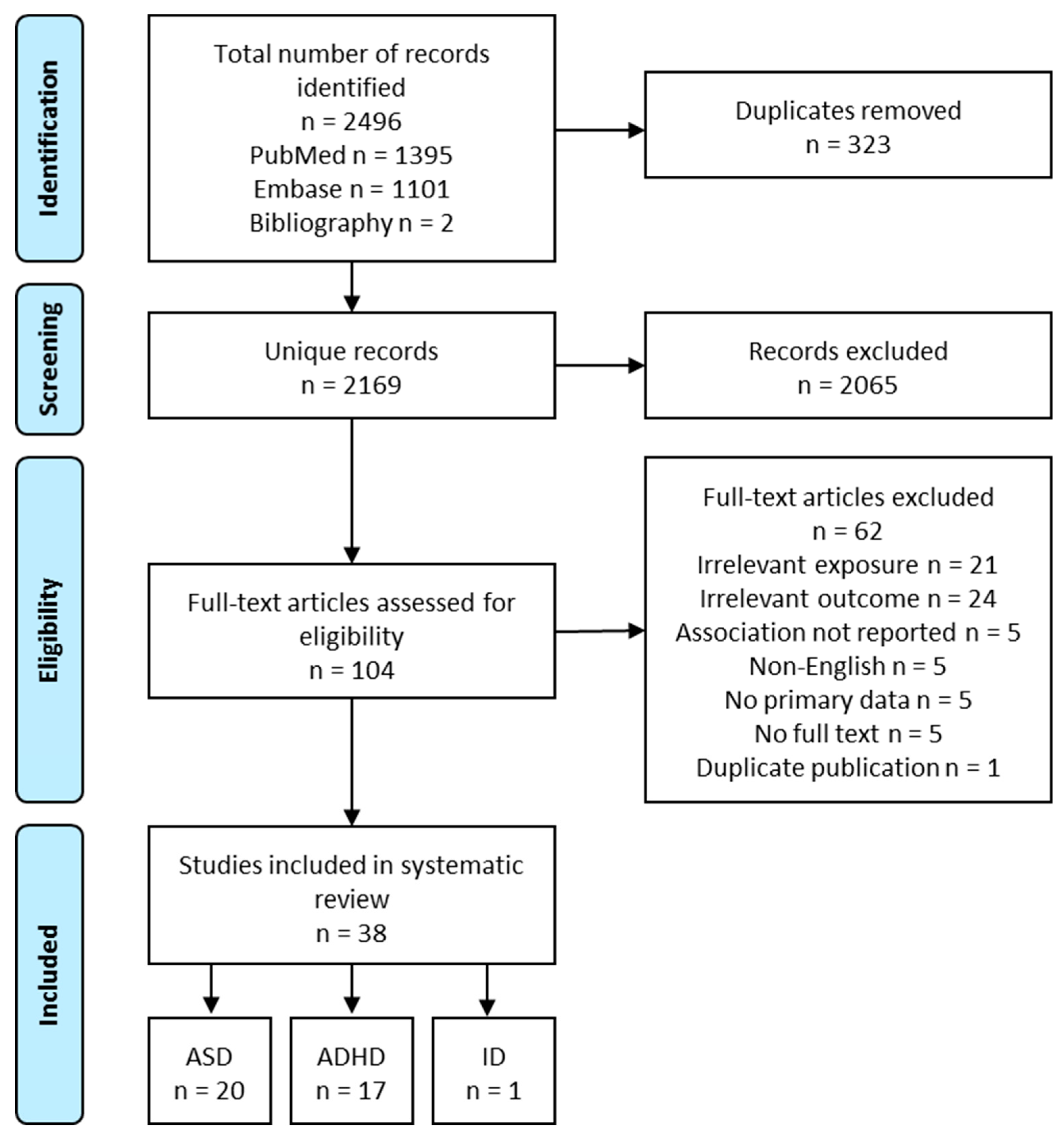
2.2. Literature Search, Screening and Abstraction
2.3. Meta-Analysis
2.4. Quality Assessment
3. Results and Commentary
3.1. Maternal Nutrition and ASD Risk
3.1.1. Maternal Folate Intake/Status, Multivitamin Intake and Offspring ASD Risk
3.1.2. Maternal Iron Intake and Offspring ASD Risk
3.1.3. Maternal PUFA Intake or Status, Seafood Intake and Offspring ASD Risk
3.1.4. Maternal Dietary Patterns and Offspring risk of ASD
3.2. Maternal Nutrition and ADHD Risk
3.2.1. Maternal Folate Intake/Status, Multivitamin Intake and Offspring ADHD Risk
3.2.2. Maternal PUFA and Seafood Intake and Offspring ADHD Risk
3.2.3. Maternal Caffeine, Coffee, and Tea Intake and Offspring ADHD Risk
3.2.4. Maternal Dietary Patterns and Offspring ADHD Risk
4. Methodological Issues and Future Directions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- King, J.C. A summary of pathways or mechanisms linking preconception maternal nutrition with birth outcomes. J. Nutr. 2016, 146, 1437s–1444s. [Google Scholar] [CrossRef] [PubMed]
- Stephenson, J.; Heslehurst, N.; Hall, J.; Schoenaker, D.; Hutchinson, J.; Cade, J.E.; Poston, L.; Barrett, G.; Crozier, S.R.; Barker, M.; et al. Before the beginning: Nutrition and lifestyle in the preconception period and its importance for future health. Lancet 2018, 391, 1830–1841. [Google Scholar] [CrossRef]
- Tau, G.Z.; Peterson, B.S. Normal development of brain circuits. Neuropsychopharmacology 2010, 35, 147–168. [Google Scholar] [CrossRef] [PubMed]
- Lyall, K.; Schmidt, R.J.; Hertz-Picciotto, I. Maternal lifestyle and environmental risk factors for autism spectrum disorders. Int. J. Epidemiol. 2014, 43, 443–464. [Google Scholar] [CrossRef] [PubMed]
- Linnet, K.M.; Dalsgaard, S.; Obel, C.; Wisborg, K.; Henriksen, T.B.; Rodriguez, A.; Kotimaa, A.; Moilanen, I.; Thomsen, P.H.; Olsen, J.; et al. Maternal lifestyle factors in pregnancy risk of attention deficit hyperactivity disorder and associated behaviors: review of the current evidence. Am. J. Psychiatry 2003, 160, 1028–1040. [Google Scholar] [CrossRef] [PubMed]
- Prado, E.L.; Dewey, K.G. Nutrition and brain development in early life. Nutr. Rev. 2014, 72, 267–284. [Google Scholar] [CrossRef] [PubMed]
- Bale, T.L.; Baram, T.Z.; Brown, A.S.; Goldstein, J.M.; Insel, T.R.; McCarthy, M.M.; Nemeroff, C.B.; Reyes, T.M.; Simerly, R.B.; Susser, E.S.; et al. Early life programming and neurodevelopmental disorders. Biol. Psychiatry 2010, 68, 314–319. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S. Impact of maternal diet on the epigenome during in utero life and the developmental programming of diseases in childhood and adulthood. Nutrients 2015, 7, 9492–9507. [Google Scholar] [CrossRef] [PubMed]
- Steegers-Theunissen, R.P.; Obermann-Borst, S.A.; Kremer, D.; Lindemans, J.; Siebel, C.; Steegers, E.A.; Slagboom, P.E.; Heijmans, B.T. Periconceptional maternal folic acid use of 400 microg per day is related to increased methylation of the IGF2 gene in the very young child. PLoS ONE 2009, 4, e7845. [Google Scholar] [CrossRef]
- Craciunescu, C.N.; Brown, E.C.; Mar, M.H.; Albright, C.D.; Nadeau, M.R.; Zeisel, S.H. Folic acid deficiency during late gestation decreases progenitor cell proliferation and increases apoptosis in fetal mouse brain. J. Nutr. 2004, 134, 162–166. [Google Scholar] [CrossRef]
- Jadavji, N.M.; Deng, L.; Malysheva, O.; Caudill, M.A.; Rozen, R. MTHFR deficiency or reduced intake of folate or choline in pregnant mice results in impaired short-term memory and increased apoptosis in the hippocampus of wild-type offspring. Neuroscience 2015, 300, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Pitkin, R.M. Folate and neural tube defects. Am. J. Clin. Nutr. 2007, 85, 285s–288s. [Google Scholar] [CrossRef] [PubMed]
- Yehuda, S.; Rabinovitz, S.; Mostofsky, D.I. Essential fatty acids are mediators of brain biochemistry and cognitive functions. J. Neurosci. Res. 1999, 56, 565–570. [Google Scholar] [CrossRef]
- Martinez, M. Tissue levels of polyunsaturated fatty acids during early human development. J. Pediatrics 1992, 120, S129–S138. [Google Scholar] [CrossRef]
- Lauritzen, L.; Brambilla, P.; Mazzocchi, A.; Harsløf, L.; Ciappolino, V.; Agostoni, C. DHA effects in brain development and function. Nutrients 2016, 8, 6. [Google Scholar] [CrossRef]
- McCann, J.C.; Ames, B.N. Is docosahexaenoic acid, an n-3 long-chain polyunsaturated fatty acid, required for development of normal brain function? An overview of evidence from cognitive and behavioral tests in humans and animals. Am. J. Clin. Nutr. 2005, 82, 281–295. [Google Scholar] [CrossRef] [PubMed]
- Carlson, S.E. Docosahexaenoic acid and arachidonic acid in infant development. Semin. Neonatol. 2001, 6, 437–449. [Google Scholar] [CrossRef]
- Fretham, S.J.; Carlson, E.S.; Georgieff, M.K. The role of iron in learning and memory. Adv. Nutr. 2011, 2, 112–121. [Google Scholar] [CrossRef]
- Skeaff, S.A. Iodine deficiency in pregnancy: The effect on neurodevelopment in the child. Nutrients 2011, 3, 265–273. [Google Scholar] [CrossRef]
- Porciúncula, L.O.; Sallaberry, C.; Mioranzza, S.; Botton, P.H.S.; Rosemberg, D.B. The Janus face of caffeine. Neurochem. Int. 2013, 63, 594–609. [Google Scholar] [CrossRef]
- Bougma, K.; Aboud, F.E.; Harding, K.B.; Marquis, G.S. Iodine and mental development of children 5 years old and under: A systematic review and meta-analysis. Nutrients 2013, 5, 1384–1416. [Google Scholar] [CrossRef] [PubMed]
- Borge, T.C.; Aase, H.; Brantsaeter, A.L.; Biele, G. The importance of maternal diet quality during pregnancy on cognitive and behavioural outcomes in children: A systematic review and meta-analysis. BMJ Open 2017, 7, e016777. [Google Scholar] [CrossRef] [PubMed]
- Freedman, R.; Hunter, S.K.; Hoffman, M.C. Prenatal primary prevention of mental illness by micronutrient supplements in pregnancy. Am. J. Psychiatry 2018, 175, 607–619. [Google Scholar] [CrossRef] [PubMed]
- Starling, P.; Charlton, K.; McMahon, A.T.; Lucas, C. Fish intake during pregnancy and foetal neurodevelopment—A systematic review of the evidence. Nutrients 2015, 7, 2001–2014. [Google Scholar] [CrossRef] [PubMed]
- Gould, J.F.; Smithers, L.G.; Makrides, M. The effect of maternal omega-3 (n-3) LCPUFA supplementation during pregnancy on early childhood cognitive and visual development: A systematic review and meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2013, 97, 531–544. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Li, K.; Zhao, D.; Li, L. The association between maternal use of folic acid supplements during pregnancy and risk of autism spectrum disorders in children: A meta-analysis. Mol. Autism 2017, 8, 51. [Google Scholar] [CrossRef] [PubMed]
- DeVilbiss, E.A.; Gardner, R.M.; Newschaffer, C.J.; Lee, B.K. Maternal folate status as a risk factor for autism spectrum disorders: A review of existing evidence. Br. J. Nutr. 2015, 114, 663–672. [Google Scholar] [CrossRef]
- Bayer, S.A.; Altman, J.; Russo, R.J.; Zhang, X. Timetables of neurogenesis in the human brain based on experimentally determined patterns in the rat. Neurotoxicology 1993, 14, 83–144. [Google Scholar]
- Centers for Disease Control and Prevention. Data & Statistics on Autism Spectrum Disorder. Available online: https://www.cdc.gov/ncbddd/autism/data.html (accessed on 5 June 2019).
- Centers for Disease Control and Prevention. Data & Statistics about ADHD. Available online: https://www.cdc.gov/ncbddd/adhd/data.html (accessed on 5 June 2019).
- Tierney, J.F.; Stewart, L.A.; Ghersi, D.; Burdett, S.; Sydes, M.R. Practical methods for incorporating summary time-to-event data into meta-analysis. Trials 2007, 8, 16. [Google Scholar] [CrossRef]
- StataCorp LP. Stata Statistical Software: Release 14. (computer program); StataCorp LP: College Station, TX, USA, 2015. [Google Scholar]
- Schmidt, R.J.; Iosif, A.M.; Angel, E.G.; Ozonoff, S. Association of maternal prenatal vitamin use with risk for autism spectrum disorder recurrence in young siblings. JAMA Psychiatry 2019, 76, 391–398. [Google Scholar] [CrossRef]
- Levine, S.Z.; Kodesh, A.; Viktorin, A.; Smith, L.; Uher, R.; Reichenberg, A.; Sandin, S. Association of maternal use of folic acid and multivitamin supplements in the periods before and during pregnancy with the risk of autism spectrum disorder in offspring. JAMA Psychiatry 2018, 75, 176–184. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.M.; Shen, Y.D.; Li, Y.J.; Xun, G.L.; Liu, H.; Wu, R.R.; Ou, J.J. Maternal dietary patterns, supplements intake and autism spectrum disorders: A preliminary case-control study. Medicine 2018, 97, e13902. [Google Scholar] [CrossRef] [PubMed]
- Raghavan, R.; Riley, A.W.; Volk, H.; Caruso, D.; Hironaka, L.; Sices, L.; Hong, X.; Wang, G.; Ji, Y.; Brucato, M.; et al. Maternal multivitamin intake, plasma folate and vitamin B12 levels and autism spectrum disorder risk in offspring. Paediatr. Perinat. Epidemiol. 2018, 32, 100–111. [Google Scholar] [CrossRef]
- Strom, M.G.C.; Lyall, K.; Ascherio, A.; Olsen, S.F. Research letter: Folic acid supplementation and intake of folate in pregnancy in relation to offspring risk of autism spectrum disorder. Psychol. Med. 2018, 48, 1048–1054. [Google Scholar] [CrossRef] [PubMed]
- DeVilbiss, E.A.; Magnusson, C.; Gardner, R.M.; Rai, D.; Newschaffer, C.J.; Lyall, K.; Dalman, C.; Lee, B.K. Antenatal nutritional supplementation and autism spectrum disorders in the Stockholm youth cohort: Population based cohort study. BMJ 2017, 359, j4273. [Google Scholar] [CrossRef] [PubMed]
- Virk, J.; Liew, Z.; Olsen, J.; Nohr, E.A.; Catov, J.M.; Ritz, B. Preconceptional and prenatal supplementary folic acid and multivitamin intake and autism spectrum disorders. Autism 2016, 20, 710–718. [Google Scholar] [CrossRef] [PubMed]
- Braun, J.M.; Froehlich, T.; Kalkbrenner, A.; Pfeiffer, C.M.; Fazili, Z.; Yolton, K.; Lanphear, B.P. Brief report: Are autistic-behaviors in children related to prenatal vitamin use and maternal whole blood folate concentrations? J. Autism Dev. Disord. 2014, 44, 2602–2607. [Google Scholar] [CrossRef]
- Nilsen, R.M.; Surén, P.; Gunnes, N.; Alsaker, E.R.; Bresnahan, M.; Hirtz, D.; Roth, C. Analysis of self-selection bias in a population-based cohort study of autism spectrum disorders. Paediatr. Perinat. Epidemiol. 2013, 27, 553–563. [Google Scholar] [CrossRef]
- Suren, P.; Roth, C.; Bresnahan, M.; Haugen, M.; Hornig, M.; Hirtz, D.; Lie, K.K.; Lipkin, W.I.; Magnus, P.; Reichborn-Kjennerud, T.; et al. Association between maternal use of folic acid supplements and risk of autism spectrum disorders in children. Jama 2013, 309, 570–577. [Google Scholar] [CrossRef]
- DeSoto, M.C.; Hitlan, R.T. Synthetic folic acid supplementation during pregnancy may increase the risk of developing autism. J. Pediatric Biochem. 2012, 2, 251–261. [Google Scholar]
- Schmidt, R.J.; Tancredi, D.J.; Ozonoff, S.; Hansen, R.L.; Hartiala, J.; Allayee, H.; Schmidt, L.C.; Tassone, F.; Hertz-Picciotto, I. Maternal periconceptional folic acid intake and risk of autism spectrum disorders and developmental delay in the CHARGE (Childhood Autism Risks from Genetics and Environment) case-control study. Am. J. Clin. Nutr. 2012, 96, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, R.J.; Hansen, R.L.; Hartiala, J.; Allayee, H.; Schmidt, L.C.; Tancredi, D.J.; Tassone, F.; Hertz-Picciotto, I. Prenatal vitamins, one-carbon metabolism gene variants, and risk for autism. Epidemiology 2011, 22, 476–485. [Google Scholar] [CrossRef] [PubMed]
- Steenweg-de Graaff, J.; Ghassabian, A.; Jaddoe, V.W.; Tiemeier, H.; Roza, S.J. Folate concentrations during pregnancy and autistic traits in the offspring. The Generation R Study. Eur. J. Public Health 2015, 25, 431–433. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, R.J.; Tancredi, D.J.; Krakowiak, P.; Hansen, R.L.; Ozonoff, S. Maternal intake of supplemental iron and risk of autism spectrum disorder. Am. J. Epidemiol. 2014, 180, 890–900. [Google Scholar] [CrossRef] [PubMed]
- Lyall, K.; Munger, K.L.; O’Reilly, E.J.; Santangelo, S.L.; Ascherio, A. Maternal dietary fat intake in association with autism spectrum disorders. Am. J. Epidemiol. 2013, 178, 209–220. [Google Scholar] [CrossRef]
- Steenweg-De Graaff, J.; Tiemeier, H.; Ghassabian, A.; Rijlaarsdam, J.; Jaddoe, V.W.V.; Verhulst, F.C.; Roza, S.J. Maternal fatty acid status during pregnancy and child autistic traits: The Generation R Study. Am. J. Epidemiol. 2016, 183, 792–799. [Google Scholar] [CrossRef]
- Gao, L.; Cui, S.S.; Han, Y.; Dai, W.; Su, Y.Y.; Zhang, X. Does periconceptional fish consumption by parents affect the incidence of autism spectrum disorder and intelligence deficiency? A Case-control study in Tianjin, China. Biomed. Environ. Sci. 2016, 29, 885–892. [Google Scholar] [CrossRef]
- Julvez, J.; Mendez, M.; Fernandez-Barres, S.; Romaguera, D.; Vioque, J.; Llop, S.; Ibarluzea, J.; Guxens, M.; Avella-Garcia, C.; Tardon, A.; et al. Maternal consumption of seafood in pregnancy and child neuropsychological development: A longitudinal study based on a population with high consumption levels. Am. J. Epidemiol. 2016, 183, 169–182. [Google Scholar] [CrossRef]
- House, J.S.; Mendez, M.; Maguire, R.L.; Gonzalez-Nahm, S.; Huang, Z.; Daniels, J.; Murphy, S.K.; Fuemmeler, B.F.; Wright, F.A.; Hoyo, C. Periconceptional maternal mediterranean diet is associated with favorable offspring behaviors and altered CpG methylation of imprinted genes. Front. Cell Dev. Biol. 2018, 6, 107. [Google Scholar] [CrossRef]
- Schmidt, R.J.; Ozonoff, S.; Hansen, R.; Hartiala, J.; Allayee, H.; Schmidt, L.; Tassone, F.; Hertz-Picciotto, I. (old record 6424) Maternal periconceptional folic acid intake and risk for developmental delay and autism spectrum disorder: A case-control study. Am. J. Epidemiol. 2012, 175, S126. [Google Scholar] [CrossRef][Green Version]
- Li, Y.-M.; Ou, J.-J.; Liu, L.; Zhang, D.; Zhao, J.-P.; Tang, S.-Y. Association between maternal obesity and autism spectrum disorder in offspring: A meta-analysis. J. Autism Dev. Disord. 2016, 46, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Ueland, P.M.; Hustad, S.; Schneede, J.; Refsum, H.; Vollset, S.E. Biological and clinical implications of the MTHFR C677T polymorphism. Trends Pharmacol. Sci. 2001, 22, 195–201. [Google Scholar] [CrossRef]
- Rijlaarsdam, J.; Cecil, C.A.; Walton, E.; Mesirow, M.S.; Relton, C.L.; Gaunt, T.R.; McArdle, W.; Barker, E.D. Prenatal unhealthy diet, insulin-like growth factor 2 gene (IGF2) methylation, and attention deficit hyperactivity disorder symptoms in youth with early-onset conduct problems. J. Child Psychol. Psychiatry 2017, 58, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Pidsley, R.; Dempster, E.; Troakes, C.; Al-Sarraj, S.; Mill, J. Epigenetic and genetic variation at the IGF2/H19 imprinting control region on 11p15. 5 is associated with cerebellum weight. Epigenetics 2012, 7, 155–163. [Google Scholar] [CrossRef] [PubMed]
- Office of Dietary Supplements; National Library of Medicine. Dietary Supplement Label Database. Available online: https://www.dsld.nlm.nih.gov/dsld/index.jsp (accessed on 7 February 2019).
- Stamm, R.; Houghton, L. Nutrient intake values for folate during pregnancy and lactation vary widely around the world. Nutrients 2013, 5, 3920–3947. [Google Scholar] [CrossRef] [PubMed]
- Wilcken, B.; Bamforth, F.; Li, Z.; Zhu, H.; Ritvanen, A.; Redlund, M.; Stoll, C.; Alembik, Y.; Dott, B.; Czeizel, A. Geographical and ethnic variation of the 677C> T allele of 5, 10 methylenetetrahydrofolate reductase (MTHFR): Findings from over 7000 newborns from 16 areas world wide. J. Med Genet. 2003, 40, 619–625. [Google Scholar] [CrossRef] [PubMed]
- Schmitz, G.; Ecker, J. The opposing effects of n-3 and n-6 fatty acids. Prog. Lipid Res. 2008, 47, 147–155. [Google Scholar] [CrossRef]
- Patterson, E.; Wall, R.; Fitzgerald, G.F.; Ross, R.P.; Stanton, C. Health implications of high dietary omega-6 polyunsaturated fatty acids. J. Nutr. Metab. 2012, 2012, 539426. [Google Scholar] [CrossRef]
- D’Souza, S.W.; Waldie, K.E.; Peterson, E.R.; Underwood, L.; Morton, S.M.B. Antenatal and postnatal determinants of behavioural difficulties in early childhood: Evidence from growing up in New Zealand. Child Psychiatry Hum. Dev. 2019, 50, 45–60. [Google Scholar] [CrossRef]
- Miyake, Y.T.K.; Okubo, H.; Sasaki, S.; Arakawa, M. Maternal B vitamin intake during pregnancy and childhood behavioral problems in Japan: The Kyushu Okinawa maternal and child health study. Nutr. Neurosci. 2018, 19, 1–8. [Google Scholar] [CrossRef]
- Virk, J.; Liew, Z.; Olsen, J.; Nohr, E.A.; Catov, J.M.; Ritz, B. Pre-conceptual and prenatal supplementary folic acid and multivitamin intake, behavioral problems, and hyperkinetic disorders: A study based on the Danish National Birth Cohort (DNBC). Nutr. Neurosci. 2018, 21, 352–360. [Google Scholar] [CrossRef] [PubMed]
- Julvez, J.; Fortuny, J.; Mendez, M.; Torrent, M.; Ribas-Fitó, N.; Sunyer, J. Maternal use of folic acid supplements during pregnancy and four-year-old neurodevelopment in a population-based birth cohort. Paediatr. Perinat. Epidemiol. 2009, 23, 199–206. [Google Scholar] [CrossRef] [PubMed]
- Schlotz, W.; Jones, A.; Phillips, D.I.; Gale, C.R.; Robinson, S.M.; Godfrey, K.M. Lower maternal folate status in early pregnancy is associated with childhood hyperactivity and peer problems in offspring. J. Child Psychol. Psychiatry 2010, 51, 594–602. [Google Scholar] [CrossRef] [PubMed]
- Abel, M.H.; Ystrom, E.; Caspersen, I.H.; Meltzer, H.M.; Aase, H.; Torheim, L.E.; Askeland, R.B.; Reichborn-Kjennerud, T.; Brantsaeter, A.L. Maternal iodine intake and offspring attention-deficit/hyperactivity disorder: Results from a large prospective cohort study. Nutrients 2017, 9, 1239. [Google Scholar] [CrossRef] [PubMed]
- Miyake, Y.T.K.; Okubo, H.; Sasaki, S.; Arakawa, M. Maternal fat intake during pregnancy and behavioral problems in 5-y-old Japanese children. Nutrition 2018, 50, 91–96. [Google Scholar] [CrossRef]
- Ramakrishnan, U.; Gonzalez-Casanova, I.; Schnaas, L.; DiGirolamo, A.; Quezada, A.D.; Pallo, B.C.; Hao, W.; Neufeld, L.M.; Rivera, J.A.; Stein, A.D.; et al. Prenatal supplementation with DHA improves attention at 5 years of age: A randomized controlled trial. Am. J. Clin. Nutr. 2016, 104, 1075–1082. [Google Scholar] [CrossRef] [PubMed]
- Gale, C.R.; Robinson, S.M.; Godfrey, K.M.; Law, C.M.; Schlotz, W.; O’Callaghan, F.J. Oily fish intake during pregnancy—Association with lower hyperactivity but not with higher full-scale IQ in offspring. J. Child Psychol. Psychiatry Allied Discip. 2008, 49, 1061–1068. [Google Scholar] [CrossRef]
- Hibbeln, J.R.; Davis, J.M.; Steer, C.; Emmett, P.; Rogers, I.; Williams, C.; Golding, J. Maternal seafood consumption in pregnancy and neurodevelopmental outcomes in childhood (ALSPAC study): An observational cohort study. Lancet 2007, 369, 578–585. [Google Scholar] [CrossRef]
- Miyake, Y.T.K.; Okubo, H.; Sasaki, S.; Arakawa, M. Maternal caffeine intake in pregnancy is inversely related to childhood peer problems in Japan: The Kyushu Okinawa maternal and child health study. Nutr. Neurosci. 2018, 13, 1–8. [Google Scholar] [CrossRef]
- Del-Ponte, B.; Santos, I.S.; Tovo-Rodrigues, L.; Anselmi, L.; Munhoz, T.N.; Matijasevich, A. Caffeine consumption during pregnancy and ADHD at the age of 11 years: A birth cohort study. BMJ Open 2016, 6, e012749. [Google Scholar] [CrossRef]
- Loomans, E.M.; Hofland, L.; van der Stelt, O.; van der Wal, M.F.; Koot, H.M.; Van den Bergh, B.R.; Vrijkotte, T.G. Caffeine intake during pregnancy and risk of problem behavior in 5- to 6-year-old children. Pediatrics 2012, 130, e305–e313. [Google Scholar] [CrossRef] [PubMed]
- Hvolgaard Mikkelsen, S.; Obel, C.; Olsen, J.; Niclasen, J.; Bech, B.H. Maternal caffeine consumption during pregnancy and behavioral disorders in 11-year-old offspring: A Danish National Birth Cohort study. J. Pediatrics 2017, 189, 120–127.e121. [Google Scholar] [CrossRef] [PubMed]
- Linnet, K.M.; Wisborg, K.; Secher, N.J.; Thomsen, P.H.; Obel, C.; Dalsgaard, S.; Henriksen, T.B. Coffee consumption during pregnancy and the risk of hyperkinetic disorder and ADHD: A prospective cohort study. Acta Paediatr. 2009, 98, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Galera, C.; Heude, B.; Forhan, A.; Bernard, J.Y.; Peyre, H.; Van der Waerden, J.; Pryor, L.; Bouvard, M.P.; Melchior, M.; Lioret, S.; et al. Prenatal diet and children’s trajectories of hyperactivity-inattention and conduct problems from 3 to 8 years: The EDEN mother-child cohort. J. Child Psychol. Psychiatry 2018, 59, 1003–1011. [Google Scholar] [CrossRef] [PubMed]
- McGee, R.A.; Clark, S.E.; Symons, D.K. Does the conners’ continuous performance test aid in ADHD diagnosis? J. Abnorm. Child Psychol. 2000, 28, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Thapar, A.; Cooper, M.; Jefferies, R.; Stergiakouli, E. What causes attention deficit hyperactivity disorder? Arch. Dis. Child. 2012, 97, 260–265. [Google Scholar] [CrossRef]
- Mathias, R.A.; Pani, V.; Chilton, F.H. Genetic Variants in the FADS gene: Implications for dietary recommendations for fatty acid intake. Curr. Nutr. Rep. 2014, 3, 139–148. [Google Scholar] [CrossRef]
- Symonds, M.E.; Sebert, S.P.; Hyatt, M.A.; Budge, H. Nutritional programming of the metabolic syndrome. Nat. Rev. Endocrinol. 2009, 5, 604–610. [Google Scholar] [CrossRef]
| Source (Country) | Design & Sample | Maternal Exposures | Offspring Outcomes | Findings | Covariates |
|---|---|---|---|---|---|
| Vitamins and Minerals | |||||
| Folate and Multivitamin-ASD Diagnosis or Cutoff | |||||
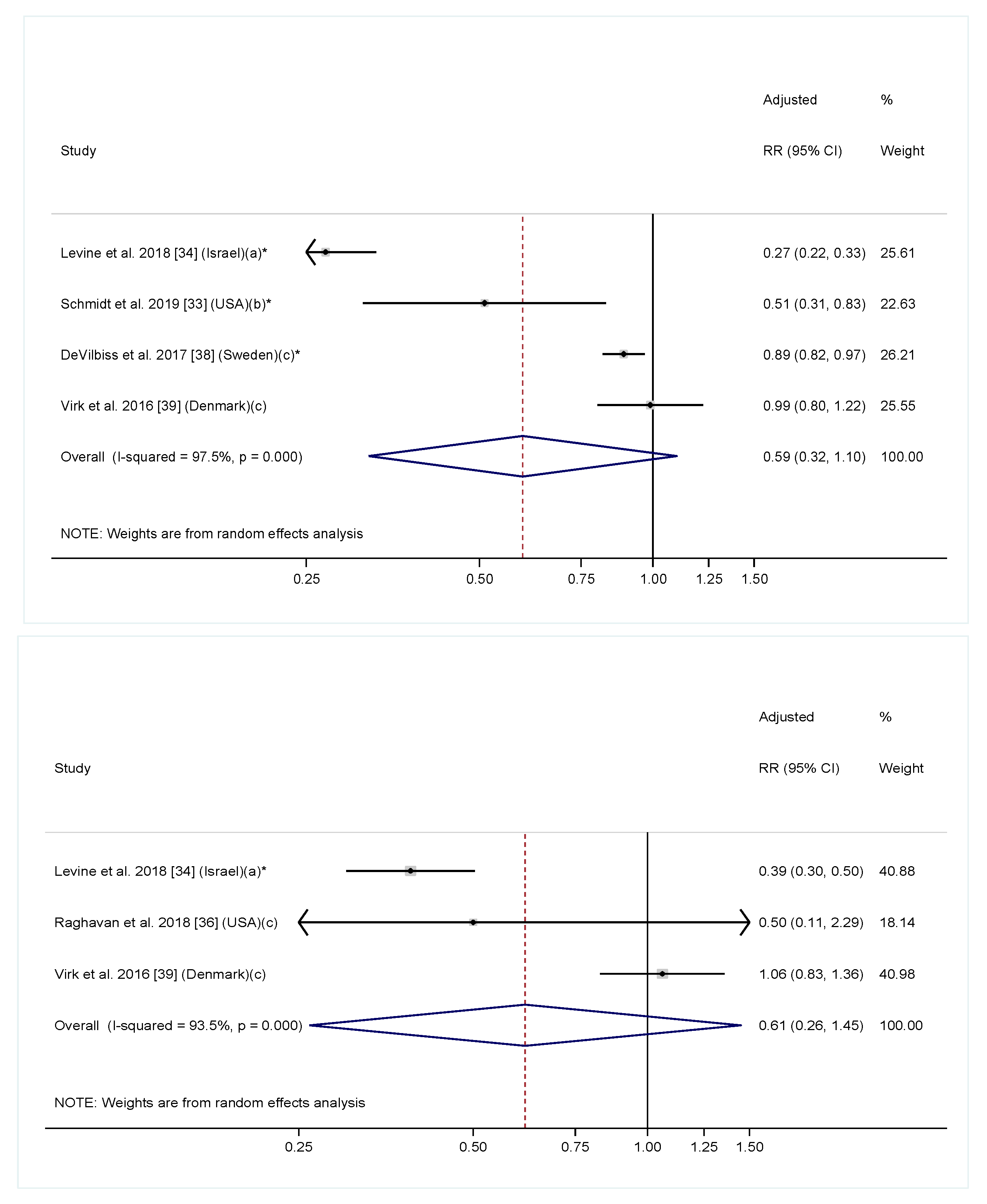
| Schmidt et al. 2019 (USA) [33] | Prospective cohort; 332 children who were younger sibling of children with ASD in the MARBLES study | Vitamin and supplement use for the 6 months preconception and each month during the pregnancy assessed in interviews in the first and second halves of pregnancy and after birth | ASD assessed by ADOS at 3 years of age | Prenatal vitamin in the first month of pregnancy was associated with lower ASD risk (RR = 0.50, 95% CI: 0.30 to 0.81) *. Folic acid supplement ≥600 mcg/day in the first month of pregnancy was associated with lower ASD risk (RR = 0.38, 95% CI: 0.16, 0.90) * | Maternal education. The folic acid model further adjusted for iron intake |
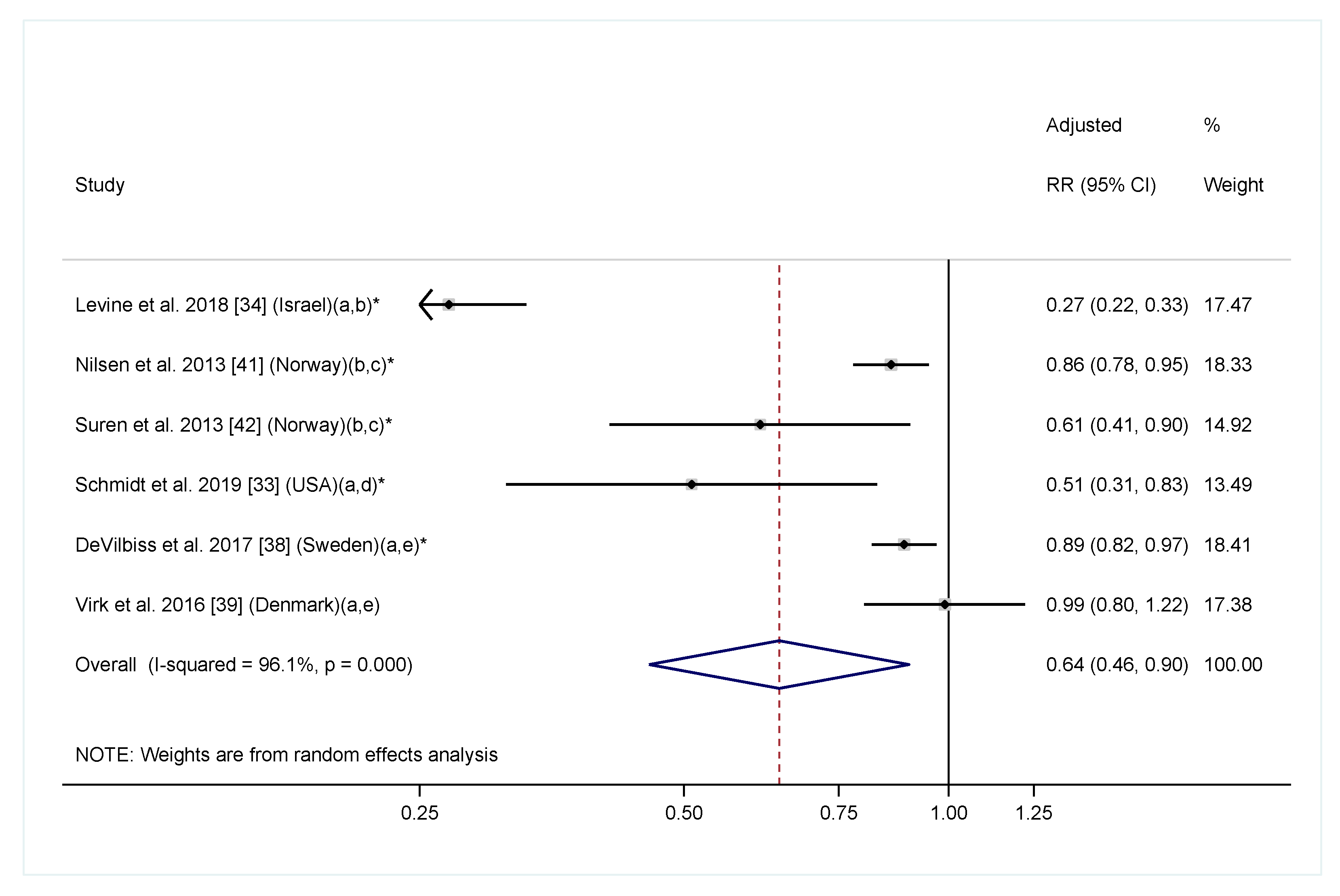
| Levine et al. 2018 (Israel) [34] | Prospective cohort; 45,300 children | Intake of folic acid and multivitamin supplements before and during pregnancy was coded using ATC from the prescription registry | ASD diagnosis identified from health care registers from the Meuhedet health care organization; children were 10 years old | Folic acid and/or multivitamin both before (RR = 0.39, 95% CI: 0.30, 0.50) * and during (RR = 0.27, 95% CI: 0.22, 0.33) * pregnancy were associated with lower risk of ASD. Results on folic acid supplements and multivitamin as two separate exposures were consistent with the main findings | sex, birth year, socioeconomic status, a maternal and paternal psychiatric diagnosis by childbirth, maternal and paternal age at childbirth, and parity |
| Li et al. 2018 (China) [35] | Retrospective case-control; 374 ASD and 354 TD in the ACED | Food preference and supplement use preconception and during pregnancy assessed 3–6 years after delivery | ASD identified from special education schools; TD identified from ordinary schools; children were 3–6 years of age | Maternal folic acid supplementation before pregnancy was not associated with ASD risk (OR = 0.95, 95% CI: 0.61, 1.50); Maternal folic acid supplementation during pregnancy was associated with lower ASD risk (OR = 0.64, 95% CI: 0.41, 1.00) * | Child’s and parental age, child’s gender, parental education, maternal BMI before conception and delivery, premature delivery, and intake of other supplements |
| Raghavan et al. 2018 (USA) [36] | Prospective cohort; 1257 children in the Boston Birth Cohort | Multivitamin supplement intake before pregnancy and in each trimester assessed 1–3 days after delivery; plasma folate and vitamin B12 levels measured 1–3 days after delivery | ASD identified from electronic medical records in Boston Medical Center | Before pregnancy, multivitamin supplement was not associated with ASD risk (HR = 0.5, 95% CI: 0.1, 2.1). In the first trimester, ≤2 (HR = 3.4, 95% CI: 1.6, 7.2) * and >5 (HR = 2.3, 95% CI: 1.2, 3.6) * times/week of multivitamin supplement were both associated with higher risk of ASD compared to 3–5 times/day. Findings are similar at second and third trimester. Very high serum folate (HRs [95% CI] for decile 1 and 10 vs. the rest were 1.2 [0.5, 2.8] and 2.5 [1.3, 4.6]) * and vitamin B12 (HRs [95% CI] for decile 1 and 10 vs. the rest were 0.7 [0.3, 1.7] and 2.5 [1.4, 4.5]) * concentrations were both associate with higher ASD risk. | Maternal age, education, parity, BMI, smoking status, diabetes status, race, and MTHFR genotype, offspring gestational age, sex, and year of birth |
| Strom et al. 2018 (Denmark) [37] | Prospective cohort; 92,676 children in the DNBC | Folic acid supplementation and folate intake from food in the previous 4 weeks assessed by FFQ at GW 25 | ASD cases identified in Danish Central Psychiatric Research Registry and the Danish National Patients Registry | Folic acid supplement at GW −4 to 8 was not associated with ASD risk (HR = 1.04, 95% CI: 0.94, 1.19). Folic acid supplement in mid pregnancy was not associated with ASD risk (HRs [95% CI] for <400 and ≥400 mcg/day vs. no intake was 1.01 [0.76, 1.34] and 0.98 [0.75, 1.29]). Folate from food was not associated with SD risk (HRs [95% CI] for quintile 2–5 were 0.82 [0.67, 1.01], 0.96 [0.78, 1.17], 0.85 [0.69, 1.04] and 0.94 [0.77, 1.16]) | Maternal age, paternal age, parity, maternal smoking during pregnancy, maternal education, family socioeconomic status, whether the pregnancy was planned, maternal prepregnancy BMI and sex of the child |
| DeVilbiss et al. 2017 (Sweden) [38] | Prospective cohort; 273,107 children in the Stockholm youth cohort | Supplement use at the first antenatal visit coded in ATC from medical birth registry | ASD identified from computerized registers covering all pathways of ASD diagnosis and care in Stockholm County; children were 4–15 years old | Multivitamin supplement was associated with lower risk of ASD compared to no vitamin/mineral supplement (OR = 0.89, 95% CI: 0.82, 0.97) *. Folic acid supplement alone (OR = 1.27, 95% CI: 1.01, 1.60), iron supplement alone (0.96, 95% CI: 0.90, 1.03) and combined folic acid and iron supplement (OR = 0.92, 0.83 to 1.02) were not associated with ASD risk | Child characteristics (sex, birth year, and years resided in Stockholm County), socioeconomic indicators (education, family income, and maternal birth country), maternal characteristics (age, BMI, parity, smoking status), medication use during pregnancy (antidepressants or antiepileptics), and maternal neuropsychiatric conditions (anxiety disorders, autism, bipolar disorder, depression, epilepsy, intellectual disability, non-affective psychotic disorders, and stress disorders) |
| Virk et al. 2016 (Denmark) [39] | Prospective cohort; 35,059 children in the DNBC | Folic acid and multivitamin supplementations from 4 weeks preconception to GW 8 assessed by questionnaire at GW 12 | ASD identified from National Hospital Register; children were 10 years of age | Folic acid supplement at GW −4 to 8 was not associated with ASD risk (RR = 1.06, 95% CI: 0.82, 1.36). Multivitamin supplement was not associated with ASD risk (RR = 1.00, 95% CI: 0.82, 1.22) | Maternal age; maternal smoking and alcohol consumption during pregnancy; household socioeconomic status-examined more but none changed estimates more than 5% |
| Braun et al. 2014 (USA) [40] | Prospective cohort; 209 children in the HOMA study | Current vitamin supplementation assessed by interviews at GW 14–39; whole blood folate concentrations measured at GW 11–21 | Autistic-behaviors assessed by SRS at 4–5 years of age; scores >60 were defined as abnormal | Vitamin supplementation was associated with lower risk of failed SRS test (weekly/daily vs. never/rare intake: OR = 0.26, 95% CI: 0.08, 0.89) *. Whole blood folate concentrations were not associated with ASD risk (OR per SD = 1.42, 95% CI: 0.81, 2.49) | Whole blood folate concentration, maternal age, race, education, household income, marital status, employment during pregnancy, insurance status, depressive symptoms, serum cotinine concentrations, food security, and fresh fruit/vegetable intake |
| Nilsen et al. 2013 (Norway) [41] | Prospective cohort; 89,836 children in the MoBa | Folic acid intake before and/or during pregnancy recorded in Medical Birth Registry of Norway | ASD identified in Norwegian Patient Registry; all children were 3 years of older | Folic acid supplement was associated with lower ASD risk (OR = 0.86, 95% CI: 0.78, 0.95) * | Year of birth, maternal age, paternal age, marital status, and parity, hospital size |
| Suren et al. 2013 (Norway) [42] | Prospective cohort; 85,176 children in the MoBa | Folic acid and other supplementations from 4 weeks preconception to GW 8 assessed by questionnaire at GW 12 | ASD identified through questionnaire screening at 3, 5 and 7 years, professional and parental referral, and the Norwegian Patient Registry when children were 3–10 years of age | Folic acid supplement was associated with lower ASD risk (OR = 0.61, 95% CI: 0.41, 0.90) *. Supplements with folic acid were associated with lower ASD risk, supplements without folic acid were not | Adjusted for year of birth, maternal education level, and parity |
| DeSoto et al. 2012 (USA) [43] | Retrospective case-control; 256 ASD and 752 TD in the Vaccine Safety Datalink project | Folic acid intake from prenatal vitamins | ASD identified based on medical record | Folic acid supplement/prenatal multivitamin was associated with higher ASD risk (OR = 2.34, 95% CI: 1.14, 4.82) * | Child and family characteristics (e.g., maternal age, birth weight, poverty ratio, birth order, breast feeding duration), maternal prenatal health care/seeking behavior (e.g., adequacy of prenatal care, cholesterol screen, pap smear, prenatal alcohol use, prenatal viral infections), and child medical conditions (e.g., anemia, pica) |
| Schmidt et al. 2012 (USA) [44] | Retrospective case-control; 429 ASD and 278 TD in CHARGE study | Folic acid intake from supplements and fortified cereals in each month from 3 months before conception to delivery assessed by interviews at 2–5 years after delivery | ASD identified from the California Regional Center System; matched TD identified from state birth files; children were 2 to 5 years old | Higher folic acid intake in the first trimester was associated with lower ASD risk (ORs [95% CI] for <500, 500–799, 800–1000 and >1000 mcg/day vs. no intake were 0.35 [0.10 1.24], 0.27 [0.06 1.36], 0.25 [0.05 1.22] and 0.18 [0.04 0.94]) * | Adjusted for maternal educational level, child’s birth year, and log-transformed total iron and vitamin E from supplements and cereals; estimates were not substantially different when further adjusted for log-transformed vitamin B-12, vitamin B-6, vitamin C, vitamin D, or calcium from supplements and cereals or when adjusted for prenatal vitamin use in the first month of pregnancy |
| Schmidt et al. 2011 (USA) [45] | Retrospective case-control; 288 Autism and 278 TD in CHARGE study | Vitamin use in each month from 3 months before conception to delivery assessed by interviews at 2–5 years after delivery | Autism identified from the California Regional Center System; matched TD identified from state birth files; children were 2 to 5 years old | Prenatal vitamin supplement was associated with lower autism risk (OR [95% CI] for any use was 0.61 [0.39, 0.97]; ORs [95% CI] for irregular or <4 days/week, 4 days/week-daily, and > daily vs. no use were 0.96 [0.18–5.0], 0.61 [0.38–0.98] and 0.51 [0.19–1.4], p-trend = 0.002) * Multivitamin supplement was not associated with ASD risk [OR 1.2, 95% CI [0.51, 2.60]). | Maternal education and the child’s year of birth |
| Folate and multivitamin-ASD traits | |||||
| Steenweg-de Graaff et al. 2015 (The Netherlands) [46] | Prospective cohort; 3893 children in the Generation R study | Plasma folate concentrations at GW 10–17; folic acid supplementation from preconception to early pregnancy assessed by questionnaire early in pregnancy | Autistic traits using the SRS short form at 6 years of age; scores >95th percentile defined as abnormality | Folic acid supplement starting before pregnancy (beta per SD = −0.042, 95% CI: −0.068, −0.017) *, before GW 10 (beta per SD = −0.041, 95% CI: −0.066, -0.016) *, and after GW 10 (beta per SD = −0.057, 95% CI: −0.089, −0.025) * in pregnancy were all associated with fewer autistic traits. Higher serum folate concentration was not associated with fewer autistic traits (beta per SD = −0.007, 95% CI: −0.016, 0.001), nor the risk of being a probable ASD case (OR per SD = 1.03, 95% CI: 0.76, 1.39). | Gestational age at venipuncture, gender and age of the child, maternal psychopathology, education and family income |
| Iron-ASD diagnosis or cutoff | |||||
| Schmidt et al. 2019 (USA) [33] | Prospective cohort; 332 children who were younger sibling of children with ASD in the MARBLES | Vitamin and supplement use for the 6 months preconception and each month during the pregnancy assessed in interviews in the first and second halves of pregnancy and after birth | ASD assessed by ADOS at 3 years of age | Iron supplement in the first month of pregnancy was not associated with ASD risk (RR = 1.47, 95% CI: 0.61, 3.55) | Maternal education and folic acid supplement |
| DeVilbiss et al. 2017 (Sweden) [38] | Prospective cohort; 273,107 children in the Stockholm Youth Cohort | Supplement use at the first antenatal visit coded in ATC from medical birth registry | ASD identified from computerized registers covering all pathways of ASD diagnosis and care in Stockholm County; children were 4–15 years old | Folic acid supplement alone (OR = 1.27, 95% CI: 1.01, 1.60), iron supplement alone (OR = 0.96, 95% CI: 0.90 to 1.03) and combined folic acid and iron supplement (OR = 0.92, 95% CI: 0.83, 1.02) were not associated with ASD risk | Child characteristics (sex, birth year, and years resided in Stockholm County), socioeconomic indicators (education, family income, and maternal birth country), maternal characteristics (age, body mass index, parity, smoking status), medication use during pregnancy (antidepressants or antiepileptics), and maternal neuropsychiatric conditions (anxiety disorders, autism, bipolar disorder, depression, epilepsy, intellectual disability, non-affective psychotic disorders, and stress disorders) |
| Schmidt et al. 2014 (USA) [47] | Retrospective case-control; 520 ASD and 346 TD in CHARGE study | Iron supplementation in each month from 3 months before conception to delivery assessed by interviews at 2–5 years after delivery | ASD identified from the California Regional Center System; matched TD identified from state birth files; children were 2 to 5 years old | Iron-specific supplement before and during pregnancy was not associated with ASD risk (ORs [95% CI] for gestational months −3 to 9 were 0.89 [0.19, 4.13], 0.89 [0.19, 4.13], and 0.78 [0.21, 2.84], 0.67 [0.29, 1.55], 0.72 [0.37, 1.39], 0.71 [0.40, 1.27], 0.64 [0.39, 1.04], 0.65 [0.41, 1.04], 0.66 [0.43, 1.02], 0.73 [0.48, 1.13], 0.77 [0.49, 1.20] and 0.64 [0.39, 1.03]). Total iron supplement before and during pregnancy were not associated with ASD risk | Maternal folic acid intake, home ownership, child’s birth year. The model on total iron supplement was adjusted for folic acid |
| Calcium-ASD diagnosis or cutoff | |||||
| Li et al. 2018 (China) [35] | Retrospective case-control; 374 ASD and 354 TD in the ACED | food preference and supplement use preconception and during pregnancy assessed 3–6 years after delivery | ASD identified from special education schools; TD identified from ordinary schools; children were 3–6 years of age | Maternal calcium supplementation before pregnancy was associated with lower ASD risk (OR = 0.48, 95% CI: 0.28, 0.84) *; during pregnancy, it was not associated with ASD risk (OR = 1.11, 95% CI: 0.70, 1.75) | Child’s and parental age, child’s gender, parental education, maternal BMI before conception and delivery, premature delivery, and intake of other supplement |
| PUFA and seafood | |||||
| PUFA-ASD diagnosis or cutoff | |||||
| Lyall et al. 2013 (USA) [48] | Prospective cohort; 18,045 children in NHSII | Fat intake before pregnancy reported in the year assessed by FFQ after delivery | Autism, Asperger Syndrome, or PDD diagnosis reported by mothers at 4 years of age | Higher total PUFA from food (RRs [95% CI] for quartile 2–4 were 0.74 [0.55, 1.00], 0.73 [0.54, 0.99] and 0.77 [0.47, 1.26], p-trend = 0.05) * was associated with lower ASD risk. Total n-3 PUFA (0.98 [0.73, 1.32], 0.78 [0.57, 1.06] and 0.90 [0.66, 1.22], p-trend = 0.12), ALA (0.91 [0.67, 1.23], 0.86 [0.64, 1.16] and 0.80 [0.58, 1.08], p-trend = 0.14), EPA (1.11 [0.80, 1.54], 1.13 [0.79, 1.61] and 1.07 [0.76, 1.51], p-trend = 0.97), and DHA (1.05 [0.78, 1.42], 0.95 [0.69, 1.31] and 1.07 [0.79, 1.45], p-trend = 0.74) from food were not associated with ASD risk. Total n-6 PUFA (1.01 [0.75, 1.36], 1.01 [0.75, 1.36] and 0.66 [0.47, 0.92], p-trend = 0.01) * and LA (1.01 [0.76, 1.35], 0.86 [0.64, 1.16], 0.86 [0.64, 1.16] and 0.66 [0.48, 0.92], p-trend = 0.008) * from food were associated with lower ASD risk; AA (0.98 [0.73, 1.32], 0.78 [0.57, 1.06] and 0.79 [0.58, 1.09], p-trend = 0.09) were not associated with ASD risk | Adjusted for total energy intake, maternal age, child’s year of birth, income level, race, body mass index, and prepregnancy smoking status. Removal of adjustment for smoking did not affect results. Additional adjustment for intake of protein, whole grains, alcohol, fruit, and vegetables, as well as for multivitamin use, physical activity, child birth order, and maternal pregnancy complications did not materially alter the results |
| PUFA-ASD traits | |||||
| Steenweg-De Graaff et al. 2016 (The Netherlands) [49] | Prospective cohort; 4624 children in the Generation R study | Plasma fatty acid profiles measured before GW 25 | Autistic traits assessed by SRS at 6 years of age | N-3 PUFA percentage was not associated with autistic trait (beta per SD = −0.002, 95% CI: −0.011, 0.006). Higher n-6 PUFA percentage was associated with fewer autistic traits (beta per SD = 0.011, 95% CI: 0.002, 0.020) *. Higher n-3 to n-6 ratio was associated with fewer autistic traits (beta per SD = −0.009, 95% CI: −0.017, −0.001) * | Gestational age at venipuncture, sex, and age of the child at assessment, maternal IQ, prepregnancy body mass index, educational level, national origin, age at enrollment, psychopathology score in mid-pregnancy, smoking, alcohol consumption, and folic acid supplement use during pregnancy, family income, child day-care attendance, and paternal educational level, national origin, and psychopathology score |
| Seafood-ASD diagnosis or cutoff | |||||
| Gao et al. 2016 (China) [50] | Retrospective case-control; 108 ASD and 108 TD | Fish intake 6 months before pregnancy until delivery, assessed by FFQ 4–17 years after delivery | ASD identified from the registry of special education schools; matched TD identified from ordinary schools; children were 4–17 years old | Maternal no habit of eating grass carp was associated with higher risk of ASD (OR = 3.59, 95% CI: 1.22, 10.51) * | Maternal habit of eating grass carp, parental habit of eating hairtail, income level at childbirth, current income level. Paternal education, maternal education; matched on child age and sex |
| Lyall et al. 2013 (USA) [48] | Prospective cohort; 18,045 children in NHSII | Fish intake before pregnancy reported in the year assessed by FFQ after delivery | Autism, Asperger Syndrome, or PDD diagnosis reported by mothers at 4 years of age | Fish intake was not associated with ASD risk (RR [95% CI] for <1, 1 and >1 time/week were 1.10 [0.73, 1.66], 0.99 [0.65, 1.50] and 1.02 [0.59, 1.75]) | Adjusted for total energy intake, maternal age, child’s year of birth, income level, race, body mass index, and prepregnancy smoking status. Removal of adjustment for smoking did not affect results. Additional adjustment for intake of protein, whole grains, alcohol, fruit, and vegetables, as well as for multivitamin use, physical activity, child birth order, and maternal pregnancy complications did not materially alter the results |
| Seafood-ASD traits | |||||
| Julvez et al. 2016 (Spain) [51] | Prospective cohort; 1589 children in the INMA study | Seafood intake in the first trimester assessed by interviews with FFQ at GW 10–13 | Autism spectrum traits assessed by the Childhood Asperger Syndrome Test based on parent report at 5 years of age | Higher total seafood intake was associated with fewer autistic traits (beta [95% CI] for quintile 2–5 vs. 1 were −0.42 [−0.90, 0.07], −0.45 [−0.95, 0.05], -0.61 [−1.12, −0.11], and −0.55 [−1.06, −0.04], p-trend = 0.04) *. Large fatty fish and lean fish were both associated with fewer autistic traits, whereas small fatty fish and shellfish were not associated | Sex of the child, age during testing, cohort, quality of the test, and maternal energy intake during pregnancy, child’s birth weight, gestational age, duration of breastfeeding, maternal age, educational level, social class, prepregnancy body mass index, parity, and country of origin/birth |
| Steenweg-De Graaff et al. 2016 (The Netherlands) [49] | Prospective cohort; 4624 children in the Generation R study | Fish intake in the past 3 months assessed by FFQ in early pregnancy | Autistic traits assessed by SRS at 6 years of age | Fish intake was not associated with autistic trait (beta = −0.022, 95% CI: −0.055, 0.010) | Gestational age at venipuncture, sex, and age of the child at assessment, maternal IQ, prepregnancy body mass index, educational level, national origin, age at enrollment, psychopathology score in mid-pregnancy, smoking, alcohol consumption, and folic acid supplement use during pregnancy, family income, child day-care attendance, and paternal educational level, national origin, and psychopathology score |
| Fish oil-ASD diagnosis or cutoff | |||||
| Suren et al. 2013 (Norway) [42] | Prospective cohort; 85,176 children in the MoBa | Folic acid and other supplementations from GW −4 to 8 weeks assessed by questionnaire at GW 12 | ASD identified through questionnaire screening at 3, 5 and 7 years, professional and parental referral, and the Norwegian Patient Registry when children were 3–10 years of age | Fish oil supplement at GW −4 to 8 was not associated with ASD risk (OR = 1.29, 95% CI: 0.88, 1.89) | Adjusted for year of birth, maternal education level, and parity |
| Fruit | |||||
| Fruit-ASD diagnosis or cutoff | |||||
| Gao et al. 2016 (China) [50] | Retrospective case-control; 108 ASD and 108 TD | Fruit intake 6 months before pregnancy until delivery, assessed by FFQ 4–17 years after delivery | ASD identified from the registry of special education schools; matched TD identified from ordinary schools; children were 4–17 years old | Maternal no habit of eating fruits was associated with higher risk of ASD (OR = 2.42, 95% CI: 1.24, 4.73) * | maternal habit of eating grass carp, parental habit of eating hairtail, income level at childbirth, current income level. Paternal education, maternal education; frequency matched on child age and sex |
| Dietary patterns | |||||
| Dietary patterns-ASD diagnosis or cutoff | |||||
| House et al. 2018 (USA) [52] | Prospective cohort; 325 children in the NEST study | adherence to Mediterranean diet periconception assessed by FFQ in the first trimester of pregnancy or at enrollment | ASD index from ITSEA administered by a parent, caregiver or staff at 1–2 years of age | Adherence to Mediterranean diet was associated with lower ASD risk (ORs [95% CI] for tertile 2 and 3 vs. 1 were 0.46 [0.23, 0.90] and 0.35 [0.15, 0.80]) *. However, the trend was not significant after FDA adjustment (p = 0.09) | Breastfeeding at least 3 months, age of child at behavioral assessment, maternal fiber intake, total calories, folate, education, diabetes, obesity, smoking, and age, as well as paternal age and child parity, premature birth, weight, race, and child sex |
| Li et al. 2018 (China) [35] | Retrospective case-control; 374 ASD and 354 TD in the ACED | “Mostly meat”, “mostly vegetable” or “both meat and vegetable” dietary patterns assessed in questionnaires 3–6 years after delivery | ASD identified from special education schools; TD identified from ordinary schools; children were 3–6 years of age | Before pregnancy, maternal mostly meat (OR = 4.01, 95% CI:1.08, 14.89) * and mostly vegetable dietary pattern (OR = 2.23, 95% CI: 1.01, 4.95) * were both associated with higher ASD risk compared to both meat and vegetable dietary pattern. During pregnancy, they were not associated with ASD risk (ORs [95% CI] were 1.36 [0.29, 6.32] and 1.20 [0.53, 2.68]) | Child’s and parental age, child’s gender, parental education, maternal BMI before conception and delivery, premature delivery, and other maternal dietary patterns |
| Source (Country) | Design & Sample | Maternal Exposures | Offspring Outcomes | Findings | Covariates |
|---|---|---|---|---|---|
| Vitamins and Minerals | |||||
| Folate and Multivitamin-ADHD Diagnosis or Cutoff | |||||
| D’Souza et al. 2019 (New Zealand) [63] | Prospective cohort; 6246 children in the Growing Up in New Zealand Study | Folic acid and multivitamin supplementation before pregnancy, during the first trimester, and after the first trimester assessed in interviews in late pregnancy | Hyperactivity-inattention symptoms assessed by SDQ using mothers’ report at 2 years of age; clinical cutoff was used to define abnormality | Folic acid intake was not associated with ADHD risk (ORs [95% CI] for first trimester only and no intake vs. intake both before pregnancy and at first trimester were 0.98 [0.74, 1.31] and 0.88 [0.57, 1.34]). Multivitamin was not associated with ADHD risk (OR = 0.97, 95% CI: 0.75, 1.24) | Mother’s ethnicity, mother’s education, mother’s age when pregnant, child’s gestational age, child’s birth weight, child’s gender, parity, planned pregnancy, mother in paid employment, area-level deprivation, and rurality |
| Miyake et al. 2018 (Japan) [64] | Prospective cohort; 1199 children in the KOMCHS | Folate and other B-vitamin intake from food in the past month assessed by FFQ at GW 5 to 39 | Hyperactivity-inattention symptoms assessed by SDQ using mothers’ report at 5 years of age; clinical cutoff was used to define abnormality | Folate from food was not associated with hyperactivity-inattention problem (ORs [95% CI] for quartile 2–4 were 0.75 [0.46, 1.21], 0.66 [0.40, 1.07], and 0.69 [0.42, 1.12], p-trend = 0.10). Vitamin B12 (ORs [95% CI] for quartile 2–4 were 0.80 [0.49, 1.29], 0.99 [0.61, 1.61] and 0.81 [0.50, 1.32], p-trend = 0.60) and B2 (ORs for quartile 2–4 were 1.09 [0.68, 1.75], 1.03 [0.64, 1.66] and 0.61 [0.36, 1.03], p-trend = 0.08) from food were not associated hyperactivity-inattention problem. Higher vitamin B6 from food was associated with lower risk of hyperactivity-inattention problem (ORs for quartile 2–4 were 0.76 [0.48, 1.21], 0.58 [0.36, 0.94] and 0.57 [0.34, 0.94], p-trend = 0.01) * | Maternal age, gestation at baseline, region of residence at baseline, number of children at baseline, maternal and paternal education, household income, maternal depressive symptoms during pregnancy, maternal alcohol intake during pregnancy, maternal vitamin B complex supplement use during pregnancy, maternal smoking during pregnancy, child’s birth weight, child’s sex, breastfeeding duration, and smoking in the household during the first year of life. |
| Virk et al. 2018 (Denmark) [65] | Prospective cohort; 35,059 children in the DNBC | Folic acid and multivitamin supplementations from GW −4 to 8 assessed by questionnaire at GW 12 | Hyperkinetic disorder and treatment for ADHD were identified from National Patient Register; children were 7 years of age. Hyperactivity-inattention symptoms assessed by SDQ at age 7 years based on parent reports, and a score ≥7 was defined as abnormal | Folic acid supplement was not associated with risk of hyperkinetic disorder diagnosis (HR = 0.87, 95% CI: 0.54, 1.41) or ADHD medication (HR = 0.96, 95% CI: 0.68, 1.37). Maternal multivitamin use was associated with lower risk of hyperkinetic disorder diagnosis (HR = 0.70, 95% CI: 0.52, 0.96) *, ADHD medication (HR = 0.78, 95% CI: 0.62, 0.98) * | Maternal age, household socio-economic status, maternal smoking and alcohol consumption during pregnancy, maternal prepregnancy body mass index, birth year, and offspring sex |
| Julvez et al. 2009 (Spain) [66] | Prospective cohort; 420 children in the Menorca cohort | Current folic acid and vitamin supplementations assessed by interviews at GW 12 | ADHD assessed by ADHD Rating Scale-IV based on teacher report at 4 years of age; scores >80th percentile was defined abnormal | Folic acid with or without other vitamins compared to no folic acid or vitamins was not associated with ADHD risk (OR = 0.74, 95% CI: 0.38, 1.47). Vitamins without folic acid compared to no folic acid or vitamins was not associated with ADHD risk (OR = 0.26, 95% CI: 0.05, 1.31) | Parental social class and level of education, mother’s parity at child’s age four, mother’s marital status, maternal tobacco smoking during pregnancy, maternal intake of supplementary calcium and iron at the same time as study determinants, gestational age at interview, child’s gender, child’s duration of breast feeding, child’s age and school season during test assessment, evaluator and child’s home location at age four |
| Folate and multivitamin-ADHD symptoms | |||||
| Schlotz et al. 2010 (UK) [67] | Prospective cohort; 139 children | Total folate intake from foods and supplements during early pregnancy assessed by FFQ at GW 14, and during late pregnancy assessed at GW 28 | Hyperactivity-inattention symptoms assessed by SDQ based on mothers’ report at 8 years | Maternal red cell folate concentration (beta per SD = −1.23, 95% CI: −2.20, −0.26) * and total folate intake from food and supplements (beta per SD = −0.75, 95% CI: −1.39, −0.11) * in early pregnancy were both associated with fewer hyperactivity-inattention symptoms. However, total folate intake from food and supplements in late pregnancy (beta per SD = 0.07, 95% CI: −0.80, 0.93) was not associated with hyperactivity-inattention symptoms | Analysis of red cell folate: child’s sex, mother’s smoking and drinking alcohol during pregnancy, and mother’s educational attainment. Analysis of total folate intake: daily energy, child’s sex |
| Iodine-ADHD diagnosis or cutoff and ADHD symptoms | |||||
| Abel et al. 2017 (Norway) [68] | Prospective cohort; 77,164 children in the MoBa | Iodine intake from foods and supplements assessed by FFQ at GW 22 | ADHD identified from Norwegian Patient Registry; ADHD symptom assessed by the ADHD Rating Scale based on mother report at 8 years | Iodine from food was not associated with ADHD diagnosis (p-overall = 0.89). Iodine supplement was not associated with ADHD diagnosis, irrespective of food iodine intake. However, higher iodine from food was associated with fewer ADHD symptoms (beta [95% CI] for 25, 50, 75, 100, 125, 200, 225, 250, 300, 350, 400 vs. 160 mcg/day were 0.05 [−0.02, 0.12], 0.06 [0.01, 0.10], 0.06 [0.03, 0.09], 0.05 [0.02, 0.09], 0.03 [0.01, 0.05], −0.01 [-0.03, −0.00], -0.02 [-0.04, 0.01], −0.01 [-0.05, 0.02], −0.01 [-0.07, 0.05], −0.01 [−0.09, 0.08] and −0.00 [−0.12, 0.11], p-overall = 0.001) *. Higher iodine supplement was associated with higher ADHD score among women with less than 160 mcg/day of food iodine (beta [95% CI] for 1–200 and >200 mcg/day were 0.06 [0.03, 0.10] and 0.06 [−0.03, 0.16]) *, but not among women with more than 160 mcg/day of food iodine | Sibling clusters, total energy intake, maternal age, BMI, parity, education, smoking in pregnancy, and fiber intake |
| PUFA and Seafood | |||||
| PUFA-ADHD Diagnosis or Cutoff | |||||
| Miyake et al. 2018 (Japan) [69] | Prospective cohort; 1199 children in the KOMCHS | Fat intake from food in the past month assessed by FFQ at GW 5–39 | Hyperactivity-inattention symptoms assessed by SDQ using mothers’ report at 5 years of age; clinical cutoff was used to define abnormality | Total n-3 PUFA (ORs [95% CI] for quartile 2–4 were 0.82 [0.51, 1.31], 0.75 [0.46, 1.22], and 0.80 [0.49, 1.29], p-trend = 0.31), ALA (0.93 [0.59, 1.49], 0.73 [0.44, 1.20], and 0.72 [0.44, 1.18], p-trend = 0.13), EPA (0.90 [0.55, 1.46], 0.92 [0.56, 1.49], and 0.97 [0.59, 1.59], p-trend = 0.91), and DHA (0.85 [0.52, 1.39], 1.01 [0.62, 1.65], and 1.07 [0.66, 1.73], p-trend = 0.66) were not associated with hyperactivity-inattention problem. Total n-6 PUFA (1.05 [0.66, 1.68], 0.75 [0.45, 1.23], and 0.81 [0.49, 1.33], p-trend = 0.22), LA (1.03 [0.65, 1.64], 0.67 [0.40, 1.11], and 0.81 [0.49, 1.32], p-trend = 0.18), and AA (0.92 [0.56, 1.51], 1.10 [0.69, 1.77], and 0.91 [0.55, 1.50], p-trend = 0.92) were not associated with hyperactivity-inattention problem. Total n-3 to n-6 ratio (1.34 [0.83, 2.18], 1.24 [0.75, 2.04], and 0.97 [0.58, 1.63], p-trend = 0.84) was not associated with hyperactivity-inattention problem | Maternal age, gestation at baseline, region of residence at baseline, number of children at baseline, maternal and paternal education, household income, maternal depressive symptoms during pregnancy, maternal alcohol intake during pregnancy, maternal vitamin B complex supplement use during pregnancy, maternal smoking during pregnancy, child’s birth weight, child’s sex, breastfeeding duration, and smoking in the household during the first year of life |
| PUFA-ADHD symptoms | |||||
| Ramakrishnan et al. 2016 (Mexico) [70] | Randomized controlled trial; 797 children in POSGRAD study | Interventions of 400 mg of DHA supplementation or placebo from GW 18–22 to delivery | Hyperactivity-inattention symptoms assessed by K-CPT at 5 years of age. >70th percentile was at clinical risk of suffering from a disorder such as ADHD | DHA supplement of 400 mg/day was not associated with overall K-CPT score >70 (7.2% and 8.1%, p = 0.62) | None |
| Seafood-ADHD diagnosis or cutoff | |||||
| Gale et al. 2008 (UK) [71] | Prospective cohort; 219 children | Fish intake in the past 3 months assessed by FFQ at GW 15 and 32 | Hyperactivity-inattention symptoms assessed by SDQ using mothers’ report at 9 years of age; clinical cutoff was used to define abnormality | More frequent oily fish intake in early pregnancy (ORs [95% CI] for <1 and ≥1 time/week vs. no intake were 0.30 [0.12, 0.76] and 0.41 [0.15, 1.12]) * and late pregnancy (ORs [95% CI] for <1 and ≥1 time/week vs. no intake were 0.40 [0.16, 0.98] and 0.72 [0.26, 1.98]) * were both associated with lower risk of hyperactivity-inattention problem. Frequency of eating all types of fish was not associated with hyperactivity-inattention problem | Maternal social class, educational qualifications, age, IQ, smoking and drinking in pregnancy, duration of breastfeeding and birthweight |
| Hibbeln et al. 2007 (UK) [72] | Prospective cohort; 8946 children in ALSPAC | Seafood intake during pregnancy assessed by FFQ at GW 32 | Hyperactivity-inattention symptoms assessed by SDQ using mothers’ report at 7 years of age; highest quartile was defined as suboptimal outcome | Seafood intake was not associated with hyperactivity-inattention problem (ORs [95% CI] for none and 1–340 g/week vs. ≥340 g/week were 1.13 [0.84, 1.53] and 0.91 [0.73, 1.12], p-trend = 0.66) | Maternal education, housing, crowding at home, life events, partner, maternal age, maternal smoking in pregnancy, maternal alcohol use in pregnancy, parity, breastfeeding, gender, ethnic origin, birthweight, preterm delivery, 12 non-fish food groups |
| Caffeine, coffee and tea | |||||
| Caffeine-ADHD diagnosis or cutoff | |||||
| Miyake et al. 2018 (Japan) [73] | Prospective cohort; 1199 children in the KOMCHS | Caffeine intake from food in the past month assessed by FFQ at GW 5–39 | Hyperactivity-inattention symptoms assessed by SDQ using mothers’ report at 5 years of age; clinical cutoff was used to define abnormality | Caffeine was not associated with hyperactivity-inattention problem (ORs [95% CI] for quartile 2–4 were 1.04 [0.64, 1.68], 0.99 [0.61, 1.62], and 0.84 [0.51, 1.38], p-trend = 0.49) | Maternal age, gestation at baseline, region of residence at baseline, number of children at baseline, maternal and paternal education, household income, maternal depressive symptoms during pregnancy, maternal alcohol intake during pregnancy, maternal vitamin B complex supplement use during pregnancy, maternal smoking during pregnancy, child’s birth weight, child’s sex, breastfeeding duration, and smoking in the household during the first year of life |
| Del-Ponte et al. 2016 (Brazil) [74] | Prospective cohort; 3485 children | Caffeine intake during each trimester assessed in interviews after delivery | ADHD assessed by DAWBA based on mother’s report at 11 years of age; clinical cutoff was used to define abnormality | Caffeine in the entire pregnancy was not associated with ADHD risk (ORs [95% CI] for 100–299 and ≥300 vs. <100 mg/day were 1.12 [0.68 to 1.84] and 0.90 [0.51 to 1.59]). Similar results were found in each of the three trimesters | Maternal mood symptoms during pregnancy, National Economic Index, paternal education level and maternal conjugal situation |
| Loomans et al. 2012 (The Netherlands) [75] | Prospective cohort; 3439 children in the ABCD study | Caffeine intake from coffee, tea and cola in the past week assessed by questionnaire at GW 16 | Hyperactivity-inattention symptoms assessed by SDQ based on mothers’ report at 5–6 years of age; clinical cutoff was used to define abnormality | Caffeine was not associated with hyperactivity-inattention problem (ORs [95% CI] for 86–255, 256–425, and >425 vs. 0–85 mg/day were 0.94 [0.68, 1.31], 0.87 [0.57, 1.33], and 1.08 [0.55, 2.12]) | Maternal age, ethnicity, maternal education, maternal anxiety, cohabitant status, smoking, alcohol, child’s gender, family size |
| Coffee and tea-ADHD diagnosis or cutoff | |||||
| Hvolgaard Mikkelsen et al. 2017 (Denmark) [76] | Prospective cohort; 47,491 children in the DNBC | Current coffee and tea intake assessed by interviews at GW 15 and 30 | Hyperactivity-inattention symptoms assessed by SDQ based on children, parents and teachers’ report at 11 years of age; computerized algorithms were used to identified ADHD | Higher coffee intake in the first trimester was associated with higher risk of hyperactivity-inattention problem (ORs [95% CI] for 1–3, 4–7 and ≥8 cups/day vs. no intake were 0.97 [0.88, 1.08], 1.09 [0.93, 1.27] and 1.47 [1.18, 1.83], p-trend = 0.03) *. In the third trimester, it was not associated (ORs [95% CI] for 1–3, 4–7 and ≥8 cups/day vs. no intake were 0.94 [0.85, 1.04], 0.96 [0.83, 1.13] and 1.21 [0.95, 1.55], p-trend = 0.88). Tea intake in the first trimester was not associated with hyperactivity-inattention problem (ORs [95% CI] for 1–3, 4–7 and ≥8 cups/day vs. no intake were 0.93 [0.85, 1.03], 0.97 [0.85, 1.12] and 1.21 [0.98, 1.49], p-trend = 0.57). In the third trimester, higher tea intake was associated with lower risk of hyperactivity-inattention problem (ORs [95% CI] for 1–3, 4–7 and ≥8 cups/day vs. no intake were 0.90 [0.81, 1.01], 0.79 [0.66, 0.94] and 0.84 [0.63, 1.11], p-trend = 0.01) * | Sex, birth year, smoking, socioeconomic status, maternal age, parity, maternal BMI, and mutually coffee or tea |
| Linnet et al. 2009 (Denmark) [77] | Prospective cohort; 24,068 children in the Aarhus Birth Cohort | Coffee intake during pregnancy assessed by a questionnaire prior to GW 16 | Hyperkinetic disorder and ADHD recorded in Danish Psychiatric Central Register; children were 3–12 years of age | Coffee was not associated with ADHD risk (RRs [95% CI] for 1–3, 4–9 and ≥10 cups of coffee were 0.9 [0.5, 1.6], 1.3 [0.7, 2.3] and 2.3 [0.9, 5.9]) | Smoking, alcohol, maternal age, gender of the child, parental years of schooling after basic school, employment status, cohabitant status and parental and sibling’s psychiatric hospitalizations or contacts as outpatients |
| Dietary patterns | |||||
| Dietary patterns-ADHD diagnosis or cutoff | |||||
| Galera et al. 2018 (France) [78] | Prospective cohort; 1242 children in the EDEN mother-child cohort | Dietary patterns in the third trimester assessed by FFQ after delivery and derived using principle component analysis | Hyperactivity-inattention symptoms assessed by SDQ based on mother’s report at 3, 5, and 8 years of age; clinical cutoff was used to define abnormality; longitudinal trajectories were derived based on mixture models | Lower scores of healthy dietary pattern (OR [95% CI] for quartile 1 vs. the rest was 1.61 [1.09, 2.37]) * and higher scores of Western dietary pattern (OR for quartile 4 vs. the rest was 1.67 [1.13, 2.47]) * were both associated with higher risk of high hyperactivity-inattention trajectory | Centre, child gender, maternal age, prepregnancy BMI, maternal smoking, maternal alcohol-drinking, gestational diabetes, multiparity, gestational length, birth weight, breastfeeding, prenatal maternal depressive symptoms, prenatal maternal anxiety, postnatal maternal depressive symptoms, parental separation, family income, maternal education, child dietary patterns at age 2, and mutual adjusted for healthy and Western dietary patterns |
| Dietary patterns-ADHD symptoms | |||||
| Rijlaarsdam et al. 2017 (UK) [56] | Prospective cohort; 83 youths with early-onset persistent conduct problems and 81 youths with low conduct problem in ALSPAC | Dietary patterns during pregnancy assessed by FFQ at GW 32 and derived using confirmatory factor analysis | ADHD symptoms assessed by DAWBA based on parent reports at 7 years of age | Unhealthy dietary pattern was indirectly associated with more ADHD symptoms through IGF2 DNA methylation at birth among youths with early-onset persistent conduct problems (beta per SD = 0.069, 95% CI: 0.003, 0.206) *, but not among youths with low conduct problem (beta per SD = −0.015, 95% CI: −0.086, 0.019) | Cumulative risk index during pregnancy and in childhood, including life events, contextual risks, parental risks, interpersonal risks, direct victimization |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, M.; Francis, E.; Hinkle, S.N.; Ajjarapu, A.S.; Zhang, C. Preconception and Prenatal Nutrition and Neurodevelopmental Disorders: A Systematic Review and Meta-Analysis. Nutrients 2019, 11, 1628. https://doi.org/10.3390/nu11071628
Li M, Francis E, Hinkle SN, Ajjarapu AS, Zhang C. Preconception and Prenatal Nutrition and Neurodevelopmental Disorders: A Systematic Review and Meta-Analysis. Nutrients. 2019; 11(7):1628. https://doi.org/10.3390/nu11071628
Chicago/Turabian StyleLi, Mengying, Ellen Francis, Stefanie N. Hinkle, Aparna S. Ajjarapu, and Cuilin Zhang. 2019. "Preconception and Prenatal Nutrition and Neurodevelopmental Disorders: A Systematic Review and Meta-Analysis" Nutrients 11, no. 7: 1628. https://doi.org/10.3390/nu11071628
APA StyleLi, M., Francis, E., Hinkle, S. N., Ajjarapu, A. S., & Zhang, C. (2019). Preconception and Prenatal Nutrition and Neurodevelopmental Disorders: A Systematic Review and Meta-Analysis. Nutrients, 11(7), 1628. https://doi.org/10.3390/nu11071628






