Comparisons of Visceral Adiposity Index, Body Shape Index, Body Mass Index and Waist Circumference and Their Associations with Diabetes Mellitus in Adults
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Population
2.2. Definition of Key Study Outcome DM
2.3. Anthropometry and Biochemical Measurements
2.4. Biochemical Measurements
2.5. Statistical Analysis
3. Results
3.1. Characteristics of the Study Population Classified According to the CVAI Quartiles
3.2. Characteristics of the Study Population Grouped by ABSI Levels
3.3. Partial Correlation Analysis with Metabolic Variables
3.4. ORs of CVAI or ABSI with Risk of Diabetes
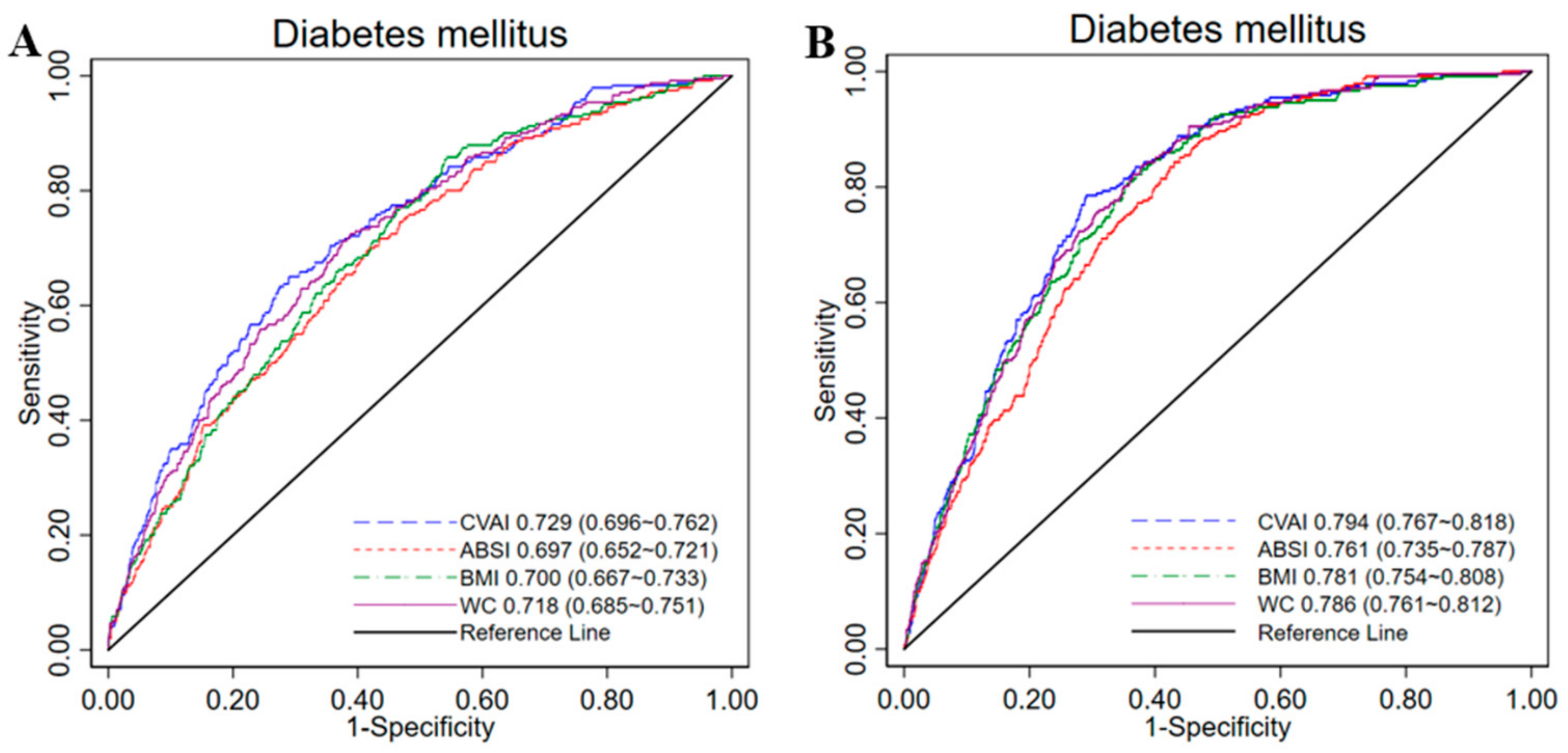
3.5. Receiver-Operating Characteristic (ROC) Curve Analysis and Optimum Thresholds for Anthropometric Indices
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hossain, P.; Kawar, B.; El Nahas, M. Obesity and diabetes in the developing world—A growing challenge. N. Engl. J. Med. 2007, 356, 213–215. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Lu, J.; Weng, J.; Jia, W.; Ji, L.; Xiao, J.; Shan, Z.; Liu, J.; Tian, H.; Ji, Q.; et al. Prevalence of diabetes among men and women in China. N. Engl. J. Med. 2010, 362, 1090–1101. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Wang, L.; He, J.; Bi, Y.; Li, M.; Wang, T.; Wang, L.; Jiang, Y.; Dai, M.; Lu, J.; et al. Prevalence and control of diabetes in Chinese adults. JAMA 2013, 310, 948–959. [Google Scholar] [CrossRef] [PubMed]
- Caprio, S.; Perry, R.; Kursawe, R. Adolescent Obesity and Insulin Resistance: Roles of Ectopic Fat Accumulation and Adipose Inflammation. Gastroenterology 2017, 152, 1638–1646. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.C.; Hayashi, T.; Fujimoto, W.Y.; Kahn, S.E.; Leonetti, D.L.; McNeely, M.J.; Boyko, E.J. Visceral abdominal fat accumulation predicts the conversion of metabolically healthy obese subjects to an unhealthy phenotype. Int. J. Obes. (Lond.) 2015, 39, 1365–1370. [Google Scholar] [CrossRef] [PubMed]
- Yun, C.H.; Bezerra, H.G.; Wu, T.H.; Yang, F.S.; Liu, C.C.; Wu, Y.J.; Kuo, J.Y.; Hung, C.L.; Lee, J.J.; Hou, C.J.; et al. The normal limits, subclinical significance, related metabolic derangements and distinct biological effects of body site-specific adiposity in relatively healthy population. PLoS ONE 2013, 8, e61997. [Google Scholar] [CrossRef] [PubMed]
- Okorodudu, D.O.; Jumean, M.F.; Montori, V.M.; Romero-Corral, A.; Somers, V.K.; Erwin, P.J.; Lopez-Jimenez, F. Diagnostic performance of body mass index to identify obesity as defined by body adiposity: A systematic review and meta-analysis. Int. J. Obes. (Lond.) 2010, 34, 791–799. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.M.; Bredlau, C.; Bosy-Westphal, A.; Mueller, M.; Shen, W.; Gallagher, D.; Maeda, Y.; McDougall, A.; Peterson, C.M.; Ravussin, E.; et al. Relationships between body roundness with body fat and visceral adipose tissue emerging from a new geometrical model. Obesity (Silver Spring) 2013, 21, 2264–2271. [Google Scholar] [CrossRef]
- Nevill, A.M.; Stewart, A.D.; Olds, T.; Holder, R. Relationship between adiposity and body size reveals limitations of BMI. Am. J. Phys. Anthropol. 2006, 129, 151–156. [Google Scholar] [CrossRef]
- Nazare, J.A.; Smith, J.; Borel, A.L.; Aschner, P.; Barter, P.; Van Gaal, L.; Tan, C.E.; Wittchen, H.U.; Matsuzawa, Y.; Kadowaki, T.; et al. Usefulness of measuring both body mass index and waist circumference for the estimation of visceral adiposity and related cardiometabolic risk profile (from the INSPIRE ME IAA study). Am. J. Cardiol. 2015, 115, 307–315. [Google Scholar] [CrossRef]
- Wang, Y.; Rimm, E.B.; Stampfer, M.J.; Willett, W.C.; Hu, F.B. Comparison of abdominal adiposity and overall obesity in predicting risk of type 2 diabetes among men. Am. J. Clin. Nutr. 2005, 81, 555–563. [Google Scholar] [CrossRef] [PubMed]
- Koh-Banerjee, P.; Wang, Y.; Hu, F.B.; Spiegelman, D.; Willett, W.C.; Rimm, E.B. Changes in body weight and body fat distribution as risk factors for clinical diabetes in US men. Am. J. Epidemiol. 2004, 159, 1150–1159. [Google Scholar] [CrossRef] [PubMed]
- Pouliot, M.C.; Despres, J.P.; Lemieux, S.; Moorjani, S.; Bouchard, C.; Tremblay, A.; Nadeau, A.; Lupien, P.J. Waist circumference and abdominal sagittal diameter: Best simple anthropometric indexes of abdominal visceral adipose tissue accumulation and related cardiovascular risk in men and women. Am. J. Cardiol. 1994, 73, 460–468. [Google Scholar] [CrossRef]
- Oh, J.Y.; Sung, Y.A.; Lee, H.J. The visceral adiposity index as a predictor of insulin resistance in young women with polycystic ovary syndrome. Obesity (Silver Spring) 2013, 21, 1690–1694. [Google Scholar] [CrossRef] [PubMed]
- Amato, M.C.; Giordano, C.; Galia, M.; Criscimanna, A.; Vitabile, S.; Midiri, M.; Galluzzo, A. AlkaMeSy Study Group. Visceral Adiposity Index: A reliable indicator of visceral fat function associated with cardiometabolic risk. Diabetes Care 2010, 33, 920–922. [Google Scholar] [CrossRef]
- Liu, P.J.; Ma, F.; Lou, H.P.; Chen, Y. Visceral Adiposity Index Is Associated with Pre-Diabetes and Type 2 Diabetes Mellitus in Chinese Adults Aged 20–50. Ann. Nutr. Metab. 2016, 68, 235–243. [Google Scholar] [CrossRef]
- Du, T.; Sun, X.; Huo, R.; Yu, X. Visceral adiposity index, hypertriglyceridemic waist and risk of diabetes: The China Health and Nutrition Survey 2009. Int. J. Obes. (Lond.) 2014, 38, 840–847. [Google Scholar] [CrossRef]
- Zhao, Q.; Zhang, K.; Li, Y.; Zhen, Q.; Shi, J.; Yu, Y.; Tao, Y.; Cheng, Y.; Liu, Y. Capacity of a body shape index and body roundness index to identify diabetes mellitus in Han Chinese people in Northeast China: A cross-sectional study. Diabet. Med. 2018, 35, 1580–1587. [Google Scholar] [CrossRef]
- Krakauer, N.Y.; Krakauer, J.C. A new body shape index predicts mortality hazard independently of body mass index. PLoS ONE 2012, 7, e39504. [Google Scholar] [CrossRef]
- Ji, M.; Zhang, S.; An, R. Effectiveness of A Body Shape Index (ABSI) in predicting chronic diseases and mortality: A systematic review and meta-analysis. Obes. Rev. 2018, 19, 737–759. [Google Scholar] [CrossRef]
- Zhang, B.; Zhai, F.Y.; Du, S.F.; Popkin, B.M. The China Health and Nutrition Survey, 1989–2011. Obes. Rev. 2014, 15 (Suppl. 1), 2–7. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Gong, L.; Li, Q.; Hu, J.; Zhang, S.; Wang, Y.; Zhou, H.S.; Yang, S.; Wang, Z. A Novel Visceral Adiposity Index for Prediction of Type 2 Diabetes and Pre-diabetes in Chinese adults: A 5-year prospective study. Sci. Rep. 2017, 7, 13784. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Fluss, R.; Faraggi, D.; Reiser, B. Estimation of the Youden Index and its associated cutoff point. Biom. J. 2005, 47, 458–472. [Google Scholar] [CrossRef] [PubMed]
- Liu, X. Classification accuracy and cut point selection. Stat. Med. 2012, 31, 2676–2686. [Google Scholar] [CrossRef] [PubMed]
- Ma, R.C.; Lin, X.; Jia, W. Causes of type 2 diabetes in China. Lancet Diabetes Endocrinol. 2014, 2, 980–991. [Google Scholar] [CrossRef]
- Xia, M.F.; Chen, Y.; Lin, H.D.; Ma, H.; Li, X.-M.; Aleteng, Q.; Li, Q.; Wang, D.Q.; Hu, Y.X.; Pan, B.-S.; et al. An indicator of visceral adipose dysfunction to evaluate metabolic health in adult Chinese. Sci. Rep. 2016, 6, 38214. [Google Scholar] [CrossRef]
- Xia, M.F.; Lin, H.D.; Chen, L.Y.; Wu, L.; Ma, H.; Li, Q.; Aleteng, Q.; Chen, Y.; Sun, Y.X.; Hu, Y.; et al. Association of visceral adiposity and its longitudinal increase with the risk of diabetes in Chinese adults: A prospective cohort study. Diabetes Metab. Res. Rev. 2018, 34, e3048. [Google Scholar] [CrossRef]
- Vazquez, G.; Duval, S.; Jacobs, D.R., Jr.; Silventoinen, K. Comparison of body mass index, waist circumference, and waist/hip ratio in predicting incident diabetes: A meta-analysis. Epidemiol. Rev. 2007, 29, 115–128. [Google Scholar] [CrossRef]
- Hartemink, N.; Boshuizen, H.C.; Nagelkerke, N.J.; Jacobs, M.A.; van Houwelingen, H.C. Combining risk estimates from observational studies with different exposure cutpoints: A meta-analysis on body mass index and diabetes type 2. Am. J. Epidemiol. 2006, 163, 1042–1052. [Google Scholar] [CrossRef]
- He, S.; Chen, X. Could the new body shape index predict the new onset of diabetes mellitus in the Chinese population? PLoS ONE 2013, 8, e50573. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Jousilahti, P.; Stehouwer, C.D.A.; Söderberg, S.; Onat, A.; Laatikainen, T.; Yudkin, J.S.; Dankner, R.; Morris, R.; Tuomilehto, J.; et al. Cardiovascular and all-cause mortality in relation to various anthropometric measures of obesity in Europeans. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 295–304. [Google Scholar] [CrossRef] [PubMed]
- He, S.; Zheng, Y.; Wang, H.; Chen, X. Assessing the relationship between a body shape index and mortality in a group of middle-aged men. Clin. Nutr. 2017, 36, 1355–1359. [Google Scholar] [CrossRef] [PubMed]
- Maessen, M.F.; Eijsvogels, T.M.; Verheggen, R.J.; Hopman, M.T.; Verbeek, A.L.; de Vegt, F. Entering a new era of body indices: The feasibility of a body shape index and body roundness index to identify cardiovascular health status. PLoS ONE 2014, 9, e107212. [Google Scholar] [CrossRef] [PubMed]
- Fujita, M.; Sato, Y.; Nagashima, K.; Takahashi, S.; Hata, A. Predictive power of a body shape index for development of diabetes, hypertension, and dyslipidemia in Japanese adults: A retrospective cohort study. PLoS ONE 2015, 10, e0128972. [Google Scholar] [CrossRef]
- Chang, Y.; Guo, X.; Chen, Y.; Guo, L.; Li, Z.; Yu, S.; Yang, H.; Sun, Y. A body shape index and body roundness index: Two new body indices to identify diabetes mellitus among rural populations in northeast China. BMC Public Health 2015, 15, 794. [Google Scholar] [CrossRef]
- Wang, H.; Liu, A.; Zhao, T.; Gong, X.; Pang, T.; Zhou, Y.; Xiao, Y.; Yan, Y.; Fan, C.; Teng, W.; et al. Comparison of anthropometric indices for predicting the risk of metabolic syndrome and its components in Chinese adults: A prospective, longitudinal study. BMJ Open 2017, 7, e016062. [Google Scholar] [CrossRef]
- Kahn, H.S.; Cheng, Y.J. Longitudinal changes in BMI and in an index estimating excess lipids among white and black adults in the United States. Int. J. Obes. (Lond.) 2008, 32, 136–143. [Google Scholar] [CrossRef]
- Zhou, B.F. Cooperative Meta-Analysis Group of the Working Group on Obesity in C. Predictive values of body mass index and waist circumference for risk factors of certain related diseases in Chinese adults—Study on optimal cut-off points of body mass index and waist circumference in Chinese adults. Biomed. Environ. Sci. 2002, 15, 83–96. [Google Scholar]
| Men | Women | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Q1(<57) | Q2(57–89) | Q3(89–122) | Q4(≥122) | p-Value | Q1(<48) | Q2(48–79) | Q3(79–109) | Q4(≥109) | p-Value | |
| n = 623 | n = 606 | n = 621 | n = 613 | n = 838 | n = 851 | n = 828 | n = 858 | |||
| Age, years | 49.2 (15.7) | 52.6 (13.8) | 55.6 (12.9) | 56.6 (12.7) | <0.001 | 36.3 (9.9) | 49.0 (10.4) | 56.0 (10.9) | 63.6 (10.5) | <0.001 |
| BMI, Kg/m2 | 20.7 (7.3) | 22.6 (2.8) | 24.2 (2.6) | 27.3 (3.8) | <0.001 | 20.6 (2.2) | 22.4 (2.5) | 24.2 (2.8) | 26.7 (3.5) | <0.001 |
| SBP, mm Hg | 121.1 (16.3) | 125.3 (16.0) | 128.9 (17.5) | 134.4 (18.5) | <0.001 | 109.8 (12.0) | 120.4 (15.8) | 127.6 (19.4) | 136.8 (20.0) | <0.001 |
| DBP, mm Hg | 77.7 (10.4) | 80.3 (10.2) | 82.8 (10.6) | 86.7 (10.8) | <0.001 | 72.4 (8.6) | 77.8 (9.7) | 80.8 (11.2) | 83.8 (11.5) | <0.001 |
| WC, cm | 72.7 (5.9) | 81.6 (3.4) | 87.8 (3.3) | 97.8 (5.8) | <0.001 | 72.1 (7.0) | 78.8 (6.7) | 84.0 (7.2) | 92.0 (8.7) | <0.001 |
| WHtR | 0.45 (0.06) | 0.49 (0.02) | 0.53 (0.02) | 0.58 (0.03) | <0.001 | 0.46 (0.04) | 0.51 (0.04) | 0.54 (0.04) | 0.60 (0.06) | <0.001 |
| WHR | 0.83 (0.06) | 0.88 (0.05) | 0.91 (0.05) | 0.95 (0.06) | <0.001 | 0.81 (0.06) | 0.86 (0.10) | 0.89 (0.10) | 0.92 (0.26) | <0.001 |
| TC mmol/L | 4.6 (0.9) | 4.8 (0.9) | 4.9 (1.0) | 5.1 (1.0) | <0.001 | 4.4 (0.9) | 4.9 (1.0) | 5.1 (1.0) | 5.4 (1.0) | <0.001 |
| Total triglycerides, mmol/L | 0.9 (0.7–1.3) | 1.2 (0.8–1.8) | 1.5 (1.0–2.2) | 2.1 (1.4–3.1) | <0.001 | 0.8 (0.6–1.1) | 1.1 (0.8–1.6) | 1.4 (1.0–1.9) | 2.0 (1.4–2.8) | <0.001 |
| HDL-C, mmol/L | 1.6 (0.5) | 1.4 (0.4) | 1.3 (0.3) | 1.2 (0.3) | <0.001 | 1.7 (0.6) | 1.5 (0.4) | 1.5 (0.3) | 1.3 (0.3) | <0.001 |
| LDL-C mmol/L | 2.8 (1.1) | 3.0 (0.8) | 3.1 (1.0) | 3.1 (1.0) | <0.001 | 2.6 (0.7) | 3.1 (0.9) | 3.3 (1.0) | 3.3 (1.0) | <0.001 |
| HbA1c, % | 5.4 (0.8) | 5.6 (0.9) | 5.7 (1.0) | 6.0 (1.1) | <0.001 | 5.3 (1.0) | 5.5 (0.6) | 5.7 (0.8) | 6.0 (1.0) | <0.001 |
| HbA1c, mmol/L | 35.8 (8.5) | 37.6 (9.3) | 38.6 (10.5) | 42.1 (11.7) | <0.001 | 34.4 (10.7) | 36.3 (6.8) | 38.7 (9.0) | 42.4 (11.4) | <0.001 |
| Glucose, mmol/L | 5.2 (1.2) | 5.4 (1.4) | 5.6 (1.6) | 6.1 (2.1) | <0.001 | 4.9 (0.6) | 5.2 (0.9) | 5.5 (1.4) | 6.0 (1.8) | <0.001 |
| Insulin, μU/mL | 8.44(6.00–11.87) | 9.18(6.44–13.30) | 10.90 (7.64–15.09) | 13.70 (9.58–20.58) | <0.001 | 8.98 (6.74–12.34) | 10.10 (7.28–13.96) | 10.87 (7.77–15.70) | 13.53 (9.37–20.52) | <0.001 |
| HOMA-IR | 1.88 (1.27–2.66) | 2.12 (1.42–3.11) | 2.58 (1.74–3.78) | 3.41 (2.28–5.60) | <0.001 | 1.97 (1.41–2.70) | 2.27 (1.62–3.20) | 2.53 (1.74–3.94) | 3.36 (2.18–5.64) | <0.001 |
| QUICKI | 2.09 (1.99–2.20) | 2.11 (2.00–2.22) | 2.09 (2.00–2.21) | 2.11 (2.01–2.26) | 0.015 | 2.04 (1.95–2.15) | 2.07 (1.98–2.17) | 2.09 (2.00–2.19) | 2.11 (2.01–2.24) | <0.001 |
| hs-CRP, mg/L | 1.0 (0.0–2.0) | 1.0 (0.0–2.0) | 1.0 (1.0–3.0) | 2.0 (1.0–3.0) | <0.001 | 0.0 (0.0–1.0) | 1.0 (0.0–2.0) | 1.0 (1.0–3.0) | 2.0 (1.0–4.0) | <0.001 |
| Uric acid, mg/L | 326.2 (89.2) | 339.0 (118.7) | 357.9 (116.3) | 395.8 (119.3) | <0.001 | 230.7 (58.7) | 249.2 (67.8) | 271.3 (72.2) | 314.4 (87.1) | <0.001 |
| Current smoking | 385 (61.8%) | 343 (56.6%) | 332 (53.5%) | 304 (49.6%) | <0.001 | 5 (0.6%) | 18 (2.1%) | 29 (3.5%) | 43 (5.0%) | <0.001 |
| Current drinking | 349 (56.0%) | 358 (59.1%) | 382 (61.5%) | 359 (58.6%) | 0.27 | 87 (10.4%) | 87 (10.2%) | 66 (8.0%) | 74 (8.6%) | 0.24 |
| DM | 21 (3.4%) | 43 (7.1%) | 58 (9.3%) | 118 (19.2%) | <0.001 | 5 (0.6%) | 22 (2.6%) | 66 (8.0%) | 149 (17.4%) | <0.001 |
| Men | Women | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Q1 | Q2 | Q3 | Q4 | p-Value | Q1 | Q2 | Q3 | Q4 | p-Value | |
| n = 608 | n = 544 | n = 744 | n = 567 | n = 899 | n = 892 | n = 767 | n = 817 | |||
| Age, years | 49.3 (14.7) | 51.8 (14.0) | 53.9 (12.9) | 59.1 (13.1) | <0.001 | 44.7 (13.5) | 49.1 (13.2) | 53.7 (12.8) | 58.6 (14.4) | <0.001 |
| BMI, Kg/m2 | 24.0 (8.4) | 23.6 (3.4) | 24.0 (3.3) | 22.9 (3.5) | <0.001 | 23.3 (3.7) | 23.7 (3.5) | 24.0 (3.4) | 23.1 (3.5) | <0.001 |
| Systolic BP, mm Hg | 124.2 (16.2) | 127.0 (17.5) | 129.0 (18.6) | 129.2 (18.0) | <0.001 | 117.6 (16.3) | 123.6 (20.1) | 125.9 (19.9) | 128.5 (21.0) | <0.001 |
| Diastolic BP, mm Hg | 80.4 (10.9) | 81.9 (11.1) | 82.5 (11.1) | 82.5 (10.9) | 0.002 | 76.3 (9.9) | 79.4 (11.7) | 79.7 (10.8) | 79.8 (11.6) | <0.001 |
| Waist circumference, cm | 77.0 (9.3) | 83.2 (8.3) | 87.8 (8.5) | 91.4 (9.3) | <0.001 | 73.5 (8.0) | 80.2 (8.4) | 84.6 (8.2) | 89.8 (9.6) | <0.001 |
| WHtR | 0.47 (0.07) | 0.50 (0.05) | 0.53 (0.05) | 0.55 (0.05) | <0.001 | 0.47 (0.05) | 0.51 (0.05) | 0.54 (0.05) | 0.58 (0.06) | <0.001 |
| WHR | 0.84 (0.07) | 0.88 (0.05) | 0.91 (0.05) | 0.95 (0.07) | <0.001 | 0.80 (0.06) | 0.85 (0.07) | 0.88 (0.05) | 0.96 (0.28) | <0.001 |
| CVAI | 53.3 (41.7) | 80.7 (39.4) | 102.0 (40.2) | 119.9 (42.0) | <0.001 | 54.0 (42.7) | 73.8 (41.7) | 89.2 (40.0) | 99.4 (41.6) | <0.001 |
| Total cholesterol, mmol/L | 4.8 (1.0) | 4.9 (0.9) | 4.9 (1.0) | 4.9 (1.0) | 0.071 | 4.7 (1.0) | 4.9 (1.0) | 5.0 (1.0) | 5.2 (1.1) | <0.001 |
| Total triglycerides, mmol/L | 1.2 (0.8–1.9) | 1.2 (0.8–2.0) | 1.4 (0.9–2.2) | 1.4 (0.9–2.3) | <0.001 | 1.1 (0.7–1.6) | 1.3 (0.8–1.9) | 1.3 (0.9–2.1) | 1.4 (0.9–2.1) | <0.001 |
| HDL-cholesterol, mmol/L | 1.4 (0.4) | 1.4 (0.5) | 1.4 (0.4) | 1.4 (0.4) | 0.17 | 1.5 (0.4) | 1.5 (0.4) | 1.5 (0.6) | 1.5 (0.5) | 0.14 |
| LDL-cholesterol, mmol/L | 2.9 (0.9) | 3.0 (1.0) | 3.0 (1.0) | 3.0 (1.1) | 0.38 | 2.9 (0.9) | 3.0 (1.0) | 3.1 (1.0) | 3.2 (1.0) | <0.001 |
| HbA1c, % | 5.6 (0.9) | 5.6 (0.8) | 5.7 (0.9) | 5.8 (1.1) | <0.001 | 5.5 (0.6) | 5.6 (1.1) | 5.7 (0.9) | 5.8 (1.0) | <0.001 |
| HbA1c, mmol/L | 37.2 (9.5) | 37.8 (8.5) | 38.8 (10.1) | 40.3 (12.6) | <0.001 | 36.1 (7.0) | 37.4 (11.7) | 38.7 (9.3) | 39.9 (11.2) | <0.001 |
| Glucose, mmol/L | 5.4 (1.6) | 5.5 (1.4) | 5.7 (1.8) | 5.7 (1.7) | <0.001 | 5.1 (0.9) | 5.3 (1.1) | 5.5 (1.3) | 5.7 (1.9) | <0.001 |
| Insulin, μU/mL | 10.10 (6.95–14.07) | 10.48 (6.97–14.99) | 10.52 (7.33–15.14) | 10.70 (7.14–15.90) | 0.13 | 10.39 (7.66–14.61) | 10.71 (7.58–15.41) | 10.61 (7.85–16.15) | 10.80 (7.58–15.81) | 0.27 |
| HOMA-IR | 2.26 (1.50–3.35) | 2.44 (1.55–3.68) | 2.52 (1.66–3.87) | 2.46 (1.63–4.15) | 0.006 | 2.26 (1.64–3.39) | 2.42 (1.68–3.69) | 2.48 (1.73–3.89) | 2.49 (1.65–4.01) | 0.003 |
| QUICKI | 2.09 (1.98–2.19) | 2.10 (2.00–2.22) | 2.11 (2.02–2.23) | 2.11 (2.00–2.24) | 0.018 | 2.05 (1.96–2.15) | 2.08 (1.99–2.17) | 2.09 (2.00–2.20) | 2.10 (2.01–2.23) | <0.001 |
| hs-CRP, mg/L | 1.0 (0.0–2.0) | 1.0 (0.0–2.0) | 1.0 (1.0–3.0) | 1.0 (1.0–3.0) | <0.001 | 1.0 (0.0–2.0) | 1.0 (0.0–2.0) | 1.0 (1.0–3.0) | 1.0 (1.0–3.0) | <0.001 |
| Uric acid, mg/L | 347.3 (127.8) | 352.1 (114.8) | 364.1 (112.0) | 352.7 (100.7) | 0.045 | 253.6 (73.5) | 263.9 (78.3) | 272.5 (80.3) | 278.3 (81.2) | <0.001 |
| Current smoking | 320 (52.6%) | 300 (55.1%) | 429 (57.7%) | 315 (55.6%) | 0.33 | 19 (2.1%) | 14 (1.6%) | 26 (3.4%) | 36 (4.4%) | 0.002 |
| Current drinking | 343 (56.4%) | 323 (59.4%) | 453 (60.9%) | 329 (58.0%) | 0.39 | 77 (8.6%) | 89 (10.0%) | 73 (9.5%) | 75 (9.2%) | 0.77 |
| DM | 38 (6.3%) | 43 (7.9%) | 82 (11.0%) | 77 (13.6%) | <0.001 | 31 (3.4%) | 48 (5.4%) | 67 (8.7%) | 96 (11.8%) | <0.001 |
| CVAI (Age-and Sex Adjusted) | ABSI (Age-and Sex Adjusted) | |||
|---|---|---|---|---|
| r | p | r | p | |
| Glucose | 0.209 | <0.001 | 0.042 | 0.002 |
| HbA1c (%) | 0.199 | <0.001 | 0.044 | <0.001 |
| Insulin | 0.133 | <0.001 | 0.029 | 0.03 |
| HOMA-IR | 0.144 | <0.001 | 0.035 | 0.008 |
| TC | 0.201 | <0.001 | 0.034 | 0.009 |
| TG | 0.451 | <0.001 | 0.054 | <0.001 |
| HDL-C | −0.420 | <0.001 | −0.005 | <0.01 |
| LDL-C | 0.127 | <0.001 | 0.022 | 0.09 |
| Uric acid | 0.292 | <0.001 | 0.027 | 0.04 |
| hs-CRP | 0.021 | 0.11 | 0.012 | 0.35 |
| Model 1 | Model 2 | Model 3 | Model 4 | |
|---|---|---|---|---|
| Visceral adiposity index (CVAI) | ||||
| Men | ||||
| Q1 | 1 | 1 | 1 | 1 |
| Q2 | 2.0 (1.2–3.5) | 2.0 (1.2–3.5) | 2.0 (1.1–3.4) | 1.9 (1.1–3.3) |
| Q3 | 2.6 (1.5–4.3) | 2.6 (1.5–4.3) | 2.4 (1.4–4.0) | 2.3 (1.4–3.9) |
| Q4 | 5.9 (3.6–9.6) | 5.9 (3.6–9.5) | 5.2 (3.1–8.5)) | 4.9 (2.9–8.1) |
| Women | ||||
| Q1 | 1 | 1 | 1 | 1 |
| Q2 | 3.6 (1.3–9.7) | 3.7 (1.4–9.9) | 3.1 (1.2–8.4) | 3.0 (1.1–8.2) |
| Q3 | 10.5 (4.1–27.1) | 10.7 (4.1–27.5) | 8.7 (3.2–21.7) | 7.7 (2.9–21.0) |
| Q4 | 22.6 (8.7–58.9) | 22.9 (8.8–59.9) | 16.2 (6.1–43.2) | 14.2 (5.3–38.2) |
| Body shape index (ABSI) | ||||
| Men | ||||
| Q1 | 1 | 1 | 1 | 1 |
| Q2 | 1.2 (0.8–1.9) | 1.3 (0.8–2.0) | 1.2 (0.8–1.9) | 1.2 (0.8–1.9) |
| Q3 | 1.7 (1.1–2.5) | 1.8 (1.2–2.6) | 1.6 (1.1–2.5) | 1.6 (1.1–2.4) |
| Q4 | 1.9 (1.2–2.8) | 1.9 (1.2–2.9) | 1.9 (1.2–2.8) | 1.8 (1.2–2.8) |
| Women | ||||
| Q1 | 1 | 1 | 1 | 1 |
| Q2 | 1.3 (0.8–2.1) | 1.4 (0.9–2.2) | 1.2 (0.8–2.0) | 1.2 (0.7–1.9) |
| Q3 | 1.9 (1.2–2.9) | 1.2 (1.2–3.0) | 1.8 (1.1–2.8) | 1.7 (1.1–2.7) |
| Q4 | 2.0 (1.3–3.1) | 2.1 (1.4–3.3) | 2.0 (1.3–3.1) | 2.0 (1.3–3.1) |
| Men | Women | |||||||
|---|---|---|---|---|---|---|---|---|
| Cut-off | Sensitivity (%) | Specificity (%) | Youden Index | Cut-off | Sensitivity (%) | Specificity (%) | Youden Index | |
| CVAI | 107.27 | 0.65 | 0.71 | 0.36 | 88.15 | 0.79 | 0.71 | 0.50 |
| ABSI | 0.08 | 0.72 | 0.57 | 0.28 | 0.08 | 0.84 | 0.57 | 0.41 |
| BMI | 24.73 | 0.86 | 0.46 | 0.32 | 23.18 | 0.82 | 0.63 | 0.45 |
| WC | 87.9 | 0.71 | 0.63 | 0.34 | 84.9 | 0.84 | 0.62 | 0.46 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wei, J.; Liu, X.; Xue, H.; Wang, Y.; Shi, Z. Comparisons of Visceral Adiposity Index, Body Shape Index, Body Mass Index and Waist Circumference and Their Associations with Diabetes Mellitus in Adults. Nutrients 2019, 11, 1580. https://doi.org/10.3390/nu11071580
Wei J, Liu X, Xue H, Wang Y, Shi Z. Comparisons of Visceral Adiposity Index, Body Shape Index, Body Mass Index and Waist Circumference and Their Associations with Diabetes Mellitus in Adults. Nutrients. 2019; 11(7):1580. https://doi.org/10.3390/nu11071580
Chicago/Turabian StyleWei, Junxiang, Xin Liu, Hong Xue, Youfa Wang, and Zumin Shi. 2019. "Comparisons of Visceral Adiposity Index, Body Shape Index, Body Mass Index and Waist Circumference and Their Associations with Diabetes Mellitus in Adults" Nutrients 11, no. 7: 1580. https://doi.org/10.3390/nu11071580
APA StyleWei, J., Liu, X., Xue, H., Wang, Y., & Shi, Z. (2019). Comparisons of Visceral Adiposity Index, Body Shape Index, Body Mass Index and Waist Circumference and Their Associations with Diabetes Mellitus in Adults. Nutrients, 11(7), 1580. https://doi.org/10.3390/nu11071580








