In Search of Panacea—Review of Recent Studies Concerning Nature-Derived Anticancer Agents
Abstract
1. Introduction
- The mitotic inhibitors:
- Vinca alkaloids–vinblastine, vincristine, vindesine, vinorelbine, vinflunine,
- colchicine,
- podophyllotoxin,
- taxanes (paclitaxel, docetaxel).
- I and II topoisomerases inhibitors:
- camptothecin,
- topotecan
- irinotecan
- etoposide
- Inducers of xenobiotic metabolism:
- allyl sulfide,
- indole-3-carbinol,
- phenethyl isothiocyanate,
- sulforaphane,
- glucoraphanin,
- iberin,
- terpenes,
- coumarins.
- Polyphenols:
- carnosol
- resveratrol
- 6-gingerol,
- honokiol,
- flavonoids.
2. Methods
3. The Mitotic Inhibitors
4. I and II Topoisomerases Inhibitors
5. Inducers of Xenobiotics Metabolism
5.1. Terpenes
5.1.1. Parthenolide
5.1.2. Betulinic Acid
5.2. Coumarins
5.3. Isothiocyanates
5.3.1. Phenethyl Isothiocyanate
5.3.2. Sulforaphane and Glucoraphanin
5.3.3. Iberin
5.4. Organosulfur Compounds
5.5. Indole-3-Carbinol
6. Polyphenols
6.1. Carnosol
6.2. Resveratrol
6.3. 6-Gingerol
6.4. Honokiol
6.5. Flavonoids
6.5.1. Green Tea Flavonoids
6.5.2. Caffeic Acid Phenethyl Ester and Caffeic Acid
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- American Cancer Society. Cancer Facts & Figures 2018; American Cancer Society: Atlanta, GA, USA, 2018. [Google Scholar]
- Prakash, O.; Kumar, A.; Kumar, P.; Ajeet, A. Anticancer Potential of Plants and Natural Products: A Review. Am. J. Pharmacol. Sci. 2013, 1, 104–115. [Google Scholar] [CrossRef]
- Demain, A.L.; Vaishnav, P. Natural products for cancer chemotherapy. Microb. Biotechnol. 2011, 4, 687–699. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Kelly, T.K.; De Carvalho, D.D.; Jones, P.A. Epigenetics Modifications as Therapeutic Targets. Nat. Biotechnol. 2010, 28, 1069. [Google Scholar] [CrossRef] [PubMed]
- Putiri, E.L.; Robertson, K.D. Epigenetic mechanisms and genome stability. Clin. Epigenetics 2011, 2, 299–314. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, Y.; Liu, X.; Cho, W. Genetic and Epigenetic Studies for Determining Molecular Targets of Natural Product Anticancer Agents. Curr. Cancer Drug Targets 2013, 13, 506–518. [Google Scholar] [CrossRef] [PubMed]
- Lichtenstein, A.V. Cancer: Evolutionary, genetic and epigenetic aspects. Clin. Epigenetics 2010, 1, 85–100. [Google Scholar] [CrossRef] [PubMed]
- Tsai, H.C.; Baylin, S.B. Cancer epigenetics: Linking basic biology to clinical medicine. Cell Res. 2011, 21, 502–517. [Google Scholar] [CrossRef]
- Huang, J.; Plass, C.; Gerhauser, C. Cancer chemoprevention by targeting the epigenome. Curr. Drug Targets 2011, 12, 1925–1956. [Google Scholar] [CrossRef]
- Stefanska, B.; Karlic, H.; Varga, F.; Fabianowska-Majewska, K.; Haslberger, A.G. Epigenetic mechanisms in anti-cancer actions of bioactive food components - The implications in cancer prevention. Br. J. Pharmacol. 2012, 167, 279–297. [Google Scholar] [CrossRef]
- Ratovitski, E. Anticancer Natural Compounds as Epigenetic Modulators of Gene Expression. Curr. Genom. 2017, 18, 175–205. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, P.; Ho, E.; Williams, D.E.; Dashwood, R.H. Dietary phytochemicals, HDAC inhibition, and DNA damage/repair defects in cancer cells. Clin. Epigenetics 2011, 3, 4. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Tollefsbol, T. Impact of Epigenetic Dietary Components on Cancer through Histone Modifications. Curr. Med. Chem. 2015, 22, 2051–2064. [Google Scholar] [CrossRef] [PubMed]
- Vahid, F.; Zand, H.; Nosrat-Mirshekarlou, E.; Najafi, R.; Hekmatdoost, A. The role dietary of bioactive compounds on the regulation of histone acetylases and deacetylases: A review. Gene 2015, 562, 8–15. [Google Scholar] [CrossRef] [PubMed]
- Lim, U.; Song, M.-A. Dietary and lifestyle factors of DNA methylation. Methods Mol. Biol. 2012, 863, 359–376. [Google Scholar] [PubMed]
- Li, Y.; Tollefsbol, T.O. Impact on DNA Methylation in Cancer Prevention and Therapy by Bioactive Dietary Components. Curr. Med. Chem. 2010, 17, 2141–2151. [Google Scholar] [CrossRef] [PubMed]
- Kong, A.-N.T.; Zhang, C.; Su, Z.-Y. Targeting Epigenetics for Cancer Prevention By Dietary Cancer Preventive Compounds--The Case of miRNA. Cancer Prev. Res. 2013, 6, 622–624. [Google Scholar] [CrossRef] [PubMed]
- Phuah, N.H.; Nagoor, N.H. Regulation of MicroRNAs by Natural Agents: A New Strategies in Cancer Therapies. Biomed. Res. Int. 2014, 2014, 804510. [Google Scholar] [CrossRef]
- Lin, Q.; Ma, L.; Liu, Z.; Yang, Z.; Wang, J.; Liu, J.; Jiang, G. Targeting microRNAs: A new action mechanism of natural compounds. Oncotarget 2017, 8, 15961–15970. [Google Scholar] [CrossRef]
- Shankar, S.; Kumar, D.; Srivastava, R.K. Epigenetic modifications by dietary phytochemicals: Implications for personalized nutrition. Pharmacol. Ther. 2013, 138, 1–17. [Google Scholar] [CrossRef]
- Li, Y.; Sarkar, F.H. Targeting Epigenetically Deregulated miRNA by Nutraceuticals: Focusing on Cancer Prevention and Treatment. Curr. Pharmacol. Rep. 2015, 1, 1–10. [Google Scholar] [CrossRef]
- De Kok, T.M.; van Breda, S.G.; Briedé, J.J. Genomics-Based Identification of Molecular Mechanisms behind the Cancer Preventive Action of Phytochemicals: Potential and Challenges. Curr. Pharm. Biotechnol. 2012, 13, 255–264. [Google Scholar] [CrossRef]
- Baylin, S.B.; Jones, P.A. A decade of exploring the cancer epigenome—Biological and translational implications. Nat. Rev. Cancer 2011, 11, 726–734. [Google Scholar] [CrossRef] [PubMed]
- Plass, C.; Pfister, S.M.; Lindroth, A.M.; Bogatyrova, O.; Claus, R.; Lichter, P. Mutations in regulators of the epigenome and their connections to global chromatin patterns in cancer. Nat. Rev. Genet. 2013, 14, 765–780. [Google Scholar] [CrossRef]
- Sarwar, M.S.; Zhang, H.-J.; Tsang, S.W. Perspectives of Plant Natural Products in Inhibition of Cancer Invasion and Metastasis by Regulating Multiple Signaling Pathways. Curr. Med. Chem. 2018, 25, 5057–5087. [Google Scholar] [CrossRef]
- Gascoigne, K.E.; Taylor, S.S. How do anti-mitotic drugs kill cancer cells? J. Cell Sci. 2009, 122, 2579–2585. [Google Scholar] [CrossRef]
- Jordan, M.A.; Wilson, L. Microtubules as a target for anticancer drugs. Nat. Rev. Cancer 2004, 4, 253–265. [Google Scholar] [CrossRef]
- Najem, S.; Langemann, D.; Appl, B.; Trochimiuk, M.; Hundsdoerfer, P.; Reinshagen, K.; Eschenburg, G. Smac mimetic LCL161 supports neuroblastoma chemotherapy in a drug class-dependent manner and synergistically interacts with ALK inhibitor TAE684 in cells with ALK mutation F1174L. Oncotarget 2016, 7, 72634–72653. [Google Scholar] [CrossRef][Green Version]
- Zhu, R.-H.; Li, H.-D.; Cai, H.-L.; Jiang, Z.-P.; Xu, P.; Dai, L.-B.; Peng, W.-X. Validated HILIC–MS/MS assay for determination of vindesine in human plasma: Application to a population pharmacokinetic study. J. Pharm. Biomed. Anal. 2014, 96, 31–36. [Google Scholar] [CrossRef]
- Almagro, L. Indole Alkaloids from Catharanthus roseus: Bioproduction and Their Effect on Human Health. Molecules 2015, 20, 2973–3000. [Google Scholar] [CrossRef]
- Rowinsky, E. The Vinca Alkaloids. In Holland-Frei Cancer Medicine, 6th ed.; BC Decker: Hamilton, ON, Canada, 2003. [Google Scholar]
- Mukhtar, E.; Adhami, V.M.; Mukhtar, H. Targeting microtubules by natural agents for cancer therapy. Mol. Cancer Ther. 2014, 13, 275–284. [Google Scholar] [CrossRef]
- Glikokoniugatów, Z.B.; Zoologii, I. Substancje pochodzenia roślinnego w terapii nowotworów Plant-derived compounds in the treatment of cancer. Postępy Fitoterapii 2017, 18, 203–208. [Google Scholar]
- Moudi, M.; Go, R.; Yien, C.Y.S.; Nazre, M. Vinca alkaloids. Int. J. Prev. Med. 2013, 4, 1231–1235. [Google Scholar]
- Allen, T.M.; Cullis, P.R. Liposomal drug delivery systems: From concept to clinical applications. Adv. Drug Deliv. Rev. 2013, 65, 36–48. [Google Scholar] [CrossRef]
- Lee, C.-T.; Huang, Y.-W.; Yang, C.-H.; Huang, K.-S. Drug delivery systems and combination therapy by using vinca alkaloids. Curr. Top. Med. Chem. 2015, 15, 1491–1500. [Google Scholar] [CrossRef]
- Slobodnick, A.; Shah, B.; Pillinger, M.H.; Krasnokutsky, S. Colchicine: Old and New. Am. J. Med. 2015, 128, 461–470. [Google Scholar] [CrossRef]
- Dalbeth, N.; Lauterio, T.J.; Wolfe, H.R. Mechanism of action of colchicine in the treatment of gout. Clin. Ther. 2014, 36, 1465–1479. [Google Scholar] [CrossRef]
- Ben-Chetrit, E.; Bergmann, S.; Sood, R. Mechanism of the anti-inflammatory effect of colchicine in rheumatic diseases: A possible new outlook through microarray analysis. Rheumatology 2006, 45, 274–282. [Google Scholar] [CrossRef]
- Özkaya, N.; Yalçinkaya, F. Colchicine treatment in children with familial Mediterranean fever. Clin. Rheumatol. 2003, 22, 314–317. [Google Scholar] [CrossRef]
- Leung, Y.Y.; Yao Hui, L.L.; Kraus, V.B. Colchicine-Update on mechanisms of action and therapeutic uses. Semin. Arthritis Rheum. 2015, 45, 341–350. [Google Scholar] [CrossRef]
- Lu, Y.; Chen, J.; Xiao, M.; Li, W.; Miller, D.D. An overview of tubulin inhibitors that interact with the colchicine binding site. Pharm. Res. 2012, 29, 2943–2971. [Google Scholar] [CrossRef]
- Maldonado, E.N.; Patnaik, J.; Mullins, M.R.; Lemasters, J.J. Free Tubulin Modulates Mitochondrial Membrane Potential in Cancer Cells. Cancer Res. 2011, 70, 10192–10201. [Google Scholar] [CrossRef]
- Lin, Z.-Y.; Kuo, C.-H.; Wu, D.-C.; Chuang, W.-L. Anticancer effects of clinically acceptable colchicine concentrations on human gastric cancer cell lines. Kaohsiung J. Med. Sci. 2016, 32, 68–73. [Google Scholar] [CrossRef]
- Banerjee, S.; Hwang, D.J.; Li, W.; Miller, D.D. Current Advances of Tubulin Inhibitors in Nanoparticle Drug Delivery and Vascular Disruption/Angiogenesis. Molecules 2016, 21, 1468. [Google Scholar] [CrossRef]
- Tangutoori, S.; Ohta, A.; Gatley, S.; Campbell, R.B. Repurposing an Erstwhile Cancer Drug: A Quantitative and Therapeutic Evaluation of Alternative Nanosystems for the Delivery of Colchicine to Solid Tumors. J. Cancer Sci. Ther. 2014, 6, 236–246. [Google Scholar] [CrossRef]
- Hearn, B.R.; Shaw, S.J.; Myles, D.C. Microtubule Targeting Agents. In Comprehensive Medicinal Chemistry II; Elsevier: Amsterdam, The Netherlands, 2007; pp. 81–110. ISBN 9780080450445. [Google Scholar]
- Zygadlo, J.A.; Gleiser, R.M. Chemical Ecology in Relation to Medicine and Pharmaceuticals. In Chemical Ecology; EOLSS Publications: Oxford, UK, 2009; pp. 491–504. [Google Scholar]
- Kumar, A.; Kumar, V.; Alegria, A.E.; Malhotra, S.V. Synthetic and application perspectives of azapodophyllotoxins: Alternative scaffolds of podophyllotoxin. Curr. Med. Chem. 2011, 18, 3853–3870. [Google Scholar] [CrossRef]
- Ardalani, H. Podophyllotoxin: A novel potential natural anticancer agent. Avicenna J. Phytomed. 2017, 7, 285–294. [Google Scholar]
- The Royal Society Of Chemistry ChemSpider. Search and Share Chemistry. Available online: http://www.chemspider.com/ (accessed on 25 March 2019).
- Rai, V.; Tandon, P.K.; Khatoon, S. Effect of chromium on antioxidant potential of Catharanthus roseus varieties and production of their anticancer alkaloids: Vincristine and vinblastine. Biomed. Res. Int. 2014, 2014, 934182. [Google Scholar] [CrossRef]
- Aapro, M.; Finek, J. Oral vinorelbine in metastatic breast cancer: A review of current clinical trial results. Cancer Treat. Rev. 2012, 38, 120–126. [Google Scholar] [CrossRef]
- Gerullis, H.; Wawroschek, F.; Köhne, C.-H.; Ecke, T.H. Vinflunine in the treatment of advanced urothelial cancer: Clinical evidence and experience. Ther. Adv. Urol. 2017, 9, 28–35. [Google Scholar] [CrossRef]
- Cortés, F.; Pastor, N.; Mateos, S.; Domínguez, I. Roles of DNA topoisomerases in chromosome segregation and mitosis. Mutat. Res. Rev. Mutat. Res. 2003, 543, 59–66. [Google Scholar] [CrossRef]
- Mittal, B.; Tulsyan, S.; Kumar, S.; Mittal, R.D.; Agarwal, G. Cytochrome P450 in Cancer Susceptibility and Treatment; Elsevier: Amsterdam, The Netherlands, 2015; ISBN 9780128022566. [Google Scholar]
- Hande, K.R. Etoposide: Four decades of development of a topoisomerase II inhibitor. Eur. J. Cancer 1998, 34, 1514–1521. [Google Scholar] [CrossRef]
- Smith, M.A. Etoposide and Teniposide. W Treatment of Acute Leukemias; Humana Press: Totowa, NJ, USA, 2003; pp. 429–437. [Google Scholar]
- Young, R.C. Etoposide in the treatment of non-Hodgkin’s lymphomas. Semin. Oncol. 1992, 19, 19–25. [Google Scholar]
- Hande, K.R. Topoisomerase II inhibitors. Update Cancer Ther. 2008, 3, 13–26. [Google Scholar] [CrossRef]
- Stanton, R.A.; Gernert, K.M.; Nettles, J.H.; Aneja, R. Drugs That Target Dynamic Microtubules: A New Molecular Perspective. Med. Res. Rev. 2011, 31, 443–481. [Google Scholar] [CrossRef]
- Schwab, C.L.; English, D.P.; Roque, D.M.; Santin, A.D. Taxanes: Their impact on gynecologic malignancy. Anticancer. Drugs 2014, 25, 522–535. [Google Scholar] [CrossRef]
- Markman, M.; Mekhail, T.M. Paclitaxel in cancer therapy. Expert Opin. Pharmacother. 2002, 3, 755–766. [Google Scholar] [CrossRef]
- Prabhash, K.; Noronha, V.; Patil, V.; Bhosale, B.; Joshi, A.; Purandare, N. Metronomic weekly paclitaxel in advanced unresectable esophageal cancer. Indian J. Cancer 2013, 50, 128. [Google Scholar] [CrossRef]
- Yared, J.A.; Tkaczuk, K.H.R. Update on taxane development: New analogs and new formulations. Drug Des. Dev. Ther. 2012, 6, 371–384. [Google Scholar]
- Rowinsky, E.K. The development and clinical utility of the taxane class of antimicrotubule chemotherapy agents. Annu. Rev. Med. 1997, 48, 353–374. [Google Scholar] [CrossRef]
- Zhang, D.; Yang, R.; Wang, S.; Dong, Z. Paclitaxel: New uses for an old drug. Drug Des. Dev. Ther. 2014, 8, 279–284. [Google Scholar]
- Crown, J.; O’Leary, M.; Ooi, W.-S. Docetaxel and paclitaxel in the treatment of breast cancer: A review of clinical experience. Oncologist 2004, 9 (Suppl. 2), 24–32. [Google Scholar] [CrossRef]
- Katsumata, N. Docetaxel: An alternative taxane in ovarian cancer. Br. J. Cancer 2003, 89, S9–S15. [Google Scholar] [CrossRef] [PubMed]
- Nabell, L.; Spencer, S. Docetaxel with concurrent radiotherapy in head and neck cancer. Semin. Oncol. 2003, 30, 89–93. [Google Scholar] [CrossRef] [PubMed]
- Heys, S.D.; Sarkar, T.; Hutcheon, A.W. Primary docetaxel chemotherapy in patients with breast cancer: Impact on response and survival. Breast Cancer Res. Treat. 2005, 90, 169–185. [Google Scholar] [CrossRef] [PubMed]
- He, X.; Wang, J.; Li, Y. Efficacy and safety of docetaxel for advanced non-small-cell lung cancer: A meta-analysis of phase iii randomized controlled trials. OncoTargets Ther. 2015, 8, 2023–2031. [Google Scholar] [CrossRef] [PubMed]
- Tetzlaff, E.D.; Cheng, J.D.; Ajani, J.A. Review of docetaxel in the treatment of gastric cancer. Ther. Clin. Risk Manag. 2008, 4, 999–1007. [Google Scholar] [CrossRef] [PubMed]
- Albany, C.; Sonpavde, G. Docetaxel for the treatment of bladder cancer. Expert Opin. Investig. Drugs 2015, 24, 1657–1664. [Google Scholar] [CrossRef] [PubMed]
- Alberts, B.; Johnson, A.; Lewis, J.; Raff, M.; Roberts, K.; Walter, P. Molecular Biology of the Cell; Garland Science: Boca Raton, FL, USA, 2002; ISBN 0815332181. [Google Scholar]
- Pommier, Y. DNA topoisomerase I inhibitors: Chemistry, biology, and interfacial inhibition. Chem. Rev. 2009, 109, 2894–2902. [Google Scholar] [CrossRef] [PubMed]
- Wieczorek, M.; Sobiak, S.; Meissner, R.K. Rozwój badań nad wykorzystaniem substancji pochodzenia roślinnego w terapii nowotworów. Now. Lek. 2006, 75, 407–413. [Google Scholar]
- O’Reilly, S.; Rowinsky, E.K. The clinical status of irinotecan (CPT-11), a novel water soluble camptothecin analogue: 1996. Crit. Rev. Oncol. Hematol. 1996, 24, 47–70. [Google Scholar] [CrossRef]
- Creemers, G.J.; Wanders, J.; Gamucci, T.; Vallentin, S.; Dirix, L.Y.; Schöffski, P.; Hudson, I.; Verweij, J. Topotecan in colorectal cancer: A phase II study of the EORTC early clinical trials group. Ann. Oncol. 1995, 6, 844–846. [Google Scholar] [CrossRef] [PubMed]
- Sevinc, A.; Kalender, M.E.; Altinbas, M.; Ozkan, M.; Dikilitas, M.; Camci, C.; Anatolian Society of Medical Oncology (ASMO). Irinotecan as a second-line monotherapy for small cell lung cancer. Asian Pac. J. Cancer Prev. 2011, 12, 1055–1059. [Google Scholar] [PubMed]
- Wu, G.; Qi, X.; Mo, X.; Yu, G.; Wang, Q.; Zhu, T.; Gu, Q.; Liu, M.; Li, J.; Li, D. Structure-based discovery of cytotoxic dimeric tetrahydroxanthones as potential topoisomerase I inhibitors from a marine-derived fungus. Eur. J. Med. Chem. 2018, 148, 268–278. [Google Scholar] [CrossRef] [PubMed]
- Rosen, L.S. Irinotecan in lymphoma, leukemia, and breast, pancreatic, ovarian, and small-cell lung cancers. Oncology 1998, 12, 103–109. [Google Scholar] [PubMed]
- Gerrits, C.J.; Burris, H.; Schellens, J.H.; Planting, A.S.; van den Burg, M.E.; Rodriguez, G.; van Beurden, V.; Loos, W.; Hudson, I.; Fields, S. Five days of oral topotecan (hycamtin®), a phase I and pharmacological study in adult patients with solid tumours. Eur. J. Cancer 1998, 34, 1030–1035. [Google Scholar] [CrossRef]
- Björkholm, M. Etoposide and teniposide in the treatment of acute leukemia. Med. Oncol. Tumor Pharmacother. 1990, 7, 3–10. [Google Scholar]
- Slevin, M.L.; Clark, P.I.; Joel, S.P.; Malik, S.; Osborne, R.J.; Gregory, W.M.; Lowe, D.G.; Reznek, R.H.; Wrigley, P.F. A randomized trial to evaluate the effect of schedule on the activity of etoposide in small-cell lung cancer. J. Clin. Oncol. 1989, 7, 1333–1340. [Google Scholar] [CrossRef]
- Loehrer, P.J. Etoposide therapy for testicular cancer. Cancer 1991, 67, 220–224. [Google Scholar] [CrossRef]
- Taylor, R.E.; McElwain, T.J.; Barrett, A.; Peckham, M.J. Etoposide as a single agent in relapsed advanced lymphomas. Cancer Chemother. Pharmacol. 1982, 7, 175–177. [Google Scholar] [CrossRef]
- Sheweita, S.A.; Tilmisany, A.K. Cancer and phase II drug-metabolizing enzymes. Curr. Drug Metab. 2003, 4, 45–58. [Google Scholar] [CrossRef] [PubMed]
- Croom, E. Metabolism of Xenobiotics of Human Environments. In Progress in Molecular Biology and Translational Science; Academic Press: Cambridge, MA, USA, 2012; pp. 31–88. [Google Scholar]
- Hayes, J.D.; Flanagan, J.U.; Jowsey, I.R. Glutatione transperases. Annu. Rev. Pharmacol. Toxicol. 2005, 45, 51–88. [Google Scholar] [CrossRef] [PubMed]
- Usta, M.; Wortelboer, H.M.; Vervoort, J.; Boersma, M.G.; Rietjens, I.M.C.M.; van Bladeren, P.J.; Cnubben, N.H.P. Human Glutathione S-Transferase-Mediated Glutathione Conjugation of Curcumin and Efflux of These Conjugates in Caco-2 Cells. Chem. Res. Toxicol. 2007, 20, 1895–1902. [Google Scholar] [CrossRef] [PubMed]
- Tew, K.D.; Manevich, Y.; Grek, C.; Xiong, Y.; Uys, J.; Townsend, D.M. The role of glutathione S-transferase P in signaling pathways and S-glutathionylation in cancer. Free Radic. Biol. Med. 2011, 51, 299–313. [Google Scholar] [CrossRef] [PubMed]
- Ross, D. Quinone Reductases Multitasking in the Metabolic World. Drug Metab. Rev. 2004, 36, 639–654. [Google Scholar] [CrossRef] [PubMed]
- Siegel, D.; Kepa, J.K.; Ross, D. Biochemical and Genetic Analysis of NAD(P)H:Quinone Oxidoreductase 1 (NQO1). In Current Protocols in Toxicology; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2007; Chapter 4; p. Unit4.22. [Google Scholar]
- Osman, A.G.; Chittiboyina, A.G.; Khan, I.A. Cytoprotective Role of Dietary Phytochemicals Against Cancer Development via Induction of Phase II and Antioxidant Enzymes. Adv. Mol. Toxicol. 2016, 10, 99–137. [Google Scholar]
- Talalay, P. Chemoprotection against cancer by induction of phase 2 enzymes. Biofactors 2000, 12, 5–11. [Google Scholar] [CrossRef] [PubMed]
- Krajka-Kuźniak, V. Induction of phase II enzymes as a strategy in the chemoprevention of cancer and other degenerative diseases. Postepy Hig. Med. Dosw. 2007, 61, 627–638. [Google Scholar]
- Begleiter, A.; Leith, M.K.; Curphey, T.J.; Doherty, G.P. Induction of DT-diaphorase in cancer chemoprevention and chemotherapy. Oncol. Res. 1997, 9, 371–382. [Google Scholar]
- González-Burgos, E.; Gómez-Serranillos, M.P. Terpene compounds in nature: A review of their potential antioxidant activity. Curr. Med. Chem. 2012, 19, 5319–5341. [Google Scholar] [CrossRef]
- Sasaki, S.; Tozawa, T.; Sugamoto, K.; Matsushita, Y.; Satoh, T. A novel di terpene para-hydroquinone compound derived from cryptoquinone protects neuronal cells against oxidative stress and activates the Nrf2/ARE pathway. Neurosci. Lett. 2013, 548, 132–136. [Google Scholar] [CrossRef] [PubMed]
- Ma, Q. Role of nrf2 in oxidative stress and toxicity. Annu. Rev. Pharmacol. Toxicol. 2013, 53, 401–426. [Google Scholar] [CrossRef] [PubMed]
- Cavin, C.; Bezencon, C.; Guignard, G.; Schilter, B. Coffee diterpenes prevent benzo[a]pyrene genotoxicity in rat and human culture systems. Biochem. Biophys. Res. Commun. 2003, 306, 488–495. [Google Scholar] [CrossRef]
- Huber, W.W.; Prustomersky, S.; Delbanco, E.; Uhl, M.; Scharf, G.; Turesky, R.J.; Thier, R.; Schulte-Hermann, R. Enhancement of the chemoprotective enzymes glucuronosyl transferase and glutathione transferase in specific organs of the rat by the coffee components kahweol and cafestol. Arch. Toxicol. 2002, 76, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Garca-Piñeres, A.; Lindenmeyer, M.; Merfort, I. Role of cysteine residues of p65/NF-κB on the inhibition by the sesquiterpene lactone parthenolide and N-ethyl maleimide, and on its transactivating potential. Life Sci. 2004, 75, 841–856. [Google Scholar] [CrossRef]
- Guzman, M.L. The sesquiterpene lactone parthenolide induces apoptosis of human acute myelogenous leukemia stem and progenitor cells. Blood 2005, 105, 4163–4169. [Google Scholar] [CrossRef]
- Woynarowski, J.M.; Konopa, J. Inhibition of DNA biosynthesis in HeLa cells by cytotoxic and antitumor sesquiterpene lactones. Mol. Pharmacol. 1981, 19, 97–102. [Google Scholar]
- Pajak, B.; Gajkowska, B.; Orzechowski, A. Molecular basis of parthenolide-dependent proapoptotic activity in cancer cells. Folia Histochem. Cytobiol. 2008, 46, 129–135. [Google Scholar] [CrossRef]
- Gopal, Y.N.V.; Chanchorn, E.; Van Dyke, M.W. Parthenolide promotes the ubiquitination of MDM2 and activates p53 cellular functions. Mol. Cancer Ther. 2009, 8, 552–562. [Google Scholar] [CrossRef]
- Gopal, Y.N.V.; Arora, T.S.; Van Dyke, M.W. Parthenolide Specifically Depletes Histone Deacetylase 1 Protein and Induces Cell Death through Ataxia Telangiectasia Mutated. Chem. Biol. 2007, 14, 813–823. [Google Scholar] [CrossRef]
- Liu, Z.; Liu, S.; Xie, Z.; Pavlovicz, R.E.; Wu, J.; Chen, P.; Aimiuwu, J.; Pang, J.; Bhasin, D.; Neviani, P. Modulation of DNA Methylation by a Sesquiterpene Lactone Parthenolide. J. Pharmacol. Exp. Ther. 2009, 329, 505–514. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Pang, Q.; Zhou, D.; Zhang, A.; Luo, S.; Wang, Y.; Yan, X. Proteomic investigation into betulinic acid-induced apoptosis of human cervical cancer HeLa cells. PLoS ONE 2014, 9, e105768. [Google Scholar] [CrossRef] [PubMed]
- Tan, Y.; Yu, R.; Pezzuto, J.M. Betulinic acid-induced programmed cell death in human melanoma cells involves mitogen-activated protein kinase activation. Clin. Cancer Res. 2003, 9, 2866–2875. [Google Scholar] [PubMed]
- Alakurtti, S.; Mäkelä, T.; Koskimies, S.; Yli-Kauhaluoma, J. Pharmacological properties of the ubiquitous natural product betulin. Eur. J. Pharm. Sci. 2006, 29, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Fulda, S.; Scaffidi, G.; Susin, S.A.; Krammer, P.H.; Kroemer, G.; Peter, M.E.; Debatin, K.M. Activation of mitochondria and release of mitochondrial apoptogenic factors by betulinic acid. J. Biol. Chem. 1998, 273, 33942–33948. [Google Scholar] [CrossRef] [PubMed]
- Fulda, S.; Debatin, K.-M. Sensitization for anticancer drug-induced apoptosis by betulinic Acid. Neoplasia 2005, 7, 162–170. [Google Scholar] [CrossRef]
- Sawada, N.; Kataoka, K.; Kondo, K.; Arimochi, H.; Fujino, H.; Takahashi, Y.; Miyoshi, T.; Kuwahara, T.; Monden, Y.; Ohnishi, Y. Betulinic acid augments the inhibitory effects of vincristine on growth and lung metastasis of B16F10 melanoma cells in mice. Br. J. Cancer 2004, 90, 1672–1678. [Google Scholar] [CrossRef] [PubMed]
- Kostova, I.; Bhatia, S.; Grigorov, P.; Balkansky, S.; Parmar, V.S.; Prasad, A.K.; Saso, L. Coumarins as antioxidants. Curr. Med. Chem. 2011, 18, 3929–3951. [Google Scholar] [CrossRef]
- Murakami, A.; Kuki, W.; Takahashi, Y.; Yonei, H.; Nakamura, Y.; Ohto, Y.; Ohigashi, H.; Koshimizu, K. Auraptene, a citrus coumarin, inhibits 12-O-tetradecanoylphorbol-13-acetate-induced tumor promotion in ICR mouse skin, possibly through suppression of superoxide generation in leukocytes. Jpn. J. Cancer Res. 1997, 88, 443–452. [Google Scholar] [CrossRef]
- Tanaka, T.; Kawabata, K.; Kakumoto, M.; Hara, A.; Murakami, A.; Kuki, W.; Takahashi, Y.; Yonei, H.; Maeda, M.; Ota, T. Citrus auraptene exerts dose-dependent chemopreventive activity in rat large bowel tumorigenesis: The inhibition correlates with suppression of cell proliferation and lipid peroxidation and with induction of phase II drug-metabolizing enzymes. Cancer Res. 1998, 58, 2550–2556. [Google Scholar]
- Aras, U.; Gandhi, Y.A.; Masso-Welch, P.A.; Morris, M.E. Chemopreventive and anti-angiogenic effects of dietary phenethyl isothiocyanate in an N-methyl nitrosourea-induced breast cancer animal model. Biopharm. Drug Dispos. 2013, 34, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Ni, W.-Y.; Lu, H.-F.; Hsu, S.-C.; Hsiao, Y.-P.; Liu, K.-C.; Liu, J.-Y.; Ji, B.-C.; Hsueh, S.-C.; Hung, F.-M.; Shang, H.-S. Phenethyl isothiocyanate inhibits in vivo growth of subcutaneous xenograft tumors of human malignant melanoma A375.S2 cells. In Vivo 2014, 28, 891–894. [Google Scholar] [PubMed]
- Huang, S.-H.; Hsu, M.-H.; Hsu, S.-C.; Yang, J.-S.; Huang, W.-W.; Huang, A.-C.; Hsiao, Y.-P.; Yu, C.-C.; Chung, J.-G. Phenethyl isothiocyanate triggers apoptosis in human malignant melanoma A375.S2 cells through reactive oxygen species and the mitochondria-dependent pathways. Hum. Exp. Toxicol. 2014, 33, 270–283. [Google Scholar] [CrossRef] [PubMed]
- Moon, Y.J.; Brazeau, D.A.; Morris, M.E. Dietary phenethyl isothiocyanate alters gene expression in human breast cancer cells. Evid. Based Complement. Alternat. Med. 2011, 2011, 462525. [Google Scholar] [CrossRef] [PubMed]
- Yan, H.; Zhu, Y.; Liu, B.; Wu, H.; Li, Y.; Wu, X.; Zhou, Q.; Xu, K. Mitogen-activated protein kinase mediates the apoptosis of highly metastatic human non-small cell lung cancer cells induced by isothiocyanates. Br. J. Nutr. 2011, 106, 1779–1791. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Govind, S.; Sajankila, S.P.; Mi, L.; Roy, R.; Chung, F.-L. Phenethyl isothiocyanate sensitizes human cervical cancer cells to apoptosis induced by cisplatin. Mol. Nutr. Food Res. 2011, 55, 1572–1581. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.-L.; Huang, A.-C.; Yang, J.-S.; Liao, C.-L.; Lu, H.-F.; Chou, S.-T.; Ma, C.-Y.; Hsia, T.-C.; Ko, Y.-C.; Chung, J.-G. Benzyl isothiocyanate (BITC) and phenethyl isothiocyanate (PEITC)-mediated generation of reactive oxygen species causes cell cycle arrest and induces apoptosis via activation of caspase-3, mitochondria dysfunction and nitric oxide (NO) in human osteogenic. J. Orthop. Res. 2011, 29, 1199–1209. [Google Scholar] [CrossRef] [PubMed]
- Xiao, D.; Powolny, A.A.; Moura, M.B.; Kelley, E.E.; Bommareddy, A.; Kim, S.-H.; Hahm, E.-R.; Normolle, D.; Van Houten, B.; Singh, S. V Phenethyl isothiocyanate inhibits oxidative phosphorylation to trigger reactive oxygen species-mediated death of human prostate cancer cells. J. Biol. Chem. 2010, 285, 26558–26569. [Google Scholar] [CrossRef]
- Jakubikova, J.; Cervi, D.; Ooi, M.; Kim, K.; Nahar, S.; Klippel, S.; Cholujova, D.; Leiba, M.; Daley, J.F.; Delmore, J. Anti-tumor activity and signaling events triggered by the isothiocyanates, sulforaphane and phenethyl isothiocyanate, in multiple myeloma. Haematologica 2011, 96, 1170–1179. [Google Scholar] [CrossRef]
- Chou, Y.-C.; Chang, M.-Y.; Wang, M.-J.; Harnod, T.; Hung, C.-H.; Lee, H.-T.; Shen, C.-C.; Chung, J.-G. PEITC induces apoptosis of Human Brain Glioblastoma GBM8401 Cells through the extrinsic- and intrinsic -signaling pathways. Neurochem. Int. 2015, 81, 32–40. [Google Scholar] [CrossRef]
- Wang, L.G.; Beklemisheva, A.; Liu, X.M.; Ferrari, A.C.; Feng, J.; Chiao, J.W. Dual action on promoter demethylation and chromatin by an isothiocyanate restored GSTP1 silenced in prostate cancer. Mol. Carcinog. 2007, 46, 24–31. [Google Scholar] [CrossRef]
- Boyanapalli, S.S.S.; Li, W.; Fuentes, F.; Guo, Y.; Ramirez, C.N.; Gonzalez, X.-P.; Pung, D.; Kong, A.-N.T. Epigenetic reactivation of RASSF1A by phenethyl isothiocyanate (PEITC) and promotion of apoptosis in LNCaP cells. Pharmacol. Res. 2016, 114, 175–184. [Google Scholar] [CrossRef]
- Lea, M.A.; Randolph, V.M.; Lee, J.E.; DesBordes, C. Induction of histone acetylation in mouse erythroleukemia cells by some organosulfur compounds including allyl isothiocyanate. Int. J. Cancer 2001, 92, 784–789. [Google Scholar] [CrossRef] [PubMed]
- Izzotti, A.; Calin, G.A.; Steele, V.E.; Cartiglia, C.; Longobardi, M.; Croce, C.M.; De Flora, S. Chemoprevention of Cigarette Smoke-Induced Alterations of MicroRNA Expression in Rat Lungs. Cancer Prev. Res. 2010, 3, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Izzotti, A.; Larghero, P.; Cartiglia, C.; Longobardi, M.; Pfeffer, U.; Steele, V.E.; De Flora, S. Modulation of microRNA expression by budesonide, phenethyl isothiocyanate and cigarette smoke in mouse liver and lung. Carcinogenesis 2010, 31, 894–901. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Shu, L.; Kim, H.; Khor, T.O.; Wu, R.; Li, W.; Kong, A.-N.T. Phenethyl isothiocyanate (PEITC) suppresses prostate cancer cell invasion epigenetically through regulating microRNA-194. Mol. Nutr. Food Res. 2016, 60, 1427–1436. [Google Scholar] [CrossRef] [PubMed]
- van Lieshout, E.M.; Bedaf, M.M.; Pieter, M.; Ekkel, C.; Nijhoff, W.A.; Peters, W.H. Effects of dietary anticarcinogens on rat gastrointestinal glutathione S-transferase theta 1-1 levels. Carcinogenesis 1998, 19, 2055–2057. [Google Scholar] [CrossRef] [PubMed]
- FIMOGNARI, C.; HRELIA, P. Sulforaphane as a promising molecule for fighting cancer. Mutat. Res. Mutat. Res. 2007, 635, 90–104. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Zhang, T.; Korkaya, H.; Liu, S.; Lee, H.F.; Newman, B.; Yu, Y.; Clouthier, S.G.; Schwartz, S.J.; Wicha, M.S. Sulforaphane, a Dietary Component of Broccoli/Broccoli Sprouts, Inhibits Breast Cancer Stem Cells. Clin. Cancer Res. 2010, 16, 2580–2590. [Google Scholar] [CrossRef] [PubMed]
- Clarke, J.D.; Dashwood, R.H.; Ho, E. Multi-targeted prevention of cancer by sulforaphane. Cancer Lett. 2008, 269, 291–304. [Google Scholar] [CrossRef]
- Mahéo, K.; Morel, F.; Langouët, S.; Kramer, H.; Le Ferrec, E.; Ketterer, B.; Guillouzo, A. Inhibition of cytochromes P-450 and induction of glutathione S-transferases by sulforaphane in primary human and rat hepatocytes. Cancer Res. 1997, 57, 3649–3652. [Google Scholar] [PubMed]
- Abdull Razis, A.F.; Noor, N.M. Sulforaphane is superior to glucoraphanin in modulating carcinogen-metabolising enzymes in Hep G2 cells. Asian Pac. J. Cancer Prev. 2013, 14, 4235–4238. [Google Scholar] [CrossRef] [PubMed]
- Fahey, J.W.; Holtzclaw, W.D.; Wehage, S.L.; Wade, K.L.; Stephenson, K.K.; Talalay, P. Sulforaphane Bioavailability from Glucoraphanin-Rich Broccoli: Control by Active Endogenous Myrosinase. PLoS ONE 2015, 10, e0140963. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, T.A.; Fahey, J.W.; Dinkova-Kostova, A.T.; Holtzclaw, W.D.; Stephenson, K.K.; Wade, K.L.; Ye, L.; Talalay, P. Safety, Tolerance, and Metabolism of Broccoli Sprout Glucosinolates and Isothiocyanates: A Clinical Phase I Study. Nutr. Cancer 2006, 55, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Mut-Salud, N.; Álvarez, P.J.; Garrido, J.M.; Carrasco, E.; Aránega, A.; Rodríguez-Serrano, F. Antioxidant Intake and Antitumor Therapy: Toward Nutritional Recommendations for Optimal Results. Oxid. Med. Cell. Longev. 2016, 2016, 6719534. [Google Scholar] [CrossRef] [PubMed]
- Guyonnet, D.; Belloir, C.; Suschetet, M.; Siess, M.H.; Le Bon, A.M. Antimutagenic activity of organosulfur compounds from Allium is associated with phase II enzyme induction. Mutat. Res. 2001, 495, 135–145. [Google Scholar] [CrossRef]
- Munday, R.; Munday, J.S.; Munday, C.M. Comparative effects of mono-, di-, tri-, and tetrasulfides derived from plants of the Allium family: Redox cycling in vitro and hemolytic activity and Phase 2 enzyme induction in vivo. Free Radic. Biol. Med. 2003, 34, 1200–1211. [Google Scholar] [CrossRef]
- Guyonnet, D.; Siess, M.-H.; Le Bon, A.-M.; Suschetet, M. Modulation of Phase II Enzymes by Organosulfur Compounds from Allium Vegetables in Rat Tissues. Toxicol. Appl. Pharmacol. 1999, 154, 50–58. [Google Scholar] [CrossRef]
- Lea, M.A.; Rasheed, M.; Randolph, V.M.; Khan, F.; Shareef, A.; DesBordes, C. Induction of Histone Acetylation and Inhibition of Growth of Mouse Erythroleukemia Cells by S-Allylmercaptocysteine. Nutr. Cancer 2002, 43, 90–102. [Google Scholar] [CrossRef]
- DRUESNE, N.; PAGNIEZ, A.; MAYEUR, C.; THOMAS, M.; CHERBUY, C.; DUÉE, P.-H.; MARTEL, P.; CHAUMONTET, C. Repetitive Treatments of Colon HT-29 Cells with Diallyl Disulfide Induce a Prolonged Hyperacetylation of Histone H3 K14. Ann. N. Y. Acad. Sci. 2004, 1030, 612–621. [Google Scholar] [CrossRef]
- Nian, H.; Delage, B.; Ho, E.; Dashwood, R.H. Modulation of histone deacetylase activity by dietary isothiocyanates and allyl sulfides: Studies with sulforaphane and garlic organosulfur compounds. Environ. Mol. Mutagen. 2009, 50, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Lea, M.A.; Randolph, V.M.; Patel, M. Increased acetylation of histones induced by diallyl disulfide and structurally related molecules. Int. J. Oncol. 1999, 15, 347–399. [Google Scholar] [CrossRef]
- Druesne, N. Diallyl disulfide (DADS) increases histone acetylation and p21waf1/cip1 expression in human colon tumor cell lines. Carcinogenesis 2004, 25, 1227–1236. [Google Scholar] [CrossRef] [PubMed]
- Busbee, P.B.; Nagarkatti, M.; Nagarkatti, P.S. Natural indoles, indole-3-carbinol (I3C) and 3,3′-diindolylmethane (DIM), attenuate staphylococcal enterotoxin B-mediated liver injury by downregulating miR-31 expression and promoting caspase-2-mediated apoptosis. PLoS ONE 2015, 10, e0118506. [Google Scholar] [CrossRef]
- Nho, C.W.; Jeffery, E. The Synergistic Upregulation of Phase II Detoxification Enzymes by Glucosinolate Breakdown Products in Cruciferous Vegetables. Toxicol. Appl. Pharmacol. 2001, 174, 146–152. [Google Scholar] [CrossRef]
- Saw, C.L.-L.; Cintrón, M.; Wu, T.-Y.; Guo, Y.; Huang, Y.; Jeong, W.-S.; Kong, A.-N.T. Pharmacodynamics of dietary phytochemical indoles I3C and DIM: Induction of Nrf2-mediated phase II drug metabolizing and antioxidant genes and synergism with isothiocyanates. Biopharm. Drug Dispos. 2011, 32, 289–300. [Google Scholar] [CrossRef] [PubMed]
- Acharya, A.; Das, I.; Singh, S.; Saha, T. Chemopreventive properties of indole-3-carbinol, diindolylmethane and other constituents of cardamom against carcinogenesis. Recent Pat. Food Nutr. Agric. 2010, 2, 166–177. [Google Scholar] [CrossRef]
- Ashok, B.T.; Chen, Y.; Liu, X.; Bradlow, H.L.; Mittelman, A.; Tiwari, R.K. Abrogation of Estrogen-Mediated Cellular and Biochemical Effects by Indole-3-Carbinol. Nutr. Cancer 2001, 41, 180–187. [Google Scholar] [CrossRef]
- Ashok, B.T.; Chen, Y.G.; Liu, X.; Garikapaty, V.P.S.; Seplowitz, R.; Tschorn, J.; Roy, K.; Mittelman, A.; Tiwari, R.K. Multiple molecular targets of indole-3-carbinol, a chemopreventive anti-estrogen in breast cancer. Eur. J. Cancer Prev. 2002, 11 (Suppl. 2), S86–S93. [Google Scholar]
- Meng, Q.; Yuan, F.; Goldberg, I.D.; Rosen, E.M.; Auborn, K.; Fan, S. Indole-3-Carbinol Is a Negative Regulator of Estrogen Receptor-α Signaling in Human Tumor Cells. J. Nutr. 2000, 130, 2927–2931. [Google Scholar] [CrossRef]
- Li, Y.; Li, X.; Guo, B. Chemopreventive Agent 3,3′-Diindolylmethane Selectively Induces Proteasomal Degradation of Class I Histone Deacetylases. Cancer Res. 2010, 70, 646–654. [Google Scholar] [CrossRef]
- Beaver, L.M.; Yu, T.-W.; Sokolowski, E.I.; Williams, D.E.; Dashwood, R.H.; Ho, E. 3,3′-Diindolylmethane, but not indole-3-carbinol, inhibits histone deacetylase activity in prostate cancer cells. Toxicol. Appl. Pharmacol. 2012, 263, 345–351. [Google Scholar] [CrossRef]
- Li, Y.; VandenBoom, T.G.; Kong, D.; Wang, Z.; Ali, S.; Philip, P.A.; Sarkar, F.H. Up-regulation of miR-200 and let-7 by Natural Agents Leads to the Reversal of Epithelial-to-Mesenchymal Transition in Gemcitabine-Resistant Pancreatic Cancer Cells. Cancer Res. 2009, 69, 6704–6712. [Google Scholar] [CrossRef]
- Li, Y.; VandenBoom, T.G.; Wang, Z.; Kong, D.; Ali, S.; Philip, P.A.; Sarkar, F.H. miR-146a Suppresses Invasion of Pancreatic Cancer Cells. Cancer Res. 2010, 70, 1486–1495. [Google Scholar] [CrossRef]
- Kong, D.; Heath, E.; Chen, W.; Cher, M.; Powell, I.; Heilbrun, L.; Li, Y.; Ali, S.; Sethi, S.; Hassan, O. Erratum: Epigenetic silencing of miR-34a in human prostate cancer cells and tumor tissue specimens can be reversed by BR-DIM treatment. Am. J. Transl. Res. 2013, 6, 102–103. [Google Scholar]
- Melkamu, T.; Zhang, X.; Tan, J.; Zeng, Y.; Kassie, F. Alteration of microRNA expression in vinyl carbamate-induced mouse lung tumors and modulation by the chemopreventive agent indole-3-carbinol. Carcinogenesis 2010, 31, 252–258. [Google Scholar] [CrossRef]
- Jin, Y. 3,3′-Diindolylmethane inhibits breast cancer cell growth via miR-21-mediated Cdc25A degradation. Mol. Cell. Biochem. 2011, 358, 345–354. [Google Scholar] [CrossRef]
- Busbee, P.B.; Nagarkatti, M.; Nagarkatti, P.S. Natural indoles, indole-3-carbinol and 3,3′-diindolymethane, inhibit T cell activation by staphylococcal enterotoxin B through epigenetic regulation involving HDAC expression. Toxicol. Appl. Pharmacol. 2014, 274, 7–16. [Google Scholar] [CrossRef]
- Millimouno, F.M.; Dong, J.; Yang, L.; Li, J.; Li, X. Targeting apoptosis pathways in cancer and perspectives with natural compounds from mother nature. Cancer Prev. Res. 2014, 7, 1081–1107. [Google Scholar] [CrossRef]
- Zuco, V.; Supino, R.; Righetti, S.C.; Cleris, L.; Marchesi, E.; Gambacorti-Passerini, C.; Formelli, F. Selective cytotoxicity of betulinic acid on tumor cell lines, but not on normal cells. Cancer Lett. 2002, 175, 17–25. [Google Scholar] [CrossRef]
- Fulda, S.; Friesen, C.; Los, M.; Scaffidi, C.; Mier, W.; Benedict, M.; Nuã, G.; Krammer, P.H.; Peter, M.E.; Debatin, K. Betulinic Acid Triggers CD95 ( APO-1/Fas )—And p53-independent Apoptosis via Activation of Caspases in Neuroectodermal Tumors Activation of Caspases in Neuroectodermal Tumors. Cancer Res. 1997, 95, 4956–4964. [Google Scholar]
- Liu, Y.; Zhang, X.; Liu, Z.; Wang, L.; Luo, L.; Wang, M.; Wang, Q.; Gao, D. Gold nanoshell-based betulinic acid liposomes for synergistic chemo-photothermal therapy. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 1891–1900. [Google Scholar] [CrossRef]
- Mullauer, F.B.; Van Bloois, L.; Daalhuisen, J.B.; Ten Brink, M.S.; Storm, G.; Medema, J.P.; Schiffelers, R.M.; Kessler, J.H. Betulinic acid delivered in liposomes reduces growth of human lung and colon cancers in mice without causing systemic toxicity. Anticancer Drugs 2011, 22, 223–233. [Google Scholar] [CrossRef]
- Tan, J.; Karthivashan, G.; Arulselvan, P.; Fakurazi, S.; Hussein, M. Characterization and in vitro studies of the anticancer effect of oxidized carbon nanotubes functionalized with betulinic acid. Drug Des. Dev. Ther. 2014, 20, 2333–2343. [Google Scholar] [CrossRef]
- Olaharski, A.J.; Rine, J.; Marshall, B.L.; Babiarz, J.; Zhang, L.; Verdin, E.; Smith, M.T. The flavoring agent dihydrocoumarin reverses epigenetic silencing and inhibits sirtuin deacetylases. PLoS Genet. 2005, 1, 0689–0694. [Google Scholar] [CrossRef]
- Chen, C.C.; Huang, J.S.; Wang, T.H.; Kuo, C.H.; Wang, C.J.; Wang, S.H.; Leu, Y.L. Dihydrocoumarin, an HDAC inhibitor, increases DNA damage sensitivity by inhibiting Rad52. Int. J. Mol. Sci. 2017, 18, 2655. [Google Scholar] [CrossRef]
- Seidel, C.; Schnekenburger, M.; Zwergel, C.; Gaascht, F.; Mai, A.; Dicato, M.; Kirsch, G.; Valente, S.; Diederich, M. Novel inhibitors of human histone deacetylases: Design, synthesis and bioactivity of 3-alkenoylcoumarines. Bioorg. Med. Chem. Lett. 2014, 24, 3797–3801. [Google Scholar] [CrossRef]
- Abdizadeh, T.; Kalani, M.R.; Abnous, K.; Tayarani-Najaran, Z.; Khashyarmanesh, B.Z.; Abdizadeh, R.; Ghodsi, R.; Hadizadeh, F. Design, synthesis and biological evaluation of novel coumarin-based benzamides as potent histone deacetylase inhibitors and anticancer agents. Eur. J. Med. Chem. 2017, 132, 42–62. [Google Scholar] [CrossRef]
- Wu, X.; Zhou, Q.; Xu, K. Are isothiocyanates potential anti-cancer drugs? Acta Pharmacol. Sin. 2009, 30, 501–512. [Google Scholar] [CrossRef]
- Zhang, Y.; Munday, R.; Jobson, H.E.; Munday, C.M.; Lister, C.; Wilson, P.; Fahey, J.W.; Mhawech-Fauceglia, P. Induction of GST and NQO1 in Cultured Bladder Cells and in the Urinary Bladders of Rats by an Extract of Broccoli (Brassica oleracea italica) Sprouts. J. Agric. Food Chem. 2006, 54, 9370–9376. [Google Scholar] [CrossRef]
- Nune, S.K.; Gunda, P.; Thallapally, P.K.; Lin, Y.-Y.; Forrest, M.L.; Berkland, C.J. Nanoparticles for biomedical imaging. Expert Opin. Drug Deliv. 2009, 6, 1175–1194. [Google Scholar] [CrossRef]
- Khosroshahi, M.E.; Asemani, M. International Journal of Nanomaterials, Nanotechnology and Nanomedicine Synthesis, Characterization and Imaging of Fluorescine Isothiocyanate Conjugated Magnetite Nanoparticles in MCF 7 Breast Cancer Cell Lines. Int. J. Nanomater. Nanotechnol. Nanomed. 2017, 3, 44–50. [Google Scholar]
- Encinas-Basurto, D.; Ibarra, J.; Juarez, J.; Burboa, M.G.; Barbosa, S.; Taboada, P.; Troncoso-Rojas, R.; Valdez, M.A. Poly(lactic-co-glycolic acid) nanoparticles for sustained release of allyl isothiocyanate: Characterization, in vitro release and biological activity. J. Microencapsul. 2017, 34, 231–242. [Google Scholar] [CrossRef]
- Traka, M.; Gasper, A.V.; Smith, J.A.; Hawkey, C.J.; Bao, Y.; Mithen, R.F. Transcriptome analysis of human colon Caco-2 cells exposed to sulforaphane. J. Nutr. 2005, 135, 1865–1872. [Google Scholar] [CrossRef]
- Meeran, S.M.; Patel, S.N.; Tollefsbol, T.O. Sulforaphane Causes Epigenetic Repression of hTERT Expression in Human Breast Cancer Cell Lines. PLoS ONE 2010, 5, e11457. [Google Scholar] [CrossRef]
- Myzak, M.C.; Karplus, P.A.; Chung, F.-L.; Dashwood, R.H. A Novel Mechanism of Chemoprotection by Sulforaphane. Cancer Res. 2004, 64, 5767–5774. [Google Scholar] [CrossRef]
- Myzak, M.C.; Hardin, K.; Wang, R.; Dashwood, R.H.; Ho, E. Sulforaphane inhibits histone deacetylase activity in BPH-1, LnCaP and PC-3 prostate epithelial cells. Carcinogenesis 2006, 27, 811–819. [Google Scholar] [CrossRef]
- Pledgie-Tracy, A.; Sobolewski, M.D.; Davidson, N.E. Sulforaphane induces cell type-specific apoptosis in human breast cancer cell lines. Mol. Cancer Ther. 2007, 6, 1013–1021. [Google Scholar] [CrossRef]
- Wong, C.P.; Hsu, A.; Buchanan, A.; Palomera-Sanchez, Z.; Beaver, L.M.; Houseman, E.A.; Williams, D.E.; Dashwood, R.H.; Ho, E. Effects of Sulforaphane and 3,3′-Diindolylmethane on Genome-Wide Promoter Methylation in Normal Prostate Epithelial Cells and Prostate Cancer Cells. PLoS ONE 2014, 9, e86787. [Google Scholar] [CrossRef]
- Su, Z.Y.; Zhang, C.; Lee, J.H.; Shu, L.; Wu, T.Y.; Khor, T.O.; Conney, A.H.; Lu, Y.P.; Kong, A.N.T. Requirement and epigenetics reprogramming of Nrf2 in suppression of tumor promoter TPA-induced mouse skin cell transformation by sulforaphane. Cancer Prev. Res. 2014, 7, 319–329. [Google Scholar] [CrossRef]
- Myzak, M.C.; Dashwood, W.M.; Orner, G.A.; Ho, E.; Dashwood, R.H. Sulforaphane inhibits histone deacetylase in vivo and suppresses tumorigenesis in Apc min mice. FASEB J. 2006, 20, 506–508. [Google Scholar] [CrossRef]
- Mileo, A.M.; Miccadei, S. Polyphenols as Modulator of Oxidative Stress in Cancer Disease: New Therapeutic Strategies. Oxid. Med. Cell. Longev. 2016, 2016, 6475624. [Google Scholar] [CrossRef]
- Gao, L.; Cheng, D.; Yang, J.; Wu, R.; Li, W.; Kong, A.-N. Sulforaphane epigenetically demethylates the CpG sites of the miR-9-3 promoter and reactivates miR-9-3 expression in human lung cancer A549 cells. J. Nutr. Biochem. 2018, 56, 109–115. [Google Scholar] [CrossRef]
- Beaver, L.M.; Lӧhr, C.V.; Clarke, J.D.; Glasser, S.T.; Watson, G.W.; Wong, C.P.; Zhang, Z.; Williams, D.E.; Dashwood, R.H.; Shannon, J. Broccoli Sprouts Delay Prostate Cancer Formation and Decrease Prostate Cancer Severity with a Concurrent Decrease in HDAC3 Protein Expression in Transgenic Adenocarcinoma of the Mouse Prostate (TRAMP) Mice. Curr. Dev. Nutr. 2018, 2, nzy002. [Google Scholar] [CrossRef]
- Myzak, M.C.; Tong, P.; Dashwood, W.-M.; Dashwood, R.H.; Ho, E. Sulforaphane retards the growth of human PC-3 xenografts and inhibits HDAC activity in human subjects. Exp. Biol. Med. 2007, 232, 227–234. [Google Scholar]
- Jadhav, U.; Ezhilarasan, R.; Vaughn, S.F.; Berhow, M.A.; Mohanam, S. Dietary isothiocyanate iberin inhibits growth and induces apoptosis in human glioblastoma cells. J. Pharmacol. Sci. 2007, 103, 247–251. [Google Scholar] [CrossRef]
- Jakubikova, J.; Bao, Y.; Bodo, J.; Sedlak, J. Isothiocyanate iberin modulates phase II enzymes, posttranslational modification of histones and inhibits growth of Caco-2 cells by inducing apoptosis. Neoplasma 2006, 53, 463–470. [Google Scholar]
- Kore, A.M.; Jeffery, E.H.; Wallig, M.A. Effects of 1-isothiocyanato-3-(methylsulfinyl)-propane on xenobiotic metabolizing enzymes in rats. Food Chem. Toxicol. 1993, 31, 723–729. [Google Scholar] [CrossRef]
- Wang, S.; Cheng, L.; Liu, Y.; Wang, J.; Jiang, W. Indole-3-Carbinol (I3C) and its Major Derivatives: Their Pharmacokinetics and Important Roles in Hepatic Protection. Curr. Drug Metab. 2016, 17, 401–409. [Google Scholar] [CrossRef]
- Fan, S.; Meng, Q.; Auborn, K.; Carter, T.; Rosen, E.M. BRCA1 and BRCA2 as molecular targets for phytochemicals indole-3-carbinol and genistein in breast and prostate cancer cells. Br. J. Cancer 2006, 94, 407–426. [Google Scholar] [CrossRef]
- White, A.I.; Jenkins, G.L. Salvia Carnosa (Dougl.). I—A Phytochemical Study**Abstracted from a part of the thesis presented to the Graduate Faculty of the University of Minnesota by Allen I. White in partial fulfilment of the requirements for the degree of Doctor of Philosophy. J. Am. Pharm. Assoc. 1942, 31, 33–37. [Google Scholar] [CrossRef]
- Brieskorn, C.H.; Fuchs, A.; Bredenberg, J.B.S.; McChesney, J.D.; Wenkert, E. The Structure of Carnosol. J. Org. Chem. 1964, 29, 2293–2298. [Google Scholar] [CrossRef]
- Chun, K.-S.; Kundu, J.K.J.; Chae, I.G.; Kundu, J.K.J. Carnosol: A Phenolic Diterpene With Cancer Chemopreventive Potential. J. Cancer Prev. 2014, 19, 103–110. [Google Scholar] [CrossRef]
- Huang, M.T.; Ho, C.T.; Wang, Z.Y.; Ferraro, T.; Lou, Y.R.; Stauber, K.; Ma, W.; Georgiadis, C.; Laskin, J.D.; Conney, A.H. Inhibition of skin tumorigenesis by rosemary and its constituents carnosol and ursolic acid. Cancer Res. 1994, 54, 701–708. [Google Scholar]
- Samarghandian, S.; Azimi-Nezhad, M.; Farkhondeh, T. Anti-Carcinogenic Effects of Carnosol-An Updated Review. Curr. Drug Discov. Technol. 2018, 15, 32–40. [Google Scholar] [CrossRef]
- Giacomelli, C.; Daniele, S.; Natali, L.; Iofrida, C.; Flamini, G.; Braca, A.; Trincavelli, M.L.; Martini, C. Carnosol controls the human glioblastoma stemness features through the epithelial-mesenchymal transition modulation and the induction of cancer stem cell apoptosis. Sci. Rep. 2017, 7, 15174. [Google Scholar] [CrossRef]
- Wang, L.; Zhang, Y.; Liu, K.; Chen, H.; Yang, R.; Ma, X.; Kim, H.-G.; Bode, A.M.; Kim, D.J.; Dong, Z. Carnosol suppresses patient-derived gastric tumor growth by targeting RSK2. Oncotarget 2018, 9, 34200. [Google Scholar] [CrossRef]
- Singletary, K.; MacDonald, C.; Wallig, M. Inhibition by rosemary and carnosol of 7,12-dimethylbenz[a]anthracene (DMBA)-induced rat mammary tumorigenesis and in vivo DMBA-DNA adduct formation. Cancer Lett. 1996, 104, 43–48. [Google Scholar] [CrossRef]
- Vergara, D.; Simeone, P.; Toraldo, D.; Del Boccio, P.; Vergaro, V.; Leporatti, S.; Pieragostino, D.; Tinelli, A.; De Domenico, S.; Alberti, S. Resveratrol downregulates Akt/GSK and ERK signalling pathways in OVCAR-3 ovarian cancer cells. Mol. Biosyst. 2012, 8, 1078–1087. [Google Scholar] [CrossRef]
- Justo, O.R.; Simioni, P.U.; Gabriel, D.L.; Tamashiro WM DS, C.; Rosa PD, T.V.; Moraes Â, M. Evaluation of in vitro anti-inflammatory effects of crude ginger and rosemary extracts obtained through supercritical CO2 extraction on macrophage and tumor cell line: The influence of vehicle type. BMC Complement. Altern. Med. 2015, 15, 390. [Google Scholar] [CrossRef]
- Frémont, L. Biological effects of resveratrol. Life Sci. 2000, 66, 663–673. [Google Scholar] [CrossRef]
- Wang, Y.; Catana, F.; Yang, Y.; Roderick, R.; Van Breemen, R.B. An LC-MS Method for Analyzing Total Resveratrol in Grape Juice, Cranberry Juice, and in Wine. J. Agric. Food Chem. 2002, 50, 431–435. [Google Scholar] [CrossRef]
- Lyons, M.M.; Yu, C.; Toma, R.B.; Cho, S.Y.; Reiboldt, W.; Lee, J.; Van Breemen, R.B. Resveratrol in Raw and Baked Blueberries and Bilberries. J. Agric. Food Chem. 2003, 51, 5867–5870. [Google Scholar] [CrossRef]
- Tokuşoǧlu, Ö.; Ünal, M.K.; Yemiş, F. Determination of the Phytoalexin Resveratrol (3,5,4‘-Trihydroxystilbene) in Peanuts and Pistachios by High-Performance Liquid Chromatographic Diode Array (HPLC-DAD) and Gas Chromatography−Mass Spectrometry (GC-MS). J. Agric. Food Chem. 2005, 53, 5003–5009. [Google Scholar] [CrossRef]
- Siemann, E.H.; Creasy, L.L. Concentration of the Phytoalexin Resveratrol in Wine. Am. J. Enol. Vitic. 1992, 43, 49–52. [Google Scholar]
- Berman, A.Y.; Motechin, R.A.; Wiesenfeld, M.Y.; Holz, M.K. The therapeutic potential of resveratrol: a review of clinical trials. NPJ Precis. Oncol. 2017, 1, 35. [Google Scholar] [CrossRef]
- Varoni, E.M.; Faro, A.F.L.; Sharifi-Rad, J.; Iriti, M. Anticancer Molecular Mechanisms of Resveratrol. Front. Nutr. 2016, 3, 8. [Google Scholar] [CrossRef]
- Kulkarni, S.S.; Cantó, C. The molecular targets of resveratrol. Biochim. et Biophys. Acta (BBA) - Mol. Basis Dis. 2015, 1852, 1114–1123. [Google Scholar] [CrossRef]
- Sajish, M.; Schimmel, P. A human tRNA synthetase is a potent PARP1-activating effector target for resveratrol. Nature 2015, 519, 370. [Google Scholar] [CrossRef]
- Farhan, M.; Ullah, M.F.; Faisal, M.; Farooqi, A.A.; Sabitaliyevich, U.Y.; Biersack, B.; Ahmad, A. Differential Methylation and Acetylation as the Epigenetic Basis of Resveratrol’s Anticancer Activity. Medicines 2019, 6, 24. [Google Scholar] [CrossRef]
- Dhar, S.; Kumar, A.; Rimando, A.M.; Zhang, X.; Levenson, A.S. Resveratrol and pterostilbene epigenetically restore PTEN expression by targeting oncomiRs of the miR-17 family in prostate cancer. Oncotarget 2015, 6, 27214–27226. [Google Scholar] [CrossRef] [PubMed]
- Azimi, A.; Hagh, M.F.; Talebi, M.; Yousefi, B.; Hossein pour feizi, A.A.; Baradaran, B.; Movassaghpour, A.A.; Shamsasenjan, K.; Khanzedeh, T.; Ghaderi, A.H. Time-and Concentration-Dependent Effects of Resveratrol on miR 15a and miR16-1 Expression and Apoptosis in the CCRF-CEM Acute Lymphoblastic Leukemia Cell Line. Asian Pac. J. Cancer Prev. 2015, 16, 6463–6468. [Google Scholar] [CrossRef] [PubMed]
- Qin, W.; Zhang, K.; Clarke, K.; Weiland, T.; Sauter, E.R. Methylation and miRNA Effects of Resveratrol on Mammary Tumors vs. Normal Tissue. Nutr. Cancer 2014, 66, 270–277. [Google Scholar] [CrossRef] [PubMed]
- Summerlin, N.; Soo, E.; Thakur, S.; Qu, Z.; Jambhrunkar, S.; Popat, A. Resveratrol nanoformulations: Challenges and opportunities. Int. J. Pharm. 2015, 479, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Krüger, S.; Bergin, A.; Morlock, G.E. Effect-directed analysis of ginger (Zingiber officinale) and its food products, and quantification of bioactive compounds via high-performance thin-layer chromatography and mass spectrometry. Food Chem. 2018, 243, 258–268. [Google Scholar] [CrossRef] [PubMed]
- Zadeh, J.B.; Kor, N.M. Physiological and pharmaceutical effects of Ginger (Zingiber officinale Roscoe) as a valuable medicinal plant. Eur. J. Exp. Biol. 2014, 4, 87–90. [Google Scholar]
- Chrubasik, S.; Pittler, M.H.; Roufogalis, B.D. Zingiberis rhizoma: A comprehensive review on the ginger effect and efficacy profiles. Phytomedicine 2005, 12, 684–701. [Google Scholar] [CrossRef]
- Ali, B.H.; Blunden, G.; Tanira, M.O.; Nemmar, A. Some phytochemical, pharmacological and toxicological properties of ginger (Zingiber officinale Roscoe): A review of recent research. Food Chem. Toxicol. 2008, 46, 409–420. [Google Scholar] [CrossRef]
- Swapna Sonale, R.; Kadimi, U.S. Characterization of gingerol analogues in supercritical carbon dioxide (SC CO2) extract of ginger (Zingiber officinale, R.,). J. Food Sci. Technol. 2014, 51, 3383–3389. [Google Scholar] [CrossRef]
- Kim, S.O.; Kundu, J.K.; Shin, Y.K.; Park, J.H.; Cho, M.H.; Kim, T.Y.; Surh, Y.J. [6]-Gingerol inhibits COX-2 expression by blocking the activation of p38 MAP kinase and NF-κB in phorbol ester-stimulated mouse skin. Oncogene 2005, 24, 2558–2567. [Google Scholar] [CrossRef]
- Surh, Y.J. Anti-tumor promoting potential of selected spice ingredients with antioxidative and anti-inflammatory activities: A short review. Food Chem. Toxicol. 2002, 40, 1091–1097. [Google Scholar] [CrossRef]
- Kim, E.C.; Min, J.K.; Kim, T.Y.; Lee, S.J.; Yang, H.O.; Han, S.; Kim, Y.M.; Kwon, Y.G. [6]-Gingerol, a pungent ingredient of ginger, inhibits angiogenesis in vitro and in vivo. Biochem. Biophys. Res. Commun. 2005, 335, 300–308. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Wei, Q.; Yang, Q.; Cao, X.; Li, Q.; Shi, F.; Tong, S.S.; Feng, C.; Yu, Q.; Yu, J. A novel formulation of [6]-gingerol: Proliposomes with enhanced oral bioavailability and antitumor effect. Int. J. Pharm. 2018, 535, 308–315. [Google Scholar] [CrossRef] [PubMed]
- Rastogi, N.; Gara, R.K.; Trivedi, R.; Singh, A.; Dixit, P.; Maurya, R.; Duggal, S.; Bhatt, M.L.B.; Singh, S.; Mishra, D.P. (6)-Gingerolinduced myeloid leukemia cell death is initiated by reactive oxygen species and activation of miR-27b expression. Free Radic. Biol. Med. 2014, 68, 288–301. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.S.; Seo, E.Y.; Kang, N.E.; Kim, W.K. [6]-Gingerol inhibits metastasis of MDA-MB-231 human breast cancer cells. J. Nutr. Biochem. 2008, 19, 313–319. [Google Scholar] [CrossRef] [PubMed]
- Shukla, Y.; Prasad, S.; Tripathi, C.; Singh, M.; George, J.; Kalra, N. In vitro and in vivo modulation of testosterone mediated alterations in apoptosis related proteins by [6]-gingerol. Mol. Nutr. Food Res. 2007, 51, 1492–1502. [Google Scholar] [CrossRef] [PubMed]
- Weng, C.J.; Wu, C.F.; Huang, H.W.; Ho, C.T.; Yen, G.C. Anti-invasion effects of 6-shogaol and 6-gingerol, two active components in ginger, on human hepatocarcinoma cells. Mol. Nutr. Food Res. 2010, 54, 1618–1627. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.B.; Lin, C.C.; Tsay, G.J. 6-gingerol inhibits growth of colon cancer cell LoVo via induction of G2/M arrest. Evid.-Based Complement. Altern. Med. 2012, 2012, 326096. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.H.; Kim, D.W.; Jung, C.H.; Lee, Y.J.; Park, D. Gingerol sensitizes TRAIL-induced apoptotic cell death of glioblastoma cells. Toxicol. Appl. Pharmacol. 2014, 279, 253–265. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.J.; Wen, J.; Bang, S.; Park, S.W.; Song, S.Y. [6]-Gingerol Induces Cell Cycle Arrest and Cell Death of Mutant p53-expressing Pancreatic Cancer Cells. Yonsei Med. J. 2006, 47, 688–697. [Google Scholar] [CrossRef]
- Oyagbemi, A.A.; Saba, A.B.; Azeez, O.I. Molecular targets of [6]-gingerol: Its potential roles in cancer chemoprevention. BioFactors 2010, 36, 169–178. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.Y.; Lee, K.C.; Chen, S.Y.; Chang, H.H. 6-Gingerol inhibits ROS and iNOS through the suppression of PKC-α and NF-κB pathways in lipopolysaccharide-stimulated mouse macrophages. Biochem. Biophys. Res. Commun. 2009, 382, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, D.; Bishayee, K.; Ghosh, S.; Biswas, R.; Kumar Mandal, S.; Rahman Khuda-Bukhsh, A. [6]-Gingerol induces caspase 3 dependent apoptosis and autophagy in cancer cells: Drug-DNA interaction and expression of certain signal genes in HeLa cells. Eur. J. Pharmacol. 2012, 694, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Nigam, N.; George, J.; Srivastava, S.; Roy, P.; Bhui, K.; Singh, M.; Shukla, Y. Induction of apoptosis by [6]-gingerol associated with the modulation of p53 and involvement of mitochondrial signaling pathway in B[a]P-induced mouse skin tumorigenesis. Cancer Chemother. Pharmacol. 2010, 65, 687–696. [Google Scholar] [CrossRef] [PubMed]
- Bode, A.M.; Ma, W.Y.; Surh, Y.J.; Dong, Z. Inhibition of epidermal growth factor-induced cell transformation and activator protein 1 activation by [6]-gingerol. Cancer Res. 2001, 61, 850–853. [Google Scholar] [PubMed]
- Citronberg, J.; Bostick, R.; Ahearn, T.; Turgeon, D.K.; Ruffin, M.T.; Djuric, Z.; Sen, A.; Brenner, D.E.; Zick, S.M. Effects of ginger supplementation on cell-cycle biomarkers in the normal-appearing colonic mucosa of patients at increased risk for colorectal cancer: Results from a pilot, randomized, and controlled trial. Cancer Prev. Res. 2013, 6, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Plengsuriyakarn, T.; Viyanant, V.; Eursitthichai, V.; Picha, P.; Kupradinun, P.; Itharat, A.; Na-Bangchang, K. Anticancer activities against cholangiocarcinoma, toxicity and pharmacological activities of Thai medicinal plants in animal models. BMC Complement. Altern. Med. 2012, 12, 23. [Google Scholar] [CrossRef]
- Habib, S.H.M.; Makpol, S.; Hamid, N.A.A.; Das, S.; Ngah, W.Z.W.; Yusof, Y.A.M. Ginger extract (Zingiber officinale) has anti-cancer and anti-inflammatory effects on ethionine-induced hepatoma rats. Clinics 2008, 63, 807–813. [Google Scholar] [CrossRef]
- Prasad, S.; Tyagi, A.K. Ginger and its constituents: Role in prevention and treatment of gastrointestinal cancer. Gastroenterol. Res. Pract. 2015, 2015, 142979. [Google Scholar] [CrossRef]
- Ko, J.K.; Leung, C.C. Ginger extract and polaprezinc exert gastroprotective actions by anti-oxidant and growth factor modulating effects in rats. J. Gastroenterol. Hepatol. 2010, 25, 1861–1869. [Google Scholar] [CrossRef]
- Manatunga, D.C.; de Silva, R.M.; de Silva, K.M.N.; de Silva, N.; Bhandari, S.; Yap, Y.K.; Costha, N.P. pH responsive controlled release of anti-cancer hydrophobic drugs from sodium alginate and hydroxyapatite bi-coated iron oxide nanoparticles. Eur. J. Pharm. Biopharm. 2017, 117, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Deol, P.K.; Kaur, I.P. Improving the therapeutic efficiency of ginger extract for treatment of colon cancer using a suitably designed multiparticulate system. J. Drug Target. 2013, 21, 855–865. [Google Scholar] [CrossRef] [PubMed]
- Arora, S.; Singh, S.; Piazza, G.A.; Contreras, C.M.; Panyam, J.; Singh, A.P. Honokiol: A novel natural agent for cancer prevention and therapy. Curr. Mol. Med. 2012, 12, 1244–1252. [Google Scholar] [CrossRef] [PubMed]
- Kong, Z.-L.; Tzeng, S.-C.; Liu, Y.-C. Cytotoxic neolignans: An SAR study. Bioorg. Med. Chem. Lett. 2005, 15, 163–166. [Google Scholar] [CrossRef] [PubMed]
- Guillermo-Lagae, R.; Santha, S.; Thomas, M.; Zoelle, E.; Stevens, J.; Kaushik, R.S.; Dwivedi, C. Antineoplastic Effects of Honokiol on Melanoma. Biomed. Res. Int. 2017, 2017, 5496398. [Google Scholar] [CrossRef] [PubMed]
- Mannal, P.W.; Schneider, J.; Tangada, A.; McDonald, D.; McFadden, D.W. Honokiol produces anti-neoplastic effects on melanoma cells in vitro. J. Surg. Oncol. 2011, 104, 260–264. [Google Scholar] [CrossRef]
- Arora, S.; Bhardwaj, A.; Srivastava, S.K.; Singh, S.; McClellan, S.; Wang, B.; Singh, A.P. Honokiol arrests cell cycle, induces apoptosis, and potentiates the cytotoxic effect of gemcitabine in human pancreatic cancer cells. PLoS ONE 2011, 6, e21573. [Google Scholar] [CrossRef]
- Chilampalli, C.; Guillermo, R.; Kaushik, R.S.; Young, A.; Chandrasekher, G.; Fahmy, H.; Dwivedi, C. Honokiol, a chemopreventive agent against skin cancer, induces cell cycle arrest and apoptosis in human epidermoid A431 cells. Exp. Biol. Med. 2011, 236, 1351–1359. [Google Scholar] [CrossRef]
- Leeman-Neill, R.J.; Cai, Q.; Joyce, S.C.; Thomas, S.M.; Bhola, N.E.; Neill, D.B.; Arbiser, J.L.; Grandis, J.R. Honokiol inhibits epidermal growth factor receptor signaling and enhances the antitumor effects of epidermal growth factor receptor inhibitors. Clin. Cancer Res. 2010, 16, 2571–2579. [Google Scholar] [CrossRef]
- Xu, D.; Lu, Q.; Hu, X. Down-regulation of P-glycoprotein expression in MDR breast cancer cell MCF-7/ADR by honokiol. Cancer Lett. 2006, 243, 274–280. [Google Scholar] [CrossRef]
- Wang, X.; Duan, X.; Yang, G.; Zhang, X.; Deng, L.; Zheng, H.; Deng, C.; Wen, J.; Wang, N.; Peng, C. Honokiol crosses BBB and BCSFB, and inhibits brain tumor growth in rat 9L intracerebral gliosarcoma model and human U251 xenograft glioma model. PLoS ONE 2011, 6, e18490. [Google Scholar] [CrossRef]
- Woodbury, A.; Yu, S.P.; Wei, L.; García, P. Neuro-modulating effects of honokiol: A review. Front. Neurol. 2013, 4, 130. [Google Scholar] [CrossRef]
- Godugu, C.; Doddapaneni, R.; Singh, M. Honokiol nanomicellar formulation produced increased oral bioavailability and anticancer effects in triple negative breast cancer (TNBC). Colloids Surf. B Biointerfaces 2017, 153, 208–219. [Google Scholar] [CrossRef]
- Frydoonfar, H.R.; McGrath, D.R.; Spigelman, A.D. The variable effect on proliferation of a colon cancer cell line by the citrus fruit flavonoid Naringenin. Color. Dis. 2003, 5, 149–152. [Google Scholar] [CrossRef]
- Manthey, J.A.; Guthrie, N. Antiproliferative activities of citrus flavonoids against six human cancer cell lines. J. Agric. Food Chem. 2002, 50, 5837–5843. [Google Scholar] [CrossRef]
- Harmon, A.W.; Patel, Y.M. Naringenin Inhibits Glucose Uptake in MCF-7 Breast Cancer Cells: A Mechanism for Impaired Cellular Proliferation. Breast Cancer Res. Treat. 2004, 85, 103–110. [Google Scholar] [CrossRef]
- Harmon, A.W.; Patel, Y.M. Naringenin inhibits phosphoinositide 3-kinase activity and glucose uptake in 3T3-L1 adipocytes. Biochem. Biophys. Res. Commun. 2003, 305, 229–234. [Google Scholar] [CrossRef]
- Galluzzo, P.; Ascenzi, P.; Bulzomi, P.; Marino, M. The Nutritional Flavanone Naringenin Triggers Antiestrogenic Effects by Regulating Estrogen Receptor α-Palmitoylation. Endocrinology 2008, 149, 2567–2575. [Google Scholar] [CrossRef]
- Zhang, F.Y.; Du, G.J.; Zhang, L.; Zhang, C.L.; Lu, W.L.; Liang, W. Naringenin Enhances the Anti-Tumor Effect of Doxorubicin Through Selectively Inhibiting the Activity of Multidrug Resistance-Associated Proteins but not P-glycoprotein. Pharm. Res. 2009, 26, 914–925. [Google Scholar] [CrossRef]
- Krishnakumar, N.; Sulfikkarali, N.; RajendraPrasad, N.; Karthikeyan, S. Enhanced anticancer activity of naringenin-loaded nanoparticles in human cervical (HeLa) cancer cells. Biomed. Prev. Nutr. 2011, 1, 223–231. [Google Scholar] [CrossRef]
- Narumi, K.; Sonoda, J.I.; Shiotani, K.; Shigeru, M.; Shibata, M.; Kawachi, A.; Tomishige, E.; Sato, K.; Motoya, T. Simultaneous detection of green tea catechins and gallic acid in human serum after ingestion of green tea tablets using ion-pair high-performance liquid chromatography with electrochemical detection. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2014, 945–946, 147–153. [Google Scholar] [CrossRef]
- Song, Y.; Sun, H.; Zhang, A.; Yan, G.; Han, Y.; Wang, X. Plant-derived natural products as leads to anti-cancer drugs. J. Med. Plant Herb. Ther. Res. 2014, 2, 6–15. [Google Scholar]
- Hosseinimehr, S.J.; Rostamnejad, M.; Ghaffari-rad, V. Epicatechin enhances anti-proliferative effect of bleomycin in ovarian cancer cell. Res. Mol. Med. 2013, 1, 24–27. [Google Scholar] [CrossRef]
- Hagen, R.M.; Chedea, V.S.; Mintoff, C.P.; Bowler, E.; Morse, H.R.; Ladomery, M.R. Epigallocatechin-3-gallate promotes apoptosis and expression of the caspase 9a splice variant in PC3 prostate cancer cells. Int. J. Oncol. 2013, 43, 194–200. [Google Scholar] [CrossRef]
- Okabe, S.; Ochiai, Y.; Aida, M.; Park, K.; Kim, S.J.; Nomura, T.; Suganuma, M.F.H. Mechanistic aspects of green tea as a cancer preventive: Effect of components on human stomach cancer cell lines. Jpn. J. Cancer Res. 1999, 90, 733–739. [Google Scholar] [CrossRef]
- Cao, J.; Han, J.; Xiao, H.; Qiao, J.; Han, M. Effect of tea polyphenol compounds on anticancer drugs in terms of anti-tumor activity, toxicology, and pharmacokinetics. Nutrients 2016, 8, 762. [Google Scholar] [CrossRef]
- Rao, S.D.; Pagidas, K. Epigallocatechin-3-gallate, a Natural Polyphenol, Inhibits Cell Proliferation and Induces Apoptosis in Human Ovarian Cancer Cells. Anticancer Res. 2010, 30, 2519–2523. [Google Scholar]
- Wang, C.; Luo, H.; Zhu, L.; Yang, F.; Chu, Z.; Tian, H.; Feng, M.; Zhao, Y.; Shang, P. Microgravity inhibition of lipopolysaccharide-induced tumor necrosis factor-α expression in macrophage cells. Inflamm. Res. 2014, 63, 91–98. [Google Scholar] [CrossRef]
- Shankar, S.; Ganapathy, S.; Hingorani, S.R.; Srivastava, R.K. EGCG inhibits growth, invasion, angiogenesis and metastasis of pancreatic cancer. Front. Biosci. 2008, 13, 440–452. [Google Scholar] [CrossRef]
- Sugihara, N.; Kuroda, N.; Watanabe, F.; Choshi, T.; Kamishikiryo, J.; Seo, M. Effects of Catechins and Their Related Compounds on Cellular Accumulation and Efflux Transport of Mitoxantrone in Caco-2 Cell Monolayers. J. Food Sci. 2017, 82, 1224–1230. [Google Scholar] [CrossRef]
- Przystupski, D.; Michel, O.; Rossowska, J.; Kwiatkowski, S.; Saczko, J.; Kulbacka, J. The modulatory effect of green tea catechin on drug resistance in human ovarian cancer cells. Med. Chem. Res. 2019, 28, 657–667. [Google Scholar] [CrossRef]
- Wang, W.; Ke, S.; Chen, G.; Gao, Q.; Wu, S.; Wang, S.; Zhou, J.; Yang, X.; Lu, Y.; Ma, D. Effect of lung resistance-related protein on the resistance to cisplatin in human ovarian cancer cell lines. Oncol. Rep. 2004, 12, 1365–1370. [Google Scholar] [CrossRef]
- Kartalou, M.; Essigmann, J.M. Mechanisms of resistance to cisplatin. Mutat. Res. Fundam. Mol. Mech. Mutagen. 2001, 478, 23–43. [Google Scholar] [CrossRef]
- Wang, X.; Jiang, P.; Wang, P.; Yang, C.S.; Wang, X. EGCG Enhances Cisplatin Sensitivity by Regulating Expression of the Copper and Cisplatin Influx Transporter CTR1 in Ovary Cancer. PLoS ONE 2015, 10, e0125402. [Google Scholar]
- Fang, M.Z.; Wang, Y.; Ai, N.; Hou, Z.; Sun, Y.; Lu, H.; Welsh, W.; Yang, C.S. Tea polyphenol (-)-epigallocatechin-3-gallate inhibits DNA methyltransferase and reactivates methylation-silenced genes in cancer cell lines. Cancer Res. 2003, 63, 7563–7570. [Google Scholar]
- Pandey, M.; Shukla, S.; Gupta, S. Promoter demethylation and chromatin remodeling by green tea polyphenols leads to re-expression of GSTP1 in human prostate cancer cells. Int. J. Cancer 2010, 126, 2520–2533. [Google Scholar] [CrossRef]
- Thakur, V.S.; Gupta, K.; Gupta, S. Green tea polyphenols causes cell cycle arrest and apoptosis in prostate cancer cells by suppressing class I histone deacetylases. Carcinogenesis 2012, 33, 377–384. [Google Scholar] [CrossRef]
- Li, Y.; Yuan, Y.-Y.; Meeran, S.M.; Tollefsbol, T.O. Synergistic epigenetic reactivation of estrogen receptor-alpha (ERalpha) by combined green tea polyphenol and histone deacetylase inhibitor in ERalpha-negative breast cancer cells. Mol. Cancer 2010, 9, 274. [Google Scholar] [CrossRef]
- Nandakumar, V.; Vaid, M.; Katiyar, S.K. (-)-Epigallocatechin-3-gallate reactivates silenced tumor suppressor genes, Cip1/p21 and p16INK4a, by reducing DNA methylation and increasing histones acetylation in human skin cancer cells. Carcinogenesis 2011, 32, 537–544. [Google Scholar] [CrossRef]
- Balasubramanian, S.; Adhikary, G.; Eckert, R.L. The Bmi-1 polycomb protein antagonizes the (-)-epigallocatechin-3-gallate-dependent suppression of skin cancer cell survival. Carcinogenesis 2010, 31, 496–503. [Google Scholar] [CrossRef]
- Choudhury, S.R.; Balasubramanian, S.; Chew, Y.C.; Han, B.; Marquez, V.E.; Eckert, R.L. (-)-Epigallocatechin-3-gallate and DZNep reduce polycomb protein level via a proteasome-dependent mechanism in skin cancer cells. Carcinogenesis 2011, 32, 1525–1532. [Google Scholar] [CrossRef]
- Deb, G.; Thakur, V.S.; Limaye, A.M.; Gupta, S. Epigenetic induction of tissue inhibitor of matrix metalloproteinase-3 by green tea polyphenols in breast cancer cells. Mol. Carcinog. 2015, 54, 485–499. [Google Scholar] [CrossRef]
- Jha, M.; Aggarwal, R.; Jha, A.K.; Shrivastava, A. Natural Compounds: DNA Methyltransferase Inhibitors in Oral Squamous Cell Carcinoma. Appl. Biochem. Biotechnol. 2015, 177, 577–594. [Google Scholar] [CrossRef]
- Khan, M.A.; Hussain, A.; Sundaram, M.K.; Alalami, U.; Gunasekera, D.; Ramesh, L.; Hamza, A.; Quraishi, U. (-)-Epigallocatechin-3-gallate reverses the expression of various tumor-suppressor genes by inhibiting DNA methyltransferases and histone deacetylases in human cervical cancer cells. Oncol. Rep. 2015, 33, 1976–1984. [Google Scholar] [CrossRef]
- Zhou, X.-Q.; Xu, X.-N.; Li, L.; Ma, J.-J.; Zhen, E.-M.; Han, C.-B. Epigallocatechin-3-gallate inhibits the invasion of salivary adenoid cystic carcinoma cells by reversing the hypermethylation status of the RECK gene. Mol. Med. Rep. 2015, 12, 6031–6036. [Google Scholar] [CrossRef]
- Chen, L.-L.; Han, W.-F.; Geng, Y.; Su, J.-S. A genome-wide study of DNA methylation modified by epigallocatechin-3-gallate in the CAL-27 cell line. Mol. Med. Rep. 2015, 12, 5886–5890. [Google Scholar] [CrossRef]
- Li, Q.; Kakizaki, M.; Kuriyama, S.; Sone, T.; Yan, H.; Nakaya, N.; Mastuda-Ohmori, K.; Tsuji, I. Green tea consumption and lung cancer risk: The Ohsaki study. Br. J. Cancer 2008, 99, 1179–1184. [Google Scholar] [CrossRef]
- Kang, H.; Rha, S.Y.; Oh, K.W.; Nam, C.M. Green tea consumption and stomach cancer risk: A meta-analysis. Epidemiol. Health 2010, 32, e2010001. [Google Scholar] [CrossRef]
- Ni, C.-X.; Gong, H.; Liu, Y.; Qi, Y.; Jiang, C.-L.; Zhang, J.-P. Green Tea Consumption and the Risk of Liver Cancer: A Meta-Analysis. Nutr. Cancer 2017, 69, 211–220. [Google Scholar] [CrossRef]
- Yang, G.; Zheng, W.; Xiang, Y.-B.; Gao, J.; Li, H.-L.; Zhang, X.; Gao, Y.-T.; Shu, X.-O. Green tea consumption and colorectal cancer risk: A report from the Shanghai Men’s Health Study. Carcinogenesis 2011, 32, 1684–1688. [Google Scholar] [CrossRef]
- Shrubsole, M.J.; Lu, W.; Chen, Z.; Shu, X.O.; Zheng, Y.; Dai, Q.; Cai, Q.; Gu, K.; Ruan, Z.X.; Gao, Y.-T. Drinking Green Tea Modestly Reduces Breast Cancer Risk. J. Nutr. 2009, 139, 310–316. [Google Scholar] [CrossRef]
- Kurahashi, N.; Sasazuki, S.; Iwasaki, M.; Inoue, M.; Tsugane, S.; JPHC Study Group. Green Tea Consumption and Prostate Cancer Risk in Japanese Men: A Prospective Study. Am. J. Epidemiol. 2007, 167, 71–77. [Google Scholar] [CrossRef]
- Tsao, A.S.; Liu, D.; Martin, J.; Tang, X.M.; Lee, J.J.; El-Naggar, A.K.; Wistuba, I.; Culotta, K.S.; Mao, L.; Gillenwater, A. Phase II Randomized, Placebo-Controlled Trial of Green Tea Extract in Patients with High-Risk Oral Premalignant Lesions. Cancer Prev. Res. 2009, 2, 931–941. [Google Scholar] [CrossRef]
- Hakim, I.A.; Harris, R.B.; Brown, S.; Chow, H.-H.S.; Wiseman, S.; Agarwal, S.; Talbot, W. Effect of increased tea consumption on oxidative DNA damage among smokers: A randomized controlled study. J. Nutr. 2003, 133, 3303S–3309S. [Google Scholar] [CrossRef]
- Luo, H.; Tang, L.; Tang, M.; Billam, M.; Huang, T.; Yu, J.; Wei, Z.; Liang, Y.; Wang, K.; Zhang, Z.-Q. Phase IIa chemoprevention trial of green tea polyphenols in high-risk individuals of liver cancer: Modulation of urinary excretion of green tea polyphenols and 8-hydroxydeoxyguanosine. Carcinogenesis 2006, 27, 262–268. [Google Scholar] [CrossRef]
- Bettuzzi, S.; Brausi, M.; Rizzi, F.; Castagnetti, G.; Peracchia, G.; Corti, A. Chemoprevention of Human Prostate Cancer by Oral Administration of Green Tea Catechins in Volunteers with High-Grade Prostate Intraepithelial Neoplasia: A Preliminary Report from a One-Year Proof-of-Principle Study. Cancer Res. 2006, 66, 1234–1240. [Google Scholar] [CrossRef]
- Sun, Q.; Cai, X.; Li, J.; Zheng, M.; Chen, Z.; Yu, C.-P. Green synthesis of silver nanoparticles using tea leaf extract and evaluation of their stability and antibacterial activity. Colloids Surfaces A Physicochem. Eng. Asp. 2014, 444, 226–231. [Google Scholar] [CrossRef]
- Mukherjee, S.; Ghosh, S.; Das, D.K.; Chakraborty, P.; Choudhury, S.; Gupta, P.; Adhikary, A.; Dey, S.; Chattopadhyay, S. Gold-conjugated green tea nanoparticles for enhanced anti-tumor activities and hepatoprotection—Synthesis, characterization and in vitro evaluation. J. Nutr. Biochem. 2015, 26, 1283–1297. [Google Scholar] [CrossRef]
- Nakhjavani, M.; Nikkhah, V.; Sarafraz, M.M.; Shoja, S.; Sarafraz, M. Green synthesis of silver nanoparticles using green tea leaves: Experimental study on the morphological, rheological and antibacterial behaviour. Heat Mass Transf. 2017, 53, 3201–3209. [Google Scholar] [CrossRef]
- Wang, Y.-J.; Huang, Y.; Anreddy, N.; Zhang, G.-N.; Zhang, Y.-K.; Xie, M.; Lin, D.; Yang, D.-H.; Zhang, M.; Chen, Z.-S. Tea nanoparticle, a safe and biocompatible nanocarrier, greatly potentiates the anticancer activity of doxorubicin. Oncotarget 2016, 7, 5877–5891. [Google Scholar] [CrossRef]
- Michel, O.; Przystupski, D.; Saczko, J.; Szewczyk, A.; Niedzielska, N.; Rossowska, J.; Kulbacka, J. The favourable effect of catechin in electrochemotherapy in human pancreatic cancer cells. Acta Biochim. Pol. 2018, 65, 173–184. [Google Scholar] [CrossRef]
- Hsieh, C.-H.; Lu, C.-H.; Chen, W.-T.; Ma, B.-L.; Chao, C.-Y. Application of non-invasive low strength pulsed electric field to EGCG treatment synergistically enhanced the inhibition effect on PANC-1 cells. PLoS ONE 2017, 12, e0188885. [Google Scholar] [CrossRef]
- Moreira, L.L.; Dias, T.; Dias, L.G.; Rogão, M.; Da Silva, J.P.; Estevinho, L.M. Propolis influence on erythrocyte membrane disorder (hereditary spherocytosis): A first approach. Food Chem. Toxicol. 2011, 49, 520–526. [Google Scholar] [CrossRef][Green Version]
- Rzepecka-Stojko, A.; Kabała-Dzik, A.; Moździerz, A.; Kubina, R.; Wojtyczka, R.; Stojko, R.; Dziedzic, A.; Jastrzębska-Stojko, Ż.; Jurzak, M.; Buszman, E. Caffeic Acid Phenethyl Ester and Ethanol Extract of Propolis Induce the Complementary Cytotoxic Effect on Triple-Negative Breast Cancer Cell Lines. Molecules 2015, 20, 9242–9262. [Google Scholar] [CrossRef]
- Murtaza, G.; Karim, S.; Akram, M.R.; Khan, S.A.; Azhar, S.; Mumtaz, A.; Bin Asad, M.H.H. Caffeic acid phenethyl ester and therapeutic potentials. Biomed. Res. Int. 2014, 2014, 145342. [Google Scholar] [CrossRef]
- Jo, S.-Y.; Lee, N.; Hong, S.-M.; Jung, H.H.; Chae, S.-W. Caffeic Acid Phenethyl Ester Inhibits Diesel Exhaust Particle—Induced Inflammation of Human Middle Ear Epithelial Cells via NOX4 Inhibition. Ann. Otol. Rhinol. Laryngol. 2013, 122, 595–600. [Google Scholar] [CrossRef]
- da Cunha, F.M.; Duma, D.; Assreuy, J.; Buzzi, F.C.; Niero, R.; Campos, M.M.; Calixto, J.B. Caffeic Acid Derivatives: In Vitro and In Vivo Anti-inflammatory Properties. Free Radic. Res. 2004, 38, 1241–1253. [Google Scholar] [CrossRef]
- Borrelli, F.; Maffia, P.; Pinto, L.; Ianaro, A.; Russo, A.; Capasso, F.; Ialenti, A. Phytochemical compounds involved in the anti-inflammatory effect of propolis extract. Fitoterapia 2002, 73 (Suppl. 1), S53–S63. [Google Scholar] [CrossRef]
- Hishikawa, K.; Nakaki, T.; Fujita, T. Oral Flavonoid Supplementation Attenuates Atherosclerosis Development in Apolipoprotein E-Deficient Mice. Arterioscler. Thromb. Vasc. Biol. 2004, 25, 442–446. [Google Scholar] [CrossRef]
- Park, J.H.; Lee, J.K.; Kim, H.S.; Chung, S.T.; Eom, J.H.; Kim, K.A.; Chung, S.J.; Paik, S.Y.; Oh, H.Y. Immunomodulatory effect of caffeic acid phenethyl ester in Balb/c mice. Int. Immunopharmacol. 2004, 4, 429–436. [Google Scholar] [CrossRef]
- Lee, K.J.; Choi, J.H.; Khanal, T.; Hwang, Y.P.; Chung, Y.C.; Jeong, H.G. Protective effect of caffeic acid phenethyl ester against carbon tetrachloride-induced hepatotoxicity in mice. Toxicology 2008, 248, 18–24. [Google Scholar] [CrossRef]
- Ilhan, A.; Iraz, M.; Gurel, A.; Armutcu, F.; Akyol, O. Caffeic acid phenethyl ester exerts a neuroprotective effect on CNS against pentylenetetrazol-induced seizures in mice. Neurochem. Res. 2004, 29, 2287–2292. [Google Scholar] [CrossRef]
- Orban, Z.; Mitsiades, N.; Burke, T.R.; Tsokos, M.; Chrousos, G.P. Caffeic acid phenethyl ester induces leukocyte apoptosis, modulates nuclear factor-kappa B and suppresses acute inflammation. Neuroimmunomodulation 2000, 7, 99–105. [Google Scholar] [CrossRef]
- Chinery, R.; Beauchamp, R.D.; Shyr, Y.; Kirkland, S.C.; Coffey, R.J.; Morrow, J.D.; Mestre, J.R.; Grunberger, D.; Sacks, P.G.; Tanabe, T. Antioxidants reduce cyclooxygenase-2 expression, prostaglandin production, and proliferation in colorectal cancer cells. Cancer Res. 1998, 58, 2323–2327. [Google Scholar]
- Lee, Y.-J.; Kuo, H.-C.; Chu, C.-Y.; Wang, C.-J.; Lin, W.-C.; Tseng, T.-H. Involvement of tumor suppressor protein p53 and p38 MAPK in caffeic acid phenethyl ester-induced apoptosis of C6 glioma cells. Biochem. Pharmacol. 2003, 66, 2281–2289. [Google Scholar] [CrossRef]
- Kuo, H.-C.; Kuo, W.-H.; Lee, Y.-J.; Lin, W.-L.; Chou, F.-P.; Tseng, T.-H. Inhibitory effect of caffeic acid phenethyl ester on the growth of C6 glioma cells in vitro and in vivo. Cancer Lett. 2006, 234, 199–208. [Google Scholar] [CrossRef]
- Sawicka, D.; Car, H.; Borawska, M.H.; Nikliński, J. The anticancer activity of propolis. Folia Histochem. Cytobiol. 2012, 500004, 25–37. [Google Scholar] [CrossRef]
- McEleny, K.; Coffey, R.; Morrissey, C.; Fitzpatrick, J.M.; Watson, R.W.G. Caffeic acid phenethyl ester-induced PC-3 cell apoptosis is caspase-dependent and mediated through the loss of inhibitors of apoptosis proteins. BJU Int. 2004, 94, 402–406. [Google Scholar] [CrossRef]
- Ozturk, G.; Ginis, Z.; Akyol, S.; Erden, G.; Gurel, A.; Akyol, O. The anticancer mechanism of caffeic acid phenethyl ester (CAPE): Review of melanomas, lung and prostate cancers. Eur. Rev. Med. Pharmacol. Sci. 2012, 16, 2064–2068. [Google Scholar]
- Kudugunti, S.K.; Thorsheim, H.; Yousef, M.S.; Guan, L.; Moridani, M.Y. The metabolic bioactivation of caffeic acid phenethyl ester (CAPE) mediated by tyrosinase selectively inhibits glutathione S-transferase. Chem. Biol. Interact. 2011, 192, 243–256. [Google Scholar] [CrossRef]
- Hwang, H.J.; Park, H.J.; Chung, H.-J.; Min, H.-Y.; Park, E.-J.; Hong, J.-Y.; Lee, S.K. Inhibitory effects of caffeic acid phenethyl ester on cancer cell metastasis mediated by the down-regulation of matrix metalloproteinase expression in human HT1080 fibrosarcoma cells. J. Nutr. Biochem. 2006, 17, 356–362. [Google Scholar] [CrossRef]
- Lee, W.J.; Zhu, B.T. Inhibition of DNA methylation by caffeic acid and chlorogenic acid, two common catechol-containing coffee polyphenols. Carcinogenesis 2006, 27, 269–277. [Google Scholar] [CrossRef]
- Omene, C.; Kalac, M.; Wu, J.; Marchi, E.; Frenkel, K.; O’Connor, O.A. Propolis and its active component, Caffeic acid phenethyl ester (CAPE), modulate breast cancer therapeutic targets via an epigenetically mediated mechanism of action. J. Cancer Sci. Ther. 2013, 5, 334–342. [Google Scholar]
- Pellerito, C.; Morana, O.; Ferrante, F.; Calvaruso, G.; Notaro, A.; Sabella, S.; Fiore, T. Synthesis, chemical characterization, computational studies and biological activity of new DNA methyltransferases (DNMTs) specific inhibitor. Epigenetic regulation as a new and potential approach to cancer therapy. J. Inorg. Biochem. 2015, 150, 18–27. [Google Scholar] [CrossRef]
- Eanes, L.; Patel, Y.M. Inhibition of the MAPK pathway alone is insufficient to account for all of the cytotoxic effects of naringenin in MCF-7 breast cancer cells. Biochim. Open 2016, 3, 64–71. [Google Scholar] [CrossRef]
- Frezza, M.; Garay, J.; Chen, D.; Cui, C.; Turos, E.; Dou, Q.P. Induction of tumor cell apoptosis by a novel class of N-thiolated beta-lactam antibiotics with structural modifications at N1 and C3 of the lactam ring. Int. J. Mol. Med. 2008, 21, 689–695. [Google Scholar]
- Kubina, R.; Kabała-Dzik, A.; Dziedzic, A.; Bielec, B.; Wojtyczka, R.D.; Bułdak, R.J.; Wyszyńska, M.; Stawiarska-Pięta, B.; Szaflarska-Stojko, E. The ethanol extract of polish propolis exhibits anti-proliferative and/or pro-apoptotic effect on HCT 116 colon cancer and Me45 Malignant melanoma cells in vitro conditions. Adv. Clin. Exp. Med. 2015, 24, 203–212. [Google Scholar] [CrossRef]
- Patel, S. Emerging Adjuvant Therapy for Cancer: Propolis and its Constituents. J. Diet. Suppl. 2016, 13, 245–268. [Google Scholar] [CrossRef]
- Turan, I.; Demir, S.; Misir, S.; Kilinc, K.; Mentese, A.; Aliyazicioglu, Y.; Deger, O. Cytotoxic effect of Turkish propolis on liver, colon, breast, cervix and prostate cancer cell lines. Trop. J. Pharm. Res. 2015, 14, 777–782. [Google Scholar] [CrossRef]
- Khan, T.; Gurav, P. PhytoNanotechnology: Enhancing delivery of plant based anti-cancer drugs. Front. Pharmacol. 2018, 8, 1002. [Google Scholar] [CrossRef]
- Bhavana, V.; Sudharshan, S.J.S.; Madhu, D. Natural Anticancer Compounds and Their Derivatives in Clinical Trials; Springer: Singapore, 2018; ISBN 9789811082160. [Google Scholar]
- Nguyen, M.M.; Ahmann, F.R.; Nagle, R.B.; Hsu, C.-H.; Tangrea, J.A.; Parnes, H.L.; Sokoloff, M.H.; Gretzer, M.B.; Chow, H.-H.S. Randomized, double-blind, placebo-controlled trial of polyphenon E in prostate cancer patients before prostatectomy: Evaluation of potential chemopreventive activities. Cancer Prev. Res. 2012, 5, 290–298. [Google Scholar] [CrossRef]
- Reed, G.A.; Peterson, K.S.; Smith, H.J.; Gray, J.C.; Sullivan, D.K.; Mayo, M.S.; Crowell, J.A.; Hurwitz, A. A Phase I Study of Indole-3-Carbinol in Women: Tolerability and Effects. Cancer Epidemiol. Biomark. Prev. 2005, 14, 1953–1960. [Google Scholar] [CrossRef]
- Lozanovski, V.J.; Houben, P.; Hinz, U.; Hackert, T.; Herr, I.; Schemmer, P. Pilot study evaluating broccoli sprouts in advanced pancreatic cancer (POUDER trial) - study protocol for a randomized controlled trial. Trials 2014, 15, 204. [Google Scholar] [CrossRef]
- Gezici, S.; Şekeroğlu, N. Current Perspectives in the Application of Medicinal Plants Against Cancer: Novel Therapeutic Agents. Anticancer. Agents Med. Chem. 2019, 19, 101–111. [Google Scholar] [CrossRef]
- Zhang, Y.; Xu, Q.; Lu, J.; Wang, P.; Zhang, H. Tea consumption and the incidence of cancer: A systematic review and meta-analysis of prospective observational studies. Eur. J. Cancer Prev. 2015, 24, 353–362. [Google Scholar] [CrossRef]
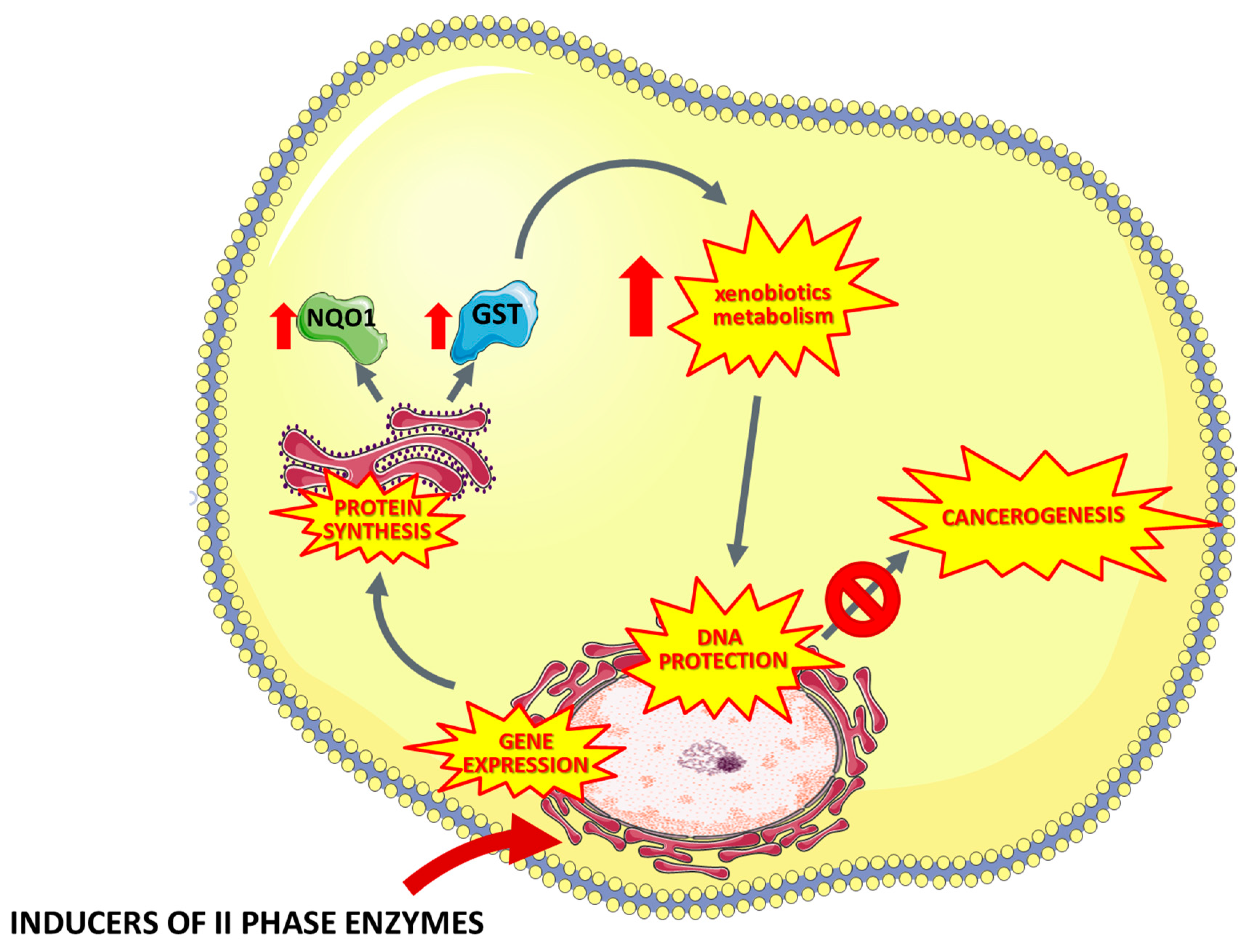
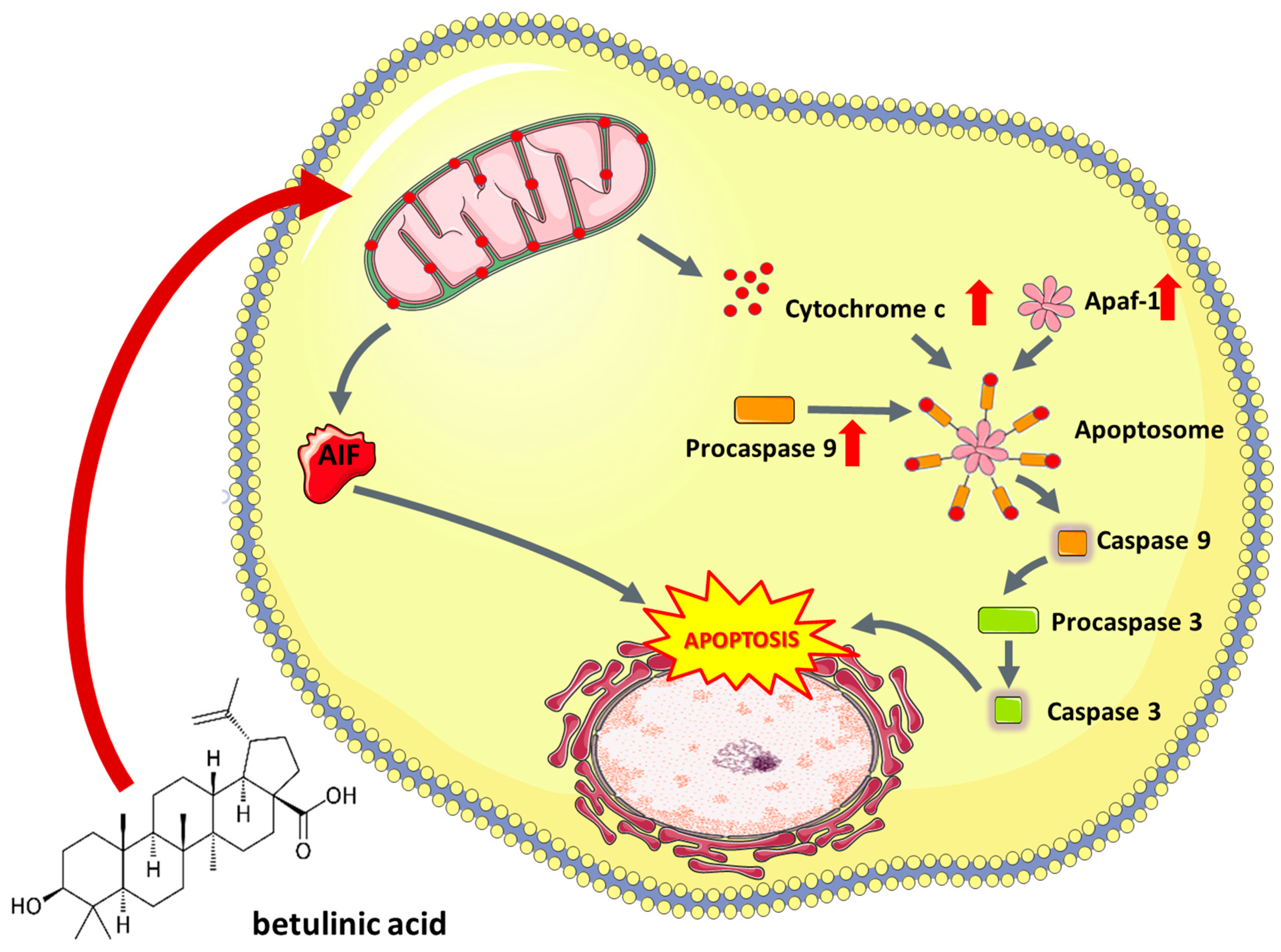
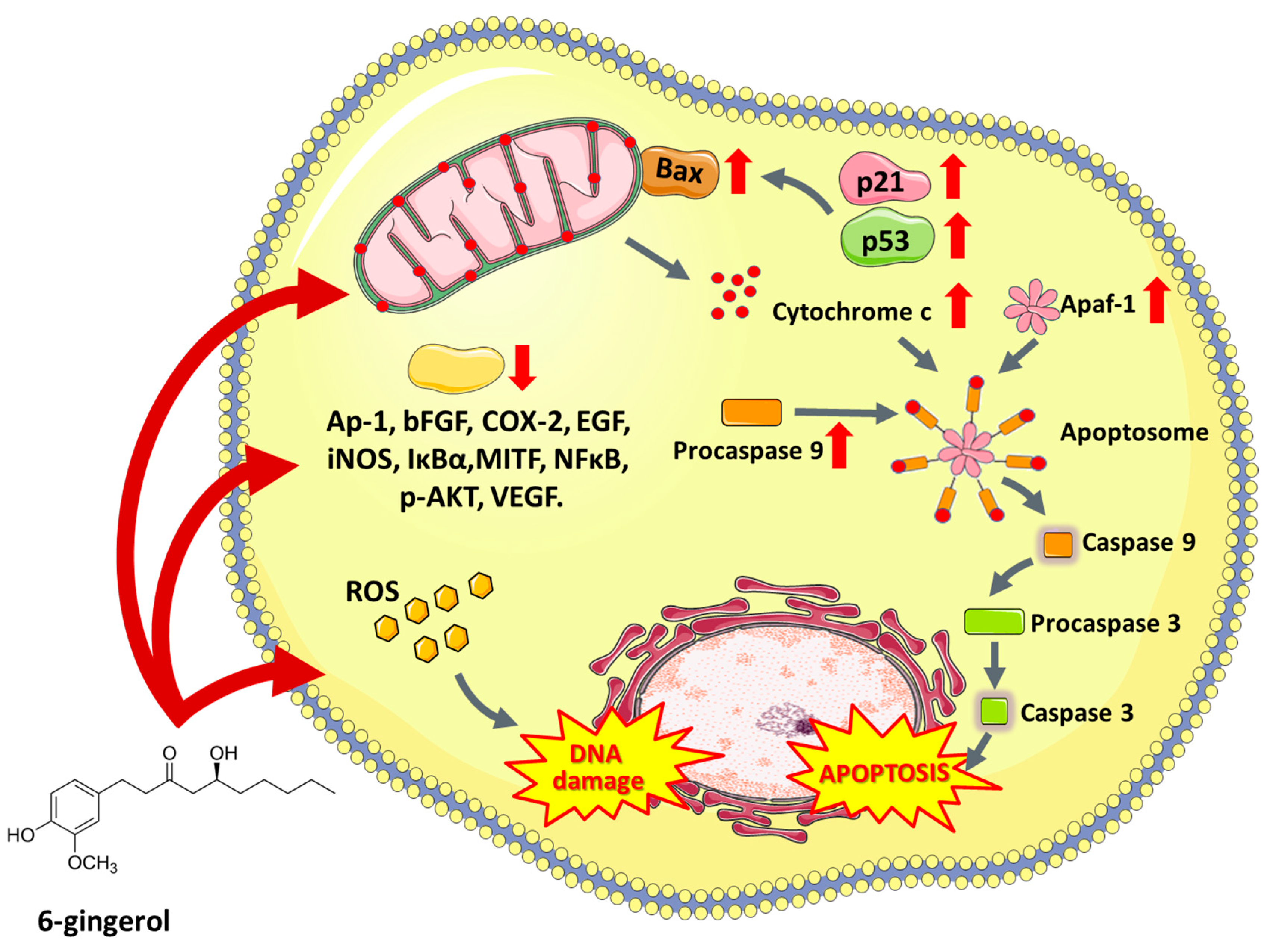
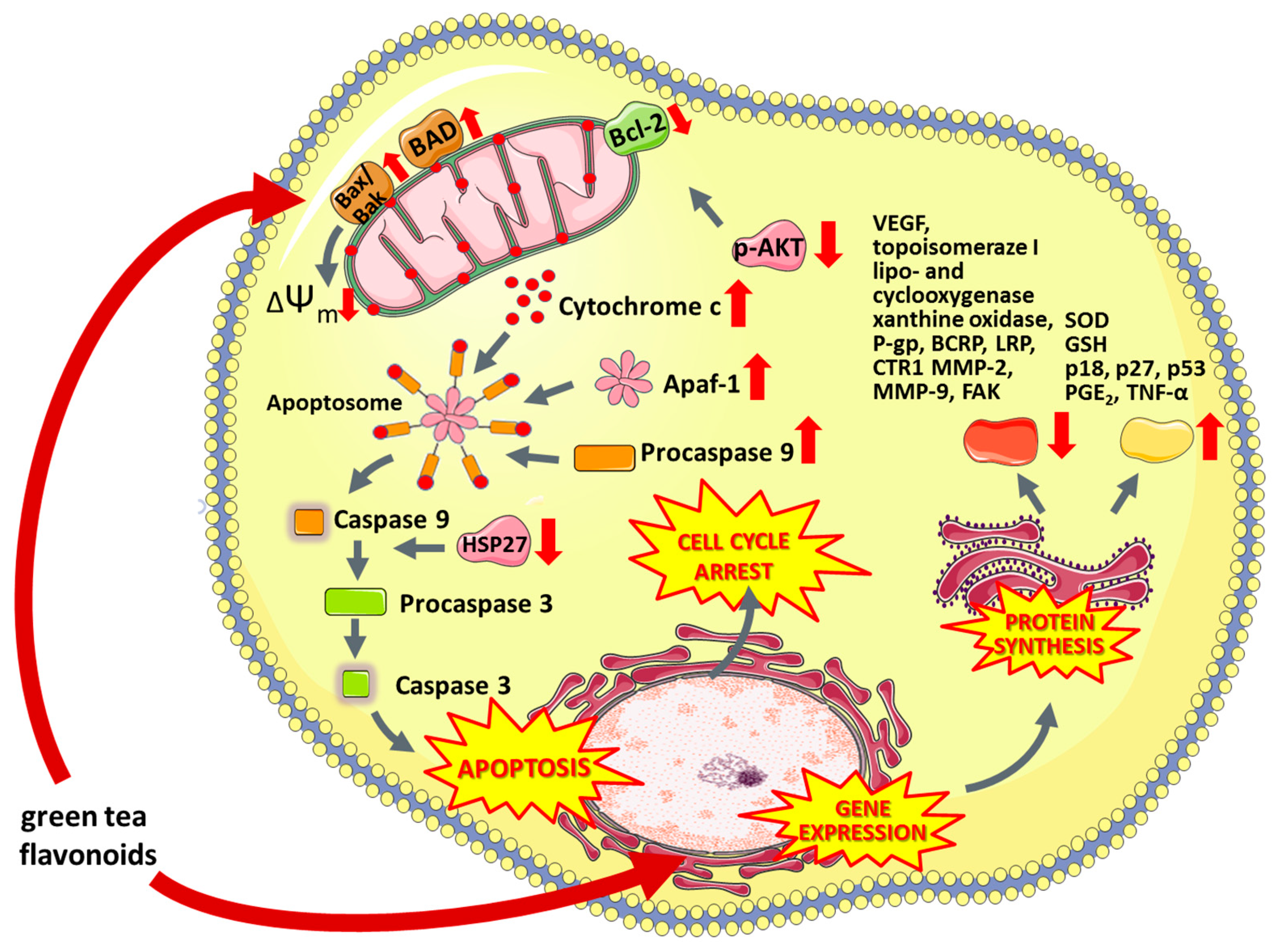
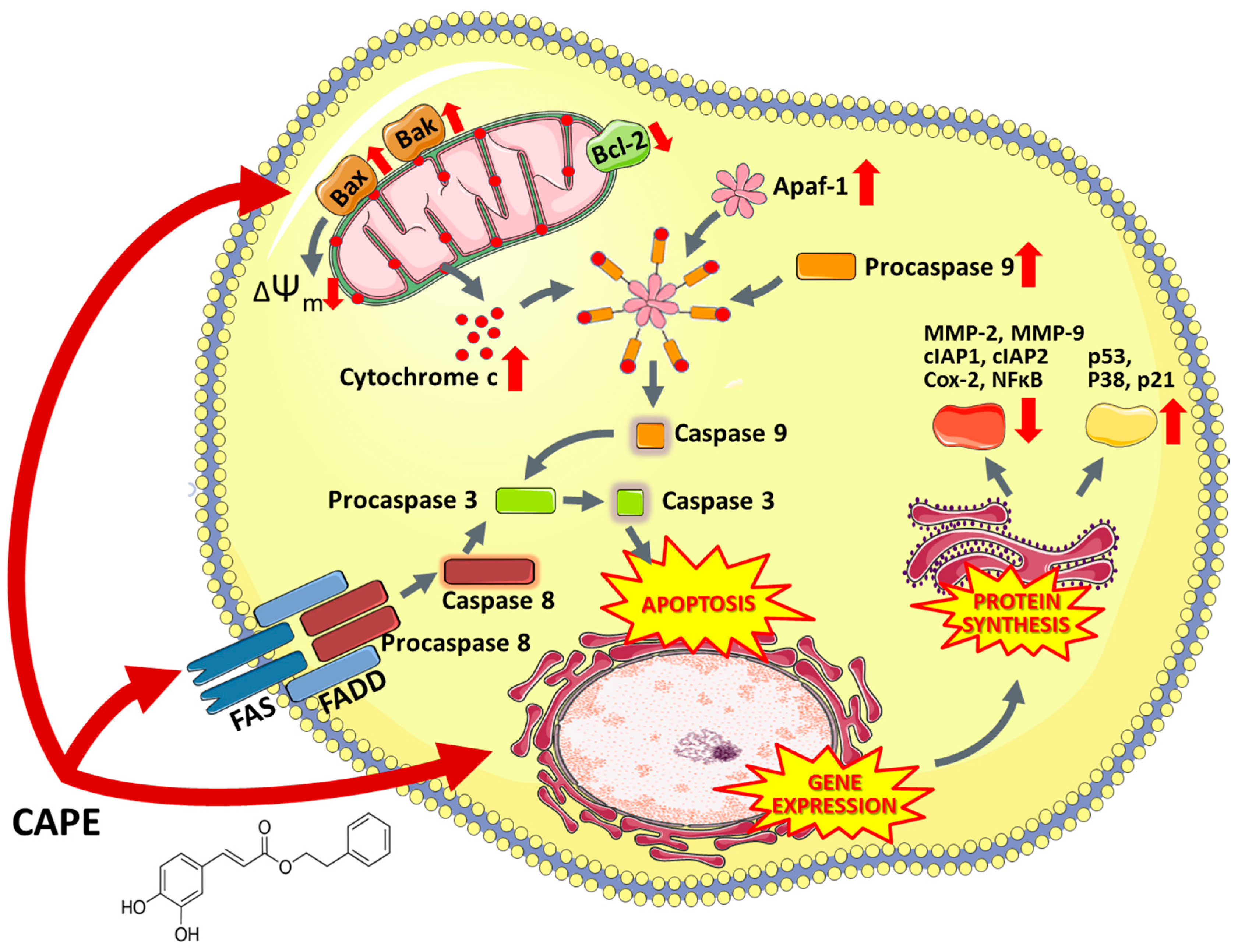
| Structure/Name | Mechanism(s) | Experimental Model | Compound Source | Ref. |
|---|---|---|---|---|
 Vinblastine (above), vincristine, vindesine, vinorelbine | ↓ of microtubule polymerization, ↑ apoptosis, ↑ microtubule depolymerization | HeLa cells, breast and lung cancer, Hodgkin’s disease, lymphosarcoma, chronic lymphocytic leukemia, acute lymphoblastic leukemia, Wilms’ tumor, rhabdo-myosarcoma, chorio-carcinoma, neuroblastoma | pink periwinkle plant Catharanthus roseus | [29,30,33,34,35,53,54,55] |
 Colchicine | ↓ mitosis, ↑ microtubules depolymerization | Due to the low therapeutic index, currently colchicine is not used as an anticancer agent | Meadow saffron Colchicum autumnale L. | [39,40,41,42,45] |
 podophyllotoxin (above), etoposide and teniposide (semi-synthetic derivatives of podophyllotoxin) | ↓ microtubule polymerization; ↓ topoisomerase II (etoposide and teniposide) | Etoposide and teniposide: germ-cell malignancies, lung cancer, Kaposi’s sarcoma, soft tissue sarcomas, leukemias, non-Hodgkin’s lymphoma, neuroblastoma | Podophyllum peltatum L., Podophyllum emodi | [50,56,57,58,59,60,61] |
 Paclitaxel (above), docetaxel | ↑ microtubule polymerization | Kaposi’s sarcoma, breast, head, neck, lung, ovarian esophageal, prostate and bladder cancer | Pacific yew trees (Taxus brevifolia) | [62,63,64,65,66,67,68,69,70,71,72,73,74,75] |
| Structure/Name | Mechanism(s) | Experimental Model | Compound Source | Ref. |
|---|---|---|---|---|
 Camptothecin (above), topotecan and irinotecan (semisynthetic derivatives) | ↓ topoisomerase I | Colorectal cancer, small cell lung cancer, leukemia | Tibetan tree Camptotheca acuminate | [79,80,81,83,84] |
 Etoposide | ↓Topoisomerase II | Leukemia, small cell lung cancer, testicular tumors, Hodgkin’s lymphoma | Podophyllum peltatum | [85,86,87,88] |
| Structure/Name | Mechanism(s) | Experimental Model | Compound Source | Ref. |
|---|---|---|---|---|
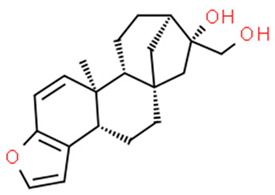 Kahweol (above) and other terpenes (diterpenes, kafestol) | ↑ II phase enzymes (via NRF2 transcription factor) | Mice and rat liver and kidney cells | Green coffee seeds | [100,101,102,103,104] |
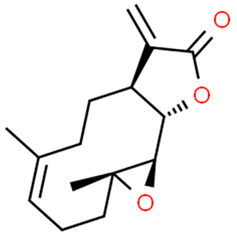 Parthenolide | ↑ cell cycle arrest, cell differentiation, apoptosis, epigenetics: ↓ HDAC1 and DNMT1 | Leukemia, cervical cancer, breast cancer | Tanacetum parthenium feverfew | [105,106,107,108,109,110,111] |
 Betulinic acid | ↑ apoptosis, mitochondrial membrane permeabilization, cell cycle arrest | Lung, cervical and ovarian cancer, melanoma, rhabdomyo-sarcoma, neuroblastoma, leukemia, epidermoid carcinoma | bark of Betula pubescens | [112,113,114,115,116,117] |
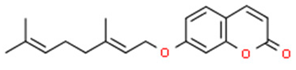 Auraptene (above) and other coumarins (furanocoumarin, pyranocoumarin) | ↑ of II phase enzymes DHC: ↓HDAC (↓Rad52) ↓SIRT1 deacetylases | In vivo and in vitro animal model, yeast, human lymphoblastoid, colon, prostate ovarian, breast, non-small cell lung cancer, leukemia cell line | Citrus fruits and vegetables such as parsley, celery, parsnip | [118,119,120] |
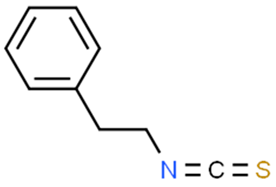 Phenethyl isothiocyanate (PEITC) | ↑ of II phase enzymes, ↑DNMT1, ↑GSTP1; ↓HDAC, miRNAs modulation (cell proliferation, apoptosis and angiogenesis, cell invasiveness; ↓MMPs) | Animal and human cancer cells in vitro: breast, cervical, non-small cell lung. Prostate cancer, osteogenic sarcoma, myeloma cell lines | Spices, such as mustard, wasabi, and horseradish | [121,122,123,124,125,126,127,128,129,130,131,132,133,134,135,136] |
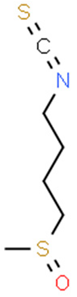 Sulforaphane | ↑ II phase enzymes; ↑ apoptosis; ↓DNMT1s, ↓HDAC | Human cancer cells in vitro: breast, colon cancer, glioblastoma | Broccoli | [137,138,139,140,141] |
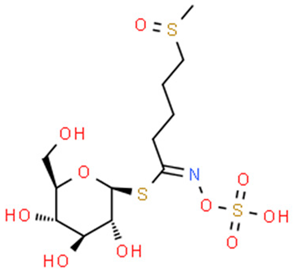 Glucoraphanin | ↑ II phase enzymes | Weaker chemo-preventive properties than sulforaphane | Plant precursor of sulforaphane | [142,143,144] |
 Iberin | ↑ II phase enzymes; ↑apoptosis | Human cancer cells in vitro: breast, colon cancer, glioblastoma | Broccoli | [138,139,140,141] |
 Allyl sulfide (above), diallyl sulfide, | ↑ phase II enzymes DADS: ↓HDAC (↓ cell proliferation, ↑cell cycle arrest) | Rat intestine and liver cells, erythroleukemia, colon cancer cells | Genus Allium: garlic and onions etc. | [145,146,147,148,149,150,151,152,153] |
 Indole-3-carbinol | ↑ phase II enzymes ↓HDAC ↑ cell cycle arrest, apoptosis miRNAs modulation (↓cell invasiveness) | Rat liver cells, human cancer cells in vitro: breast, prostate, ovarian cancer | Vegetables of Cruciferae family, such as: cabbage, kale, brussels sprouts, broccoli | [154,155,156,157,158,159,160,161,162,163,164,165,166,167,168] |
| Structure/Name | Mechanism(s) | Experimental Model | Compound Source | Ref. |
|---|---|---|---|---|
 Carnosol | ↓ CDKs, JAK2-STAT3, MAPK, Akt pathways; ↑ cycle arrest, p53 pathway | (GBM), breast, colon, skin, ovarian, prostate cancer, leukemia macrophage cell lines, | (Rosmarinus officinalis), oregano (Origanum vulgare), sage (Salvia carnosa | [201,202,203,204,205,206,207,208,210] |
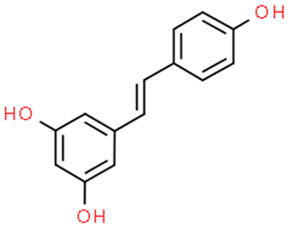 Resveratrol | Targeting COX (↓ tumor proliferation) ↑ cell cycle arrest ↓ NF-κB AP-1, HIF-1α, MMPs ↑ BRCA-1, NRF2, RASSF-1α; miRNAs modulation; ↓HDACs, DNMTs | many cancers in vitro and in vivo (e.g., prostate, breast, colorectal cancer) Clinical trials | Polygonum cuspidatum root, Vitis vinifera (common grape) | [22,216,217,220] |
 6-gingerol | ↓angiogenesis, metastasis; ↑ apoptosis antitumor, antioxidant activity miRNAs modulation | Leukemia, breast, prostate, liver, colon, glioblastoma and pancreatic cancer | Rhizome of ginger (Zingiber officinale Roscoe) | [234,235,236,237,238,239,240] |
 Honokiol | ↑ apoptosis; ↓ EGFR signaling, P-glycoprotein | Hepatocellular carcinoma, melanoma, pancreas, epidermoid squamous skin cancer, glioma, head and neck squamous cancer | Cortex, cones and leaves of Magnolia officinalis | [254,255,256,257,258,261] |
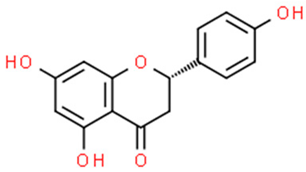 Naringenin | ↓ proliferation, kinase and glucose uptake; ↑apoptosis; antioxidant activities; MDR modulation | Breast and colon cancer | Grapefruit, orange etc., | [264,265,266,267,268,269,335,336] |
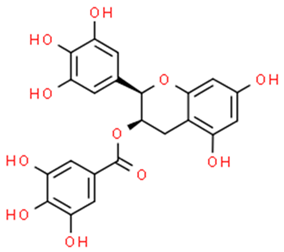 Green tea flavonoids epigallocatechin gallate (above), epicatechin gallate, epigallocatechin, epicatechin and catechin | ↓ proliferation, angiogenesis, DNA synthesis, ↑apoptosis, cell cycle arrest; ↓HDACs, (↓cell invasiveness) DNMTs antioxidant activities; MDR modulation, PcGs modulation. | Many cancers in vitro and in vivo (e.g., lung ovarian, breast, prostate); Clinical trials | Green tea | [272,273,274,275,276,277,278,279,280,282,283,284,285,286,287,288,289,290,291,292,293,294,295,296,297,298,299,300,301,302,305,306] |
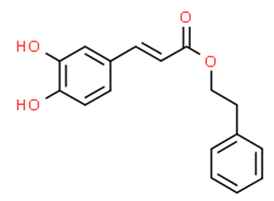 Caffeic acid phenethyl ester, caffeic acid | Cytostatic properties; ↑apoptosis; MDR modulation, ↓ MMPs; ↓HDACs, DNMTs | Laryngeal, pancreatic, brain, kidney, breast, lung, bladder, colorectal, prostate, head and neck cancer, melanoma | Propolis | [314,322,325,326,337,338,339] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Przystupski, D.; Niemczura, M.J.; Górska, A.; Supplitt, S.; Kotowski, K.; Wawryka, P.; Rozborska, P.; Woźniak, K.; Michel, O.; Kiełbik, A.; et al. In Search of Panacea—Review of Recent Studies Concerning Nature-Derived Anticancer Agents. Nutrients 2019, 11, 1426. https://doi.org/10.3390/nu11061426
Przystupski D, Niemczura MJ, Górska A, Supplitt S, Kotowski K, Wawryka P, Rozborska P, Woźniak K, Michel O, Kiełbik A, et al. In Search of Panacea—Review of Recent Studies Concerning Nature-Derived Anticancer Agents. Nutrients. 2019; 11(6):1426. https://doi.org/10.3390/nu11061426
Chicago/Turabian StylePrzystupski, Dawid, Magdalena J. Niemczura, Agata Górska, Stanisław Supplitt, Krzysztof Kotowski, Piotr Wawryka, Paulina Rozborska, Kinga Woźniak, Olga Michel, Aleksander Kiełbik, and et al. 2019. "In Search of Panacea—Review of Recent Studies Concerning Nature-Derived Anticancer Agents" Nutrients 11, no. 6: 1426. https://doi.org/10.3390/nu11061426
APA StylePrzystupski, D., Niemczura, M. J., Górska, A., Supplitt, S., Kotowski, K., Wawryka, P., Rozborska, P., Woźniak, K., Michel, O., Kiełbik, A., Bartosik, W., Saczko, J., & Kulbacka, J. (2019). In Search of Panacea—Review of Recent Studies Concerning Nature-Derived Anticancer Agents. Nutrients, 11(6), 1426. https://doi.org/10.3390/nu11061426








