Impairment between Oxidant and Antioxidant Systems: Short- and Long-term Implications for Athletes’ Health
Abstract
1. Reactive Oxygen Species: The Patho-physiological Role
1.1. ROS-Mediated Cellular Signaling
1.2. Endogenous and Exogenous Antioxidants
1.3. Oxidative Stress and Correlated Diseases
2. The Physiological and Pathological Role of Oxidative Stress in Physical Exercise
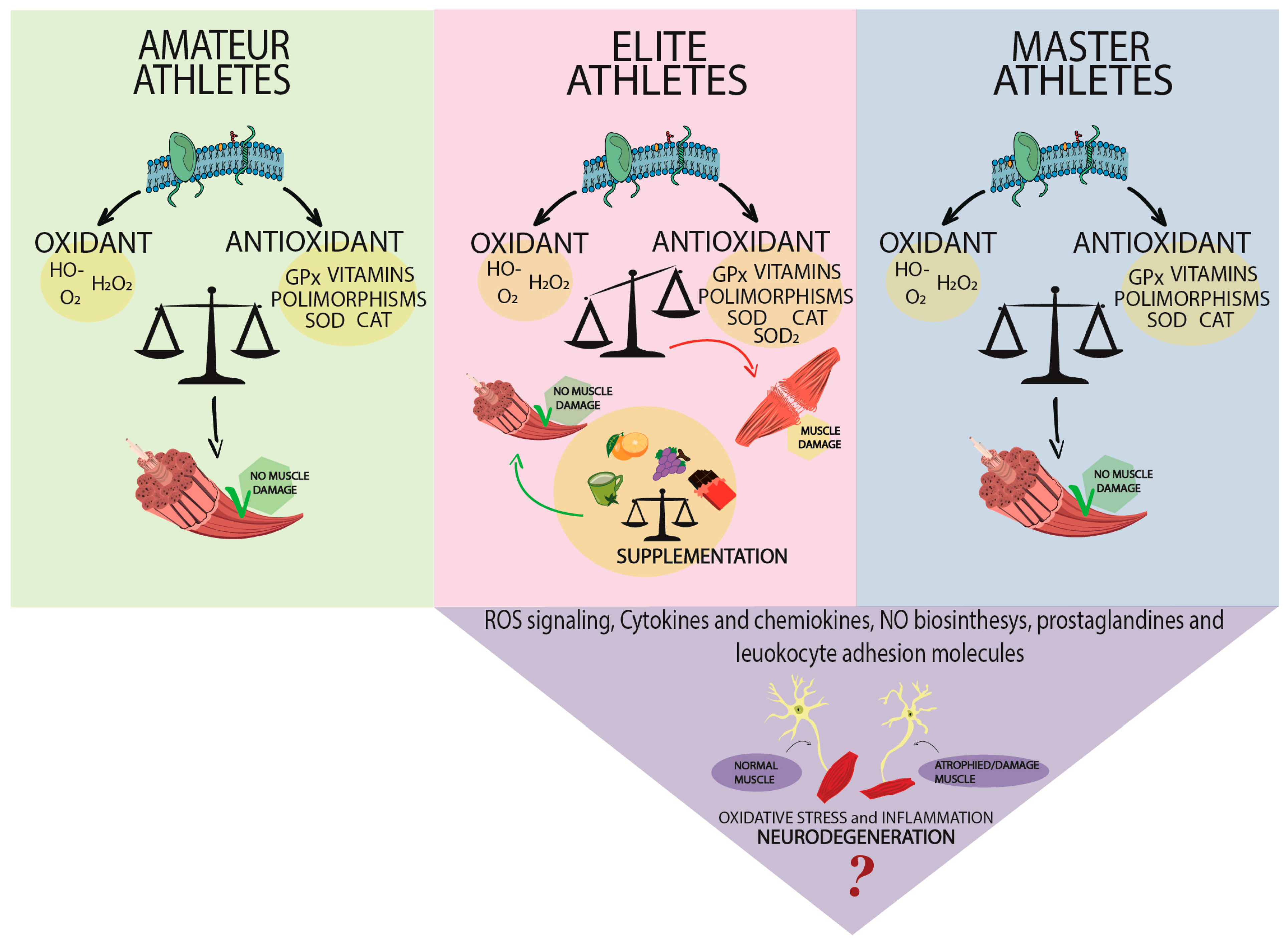
2.1. Oxidative Stress in Amateur Trainers
2.2. Oxidative Stress and Elite Athletes
2.3. Oxidative Stress and Master Athletes
3. Muscle Damage in Athletes Induced by Redox Imbalance during Intensive Exercise
4. Muscle Damage and Neurodegeneration in Athletes
5. Impact of Antioxidant Supplementation in the Athlete Population
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Halliwell, B. Free Radicals and Other Reactive Species in Disease, Encyclopedia of Life Sciences; John Wiley & Sons, Ltd.: Chichester, UK, 2005. [Google Scholar]
- Di Meo, S.; Reed, T.T.; Venditti, P.; Victor, V.M. Role of ROS and RNS Sources in Physiological and Pathological Conditions. Oxidative Med. Cell. Longev. 2016, 2016, 1245049. Available online: http://www.ncbi.nlm.nih.gov/pubmed/27478531 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Thannickal, V.J.; Fanburg, B.L. Reactive oxygen species in cell signaling. Am. J. Physiol. Cell. Mol. Physiol. 2000, 279, L1005–L1028. Available online: http://www.ncbi.nlm.nih.gov/pubmed/11076791 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Zeeshan, H.M.A.; Lee, G.H.; Kim, H.-R.; Chae, H.-J. Endoplasmic Reticulum Stress and Associated ROS. Int. J. Mol. Sci. 2016, 17, 327. Available online: http://www.ncbi.nlm.nih.gov/pubmed/26950115 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Abdal Dayem, A.; Hossain, M.K.; Lee, S.B.; Kim, K.; Saha, S.K.; Yang, G.-M.; Choi, H.Y.; Cho, S.-G. The Role of Reactive Oxygen Species (ROS) in the Biological Activities of Metallic Nanoparticles. Int. J. Mol. Sci. 2017, 18, 120. Available online: http://www.ncbi.nlm.nih.gov/pubmed/28075405 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Violi, F.; Loffredo, L.; Carnevale, R.; Pignatelli, P.; Pastori, D. Atherothrombosis and Oxidative Stress: Mechanisms and Management in Elderly. Antioxid. Redox Signal. 2017, 27, 1083–1124. Available online: http://www.ncbi.nlm.nih.gov/pubmed/28816059 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Brown, D.I.; Griendling, K.K. Nox proteins in signal transduction. Free Radic. Biol. Med. 2009, 47, 1239–1253. Available online: http://www.ncbi.nlm.nih.gov/pubmed/19628035 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Bae, Y.S.; Oh, H.; Rhee, S.G.; Do Yoo, Y. Regulation of reactive oxygen species generation in cell signaling. Mol. Cells 2011, 32, 491–509. Available online: http://www.ncbi.nlm.nih.gov/pubmed/22207195 (accessed on 15 May 2019). [CrossRef]
- Zhang, J.; Wang, X.; Vikash, V.; Ye, Q.; Wu, D.; Liu, Y.; Dong, W. ROS and ROS-Mediated Cellular Signaling. Oxidative Med. Cell. Longev. 2016, 2016, 4350965. Available online: http://www.ncbi.nlm.nih.gov/pubmed/26998193 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Phaniendra, A.; Jestadi, D.B.; Periyasamy, L. Free radicals: Properties, sources, targets, and their implication in various diseases. Indian J. Clin. Biochem. 2015, 30, 11–26. Available online: http://www.ncbi.nlm.nih.gov/pubmed/25646037 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Aguilar, T.A.F.; Navarro, B.C.H.; Pérez, J.A.M. Endogenous Antioxidants: A Review of their Role in Oxidative Stress. In A Master Regulator of Oxidative Stress—The Transcription Factor Nrf2; IntechOpen: London, UK, 2016; Available online: http://www.intechopen.com/books/a-master-regulator-of-oxidative-stress-the-transcription-factor-nrf2/endogenous-antioxidants-a-review-of-their-role-in-oxidative-stress (accessed on 15 May 2019).
- Lobo, V.; Patil, A.; Phatak, A.; Chandra, N. Free radicals, antioxidants and functional foods: Impact on human health. Pharmacogn. Rev. 2010, 4, 118–126. Available online: http://www.ncbi.nlm.nih.gov/pubmed/22228951 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Carlsen, M.H.; Halvorsen, B.L.; Holte, K.; Bøhn, S.K.; Dragland, S.; Sampson, L.; Willey, C.; Senoo, H.; Umezono, Y.; Sanada, C.; et al. The total antioxidant content of more than 3100 foods, beverages, spices, herbs and supplements used worldwide. Nutr. J. 2010, 9, 3. [Google Scholar] [CrossRef] [PubMed]
- Zujko, M.; Witkowska, A.; Waśkiewicz, A.; Sygnowska, E. Estimation of dietary intake and patterns of polyphenol consumption in Polish adult population. Adv. Med. Sci. 2012, 57, 375–384. Available online: https://www.sciencedirect.com/science/article/pii/S1896112614601000 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Wilson, D.; Nash, P.; Buttar, H.; Griffiths, K.; Singh, R.; De Meester, F.; Horiuchi, R.; Takahashi, T. The Role of Food Antioxidants, Benefits of Functional Foods, and Influence of Feeding Habits on the Health of the Older Person: An Overview. Antioxidants 2017, 6, 81. Available online: http://www.ncbi.nlm.nih.gov/pubmed/29143759 (accessed on 15 May 2019).
- Carnevale, R.; Loffredo, L.; Pignatelli, P.; Nocella, C.; Bartimoccia, S.; Di Santo, S.; Martino, F.; Catasca, E.; Perri, L.; Violi, F. Dark chocolate inhibits platelet isoprostanes via NOX2 down-regulation in smokers. J. Thromb. Haemost. 2012, 10, 125–132. Available online: http://www.ncbi.nlm.nih.gov/pubmed/22066819 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Aboul-Enein, H.Y.; Kruk, I.; Kładna, A.; Lichszteld, K.; Michalska, T. Scavenging effects of phenolic compounds on reactive oxygen species. Biopolymers 2007, 86, 222–230. Available online: http://www.ncbi.nlm.nih.gov/pubmed/17373654 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Li, Y.; Cao, Z.; Zhu, H. Upregulation of endogenous antioxidants and phase 2 enzymes by the red wine polyphenol, resveratrol in cultured aortic smooth muscle cells leads to cytoprotection against oxidative and electrophilic stress. Pharmacol. Res. 2006, 53, 6–15. Available online: http://www.ncbi.nlm.nih.gov/pubmed/16169743 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Nocella, C.; Cammisotto, V.; Fianchini, L.; D’Amico, A.; Novo, M.; Castellani, V.; Stefanini, L.; Violi, F.; Carnevale, R. Extra Virgin Olive Oil and Cardiovascular Diseases: Benefits for Human Health. Endocr. Metab. Immune Disord. Drug Targets 2017, 18, 4–13. Available online: http://www.ncbi.nlm.nih.gov/pubmed/29141571 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Khansari, N.; Shakiba, Y.; Mahmoudi, M. Chronic inflammation and oxidative stress as a major cause of age-related diseases and cancer. Recent Pat. Inflamm. Allergy Drug Discov. 2009, 3, 73–80. Available online: http://www.ncbi.nlm.nih.gov/pubmed/19149749 (accessed on 15 May 2019). [CrossRef]
- Gracia, K.C.; Llanas-Cornejo, D.; Husi, H. CVD and Oxidative Stress. J. Clin. Med. 2017, 6, 22. Available online: http://www.ncbi.nlm.nih.gov/pubmed/28230726 (accessed on 15 May 2019).
- Uttara, B.; Singh, A.V.; Zamboni, P.; Mahajan, R.T. Oxidative stress and neurodegenerative diseases: A review of upstream and downstream antioxidant therapeutic options. Curr. Neuropharmacol. 2009, 7, 65–74. Available online: http://www.ncbi.nlm.nih.gov/pubmed/19721819 (accessed on 15 May 2019). [CrossRef]
- Valko, M.; Izakovic, M.; Mazur, M.; Rhodes, C.J.; Telser, J. Role of oxygen radicals in DNA damage and cancer incidence. Mol. Cell. Biochem. 2004, 266, 37–56. Available online: http://www.ncbi.nlm.nih.gov/pubmed/15646026 (accessed on 15 May 2019). [CrossRef]
- Nowak, W.N.; Deng, J.; Ruan, X.Z.; Xu, Q. Reactive Oxygen Species Generation and Atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 2017, 37, e41–e52. [Google Scholar] [CrossRef] [PubMed]
- Pan, X.; Zhu, Y.; Lin, N.; Zhang, J.; Ye, Q.; Huang, H.; Chen, X. Microglial phagocytosis induced by fibrillar β-amyloid is attenuated by oligomeric β-amyloid: Implications for Alzheimer’s disease. Mol. Neurodegener. 2011, 6, 45. Available online: http://www.ncbi.nlm.nih.gov/pubmed/21718498 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Sevcsik, E.; Trexler, A.J.; Dunn, J.M.; Rhoades, E. Allostery in a disordered protein: Oxidative modifications to α-synuclein act distally to regulate membrane binding. J. Am. Chem. Soc. 2011, 133, 7152–7158. Available online: http://www.ncbi.nlm.nih.gov/pubmed/21491910 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Zhao, W.; Varghese, M.; Yemul, S.; Pan, Y.; Cheng, A.; Marano, P.; Hassan, S.; Vempati, P.; Chen, F.; Qian, X.; et al. Peroxisome proliferator activator receptor gamma coactivator-1alpha (PGC-1α) improves motor performance and survival in a mouse model of amyotrophic lateral sclerosis. Mol. Neurodegener. 2011, 6, 51. [Google Scholar] [CrossRef] [PubMed]
- Witherick, J.; Wilkins, A.; Scolding, N.; Kemp, K. Mechanisms of oxidative damage in multiple sclerosis and a cell therapy approach to treatment. Autoimmune Dis. 2010, 2011, 164608. Available online: http://www.ncbi.nlm.nih.gov/pubmed/21197107 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Islam, M.T. Oxidative stress and mitochondrial dysfunction-linked neurodegenerative disorders. Neurol. Res. 2017, 39, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Carnevale, R.; Bartimoccia, S.; Nocella, C.; Di Santo, S.; Loffredo, L.; Illuminati, G.; Lombardi, E.; Boz, V.; Del Ben, M.; De Marco, L.; et al. LDL oxidation by platelets propagates platelet activation via an oxidative stress-mediated mechanism. Atherosclerosis 2014, 237, 108–116. Available online: http://www.ncbi.nlm.nih.gov/pubmed/25238217 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Abruzzo, P.; Esposito, F.; Marchionni, C.; di Tullio, S.; Belia, S.; Fulle, S.; Veicsteinas, A.; Marini, M. Moderate Exercise Training Induces ROS-Related Adaptations to Skeletal Muscles. Int. J. Sports Med. 2013, 34, 676–687. Available online: http://www.ncbi.nlm.nih.gov/pubmed/23325712 (accessed on 15 May 2019). [CrossRef]
- Jornayvaz, F.R.; Shulman, G.I. Regulation of mitochondrial biogenesis. Essays Biochem. 2010, 47, 69–84. Available online: http://www.ncbi.nlm.nih.gov/pubmed/20533901 (accessed on 15 May 2019). [CrossRef]
- Samjoo, I.A.; Safdar, A.; Hamadeh, M.J.; Raha, S.; Tarnopolsky, M.A. The effect of endurance exercise on both skeletal muscle and systemic oxidative stress in previously sedentary obese men. Nutr. Diabetes 2013, 3, e88. Available online: http://www.ncbi.nlm.nih.gov/pubmed/24042701 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Lorenz, D.S.; Reiman, M.P.; Lehecka, B.J.; Naylor, A. What performance characteristics determine elite versus nonelite athletes in the same sport? Sports Health 2013, 5, 542–547. Available online: http://www.ncbi.nlm.nih.gov/pubmed/24427430 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Cavarretta, E.; Peruzzi, M.; Frati, G.; Sciarretta, S. When enough is more than enough: The hidden side of the cardiac effects of intense physical exercise. Int. J. Cardiol. 2018, 258, 224–225. Available online: http://www.ncbi.nlm.nih.gov/pubmed/29544936 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Radak, Z.; Ishihara, K.; Tekus, E.; Varga, C.; Posa, A.; Balogh, L.; Boldogh, I.; Koltai, E. Exercise, oxidants, and antioxidants change the shape of the bell-shaped hormesis curve. Redox Biol. 2017, 12, 285–290. Available online: http://www.ncbi.nlm.nih.gov/pubmed/28285189 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Falone, S.; Mirabilio, A.; Pennelli, A.; Cacchio, M.; Di Baldassarre, A.; Gallina, S.; Passerini, A.; Amicarelli, F. Differential Impact of Acute Bout of Exercise on Redox-and Oxidative Damage-Related Profiles Between Untrained Subjects and Amateur Runners. Physiol. Res. 2010, 59, 953–961. Available online: www.biomed.cas.cz/physiolres (accessed on 15 May 2019). [PubMed]
- Seifi-Skishahr, F.; Damirchi, A.; Farjaminezhad, M.; Babaei, P. Physical Training Status Determines Oxidative Stress and Redox Changes in Response to an Acute Aerobic Exercise. Biochem. Res. Int. 2016, 2016, 3757623. Available online: http://www.hindawi.com/journals/bri/2016/3757623/ (accessed on 15 May 2019). [CrossRef]
- Bloomer, R.J.; Goldfarb, A.H.; Wideman, L.; McKenzie, M.J.; Consitt, L.A. Effects of acute aerobic and anaerobic exercise on blood markers of oxidative stress. J. Strength Cond. Res. 2005, 19, 276–285. Available online: http://www.ncbi.nlm.nih.gov/pubmed/15903362 (accessed on 15 May 2019).
- Arsic, A.; Vucic, V.; Glibetic, M.; Popovic, T.; Debeljak-Martacic, J.; Cubrilo, D.; Ahmetovic, Z.; Peric, D.; Borozan, S.; Djuric, D.; et al. Redox balance in elite female athletes: Differences based on sport types. J. Sports Med. Phys. Fit. 2019, 56, 1–8. Available online: http://www.ncbi.nlm.nih.gov/pubmed/25812706 (accessed on 15 May 2019).
- Bloomer, R.; Davis, P.; Consitt, L.; Wideman, L. Plasma Protein Carbonyl Response to Increasing Exercise Duration in Aerobically Trained Men and Women. Int. J. Sports Med. 2007, 28, 21–25. [Google Scholar] [CrossRef] [PubMed]
- Goto, C.; Nishioka, K.; Umemura, T.; Jitsuiki, D.; Sakagutchi, A.; Kawamura, M.; Chayama, K.; Yoshizumi, M.; Higashi, Y. Acute Moderate-Intensity Exercise Induces Vasodilation Through an Increase in Nitric Oxide Bioavailiability in Humans. Am. J. Hypertens. 2007, 20, 825–830. [Google Scholar] [CrossRef]
- Goto, C.; Higashi, Y.; Kimura, M.; Noma, K.; Hara, K.; Nakagawa, K.; Kawamura, M.; Chayama, K.; Yoshizumi, M.; Nara, I. Effect of different intensities of exercise on endothelium-dependent vasodilation in humans: Role of endothelium-dependent nitric oxide and oxidative stress. Circulation 2003, 108, 530–535. [Google Scholar] [CrossRef] [PubMed]
- Sacheck, J.M.; Cannon, J.G.; Hamada, K.; Vannier, E.; Blumberg, J.B.; Roubenoff, R. Age-related loss of associations between acute exercise-induced IL-6 and oxidative stress. Am. J. Physiol. Endocrinol. Metab. 2006, 291, E340–E349. [Google Scholar] [CrossRef]
- Bloomer, R.J.; Fry, A.C.; Falvo, M.J.; Moore, C.A. Protein carbonyls are acutely elevated following single set anaerobic exercise in resistance trained men. J. Sci. Med. Sport 2007, 10, 411–417. Available online: https://linkinghub.elsevier.com/retrieve/pii/S144024400600185X (accessed on 15 May 2019). [CrossRef] [PubMed]
- McAnulty, S.R.; McAnulty, L.S.; Nieman, D.C.; Morrow, J.D.; Utter, A.C.; Dumke, C.L. Effect of resistance exercise and carbohydrate ingestion on oxidative stress. Free Radic. Res. 2005, 39, 1219–1224. [Google Scholar] [CrossRef] [PubMed]
- Watson, T.A.; Callister, R.; Taylor, R.D.; Sibbritt, D.W.; MacDonald-Wicks, L.K.; Garg, M.L. Antioxidant restriction and oxidative stress in short-duration exhaustive exercise. Med. Sci. Sports Exerc. 2005, 37, 63–71. Available online: http://www.ncbi.nlm.nih.gov/pubmed/15632670 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Becatti, M.; Mannucci, A.; Barygina, V.; Mascherini, G.; Emmi, G.; Silvestri, E.; Wright, D.; Taddei, N.; Galanti, G.; Fiorillo, C. Redox status alterations during the competitive season in élite soccer players: Focus on peripheral leukocyte-derived ROS. Intern. Emerg. Med. 2017, 12, 777–788. Available online: http://www.ncbi.nlm.nih.gov/pubmed/28361355 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Cavarretta, E.; Peruzzi, M.; Del Vescovo, R.; Di Pilla, F.; Gobbi, G.; Serdoz, A.; Ferrara, R.; Schirone, L.; Sciarretta, S.; Nocella, C.; et al. Dark Chocolate Intake Positively Modulates Redox Status and Markers of Muscular Damage in Elite Football Athletes: A Randomized Controlled Study. Oxid. Med. Cell. Longev. 2018, 2018, 4061901. Available online: http://www.ncbi.nlm.nih.gov/pubmed/30584461 (accessed on 15 May 2019). [CrossRef]
- Kozakiewicz, M.; Rowiński, R.; Kornatowski, M.; Dąbrowski, A.; Kędziora-Kornatowska, K.; Strachecka, A. Relation of Moderate Physical Activity to Blood Markers of Oxidative Stress and Antioxidant Defense in the Elderly. Oxid. Med. Cell. Longev. 2019, 2019, 5123628. Available online: http://www.ncbi.nlm.nih.gov/pubmed/30886673 (accessed on 15 May 2019).
- Barranco-Ruiz, Y.; Aragón-Vela, J.; Casals, C.; Martínez-Amat, A.; Villa-González, E.; Huertas, J.R. Lifelong amateur endurance practice attenuates oxidative stress and prevents muscle wasting in senior adults. J. Sports Med. Phys. Fit. 2017, 57, 670–677. Available online: http://www.ncbi.nlm.nih.gov/pubmed/27045740 (accessed on 15 May 2019).
- Mrakic-Sposta, S.; Gussoni, M.; Porcelli, S.; Pugliese, L.; Pavei, G.; Bellistri, G.; Montorsi, M.; Tacchini, P.; Vezzoli, A. Training Effects on ROS Production Determined by Electron Paramagnetic Resonance in Master Swimmers. Oxid. Med. Cell. Longev. 2015, 2015, 804794. Available online: http://www.ncbi.nlm.nih.gov/pubmed/25874024 (accessed on 15 May 2019). [CrossRef]
- Vezzoli, A.; Pugliese, L.; Marzorati, M.; Serpiello, F.R.; La Torre, A.; Porcelli, S. Time-Course Changes of Oxidative Stress Response to High-Intensity Discontinuous Training versus Moderate-Intensity Continuous Training in Masters Runners. PLoS ONE 2014, 9, e87506. Available online: http://www.ncbi.nlm.nih.gov/pubmed/24498121 (accessed on 15 May 2019). [CrossRef]
- Sousa, C.V.; Aguiar, S.S.; Santos, P.A.; Barbosa, L.P.; Knechtle, B.; Nikolaidis, P.T.; Deus, L.A.; Sales, M.M.; Rosa, E.C.; Rosa, T.S.; et al. Telomere length and redox balance in master endurance runners: The role of nitric oxide. Exp. Gerontol. 2019, 117, 113–118. Available online: http://www.ncbi.nlm.nih.gov/pubmed/30481549 (accessed on 15 May 2019). [CrossRef]
- Marzatico, F.; Pansarasa, O.; Bertorelli, L.; Somenzini, L.; Della Valle, G. Blood free radical antioxidant enzymes and lipid peroxides following long-distance and lactacidemic performances in highly trained aerobic and sprint athletes. J. Sports Med. Phys. Fit. 1997, 37, 235–239. Available online: http://www.ncbi.nlm.nih.gov/pubmed/9509820 (accessed on 15 May 2019).
- Dillard, C.J.; Litov, R.E.; Savin, W.M.; Dumelin, E.E.; Tappel, A.L. Effects of exercise, vitamin E, and ozone on pulmonary function and lipid peroxidation. J. Appl. Physiol. 1978, 45, 927–932. Available online: http://www.ncbi.nlm.nih.gov/pubmed/730598 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Brown, S.J.; Child, R.B.; Day, S.H.; Donnelly, A.E. Indices of skeletal muscle damage and connective tissue breakdown following eccentric muscle contractions. Eur. J. Appl. Physiol. Occup. Physiol. 1997, 75, 369–374. [Google Scholar] [CrossRef] [PubMed]
- Kjaer, M.; Magnusson, P.; Krogsgaard, M.; Boysen Møller, J.; Olesen, J.; Heinemeier, K.; Hansen, M.; Haraldsson, B.; Koskinen, S.; Esmarck, B.; et al. Extracellular matrix adaptation of tendon and skeletal muscle to exercise. J. Anat. 2006, 208, 445–450. Available online: http://www.ncbi.nlm.nih.gov/pubmed/16637870 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Fridén, J.; Lieber, R.L. Structural and mechanical basis of exercise-induced muscle injury. Med. Sci. Sports Exerc. 1992, 24, 521–530. Available online: http://www.ncbi.nlm.nih.gov/pubmed/1569848 (accessed on 15 May 2019). [CrossRef]
- McGinley, C.; Shafat, A.; Donnelly, A.E. Does Antioxidant Vitamin Supplementation Protect against Muscle Damage? Sport Med. 2009, 39, 1011–1032. Available online: http://www.ncbi.nlm.nih.gov/pubmed/19902983 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Butterfield, T.A. Eccentric Exercise In Vivo. Exerc. Sport Sci. Rev. 2010, 38, 51–60. Available online: http://www.ncbi.nlm.nih.gov/pubmed/20335736 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Smith, M.A.; Reid, M.B. Redox modulation of contractile function in respiratory and limb skeletal muscle. Respir. Physiol. Neurobiol. 2006, 151, 229–241. Available online: https://linkinghub.elsevier.com/retrieve/pii/S1569904806000036 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Radak, Z.; Taylor, A.W.; Ohno, H.; Goto, S. Adaptation to exercise-induced oxidative stress: From muscle to brain. Exerc. Immunol. Rev. 2001, 7, 90–107. Available online: http://www.ncbi.nlm.nih.gov/pubmed/11579750 (accessed on 15 May 2019).
- Reid, M.B. Free radicals and muscle fatigue: Of ROS, canaries, and the IOC. Free Radic. Biol. Med. 2008, 44, 169–179. Available online: https://linkinghub.elsevier.com/retrieve/pii/S0891584907001797 (accessed on 15 May 2019). [CrossRef]
- Akimoto, A.K.; Miranda-Vilela, A.L.; Alves, P.C.Z.; Pereira, L.C.; Lordelo, G.S.; Hiragi, C.; da Silva, I.C.R.; Grisolia, C.K.; Klautau-Guimarães, M. Evaluation of gene polymorphisms in exercise-induced oxidative stress and damage. Free Radic. Res. 2010, 44, 322–331. [Google Scholar] [CrossRef] [PubMed]
- Ahmetov, I.I.; Naumov, V.A.; Donnikov, A.E.; Maciejewska-Karłowska, A.; Kostryukova, E.S.; Larin, A.K.; Maykova, E.V.; Alexeev, D.G.; Fedotovskaya, O.N.; Generozov, E.V.; et al. SOD2 gene polymorphism and muscle damage markers in elite athletes. Free Radic. Res. 2014, 48, 948–955. [Google Scholar] [CrossRef] [PubMed]
- Skenderi, K.P.; Tsironi, M.; Lazaropoulou, C.; Anastasiou, C.A.; Matalas, A.L.; Kanavaki, I.; Thalmann, M.; Goussetis, E.; Papassotiriou, I.; Chrousos, G.P. Changes in free radical generation and antioxidant capacity during ultramarathon foot race. Eur. J. Clin. Investig. 2008, 38, 159–165. [Google Scholar] [CrossRef]
- Perrea, A.; Vlachos, I.S.; Korou, L.M.; Doulamis, I.P.; Exarhopoulou, K.; Kypraios, G.; Kalofoutis, A.; Perrea, D.N. Comparison of the short-term oxidative stress response in National League basketball and soccer adolescent athletes. Angiology 2014, 65, 624–629. [Google Scholar] [CrossRef] [PubMed]
- Myburgh, K.H. Polyphenol supplementation: Benefits for exercise performance or oxidative stress? Sports Med. 2014, 44, S57–S70. Available online: http://www.ncbi.nlm.nih.gov/pubmed/24791917 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Mello, R.; Gomes, D.; Paz, G.A.; Nasser, I.; Miranda, H.; Salerno, V.P. Oxidative stress and antioxidant biomarker responses after a moderate-intensity soccer training session. Res. Sport Med. 2017, 25, 322–332. [Google Scholar] [CrossRef]
- Spanidis, Y.; Goutzourelas, N.; Stagos, D.; Mpesios, A.; Priftis, A.; Bar-Or, D.; Spandidos, D.A.; Tsatsakis, A.M.; Leon, G.; Kouretas, D. Variations in oxidative stress markers in elite basketball players at the beginning and end of a season. Exp. Ther. Med. 2016, 11, 147–153. [Google Scholar] [CrossRef][Green Version]
- Djordjevic, D.; Cubrilo, D.; Macura, M.; Barudzic, N.; Djuric, D.; Jakovljevic, V. The influence of training status on oxidative stress in young male handball players. Mol. Cell. Biochem. 2011, 351, 251–259. [Google Scholar] [CrossRef]
- Hadžović-Džuvo, A.; Valjevac, A.; Lepara, O.; Pjanić, S.; Hadžimuratović, A.; Mekić, A. Oxidative stress status in elite athletes engaged in different sport disciplines. Bosn. J. Basic Med. Sci. 2014, 14, 56–62. Available online: http://www.ncbi.nlm.nih.gov/pubmed/24856375 (accessed on 15 May 2019). [CrossRef][Green Version]
- Marin, D.P.; Bolin, A.P.; Campoio, T.R.; Guerra, B.A.; Otton, R. Oxidative stress and antioxidant status response of handball athletes: Implications for sport training monitoring. Int. Immunopharmacol. 2013, 17, 462–470. Available online: https://www.sciencedirect.com/science/article/pii/S1567576913002944?via%3Dihub (accessed on 15 May 2019). [CrossRef]
- Marin, D.P.; dos Santos, R.; Bolin, A.P.; Guerra, B.A.; Hatanaka, E.; Otton, R. Cytokines and oxidative stress status following a handball game in elite male players. Oxid. Med. Cell. Longev. 2011, 2011, 804873. Available online: http://www.hindawi.com/journals/omcl/2011/804873/ (accessed on 15 May 2019). [CrossRef] [PubMed]
- León-López, J.; Calderón-Soto, C.; Pérez-Sánchez, M.; Feriche, B.; Iglesias, X.; Chaverri, D.; Rodríguez, F.A. Oxidative stress in elite athletes training at moderate altitude and at sea level. Eur. J. Sport Sci. 2018, 18, 832–841. Available online: http://www.ncbi.nlm.nih.gov/pubmed/29575975 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Vecchio, M.; Currò, M.; Trimarchi, F.; Naccari, S.; Caccamo, D.; Ientile, R.; Barreca, D.; Di Mauro, D. The Oxidative Stress Response in Elite Water Polo Players: Effects of Genetic Background. Biomed. Res. Int. 2017, 2017, 7019694. Available online: https://www.hindawi.com/journals/bmri/2017/7019694/ (accessed on 15 May 2019). [CrossRef] [PubMed]
- Dinarello, C.A. Immunological and Inflammatory Functions of the Interleukin-1 Family. Annu. Rev. Immunol. 2009, 27, 519–550. Available online: http://www.ncbi.nlm.nih.gov/pubmed/19302047 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Santtila, S.; Savinainen, K.; Hurme, M. Presence of the IL-1RA allele 2 (IL1RN*2) is associated with enhanced IL-1beta production in vitro. Scand. J. Immunol. 1998, 47, 195–198. Available online: http://www.ncbi.nlm.nih.gov/pubmed/9519856 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Hill, M.; Goldspink, G. Expression and splicing of the insulin-like growth factor gene in rodent muscle is associated with muscle satellite (stem) cell activation following local tissue damage. J. Physiol. 2003, 549, 409–418. Available online: http://www.ncbi.nlm.nih.gov/pubmed/12692175 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Dobrowolny, G.; Giacinti, C.; Pelosi, L.; Nicoletti, C.; Winn, N.; Barberi, L.; Molinaro, M.; Rosenthal, N.; Musarò, A. Muscle expression of a local Igf-1 isoform protects motor neurons in an ALS mouse model. J. Cell Biol. 2005, 168, 193–199. Available online: http://www.ncbi.nlm.nih.gov/pubmed/15657392 (accessed on 15 May 2019). [CrossRef]
- Bedair, H.S.; Karthikeyan, T.; Quintero, A.; Li, Y.; Huard, J. Angiotensin II Receptor Blockade Administered after Injury Improves Muscle Regeneration and Decreases Fibrosis in Normal Skeletal Muscle. Am. J. Sports Med. 2008, 36, 1548–1554. [Google Scholar] [CrossRef]
- Al-Chalabi, A.; Leigh, P.N. Trouble on the pitch: Are professional football players at increased risk of developing amyotrophic lateral sclerosis? Brain 2005, 128, 451–453. [Google Scholar] [CrossRef][Green Version]
- Liu, Z.; Zhou, T.; Ziegler, A.C.; Dimitrion, P.; Zuo, L. Oxidative Stress in Neurodegenerative Diseases: From Molecular Mechanisms to Clinical Applications. Oxid. Med. Cell. Longev. 2017, 2017, 2525967. Available online: https://www.hindawi.com/journals/omcl/2017/2525967/ (accessed on 15 May 2019). [CrossRef]
- Albers, D.S.; Flint Beal, M. Mitochondrial dysfunction and oxidative stress in aging and neurodegenerative disease. In Advances in Dementia Research; Springer: Vienna, Austria, 2000; pp. 133–154. [Google Scholar]
- Mitchell, J.D. Amyotrophic lateral sclerosis: Toxins and environment. Amyotroph. Lateral Scler. Other Mot. Neuron Disord 2000, 1, 235–250. Available online: http://www.ncbi.nlm.nih.gov/pubmed/11465017 (accessed on 15 May 2019). [CrossRef]
- Armon, C. An Evidence-Based Medicine Approach to the Evaluation of the Role of Exogenous Risk Factors in Sporadic Amyotrophic Lateral Sclerosis. Neuroepidemiology 2003, 22, 217–228. Available online: https://www.karger.com/Article/FullText/70562 (accessed on 15 May 2019). [CrossRef]
- Gotkine, M.; Friedlander, Y.; Hochner, H. Triathletes are over-represented in a population of patients with ALS. Amyotroph. Lateral Scler. Front. Degener. 2014, 15, 534–536. Available online: http://www.ncbi.nlm.nih.gov/pubmed/25007701 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Pupillo, E.; Messina, P.; Logroscino, G.; Zoccolella, S.; Chiò, A.; Calvo, A.; Corbo, M.; Lunetta, C.; Micheli, A.; Millul, A.; et al. Trauma and amyotrophic lateral sclerosis: A case-control study from a population-based registry. Eur. J. Neurol. 2012, 19, 1509–1517. Available online: http://www.ncbi.nlm.nih.gov/pubmed/22537412 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Siciliano, G.; D’Avino, C.; Corona, A.; Barsacchi, R.; Kusmic, C.; Rocchi, A.; Pastorini, E.; Murri, L. Impaired oxidative metabolism and lipid peroxidation in exercising muscle from ALS patients. Amyotroph. Lateral Scler. Other Mot. Neuron Disord. 2002, 3, 57–62. [Google Scholar] [CrossRef] [PubMed]
- Serbest, G.; Burkhardt, M.F.; Siman, R.; Raghupathi, R.; Saatman, K.E. Temporal Profiles of Cytoskeletal Protein Loss following Traumatic Axonal Injury in Mice. Neurochem. Res. 2007, 32, 2006–2014. [Google Scholar] [CrossRef]
- Cole, K.; Perez-Polo, J.R. Neuronal trauma model: In search of Thanatos. Int. J. Dev. Neurosci. 2004, 22, 485–496. Available online: https://linkinghub.elsevier.com/retrieve/pii/S0736574804000863 (accessed on 15 May 2019). [CrossRef]
- Chiò, A.; Benzi, G.; Dossena, M.; Mutani, R.; Mora, G. Severely increased risk of amyotrophic lateral sclerosis among Italian professional football players. Brain 2005, 128, 472–476. [Google Scholar] [CrossRef]
- Chio, A.; Calvo, A.; Dossena, M.; Ghiglione, P.; Mutani, R.; Mora, G. ALS in Italian professional soccer players: The risk is still present and could be soccer-specific. Amyotroph. Lateral Scler. 2009, 10, 205–209. [Google Scholar] [CrossRef]
- Lehman, E.J.; Hein, M.J.; Baron, S.L.; Gersic, C.M. Neurodegenerative causes of death among retired National Football League players. Neurology 2012, 79, 1970–1974. Available online: http://www.ncbi.nlm.nih.gov/pubmed/22955124 (accessed on 15 May 2019). [CrossRef]
- Abel, E.L. Football Increases the Risk for Lou Gehrig’s Disease, Amyotrophic Lateral Sclerosis. Percept. Mot. Ski. 2007, 104, 1251–1254. Available online: http://www.ncbi.nlm.nih.gov/pubmed/17879657 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Valenti, M.; Pontieri, F.E.; Conti, F.; Altobelli, E.; Manzoni, T.; Frati, L. Amyotrophic lateral sclerosis and sports: A case-control study. Eur. J. Neurol. 2005, 12, 223–225. Available online: http://www.ncbi.nlm.nih.gov/pubmed/15693813 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Asan, Z. Spinal Concussion in Adults: Transient Neuropraxia of Spinal Cord Exposed to Vertical Forces. World Neurosurg. 2018, 114, e1284–e1289. Available online: http://www.ncbi.nlm.nih.gov/pubmed/29626691 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Blecher, R.; Elliott, M.A.; Yilmaz, E.; Dettori, J.R.; Oskouian, R.J.; Patel, A.; Clarke, A.; Hutton, M.; McGuire, R.; Dunn, R.; et al. Contact Sports as a Risk Factor for Amyotrophic Lateral Sclerosis: A Systematic Review. Glob. Spine J. 2019, 9, 104–118. Available online: http://www.ncbi.nlm.nih.gov/pubmed/30775214 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Margaritis, I.; Rousseau, A.S. Does physical exercise modify antioxidant requirements? Nutr. Res. Rev. 2008, 21, 3–12. Available online: http://www.ncbi.nlm.nih.gov/pubmed/19079851 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Mastaloudis, A.; Morrow, J.D.; Hopkins, D.W.; Devaraj, S.; Traber, M.G. Antioxidant supplementation prevents exercise-induced lipid peroxidation, but not inflammation, in ultramarathon runners. Free Radic. Biol. Med. 2004, 36, 1329–1341. Available online: http://www.ncbi.nlm.nih.gov/pubmed/15110397 (accessed on 15 May 2019). [CrossRef]
- Jówko, E.; Długołęcka, B.; Makaruk, B.; Cieśliński, I. The effect of green tea extract supplementation on exercise-induced oxidative stress parameters in male sprinters. Eur. J. Nutr. 2015, 54, 783–791. Available online: http://www.ncbi.nlm.nih.gov/pubmed/25120110 (accessed on 15 May 2019). [CrossRef]
- Andújar, I.; Recio, M.C.; Giner, R.M.; Ríos, J.L. Cocoa Polyphenols and Their Potential Benefits for Human Health. Oxid. Med. Cell. Longev. 2012, 2012, 906252. Available online: http://www.hindawi.com/journals/omcl/2012/906252/ (accessed on 15 May 2019). [CrossRef]
- Slattery, K.M.; Dascombe, B.; Wallace, L.K.; Bentley, D.J.; Coutts, A.J. Effect of N-acetylcysteine on Cycling Performance after Intensified Training. Med. Sci. Sport Exerc. 2014, 46, 1114–1123. Available online: http://www.ncbi.nlm.nih.gov/pubmed/24576857 (accessed on 15 May 2019). [CrossRef]
- Gross, M.; Baum, O.; Hoppeler, H. Antioxidant supplementation and endurance training: Win or loss? Eur. J. Sport Sci. 2011, 11, 27–32. [Google Scholar] [CrossRef]
- Chou, C.C.; Sung, Y.C.; Davison, G.; Chen, C.Y.; Liao, Y.H. Short-Term High-Dose Vitamin C and E Supplementation Attenuates Muscle Damage and Inflammatory Responses to Repeated Taekwondo Competitions: A Randomized Placebo-Controlled Trial. Int. J. Med. Sci. 2018, 15, 1217–1226. Available online: http://www.ncbi.nlm.nih.gov/pubmed/30123060 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Braakhuis, A.J.; Hopkins, W.G. Impact of Dietary Antioxidants on Sport Performance: A Review. Sport Med. 2015, 45, 939–955. Available online: http://www.ncbi.nlm.nih.gov/pubmed/25790792 (accessed on 15 May 2019). [CrossRef] [PubMed]
- de Oliveira, D.C.X.; Rosa, F.T.; Simões-Ambrósio, L.; Jordao, A.A.; Deminice, R. Antioxidant vitamin supplementation prevents oxidative stress but does not enhance performance in young football athletes. Nutrition 2019, 63–64, 29–35. Available online: http://www.ncbi.nlm.nih.gov/pubmed/30927644 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Manach, C.; Scalbert, A.; Morand, C.; Rémésy, C.; Jiménez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. Available online: http://www.ncbi.nlm.nih.gov/pubmed/15113710 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Juszkiewicz, A.; Glapa, A.; Basta, P.; Petriczko, E.; Żołnowski, K.; Machaliński, B.; Trzeciak, J.; Łuczkowska, K.; Skarpańska-Stejnborn, A. The effect of L-theanine supplementation on the immune system of athletes exposed to strenuous physical exercise. J. Int. Soc. Sports Nutr. 2019, 16, 7. Available online: http://www.ncbi.nlm.nih.gov/pubmed/30770758 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Machado, Á.S.; da Silva, W.; Souza, M.A.; Carpes, F.P. Green Tea Extract Preserves Neuromuscular Activation and Muscle Damage Markers in Athletes under Cumulative Fatigue. Front. Physiol. 2018, 9, 1137. Available online: http://www.ncbi.nlm.nih.gov/pubmed/30174618 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Medved, I.; Brown, M.J.; Bjorksten, A.R.; Murphy, K.T.; Petersen, A.C.; Sostaric, S.; Gong, X.; McKenna, M.J. N-acetylcysteine enhances muscle cysteine and glutathione availability and attenuates fatigue during prolonged exercise in endurance-trained individuals. J. Appl. Physiol. 2004, 97, 1477–1485. Available online: http://www.ncbi.nlm.nih.gov/pubmed/15194675 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Holdiness, M.R. Clinical Pharmacokinetics of N-Acetylcysteine. Clin. Pharmacokinet. 1991, 20, 123–134. Available online: http://www.ncbi.nlm.nih.gov/pubmed/2029805 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Antonioni, A.; Fantini, C.; Dimauro, I.; Caporossi, D. Redox homeostasis in sport: Do athletes really need antioxidant support? Res. Sport Med. 2019, 27, 147–165. Available online: http://www.ncbi.nlm.nih.gov/pubmed/30596287 (accessed on 15 May 2019). [CrossRef]
- Poljsak, B.; Šuput, D.; Milisav, I. Achieving the balance between ROS and antioxidants: When to use the synthetic antioxidants. Oxid. Med. Cell. Longev. 2013, 2013, 956792. Available online: http://www.ncbi.nlm.nih.gov/pubmed/23738047 (accessed on 15 May 2019). [CrossRef]
- Merry, T.L.; Ristow, M. Do antioxidant supplements interfere with skeletal muscle adaptation to exercise training? J. Physiol. 2016, 594, 5135–5147. Available online: http://www.ncbi.nlm.nih.gov/pubmed/26638792 (accessed on 15 May 2019). [CrossRef]
- Margaritelis, N.V.; Paschalis, V.; Theodorou, A.A.; Kyparos, A.; Nikolaidis, M.G. Antioxidants in Personalized Nutrition and Exercise. Adv. Nutr. 2018, 9, 813–823. Available online: https://academic.oup.com/advances/article/9/6/813/5106979 (accessed on 15 May 2019). [CrossRef]
- Moraes, A.; Andreato, L.V.; Branco, B.H.M.; Silva, E.L.; Gonçalves, M.A.; Santos, R.Z.; Becker, A.M.; Cavalcante, L.; Casagrande, F.; Benetti, M. Effects of N-acetylcysteine supplementation on cellular damage and oxidative stress indicators in volleyball athletes. J. Exerc. Rehabil. 2018, 14, 802. Available online: http://www.ncbi.nlm.nih.gov/pubmed/30443526 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Riva, A.; Vitale, J.A.; Belcaro, G.; Hu, S.; Feragalli, B.; Vinciguerra, G.; Cacchio, M.; Bonanni, E.; Giacomelli, L.; Eggenhöffner, R.; et al. Quercetin phytosome® in triathlon athletes: A pilot registry study. Minerva Med. 2018, 109, 285–289. Available online: http://www.ncbi.nlm.nih.gov/pubmed/29947492 (accessed on 15 May 2019).
- Taghizadeh, M.; Malekian, E.; Memarzadeh, M.R.; Mohammadi, A.A.; Asemi, Z. Grape Seed Extract Supplementation and the Effects on the Biomarkers of Oxidative Stress and Metabolic Profiles in Female Volleyball Players: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Iran. Red Crescent Med. J. 2016, 18, e31314. Available online: http://ircmj.neoscriber.org/en/articles/16766.html (accessed on 15 May 2019). [CrossRef]
- Capó, X.; Martorell, M.; Busquets-Cortés, C.; Sureda, A.; Riera, J.; Drobnic, F.; Tur, J.A.; Pons, A. Effects of dietary almond- and olive oil-based docosahexaenoic acid- and vitamin E-enriched beverage supplementation on athletic performance and oxidative stress markers. Food Funct. 2016, 7, 4920–4934. Available online: http://www.ncbi.nlm.nih.gov/pubmed/27841405 (accessed on 15 May 2019). [CrossRef]
- Hadi, A.; Pourmasoumi, M.; Kafeshani, M.; Karimian, J.; Maracy, M.R.; Entezari, M.H. The Effect of Green Tea and Sour Tea (Hibiscus sabdariffa L.) Supplementation on Oxidative Stress and Muscle Damage in Athletes. J. Diet. Suppl. 2017, 14, 346–357. Available online: http://www.ncbi.nlm.nih.gov/pubmed/27736246 (accessed on 15 May 2019). [CrossRef]
- Żychowska, M.; Jastrzębski, Z.; Chruściński, G.; Michałowska-Sawczyn, M.; Nowak-Zaleska, A. Vitamin C, A and E supplementation decreases the expression of HSPA1A and HSPB1 genes in the leukocytes of young polish figure skaters during a 10-day training camp. J. Int. Soc. Sports Nutr. 2015, 12, 9. Available online: http://www.ncbi.nlm.nih.gov/pubmed/25722659 (accessed on 15 May 2019). [CrossRef]
- Skarpańska-Stejnborn, A.; Basta, P.; Sadowska, J.; Pilaczyńska-Szczeńniak, Ł. Effect of supplementation with chokeberry juice on the inflammatory status and markers of iron metabolism in rowers. J. Int. Soc. Sports Nutr. 2014, 11, 48. Available online: http://www.ncbi.nlm.nih.gov/pubmed/25298754 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Nieman, D.C.; Gillitt, N.D.; Knab, A.M.; Shanely, R.A.; Pappan, K.L.; Jin, F.; Lila, M.A. Influence of a Polyphenol-Enriched Protein Powder on Exercise-Induced Inflammation and Oxidative Stress in Athletes: A Randomized Trial Using a Metabolomics Approach. PLoS ONE 2013, 8, e72215. Available online: http://www.ncbi.nlm.nih.gov/pubmed/23967286 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Askari, G.; Ghiasvand, R.; Feizi, A.; Ghanadian, S.M.; Karimian, J. The effect of quercetin supplementation on selected markers of inflammation and oxidative stress. J. Res. Med. Sci. 2012, 17, 637–641. Available online: http://www.ncbi.nlm.nih.gov/pubmed/23798923 (accessed on 15 May 2019). [PubMed]
- Askari, G.; Hajishafiee, M.; Ghiasvand, R.; Hariri, M.; Darvishi, L.; Ghassemi, S.; Iraj, B.; Hovsepian, V. Quercetin and vitamin C supplementation: Effects on lipid profile and muscle damage in male athletes. Int. J. Prev. Med. 2013, 4, S58–S62. Available online: http://www.ncbi.nlm.nih.gov/pubmed/23717772 (accessed on 15 May 2019). [PubMed]
- Taghiyar, M.; Darvishi, L.; Askari, G.; Feizi, A.; Hariri, M.; Mashhadi, N.S.; Ghiasvand, R. The effect of vitamin C and e supplementation on muscle damage and oxidative stress in female athletes: A clinical trial. Int. J. Prev. Med. 2013, 4, S16–S23. Available online: http://www.ncbi.nlm.nih.gov/pubmed/23717764 (accessed on 15 May 2019). [PubMed]
- Díaz-Castro, J.; Guisado, R.; Kajarabille, N.; García, C.; Guisado, I.M.; de Teresa, C.; Ochoa, J.J. Coenzyme Q10 supplementation ameliorates inflammatory signaling and oxidative stress associated with strenuous exercise. Eur. J. Nutr. 2012, 51, 791–799. Available online: http://www.ncbi.nlm.nih.gov/pubmed/21990004 (accessed on 15 May 2019). [CrossRef] [PubMed]
- Nishizawa, M.; Hara, T.; Miura, T.; Fujita, S.; Yoshigai, E.; Ue, H.; Hayashi, Y.; Kwon, A.H.; Okumura, T.; Isaka, T. Supplementation with a Flavanol-rich Lychee Fruit Extract Influences the Inflammatory Status of Young Athletes. Phyther. Res. 2011, 25, 1486–1493. Available online: http://www.ncbi.nlm.nih.gov/pubmed/21780209 (accessed on 15 May 2019). [CrossRef]
- Teixeira, V.H.; Valente, H.F.; Casal, S.I.; Marques, A.F.; Moreira, P.A. Antioxidants do not prevent postexercise peroxidation and may delay muscle recovery. Med. Sci. Sports Exerc. 2009, 41, 1752–1760. Available online: https://insights.ovid.com/crossref?an=00005768-200909000-00010 (accessed on 15 May 2019). [CrossRef]
- Ristow, M.; Zarse, K.; Oberbach, A.; Klöting, N.; Birringer, M.; Kiehntopf, M.; Stumvoll, M.; Kahn, C.R.; Blüher, M. Antioxidants prevent health-promoting effects of physical exercise in humans. Proc. Natl. Acad. Sci. USA 2009, 106, 8665–8670. [Google Scholar] [CrossRef]
- Peternelj, T.T.; Coombes, J.S. Antioxidant supplementation during exercise training: Beneficial or detrimental? Sports Med. 2011, 41, 1043–1069. [Google Scholar] [CrossRef]
- Dickinson, B.C.; Peltier, J.; Stone, D.; Schaffer, D.V.; Chang, C.J. Nox2 redox signaling maintains essential cell populations in the brain. Nat. Chem. Biol. 2011, 7, 106–112. Available online: http://www.ncbi.nlm.nih.gov/pubmed/21186346 (accessed on 15 May 2019). [CrossRef]
- Zhang, L.; Jope, R.S. Oxidative stress differentially modulates phosphorylation of ERK, p38 and CREB induced by NGF or EGF in PC12 cells. Neurobiol. Aging 2019, 20, 271–278. Available online: http://www.ncbi.nlm.nih.gov/pubmed/10588574 (accessed on 15 May 2019). [CrossRef]
- Pham-Huy, L.A.; He, H.; Pham-Huy, C. Free radicals, antioxidants in disease and health. Int. J. Biomed. Sci. 2008, 4, 89–96. Available online: http://www.ncbi.nlm.nih.gov/pubmed/23675073 (accessed on 15 May 2019). [PubMed]
| N of Subjects Male/Female Age (y) | Type and Time of Exercise | Type of Meal | Sampling | Effects | References |
|---|---|---|---|---|---|
| AMATEURS | |||||
| 18 16/24 2.8 ± 1.4 | Running/ 33.3 ± 0.5 h | Fluids (water, beverages and sports drinks) Carbohydrate-rich food (bread, fruits, cookies and pasta) | Serum and plasma 15 min and 48 h after the end of the race | TEAC ↑ TAC ↑ TBARS ↑ AT/TBARS ↓ PCC ↓ MDA ↑ 8-iso-PGF2a ↑ GSSG and GSSG/GSH ↑ | [67] |
| 23 23/0 60 ± 1.8 | Cycling/ 8 times week | ND | Serum and plasma Baseline (vs. control group) | MDA ↓ GPx ↑ Antioxidant activity↑ | [51] |
| 33 33/0 42 ± 1.0 | Treadmill test/ 7 stages (3 min each) | 200 ml pear juice Two slices of bread | Serum Before and 30 min post exercise | TEAC ↔ TBARS ↓ GSH/GSSG ↑ | [37] |
| 10 10/0 21.1 ± 1.7 | Treadmill test/ 35 min | Recorded normal diet | Plasma and serum Before, 10 min and 30 min post exercise | SOD ↑ CAT ↑ | [38] |
| ELITE | |||||
| 13 13/0 20.7 ± 2.5 | Basket/ National League One Game | ND | Serum 40 min before and 45 min after game | TSP ↔ MPO ↔ | [68] |
| 35 35/0 21.6 ± 1.9 | Soccer/ National League One Game | ND | Serum 40 min before and 45 min after game | TSP ↓ MPO ↔ | [69] |
| 22 22/0 26.5 ± 1.9 | Soccer/ 15 min warmup and 90 min match | ND | Plasma Before and 5 min after training | Lactate↑ Glucose ↑ TAC↔ AST and ALT ↔ CK ↔ TBARS ↔ | [70] |
| 14 14/0 26.8 ± 1.2 | Basket/ 1 regular season | ND | Plasma Before and after season (6 month, 59 matches) | TAC ↑↔ TBARS ↔ GSH ↓ CAT↔ | [71] |
| 33 33/0 17.1 ± 1.1 | Handball/ Maximal progressive excercize test (bicyle ergometer) | ND | Plasma Post exercise test | SOD↑ CAT↓ VO2 max ↓ H2O2↑ NO2-↑ | [72] |
| 12 12/0 21.7 ± 6.0 | Wrestlers | ND | Plasma Baseline (vs. baseline soccer and basketball players) | AOPP ↔ MDA ↔ TAC ↔ | [73] |
| 14 14/0 22.1 ± 4.4 | Soccer | ND | Plasma Baseline (vs. baseline wrestlers and basketball players) | AOPP ↔ MDA↔ (vs. wrestlers), ↓ (vs. basketball) TAC ↔ | [73] |
| 13 13/0 20.2 ± 2.3 | Basketball | ND | Plasma Baseline (vs. wrestlers and soccer players) | AOPP ↔ MDA↔ (vs. wrestlers), ↑ (vs. soccer) TAC ↔ | [73] |
| 15 0/15 20–23 | Water polo | Dietary intake (2300-2400 Kcal/day) | Plasma and erythrocytes Baseline (vs. control group) | MDA ↑ TAS ↑ GSSG ↑ H2O2 ↑ O2-↑ SOD activity ↑ GPx ↔ | [40] |
| 19 0/19 20–23 | Football | ||||
| 10 10/0 25 ± 4.5 | Handball/ Three training periods T2–T4 (8 week/each) | ND | Plasma, erythrocytes, neutrophils and lymphocyte Before, T2, T3, T4, after T4 | TBARs ↑ Thiols ↑ CK ↑ lactate dehydrogenase ↑ aspartate aminotransferase↑ IL-6 and TNF-α ↑ uric acid ↑ | [74] |
| 14 14/0 25 ± 4.5 | Handball/ Season match (60 min) | ND | Plasma, erythrocytes Before, after 60 min and after 24h | TBARs and Thiols ↑ Antioxidant activity ↑ GSH ↓ GSSG/GSH ↔ CK ↑ Lactate dehydrogenase ↑ IL-6 and TNF-α ↑ SOD ↑ CAT↓ GPx ↔ | [75] |
| 61 27/34 21.4 ± 1.6 | Swimming/ High-intesity training session (3h, 4 weeks) | ND | Plasma, blood Before and after 4 weeks | GPx activity ↑ LPO ↑ GSSG/GSH ↑ | [76] |
| MASTER | |||||
| 16 16/0 30 ± 5 | Swimming/ High intensity discontinous training | ND | Blood Before and after 6 weeks | ROS production ↑ Antioxidant capacity ↑ | [52] |
| 20 20/0 47.8 ± 7.8 | Running/ High intensity discontinous training | ND | Plasma and urine Before and after exercise test | TBARS ↓ PC ↔ TAC ↓ 8-OH-dG ↓ | [53] |
| 10 10/0 51.6 ± 5.2 | Endurance Races | ND | Plasma Baseline (vs. control groups) | TBARS ↔ TEAC ↓ SOD ↔ CAT ↔ NO2- ↑ REDOX INDEX ↔ LTL ↓ | [54] |
| Treatment | Dose | Subjects N Kind of Sport | Study Duration | Markers | References | |
|---|---|---|---|---|---|---|
| 1 | Vitamin C and Vitamin E | 500 mg/d and 400 UI/d respectively | 21 Football athletes | 15 days | - MDA ↓ - Total lipid hydroperoxide ↓ - GSH/GSSH ↓ - FRAP ↓ - CK ↔ - VJH ↔ - Agility ↔ - Sprint test ↔ - Fatigue index ↔ - Muscle soreness ↔ | [108] |
| 2 | L-theanine | 300 mg/d | 20 Rowing athletes | 6 weeks | - IL-10 ↓ - IFN- γ ↑ - IL-2/IL-10 ↑ - IFN-γ/IL-10 ↑ - Th1/Th2 balance ↑ - CTL count ↓ - Treg/NK ↓ - Treg/ CTL ↓ - REP ↔ | [110] |
| 3 | Dark Chocolate | 40 g/d | 24 young elite male football players 15 physically active male | 30 days | - HBA ↔ - H2O2 ↓ - sNox2-dp ↔ - Myoglobin ↓ - CK ↓ - LDH ↓ | [49] |
| 4 | N-acetyl-cysteine | 1200 mg/d | 20 Male volleyball athletes | 14 days | - CK ↔ - AST ↔ - Creatinine ↓ - GPx ↔ - SOD ↔ - Glutathione ↓ - GSH ↔ - FRAP ↔ - LOOH ↔ - TBARS ↔ | [118] |
| 5 | Green Tea Extract | 500 mg/d | 22 Healthy trained men | 15 days | - CK ↓ - TBARS ↓ - Heart rate following exercise ↓ - EMG assessed neuromuscular electrical activity ↑ | [111] |
| 6 | Vitamin C and Vitamin E | 2000 mg/d and 1400 UI/d respectively | 18 Elite Taekwondo athletes | 4 days | - Myoglobin ↓ - CK ↓ - Heart rate following exercise ↔ - Blood lactate ↔ - Hemolysis ↓ - Plasma free radicals ↓ | [106] |
| 7 | Quercetin Phytosome® | 500 mg/d | 48 Amateur Thriatlon athletes | 2 weeks | - Training performance ↑ - Training efficacy ↑ - Post-run diffuse muscle pain ↓ - Cramps and localized pain ↓ - Recovery time ↓ - Plasma free radicals ↓ | [119] |
| 8 | Grape Seed Extract | 600 mg/d | 40 Female Volleyball Players | 8 weeks | - GSH ↑ - MDA ↓ - Serum insulin ↓ - HOMA-IR ↓ - CPK ↔ - TAC ↔ - NO ↔ - FPG ↔ | [120] |
| 9 | Docosahexaenoic acid- and vitamin E | 1 liter/d of isotonic beverage (278 mOsm/kg) | 10 Young male Taekwondo athletes 8 Senior athletes | 5 weeks | - Performance ↔ - Fatigue perception ↓ - Total polyphenol ↑ - MUFA and PUFA ↔ - MDA ↓ - Nitrotyrosine plasma levels ↓ - Antioxidant gene expression in PBMC ↔ | [121] |
| 10 | Green Tea and Sour Tea (Hibiscus sabdariffa L.) | 450 mg/d and 450 mg/d respectively | 54 Male soccer player | 6 weeks | - MDA ↓ - TAC ↑ - AST ↔ - CK ↔ - LDH ↔ | [122] |
| 11 | Vitamin C, Vitamin A and Vitamin E | 8 mg/kg/d, 16 ug/kg/d and 1 mg/kg/d respectively | 14 Junior female figure skaters athletes | 20 days | - HSPA1A gene expression ↓ - HSPB1 gene expression ↓ | [123] |
| 12 | Chokeberry juice | 150 ml/d | 19 Rowing athletes | 8 weeks | - IL-6 ↔ - TNF-α ↓ - TAC ↑ - UA ↔ - Myoglobin ↔ | [124] |
| 13 | Green Tea Extract | 980 mg/d | 16 Sprinter athletes | 8 weeks | - SOD ↓ - GPx ↔ - Total polyphenols ↑ - TAC ↔ - UA ↓ - MDA ↓ - CK ↔ - Lactate ↔ - Performance ↔ | [102] |
| 14 | N-acetyl-cysteine | 1200 mg/d | 10 Male thriathletes | 9 days | - Performance ↑ - TAC ↑ - TBARS ↓ - IL-6 ↓ - MPC-1 ↓ - NF-kB activity ↑ | [125] |
| 15 | Resveratrol and Quercetin | 120 mg/d and 225 mg/d respectively for 6 days; 240 mg/d and 450 mg/d respectively on day 7 | 14 Trained male adults | 7 days | - F2-isoprostanes ↓ - FRAP ↔ - TEAC ↔ - ORAC ↔ - IL-8 ↔ - CRP ↔ | [46] |
| 16 | Polyphenol-enriched protein powder (PSPC) | 40 g/d of PSPC (2136 mg/d gallic acid equivalents) | 38 Long distance runners | 17 days | - CRP ↔ - Cytokines ↔ | [125] |
| 17 | Quercetin and Vitamin C | 500 mg/d and/or 250 mg/d respectively | 60 Non-professional athletes | 8 weeks | - IL-6 ↓ - CRP ↓ - E-selectin ↔ - F2-isoprostanes ↓ | [126] |
| 18 | Quercetin and Vitamin C | 500 mg/d and 200 mg/d respectively or 500 mg/d Quercetin or 500 mg/d Vitamin C | 60 Non-professional athletes | 8 weeks | - LDH ↓ | [127] |
| 19 | Vitamin C and Vitamin E | 250 mg/d and/or 400 UI/d respectively | 64 Trained female athletes | 4 weeks | - Performance ↔ - Myoglobin ↔ | [128] |
| 20 | Coenzyme Q (10) | 30 mg on day 1 90 mg on day 2 30 mg on day 3 | 20 Amateur running athletes | 3 days | - IL-6 ↔ - TNF-α ↓ - GPx ↔ - H2O2 ↓ - CAT ↑ - TAS ↑ - Isoprostanes ↓ - 8-OHdG ↓ | [129] |
| 21 | Flavanol-rich Lychee fruit extract | 50 mg/d | 20 Male long-distance runners | 2 months | - Performance ↔ - NO ↔ - LDH ↔ - CPK ↔ - CRP ↔ - IL-6 ↔ - IL-10 ↔ - TGF-β ↑ - UA ↔ - ORAC ↔ | [130] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nocella, C.; Cammisotto, V.; Pigozzi, F.; Borrione, P.; Fossati, C.; D’Amico, A.; Cangemi, R.; Peruzzi, M.; Gobbi, G.; Ettorre, E.; et al. Impairment between Oxidant and Antioxidant Systems: Short- and Long-term Implications for Athletes’ Health. Nutrients 2019, 11, 1353. https://doi.org/10.3390/nu11061353
Nocella C, Cammisotto V, Pigozzi F, Borrione P, Fossati C, D’Amico A, Cangemi R, Peruzzi M, Gobbi G, Ettorre E, et al. Impairment between Oxidant and Antioxidant Systems: Short- and Long-term Implications for Athletes’ Health. Nutrients. 2019; 11(6):1353. https://doi.org/10.3390/nu11061353
Chicago/Turabian StyleNocella, Cristina, Vittoria Cammisotto, Fabio Pigozzi, Paolo Borrione, Chiara Fossati, Alessandra D’Amico, Roberto Cangemi, Mariangela Peruzzi, Giuliana Gobbi, Evaristo Ettorre, and et al. 2019. "Impairment between Oxidant and Antioxidant Systems: Short- and Long-term Implications for Athletes’ Health" Nutrients 11, no. 6: 1353. https://doi.org/10.3390/nu11061353
APA StyleNocella, C., Cammisotto, V., Pigozzi, F., Borrione, P., Fossati, C., D’Amico, A., Cangemi, R., Peruzzi, M., Gobbi, G., Ettorre, E., Frati, G., Cavarretta, E., Carnevale, R., & SMiLe Group. (2019). Impairment between Oxidant and Antioxidant Systems: Short- and Long-term Implications for Athletes’ Health. Nutrients, 11(6), 1353. https://doi.org/10.3390/nu11061353









