Association between 25-Hydroxyvitamin D, Parathyroid Hormone, Vitamin D and Calcium Intake, and Bone Density in Healthy Adult Women: A Cross-Sectional Analysis from the D-SOL Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Exclusion Criteria
2.2. Data Collection
2.3. Laboratory Analysis
2.4. Bone Measurements
2.5. Statistical Analysis
3. Results
3.1. Participant Characteristics
3.2. Bone Density
3.3. Correlations between Bone Parameters and Age, Weight, and BMI
3.4. Correlations between Bone Parameters and Habitual Dietary Vitamin D and Calcium Intakes
3.5. Correlations between Bone Parameters and 25(OH)D Concentrations
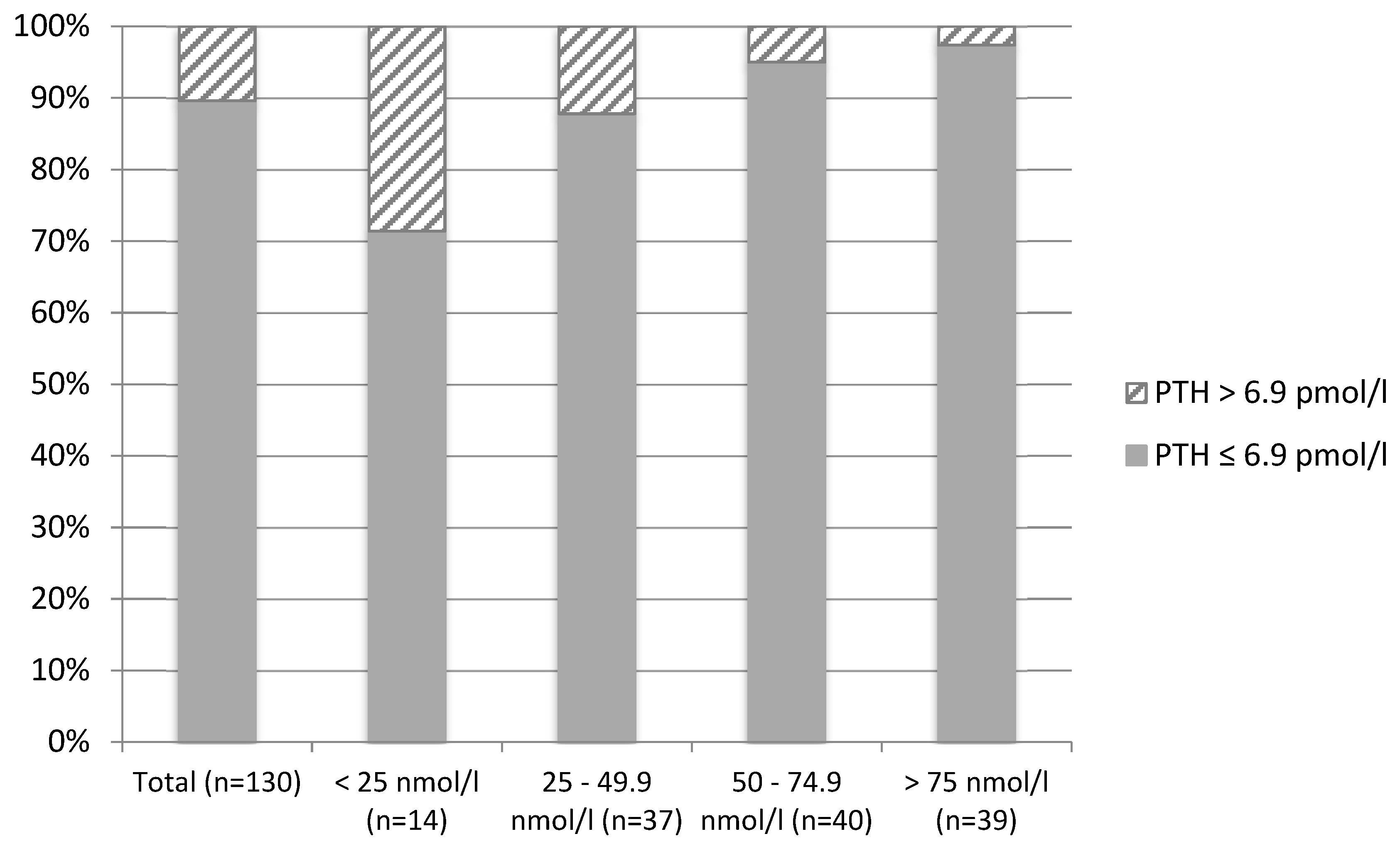
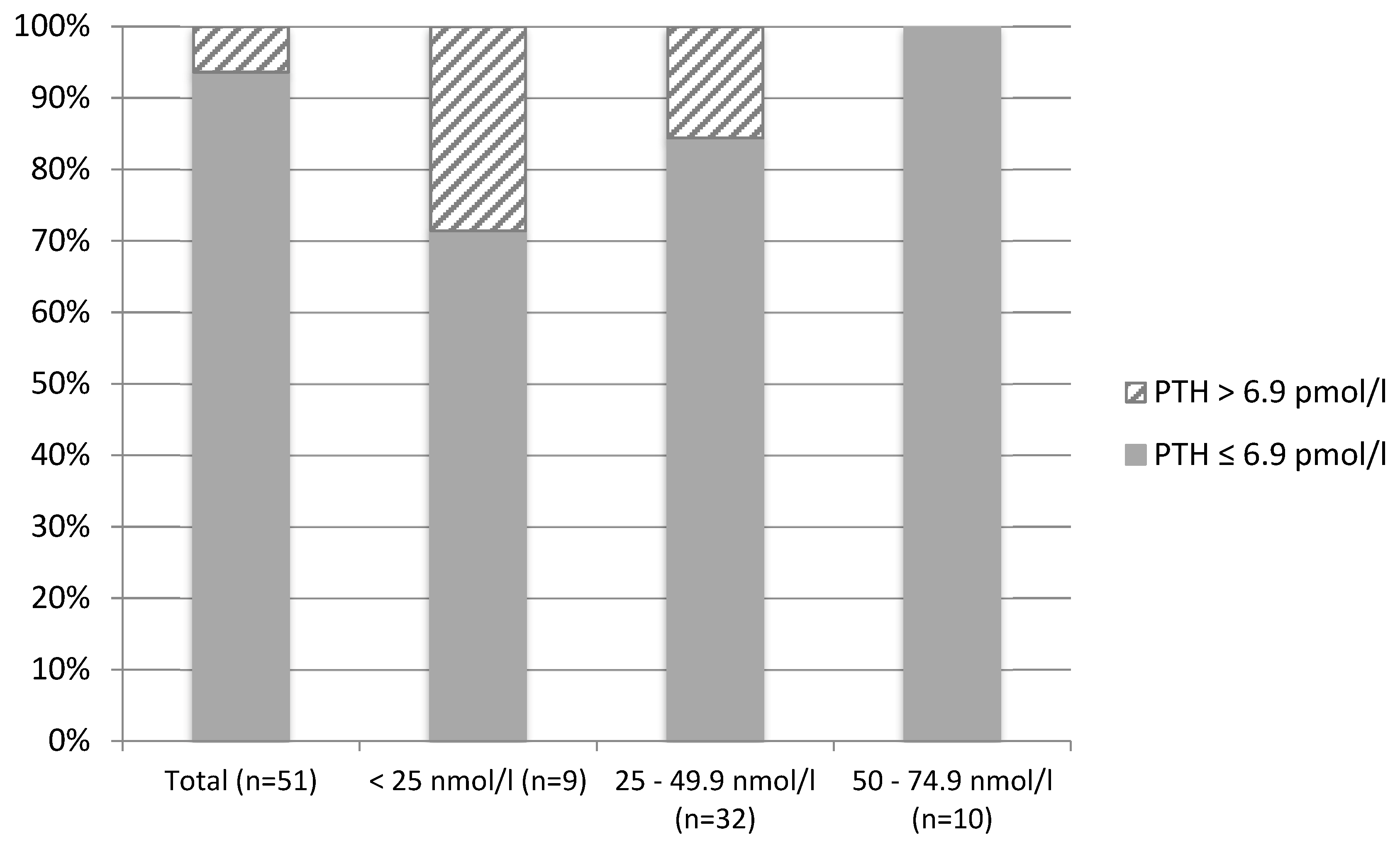
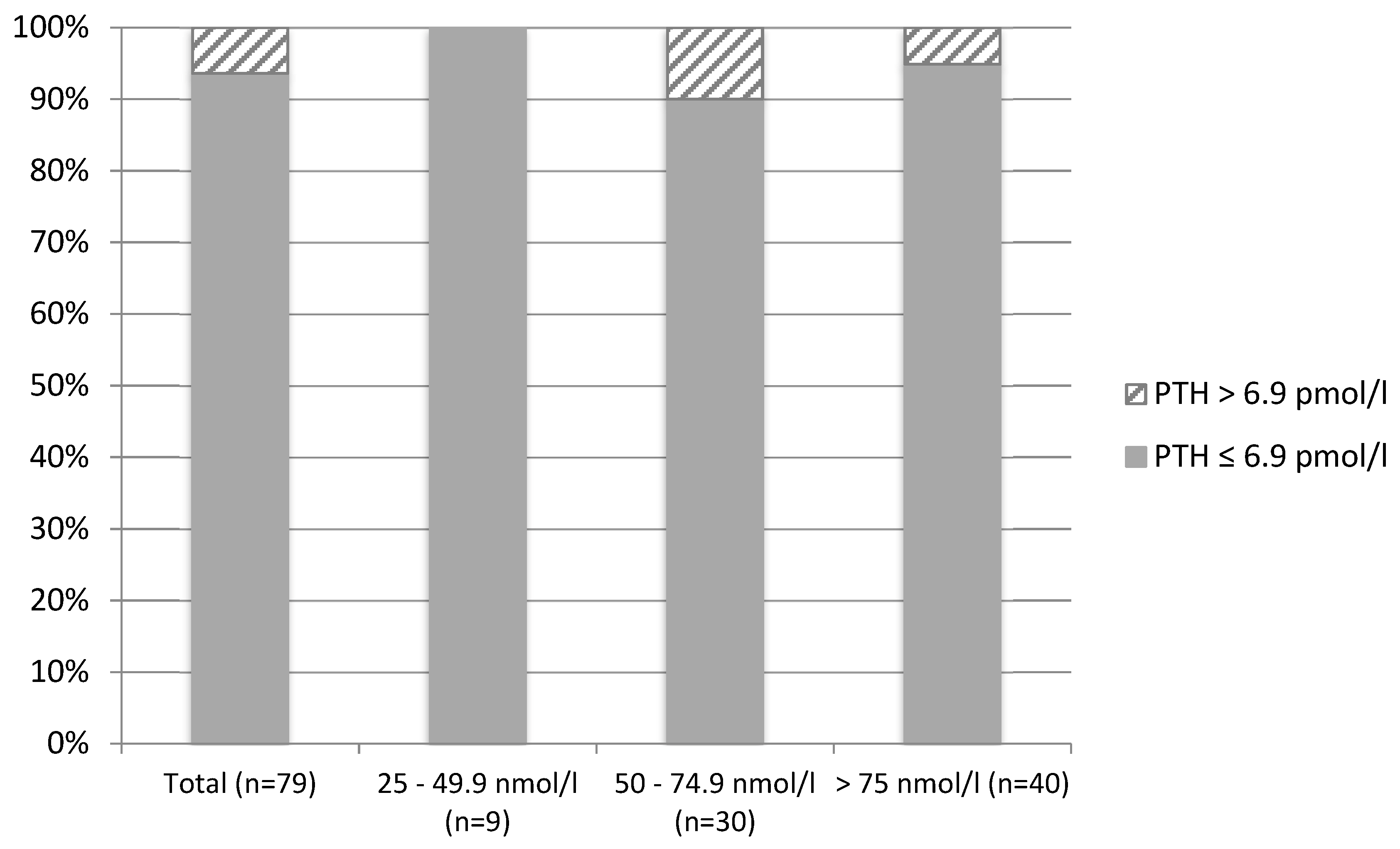
3.6. Correlations between Bone Parameters and PTH Concentrations
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Heaney, R. Calcium in the treatment of osteoporosis. In Osteoporosis: Fourth Edition; Marcus, R., Dempster, D., Cauley, J., Feldman, D., Eds.; Academic Press: Cambridge, MA, USA, 2013; pp. 1691–1700. [Google Scholar]
- Peacock, M. Calcium metabolism in health and disease. Clin. J. Am. Soc. Nephrol. 2010, 5, S23–S30. [Google Scholar] [CrossRef] [PubMed]
- DeLuca, H.F. Overview of general physiologic features and functions of vitamin D. Am. J. Clin. Nutr. 2004, 80, 1689S–1696S. [Google Scholar] [CrossRef] [PubMed]
- Reid, I.R.; Bolland, M.J.; Grey, A. Effects of vitamin D supplements on bone mineral density: A systematic review and meta-analysis. Lancet 2014, 383, 146–155. [Google Scholar] [CrossRef]
- Tang, B.M.; Eslick, G.D.; Nowson, C.; Smith, C.; Bensoussan, A. Use of calcium or calcium in combination with vitamin D supplementation to prevent fractures and bone loss in people aged 50 years and older: A meta-analysis. Lancet 2007, 370, 657–666. [Google Scholar] [CrossRef]
- Silk, L.N.; Greene, D.A.; Baker, M.K. The effect of calcium or calcium and vitamin D supplementation on bone mineral density in healthy males: A systematic review and meta-analysis. Int. J. Sport Nutr. Exerc. Metab. 2015, 25, 510–524. [Google Scholar] [CrossRef] [PubMed]
- Jackson, R.D.; LaCroix, A.Z.; Gass, M.; Wallace, R.B.; Robbins, J.; Lewis, C.E.; Bassford, T.; Beresford, S.A.; Black, H.R.; Blanchette, P.; et al. Calcium plus vitamin D supplementation and the risk of fractures. N. Engl. J. Med. 2006, 354, 669–683. [Google Scholar] [CrossRef] [PubMed]
- Chapuy, M.C.; Pamphile, R.; Paris, E.; Kempf, C.; Schlichting, M.; Arnaud, S.; Garnero, P.; Meunier, P.J. Combined calcium and vitamin D3 supplementation in elderly women: Confirmation of reversal of secondary hyperparathyroidism and hip fracture risk: The decalyos ii study. Osteoporos. Int. 2002, 13, 257–264. [Google Scholar] [CrossRef]
- Dawson-Hughes, B.; Harris, S.S.; Krall, E.A.; Dallal, G.E. Effect of calcium and vitamin D supplementation on bone density in men and women 65 years of age or older. New Engl. J. Med. 1997, 337, 670–676. [Google Scholar] [CrossRef]
- Vieth, R.; Ladak, Y.; Walfish, P.G. Age-related changes in the 25-hydroxyvitamin D versus parathyroid hormone relationship suggest a different reason why older adults require more vitamin D. J. Clin. Endocr. Metab. 2003, 88, 185–191. [Google Scholar] [CrossRef]
- Lips, P.; Duong, T.; Oleksik, A.; Black, D.; Cummings, S.; Cox, D.; Nickelsen, T.; Ste-Marie, L.G.; Evalu, M.O.R. A global study of vitamin D status and parathyroid function in postmenopausal women with osteoporosis: Baseline data from the multiple outcomes of raloxifene evaluation clinical trial. J. Clin. Endocr. Metab. 2001, 86, 1212–1221. [Google Scholar] [CrossRef]
- Saraiva, G.L.; Cendoroglo, M.S.; Ramos, L.R.; Araujo, L.M.; Vieira, J.G.; Maeda, S.S.; Borba, V.Z.; Kunii, I.; Hayashi, L.F.; Lazaretti-Castro, M. Prevalence of vitamin D deficiency, insufficiency and secondary hyperparathyroidism in the elderly inpatients and living in the community of the city of Sao Paulo, Brazil. Arq. Bras. Endocrinol. Metabol. 2007, 51, 437–442. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Moreno-Reyes, R.; Carpentier, Y.A.; Boelaert, M.; El Moumni, K.; Dufourny, G.; Bazelmans, C.; Leveque, A.; Gervy, C.; Goldman, S. Vitamin D deficiency and hyperparathyroidism in relation to ethnicity: A cross-sectional survey in healthy adults. Eur. J. Nutr. 2009, 48, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Melin, A.L.; Wilske, J.; Ringertz, H.; Saaf, M. Vitamin D status, parathyroid function and femoral bone density in an elderly swedish population living at home. Aging Clin. Exp. Res. 1999, 11, 200–207. [Google Scholar] [CrossRef]
- Russo, L.A.; Gregorio, L.H.; Lacativa, P.G.; Marinheiro, L.P. Concentration of 25-hydroxyvitamin D in postmenopausal women with low bone mineral density. Arq. Bras. Endocrinol. Metabol. 2009, 53, 1079–1087. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.C.; Chimeh, F.; Lu, Z.R.; Mathieu, J.; Person, K.S.; Zhang, A.Q.; Kohn, N.; Martinello, S.; Berkowitz, R.; Holick, M.F. Factors that influence the cutaneous synthesis and dietary sources of vitamin D. Arch. Biochem. Biophys. 2007, 460, 213–217. [Google Scholar] [CrossRef]
- Marfell-Jones, M.J.; Stewart, A.D.; De Ridder, J.H. International Standards for Anthropometric Assessment; International Society for the Advancement of Kinanthropometry: Lower Hutt, New Zealand, 2012. [Google Scholar]
- Burt, L.A.; Macdonald, H.M.; Hanley, D.A.; Boyd, S.K. Bone microarchitecture and strength of the radius and tibia in a reference population of young adults: An hr-pqct study. Arch. Osteoporos. 2014, 9, 183. [Google Scholar] [CrossRef]
- Looker, A.C.; Wahner, H.W.; Dunn, W.L.; Calvo, M.S.; Harris, T.B.; Heyse, S.P.; Johnston, C.C., Jr.; Lindsay, R. Updated data on proximal femur bone mineral levels of us adults. Osteoporos. Int. 1998, 8, 468–489. [Google Scholar] [CrossRef]
- Scientific Advisory Committee on Nutrition (SCAN). Vitamin D and Health; Scientific Advisory Committee on Nutrition: London, UK, 2016.
- Ross, A.C.; Manson, J.E.; Abrams, S.A.; Aloia, J.F.; Brannon, P.M.; Clinton, S.K.; Durazo-Arvizu, R.A.; Gallagher, J.C.; Gallo, R.L.; Jones, G.; et al. The 2011 report on dietary reference intakes for calcium and vitamin D from the institute of medicine: What clinicians need to know. J. Clin. Endocrinol. Metab. 2011, 96, 53–58. [Google Scholar] [CrossRef]
- Public Health England. Government recommendations for energy and nutrients for males and females aged 1–18 years and 19+ years. In Government Dietary Recommendations; Public Health England: London, UK, 2016. [Google Scholar]
- Felson, D.T.; Zhang, Y.; Hannan, M.T.; Anderson, J.J. Effects of weight and body mass index on bone mineral density in men and women: The framingham study. J. Bone Miner. Res. 1993, 8, 567–573. [Google Scholar] [CrossRef]
- Johansson, H.; Kanis, J.A.; Oden, A.; McCloskey, E.; Chapurlat, R.D.; Christiansen, C.; Cummings, S.R.; Diez-Perez, A.; Eisman, J.A.; Fujiwara, S.; et al. A meta-analysis of the association of fracture risk and body mass index in women. J. Bone Miner. Res. 2014, 29, 223–233. [Google Scholar] [CrossRef]
- De Laet, C.; Kanis, J.A.; Oden, A.; Johanson, H.; Johnell, O.; Delmas, P.; Eisman, J.A.; Kroger, H.; Fujiwara, S.; Garnero, P.; et al. Body mass index as a predictor of fracture risk: A meta-analysis. Osteoporosis. Int. 2005, 16, 1330–1338. [Google Scholar] [CrossRef] [PubMed]
- Nahas, E.A.P.; Kawakami, M.S.; Nahas-Neto, J.; Buttros, D.D.; Cangussu, L.; Rodrigues, A.B. Assessment of risk factors for low bone mineral density in brazilian postmenopausal women. Climacteric 2011, 14, 220–227. [Google Scholar] [CrossRef]
- Mazocco, L.; Chagas, P. Association between body mass index and osteoporosis in women from northwestern Rio Grande do Sul. Rev. Bras. Reumatol. 2017, 57, 299–305. [Google Scholar] [CrossRef]
- Frazao, P.; Naveira, M. Factors associated with low bone mineral density among white women. Rev. Saude Publica 2007, 41, 740–748. [Google Scholar] [PubMed]
- Barbour, K.E.; Zmuda, J.M.; Strotmeyer, E.S.; Horwitz, M.J.; Boudreau, R.; Evans, R.W.; Ensrud, K.E.; Petit, M.A.; Gordon, C.L.; Cauley, J.A. Correlates of trabecular and cortical volumetric bone mineral density of the radius and tibia in older men: The Osteoporotic Fractures in Men Study. J. Bone Miner. Res. 2010, 25, 1017–1028. [Google Scholar] [CrossRef]
- Sheu, Y.; Cauley, J.A.; Bunker, C.H.; Wheeler, V.W.; Patrick, A.L.; Gordon, C.L.; Kammerer, C.M.; Zmuda, J.M. Correlates of trabecular and cortical volumetric BMD in men of African ancestry. J. Bone Miner. Res. 2009, 24, 960–968. [Google Scholar] [CrossRef]
- Burr, D.B. Remodeling and the repair of fatigue damage. Calcif. Tissue Int. 1993, 53, S75–S80. [Google Scholar] [CrossRef]
- Lorentzon, M.; Landin, K.; Mellström, D.; Ohlsson, C. Leptin is a negative independent predictor of areal BMD and cortical bone size in young adult Swedish men. J. Bone Miner. Res. 2006, 21, 1871–1878. [Google Scholar] [CrossRef]
- Warensjo, E.; Byberg, L.; Melhus, H.; Gedeborg, R.; Mallmin, H.; Wolk, A.; Michaelsson, K. Dietary calcium intake and risk of fracture and osteoporosis: Prospective longitudinal cohort study. BMJ 2011, 342, d1473. [Google Scholar] [CrossRef]
- Kim, K.M.; Choi, S.H.; Lim, S.; Moon, J.H.; Kim, J.H.; Kim, S.W.; Jang, H.C.; Shin, C.S. Interactions between dietary calcium intake and bone mineral density or bone geometry in a low calcium intake population (KNHANES IV 2008–2010). J. Clin. Endocr. Metab. 2014, 99, 2409–2417. [Google Scholar] [CrossRef]
- Bischoff-Ferrari, H.A.; Kiel, D.P.; Dawson-Hughes, B.; Orav, J.E.; Li, R.; Spiegelman, D.; Dietrich, T.; Willett, W.C. Dietary calcium and serum 25-hydroxyvitamin D status in relation to BMD among US adults. J. Bone Miner. Res. 2009, 24, 935–942. [Google Scholar] [CrossRef] [PubMed]
- Mezquita-Raya, P.; Munoz-Torres, M.; Luna, J.D.; Luna, V.; Lopez-Rodriguez, F.; Torres-Vela, E.; Escobar-Jimenez, F. Relation between vitamin D insufficiency, bone density, and bone metabolism in healthy postmenopausal women. J. Bone Miner. Res. 2001, 16, 1408–1415. [Google Scholar] [CrossRef] [PubMed]
- POF. Pesquisa de orçamentos familiares 2008–2009. In Análise do Consumo Alimentar Pessoal no Brasil; Instituto Brasileiro de Geografia e Estatística – IBGE: Rio de Janeiro, Brazil, 2011. [Google Scholar]
- Lips, P. Vitamin D deficiency and secondary hyperparathyroidism in the elderly: Consequences for bone loss and fractures and therapeutic implications. Endocr. Rev. 2001, 22, 477–501. [Google Scholar] [CrossRef] [PubMed]
- Bouillon, R.; Van Schoor, N.M.; Gielen, E.; Boonen, S.; Mathieu, C.; Vanderschueren, D.; Lips, P. Optimal vitamin D status: A critical analysis on the basis of evidence-based medicine. J. Clin. Endocr. Metab. 2013, 98, E1283–E1304. [Google Scholar] [CrossRef] [PubMed]
- Bischof Kota, S.; Jammula, S.; Kota, S.; Meher, L.; Modi, K. Correlation of vitamin D, bone mineral density and parathyroid hormone levels in adults with low bone density. Indian J. Orthop. 2013, 47, 402–407. [Google Scholar] [CrossRef] [PubMed]
- Sahota, O.; Mundey, M.K.; San, P.; Godber, I.M.; Lawson, N.; Hosking, D.J. The relationship between vitamin D and parathyroid hormone: Calcium homeostasis, bone turnover, and bone mineral density in postmenopausal women with established osteoporosis. Bone 2004, 35, 312–319. [Google Scholar] [CrossRef]
- Iuliano-Burns, S.; Ayton, J.; Hillam, S.; Jones, G.; King, K.; Macleod, S.; Seeman, E. Skeletal and hormonal responses to vitamin D supplementation during sunlight deprivation in antarctic expeditioners. Osteoporosis. Int. 2012, 23, 2461–2467. [Google Scholar] [CrossRef]
- Villareal, D.T.; Civitelli, R.; Chines, A.; Avioli, L.V. Subclinical vitamin-d deficiency in postmenopausal women with low vertebral bone mass. J. Clin. Endocr. Metab. 1991, 72, 628–634. [Google Scholar] [CrossRef]
- Bischoff-Ferrari, H.A.; Dietrich, T.; Orav, E.J.; Dawson-Hughes, B. Positive association between 25-hydroxy, vitamin D levels and bone mineral density: A population-based study of younger and older adults. Am. J. Med. 2004, 116, 634–639. [Google Scholar] [CrossRef]
- Smith, L.M.; Gallagher, J.C.; Kaufmann, M.; Jones, G. Effect of increasing doses of vitamin D on bone mineral density and serum n-terminal telopeptide in elderly women: A randomized controlled trial. J. Intern. Med. 2018, 284, 685–693. [Google Scholar] [CrossRef]
- Gannage-Yared, M.H.; Chemali, R.; Yaacoub, N.; Halaby, G. Hypovitaminosis d in a sunny country: Relation to lifestyle and bone markers. J. Bone Miner. Res. 2000, 15, 1856–1862. [Google Scholar] [CrossRef] [PubMed]
- Olmos, J.M.; Hernandez, J.L.; Garcia-Velasco, P.; Martinez, J.; Llorca, J.; Gonzalez-Macias, J. Serum 25-hydroxyvitamin d, parathyroid hormone, calcium intake, and bone mineral density in Spanish adults. Osteoporos. Int. 2016, 27, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.S.; Cohen, A.; Shane, E.; Yin, P.T.; Stein, E.M.; Rogers, H.; Kokolus, S.L.; McMahon, D.J.; Lappe, J.M.; Recker, R.R.; et al. Bone Density, Geometry, Microstructure and Stiffness: Relationships Between Peripheral and Central Skeletal Sites Assessed by DXA, HR-pQCT, and cQCT in Premenopausal Women. J. Bone Miner. Res. 2010, 25, 2229–2238. [Google Scholar] [CrossRef] [PubMed]
| England (n = 51) | Brazil (n = 79) | p Value 2 | |
|---|---|---|---|
| Age (years) | 33 (28, 38) | 27 (24, 31) | <0.001 |
| Height (m) | 1.64 ± 0.05 | 1.62 ± 0.59 | 0.094 |
| Weight (kg) | 70.11 ± 14.22 | 63.21 ± 13.07 | 0.002 |
| Waist Circumference (cm) | 86.3 (77.0, 96.0) | 70.4 (66.1, 77.2) | <0.001 |
| BMI (kg/m2) | 25.94 ± 5.30 | 24.01 ± 4.88 | 0.009 |
| Body fat (%) ≠ | 30.95 ± 6.00 | 38.65 ± 8.52 | <0.001 a |
| Vitamin D intake (μg/day) | 2.59 (1.55, 3.92) | 1.57 (0.73, 2.79) | <0.001 |
| Calcium intake (mg/day) | 711.76 (537.22, 879.51) | 479.97 (347.42, 704.92) | 0.003 |
| Serum 25(OH)D (nmol/L) | 35.22 ± 14.89 | 75.00 ± 22.13 | <0.001 a |
| Plasma PTH (pmol/L) | 5.39 ± 2.07 | 4.49 ± 1.47 | 0.004 a |
| Serum Calcium (mmol/L) 3 | 2.30 ± 0.07 | 2.28 ± 0.06 | 0.066 a |
| Bone Parameter | All | Age | p Value 2 | ||
|---|---|---|---|---|---|
| Tertile 1 | Tertile 2 | Tertile 3 | |||
| (≤29 Years) | (30–36 Years) | (>37 Years) | |||
| Number (n) | 51 | 18 | 16 | 17 | |
| Wegiht (kg) | 70.11 ± 14.22 | 64.70 ± 11.24 | 72.71 ± 15.68 | 73.38 ± 14.69 | 0.132 |
| Height (m) | 1.64 ± 0.05 | 1.63 ± 0.05 | 1.65 ± 0.06 | 1.64 ± 0.04 | 0.518 |
| Serum 25(OH)D (nmol/L) | 35.22 ± 14.89 | 36.78 ± 13.20 | 33.74 ± 15.52 | 34.95 ± 16.64 | 0.84 |
| Plasma PTH (pmol/L) | 5.39 ± 2.07 | 5.31 ± 2.00 | 5.29 ± 2.54 | 5.56 ± 1.74 | 0.92 |
| Serum calcium (mmol/L) | 2.30 ± 0.07 | 2.31 ± 0.08 | 2.28 ± 0.06 | 2.29 ± 0.07 | 0.388 |
| 4% site | |||||
| BMC (g/cm) | 1.07 ± 0.14 | 1.05 ± 0.15 | 1.08 ± 0.13 | 1.08 ± 0.13 | 0.821 |
| vBMD (mg/cm3): | |||||
| Total | 353.20± 51.81 | 380.72 ± 60.82 a | 329.97 ± 37.07 a | 346.12 ± 41.47 | 0.011 |
| Trabecular | 199.29 ± 57.78 | 205.61 ± 73.89 | 189.48 ± 44.14 | 201.85 ± 51.53 | 0.709 |
| 66% site | |||||
| BMC (g/cm) | 0.96 ± 0.14 | 0.93 ± 0.15 | 0.97 ± 0.12 | 0.97 ± 0.14 | 0.557 |
| vBMD (mg/cm3): | |||||
| Total | 714.38 ±86.33 | 753.33 ± 68.57 | 693.10 ± 90.80 | 693.17 ± 89.02 | 0.056 |
| Cortical | 1127.23 ± 36.44 | 1140.09 ± 27.03 | 1124.26 ± 37.61 | 1116.43 ± 41.59 | 0.147 |
| Z-score n (%) | |||||
| Total vBMD (4%) | |||||
| Normal (>–2.0) | 51 (100%) | 18 (100%) | 16 (100%) | 17 (100%) | |
| Low bone mineral (≤–2.0) | 0 | 0 | 0 | 0 | |
| Trabecular vBMD (4%) | |||||
| Normal (>–2.0) | 51 (100%) | 18 (100%) | 16 (100%) | 17 (100%) | |
| Low bone mineral (≤–2.0) | 0 | 0 | 0 | 0 | |
| Total vBMD (66%) | |||||
| Normal (>–2.0) | 48 (94.2%) | 18 (100%) | 13 (94.8%) | 17 (100%) | |
| Low bone mineral (≤–2.0) | 3 (5.8%) | 0 | 3 (18.7%) | 0 | |
| Cortical vBMD (66%) | |||||
| Normal (>−1.0) | 51 (100%) | 18 (100%) | 16 (100%) | 17 (100%) | |
| Low bone mineral (≤−2.0) | 0 | 0 | 0 | 0 | |
| Bone Parameter | All | Age (Years) | p Value 2 | ||
|---|---|---|---|---|---|
| Tertile 1 ≤25 | Tertile 2 26–30 | Tertile 3 ≥31 | |||
| Number (n) | 79 | 33 | 25 | 21 | |
| Weight (kg) | 63.21 ± 13.07 | 60.41 ± 12.58 | 64.30 ± 12.46 | 66.32 ± 14.20 | 0.239 |
| Height (m) | 1.62 ± 0.59 | 1.61 ± 0.05 | 1.64 ± 0.06 | 1.60 ± 0.06 | 0.157 |
| Serum 25(OH)D (nmol/L) | 75.00 ± 22.13 | 78.87 ± 27.23 | 74.68 ± 18.39 | 77.15 ± 17.61 | 0.868 |
| Plasma PTH (pmol/L) | 4.49 ±1.47 | 3.96 ± 1.25 a | 4.79 ± 1.61 | 4.99 ± 1.42 a | 0.021 |
| Serum calcium (mmol/L) | 2.28 ± 0.06 | 2.29 ± 0.07 | 2.29 ± 0.05 | 2.25 ± 0.07 | 0.26 |
| Lumbar Spine | |||||
| Number (n) | 79 | 33 | 25 | 21 | 0.537 |
| BMD (g/cm2) | 1.13 ± 0.11 | 1.12 ± 0.12 | 1.13 ± 0.11 | 1.15 ± 0.11 | |
| Femur | |||||
| Number (n) | 64 | 27 | 20 | 17 | 0.495 |
| BMD (g/cm2) | 0.98 ± 0.10 | 0.97 ± 0.10 | 0.97 ± 0.12 | 1.01 ± 0.11 | |
| Total BMC | |||||
| Number (n) | 79 | 33 | 25 | 21 | 0.204 |
| BMC (g) | 2304.87 ± 401.76 | 2304.87 ± 401.76 | 2459.92 ± 377.29 | 2460.95 ± 338.42 | |
| Z-score n (%) | |||||
| Lumbar Spine BMD | |||||
| Normal (>−2.0) | 79 (97.5%) | 31 (94%) | 25 (100%) | 21 (100%) | |
| Low bone mineral (≤−2.0) | 2 (2.5%) | 2 (6%) | 0 | 0 | |
| Z-score n (%) | |||||
| Femur BMD | |||||
| Normal (>−2.0) | 64 (100%) | 27 (100%) | 20 (100%) | 17 (100%) | |
| Low bone mineral (≤−2.0) | 0 | 0 | 0 | 0 | |
| Bone Parameter | Age (Years) | Weight (kg) | BMI (kg/m2) | Body Fat (%) | ||||
|---|---|---|---|---|---|---|---|---|
| r Value | p Value | r Value | p Value | r Value | p Value | r Value | p Value | |
| 4% site | ||||||||
| BMC (g/cm) | 0.186 | 0.191 | 0.323 | 0.021 | 0.317 | 0.024 | 0.118 | 0.386 |
| vBMD (mg/cm3): | ||||||||
| Total | −0.143 | 0.316 | −0.005 | 0.97 | −0.028 | 0.847 | 0.034 | 0.814 |
| Trabecular | −0.007 | 0.962 | −0.118 | 0.41 | −0.112 | 0.435 | 0.085 | 0.551 |
| 66% site | ||||||||
| BMC (g/cm) | 0.216 | 0.128 | 0.256 | 0.07 | 0.253 | 0.074 | −0.063 | 0.345 |
| vBMD (mg/cm3): | ||||||||
| Total | −0.213 | 0.134 | −0.335 | 0.016 | −0.330 | 0.018 | −0.159 | 0.266 |
| Cortical | −0.223 | 0.116 | −0.313 | 0.025 | −0.309 | 0.027 | −0.122 | 0.393 |
| Bone Parameter | Age (Years) | Weight (kg) | BMI (kg/m2) | Body Fat (%) | ||||
|---|---|---|---|---|---|---|---|---|
| r Value | p Value | r Value | p Value | r Value | p Value | r Value | p Value | |
| Lumbar Spine BMD (mg/cm2) | 0.120 | 0.292 | 0.337 | 0.002 | 0.298 | 0.008 | 0.252 | 0.025 |
| Femur BMD (mg/cm2) | 0.273 | 0.029 | 0.583 | <0.001 | 0.612 | <0.001 | 0.378 | 0.002 |
| Total BMC (g) | 0.267 | 0.017 | 0.606 | <0.001 | 0.585 | <0.001 | 0.412 | <0.001 |
| Bone Parameter | All | 25(OH)D in nmol/L | p Value 2 | ||
|---|---|---|---|---|---|
| <25 | 25–49.99 | 50–74.99 | |||
| Number (n) | 51 | 14 | 28 | 9 | |
| Weight (kg) | 70.11 ± 14.22 | 76.01 ± 17.72 | 66.44 ± 12.6 | 72.33 ± 10.20 | 0.104 |
| Height (m) | 1.64 ± 0.05 | 1.63 ± 0.04 | 1.64 ± 0.05 | 1.67 ± 0.05 | 0.340 |
| BMI (kg/m2) | 25.94 ± 5.30 | 28.28 ± 6.13 | 24.73 ± 4.84 | 26.09 ± 4.54 | 0.123 |
| Serum 25(OH)D (nmol/L) | 35.22 ± 14.89 | 20.45 ± 5.33 | 34.29 ± 5.69 | 61.07 ± 9.74 | >0.001 |
| Plasma PTH (pmol/L) | 5.39 ± 2.07 | 6.60 ± 2.47 | 5.00 ± 1.92 | 4.69 ± 0.92 | 0.030 |
| Serum calcium (mmol/L) | 2.30 ± 0.07 | 2.28 ± 0.05 | 2.29 ± 0.08 | 2.31 ± 0.05 | 0.634 |
| 4% site | |||||
| BMC (g/cm) | 1.07 ± 0.14 | 1.06 ± 0.12 | 1.07 ± 0.15 | 1.10 ± 0.13 | 0.782 |
| vBMD (mg/cm3): | |||||
| Total | 352.23 ± 51.81 | 355.81 ± 40.47 | 356.86 ± 57.76 | 337.95 ± 50.35 | 0.630 |
| Trabecular | 199.29 ± 57.78 | 189.56 ± 25.61 | 208.46 ± 63.39 | 185.91 ± 57.87 | 0.462 |
| 66% site | |||||
| BMC (g/cm) | 0.96 ± 0.14 | 0.97 ± 0.13 | 0.96 ± 0.16 | 0.92 ± 0.05 | 0.642 |
| vBMD (mg/cm3): | |||||
| Total | 714.39 ± 86.33 | 658.54 ± 104.23 a,b | 732.91 ± 73.85 a | 743.62 ± 53.80 b | 0.014 |
| Cortical | 1127.23 ± 36.44 | 1107.74 ± 44.43 c | 1136.66 ± 31.41 c | 1128.24 ± 28.01 | 0.049 |
| Bone Parameter | All | 25(OH)D in nmol/L | p Value 2 | ||
|---|---|---|---|---|---|
| 25–49.99 | 50–74.99 | >75 | |||
| Weight (kg) | 63.21 ± 13.07 | 57.49 ± 10.73 | 62.51 ± 12.24 | 65.03 ± 13.97 | 0.278 |
| Height (m) | 1.62 ± 0.59 | 1.60 ± 0.06 | 1.62 ± 0.06 | 1.62 ± 0.05 | 0.625 |
| BMI (kg/m2) | 24.01 ± 4.88 | 22.4 ± 3.09 | 23.65 ± 4.71 | 24.64 ± 5.29 | 0.409 |
| Serum 25(OH)D (nmol/L) | 75.00 ± 22.13 | 42.77 ± 4.12 | 61.86 ± 7.05 | 92.11 ± 16.31 | >0.001 |
| Plasma PTH (pmol/L) | 4.49 ± 1.47 | 4.73 ± 1.23 | 4.50 ± 1.53 | 4.42 ± 1.51 | 0.86 |
| Serum calcium (mmol/L) | 2.28 ± 0.06 | 2.28 ± 0.06 | 2.28 ± 0.07 | 2.28 ± 0.06 | 0.993 |
| Lumbar Spine | |||||
| Number (n) | 79 | 9 | 30 | 40 | 0.716 |
| BMD (mg/cm2) | 1.13 ± 0.11 | 1.11 ± 0.11 | 1.13 ± 0.12 | 1.14 ± 0.10 | |
| Femur | |||||
| Number (n) | 64 | 8 | 25 | 31 | 0.728 |
| BMD (mg/cm2) | 0.98 ± 0.10 | 0.94 ± 0.10 | 0.97 ± 0.10 | 1.00 ± 0.11 | |
| Total | |||||
| Number (n) | 79 | 9 | 30 | 40 | 0.335 |
| BMC (g) | 2395.43 ± 381.24 | 2305.66 ± 372.97 | 2391.46 ± 448.47 | 2418.60 ± 331.93 | |
| Bone Parameter | PTH | |
|---|---|---|
| r Value | p Value | |
| 4% site | ||
| BMC (g/cm) | 0.348 | 0.012 |
| Total vBMD (mg/cm3) | 0.108 | 0.45 |
| Trabecular vBMD (mg/cm3) | 0.085 | 0.553 |
| 66% site | ||
| BMC (g/cm) | 0.435 | 0.001 |
| Total vBMD (mg/cm3) | −0.312 | 0.026 |
| Cortical vBMD (mg/cm3) | −0.184 | 0.197 |
| Bone Parameter | PTH | |
|---|---|---|
| r Value | p Value | |
| Lumbar Spine BMD (mg/cm2) (n = 78) | 0.004 | 0.976 |
| Femur BMD (mg/cm2) (n = 63) | 0.215 | 0.900 |
| Total BMC (g) (n = 78) | 0.114 | 0.319 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mendes, M.M.; Hart, K.H.; Lanham-New, S.A.; Botelho, P.B. Association between 25-Hydroxyvitamin D, Parathyroid Hormone, Vitamin D and Calcium Intake, and Bone Density in Healthy Adult Women: A Cross-Sectional Analysis from the D-SOL Study. Nutrients 2019, 11, 1267. https://doi.org/10.3390/nu11061267
Mendes MM, Hart KH, Lanham-New SA, Botelho PB. Association between 25-Hydroxyvitamin D, Parathyroid Hormone, Vitamin D and Calcium Intake, and Bone Density in Healthy Adult Women: A Cross-Sectional Analysis from the D-SOL Study. Nutrients. 2019; 11(6):1267. https://doi.org/10.3390/nu11061267
Chicago/Turabian StyleMendes, Marcela M., Kathryn H. Hart, Susan A. Lanham-New, and Patrícia B. Botelho. 2019. "Association between 25-Hydroxyvitamin D, Parathyroid Hormone, Vitamin D and Calcium Intake, and Bone Density in Healthy Adult Women: A Cross-Sectional Analysis from the D-SOL Study" Nutrients 11, no. 6: 1267. https://doi.org/10.3390/nu11061267
APA StyleMendes, M. M., Hart, K. H., Lanham-New, S. A., & Botelho, P. B. (2019). Association between 25-Hydroxyvitamin D, Parathyroid Hormone, Vitamin D and Calcium Intake, and Bone Density in Healthy Adult Women: A Cross-Sectional Analysis from the D-SOL Study. Nutrients, 11(6), 1267. https://doi.org/10.3390/nu11061267







