The Relationship between Whole Grain Intake and Body Weight: Results of Meta-Analyses of Observational Studies and Randomized Controlled Trials
Abstract
1. Introduction
2. Methods
2.1. Literature Searches
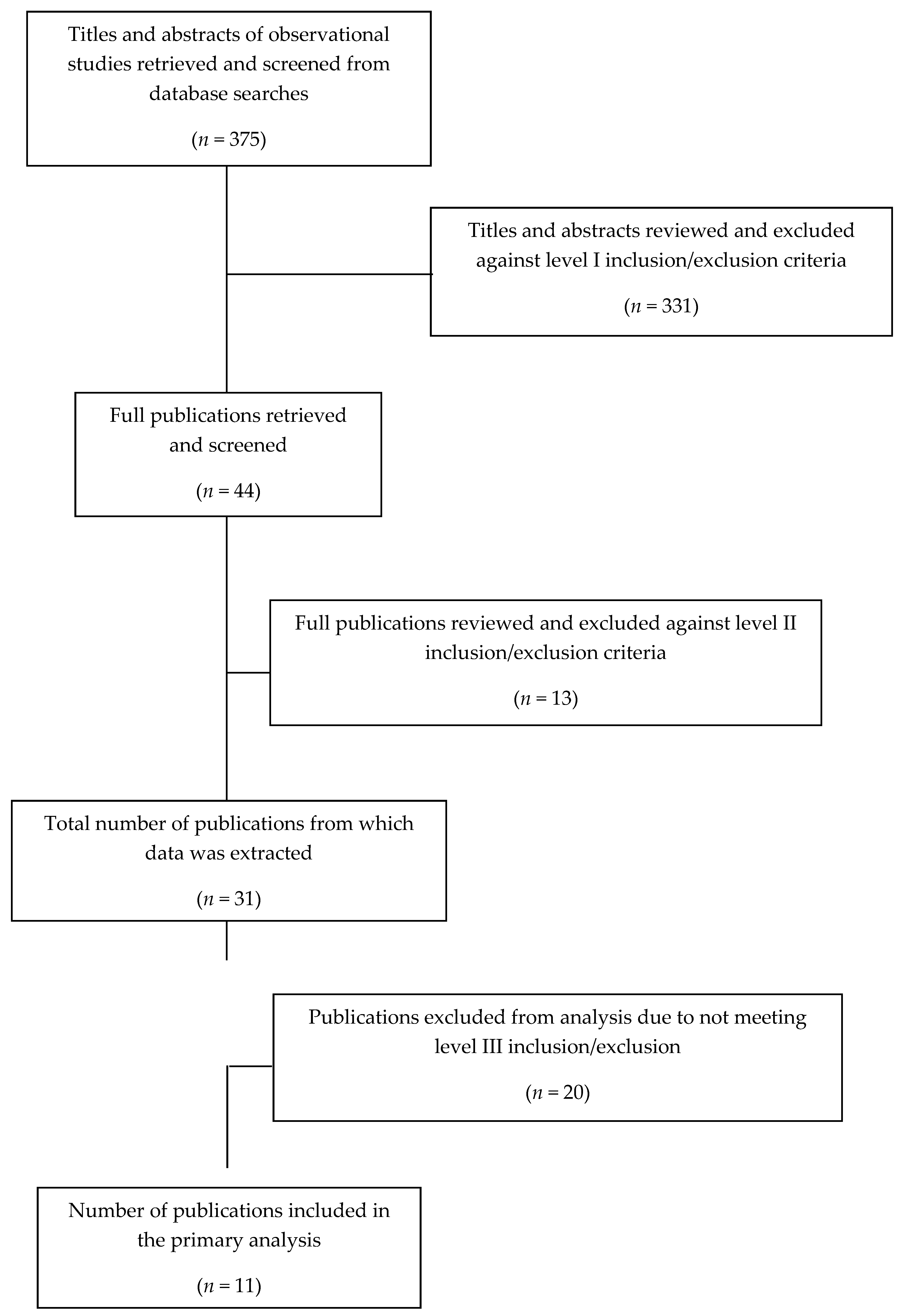
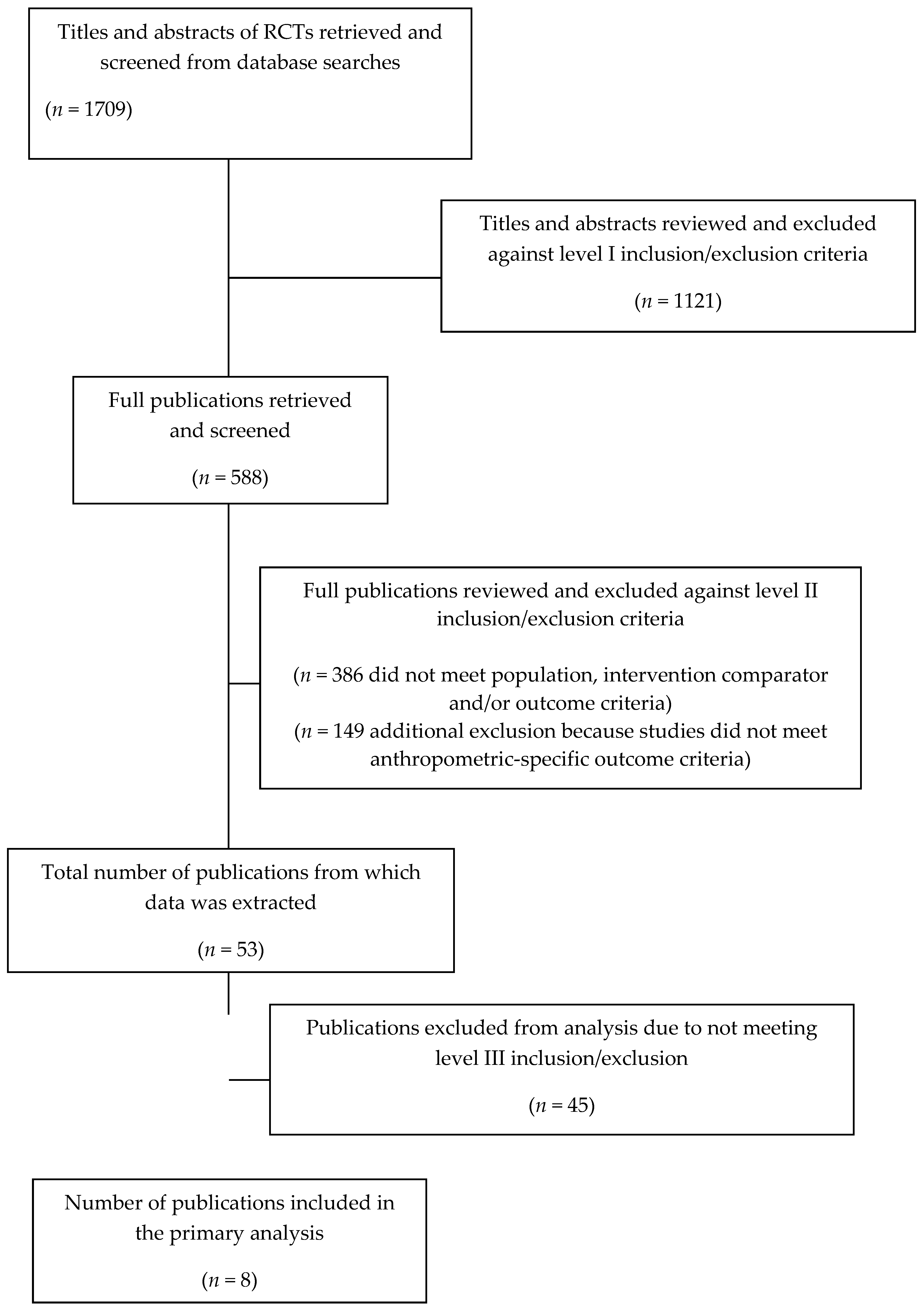
2.2. Inclusion and Exclusion Criteria Screening
2.3. Meta-Regression Analysis of WG Intake: Cross-Sectional Studies
2.4. Meta-Regression Analysis of WG Intake: RCTs
2.5. Statistical Analyses
3. Results
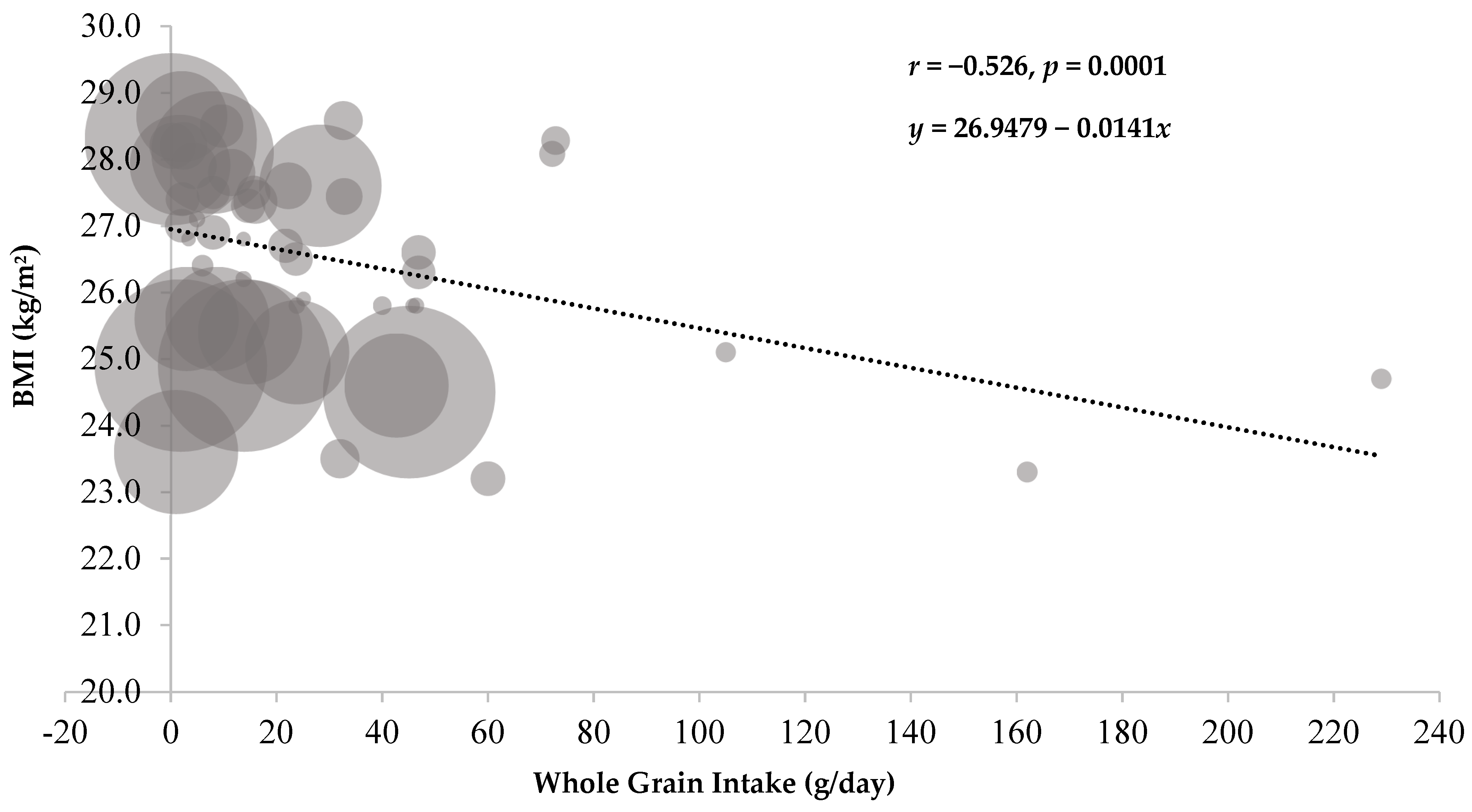
3.1. Meta-Regression Analysis: Cross-Sectional Studies
3.2. Qualitative Analysis: Prospective Cohort Studies
3.3. Meta-Analysis: RCTs
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BMI | body mass index |
| CI | confidence interval |
| HPFS | Health Professionals Follow-up Study |
| MD | mean difference |
| NHS | Nurses’ Health Study |
| NSHD | Northern Sweden Health and Disease Study |
| PHS | Physicians’ Health Study |
| RCT | randomized controlled trial |
| RG | refined grain |
| SD | standard deviation |
| SE | standard error |
| SEM | standard error of the mean |
| SMD | standardized mean difference |
| SUN | Seguimiento Universidad de Navarra |
| WG | whole grain |
References
- U.S. Department of Health and Human Services and U.S. Department of Agriculture. 2015–2020 Dietary Guidelines for Americans. Available online: http://health.gov/dietaryguidelines/2015/guidelines/ (accessed on 14 November 2018).
- Van der Kamp, J.W.; Poutanen, K.; Seal, C.J.; Richardson, D.P. The HEALTHGRAIN definition of “whole grain”. Food Nutr. Res. 2014, 58. [Google Scholar] [CrossRef]
- De la Fuente-Arrillaga, C.; Martinez-Gonzalez, M.A.; Zazpe, I.; Vazquez-Ruiz, Z.; Benito-Corchon, S.; Bes-Rastrollo, M. Glycemic load, glycemic index, bread and incidence of overweight/obesity in a Mediterranean cohort: The SUN project. BMC Public Health 2014, 14, 1091. [Google Scholar] [CrossRef] [PubMed]
- Reicks, M.; Jonnalagadda, S.; Albertson, A.M.; Joshi, N. Total dietary fiber intakes in the US population are related to whole grain consumption: Results from the National Health and Nutrition Examination Survey 2009 to 2010. Nutr. Res. 2014, 34, 226–234. [Google Scholar] [CrossRef]
- Mozaffarian, D. Dietary and Policy Priorities for Cardiovascular Disease, Diabetes, and Obesity: A Comprehensive Review. Circulation 2016, 133, 187–225. [Google Scholar] [CrossRef]
- Giacco, R.; Della Pepa, G.; Luongo, D.; Riccardi, G. Whole grain intake in relation to body weight: From epidemiological evidence to clinical trials. Nutr. Metab. Cardiovasc. Dis. 2011, 21, 901–908. [Google Scholar] [CrossRef]
- Karl, J.P.; Saltzman, E. The role of whole grains in body weight regulation. Adv. Nutr. 2012, 3, 697–707. [Google Scholar] [CrossRef] [PubMed]
- Thielecke, F.; Jonnalagadda, S.S. Can whole grain help in weight management? J. Clin. Gastroenterol. 2014, 48, S70–S77. [Google Scholar] [CrossRef]
- Pol, K.; Christensen, R.; Bartels, E.M.; Raben, A.; Tetens, I.; Kristensen, M. Whole grain and body weight changes in apparently healthy adults: A systematic review and meta-analysis of randomized controlled studies. Am. J. Clin. Nutr. 2013, 98, 872–884. [Google Scholar] [CrossRef] [PubMed]
- US Department of Agriculture (USDA), Agricultural Research Service, Nutrient Data Laboratory. USDA National Nutrient Database for Standard Reference, Legacy. Available online: http://www.ars.usda.gov/nutrientdata (accessed on 24 September 2018).
- Kranz, S.; Dodd, K.W.; Juan, W.Y.; Johnson, L.K.; Jahns, L. Whole Grains Contribute Only a Small Proportion of Dietary Fiber to the U.S. Diet. Nutrients 2017, 9, 153. [Google Scholar] [CrossRef]
- Center for Nutrition Policy and Promotion; U.S. Department of Agriculture Website. Nutrient Content of the U.S. Food Supply, 1909–2010. Fiber Content from Major Food Groups. Calculated by USDA/Center for Nutrition Policy and Promotion. Available online: https://www.cnpp.usda.gov/USFoodSupply-1909–2010 (accessed on 5 December 2018).
- Office of Nutrition and Food Labeling. The Declaration of Certain Isolated or Synthetic Non-Digestible Carbohydrates as Dietary Fiber on Nutrition and Supplement Facts Labels: Guidance for Industry; Food and Drug Administration: College Park, MD, USA, 2018.
- Office of Nutrition and Food Labeling. Review of the Scientific Evidence on the Physiological Effects of Certain Non-Digestible Carbohydrates; Food and Drug Administration: College Park, MD, USA, 2018.
- Jovanovski, E.; Khayyat, R.; Zurbau, A.; Komishon, A.; Mazhar, N.; Sievenpiper, J.L.; Blanco Mejia, S.; Ho, H.V.T.; Li, D.; Jenkins, A.L.; et al. Should Viscous Fiber Supplements Be Considered in Diabetes Control? Results from a Systematic Review and Meta-analysis of Randomized Controlled Trials. Diabetes Care 2019. [Google Scholar] [CrossRef] [PubMed]
- Stewart, M.L.; Wilcox, M.L.; Bell, M.; Buggia, M.A.; Maki, K.C. Type-4 Resistant Starch in Substitution for Available Carbohydrate Reduces Postprandial Glycemic Response and Hunger in Acute, Randomized, Double-Blind, Controlled Study. Nutrients 2018, 10, 129. [Google Scholar] [CrossRef]
- Rebello, C.J.; O’Neil, C.E.; Greenway, F.L. Dietary fiber and satiety: The effects of oats on satiety. Nutr. Rev. 2016, 74, 131–147. [Google Scholar] [CrossRef]
- Hu, Y.; Song, Y.; Franke, A.A.; Hu, F.B.; van Dam, R.M.; Sun, Q. A Prospective Investigation of the Association Between Urinary Excretion of Dietary Lignan Metabolites and Weight Change in US Women. Am. J. Epidemiol. 2015, 182, 503–511. [Google Scholar] [CrossRef]
- Xu, C.; Liu, Q.; Zhang, Q.; Gu, A.; Jiang, Z.Y. Urinary enterolactone is associated with obesity and metabolic alteration in men in the US National Health and Nutrition Examination Survey 2001–10. Br. J. Nutr. 2015, 113, 683–690. [Google Scholar] [CrossRef]
- Rietjens, I.M.C.M.; Louisse, J.; Beekmann, K. The potential health effects of dietary phytoestrogens. Br. J. Pharmacol. 2017, 174, 1263–1280. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Sawicki, C.M.; Livingston, K.A.; Ross, A.B.; Jacques, P.F.; Koecher, K.; McKeown, N.M. Evaluating Whole Grain Intervention Study Designs and Reporting Practices Using Evidence Mapping Methodology. Nutrients 2018, 10, 1052. [Google Scholar] [CrossRef]
- Albertson, A.M.; Reicks, M.; Joshi, N.; Gugger, C.K. Whole grain consumption trends and associations with body weight measures in the United States: Results from the cross sectional National Health and Nutrition Examination Survey 2001–2012. Nutr. J. 2016, 15, 8. [Google Scholar] [CrossRef]
- O’Neil, C.E.; Zanovec, M.; Cho, S.S.; Nicklas, T.A. Whole grain and fiber consumption are associated with lower body weight measures in US adults: National Health and Nutrition Examination Survey 1999–2004. Nutr. Res. 2010, 30, 815–822. [Google Scholar] [CrossRef]
- Higgins, J.P.; Altman, D.G.; Gotzsche, P.C.; Juni, P.; Moher, D.; Oxman, A.D.; Savovic, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef]
- Lichtenstein, A.H.; Yetley, E.A.; Lau, J. Application of systematic review methodology to the field of nutrition. J. Nutr. 2008, 138, 2297–2306. [Google Scholar] [CrossRef]
- Der Simonian, R.; Laird, N. Meta-analysis in clinical trials. Control Clin. Trials 1986, 7, 177–188. [Google Scholar] [CrossRef]
- Heyland, D.K.; Novak, F.; Drover, J.W.; Jain, M.; Su, X.; Suchner, U. Should immunonutrition become routine in critically ill patients? A systematic review of the evidence. JAMA 2001, 286, 944–953. [Google Scholar] [CrossRef]
- Thompson, S.V.; Hannon, B.A.; An, R.; Holscher, H.D. Effects of isolated soluble fiber supplementation on body weight, glycemia, and insulinemia in adults with overweight and obesity: A systematic review and meta-analysis of randomized controlled trials. Am. J. Clin. Nutr. 2017, 106, 1514–1528. [Google Scholar] [CrossRef]
- Bax, L. MIX 2.0—Professional software for meta-analysis in Excel. Version 2.015. BiostatXL. Available online: https://www.meta-analysis-made-easy.com (accessed on 5 December 2018).
- Balk, E.M.; Earley, A.; Patel, K.; Trikalinos, T.A.; Dahabreh, I.J. Empirical Assessment of Within-Arm Correlation Imputation in Trials of Continuous Outcomes; Methods Research Report Prepared by the Tufts Evidence-based Practice Center under Contract No. 290-2007-10055-I, AHRQ Publication No. 12(13)-EHC141-EF; Agency for Healthcare Research and Quality: Rockville, MD, USA, 2012.
- Higgins, J.P.; Thompson, S.G. Quantifying heterogeneity in a meta-analysis. Stat. Med. 2002, 21, 1539–1558. [Google Scholar] [CrossRef]
- Sahyoun, N.R.; Jacques, P.F.; Zhang, X.L.; Juan, W.; McKeown, N.M. Whole-grain intake is inversely associated with the metabolic syndrome and mortality in older adults. Am. J. Clin. Nutr. 2006, 83, 124–131. [Google Scholar] [CrossRef]
- McKeown, N.M.; Meigs, J.B.; Liu, S.; Wilson, P.W.; Jacques, P.F. Whole-grain intake is favorably associated with metabolic risk factors for type 2 diabetes and cardiovascular disease in the Framingham Offspring Study. Am. J. Clin. Nutr. 2002, 76, 390–398. [Google Scholar] [CrossRef]
- McKeown, N.M.; Troy, L.M.; Jacques, P.F.; Hoffmann, U.; O’Donnell, C.J.; Fox, C.S. Whole- and refined-grain intakes are differentially associated with abdominal visceral and subcutaneous adiposity in healthy adults: The Framingham Heart Study. Am. J. Clin. Nutr. 2010, 92, 1165–1171. [Google Scholar] [CrossRef]
- McKeown, N.M.; Yoshida, M.; Shea, M.K.; Jacques, P.F.; Lichtenstein, A.H.; Rogers, G.; Booth, S.L.; Saltzman, E. Whole-grain intake and cereal fiber are associated with lower abdominal adiposity in older adults. J. Nutr. 2009, 139, 1950–1955. [Google Scholar] [CrossRef]
- Liu, S.; Willett, W.C.; Manson, J.E.; Hu, F.B.; Rosner, B.; Colditz, G. Relation between changes in intakes of dietary fiber and grain products and changes in weight and development of obesity among middle-aged women. Am. J. Clin. Nutr. 2003, 78, 920–927. [Google Scholar] [CrossRef]
- Lutsey, P.L.; Jacobs, D.R., Jr.; Kori, S.; Mayer-Davis, E.; Shea, S.; Steffen, L.M.; Szklo, M.; Tracy, R. Whole grain intake and its cross-sectional association with obesity, insulin resistance, inflammation, diabetes and subclinical CVD: The MESA Study. Br. J. Nutr. 2007, 98, 397–405. [Google Scholar] [CrossRef]
- Esmaillzadeh, A.; Mirmiran, P.; Azizi, F. Whole-grain consumption and the metabolic syndrome: A favorable association in Tehranian adults. Eur. J. Clin. Nutr. 2005, 59, 353–362. [Google Scholar] [CrossRef]
- Koh-Banerjee, P.; Franz, M.; Sampson, L.; Liu, S.; Jacobs, J.D.R.; Spiegelman, D.; Willett, W.; Rimm, E. Changes in whole-grain, bran, and cereal fiber consumption in relation to 8-y weight gain among men. Am. J. Clin. Nutr. 2004, 80, 1237–1245. [Google Scholar] [CrossRef]
- Bazzano, L.A.; Song, Y.; Bubes, V.; Good, C.K.; Manson, J.E.; Liu, S. Dietary intake of whole and refined grain breakfast cereals and weight gain in men. Obes. Res. 2005, 13, 1952–1960. [Google Scholar] [CrossRef]
- Winkvist, A.; Klingberg, S.; Nilsson, L.M.; Wennberg, M.; Renstrom, F.; Hallmans, G.; Boman, K.; Johansson, I. Longitudinal 10-year changes in dietary intake and associations with cardio-metabolic risk factors in the Northern Sweden Health and Disease Study. Nutr. J. 2017, 16, 20. [Google Scholar] [CrossRef]
- Mozaffarian, D.; Hao, T.; Rimm, E.B.; Willett, W.C.; Hu, F.B. Changes in diet and lifestyle and long-term weight gain in women and men. N. Engl. J. Med. 2011, 364, 2392–2404. [Google Scholar] [CrossRef]
- Melanson, K.J.; Angelopoulos, T.J.; Nguyen, V.T.; Martini, M.; Zukley, L.; Lowndes, J.; Dube, T.J.; Fiutem, J.J.; Yount, B.W.; Rippe, J.M. Consumption of whole-grain cereals during weight loss: Effects on dietary quality, dietary fiber, magnesium, vitamin B-6, and obesity. J. Am. Diet. Assoc. 2006, 106, 1380–1388. [Google Scholar] [CrossRef]
- Katcher, H.I.; Legro, R.S.; Kunselman, A.R.; Gillies, P.J.; Demers, L.M.; Bagshaw, D.M.; Kris-Etherton, P.M. The effects of a whole grain-enriched hypocaloric diet on cardiovascular disease risk factors in men and women with metabolic syndrome. Am. J. Clin. Nutr. 2008, 87, 79–90. [Google Scholar] [CrossRef]
- Maki, K.C.; Beiseigel, J.M.; Jonnalagadda, S.S.; Gugger, C.K.; Reeves, M.S.; Farmer, M.V.; Kaden, V.N.; Rains, T.M. Whole-grain ready-to-eat oat cereal, as part of a dietary program for weight loss, reduces low-density lipoprotein cholesterol in adults with overweight and obesity more than a dietary program including low-fiber control foods. J. Am. Diet. Assoc. 2010, 110, 205–214. [Google Scholar] [CrossRef]
- Kristensen, M.; Pelletier, X.; Ross, A.B.; Thielecke, F. A High Rate of Non-Compliance Confounds the Study of Whole Grains and Weight Maintenance in a Randomised Intervention Trial-The Case for Greater Use of Dietary Biomarkers in Nutrition Intervention Studies. Nutrients 2017, 9, 55. [Google Scholar] [CrossRef]
- Kristensen, M.; Toubro, S.; Jensen, M.G.; Ross, A.B.; Riboldi, G.; Petronio, M.; Bugel, S.; Tetens, I.; Astrup, A. Whole grain compared with refined wheat decreases the percentage of body fat following a 12-week, energy-restricted dietary intervention in postmenopausal women. J. Nutr. 2012, 142, 710–716. [Google Scholar] [CrossRef]
- Chang, H.C.; Huang, C.N.; Yeh, D.M.; Wang, S.J.; Peng, C.H.; Wang, C.J. Oat prevents obesity and abdominal fat distribution, and improves liver function in humans. Plant Foods Hum. Nutr. 2013, 68, 18–23. [Google Scholar] [CrossRef]
- Harris Jackson, K.; West, S.G.; Vanden Heuvel, J.P.; Jonnalagadda, S.S.; Ross, A.B.; Hill, A.M.; Grieger, J.A.; Lemieux, S.K.; Kris-Etherton, P.M. Effects of whole and refined grains in a weight-loss diet on markers of metabolic syndrome in individuals with increased waist circumference: A randomized controlled-feeding trial. Am. J. Clin. Nutr. 2014, 100, 577–586. [Google Scholar] [CrossRef]
- Brownlee, I.A.; Moore, C.; Chatfield, M.; Richardson, D.P.; Ashby, P.; Kuznesof, S.A.; Jebb, S.A.; Seal, C.J. Markers of cardiovascular risk are not changed by increased whole-grain intake: The WHOLEheart study, a randomised, controlled dietary intervention. Br. J. Nutr. 2010, 104, 125–134. [Google Scholar] [CrossRef]
- Schlesinger, S.; Neuenschwander, M.; Schwedhelm, C.; Hoffmann, G.; Bechthold, A.; Boeing, H.; Schwingshackl, L. Food Groups and Risk of Overweight, Obesity, and Weight Gain: A Systematic Review and Dose-Response Meta-Analysis of Prospective Studies. Adv. Nutr. 2019, 10, 205–218. [Google Scholar] [CrossRef]
- Bautista-Castano, I.; Sanchez-Villegas, A.; Estruch, R.; Martinez-Gonzalez, M.A.; Corella, D.; Salas-Salvado, J.; Covas, M.I.; Schroder, H.; Alvarez-Perez, J.; Quilez, J.; et al. Changes in bread consumption and 4-year changes in adiposity in Spanish subjects at high cardiovascular risk. Br. J. Nutr. 2013, 110, 337–346. [Google Scholar] [CrossRef]
- Reynolds, A.; Mann, J.; Cummings, J.; Winter, N.; Mete, E.; Te Morenga, L. Carbohydrate quality and human health: A series of systematic reviews and meta-analyses. Lancet 2019, 393, 434–445. [Google Scholar] [CrossRef]
- Ampatzoglou, A.; Atwal, K.K.; Maidens, C.M.; Williams, C.L.; Ross, A.B.; Thielecke, F.; Jonnalagadda, S.S.; Kennedy, O.B.; Yaqoob, P. Increased whole grain consumption does not affect blood biochemistry, body composition, or gut microbiology in healthy, low-habitual whole grain consumers. J. Nutr. 2015, 145, 215–221. [Google Scholar] [CrossRef]
- Lappi, J.; Mykkanen, H.; Bach Knudsen, K.E.; Kirjavainen, P.; Katina, K.; Pihlajamaki, J.; Poutanen, K.; Kolehmainen, M. Postprandial glucose metabolism and SCFA after consuming wholegrain rye bread and wheat bread enriched with bioprocessed rye bran in individuals with mild gastrointestinal symptoms. Nutr. J. 2014, 13, 104. [Google Scholar] [CrossRef]
- Leinonen, K.; Liukkonen, K.; Poutanen, K.; Uusitupa, M.; Mykkanen, H. Rye bread decreases postprandial insulin response but does not alter glucose response in healthy Finnish subjects. Eur. J. Clin. Nutr. 1999, 53, 262–267. [Google Scholar] [CrossRef]
- Tucker, A.J.; Mackay, K.A.; Robinson, L.E.; Graham, T.E.; Bakovic, M.; Duncan, A.M. The effect of whole grain wheat sourdough bread consumption on serum lipids in healthy normoglycemic/normoinsulinemic and hyperglycemic/hyperinsulinemic adults depends on presence of the APOE E3/E3 genotype: A randomized controlled trial. Nutr. Metab. 2010, 7, 37. [Google Scholar] [CrossRef]
- Giacco, R.; Lappi, J.; Costabile, G.; Kolehmainen, M.; Schwab, U.; Landberg, R.; Uusitupa, M.; Poutanen, K.; Pacini, G.; Rivellese, A.A.; et al. Effects of rye and whole wheat versus refined cereal foods on metabolic risk factors: A randomised controlled two-centre intervention study. Clin. Nutr. 2013, 32, 941–949. [Google Scholar] [CrossRef]
- Bui, T.N.; Le, T.H.; Nguyen do, H.; Tran, Q.B.; Nguyen, T.L.; Le, D.T.; Nguyen do, V.A.; Vu, A.L.; Aoto, H.; Okuhara, Y.; et al. Pre-germinated brown rice reduced both blood glucose concentration and body weight in Vietnamese women with impaired glucose tolerance. J. Nutr. Sci. Vitaminol. 2014, 60, 183–187. [Google Scholar] [CrossRef]
- Chen, J.; He, J.; Wildman, R.P.; Reynolds, K.; Streiffer, R.H.; Whelton, P.K. A randomized controlled trial of dietary fiber intake on serum lipids. Eur. J. Clin. Nutr. 2006, 60, 62–68. [Google Scholar] [CrossRef]
- Pins, J.J.; Geleva, D.; Keenan, J.M.; Frazel, C.; O’Connor, P.J.; Cherney, L.M. Do whole-grain oat cereals reduce the need for antihypertensive medications and improve blood pressure control? J. Fam. Pract. 2002, 51, 353–359. [Google Scholar]
- Yusuf, S.; Dagenais, G.; Pogue, J.; Bosch, J.; Sleight, P. Vitamin E supplementation and cardiovascular events in high-risk patients. N. Engl. J. Med. 2000, 342, 154–160. [Google Scholar] [CrossRef]
- Nicklas, T.A.; O’Neil, C.E.; Fulgoni, V.L., 3rd. Diet quality is inversely related to cardiovascular risk factors in adults. J. Nutr. 2012, 142, 2112–2118. [Google Scholar] [CrossRef]
- Piercy, K.L.; Troiano, R.P.; Ballard, R.M.; Carlson, S.A.; Fulton, J.E.; Galuska, D.A.; George, S.M.; Olson, R.D. The Physical Activity Guidelines for Americans. JAMA 2018, 320, 2020–2028. [Google Scholar] [CrossRef]
- Magee, L.; Hale, L. Longitudinal associations between sleep duration and subsequent weight gain: A systematic review. Sleep Med. Rev. 2012, 16, 231–241. [Google Scholar] [CrossRef]
- Fulgoni, V.L., 3rd; Brauchla, M.; Fleige, L.; Chu, Y. Oatmeal-Containing Breakfast is Associated with Better Diet Quality and Higher Intake of Key Food Groups and Nutrients Compared to Other Breakfasts in Children. Nutrients 2019, 11, 964. [Google Scholar] [CrossRef]
- O’Neil, C.E.; Nicklas, T.A.; Zanovec, M.; Cho, S. Whole-grain consumption is associated with diet quality and nutrient intake in adults: The National Health and Nutrition Examination Survey, 1999–2004. J. Am. Diet. Assoc. 2010, 110, 1461–1468. [Google Scholar] [CrossRef]
- Mann, K.D.; Pearce, M.S.; McKevith, B.; Thielecke, F.; Seal, C.J. Whole grain intake and its association with intakes of other foods, nutrients and markers of health in the National Diet and Nutrition Survey rolling programme 2008–11. Br. J. Nutr. 2015, 113, 1595–1602. [Google Scholar] [CrossRef]
- Maki, K.C.; Slavin, J.L.; Rains, T.M.; Kris-Etherton, P.M. Limitations of observational evidence: Implications for evidence-based dietary recommendations. Adv. Nutr. 2014, 5, 7–15. [Google Scholar] [CrossRef]
- Isaksson, H.; Tillander, I.; Andersson, R.; Olsson, J.; Fredriksson, H.; Webb, D.L.; Aman, P. Whole grain rye breakfast - sustained satiety during three weeks of regular consumption. Physiol. Behav. 2012, 105, 877–884. [Google Scholar] [CrossRef]
- Gerard, P. Gut microbiota and obesity. Cell Mol. Life Sci. 2016, 73, 147–162. [Google Scholar] [CrossRef]
- Blaut, M. Gut microbiota and energy balance: Role in obesity. Proc. Nutr. Soc. 2015, 74, 227–234. [Google Scholar] [CrossRef]
- Sandberg, J.C.; Bjorck, I.M.E.; Nilsson, A.C. Effects of whole grain rye, with and without resistant starch type 2 supplementation, on glucose tolerance, gut hormones, inflammation and appetite regulation in an 11–14.5 hour perspective; a randomized controlled study in healthy subjects. Nutr. J. 2017, 16, 25. [Google Scholar] [CrossRef]
- De Moura, F.F.; Lewis, K.D.; Falk, M.C. Applying the FDA definition of whole grains to the evidence for cardiovascular disease health claims. J. Nutr. 2009, 139, 2220S–2226S. [Google Scholar] [CrossRef]
- Wang, Q.; Ellis, P.R. Oat beta-glucan: Physico-chemical characteristics in relation to its blood-glucose and cholesterol-lowering properties. Br. J. Nutr. 2014, 112, S4–S13. [Google Scholar] [CrossRef]
- Calinoiu, L.F.; Vodnar, D.C. Whole Grains and Phenolic Acids: A Review on Bioactivity, Functionality, Health Benefits and Bioavailability. Nutrients 2018, 10, 1615. [Google Scholar] [CrossRef]
- Nayak, B.; Liu, R.H.; Tang, J. Effect of processing on phenolic antioxidants of fruits, vegetables, and grains—A review. Crit. Rev. Food Sci. Nutr. 2015, 55, 887–919. [Google Scholar] [CrossRef]
| Study Author, Year | Cohort (Country) | Subject Number | Follow-Up (years) | WG Exposure | Main Weight Outcome |
|---|---|---|---|---|---|
| Liu, 2003 [37] | NHS, females (United States) | 74,091 | 12 | Dark bread, WG cereals, popcorn, wheat germ, brown rice, bran, bulgur, kasha, couscous, etc. | WG intake inversely associated with weight gain |
| Koh-Banerjee, 2004 [40] | HPFS, males (United States) | 27,082 | 8 | WG foods with at least 51% WG content by weight | WG intake inversely associated with weight gain |
| Bazzano, 2005 [41] | PHS, males (United States) | 17,881 | 13 | WG ready-to-eat breakfast cereals | WG breakfast cereal intake inversely linked to weight gain |
| Mozaffarian, 2011 [43] | NHS, NHS II, HPFS (collectively males and females) (United States) | 120,877 | 20 | Bran, brown rice, cold breakfast cereal, cooked oatmeal, other cooked breakfast cereal, dark bread, and wheat germ | WG intake inversely associated with the among of weight gain |
| De la Feuente-Arrillaga, 2014 [3] | SUN Project, males and females (Spain) | 9,267 | 5 | WG bread | No association of WG bread intake with weight change |
| Winkvist, [42] 2017 | NSHD, males and females (Sweden) | 15,995 | 10 | NR | WG intake inversely associated with BMI change in men only |
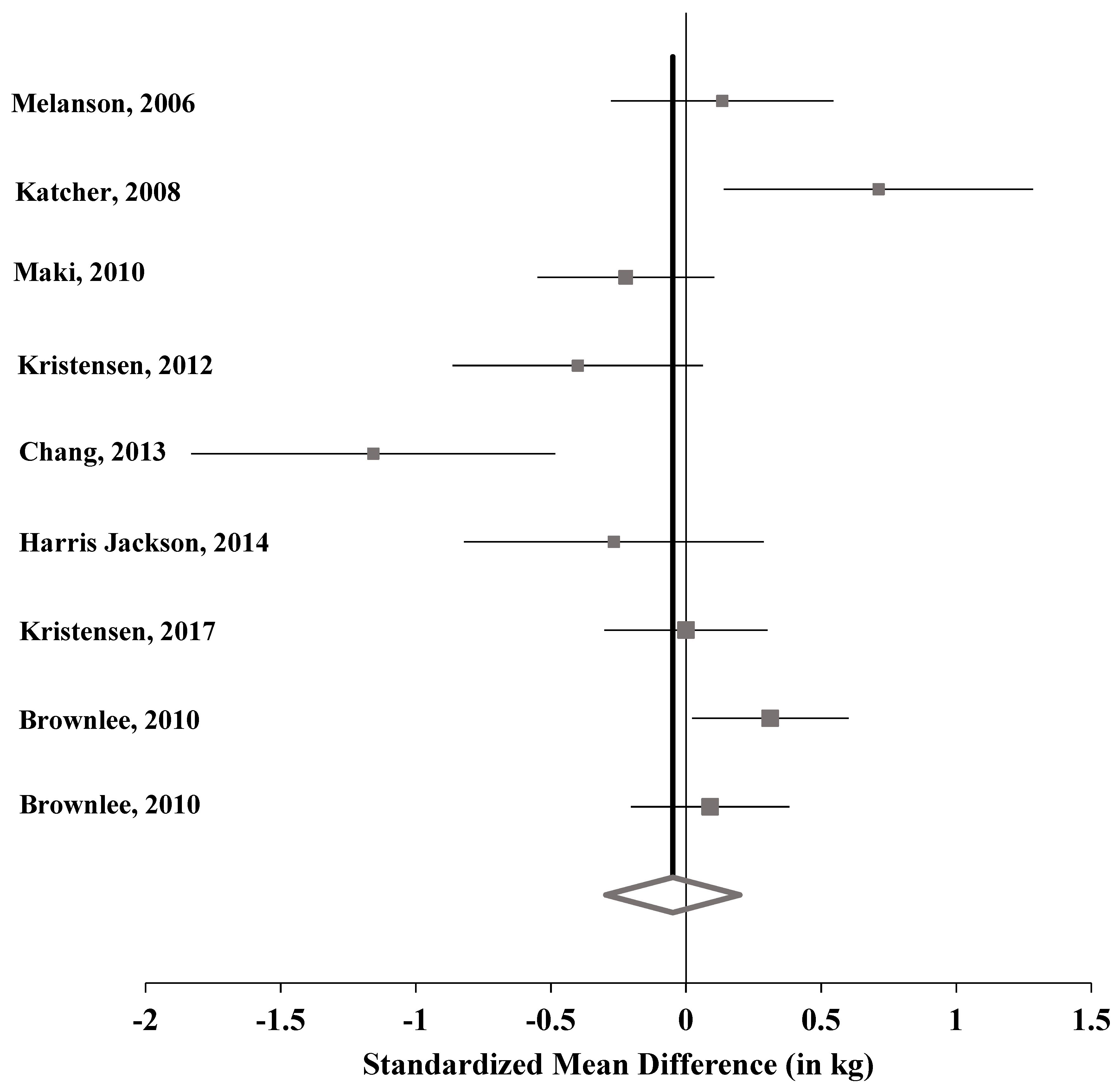
| Study Author, Year | Subjects | SMD | 95% CI | p-Value | Weight | |
|---|---|---|---|---|---|---|
| Melanson, 2006 [44] | 91 | 0.134 | −0.277, 0.545 | 0.524 | 11.35% | |
| Katcher, 2008 [45] | 47 | 0.712 | 0.140, 1.284 | 0.015 | 8.78% | |
| Maki, 2010 [46] | 144 | −0.223 | −0.550, 0.105 | 0.183 | 12.81% | |
| Kristensen, 2012 [48] | 72 | −0.401 | −0.863, 0.062 | 0.090 | 10.47% | |
| Chang, 2013 [49] | 34 | −1.158 | −1.831, -0.484 | 0.001 | 7.43% | |
| Harris Jackson, 2014 [50] | 50 | -0.267 | −0.822, 0.287 | 0.345 | 9.03% | |
| Kristensen, 2017 [47] | 169 | 0.000 | −0.302, 0.302 | 1.000 | 13.25% | |
| Brownlee, 2010 [51] | 185 | 0.312 | 0.023, 0.601 | 0.035 | 13.47% | |
| Brownlee, 2010 [51] | 181 | 0.089 | −0.204, 0.382 | 0.551 | 13.41% | |
| Pooled | 973 | −0.049 | 0.199, −0.388 | 0.698 | 100.00% |
| Secondary Analysis | Included Studies | Subjects | SMD | 95% CI | p-Value |
|---|---|---|---|---|---|
| Waist Circumference | Katcher, 2008 [45] Maki, 2010 [46] Kristensen, 2012 [48] Harris Jackson, 2014 [50] Kristensen, 2017 [47] | 482 | 0.276 | −0.436, 0.989 | 0.447 |
| Body Fat Percentage | Katcher, 2008 [45] Chang, 2013 [49] Harris Jackson, 2014 [50] Kristensen, 2017 [47] Brownlee, 2010 [51] Brownlee, 2010 [51] | 666 | 0.042 | −0.573, 0.656 | 0.895 |
| Mixed Population | Melanson, 2006 [44] Katcher, 2008 [45] Maki, 2010 [46] Chang, 2013 [49] Harris Jackson, 2014 [50] Brownlee, 2010 [51] Brownlee, 2010 [51] | 732 | −0.016 | −0.329, 0.297 | 0.921 |
| Hypocaloric Diet | Melanson, 2006 [44] Katcher, 2008 [45] Maki, 2010 [46] Kristensen, 2012 [48] Harris Jackson, 2014 [50] Kristensen, 2017 [47] | 573 | −0.031 | −0.291, 0.229 | 0.814 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maki, K.C.; Palacios, O.M.; Koecher, K.; Sawicki, C.M.; Livingston, K.A.; Bell, M.; Nelson Cortes, H.; McKeown, N.M. The Relationship between Whole Grain Intake and Body Weight: Results of Meta-Analyses of Observational Studies and Randomized Controlled Trials. Nutrients 2019, 11, 1245. https://doi.org/10.3390/nu11061245
Maki KC, Palacios OM, Koecher K, Sawicki CM, Livingston KA, Bell M, Nelson Cortes H, McKeown NM. The Relationship between Whole Grain Intake and Body Weight: Results of Meta-Analyses of Observational Studies and Randomized Controlled Trials. Nutrients. 2019; 11(6):1245. https://doi.org/10.3390/nu11061245
Chicago/Turabian StyleMaki, Kevin C., Orsolya M. Palacios, Katie Koecher, Caleigh M. Sawicki, Kara A. Livingston, Marjorie Bell, Heather Nelson Cortes, and Nicola M. McKeown. 2019. "The Relationship between Whole Grain Intake and Body Weight: Results of Meta-Analyses of Observational Studies and Randomized Controlled Trials" Nutrients 11, no. 6: 1245. https://doi.org/10.3390/nu11061245
APA StyleMaki, K. C., Palacios, O. M., Koecher, K., Sawicki, C. M., Livingston, K. A., Bell, M., Nelson Cortes, H., & McKeown, N. M. (2019). The Relationship between Whole Grain Intake and Body Weight: Results of Meta-Analyses of Observational Studies and Randomized Controlled Trials. Nutrients, 11(6), 1245. https://doi.org/10.3390/nu11061245





