Preventive Effects and Mechanisms of Garlic on Dyslipidemia and Gut Microbiome Dysbiosis
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Mouse Model
2.3. Measurement of Serum Biochemical Indicators
2.4. Histomorphology
2.5. Cecal Organic Acid Analyses
2.6. Characterization of the Gut Microbiome by 16S rRNA Gene Sequencing
2.7. Statistical Analysis
3. Result
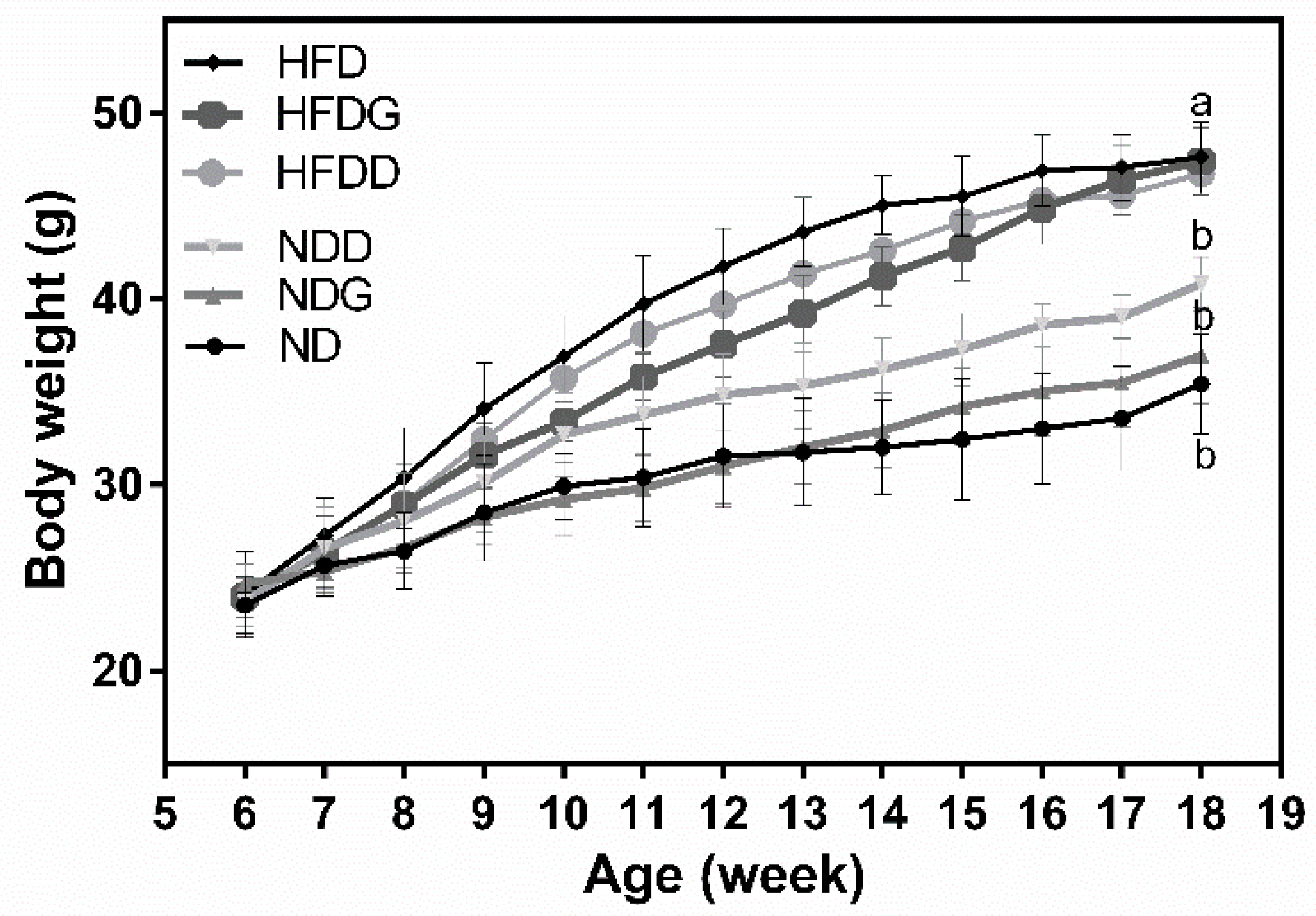
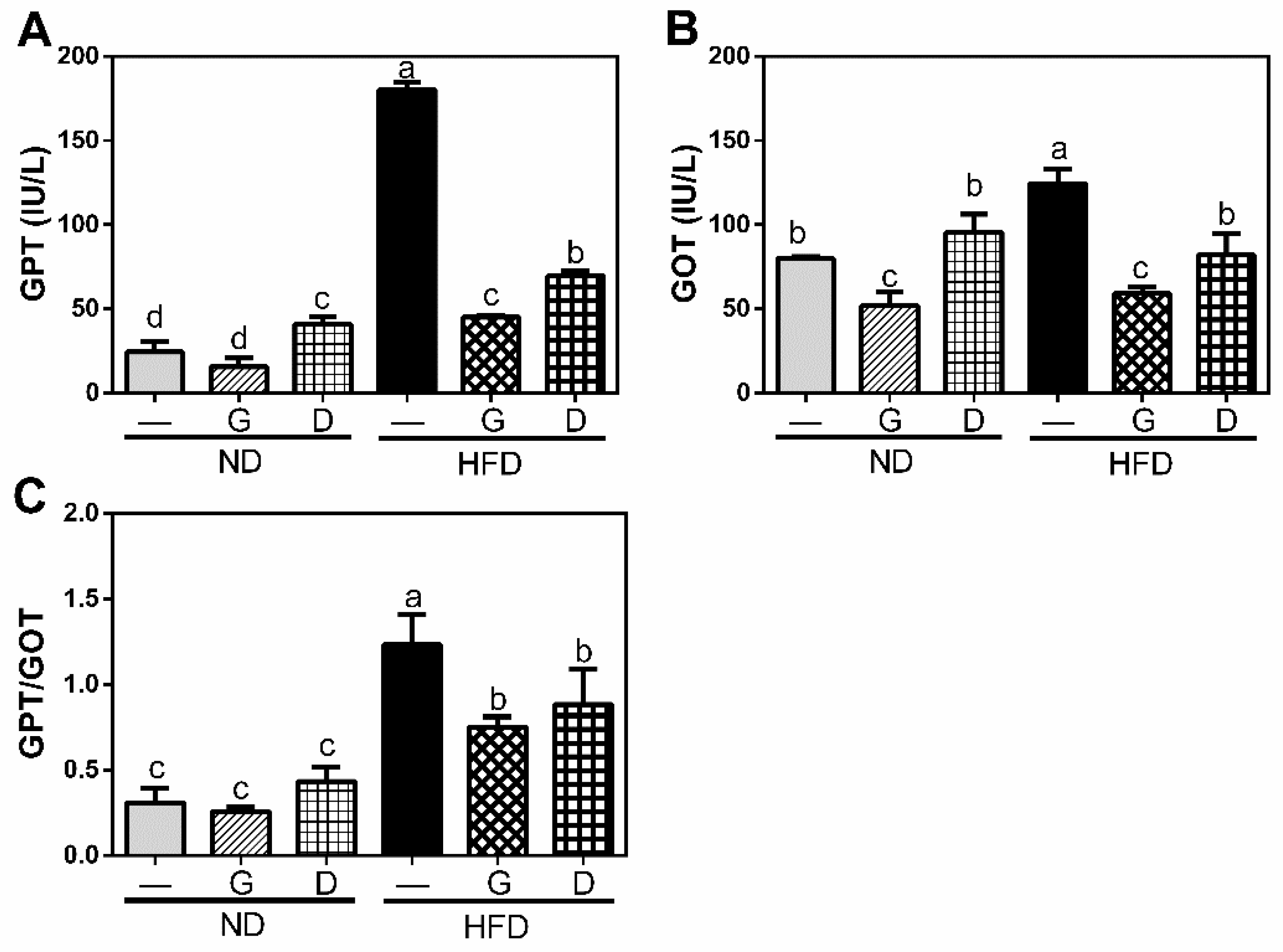
3.1. Body Weight and Index of Liver Injury
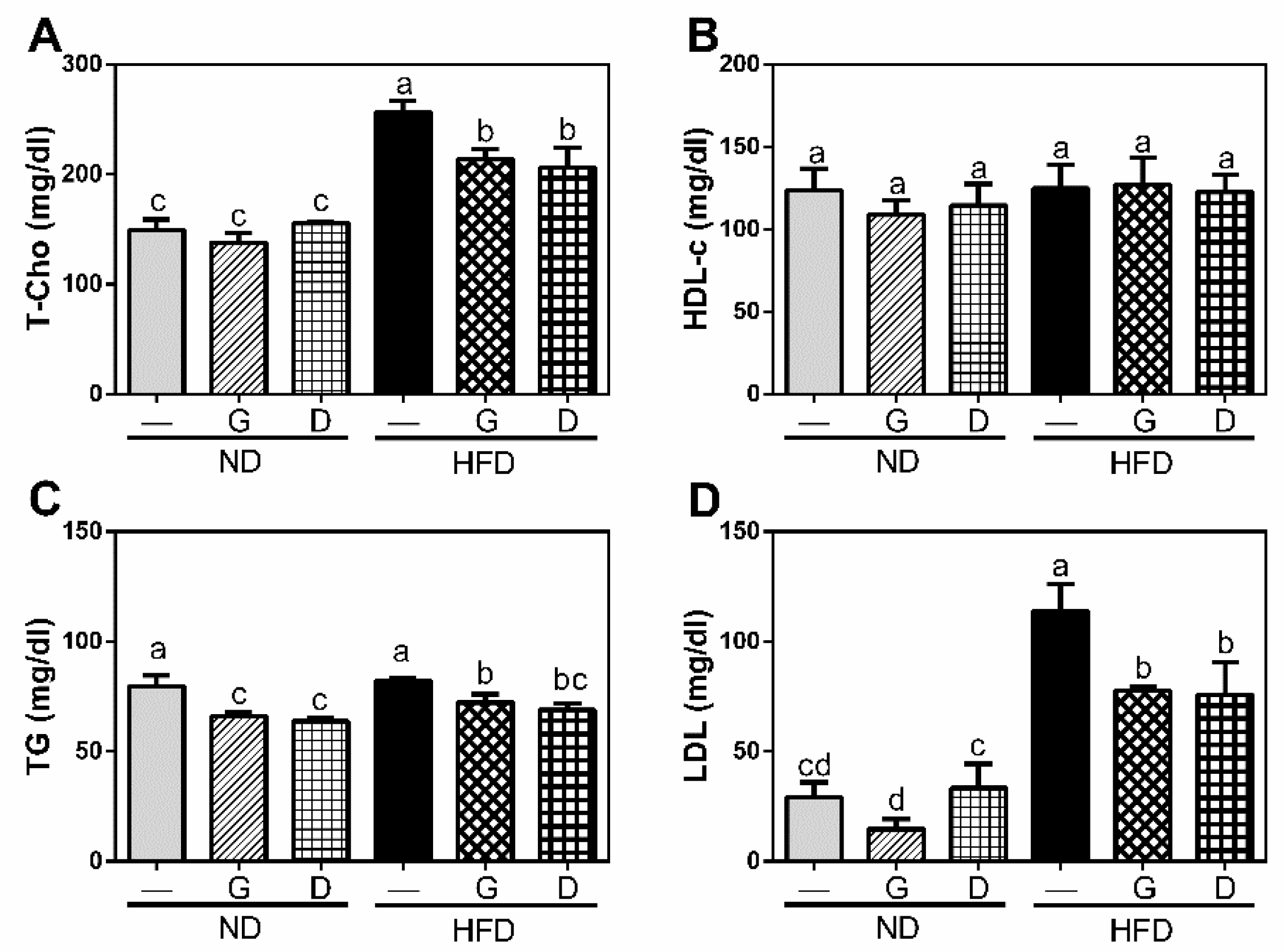
3.2. Effect of Garlic on Metabolism of Lipid and Glucose
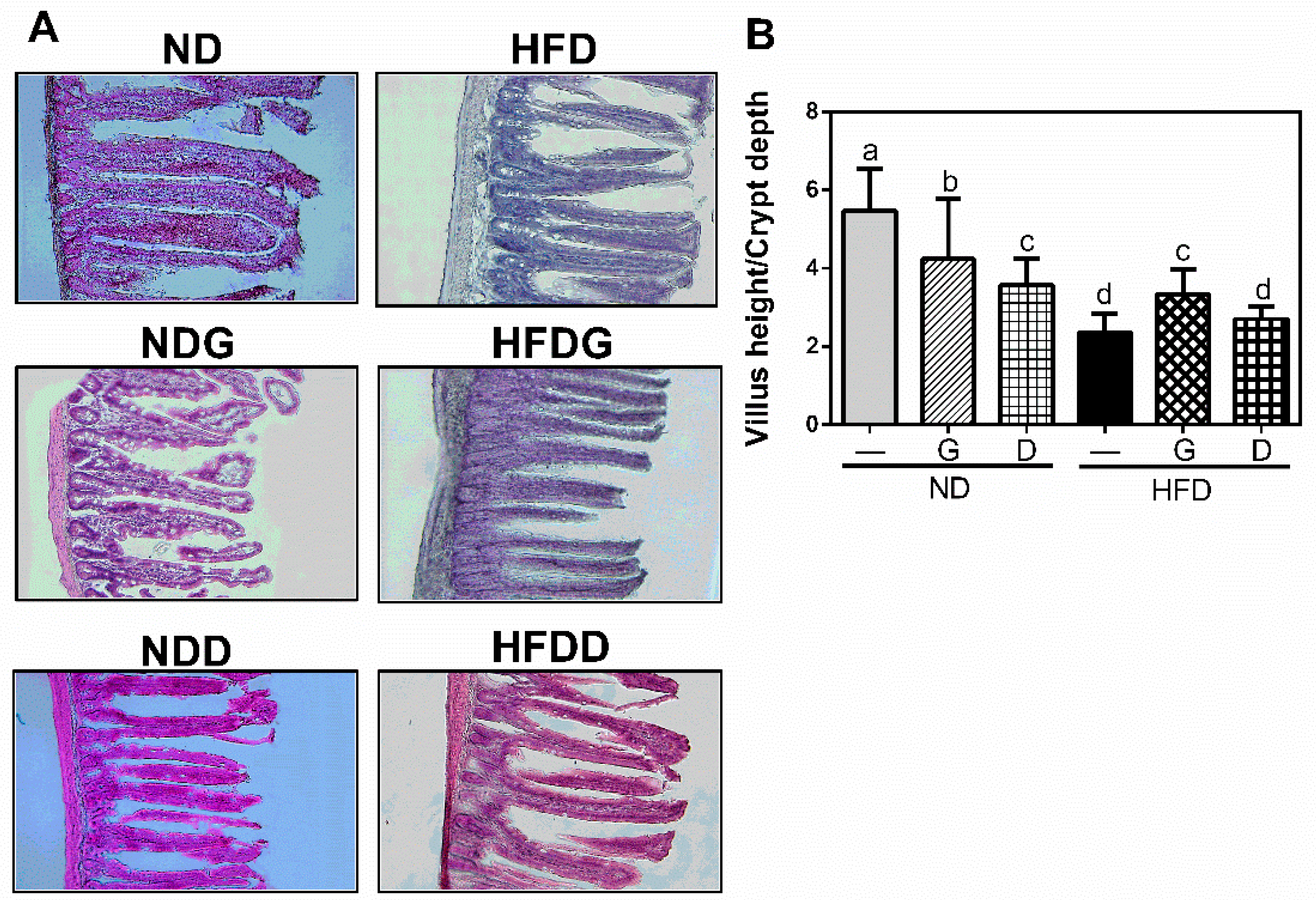
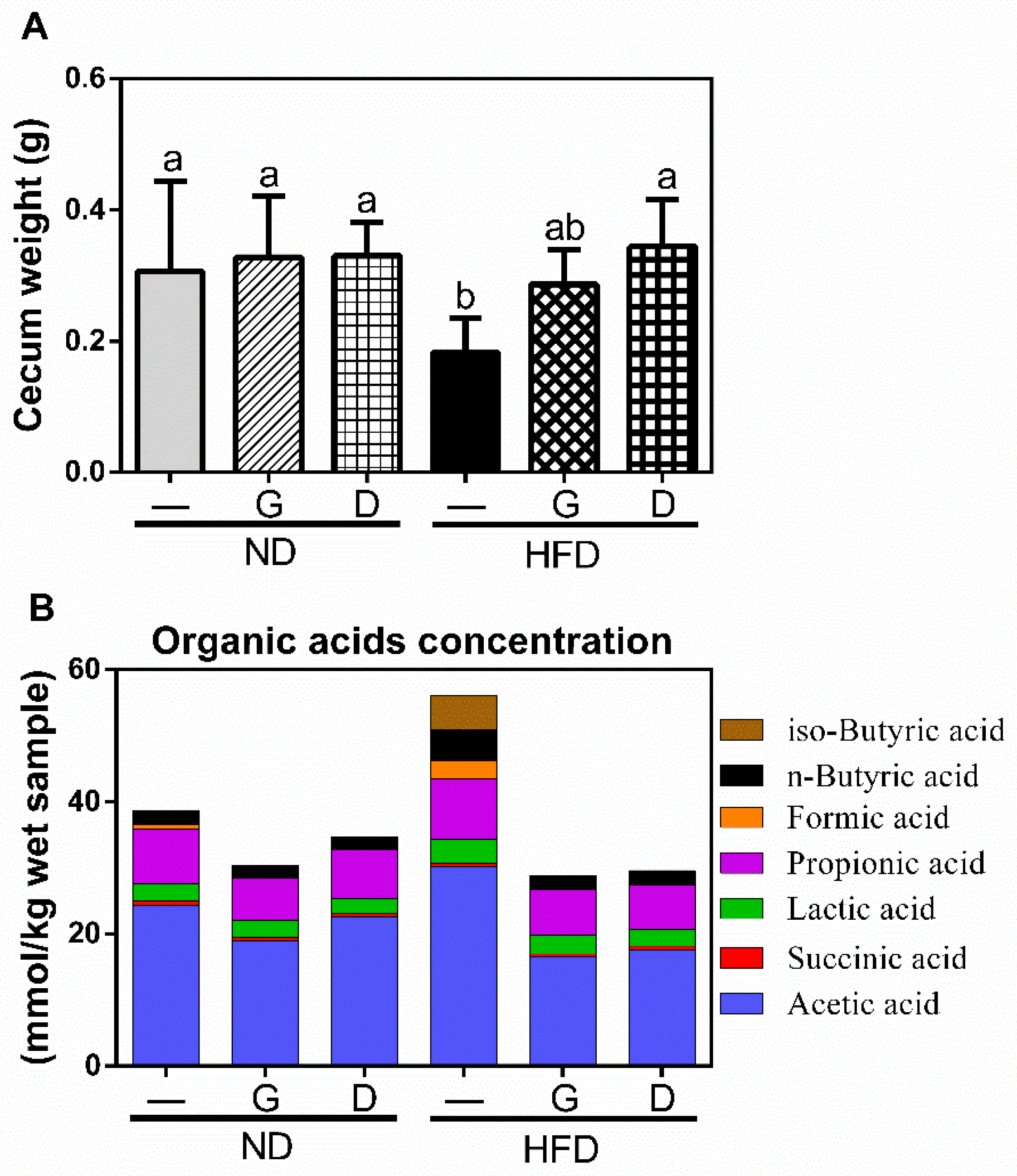
3.3. Effect of Garlic on Terminal Ileum Histomorphology and the Concentration of Organic Acids on Cecum
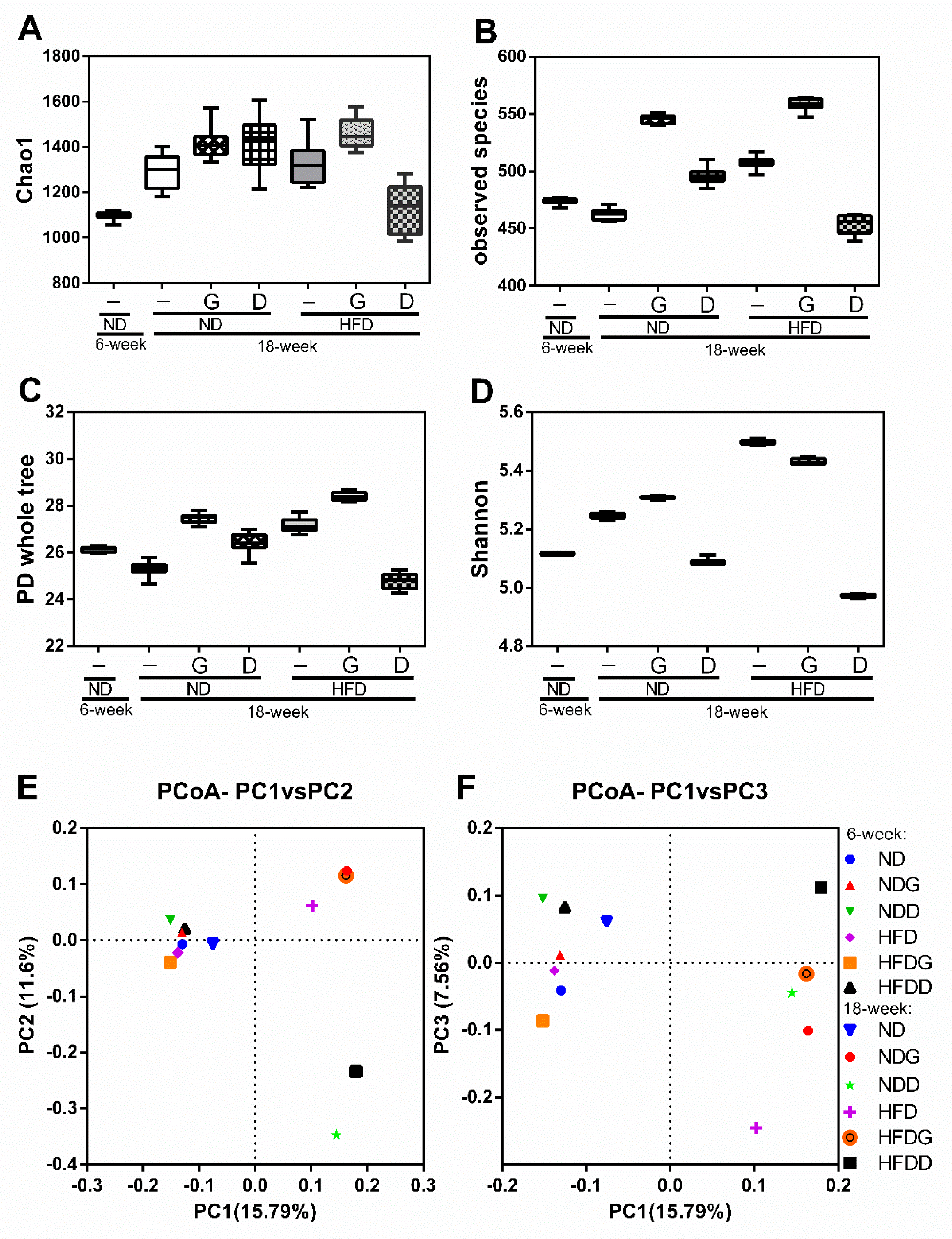
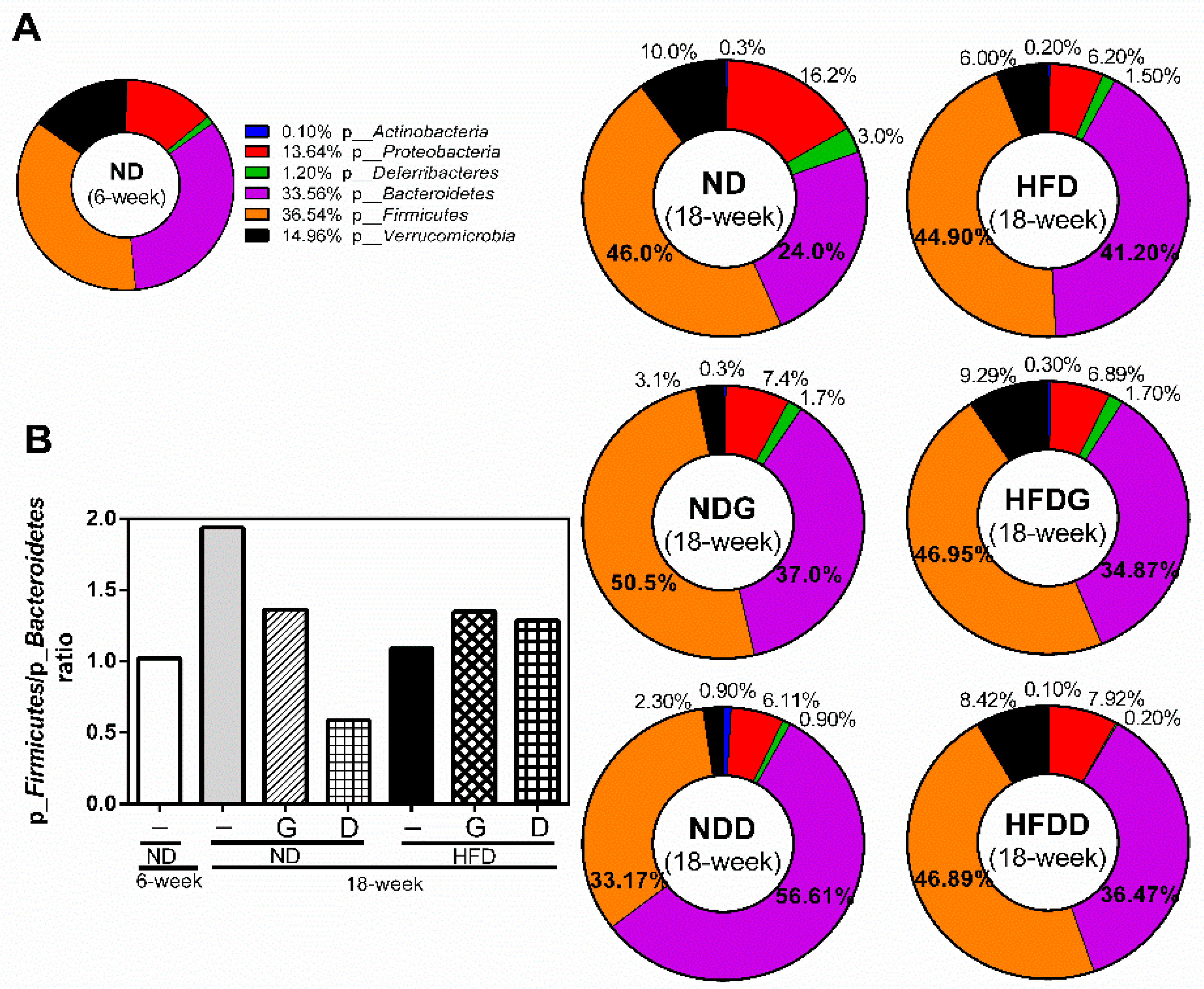
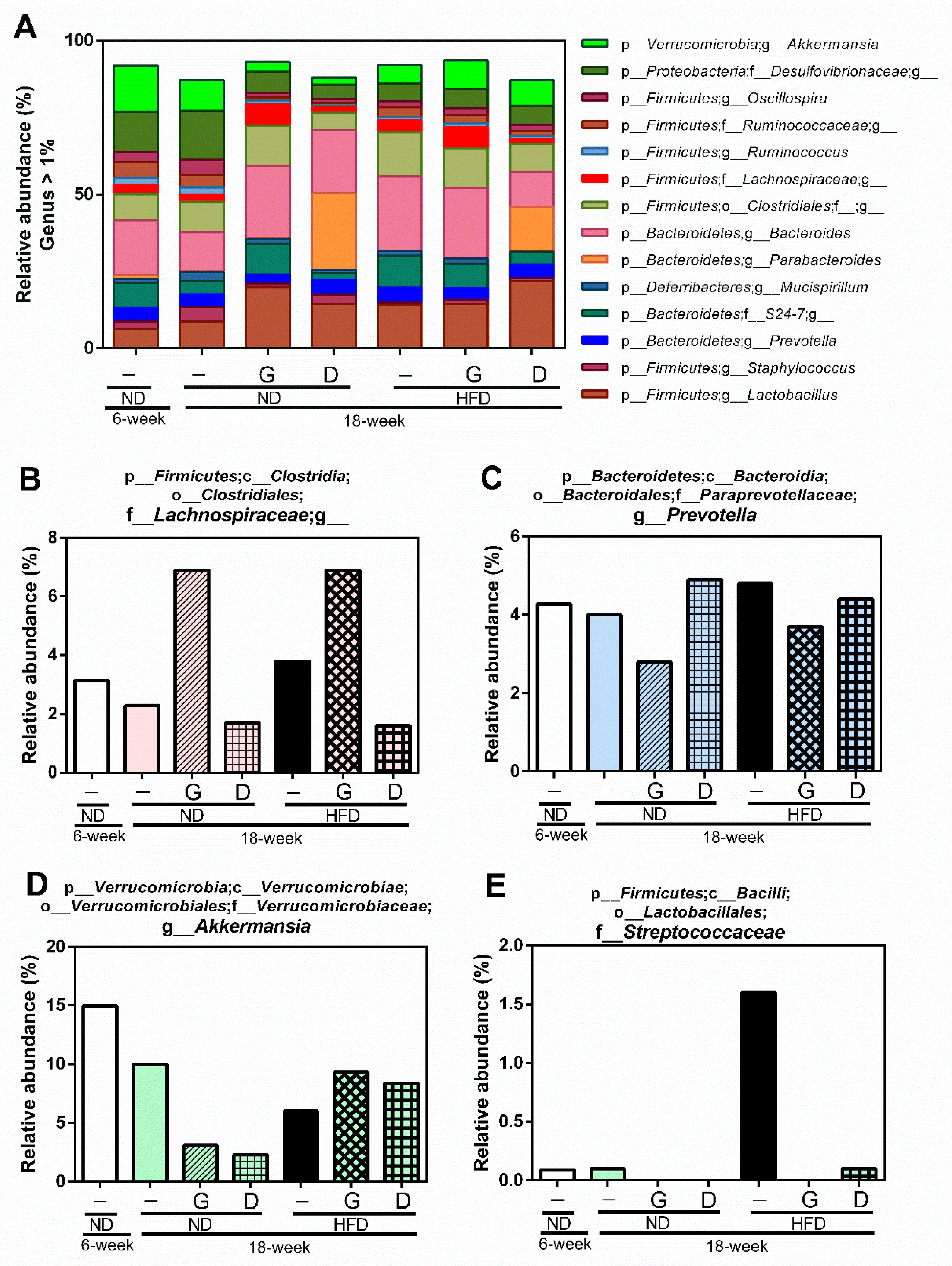
3.4. Modulation of the Gut Microbiome by Garlic
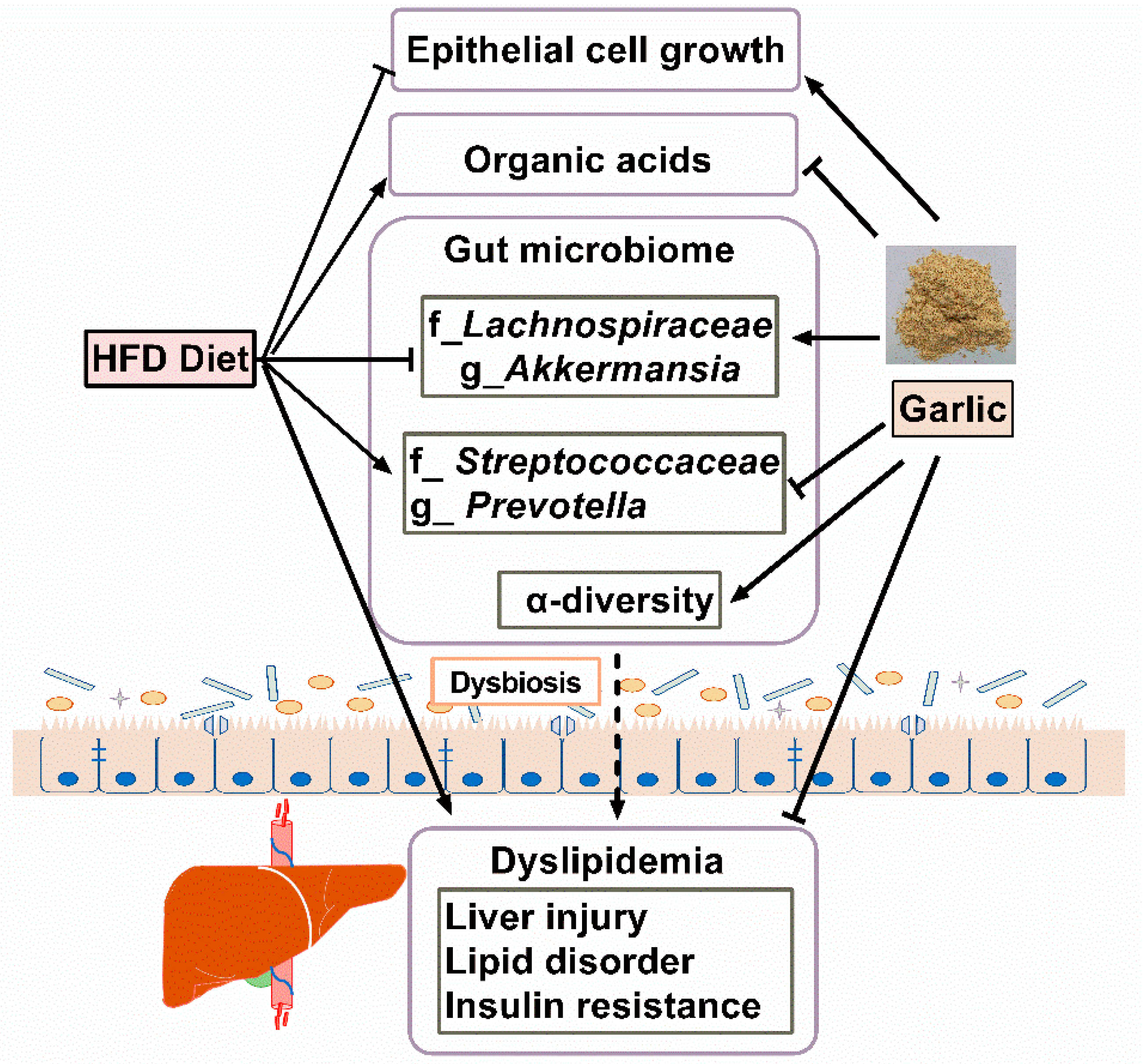
4. Discussion
Author Contributions
Funding
Conflicts of Interest
Appendix A
| G: Garlic Supplementation | ||
|---|---|---|
| Nutrients | Value | Method |
| Calorie | 370 kcal/100 g | Calorie = Protein × 4 + Lipid × 9 + Carbohydrates × 4 |
| Protein | 16.2 g/100 g | Kaida’s method |
| Lipid | 1.1 g/100 g | Acid decomposition method |
| Carbohydrates | 73.9 g/100 g | 100 − (protein + lipid + moisture + ash) |
| Salt | 0.02 g/100 g | Sodium conversion |
| Sodium | 7 mg/100 g | Atomic absorption spectroscopy |
| Moisture | 4.8 g/100 g | High pressure drying |
| Ash | 4.0 g/100 g | Ashing method |
| Fructan | 54.8 g/100 g | Fructan analysis kit |
| G-SAC | 3.385 mg/g | HPLC |
| Alliin | 13.261 mg/g | HPLC |
| SAC | 1.867 mg/g | HPLC |
| Allicin | 4.714 mg/g (wet) | HPLC [66] |
| Components (%) | ND | NDG | NDD | HFD | HFDG | HFDD |
|---|---|---|---|---|---|---|
| Lard | 6.0 | 5.9 | 6.0 | 40.0 | 39.9 | 40.0 |
| Corn starch | 54.0 | 50.2 | 54.0 | 20.0 | 16.2 | 20.0 |
| Casein | 21.0 | 20.2 | 21.0 | 21.0 | 20.2 | 21.0 |
| Sucrose | 10.0 | 9.8 | 10.0 | 10.0 | 9.8 | 10.0 |
| Cellulose | 4.0 | 3.9 | 0.0 | 4.0 | 3.9 | 0.0 |
| Mineral mix | 3.5 | 3.5 | 3.5 | 3.5 | 3.5 | 3.5 |
| Vitamin mix | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 | 1.0 |
| Choline chloride | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 | 0.2 |
| Methionine | 0.3 | 0.3 | 0.3 | 0.3 | 0.3 | 0.3 |
| G (Garlic) | — | 5.0 | — | — | 5.0 | — |
| D (Dextrin) | — | — | 4.0 | — | — | 4.0 |
| Total calories (kcal/100 g) | 367.8 | 368.5 | 367.8 | 567.4 | 567.9 | 568.1 |
References
- Koch, H.P.; Lawson, L.D. (Eds.) Garlic: The Science and Therapeutic Application of Allium sativum L. and Related Species, 2nd ed.; Williams and Wilkins: Baltimore, MD, USA, 1996; pp. 1–329. [Google Scholar]
- Lawson, L.D.; Wang, Z.Y.J. Changes in the organosulfur compounds released from garlic during aging in water, dilute ethanol, or dilute acetic acid. J. Toxicol. 1995, 14, 214. [Google Scholar]
- Baumgartner, S.; Dax, T.G.; Praznik, W.; Falk, H. Characterization of the highmolecular weight fructan isolated from garlic (Allium sativum L.). Carbohydr. Res. 2000, 328, 177–183. [Google Scholar] [CrossRef]
- Kelly, G.S. Larch arabinogalactan: Clinical relevance of a novel immune-enhancing polysaccharide. Altern. Med. Rev. 1999, 4, 96–103. [Google Scholar]
- Paulsen, B.S. Plant polysaccharides with immunostimulatory activities. Curr. Org. Chem. 2001, 5, 939–950. [Google Scholar] [CrossRef]
- Block, K.I.; Mead, M.N. Immune system effects of Echinacea, ginseng, and astragalus: A review. Integr. Cancer Ther. 2003, 2, 247–267. [Google Scholar] [CrossRef]
- Tsai, C.C.; Lin, C.R.; Tsai, H.Y.; Chen, C.J.; Li, W.T.; Yu, H.M.; Ke, Y.Y.; Hsieh, W.Y.; Chang, C.Y.; Wu, C.Y.; et al. The immunologically active oligosaccharides isolated from wheatgrass modulate monocytes via toll-like receptor-2 signaling. J. Biol. Chem. 2013, 288, 17689–17697. [Google Scholar] [CrossRef] [PubMed]
- Vogt, L.; Meyer, D.; Pullens, G.; Faas, M.; Smelt, M.; Venema, K.; Ramasamy, U.; Schols, H.A.; De Vos, P. Immunological properties of inulin-type fructans. Crit. Rev. Food Sci. Nutr. 2014, 55, 414–436. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Yajima, T.; Li, W.; Saito, K.; Ohshima, Y.; Yoshikai, Y. Levan (β-2, 6-fructan), a major fraction of fermented soybean mucilage, displays immunostimulating properties via Toll-like receptor 4 signalling: Induction of interleukin-12 production and suppression of T-helper type 2 response and immunoglobulin E production. Clin. Exp. Allergy 2006, 36, 94–101. [Google Scholar] [PubMed]
- Franco-Robles, E.; López, M.G. Implication of Fructans in Health: Immunomodulatory and Antioxidant Mechanisms. Sci. World J. 2015, 2015, 289267. [Google Scholar] [CrossRef]
- Matsuura, H.; Lachance, P.A. (Eds.) Phytochemistry of Garlic Horticultural and Processing Procedures; Food and Nutrition Press: Trumbull, CT, USA, 1997; pp. 55–69. [Google Scholar]
- Amagase, H. Clarifying the real bioactive constituents of garlic. J. Nutr. 2006, 136, 716S–725S. [Google Scholar] [CrossRef] [PubMed]
- Larry, D.; Lawson, S.; Hunsaker, M. Allicin Bioavailability and Bioequivalence from Garlic Supplements and Garlic Foods. Nutrients 2018, 10, 812. [Google Scholar]
- Gardner, C.D.; Lawson, L.D.; Block, E.; Chatterjee, L.M.; Kiazand, A.; Balise, R.R.; Kraemer, H.C. Effect of raw garlic vs. commercial garlic supplements on plasma lipid concentrations in adults with moderate hypercholesterolemia: A randomized clinical trial. Arch. Int. Med. 2007, 167, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Chu, F.; Shi, M.; Lang, Y.; Shen, D.; Jin, T.; Zhu, J.; Cui, L. Gut Microbiota in Multiple Sclerosis and Experimental Autoimmune Encephalomyelitis: Current Applications and Future Perspectives. Mediat. Inflamm. 2018, 8168717. [Google Scholar] [CrossRef] [PubMed]
- Sheflin, A.M.; Borresen, E.C.; Kirkwood, J.S.; Boot, C.M.; Whitney, A.K.; Lu, S.; Brown, R.J.; Broeckling, C.D.; Ryan, E.P.; Weir, T.L. Dietary supplementation with rice bran or navy bean alters gut bacterial metabolism in colorectal cancer survivors. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- Li, X.W.; Chen, H.P.; He, Y.Y.; Chen, W.L.; Chen, J.W.; Gao, L.; Hu, H.Y.; Wang, J. Effects of rich-polyphenols extract of dendrobium loddigesii on anti-diabetic, anti-inflammatory, anti-oxidant, and gut microbiota modulation in db/db mice. Molecules 2018, 23, 3245. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Jin, X.; Li, Q.; Sawaya, A.C.H.F.; Le Leu, R.K.; Conlon, M.A.; Wu, L.; Hu, F. Propolis from Different Geographic Origins Suppress Intestinal Inflammation in a Model of DSS-Induced Colitis is Associated with Decreased Bacteroides spp. in the Gut. Mol. Nutr. Food Res. 2018. [CrossRef]
- Fontana, L.; Partridge, L. Promoting health and longevity through diet: From model organisms to humans. Cell 2015, 161, 106–118. [Google Scholar] [CrossRef] [PubMed]
- Cani, P.D.; Delzenne, N.M. Gut microflora as a target for energy and metabolic homeostasis. Curr. Opin. Clin. Nutr. Metab. Care. 2007, 10, 729–734. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Zhang, M.; Wang, S.; Han, R.; Cao, Y.; Hua, W.; Mao, Y.; Zhang, X.; Pang, X.; Wei, C.; et al. Interactions between gut microbiota, host genetics and diet relevant to development of metabolic syndromes in mice. ISME J. 2010, 4, 232–241. [Google Scholar] [CrossRef]
- Peshev, D.; Van den Ende, W. Fructans: Prebiotics and immunomodulators. J. Funct. Foods 2014, 8, 348–357. [Google Scholar] [CrossRef]
- Leontiev, R.; Hohaus, N.; Jacob, C.; Gruhlke, M.C.H.; Slusarenko, A.J. A Comparison of the Antibacterial and Antifungal Activities of Thiosulfinate Analogues of Allicin. Sci. Rep. 2018. [Google Scholar] [CrossRef] [PubMed]
- Jakobsen, T.H.; van Gennip, M.; Phipps, R.K.; Shanmugham, M.S.; Christensen, L.D.; Alhede, M.; Skindersoe, M.E.; Rasmussen, T.B.; Friedrich, K.; Uthe, F.; et al. Ajoene, a Sulfur-Rich Molecule from Garlic, Inhibits Genes Controlled by Quorum Sensing. Antimicrob. Agents Chemother. 2012, 56, 2314–2325. [Google Scholar] [CrossRef] [PubMed]
- Slavin, J. Fiber and prebiotics: Mechanisms and health benefits. Nutrients 2013, 5, 1417–1435. [Google Scholar] [CrossRef] [PubMed]
- Barczynska, R.; Kapusniak, J.; Litwin, M.; Slizewska, K.; Szalecki, M. Dextrins from Maize Starch as Substances Activating the Growth of Bacteroidetes and Actinobacteria Simultaneously Inhibiting the Growth of Firmicutes, Responsible for the Occurrence of Obesity. Plant Foods Hum. Nutr. 2016, 71, 190–196. [Google Scholar] [CrossRef]
- Elli, M.; Cattivelli, D.; Soldi, S.; Bonatti, M.; Morelli, L. Evaluation of Prebiotic Potential of Refined Psyllium (Plantago ovata) Fiber in Healthy Women. J. Clin. Gastroenterol. 2010, 42, S174–S176. [Google Scholar] [CrossRef]
- Warnick, G.R.; Knopp, R.H.; Fitzpatrick, V.; Branson, L. Estimating low-density lipoprotein cholesterol by the Friedewald equation is adequate for classifying patients on the basis of nationally recommended cutpoints. Clin. Chem. 1990, 36, 15–19. [Google Scholar] [PubMed]
- Turner, R.C.; Holman, R.R.; Matthews, D.; Hockaday, T.D.R.; Peto, J. Insulin deficiency and insulin resistance interaction in diabetes: Estimation of their relative contribution by feedback analysis from basal plasma insulin and glucose concentrations. Metabolism 1979, 28, 1086–1096. [Google Scholar] [CrossRef]
- Tsukahara, T.; Matsukawa, N.; Tomonaga, S.; Inoue, R.; Ushida, K.; Ochiai, K. High-sensitivity detection of short-chain fatty acids in porcine ileal, cecal, portal and abdominal blood by gas chromatography-mass spectrometry. Anim. Sci. J. 2014, 85, 494–498. [Google Scholar] [CrossRef]
- Wu, S.; Hu, R.; Nakano, H.; Chen, K.; Liu, M.; He, X.; Zhang, H.; He, J.; Hou, D.-X. Modulation of Gut Microbiota by Lonicera caerulea L. Berry Polyphenols in a Mouse Model of Fatty Liver Induced by High Fat Diet. Molecules 2018, 23, 3213. [Google Scholar] [CrossRef]
- Turnbaugh, P.J.; Ley, R.E.; Mahowald, M.A.; Magrini, V.; Mardis, E.R.; Gordon, J.I. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 2006, 444, 1027–1031. [Google Scholar] [CrossRef]
- Lin, H.V.; Frassetto, A.; Kowalik, E.J.; Nawrocki, A.R.; Lu, M.M.; Kosinski, J.R.; Hubert, J.A.; Szeto, D.; Yao, X.; Forrest, G.; et al. Butyrate and propionate protect against diet-induced obesity and regulate gut hormones via free fatty acid receptor 3-independent mechanisms. PLoS ONE 2012, 7, e35240. [Google Scholar] [CrossRef]
- Brown, A.J.; Goldsworthy, S.M.; Barnes, A.A.; Eilert, M.M.; Tcheang, L.; Daniels, D.; Muir, A.I.; Wigglesworth, M.J.; Kinghorn, I.; Fraser, N.J.; et al. The orphan G protein-coupled receptors GPR41 and GPR43 are activated by propionate and other short chain carboxylic acids. J. Biol. Chem. 2003, 278, 11312–11319. [Google Scholar] [CrossRef]
- Le Poul, E.; Loison, C.; Struyf, S.; Springael, J.Y.; Lannoy, V.; Decobecq, M.E.; Brezillon, S.; Dupriez, V.; Vassart, G.; Van Damme, J.; et al. Functional characterization of human receptors for short chain fatty acids and their role in polymorphonuclear cell activation. J. Biol. Chem. 2003, 278, 25481–25489. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Obin, M.S.; Zhao, L. The gut microbiota, obesity and insulin resistance. Mol. Aspects Med. 2013, 34, 39–58. [Google Scholar] [CrossRef] [PubMed]
- Granado-Serrano, A.B.; Martín-Garí, M.; Sánchez, V.; Riart Solans, M.; Berdún, R.; Ludwig, I.A.; Rubió, L.; Vilaprinyó, E.; Portero-Otín, M.; Serrano, J.C.E. Faecal bacterial and short-chain fatty acids signature in hypercholesterolemia. Sci. Rep. 2019, 9, 1772. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; Playford, R.J. Bioactive natural compounds for the treatment of gastrointestinal disorders. Clin. Sci. 2003, 104, 547–556. [Google Scholar] [CrossRef] [PubMed]
- Munday, R.; Munday, C.M. Low doses of diallyl disulfide, a compound derived from garlic, increase tissue activities of quinone reductase and glutathione transferase in the gastrointestinal tract of the rat. Nutr. Cancer 1999, 34, 42–48. [Google Scholar] [CrossRef]
- Lin, M.; Zhang, B.; Yu, C.; Li, J.; Zhang, L.; Sun, H.; Gao, F.; Zhou, G. L-Glutamate supplementation improves small intestinal architecture and enhances the expressions of jejunal mucosa amino acid receptors and transporters in weaning piglets. PLoS ONE 2014, 9, e111950. [Google Scholar] [CrossRef]
- Hoshino, T.; Kashimoto, N.; Kasuga, S. Recent Advances on the Nutritional Effects Associated with the Use of Garlic as a Supplement Effects of Garlic Preparations on the Gastrointestinal Mucosa 1. J. Nutr. 2001, 131, 1109–1113. [Google Scholar] [CrossRef]
- Sood, D.R.; Chhokar, V.; Shilpa. Effect of garlic (Allium sativum L.) extract on degree of hydration, fructose, sulphur and phosphorus contents of rat eyelens and intestinal absorption of nutrients. Indian J. Clin. Biochem. 2003, 18, 190–196. [Google Scholar] [CrossRef]
- Chao, A. Non-parametric estimation of the number of classes in a population. Scand. J. Stat. 1984, 11, 265–270. [Google Scholar]
- Chao, A. Species richness estimation. Encycl. Stat. Sci. 2005, 12, 7909–7916. [Google Scholar]
- Forest, F.; Grenyer, R.; Rouget, M.; Davies, T.J.; Cowling, R.M.; Faith, D.P.; Balmford, A.; Manning, J.C.; Procheş, Ş.; Van Der Bank, M.; et al. Preserving the evolutionary potential of floras in biodiversity hotspots. Nature 2007, 445, 757–760. [Google Scholar] [CrossRef]
- Shannon, C.E. A Mathematical Theory of Communication. Bell Syst. Tech. J. 1948, 27, 379–423. [Google Scholar] [CrossRef]
- Pedersen, H.K.; Gudmundsdottir, V.; Nielsen, H.B.; Hyotylainen, T.; Nielsen, T.; Jensen, B.A.H.; Forslund, K.; Hildebrand, F.; Prifti, E.; Falony, G.; et al. Human gut microbes impact host serum metabolome and insulin sensitivity. Nature 2016, 535, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Jang, C.; Oh, S.F.; Wada, S.; Rowe, G.C.; Liu, L.; Chan, M.C.; Rhee, J.; Hoshino, A.; Kim, B.; Ibrahim, A.; et al. A branched-chain amino acid metabolite drives vascular fatty acid transport and causes insulin resistance. Nat. Med. 2016, 22, 421–426. [Google Scholar] [CrossRef]
- Atarashi, K.; Tanoue, T.; Shima, T.; Imaoka, A.; Kuwahara, T.; Momose, Y.; Cheng, G.; Yamasaki, S.; Saito, T.; Ohba, Y.; et al. Induction of colonic regulatory T cells by indigenous Clostridium species. Science 2011, 331, 337–341. [Google Scholar] [CrossRef] [PubMed]
- Reeves, A.E.; Koenigsknecht, M.J.; Bergin, I.L.; Young, V.B. Suppression of Clostridium difficile in the Gastrointestinal Tracts of Germfree Mice Inoculated with a Murine Isolate from the Family Lachnospiraceae. Infect. Immun. 2012, 80, 3786–3794. [Google Scholar] [CrossRef]
- Lin, Z.; Ye, W.; Zu, X.; Xie, H.; Li, H.; Li, Y.; Zhang, W. Integrative metabolic and microbial profiling on patients with Spleen-yang-deficiency syndrome. Sci. Rep. 2018, 8. [Google Scholar] [CrossRef]
- Cho, I.; Yamanishi, S.; Cox, L.; Methé, B.A.; Zavadil, J.; Li, K.; Gao, Z.; Mahana, D.; Raju, K.; Teitler, I.; et al. Antibiotics in early life alter the murine colonic microbiome and adiposity. Nature 2012, 488, 621–626. [Google Scholar] [CrossRef] [PubMed]
- Méndez-Salazar, E.O.; Ortiz-López, M.G.; Granados-Silvestre, M.D.L.Á.; Palacios-González, B.; Menjivar, M. Altered gut microbiota and compositional changes in firmicutes and proteobacteria in mexican undernourished and obese children. Front. Microbiol. 2018, 9, 2494. [Google Scholar] [CrossRef] [PubMed]
- Carrillo-Navarrete, F.; Mercado-Silva, E.; Rivera-Pastrana, D.M.; Nava-Morales, G.M.; Reynoso-Camacho, R.; Castaño-Tostado, E.; Vázquez-Barrios, M.E. Extraction and characterization of fructans from non-differentiated garlic (Allium sativum L.) and evaluation of its prebiotic effect. Acta Hortic. 2018, 1194, 991–998. [Google Scholar] [CrossRef]
- Wang, Y.; Guan, M.; Zhao, X.; Li, X. Effects of garlic polysaccharide on alcoholic liver fibrosis and intestinal microflora in mice. Pharm. Biol. 2018, 56, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Zhai, B.; Zhang, C.; Sheng, Y.; Zhao, C.; He, X.; Xu, W.; Huang, K.; Luo, Y. Hypoglycemic and hypolipidemic effect of S-allyl-cysteine sulfoxide (alliin) in DIO mice. Sci. Rep. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Yue, S.-J.; Liu, J.; Wang, A.-T.; Meng, X.-T.; Yang, Z.-R.; Peng, C.; Guan, H.-S.; Wang, C.-Y.; Yan, D. Berberine alleviates insulin resistance by reducing peripheral branched-chain amino acids. Am. J. Physiol. Metab. 2019, 316, E73–E85. [Google Scholar] [CrossRef]
- Chen, Y.; Yang, F.; Lu, H.; Wang, B.; Chen, Y.; Lei, D.; Wang, Y.; Zhu, B.; Li, L. Characterization of fecal microbial communities in patients with liver cirrhosis. Hepatology 2011, 54, 562–572. [Google Scholar] [CrossRef] [PubMed]
- Tilg, H.; Moschen, A.R. Microbiota and diabetes: An evolving relationship. Gut 2014, 63, 1513–1521. [Google Scholar] [CrossRef]
- Ganesh, B.P.; Klopfleisch, R.; Loh, G.; Blaut, M. Commensal Akkermansia muciniphila Exacerbates Gut Inflammation in Salmonella Typhimurium-Infected Gnotobiotic Mice. PLoS ONE 2013, 8, e74963. [Google Scholar] [CrossRef]
- Holscher, H.D. Dietary fiber and prebiotics and the gastrointestinal microbiota. Gut Microbes 2017, 8, 172–184. [Google Scholar] [CrossRef]
- Gamage, H.K.A.H.; Tetu, S.G.; Chong, R.W.W.; Bucio-Noble, D.; Rosewarne, C.P.; Kautto, L.; Ball, M.S.; Molloy, M.P.; Packer, N.H.; Paulsen, I.T. Fiber supplements derived from sugarcane stem, wheat dextrin and psyllium husk have different in vitro effects on the human gut microbiota. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef]
- Xiao, L.; Yang, B.; Liu, X.; Luo, Y.; Ji, Q.; Wen, Z.; Liu, Z.; Yang, P.-C. Kinetic changes of intestinal microbiota in the course of intestinal sensitization. Oncotarget 2016, 7, 81197–81207. [Google Scholar] [CrossRef] [PubMed]
- Noval Rivas, M.; Burton, O.T.; Wise, P.; Zhang, Y.Q.; Hobson, S.A.; Garcia Lloret, M.; Chehoud, C.; Kuczynski, J.; Desantis, T.; Warrington, J.; et al. A microbiota signature associated with experimental food allergy promotes allergic sensitization and anaphylaxis. J. Allergy Clin. Immunol. 2013, 131, 201–212. [Google Scholar] [CrossRef]
- Hiippala, K.; Kainulainen, V.; Kalliomäki, M.; Arkkila, P.; Satokari, R. Mucosal prevalence and interactions with the epithelium indicate commensalism of Sutterella spp. Front. Microbiol. 2016, 7, 1706. [Google Scholar] [CrossRef] [PubMed]
- Fujigawa, K.; Nakao, I.; Changko, I.; Nieko, H. Report of the Food Research Institute and the Fermentation & Food Experimental Station; Kagawa Prefectural Government: Kagawa, Japan, 1999; ISSN 13465236.

© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, K.; Xie, K.; Liu, Z.; Nakasone, Y.; Sakao, K.; Hossain, M.A.; Hou, D.-X. Preventive Effects and Mechanisms of Garlic on Dyslipidemia and Gut Microbiome Dysbiosis. Nutrients 2019, 11, 1225. https://doi.org/10.3390/nu11061225
Chen K, Xie K, Liu Z, Nakasone Y, Sakao K, Hossain MA, Hou D-X. Preventive Effects and Mechanisms of Garlic on Dyslipidemia and Gut Microbiome Dysbiosis. Nutrients. 2019; 11(6):1225. https://doi.org/10.3390/nu11061225
Chicago/Turabian StyleChen, Keyu, Kun Xie, Zhuying Liu, Yasushi Nakasone, Kozue Sakao, Md. Amzad Hossain, and De-Xing Hou. 2019. "Preventive Effects and Mechanisms of Garlic on Dyslipidemia and Gut Microbiome Dysbiosis" Nutrients 11, no. 6: 1225. https://doi.org/10.3390/nu11061225
APA StyleChen, K., Xie, K., Liu, Z., Nakasone, Y., Sakao, K., Hossain, M. A., & Hou, D.-X. (2019). Preventive Effects and Mechanisms of Garlic on Dyslipidemia and Gut Microbiome Dysbiosis. Nutrients, 11(6), 1225. https://doi.org/10.3390/nu11061225






