Health-Promoting Components in Fermented Foods: An Up-to-Date Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Including and Excluding Criteria, Paper Selection and Software
3. Results and Discussion
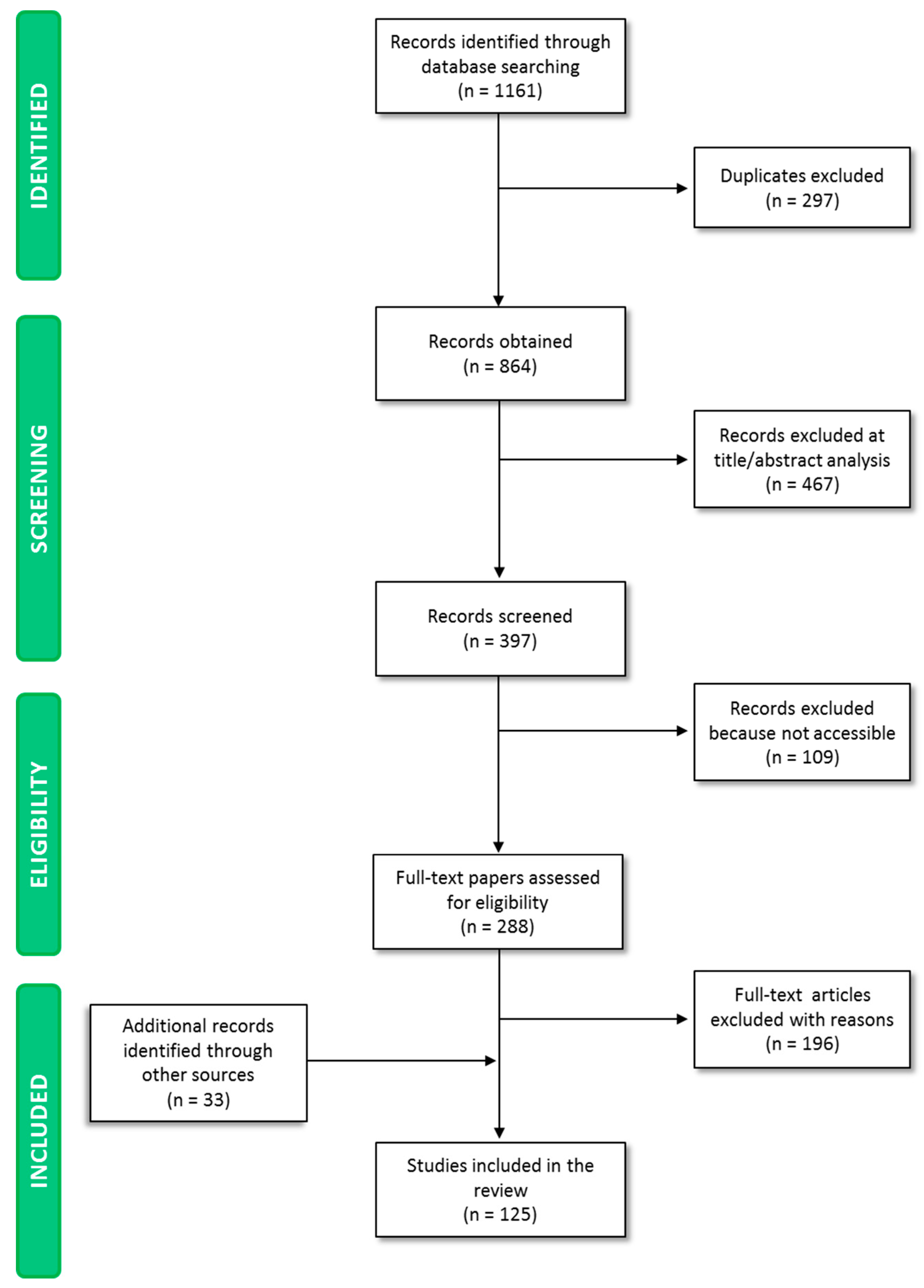
3.1. Literature Search Results
3.2. Health-Promoting Components and Activity Thereof in Fermented Foods
3.2.1. Fermented Milks
Antioxidant Compounds
Anti-Hypertensive Components
Vitamin Content
Improved Protein Digestibility
Alleviation of Lactose Intolerance
Probiotic Activity
3.2.2. Fermented Grain-Based Products
Antioxidant Compounds
Anti-Hypertensive Components
Anti-Diabetic Properties
Vitamin Content
Improved Protein Digestibility
FODMAP Reduction
3.2.3. Fermented Fruit and Vegetables
Antioxidant Compounds
Probiotic Activity
Vitamin Content
Protein Content
3.2.4. Fermented Meat and Fish
Antioxidant Compounds
Fatty Acid Profile
3.3. Fermented Foods, Gut Microbiota and Well-Being
3.4. Fermented Foods in National Dietary Guidelines
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Frías, J.; Martínez-Villaluenga, C.; Peñas, E. Fermented Foods in Health and Disease Prevention, 1st ed.; Academic Press: Boston, MA, USA, 2016. [Google Scholar]
- Kabak, B.; Dobson, A.D.W. An Introduction to the Traditional Fermented Foods and Beverages of Turkey. Crit. Rev. Food Sci. Nutr. 2011, 51, 248–260. [Google Scholar] [CrossRef] [PubMed]
- Joshi, V.K. (Ed.) Indigenous Fermented Foods of South Asia, 1st ed.; CRC Press: Boca Raton, FL, USA, 2016; p. 886. [Google Scholar]
- Marco, M.L.; Heeney, D.; Binda, S.; Cifelli, C.J.; Cotter, P.D.; Foligné, B.; Gänzle, M.; Kort, R.; Pasin, G.; Pihlanto, A.; et al. Health Benefits of Fermented Foods: Microbiota and Beyond. Curr. Opin. Biotechnol. 2017, 44, 94–102. [Google Scholar] [CrossRef]
- Fardet, A.; Rock, E. In vitro and in vivo Antioxidant Potential of Milks, Yoghurts, Fermented Milks and Cheeses: A Narrative Review of Evidence. Nutr. Res. Rev. 2018, 31, 52–70. [Google Scholar] [CrossRef] [PubMed]
- Pessione, E.; Cirrincione, S. Bioactive Molecules Released in Food by Lactic Acid Bacteria: Encrypted Peptides and Biogenic Amines. Front. Microbiol. 2016, 7, 876. [Google Scholar] [CrossRef] [PubMed]
- Rahman, U.; Khan, M.I.; Sohaib, M.; Sahar, A.; Ishaq, A. Exploiting Microorganisms to Develop Improved Functional Meat Sausages: A Review. Food Rev. Int. 2017, 33, 195–215. [Google Scholar] [CrossRef]
- Linares, D.M.; Gómez, C.; Renes, E.; Fresno, J.M.; Tornadijo, M.E.; Ross, R.P.; Stanton, C. Lactic Acid Bacteria and Bifidobacteria with Potential to Design Natural Biofunctional Health-Promoting Dairy Foods. Front. Microbiol. 2017, 8, 846. [Google Scholar] [CrossRef] [PubMed]
- Ogunremi, O.R.; Banwo, K.; Sanni, A.I. Starter-Culture to Improve the Quality of Cereal-based Fermented Foods: Trends in Selection and Application. Curr. Opin. Food Sci. 2017, 13, 38–43. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred Reporting Items for Systematic Reviews and Meta-analyses: The PRISMA Statement. Ann. Intern. Med. 2009, 151, 264–269. [Google Scholar] [CrossRef]
- Scopus. Available online: https://www.scopus.com/search/form.uri?display=basic (accessed on 27 May 2019).
- Sivamaruthi, B.S.; Kesika, P.; Prasanth, M.I.; Chaiyasut, C. A Mini Review on Antidiabetic Properties of Fermented Foods. Nutrients 2018, 10, 1973. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Hwang, C.E.; Cho, E.J.; Song, Y.H.; Kim, S.C.; Cho, K.M. Improvement of Nutritional Components and in vitro Antioxidative Properties of Soy-powder Yogurts using Lactobacillus plantarum. J. Food Drug Anal. 2018, 26, 1054–1065. [Google Scholar] [CrossRef]
- Lim, S.M. Microbiological, Physicochemical, and Antioxidant Properties of Plain Yogurt and Soy Yogurt. Korean J. Microbiol. 2013, 49, 403–414. [Google Scholar] [CrossRef]
- Tavakoli, M.; Habibi Najafi, M.B.; Mohebbi, M. Effect of the Milk Fat Content and Starter Culture Selection on Proteolysis and Antioxidant Activity of Probiotic Yogurt. Heliyon 2019, 5, e01204. [Google Scholar] [CrossRef] [PubMed]
- Solieri, L.; Rutella, G.S.; Tagliazucchi, D. Impact of Non-Starter Lactobacilli on Release of Peptides with Angiotensin-converting Enzyme Inhibitory and Antioxidant Activities During Bovine Milk Fermentation. Food Microbiol. 2015, 51, 108–116. [Google Scholar] [CrossRef] [PubMed]
- Qian, B.; Xing, M.; Cui, L.; Deng, Y.; Xu, Y.; Huang, M.; Zhang, S. Antioxidant, Antihypertensive, and Immunomodulatory Activities of Peptide Fractions from Fermented Skim Milk with Lactobacillus delbrueckii ssp. bulgaricus LB340. J. Dairy Res. 2011, 78, 72–79. [Google Scholar] [CrossRef]
- Widodo, K.; Sukarno, A.S.; Estancia, K.; Widianto, D. Conjugated Linoleic Acid Synthesis in Milk Fermented with Lactobacillus casei strain AG. Pak. J. Nutr. 2018, 17, 524–529. [Google Scholar]
- Tidona, F.; Meucci, A.; Povolo, M.; Pelizzola, V.; Zago, M.; Contarini, G.; Carminati, D.; Giraffa, G. Applicability of Lactococcus hircilactis and Lactococcus laudensis as Dairy Cultures. Int. J. Food Microbiol. 2018, 271, 1–7. [Google Scholar] [CrossRef]
- Gholamhosseinpour, A.; Hashemi, S.M.B. Ultrasound Pretreatment of Fermented Milk Containing Probiotic Lactobacillus plantarum AF1: Carbohydrate Metabolism and Antioxidant Activity. J. Food Process Eng. 2019, 42, e12930. [Google Scholar] [CrossRef]
- Beltrán-Barrientos, L.M.; González-Córdova, A.F.; Hernández-Mendoza, A.; Torres-Inguanzo, E.H.; Astiazarán-García, H.; Esparza-Romero, J.; Vallejo-Cordoba, B. Randomized Double-blind Controlled Clinical Trial of the Blood Pressure-lowering Effect of Fermented Milk with Lactococcus lactis: A Pilot Study. J. Dairy Sci. 2018, 101, 2819–2825. [Google Scholar] [CrossRef]
- Beltrán-Barrientos, L.M.; Hernández-Mendoza, A.; González-Córdova, A.F.; Astiazarán-García, H.; Esparza-Romero, J.; Vallejo-Córdoba, B. Mechanistic Pathways Underlying the Antihypertensive Effect of Fermented Milk with Lactococcus lactis NRRL B-50571 in Spontaneously Hypertensive Rats. Nutrients 2018, 10, 262. [Google Scholar] [CrossRef] [PubMed]
- Nejati, F.; Rizzello, C.G.; Di Cagno, R.; Sheikh-Zeinoddin, M.; Diviccaro, A.; Minervini, F.; Gobbetti, M. Manufacture of a Functional Fermented Milk Enriched of Angiotensin-I Converting Enzyme (ACE)-Inhibitory Peptides and γ-Amino Butyric Acid (GABA). LWT Food Sci. Technol. 2013, 51, 183–189. [Google Scholar] [CrossRef]
- Chen, L.; Alcazar, J.; Yang, T.; Lu, Z.; Lu, Y. Optimized Cultural Conditions of Functional Yogurt for γ-Aminobutyric Acid Augmentation Using Response Surface Methodology. J. Dairy Sci. 2018, 101, 10685–10693. [Google Scholar] [CrossRef]
- Revuelta, J.L.; Serrano-Amatriain, C.; Ledesma-Amaro, R.; Jiménez, A. Formation of Folates by Microorganisms: Towards the Biotechnological Production of this Vitamin. Appl. Microbiol. Biotechnol. 2018, 102, 8613–8620. [Google Scholar] [CrossRef] [PubMed]
- Saubade, F.; Hemery, Y.M.; Rochette, I.; Guyot, J.-P.; Humblot, C. Influence of Fermentation and Other Processing Steps on the Folate Content of a Traditional African Cereal-based Fermented Food. Int. J. Food Microbiol. 2018, 266, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Wouters, J.T.M.; Ayad, E.H.E.; Hugenholtz, J.; Smit, G. Microbes from Raw Milk for Fermented Dairy Products. Int. Dairy J. 2002, 12, 91–109. [Google Scholar] [CrossRef]
- Fu, X.; Harshman, S.G.; Shen, X.; Haytowitz, D.B.; Karl, J.P.; Wolfe, B.E.; Booth, S.L. Multiple Vitamin K Forms Exist in Dairy Foods. Curr. Dev. Nutr. 2017, 1, e000638. [Google Scholar] [CrossRef]
- Lorusso, A.; Coda, R.; Montemurro, M.; Rizzello, C.G. Use of Selected Lactic Acid Bacteria and Quinoa Flour for Manufacturing Novel Yogurt-Like Beverages. Foods 2018, 7, 51. [Google Scholar] [CrossRef]
- Perna, A.; Intaglietta, I.; Simonetti, A.; Gambacorta, E. Donkey Milk for Manufacture of Novel Functional Fermented Beverages. J. Food Sci. 2015, 80, S1352–S1359. [Google Scholar] [CrossRef]
- Champagne, C.P.; Raymond, Y.; Guertin, N.; Martoni, C.J.; Jones, M.L. Growth of Lactobacillus reuteri NCIMB 30242 During Yogurt Fermentation and Bile Salt Hydrolysis Activity in the Product. Dairy Sci. Technol. 2016, 96, 173–184. [Google Scholar] [CrossRef]
- Angelov, A.; Yaneva-Marinova, T.; Gotcheva, V. Oats as a Matrix of Choice for Developing Fermented Functional Beverages. J. Food Sci. Technol. 2018, 55, 2351–2360. [Google Scholar] [CrossRef]
- Joshi, S.; Biswas, K. Antioxidants in Fermented Foods. In Health Benefits of Fermented Foods and Beverages, 1st ed.; Tamang, J.P., Ed.; CRC Press: Boca Raton, FL, USA, 2015; pp. 553–565. [Google Scholar]
- Khan, M.K.; Karnpanit, W.; Nasar-Abbas, S.M.; Huma, Z.-E.; Jayasena, V. Development of a Fermented Product with Higher Phenolic Compounds and Lower Anti-nutritional Factors from Germinated Lupin (Lupinus. angustifolius L.). J. Food Process. Preserv. 2018, 42, e13843. [Google Scholar] [CrossRef]
- Habibi Najafi, M.B.; Fatemizadeh, S.S.; Tavakoli, M. Release of Proteolysis Products with ACE-Inhibitory and Antioxidant Activities in Probiotic Yogurt Containing Different Levels of Fat and Prebiotics. Int. J. Pept. Res. Ther. 2019, 25, 367–377. [Google Scholar] [CrossRef]
- Ramesh, V.; Kumar, R.; Singh, R.R.B.; Kaushik, J.K.; Mann, B. Comparative Evaluation of Selected Strains of Lactobacilli for the Development of Antioxidant Activity in Milk. Dairy Sci. Technol. 2012, 92, 179–188. [Google Scholar] [CrossRef]
- Behera, S.S.; Ray, R.C.; Zdolec, N. Lactobacillus plantarum with Functional Properties: An Approach to Increase Safety and Shelf-Life of Fermented Foods. Biomed. Res. Int. 2018, 2018, 9361614. [Google Scholar] [CrossRef]
- Grażyna, C.; Hanna, C.; Adam, A.; Magdalena, B.M. Natural Antioxidants in Milk and Dairy Products. Int. J. Dairy Technol. 2017, 70, 165–178. [Google Scholar] [CrossRef]
- Homayouni Rad, A.; Yari Khosroushahi, A.; Khalili, M.; Jafarzadeh, S. Folate Bio-fortification of Yoghurt and Fermented Milk: A Review. Dairy Sci. Technol. 2016, 96, 427–441. [Google Scholar] [CrossRef]
- Laiño, J.E.; LeBlanc, J.G.; Savoy de Giori, G. Production of Natural Folates by Lactic Acid Bacteria Starter Cultures Isolated from Artisanal Argentinean Yogurts. Can. J. Microbiol. 2012, 58, 581–588. [Google Scholar] [CrossRef]
- Yilmaz-Ersan, L.; Ozcan, T.; Akpinar-Bayizit, A.; Sahin, S. Comparison of Antioxidant Capacity of Cow and Ewe Milk Kefirs. J. Dairy Sci. 2018, 101, 3788–3798. [Google Scholar] [CrossRef]
- Jauhiainen, T.; Korpela, R. Milk Peptides and Blood Pressure. J. Nutr. 2007, 137, 825–829. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Figueroa, J.C.; González-Córdova, A.F.; Astiazaran-García, H.; Vallejo-Cordoba, B. Hypotensive and heart rate-lowering effects in rats receiving milk fermented by specific Lactococcus lactis strains. Br. J. Nutr. 2013, 109, 827–833. [Google Scholar] [CrossRef]
- Rodríguez-Figueroa, J.C.; González-Córdova, A.F.; Astiazaran-García, H.; Hernández-Mendoza, A.; Vallejo-Cordoba, B. Antihypertensive and hypolipidemic effect of milk fermented by specific Lactococcus lactis strains. J. Dairy Sci. 2013, 96, 4094–4099. [Google Scholar] [CrossRef]
- Rossi, M.; Raimondi, S.; Costantino, L.; Amaretti, A. Folate: Relevance of Chemical and Microbial Production. In Industrial Biotechnology of Vitamins, Biopigments, and Antioxidants; Wiley-VCH Verlag GmbH & Co.: Weinheim, Germany, 2016; pp. 103–128. [Google Scholar]
- Saini, R.K.; Nile, S.H.; Keum, Y.-S. Folates: Chemistry, Analysis, Occurrence, Biofortification and Bioavailability. Food Res. Int. 2016, 89, 1–13. [Google Scholar] [CrossRef]
- LeBlanc, J.G.; Laiño, J.E.; del Valle, M.J.; Vannini, V.; van Sinderen, D.; Taranto, M.P.; de Valdez, G.F.; de Giori, G.S.; Sesma, F. B-Group Vitamin Production by Lactic Acid Bacteria—Current Knowledge and Potential Applications. J. Appl. Microbiol. 2011, 111, 1297–1309. [Google Scholar] [CrossRef]
- Manoury, E.; Jourdon, K.; Boyaval, P.; Fourcassié, P. Quantitative Measurement of Vitamin K2 (menaquinones) in Various Fermented Dairy Products Using a Reliable High-Performance Liquid Chromatography Method. J. Dairy Sci. 2013, 96, 1335–1346. [Google Scholar] [CrossRef] [PubMed]
- Walther, B.; Karl, J.P.; Booth, S.L.; Boyaval, P. Menaquinones, Bacteria, and the Food Supply: The Relevance of Dairy and Fermented Food Products to Vitamin K Requirements123. Adv. Nutr. 2013, 4, 463–473. [Google Scholar] [CrossRef]
- Zironi, E.; Gazzotti, T.; Barbarossa, A.; Farabegoli, F.; Serraino, A.; Pagliuca, G. Determination of Vitamin B12 in Dairy Products by Ultra Performance Liquid Chromatography-Tandem Mass Spectrometry. Ital. J. Food Saf. 2014, 3, 4513. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, D.; Chattoraj, D.K.; Chattopadhyay, P. Studies on Changes in Microstructure and Proteolysis in Cow and Soy Milk Curd During Fermentation Using Lactic Cultures For Improving Protein Bioavailability. J. Food Sci. Technol. 2013, 50, 979–985. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Misselwitz, B.; Dai, N.; Fox, M. Lactose Intolerance in Adults: Biological Mechanism and Dietary Management. Nutrients 2015, 7, 8020–8035. [Google Scholar] [CrossRef] [PubMed]
- FAO/WHO. Health and Nutritional Properties of Probiotics in Food Including Powder Milk with Live Lactic Acid Bacteria – Joint FAO/WHO Expert Consultation. Available online: Health_and_Nutritional_Properties_of_Probiotics_in_Food_Including_Powder_Milk_with_Live_Lactic_Acid_Bacteria_-_Joint_FAOWHO_Expert_Consultation (accessed on 31 March 2019).
- Bull, M.; Plummer, S.; Marchesi, J.; Mahenthiralingam, E. The Life History of Lactobacillus acidophilus as a Probiotic: a Tale of Revisionary Taxonomy, Misidentification and Commercial Success. FEMS Microbiol. Lett. 2013, 349, 77–87. [Google Scholar] [CrossRef]
- Sharma, H.K.; Panesar, P.S. Technologies in Food Processing, 1st ed.; Apple Academic Press: Palm Bay, FL, USA, 2018. [Google Scholar]
- Blandino, A.; Al-Aseeri, M.E.; Pandiella, S.S.; Cantero, D.; Webb, C. Cereal-based Fermented Foods and Beverages. Food Res. Int. 2003, 36, 527–543. [Google Scholar] [CrossRef]
- Rocchetti, G.; Miragoli, F.; Zacconi, C.; Lucini, L.; Rebecchi, A. Impact of Cooking and Fermentation by Lactic Acid Bacteria on Phenolic Profile of Quinoa and Buckwheat Seeds. Food Res. Int. 2019, 119, 886–894. [Google Scholar] [CrossRef]
- Đorđević, T.M.; Šiler-Marinković, S.S.; Dimitrijević-Branković, S.I. Effect of Fermentation on Antioxidant Properties of Some Cereals and Pseudo Cereals. Food Chem. 2010, 119, 957–963. [Google Scholar] [CrossRef]
- Lorusso, A.; Verni, M.; Montemurro, M.; Coda, R.; Gobbetti, M.; Rizzello, C.G. Use of Fermented Quinoa Flour for Pasta Making and Evaluation of the Technological and Nutritional Features. LWT Food Sci. Technol. 2017, 78, 215–221. [Google Scholar] [CrossRef]
- Villalva, M.F.A.; González-Aguilar, G.; Sández, O.R.; García, H.A.; Osuna, A.I.L.; López-Ahumada, G.A.; Robles-Sánchez, R.M. Bioprocessing of Wheat (Triticum. aestivum cv. Kronstad) Bran from Northwest Mexico: Effects on Ferulic Acid Bioaccessibility in Breads. CyTA J. Food 2018, 16, 570–579. [Google Scholar] [CrossRef]
- Rashid, N.Y.A.; Razak, D.L.A.; Jamaluddin, A.; Sharifuddin, S.A.; Long, K. Bioactive Compounds and Antioxidant Activity of Rice Bran Fermented with Lactic Acid Bacteria. Malaysian J. Microbiol. 2015, 11, 156–162. [Google Scholar]
- Ryan, E.P.; Heuberger, A.L.; Weir, T.L.; Barnett, B.; Broeckling, C.D.; Prenni, J.E. Rice Bran Fermented with Saccharomyces boulardii Generates Novel Metabolite Profiles with Bioactivity. J. Agric. Food Chem. 2011, 59, 1862–1870. [Google Scholar] [CrossRef]
- Peñas, E.; Diana, M.; Frias, J.; Quílez, J.; Martínez-Villaluenga, C. A Multistrategic Approach in the Development of Sourdough Bread Targeted Towards Blood Pressure Reduction. Plant. Foods Hum. Nutr. 2015, 70, 97–103. [Google Scholar] [CrossRef] [PubMed]
- Curiel, J.A.; Coda, R.; Centomani, I.; Summo, C.; Gobbetti, M.; Rizzello, C.G. Exploitation of the Nutritional and Functional Characteristics of Traditional Italian Legumes: The Potential of Sourdough Fermentation. Int. J. Food Microbiol. 2015, 196, 51–61. [Google Scholar] [CrossRef]
- Rodríguez, L.R.; Pingitore, E.V.; Rollan, G.; Cocconcelli, P.S.; Fontana, C.; Saavedra, L.; Vignolo, G.; Hebert, E.M. Biodiversity and Technological-functional Potential of Lactic Acid Bacteria Isolated from Spontaneously Fermented Quinoa Sourdoughs. J. Appl. Microbiol. 2016, 120, 1289–1301. [Google Scholar] [CrossRef]
- Gobbetti, M.; Rizzello, C.G.; Di Cagno, R.; De Angelis, M. How the Sourdough May Affect the Functional Features of Leavened Baked Goods. Food Microbiol. 2014, 37, 30–40. [Google Scholar] [CrossRef]
- Bautista-Expósito, S.; Peñas, E.; Silván, J.M.; Frias, J.; Martínez-Villaluenga, C. pH-controlled fermentation in mild alkaline conditions enhances bioactive compounds and functional features of lentil to ameliorate metabolic disturbances. Food Chem. 2018, 248, 262–271. [Google Scholar] [CrossRef]
- Signorini, C.; Carpen, A.; Coletto, L.; Borgonovo, G.; Galanti, E.; Capraro, J.; Magni, C.; Abate, A.; Johnson, S.K.; Duranti, M.; et al. Enhanced Vitamin B12 Production in an Innovative Lupin Tempeh is due to Synergic Effects of Rhizopus. and Propionibacterium. in Cofermentation. Int. J. Food Sci. Nutr. 2018, 69, 451–457. [Google Scholar] [CrossRef]
- Wolkers – Rooijackers, J.C.M.; Endika, M.F.; Smid, E.J. Enhancing Vitamin B12 in Lupin Tempeh by in situ Fortification. LWT 2018, 96, 513–518. [Google Scholar] [CrossRef]
- Chamlagain, B.; Sugito, T.A.; Deptula, P.; Edelmann, M.; Kariluoto, S.; Varmanen, P.; Piironen, V. In situ Production of Active Vitamin B12 in Cereal Matrices Using Propionibacterium freudenreichii. Food Sci. Nutr. 2018, 6, 67–76. [Google Scholar] [CrossRef]
- Tarvainen, M.; Fabritius, M.; Yang, B. Determination of Vitamin K Composition of Fermented Food. Food Chem. 2019, 275, 515–522. [Google Scholar] [CrossRef]
- Çabuk, B.; Nosworthy, M.G.; Stone, A.K.; Korber, D.R.; Tanaka, T.; House, J.D.; Nickerson, M.T. Effect of Fermentation on the Protein Digestibility and Levels of Non-Nutritive Compounds of Pea Protein Concentrate. Food Technol. Biotechnol. 2018, 56, 257–264. [Google Scholar] [CrossRef] [PubMed]
- Montemurro, M.; Pontonio, E.; Gobbetti, M.; Rizzello, C.G. Investigation of the Nutritional, Functional and Technological Effects of the Sourdough Fermentation of Sprouted Flours. Int. J. Food Microbiol. 2018. [Google Scholar] [CrossRef]
- Struyf, N.; Verspreet, J.; Courtin, C.M. FODMAP Reduction in Yeast-Leavened Whole Wheat Bread. Available online: https://www.aaccnet.org/publications/cfw/2018/jul-aug/Pages/CFW-63-4-0152.aspx (accessed on 31 March 2019).
- Struyf, N.; Laurent, J.; Verspreet, J.; Verstrepen, K.J.; Courtin, C.M. Saccharomyces cerevisiae and Kluyveromyces. marxianus Cocultures Allow Reduction of Fermentable Oligo-, Di-, and Monosaccharides and Polyols Levels in Whole Wheat Bread. J. Agric. Food Chem. 2017, 65, 8704–8713. [Google Scholar] [CrossRef] [PubMed]
- Struyf, N.; Vandewiele, H.; Herrera-Malaver, B.; Verspreet, J.; Verstrepen, K.J.; Courtin, C.M. Kluyveromyces. marxianus Yeast Enables the Production of Low FODMAP Whole Wheat Breads. Food Microbiol. 2018, 76, 135–145. [Google Scholar] [CrossRef]
- Fraberger, V.; Call, L.-M.; Domig, K.J.; D’Amico, S. Applicability of Yeast Fermentation to Reduce Fructans and Other FODMAPs. Nutrients 2018, 10, 1247. [Google Scholar] [CrossRef]
- Menezes, L.A.A.; Minervini, F.; Filannino, P.; Sardaro, M.L.S.; Gatti, M.; Lindner, J.D.D. Effects of Sourdough on FODMAPs in Bread and Potential Outcomes on Irritable Bowel Syndrome Patients and Healthy Subjects. Front. Microbiol. 2018, 9, 1972. [Google Scholar] [CrossRef]
- Kadiri, O. A Review on the Status of the Phenolic Compounds and Antioxidant Capacity of the Flour: Effects of Cereal Processing. Int. J. Food Prop. 2017, 20, S798–S809. [Google Scholar] [CrossRef]
- Hole, A.S.; Rud, I.; Grimmer, S.; Sigl, S.; Narvhus, J.; Sahlstrøm, S. Improved Bioavailability of Dietary Phenolic Acids in Whole Grain Barley and Oat Groat Following Fermentation with Probiotic Lactobacillus acidophilus, Lactobacillus johnsonii, and Lactobacillus reuteri. J. Agric. Food Chem. 2012, 60, 6369–6375. [Google Scholar] [CrossRef]
- Nkhata, S.G.; Ayua, E.; Kamau, E.H.; Shingiro, J. Fermentation and Germination Improve Nutritional Value of Cereals and Legumes through Activation of Endogenous Enzymes. Food Sci. Nutr. 2018, 6, 2446–2458. [Google Scholar] [CrossRef] [PubMed]
- Galli, V.; Mazzoli, L.; Luti, S.; Venturi, M.; Guerrini, S.; Paoli, P.; Vincenzini, M.; Granchi, L.; Pazzagli, L. Effect of Selected Strains of Lactobacilli on the Antioxidant and Anti-inflammatory Properties of Sourdough. Int. J. Food Microbiol. 2018, 286, 55–65. [Google Scholar] [CrossRef]
- Gobbetti, M.; De Angelis, M.; Di Cagno, R.; Calasso, M.; Archetti, G.; Rizzello, C.G. Novel Insights on the Functional/Nutritional Features of the Sourdough Fermentation. Int. J. Food Microbiol. 2018. [Google Scholar] [CrossRef] [PubMed]
- Harvard School. Health Information and Medical Information. Available online: https://www.health.harvard.edu/ (accessed on 9 May 2019).
- Kariluoto, S.; Edelmann, M.; Nyström, L.; Sontag-Strohm, T.; Salovaara, H.; Kivelä, R.; Herranen, M.; Korhola, M.; Piironen, V. In situ Enrichment of Folate by Microorganisms in Beta-Glucan Rich Oat and Barley Matrices. Int. J. Food Microbiol. 2014, 176, 38–48. [Google Scholar] [CrossRef]
- Saubade, F.; Hemery, Y.M.; Guyot, J.-P.; Humblot, C. Lactic Acid Fermentation as a Tool for Increasing the Folate Content of Foods. Crit. Rev. Food Sci. Nutr. 2017, 57, 3894–3910. [Google Scholar] [CrossRef]
- Laurent-Babot, C.; Guyot, J.-P. Should Research on the Nutritional Potential and Health Benefits of Fermented Cereals Focus More on the General Health Status of Populations in Developing Countries? Microorganisms 2017, 5, 40. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.H. Health-Promoting Components of Fruits and Vegetables in the Diet12. Adv. Nutr. 2013, 4, 384S–392S. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhou, J.; Fan, L.; Qin, Z.; Chen, Q.; Zhao, L. Antioxidant properties of a vegetable-fruit beverage fermented with two Lactobacillus plantarum strains. Food Sci. Biotechnol. 2018, 27, 1719–1726. [Google Scholar] [CrossRef]
- Kaprasob, R.; Kerdchoechuen, O.; Laohakunjit, N.; Thumthanaruk, B.; Shetty, K. Changes in physico-chemical, astringency, volatile compounds and antioxidant activity of fresh and concentrated cashew apple juice fermented with Lactobacillus plantarum. J. Food Sci. Technol. 2018, 55, 3979–3990. [Google Scholar] [CrossRef]
- Li, Z.; Teng, J.; Lyu, Y.; Hu, X.; Zhao, Y.; Wang, M. Enhanced Antioxidant Activity for Apple Juice Fermented with Lactobacillus plantarum ATCC14917. Molecules 2019, 24, 51. [Google Scholar] [CrossRef]
- Mantzourani, I.; Terpou, A.; Alexopoulos, A.; Kimbaris, A.; Bezirtzoglou, E.; Koutinas, A.A.; Plessas, S. Production of a Potentially Symbiotic Pomegranate Beverage by Fermentation with Lactobacillus plantarum ATCC 14917 Adsorbed on a Prebiotic Carrier. Appl. Biochem. Biotechnol. 2019. [Google Scholar] [CrossRef]
- Zhang, Z.-P.; Ma, J.; He, Y.-Y.; Lu, J.; Ren, D.-F. Antioxidant and Hypoglycemic Effects of Diospyros lotus Fruit Fermented with Microbacterium flavum and Lactobacillus plantarum. J. Biosci. Bioeng. 2018, 125, 682–687. [Google Scholar] [CrossRef] [PubMed]
- Sirilun, S.; Sivamaruthi, B.S.; Kesika, P.; Peerajan, S.; Chaiyasut, C. Lactobacillus paracasei HII01 Mediated Fermentation of Syzygium cumini L. Fruits: Assessment of Changes in Phenolic Content and Antioxidant Capacity. Asian J. Pharm. Clin. Res. 2018, 11, 304–308. [Google Scholar] [CrossRef]
- Shukla, S.; Park, J.; Park, J.H.; Lee, J.S.; Kim, M. Development of Lotus Root Fermented Sugar Syrup as a Functional Food Supplement/Condiment and Evaluation of its Physicochemical, Nutritional and Microbiological Properties. J. Food Sci. Technol. 2018, 55, 619–629. [Google Scholar] [CrossRef]
- Cusano, E.; Simonato, B.; Consonni, R. Fermentation Process of Apple Juice Investigated by NMR Spectroscopy. LWT Food Sci. and Technol. 2018, 96, 147–151. [Google Scholar] [CrossRef]
- Bah, A.; Ferjani, R.; Fhoula, I.; Gharbi, Y.; Najjari, A.; Boudabous, A.; Ouzari, H.I. Microbial Community Dynamic in Tomato Fruit During Spontaneous Fermentation and Biotechnological Characterization of Indigenous Lactic Acid Bacteria. Ann. Microbiol. 2019, 69, 41–49. [Google Scholar] [CrossRef]
- Wiczkowski, W.; Szawara-Nowak, D.; Romaszko, J. The Impact of Red Cabbage Fermentation on Bioavailability of Anthocyanins and Antioxidant Capacity of Human Plasma. Food Chem. 2016, 190, 730–740. [Google Scholar] [CrossRef] [PubMed]
- Septembre-Malaterre, A.; Remize, F.; Poucheret, P. Fruits and Vegetables, as a Source of Nutritional Compounds and Phytochemicals: Changes in Bioactive Compounds During Lactic Fermentation. Food Res. Int. 2018, 104, 86–99. [Google Scholar] [CrossRef]
- Rios-Corripio, G.; Guerrero-Beltrán, J.Á. Antioxidant and Physicochemical Characteristics of Unfermented and Fermented Pomegranate (Punica granatum L.) Beverages. J. Food Sci. Technol. 2019, 56, 132–139. [Google Scholar] [CrossRef] [PubMed]
- Bujna, E.; Farkas, N.A.; Tran, A.M.; Dam, M.S.; Nguyen, Q.D. Lactic acid Fermentation of Apricot Juice by Mono- and Mixed Cultures of Probiotic Lactobacillus and Bifidobacterium Strains. Food Sci. Biotechnol. 2018, 27, 547–554. [Google Scholar] [CrossRef] [PubMed]
- Oh, M.-R.; Park, S.-H.; Kim, S.-Y.; Back, H.-I.; Kim, M.-G.; Jeon, J.-Y.; Ha, K.-C.; Na, W.-T.; Cha, Y.-S.; Park, B.-H.; et al. Postprandial Glucose-lowering Effects of Fermented Red Ginseng in Subjects with Impaired Fasting Glucose or Type 2 Diabetes: A Randomized, Double-blind, Placebo-controlled Clinical Trial. BMC Complement. Altern. Med. 2014, 14, 237. [Google Scholar] [CrossRef]
- Mustafa, S.M.; Chua, L.S.; El-Enshasy, H.A.; Abd Majid, F.A.; Hanapi, S.Z.; Abdul Malik, R. Effect of Temperature and pH on the Probiotication of Punica granatum Juice using Lactobacillus Species. J. Food Biochem. 2019. [Google Scholar] [CrossRef]
- Xing, L.; Liu, R.; Cao, S.; Zhang, W.; Guanghong, Z. Meat Protein-based Bioactive Peptides and their Potential Functional Activity: A Review. Int. J. of Food Sci. Technol. 2019. [Google Scholar] [CrossRef]
- Kęska, P.; Stadnik, J. Stability of Antiradical Activity of Protein Extracts and Hydrolysates from Dry-Cured Pork Loins with Probiotic Strains of LAB. Nutrients 2018, 10, 521. [Google Scholar] [CrossRef]
- Song, M.-Y.; Van-Ba, H.; Park, W.-S.; Yoo, J.-Y.; Kang, H.-B.; Kim, J.-H.; Kang, S.-M.; Kim, B.-M.; Oh, M.-H.; Ham, J.-S. Quality Characteristics of Functional Fermented Sausages Added with Encapsulated Probiotic Bifidobacterium longum KACC 91563. Korean J. Food Sci. Anim. Resour. 2018, 38, 981–994. [Google Scholar] [CrossRef]
- Pérez-Burillo, S.; Mehta, T.; Pastoriza, S.; Kramer, D.L.; Paliy, O.; Rufián-Henares, J.Á. Potential Probiotic Salami with Dietary Fiber Modulates Antioxidant Capacity, Short Chain Fatty Acid Production and Gut Microbiota Community Structure. LWT Food Sci. Technol. 2019, 105, 355–362. [Google Scholar] [CrossRef]
- Xu, Y.; Li, L.; Xia, W.; Zang, J.; Gao, P. The Role of Microbes in Free Fatty Acids Release and Oxidation in Fermented Fish Paste. LWT Food Sci. Technol. 2019, 101, 323–330. [Google Scholar] [CrossRef]
- WHO. Constitution. Available online: https://www.who.int/about/who-we-are/constitution (accessed on 5 May 2019).
- Rezac, S.; Kok, C.R.; Heermann, M.; Hutkins, R. Fermented Foods as a Dietary Source of Live Organisms. Front. Microbiol. 2018, 9, 1785. [Google Scholar] [CrossRef]
- Bell, V.; Ferrão, J.; Pimentel, L.; Pintado, M.; Fernandes, T. One Health, Fermented Foods and Gut Microbiota. Foods 2018, 7, 195. [Google Scholar] [CrossRef]
- Prasad, K.N.; Bondy, S.C. Dietary Fibers and their Fermented Short-Chain Fatty Acids in Prevention of Human Diseases. Bioact. Carbohydrates Dietary Fibre 2019, 17, 100170. [Google Scholar] [CrossRef]
- McRorie, J.W.; McKeown, N.M. Understanding the Physics of Functional Fibers in the Gastrointestinal Tract: An Evidence-Based Approach to Resolving Enduring Misconceptions about Insoluble and Soluble Fiber. J. Acad. Nutr. Diet. 2017, 117, 251–264. [Google Scholar] [CrossRef]
- Mota de Carvalho, N.; Costa, E.M.; Silva, S.; Pimentel, L.; Fernandes, T.H.; Pintado, M.E. Fermented Foods and Beverages in Human Diet and Their Influence on Gut Microbiota and Health. Fermentation 2018, 4, 90. [Google Scholar] [CrossRef]
- Reis, D.J.; Ilardi, S.S.; Punt, S.E.W. The Anxiolytic Effect of Probiotics: A Systematic Review and Meta-analysis of the Clinical and Preclinical Literature. PLoS ONE 2018, 13, e0199041. [Google Scholar] [CrossRef]
- Wang, H.; Lee, I.-S.; Braun, C.; Enck, P. Effect of Probiotics on Central Nervous System Functions in Animals and Humans: A Systematic Review. J. Neurogastroenterol. Motil. 2016, 22, 589–605. [Google Scholar] [CrossRef]
- Aslam, H.; Green, J.; Jacka, F.N.; Collier, F.; Berk, M.; Pasco, J.; Dawson, S.L. Fermented Foods, the Gut and Mental Health: A Mechanistic Overview with Implications for Depression and Anxiety. Nutr. Neurosci. 2018, 11, 1–13. [Google Scholar] [CrossRef]
- Kim, B.; Hong, V.M.; Yang, J.; Hyun, H.; Im, J.J.; Hwang, J.; Yoon, S.; Kim, J.E. A Review of Fermented Foods with Beneficial Effects on Brain and Cognitive Function. Prev. Nutr. Food Sci. 2016, 21, 297–309. [Google Scholar] [CrossRef]
- Giau, V.V.; Wu, S.Y.; Jamerlan, A.; An, S.S.A.; Kim, S.; Hulme, J. Gut Microbiota and Their Neuroinflammatory Implications in Alzheimer’s Disease. Nutrients 2018, 10, 1765. [Google Scholar] [CrossRef]
- Selhub, E.M.; Logan, A.C.; Bested, A.C. Fermented Foods, Microbiota, and Mental Health: Ancient Practice Meets Nutritional Psychiatry. J. Physiol. Anthropol. 2014, 33, 2. [Google Scholar] [CrossRef]
- Akbari, E.; Asemi, Z.; Daneshvar Kakhaki, R.; Bahmani, F.; Kouchaki, E.; Tamtaji, O.R.; Hamidi, G.A.; Salami, M. Effect of Probiotic Supplementation on Cognitive Function and Metabolic Status in Alzheimer’s Disease: A Randomized, Double-Blind and Controlled Trial. Front. Aging Neurosci. 2016, 8, 256. [Google Scholar] [CrossRef]
- Tseng, W.-T.; Hsu, Y.-W.; Pan, T.-M. The Ameliorative Effect of Monascus purpureus NTU 568-fermented Rice Extracts on 6-Hydroxydopamine-induced Neurotoxicity in SH-SY5Y Cells and the Rat Model of Parkinson’s Disease. Food Funct. 2016, 7, 752–762. [Google Scholar] [CrossRef]
- Tseng, W.-T.; Hsu, Y.-W.; Pan, T.-M. Neuroprotective Effects of Dimerumic Acid and Deferricoprogen from Monascus purpureus NTU 568-fermented Rice against 6-Hydroxydopamine-induced Oxidative Stress and Apoptosis in Differentiated Pheochromocytoma PC-12 Cells. Pharm. Biol. 2016, 54, 1434–1444. [Google Scholar] [CrossRef]
- Sivamaruthi, B.S.; Kesika, P.; Chaiyasut, C. Impact of Fermented Foods on Human Cognitive Function—A Review of Outcome of Clinical Trials. Sci. Pharm. 2018, 86, 22. [Google Scholar] [CrossRef] [PubMed]
- EFSA. Scientific Opinion on establishing Food-Based Dietary Guidelines. EFSA J. 2010, 8, 1460. [Google Scholar]
- Bell, V.; Ferrão, J.; Fernandes, T. Nutritional Guidelines and Fermented Food Frameworks. Foods 2017, 6, 65. [Google Scholar] [CrossRef] [PubMed]
| Query String | No. of Documents Found |
|---|---|
| (TITLE-ABS-KEY (fermented AND food) OR TITLE-ABS-KEY (fermentation) AND TITLE-ABS-KEY (healthy AND component) OR TITLE-ABS-KEY (health AND benefit) AND TITLE-ABS-KEY (milk)) AND DOCTYPE (ar) AND PUBYEAR > 2016 | 58 |
| (TITLE-ABS-KEY (fermented AND food) OR TITLE-ABS-KEY (fermentation) AND TITLE-ABS-KEY (healthy AND component) OR TITLE-ABS-KEY (health AND benefit) AND TITLE-ABS-KEY (cereals)) AND DOCTYPE (ar) AND PUBYEAR > 2016 | 16 |
| (TITLE-ABS-KEY (fermented AND food) OR TITLE-ABS-KEY (fermentation) AND TITLE-ABS-KEY (healthy AND component) OR TITLE-ABS-KEY (health AND benefit) AND TITLE-ABS-KEY (pulses)) AND DOCTYPE (ar) AND PUBYEAR > 2016 | 1 |
| (TITLE-ABS-KEY (fermented AND food) OR TITLE-ABS-KEY (fermentation) AND TITLE-ABS-KEY (healthy AND component) OR TITLE-ABS-KEY (health AND benefit) AND TITLE-ABS-KEY (legumes)) AND DOCTYPE (ar) AND PUBYEAR > 2016 | 5 |
| (TITLE-ABS-KEY (fermented AND food) OR TITLE-ABS-KEY (fermentation) AND TITLE-ABS-KEY (healthy AND component) OR TITLE-ABS-KEY (health AND benefit) AND TITLE-ABS-KEY (grains)) AND DOCTYPE (ar) AND PUBYEAR > 2016 | 27 |
| (TITLE-ABS-KEY (fermented AND food) OR TITLE-ABS-KEY (fermentation) AND TITLE-ABS-KEY (healthy AND component) OR TITLE-ABS-KEY (health AND benefit) AND TITLE-ABS-KEY (meat)) AND DOCTYPE (ar) AND PUBYEAR > 2016 | 11 |
| (TITLE-ABS-KEY (fermented AND food) OR TITLE-ABS-KEY (fermentation) AND TITLE-ABS-KEY (healthy AND component) OR TITLE-ABS-KEY (health AND benefit) AND TITLE-ABS-KEY (fish)) AND DOCTYPE (ar) AND PUBYEAR > 2016 | 9 |
| (TITLE-ABS-KEY (fermented AND food) OR TITLE-ABS-KEY (fermentation) AND TITLE-ABS-KEY (healthy AND component) OR TITLE-ABS-KEY (health AND benefit) AND TITLE-ABS-KEY (fruit)) AND DOCTYPE (ar) AND PUBYEAR > 2016 | 39 |
| (TITLE-ABS-KEY (fermented AND food) OR TITLE-ABS-KEY (fermentation) AND TITLE-ABS-KEY (healthy AND component) OR TITLE-ABS-KEY (health AND benefit) AND TITLE-ABS-KEY (vegetable)) AND DOCTYPE (ar) AND PUBYEAR > 2016 | 28 |
| (TITLE-ABS-KEY (fermented AND food) OR TITLE-ABS-KEY (fermentation) AND TITLE-ABS-KEY (antioxidants) AND TITLE-ABS-KEY (milk) OR TITLE-ABS-KEY (cereals) OR TITLE-ABS-KEY (pulses) OR TITLE-ABS-KEY (legumes) OR TITLE-ABS-KEY (grains) OR TITLE-ABS-KEY (meat) OR TITLE-ABS-KEY (fish) OR TITLE-ABS-KEY (fruit) OR TITLE-ABS-KEY (vegetable)) AND DOCTYPE (ar) AND PUBYEAR > 2016 | 383 |
| (TITLE-ABS-KEY (fermented AND food) OR TITLE-ABS-KEY (fermentation) AND TITLE-ABS-KEY (anti-hypertensive) AND TITLE-ABS-KEY (milk) OR TITLE-ABS-KEY (cereals) OR TITLE-ABS-KEY (pulses) OR TITLE-ABS-KEY (legumes) OR TITLE-ABS-KEY (grains) OR TITLE-ABS-KEY (meat) OR TITLE-ABS-KEY (fish) OR TITLE-ABS-KEY (fruit) OR TITLE-ABS-KEY (vegetable)) AND DOCTYPE (ar) AND PUBYEAR > 2016 | 7 |
| (TITLE-ABS-KEY (fermented AND food) OR TITLE-ABS-KEY (fermentation) AND TITLE-ABS-KEY (vitamins) AND TITLE-ABS-KEY (milk) OR TITLE-ABS-KEY (cereals) OR TITLE-ABS-KEY (pulses) OR TITLE-ABS-KEY (legumes) OR TITLE-ABS-KEY (grains) OR TITLE-ABS-KEY (meat) OR TITLE-ABS-KEY (fish) OR TITLE-ABS-KEY (fruit) OR TITLE-ABS-KEY (vegetable)) AND DOCTYPE (ar) AND PUBYEAR > 2016 | 117 |
| (TITLE-ABS-KEY (fermented AND food) OR TITLE-ABS-KEY (fermentation) AND TITLE-ABS-KEY (probiotic) AND TITLE-ABS-KEY (milk) OR TITLE-ABS-KEY (cereals) OR TITLE-ABS-KEY (pulses) OR TITLE-ABS-KEY (legumes) OR TITLE-ABS-KEY (grains) OR TITLE-ABS-KEY (meat) OR TITLE-ABS-KEY (fish) OR TITLE-ABS-KEY (fruit) OR TITLE-ABS-KEY (vegetable)) AND DOCTYPE (ar) AND PUBYEAR > 2016 | 430 |
| (TITLE-ABS-KEY (fermented AND food) OR TITLE-ABS-KEY (fermentation) AND TITLE-ABS-KEY (lactose intolerance) AND TITLE-ABS-KEY (milk) OR TITLE-ABS-KEY (cereals) OR TITLE-ABS-KEY (pulses) OR TITLE-ABS-KEY (legumes) OR TITLE-ABS-KEY (grains) OR TITLE-ABS-KEY (meat) OR TITLE-ABS-KEY (fish) OR TITLE-ABS-KEY (fruit) OR TITLE-ABS-KEY (vegetable)) AND DOCTYPE (ar) AND PUBYEAR > 2016 | 12 |
| (TITLE-ABS-KEY (fermented AND foods) AND TITLE-ABS-KEY (well-being)) AND DOCTYPE (ar) AND PUBYEAR > 2016 | 18 |
| 1161 |
| Health-Promoting Activity | Health-Promoting Compounds | Raw Food Matrices | Fermenting Microorganism(s) | References |
|---|---|---|---|---|
| Antioxidant activity | Phenolic compounds, GABA 1, peptides, CLA 2, folates (vitamin B9) | Goat milk | Pediococcus pentosaceus | [5] |
| Camel milk | Lactobacillus rhamnosus strain PTCC 1637 | [5] | ||
| Milk | Lactobacillus acidophilus strain PC16 | [14] | ||
| Milk | Lb. acidophilus | [15] | ||
| Milk | Lactobacillus casei strain PRA205 | [16] | ||
| Skimmed milk | Lactobacillus delbrueckii spp. bulgaricus strain LB340 | [17] | ||
| Milk | Lb. casei strain AG | [18] | ||
| Milk | Lactococcus hircilactis/Lactococcus laudensis | [19] | ||
| Milk | Lactobacillus plantarum strain AF1 | [20] | ||
| Anti-hypertensive activity | ACE 3 inhibitory peptides and GABA | Skimmed milk | Lactococcus lactis strain NRRL B-50571 | [21,22] |
| Milk | Lactobacillus spp. | [23] | ||
| Milk | Streptococcus salivarius subsp. thermophilus strain fmb5 | [24] | ||
| Increase of vitamin content | Folate (vitamin B9), vitamin K, riboflavin (vitamin B2) | Milk | LAB and Bifidobacteria species | [1,25,26] |
| Milk processed into yogurt | - | [27] | ||
| Milk | Species of the genera Carnobacterium, Enterococcus, Lactobacillus, Lactococcus, Leuconostoc, Oenococcus, Pediococcus, Streptococcus, Tetragenococcus, Vagococcus, and Weissella | [28] | ||
| Improvement of protein digestibility | - | Kefir | Lb. rhamnosus SP1 Weissella confusa DSM 20194 Lb. plantarum | [29] |
| Alleviation of lactose intolerance | Donkey milk | Lb. acidophilus and Lb. casei | [30] | |
| Probiotic activity | Milk | Lactobacillus reuteri | [31] | |
| Oat-based dairy fermented beverages | - | [32] |
| Health-Promoting Activity | Health-Promoting Compounds | Raw Food Matrices | Fermenting Microorganism(s) | References |
|---|---|---|---|---|
| Antioxidant activity | Phenolic compounds, GABA 1, peptides, CLA 2, folates (vitamin B9) | Quinoa and buckwheat | P. pentosaceus and Lactobacillus paracasei | [57] |
| Wheat germ, barley, rye and buckwheat | Lb. rhamnosus and Saccharomyces cerevisiae | [58] | ||
| Quinoa fermented | Lb. plantarum strain T6B10 and Lactobacillus rossiae strain T0B10 | [59] | ||
| Bread supplemented with bioprocessed Bran | Yeast, xylanase enzyme and combination thereof | [60] | ||
| Rice bran | Pediococcus acidilactici, L. lactis and Pediococcus pentoseous | [61] | ||
| Rice bran | Saccharomyces boulardii | [62] | ||
| Bread prepared with wheat sourdough | Lb. brevis CECT 8183 and a commercial protease | [63] | ||
| Sourdough and legume flours | Lb. brevis strain AM7 and Lb. plantarum strain C48 | [64] | ||
| Spontaneous fermented quinoa sourdough | Lb. plantarum strain CRL1905, Leuconostoc mesenteroides strain CRL1907 and Lb. brevis | [65] | ||
| Rye malt sourdoughs | Lb. reuteri | [66] | ||
| Lentils | Lb. plantarum and Savinase® | [67] | ||
| Anti-hypertensive activity | ACE 3 inhibitory peptides and GABA | Whole meal wheat sourdough | Lb. brevis CECT 8183 and protease | [63] |
| Vitamin content | Folate (vitamin B9), vitamin K, riboflavin (vitamin B2) | Lupin - tempeh | Co-culture of Propionibacterium freudenreichii and Rhizopus oryzae | [68,69] |
| Rhizopus and Propionibacterium | [68] | |||
| Cereal-based matrices (malted barley flour, barley flour and wheat aleurone) | Three strains of P. freudenreichii | [70] | ||
| Natto | - | [71] | ||
| Protein hydrolysis | Pea proteins | - | [72] | |
| Germination and sourdough fermentation | - | [73] | ||
| Anti-diabetic properties | Pasta formulated with 20% fermented quinoa flour | - | [59] | |
| FODMAP Reduction | Whole wheat bread | Kluyveromyces marxianus yeast strains | [74,75,76] | |
| Austrian traditional sourdough | S. cerevisiae and Torulaspora delbrueckii | [77] | ||
| LAB and yeasts | [78] |
| Health-Promoting Activity | Health-Promoting Compounds | Raw Food Matrices | Fermenting Microorganism(s) | References |
|---|---|---|---|---|
| Antioxidant activity | Phenolic compounds, GABA 1, peptides, CLA 2, folates (vitamin B9) | Beverage containing apples, pears, and carrots | Two strains of Lb. plantarum | [89] |
| Cashew-apple-juice | Lb. plantarum | [90] | ||
| Apple juice | Lb. plantarum strain ATCC14917 | [91] | ||
| Pomegranate juice fermented | Lb. plantarum strain ATCC 14917 | [92] | ||
| Diospyros lotus L. fruit | Lb. plantarum strain B7 | [93] | ||
| Syzygium cumini L fruit juice | Lb. paracasei strain HII01 | [94] | ||
| Apricot juice | Mono- and mixed cultures of probiotic Lactobacillus and Bifidobacterium strains | [95] | ||
| Apple juice | - | [96] | ||
| Tomato | LAB | [97] | ||
| Red cabbage | - | [98] | ||
| Vitamin content | Vitamin K (Phylloquinone) | Cashew apple juice | 5 probiotic strains (Lb. acidophilus, Lb. casei, Lb. plantarum, Lb. mesenteroides and B. longum) | [90] |
| Kimchi | [71] | |||
| Protein hydrolysis | Lotus (Nelumbo nucifera Gaertn.) root | - | [95] |
| Health-Promoting Activity | Health-Promoting Compounds | Raw Food Matrices | Fermenting Microorganism(s) | References |
|---|---|---|---|---|
| Antioxidant activity | Phenolic compounds, GABA 1, peptides, CLA 2, folates (vitamin B9) | Dry-cured pork loins | Probiotic Strains of LAB | [105] |
| Functional fermented sausages | Encapsulation of probiotic Bifidobacterium longum | [106] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Melini, F.; Melini, V.; Luziatelli, F.; Ficca, A.G.; Ruzzi, M. Health-Promoting Components in Fermented Foods: An Up-to-Date Systematic Review. Nutrients 2019, 11, 1189. https://doi.org/10.3390/nu11051189
Melini F, Melini V, Luziatelli F, Ficca AG, Ruzzi M. Health-Promoting Components in Fermented Foods: An Up-to-Date Systematic Review. Nutrients. 2019; 11(5):1189. https://doi.org/10.3390/nu11051189
Chicago/Turabian StyleMelini, Francesca, Valentina Melini, Francesca Luziatelli, Anna Grazia Ficca, and Maurizio Ruzzi. 2019. "Health-Promoting Components in Fermented Foods: An Up-to-Date Systematic Review" Nutrients 11, no. 5: 1189. https://doi.org/10.3390/nu11051189
APA StyleMelini, F., Melini, V., Luziatelli, F., Ficca, A. G., & Ruzzi, M. (2019). Health-Promoting Components in Fermented Foods: An Up-to-Date Systematic Review. Nutrients, 11(5), 1189. https://doi.org/10.3390/nu11051189







