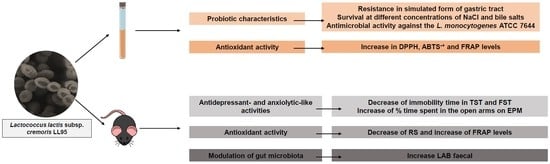
In Vitro Probiotic and Antioxidant Potential of Lactococcus lactis subsp. cremoris LL95 and Its Effect in Mice Behaviour
Abstract
1. Introduction
2. Materials and Methods
2.1. Drugs
2.2. Bacterial Strain and Growth Conditions
2.3. In Vitro Evaluation of Probiotic Characteristics
2.3.1. Survival in Simulated Gastrointestinal Tract Conditions
2.3.2. Evaluation of The Resistance to Different Concentrations of NaCl, pH and Bile Salts
2.3.3. Autoaggregation and Coaggregation Abilities
2.3.4. Antimicrobial Activity
2.3.5. Antibiotic Susceptibility Tests
2.4. Determination of In Vitro Antioxidant activities of L. lactis subsp. cremoris LL 95
2.4.1. Preparation of Intact, Heat-Killed, and Lyophilised Cells
2.4.2. DPPH Radical Scavenging Activity Assay
2.4.3. Scavenging of ABTS•+ Radical
2.4.4. Ferric Ion Reducing Antioxidant Power (FRAP)
2.5. In Vivo Experiments
2.5.1. Animals
2.5.2. Experimental Procedure
2.5.3. Antidepressant- and Anxiolytic-Like Evaluation
Open-Field Test (OFT)
Elevated Plus Maze (EPM)
Tail Suspension Test (TST)
Forced Swim Test (FST)
2.5.4. Hippocampus Weight and Tissue Preparation
2.5.5. Reactive Species Quantification
2.5.6. Ferric Reducing Antioxidant Power (FRAP) Assay
2.5.7. Quantification of Faecal LAB
2.6. Data Presentation and Statistical Analysis
3. Results and Discussion
3.1. Evaluation of Probiotic Characteristics
3.1.1. Survival in Simulated Gastrointestinal Tract Conditions and Resistance to Different Concentrations of Salt (NaCl), pH and Bile Salts
3.1.2. Autoaggregation and Coaggregation Abilities
3.1.3. Antimicrobial Activity
3.1.4. Antibiotic Susceptibility Tests
3.2. In Vitro Antioxidant Activity Experiments
Determination of In Vitro Antioxidant Activities of Lactococcus lactis subsp. cremoris LL95
3.3. In Vivo Experiments
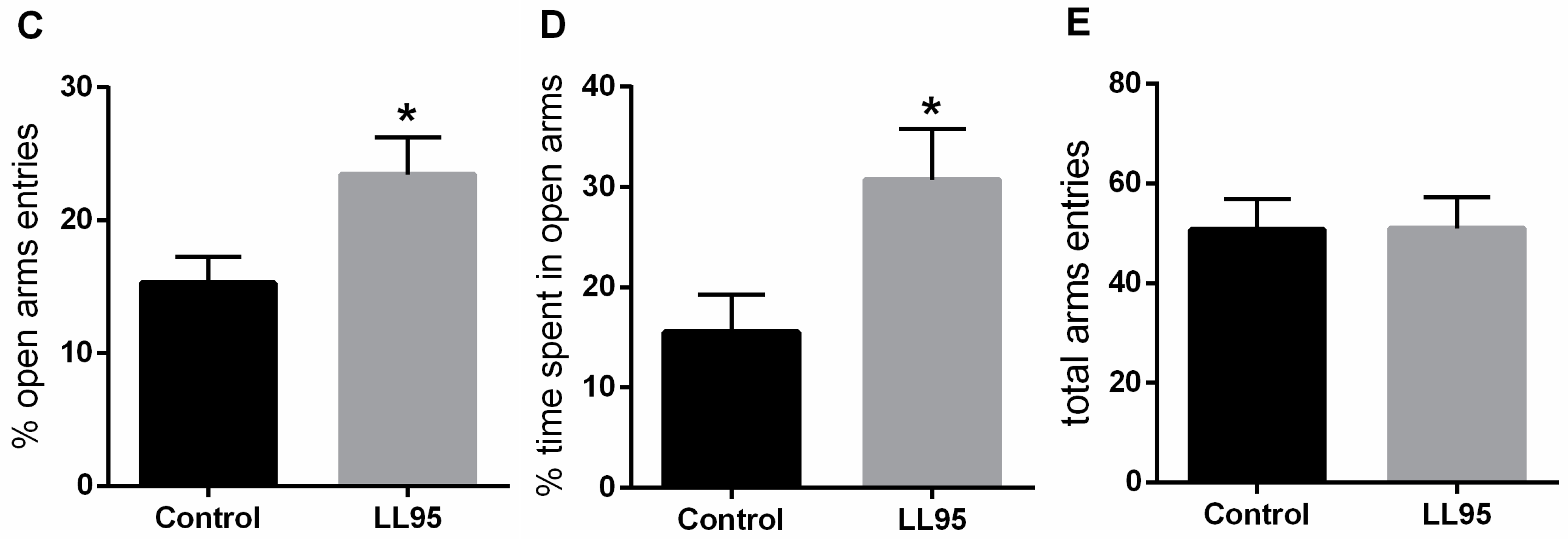
3.3.1. Lactococcus lactis subsp. cremoris LL95 Had No Effect on Locomotor Activity and Reduced the Anxiety-Like Behaviour
3.3.2. Lactococcus lactis subsp. cremoris LL95 Reduces Depressive-Like Behaviour in Mice
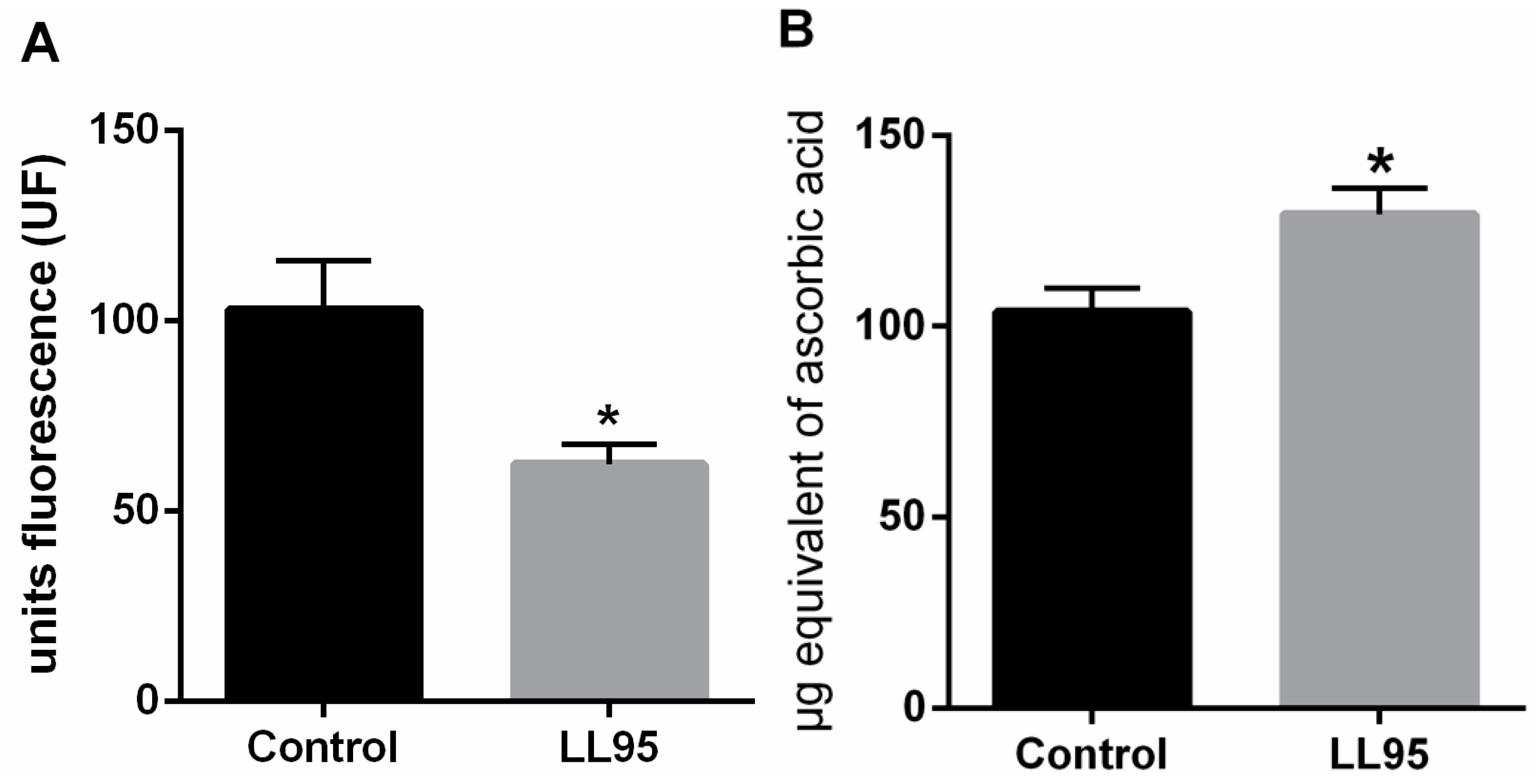
3.3.3. Lactococcus lactis subsp. cremoris LL95 Increased Hippocampal Weight and Prevented Oxidative Stress in the Hippocampus in Mice
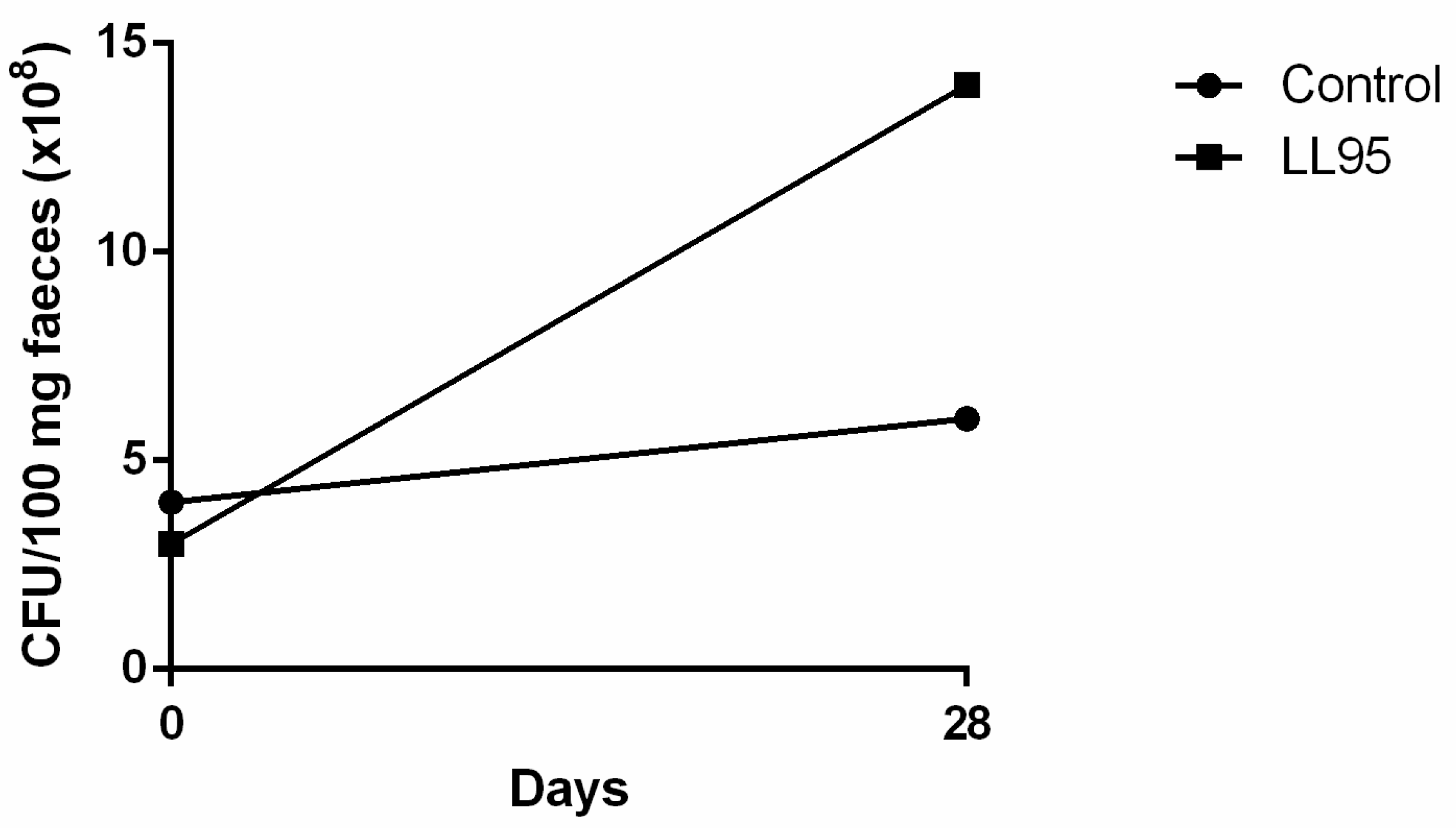
3.3.4. Administration of Lactococcus lactis subsp. cremoris LL95 Increased the Faecal Content of LAB
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bischoff, S.C.; Barbara, G.; Buurman, W.; Ockhuizen, T.; Schulzke, J.-D.; Serino, M.; Tilg, H.; Watson, A.; Wells, J.M. Intestinal permeability—A new target for disease prevention and therapy. BMC Gastroenterol. 2014, 14, 189. [Google Scholar] [CrossRef]
- Carding, S.; Verbeke, K.; Vipond, D.T.; Corfe, B.M.; Owen, L.J. Dysbiosis of the gut microbiota in disease. Microb. Ecol. Health Dis. 2015, 26, 3854. [Google Scholar] [CrossRef] [PubMed]
- DeGruttola, A.K.; Low, D.; Mizoguchi, A.; Mizoguchi, E. Current understanding of dysbiosis in disease in human and animal models. Inflamm. Bowel Dis. 2016, 22, 1137–1150. [Google Scholar] [CrossRef] [PubMed]
- Natividad, J.M.; Verdu, E.F. Modulation of intestinal barrier by intestinal microbiota: Pathological and therapeutic implications. Pharmacol. Res. 2013, 69, 42–51. [Google Scholar] [CrossRef] [PubMed]
- FAO; WHO. Joint FAO/WHO Expert Consultation on Evaluation of Health and Nutritional Properties of Probiotics in Food Including Powder Milk with Live Lactic Acid Bacteria; FAO: Rome, Italy; WHO: Rome, Italy, 2001; pp. 1–4. [Google Scholar]
- Reis, J.A.; Paula, A.T.; Casarotti, S.N.; Penna, A.L.B. Lactic Acid Bacteria Antimicrobial Compounds: Characteristics and Applications. Eng. Rev. 2012, 4, 124–140. [Google Scholar] [CrossRef]
- Saad, N.; Delattre, C.; Urdaci, M.; Schmitter, J.; Bressollier, P. An overview of the last advances in probiotic and prebiotic field. LWT Food Sci. Technol. 2013, 50, 1–16. [Google Scholar] [CrossRef]
- Pereira, G.V.D.M.; Coelho, B.D.O.; Júnior, A.I.M.; Thomaz-Soccol, V.; Soccol, C.R. How to select a probiotic? A review and update of methods and criteria. Biotechnol. Adv. 2018, 36, 2060–2076. [Google Scholar] [CrossRef]
- Dinan, T.G.; Cryan, J.F. Brain-Gut-Microbiota Axis and Mental Health. Psychosom. Med. 2017, 79, 920–926. [Google Scholar] [CrossRef]
- Bravo, J.A.; Forsythe, P.; Chew, M.V.; Escaravage, E.; Savignac, H.M.; Dinan, T.G.; Bienenstock, J.; Cryan, J.F. Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proc. Natl. Acad. Sci. USA 2011, 108, 16050–16055. [Google Scholar] [CrossRef] [PubMed]
- Cryan, J.F.; Dinan, T.G. Mind-altering microorganisms: The impact of the gut microbiota on brain and behaviour. Nat. Rev. Neurosci. 2012, 13, 701–712. [Google Scholar] [CrossRef]
- Desbonnet, L.; Clarke, G.; Shanahan, F.; Dinan, T.G.; Cryan, J.F.; Dinan, T.; Cryan, J. Microbiota is essential for social development in the mouse. Mol. Psychiatry 2013, 19, 146–148. [Google Scholar] [CrossRef] [PubMed]
- Desbonnet, L.; Garrett, L.; Clarke, G.; Kiely, B.; Cryan, J.; Dinan, T.; Dinan, T. Effects of the probiotic Bifidobacterium infantis in the maternal separation model of depression. Neuroscience 2010, 170, 1179–1188. [Google Scholar] [CrossRef] [PubMed]
- Liang, S.; Wang, T.; Hu, X.; Luo, J.; Li, W.; Wu, X.; Duan, Y.; Jin, F. Administration of Lactobacillus helveticus NS8 improves behavioral, cognitive, and biochemical aberrations caused by chronic restraint stress. Neuroscience 2015, 310, 561–577. [Google Scholar] [CrossRef]
- Liu, Y.-W.; Liu, W.-H.; Wu, C.-C.; Juan, Y.-C.; Wu, Y.-C.; Tsai, H.-P.; Wang, S.; Tsai, Y.-C. Psychotropic effects of Lactobacillus plantarum PS128 in early life-stressed and naïve adult mice. Brain Res. 2016, 1631, 1–12. [Google Scholar] [CrossRef]
- Savignac, H.M.; Kiely, B.; Dinan, T.; Cryan, J.F. Bifidobacteria exert strain-specific effects on stress-related behavior and physiology in BALB/c mice. Neurogastroenterol. Motil. 2014, 26, 1615–1627. [Google Scholar] [CrossRef]
- Abildgaard, A.; Elfving, B.; Hokland, M.; Wegener, G.; Lund, S. Probiotic treatment reduces depressive-like behaviour in rats independently of diet. Psychoneuroendocrinology 2017, 79, 40–48. [Google Scholar] [CrossRef]
- Abdrabou, A.M.; Osman, E.Y.; Aboubakr, O.A. Comparative therapeutic efficacy study of Lactobacilli probiotics and citalopram in treatment of acute stress-induced depression in lab murine models. Hum. Microbiome J. 2018, 10, 33–36. [Google Scholar] [CrossRef]
- WHO. Depression and Other Common Mental Disorders: Global Health Estimates; World Health Organization: Geneva, Switzerland, 2017; pp. 1–24. Available online: http://apps.who.int/iris/bitstream/handle/10665/254610/WHO-MSD-MER-2017.2-eng.pdf (accessed on 24 September 2018).
- Zhou, Y.; Cao, Z.; Yang, M.; Xi, X.; Guo, Y.; Fang, M.; Cheng, L.; Du, Y. Comorbid generalized anxiety disorder and its association with quality of life in patients with major depressive disorder. Sci. Rep. 2017, 7, 40511. [Google Scholar] [CrossRef]
- Anderson, H.D.; Pace, W.D.; Libby, A.M.; West, D.R.; Valuck, R.J. Rates of 5 Common Antidepressant Side Effects Among New Adult and Adolescent Cases of Depression: A Retrospective US Claims Study. Clin. Ther. 2012, 34, 113–123. [Google Scholar] [CrossRef] [PubMed]
- Vlieg, J.E.V.H.; Rademaker, J.L.; Bachmann, H.; Molenaar, D.; Kelly, W.J.; Siezen, R.J. Natural diversity and adaptive responses of Lactococcus lactis. Curr. Opin. Biotechnol. 2006, 17, 183–190. [Google Scholar] [CrossRef]
- Kosaka, A.; Yan, H.; Ohashi, S.; Gotoh, Y.; Sato, A.; Tsutsui, H.; Kaisho, T.; Toda, T.; Tsuji, N.M. Lactococcus lactis subsp. cremoris FC triggers IFN-γ production from NK and T cells via IL-12 and IL-18. Int. Immunopharmacol. 2012, 14, 729–733. [Google Scholar] [CrossRef] [PubMed]
- Maruo, T.; Gotoh, Y.; Nishimura, H.; Ohashi, S.; Toda, T.; Takahashi, K. Oral administration of milk fermented with Lactococcus lactis subsp. cremoris FC protects mice against influenza virus infection. Lett. Appl. Microbiol. 2012, 55, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Hamed, H.; Chaari, F.; Ghannoudi, Z.; Dhouib, K.; Chaabouni, S.; El Feki, A.; Gargouri, A. Fermented camel milk by Lactococcus lactis subsp. cremoris attenuates erythrocytes oxidative stress-induced hematological and immunological damage in CCl4-intoxicated mice. Environ. Sci. Pollut. Res. 2018, 25, 25983–25993. [Google Scholar] [CrossRef]
- Hamed, H.; Chaari, F.; Ghannoudi, Z.; Elfeki, A.; Ellouz, S.C.; Gargouri, A. Beneficial effects of fermented camel milk by Lactococcus lactis subsp. cremoris on cardiotoxicity induced by carbon tetrachloride in mice. Biomed. Pharmacother. 2018, 97, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Figueroa-González, I.; Quijano, G.; Ramirez, G.; Cruz-Guerrero, A.; Figueroa-González, I.; Cruz-Guerrero, A. Probiotics and prebiotics-perspectives and challenges. J. Sci. Agric. 2011, 91, 1341–1348. [Google Scholar] [CrossRef]
- Huang, Y.; Adams, M.C. In vitro assessment of the upper gastrointestinal tolerance of potential probiotic dairy propionibacteria. Int. J. Microbiol. 2004, 91, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Drosinos, E.; Mataragas, M.; Xiraphi, N.; Moschonas, G.; Gaitis, F.; Metaxopoulos, J. Characterization of the microbial flora from a traditional Greek fermented sausage. Meat Sci. 2005, 69, 307–317. [Google Scholar] [CrossRef]
- Collado, M.C.; Meriluoto, J.; Salminen, S. Adhesion and aggregation properties of probiotic and pathogen strains. Eur. Food Res. Technol. 2008, 226, 1065–1073. [Google Scholar] [CrossRef]
- Biscola, V.; Todorov, S.; Capuano, V.; Abriouel, H.; Gálvez, A.; Franco, B. Isolation and characterization of a nisin-like bacteriocin produced by a Lactococcus lactis strain isolated from charqui, a Brazilian fermented, salted and dried meat product. Meat Sci. 2013, 93, 607–613. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority. Opnion of the Scientific Committee on Animal Nutrition on the criteria for assessing the safety of micro-organisms resistant to antibiotics of human clinical and veterinary importance. Eur. Food Saf. Auth. J. 2003, 226, 1–16. [Google Scholar]
- Liasi, S.A.; Azmi, T.I.; Hassan, M.D.; Shuhaimi, M.; Rosfarizan, M.; Ariff, A.B. Antimicrobial activity and antibiotic sensitivity of three isolates of lactic acid bacteria from fermented fish product, Budu. Malays. J. Microbiol. 2009, 5, 33–37. [Google Scholar]
- Lim, L.H.; Li, H.Y.; Huang, C.H.; Chua, K.Y.; Lee, B.W.; Lee, Y.K. The Effects of Heat-Killed Wild-Type Lactobacillus casei Shirota on Allergic Immune Responses in an Allergy Mouse Model. Int. Arch. Immunol. 2008, 148, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Xing, J.; Wang, G.; Zhang, Q.; Liu, X.; Gu, Z.; Zhang, H.; Chen, Y.Q.; Chen, W. Determining Antioxidant Activities of Lactobacilli Cell-Free Supernatants by Cellular Antioxidant Assay: A Comparison with Traditional Methods. PLoS ONE 2015, 10, e0119058. [Google Scholar] [CrossRef] [PubMed]
- Han, Q.; Kong, B.; Chen, Q.; Sun, F.; Zhang, H. In vitro comparison of probiotic properties of lactic acid bacteria isolated from Harbin dry sausages and selected probiotics. J. Funct. Foods 2017, 32, 391–400. [Google Scholar] [CrossRef]
- Parker, G.; Brotchie, H. Gender differences in depression. Int. Rev. 2010, 22, 429–436. [Google Scholar] [CrossRef] [PubMed]
- Seibenhener, M.L.; Wooten, M.C. Use of the Open Field Maze to Measure Locomotor and Anxiety-like Behavior in Mice. J. Vis. Exp. 2015, 10, e52434. [Google Scholar] [CrossRef] [PubMed]
- Lister, R.G. The use of a plus-maze to measure anxiety in the mouse. Psychopharmacology 1987, 92, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Steru, L.; Chermat, R.; Thierry, B.; Simon, P. The tail suspension test: A new method for screening antidepressants in mice. Psychopharmacology 1985, 85, 367–370. [Google Scholar] [CrossRef] [PubMed]
- Porsolt, R.D.; Le Pichon, M.; Jalfre, M. Depression: A new animal model sensitive to antidepressant treatments. Nature 1977, 266, 730–732. [Google Scholar] [CrossRef] [PubMed]
- Cryan, J.F.; Markou, A.; Lucki, I. Assessing antidepressant activity in rodents: Recent developments and future needs. Pharmacol. Sci. 2002, 23, 238–245. [Google Scholar] [CrossRef]
- Loetchutinat, C.; Kothan, S.; Dechsupa, S.; Meesungnoen, J.; Jay-Gerin, J.-P.; Mankhetkorn, S. Spectrofluorometric determination of intracellular levels of reactive oxygen species in drug-sensitive and drug-resistant cancer cells using the 2′,7′-dichlorofluorescein diacetate assay. Radiat. Phys. Chem. 2005, 72, 323–331. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J. Ferric reducing/antioxidant power assay: Direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Methods Enzymol. 1999, 299, 15–27. [Google Scholar]
- Grimm, V.; Radulović, K.; Riedel, C.U. Colonization of C57BL/6 Mice by a Potential Probiotic Bifidobacterium bifidum Strain under Germ-Free and Specific Pathogen-Free Conditions and during Experimental Colitis. PLoS ONE 2015, 10, e0139935. [Google Scholar] [CrossRef]
- Maragkoudakis, P.A.; Zoumpopoulou, G.; Miaris, C.; Kalantzopoulos, G.; Pot, B.; Tsakalidou, E. Probiotic potential of Lactobacillus strains isolated from dairy products. Int. J. 2006, 16, 189–199. [Google Scholar] [CrossRef]
- Monteagudo-Mera, A.; Rodríguez-Aparicio, L.B.; Rúa, J.; Martínez-Blanco, H.; Navasa, N.; García-Armesto, M.R.; Ferrero, M. Ángel In vitro evaluation of physiological probiotic properties of different lactic acid bacteria strains of dairy and human origin. J. Funct. Foods 2012, 4, 531–541. [Google Scholar] [CrossRef]
- Charteris, W.P.; Kelly, P.M.; Morelli, L.; Collins, J.K. Ingredient selection criteria for probiotic microorganisms in functional dairy foods. Int. J. Technol. 1998, 51, 123–136. [Google Scholar] [CrossRef]
- Sgorbati, B.; Palenzona, D.; Del Re, B.; Miglioli, M. Adhesion, autoaggregation and hydrophobicity of 13 strains of Bifidobacterium longum. Lett. Appl. Microbiol. 2000, 31, 438–442. [Google Scholar]
- Kaewnopparat, S.; Dangmanee, N.; Kaewnopparat, N.; Srichana, T.; Chulasiri, M.; Settharaksa, S. In vitro probiotic properties of Lactobacillus fermentum SK5 isolated from vagina of a healthy woman. Anaerobe 2013, 22, 6–13. [Google Scholar] [CrossRef]
- Botes, M.; Loos, B.; Van Reenen, C.A.; Dicks, L.M.T.; Reenen, C.A. Adhesion of the probiotic strains Enterococcus mundtii ST4SA and Lactobacillus plantarum 423 to Caco-2 cells under conditions simulating the intestinal tract, and in the presence of antibiotics and anti-inflammatory medicaments. Arch. Microbiol. 2008, 190, 573–584. [Google Scholar] [CrossRef]
- Vandenbergh, P.A. Lactic acid bacteria, their metabolic products and interference with microbial growth. FEMS Microbiol. Rev. 1993, 12, 221–237. [Google Scholar] [CrossRef]
- Leroy, F.; Verluyten, J.; De Vuyst, L. Functional meat starter cultures for improved sausage fermentation. Int. J. Microbiol. 2006, 106, 270–285. [Google Scholar] [CrossRef] [PubMed]
- Bennish, M.L. Animals, humans, and antibiotics: Implications of the veterinary use of antibiotics on human health. Adv. Pediatr. Infect. Dis. 1999, 14, 269–290. [Google Scholar] [PubMed]
- Guarner, F.; Malagelada, J.-R. Gut flora in health and disease. Lancet 2003, 361, 512–519. [Google Scholar] [CrossRef]
- Hamilton-Miller, J. Antibiotic resistance from two perspectives: Man and microbe. Int. J. Antimicrob. Agents 2004, 23, 209–212. [Google Scholar] [CrossRef]
- Armas, F.; Camperio, C.; Marianelli, C. In Vitro Assessment of the Probiotic Potential of Lactococcus lactis LMG 7930 against Ruminant Mastitis-Causing Pathogens. PLoS ONE 2017, 12, e0169543. [Google Scholar] [CrossRef]
- Gad, G.F.M.; Abdel-Hamid, A.M.; Farag, Z.S.H. Antibiotic resistance in lactic acid bacteria isolated from some pharmaceutical and dairy products. Braz. J. Microbiol. 2014, 45, 25–33. [Google Scholar] [CrossRef]
- Teuber, M.; Meile, L.; Schwarz, F.; Veld, J.H.J.H. Acquired antibiotic resistance in lactic acid bacteria from food. Lactic Acid Bact. Genet. Metab. Appl. 1999, 76, 115–137. [Google Scholar]
- Wang, Y.; Wu, Y.; Wang, Y.; Xu, H.; Mei, X.; Yu, D.; Wang, Y.; Li, W. Antioxidant Properties of Probiotic Bacteria. Nutrients 2017, 9, 521. [Google Scholar] [CrossRef]
- Lin, M.-Y.; Yen, C.-L. Antioxidative Ability of Lactic Acid Bacteria. J. Agric. Chem. 1999, 47, 1460–1466. [Google Scholar] [CrossRef]
- Lee, J.; Hwang, K.-T.; Chung, M.-Y.; Cho, D.-H.; Park, C.-S. Resistance of Lactobacillus casei KCTC 3260 to Reactive Oxygen Species (ROS): Role for a Metal Ion Chelating Effect. J. Sci. 2005, 70, m388–m391. [Google Scholar] [CrossRef]
- Li, Y.; Hugenholtz, J.; Abee, T.; Molenaar, D. Glutathione Protects Lactococcus lactis against Oxidative Stress. Appl. Environ. Microbiol. 2003, 69, 5739–5745. [Google Scholar] [CrossRef] [PubMed]
- Bailey, K.R.; Crawley, J.N. Anxiety-Related Behaviors in Mice. In Methods of Behavior Analysis in Neuroscience; CRC Press: Boca Raton, FL, USA, 2009. [Google Scholar]
- Walf, A.; Frye, C. The use of the elevated plus maze as an assay of anxiety-related behavior in rodents. Nat. Protoc. 2007, 2, 322–328. [Google Scholar] [CrossRef] [PubMed]
- Cryan, J.F.; Holmes, A. The ascent of mouse: Advances in modelling human depression and anxiety. Nat. Rev. Drug Discov. 2005, 4, 775–790. [Google Scholar] [CrossRef]
- Slyepchenko, A.; Carvalho, A.F.; Cha, D.S.; Kasper, S.; McIntyre, R.S. Gut emotions - mechanisms of action of probiotics as novel therapeutic targets for depression and anxiety disorders. CNS Neurol. Disord. Drug Targets 2014, 13, 1770–1786. [Google Scholar] [CrossRef] [PubMed]
- Avolio, E.; Fazzari, G.; Zizza, M.; De Lorenzo, A.; Di Renzo, L.; Alo, R.; Facciolo, R.M.; Canonaco, M. Probiotics modify body weight together with anxiety states via pro-inflammatory factors in HFD-treated Syrian golden hamster. Behav. Brain Res. 2018, 356, 390–399. [Google Scholar] [CrossRef] [PubMed]
- Messaoudi, M.; LaLonde, R.; Violle, N.; Javelot, H.; Desor, D.; Nejdi, A.; Bisson, J.-F.; Rougeot, C.; Pichelin, M.; Cazaubiel, M.; et al. Assessment of psychotropic-like properties of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in rats and human subjects. Br. J. Nutr. 2010, 105, 755–764. [Google Scholar] [CrossRef]
- Steenbergen, L.; Sellaro, R.; Van Hemert, S.; Bosch, J.A.; Colzato, L.S. A randomized controlled trial to test the effect of multispecies probiotics on cognitive reactivity to sad mood. Brain Behav. Immun. 2015, 48, 258–264. [Google Scholar] [CrossRef]
- Mohammadi, A.A.; Jazayeri, S.; Khosravi-Darani, K.; Solati, Z.; Mohammadpour, N.; Asemi, Z.; Adab, Z.; Djalali, M.; Tehrani-Doost, M.; Hosseini, M.; et al. The effects of probiotics on mental health and hypothalamic–pituitary–adrenal axis: A randomized, double-blind, placebo-controlled trial in petrochemical workers. Nutr. Neurosci. 2015, 19, 387–395. [Google Scholar] [CrossRef]
- Schmidt, C. Thinking from the Gut. Nature 2015, 518, S12–S14. [Google Scholar] [CrossRef] [PubMed]
- Akkasheh, G.; Kashani-Poor, Z.; Tajabadi-Ebrahimi, M.; Jafari, P.; Akbari, H.; Taghizadeh, M.; Memarzadeh, M.R.; Asemi, Z.; Esmaillzadeh, A.; Information, P.E.K.F.C. Clinical and metabolic response to probiotic administration in patients with major depressive disorder: A randomized, double-blind, placebo-controlled trial. Nutrition 2016, 32, 315–320. [Google Scholar] [CrossRef]
- Amaretti, A.; Nunzio, M.; Pompei, A.; Raimondi, S.; Rossi, M.; Bordoni, A.; Di Nunzio, M. Antioxidant properties of potentially probiotic bacteria: In vitro and in vivo activities. Appl. Microbiol. Biotechnol. 2012, 97, 809–817. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Hu, X.-L.; Le, G.-W.; Shi, Y.-H. Inhibition of Fe-induced colon oxidative stress by lactobacilli in mice. J. Microbiol. Biotechnol. 2012, 29, 209–216. [Google Scholar] [CrossRef] [PubMed]
- Songisepp, E.; Kals, J.; Kullisaar, T.; Mändar, R.; Hütt, P.; Zilmer, M.; Mikelsaar, M. Evaluation of the functional efficacy of an antioxidative probiotic in healthy volunteers. Nutr. J. 2005, 4, 22. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Ren, P.; He, S.; Xu, L.; Yang, Y.; Gu, Z.; Zhou, Z. Comparison of adhesive gut bacteria composition, immunity, and disease resistance in juvenile hybrid tilapia fed two different Lactobacillus strains. Fish Shellfish. Immunol. 2013, 35, 54–62. [Google Scholar] [CrossRef] [PubMed]
- Kelly, J.R.; Borre, Y.; Brien, C.O.; Patterson, E.; El Aidy, S.; Deane, J.; Kennedy, P.J.; Beers, S.; Scott, K.; Moloney, G.; et al. Transferring the blues: Depression-associated gut microbiota induces neurobehavioural changes in the rat. J. Psychiatr. Res. 2016, 82, 109–118. [Google Scholar] [CrossRef] [PubMed]


| Time (min) | Log CFU/mL | |
|---|---|---|
| SS | SM | |
| 0 | 8.22 ± 0.01 a | 8.27 ± 0.02 b |
| 15 | 8.45 ± 0.04 a | 8.30 ± 0.00 b |
| 30 | 8.23 ± 0.00 a | 8.38 ± 0.13 a |
| 60 | 7.57 ± 0.11 a | 8.27 ± 0.18 b |
| 120 | 5.43 ± 0.18 a | 8.46 ± 0.11 b |
| 180 | 2.93 ± 0.31 a | 8.18 ± 0.00 b |
| 240 | 0.57 ± 0.76 a | 8.25 ± 0.10 b |
| Time (h) | LL95 | LM | ||
|---|---|---|---|---|
| 20 °C | 37 °C | 20 °C | 37 °C | |
| 2 | 22.13 ± 0.12 a | 24.10 ± 0.14 b | 12.00 ± 0.15 a | 19.26 ± 0.31 b |
| 20 | 11.70 ± 0.14 a | 16.80 ± 0.52 b | 20.18 ± 0.31 a | 22.72 ± 0.31 b |
| 24 | 15.18 ± 0.13 a | 21.32 ± 0.28 a | 18.22 ± 0.15 a | 27.10 ± 0.31 b |
| Time (h) | 20 °C | 37 °C |
|---|---|---|
| 2 | 7.90 ± 0.42 a | 0 ± 1.98 b |
| 20 | 4.95 ± 0.08 a | 12.73 ± 2.62 b |
| 24 | 0 ± 5.49 a | 18.86 ± 4.44 b |
| Antibiotics | Inhibition Zone Diameters (mm) | Classification 1 |
|---|---|---|
| Ampicillin | 23 | Sensitive |
| Chloramphenicol | 31 | Sensitive |
| Erythromycin | 23 | Sensitive |
| Penicillin | 23 | Sensitive |
| Vancomycin | 22 | Sensitive |
| Ciprofloxacin | 29 | Sensitive |
| Gentamicin | 16 | Intermediate sensitivity |
| Amikacin | 15 | Resistant |
| Trimethoprim-sulphamethoxazole | 0 | Resistant |
| Tetracycline | 15 | Resistant |
| Clindamycin | 9 | Resistant |
| Sulphonamide | 0 | Resistant |
| LL 95 cells | DPPH (U/mL) | ABTS•+ (%) | FRAP (mM) |
|---|---|---|---|
| Intact cells | 1.30 ± 0.50 a | 56.50 ± 4.68 a | 22.40 ± 0.39 a |
| Heat-killed cells | 6.85 ± 0.45 a,b | 32.34 ± 0.22 b | 13.49 ± 0.49 b |
| Lyophilised cells | 9.40 ± 1.90 b | 79.07 ± 4.73 a | 45.04 ± 0.04 c |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ramalho, J.B.; Soares, M.B.; Spiazzi, C.C.; Bicca, D.F.; Soares, V.M.; Pereira, J.G.; da Silva, W.P.; Sehn, C.P.; Cibin, F.W.S. In Vitro Probiotic and Antioxidant Potential of Lactococcus lactis subsp. cremoris LL95 and Its Effect in Mice Behaviour. Nutrients 2019, 11, 901. https://doi.org/10.3390/nu11040901
Ramalho JB, Soares MB, Spiazzi CC, Bicca DF, Soares VM, Pereira JG, da Silva WP, Sehn CP, Cibin FWS. In Vitro Probiotic and Antioxidant Potential of Lactococcus lactis subsp. cremoris LL95 and Its Effect in Mice Behaviour. Nutrients. 2019; 11(4):901. https://doi.org/10.3390/nu11040901
Chicago/Turabian StyleRamalho, Juliana B., Melina B. Soares, Cristiano C. Spiazzi, Diogo F. Bicca, Vanessa M. Soares, Juliano G. Pereira, Wladimir P. da Silva, Carla P. Sehn, and Francielli W. S. Cibin. 2019. "In Vitro Probiotic and Antioxidant Potential of Lactococcus lactis subsp. cremoris LL95 and Its Effect in Mice Behaviour" Nutrients 11, no. 4: 901. https://doi.org/10.3390/nu11040901
APA StyleRamalho, J. B., Soares, M. B., Spiazzi, C. C., Bicca, D. F., Soares, V. M., Pereira, J. G., da Silva, W. P., Sehn, C. P., & Cibin, F. W. S. (2019). In Vitro Probiotic and Antioxidant Potential of Lactococcus lactis subsp. cremoris LL95 and Its Effect in Mice Behaviour. Nutrients, 11(4), 901. https://doi.org/10.3390/nu11040901