Association of Chocolate Consumption with Hearing Loss and Tinnitus in Middle-Aged People Based on the Korean National Health and Nutrition Examination Survey 2012–2013
Abstract
1. Introduction
2. Materials and Methods
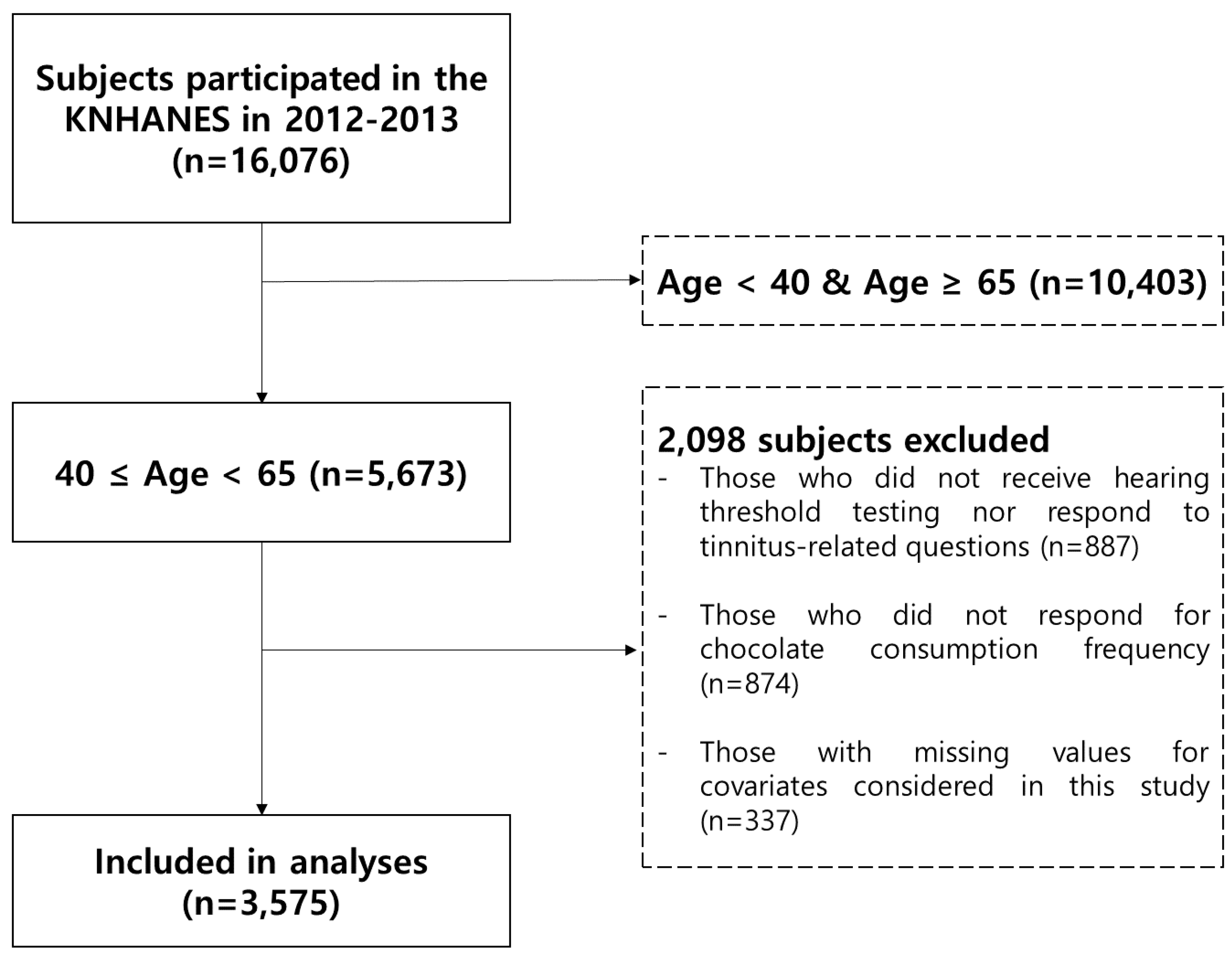
2.1. Study Population
2.2. Audiological Assessment
2.3. Potential Confounders
2.4. Assessment of Chocolate Consumption
2.5. Statistical Analysis
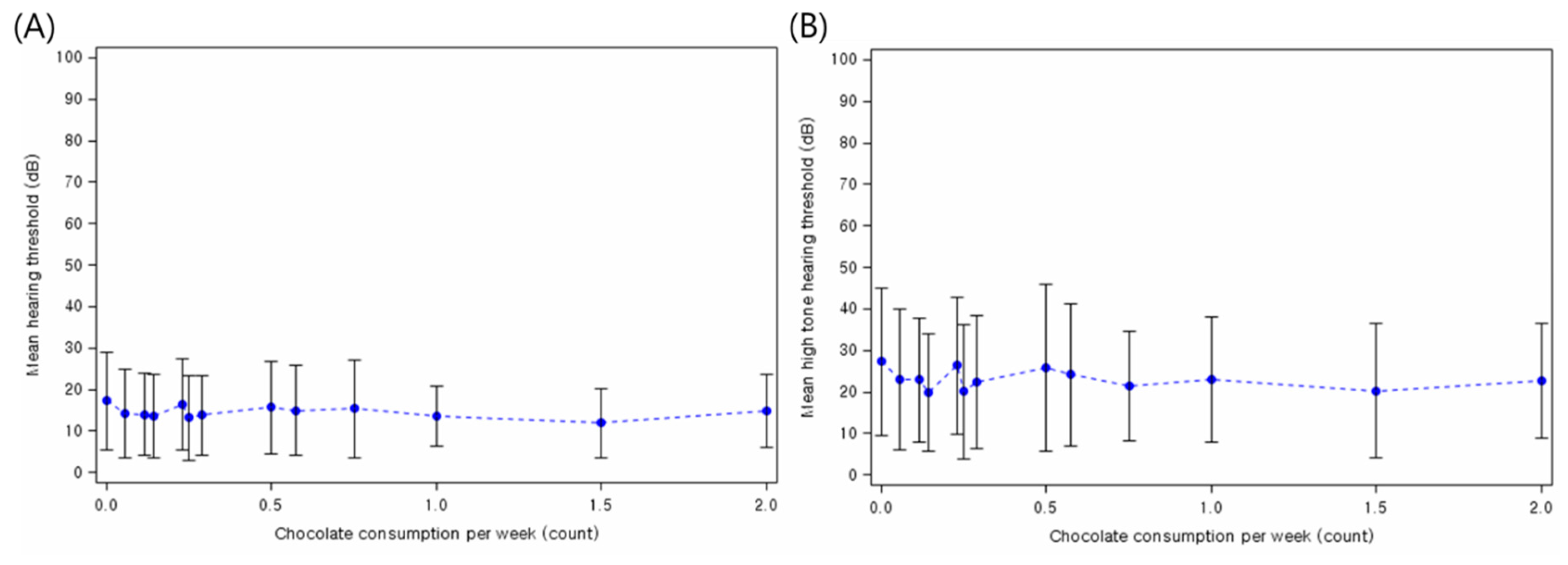
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Authors Contributions
Funding
Conflicts of Interest
References
- Ramsey, T.; Svider, P.F.; Folbe, A.J. Health burden and socioeconomic disparities from hearing loss: A global perspective. Otol. Neurotol. 2018, 39, 12–16. [Google Scholar] [CrossRef]
- Stevens, G.; Flaxman, S.; Brunskill, E.; Mascarenhas, M.; Mathers, C.D.; Finucane, M. Global Burden of Disease Hearing Loss Expert Group. Global and regional hearing impairment prevalence: An analysis of 42 studies in 29 countries. Eur. J. Public Health 2013, 23, 146–152. [Google Scholar] [CrossRef]
- Crealey, G.E.; O’Neill, C. Hearing loss, mental well-being and healthcare use: Results from the Health Survey for England (HSE). J. Public Health 2018. [Google Scholar] [CrossRef]
- Leverton, T. Depression in older adults: Hearing loss is an important factor. BMJ 2019, 364, l160. [Google Scholar] [CrossRef]
- Peracino, A. Hearing loss and dementia in the aging population. Audiol. Neurootol. 2014, 19, 6–9. [Google Scholar] [CrossRef] [PubMed]
- Lin, F.R.; Metter, E.J.; O’Brien, R.J.; Resnick, S.M.; Zonderman, A.B.; Ferrucci, L. Hearing loss and incident dementia. Arch Neurol. 2011, 68, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Nam, D.W.; Koo, J.W.; De Ridder, D.; Vanneste, S.; Song, J.J. No auditory experience, no tinnitus: Lessons from subjects with congenital- and acquired single-sided deafness. Hear. Res. 2017, 354, 9–15. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Lee, H.J.; An, S.Y.; Sim, S.; Park, B.; Kim, S.W.; Lee, J.S.; Hong, S.K.; Choi, H.G. Analysis of the prevalence and associated risk factors of tinnitus in adults. PLoS ONE 2015, 10, e0127578. [Google Scholar] [CrossRef]
- McCormack, A.; Edmondson-Jones, M.; Somerset, S.; Hall, D. A systematic review of the reporting of tinnitus prevalence and severity. Hear. Res. 2016, 337, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Aazh, H.; Danesh, A.A.; Moore, B.C.J. Parental Mental Health In Childhood As A Risk Factor For Anxiety And Depression Among People Seeking Help For Tinnitus And Hyperacusis. J. Am. Acad. Audiol. 2018. [Google Scholar] [CrossRef]
- Bauer, C.A. Tinnitus. N. Engl. J. Med. 2018, 378, 1224–1231. [Google Scholar] [CrossRef] [PubMed]
- Tunkel, D.E.; Bauer, C.A.; Sun, G.H.; Rosenfeld, R.M.; Chandrasekhar, S.S.; Cunningham, E.R., Jr.; Archer, S.M.; Blakley, B.W.; Carter, J.M.; Granieri, E.C.; et al. Clinical practice guideline: Tinnitus. Otolaryngol. Head Neck Surg. 2014, 151, S1–S40. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.Y.; Kim, S.H.; Yeo, S.G. Association of nutritional factors with hearing loss. Nutrients 2019, 11, 307. [Google Scholar] [CrossRef]
- Lee, D.Y.; Kim, Y.H. Relationship between diet and tinnitus: Korea national health and nutrition examination survey. Clin. Exp. Otorhinolaryngol. 2018, 11, 158–165. [Google Scholar] [CrossRef]
- Di Mattia, C.D.; Sacchetti, G.; Mastrocola, D.; Serafini, M. From cocoa to chocolate: The impact of processing on in vitro antioxidant activity and the effects of chocolate on antioxidant markers in vivo. Front. Immunol. 2017, 8, 1207. [Google Scholar] [CrossRef]
- Sanchez-Rodriguez, C.; Cuadrado, E.; Riestra-Ayora, J.; Sanz-Fernandez, R. Polyphenols protect against age-associated apoptosis in female rat cochleae. Biogerontology 2018, 19, 159–169. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.Y.; Han, S.Y.; Shin, H.C.; Byun, J.Y.; Rah, Y.C.; Park, M.K. Protective effect of a purified polyphenolic extract from Ecklonia cava against noise-induced hearing loss: Prevention of temporary threshold shift. Int. J. Pediatr. Otorhinolaryngol. 2016, 87, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Magrone, T.; Russo, M.A.; Jirillo, E. Cocoa and dark chocolate polyphenols: From biology to clinical applications. Front. Immunol. 2017, 8, 677. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.Y.; Jung, G.; Jang, M.J.; Suh, M.W.; Lee, J.H.; Oh, S.H.; Park, M.K. Association of coffee consumption with hearing and tinnitus based on a national population-based survey. Nutrients 2018, 10, 1429. [Google Scholar] [CrossRef]
- Sanz-Fernandez, R.; Sanchez-Rodriguez, C.; Granizo, J.J.; Durio-Calero, E.; Martin-Sanz, E. Accuracy of auditory steady state and auditory brainstem responses to detect the preventive effect of polyphenols on age-related hearing loss in Sprague-Dawley rats. Eur. Arch. Otorhinolaryngol. 2016, 273, 341–347. [Google Scholar] [CrossRef]
- Møller, A.R. Hearing: Anatomy, Physiology, and Disorders of the Auditory System; Plural Publishing: San Diego, CA, USA, 2012. [Google Scholar]
- Agrawal, Y.; Platz, E.A.; Niparko, J.K. Risk factors for hearing loss in US adults: Data from the national health and nutrition examination survey, 1999 to 2002. Otol. Neurotol. 2009, 30, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Grassi, D.; Necozione, S.; Lippi, C.; Croce, G.; Valeri, L.; Pasqualetti, P.; Desideri, G.; Blumberg, J.B.; Ferri, C. Cocoa reduces blood pressure and insulin resistance and improves endothelium-dependent vasodilation in hypertensives. Hypertension 2005, 46, 398–405. [Google Scholar] [CrossRef] [PubMed]
- Bener, A.; Al-Hamaq, A.; Abdulhadi, K.; Salahaldin, A.H.; Gansan, L. Interaction between diabetes mellitus and hypertension on risk of hearing loss in highly endogamous population. Diabetes Metab. Syndr. 2017, 11, S45–S51. [Google Scholar] [CrossRef] [PubMed]
- Lin, B.M.; Curhan, S.G.; Wang, M.; Eavey, R.; Stankovic, K.M.; Curhan, G.C. Hypertension, diuretic use, and risk of hearing loss. Am. J. Med. 2016, 129, 416–422. [Google Scholar] [CrossRef] [PubMed]
- Gianfredi, V.; Salvatori, T.; Nucci, D.; Villarini, M.; Moretti, M. Can chocolate consumption reduce cardio-cerebrovascular risk? A systematic review and meta-analysis. Nutrition 2018, 46, 103–114. [Google Scholar] [CrossRef] [PubMed]
- Williamson, G. Bioavailability and health effects of cocoa polyphenols. Inflammopharmacology 2009, 17, 111. [Google Scholar] [CrossRef]
- Vauzour, D.; Rodriguez-Mateos, A.; Corona, G.; Oruna-Concha, M.J.; Spencer, J.P. Polyphenols and human health: Prevention of disease and mechanisms of action. Nutrients 2010, 2, 1106–1131. [Google Scholar] [CrossRef] [PubMed]
- Baguley, D.M. Mechanisms of tinnitus. Br. Med. Bull. 2002, 63, 195–212. [Google Scholar] [CrossRef]
- Zheng, Y.; Stiles, L.; Chien, Y.T.; Darlington, C.L.; Smith, P.F. The effects of acute stress-induced sleep disturbance on acoustic trauma-induced tinnitus in rats. Biomed. Res. Int. 2014, 2014, 724195. [Google Scholar] [CrossRef] [PubMed]
- Gibrin, P.C.; Melo, J.J.; Marchiori, L.L. Prevalence of tinnitus complaints and probable association with hearing loss, diabetes mellitus and hypertension in elderly. Codas 2013, 25, 176–180. [Google Scholar] [CrossRef]
- McCormack, A.; Edmondson-Jones, M.; Mellor, D.; Dawes, P.; Munro, K.J.; Moore, D.R.; Fortnum, H. Association of dietary factors with presence and severity of tinnitus in a middle-aged UK population. PLoS ONE 2014, 9, e114711. [Google Scholar] [CrossRef] [PubMed]
- Steinberg, F.M.; Bearden, M.M.; Keen, C.L. Cocoa and chocolate flavonoids: Implications for cardiovascular health. J. Am. Diet. Assoc. 2003, 103, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Li, A.-N.; Li, S.; Zhang, Y.-J.; Xu, X.-R.; Chen, Y.-M.; Li, H.-B. Resources and biological activities of natural polyphenols. Nutrients 2014, 6, 6020–6047. [Google Scholar] [CrossRef] [PubMed]
- Maas, I.L.; Bruggemann, P.; Requena, T.; Bulla, J.; Edvall, N.K.; Hjelmborg, J.V.B.; Szczepek, A.J.; Canlon, B.; Mazurek, B.; Lopez-Escamez, J.A.; et al. Genetic susceptibility to bilateral tinnitus in a Swedish twin cohort. Genet. Med. 2017, 19, 1007–1012. [Google Scholar] [CrossRef] [PubMed]
| Chocolate Consumption | p-Value 1 | |||
|---|---|---|---|---|
| Total (n = 3575) | More Than Once (n = 1262) | None (n = 2313) | ||
| Hearing loss 2, n (%) | ||||
| Unilateral or bilateral | 1170 (32.73%) | 338 (26.78%) | 832 (35.97%) | <0.0001 |
| Bilateral | 638 (17.85%) | 168 (13.31%) | 470 (20.32%) | <0.0001 |
| High-tone hearing loss | 2122 (59.36%) | 651 (51.58%) | 1471 (63.60%) | <0.001 |
| Tinnitus, n (%) | 811 (22.69%) | 266 (21.08%) | 545 (23.56%) | 0.0947 |
| Tinnitus-related annoyance, n (%) | 259 (7.24%) | 78 (6.18%) | 181 (7.83%) | 0.0790 |
| Age (year), median (interquartile range, IQR) | 52 (45, 58) | 50 (44, 56) | 53 (47, 59) | <0.0001 |
| Male, n (%) | 1415 (39.58%) | 493 (39.06%) | 922 (39.86%) | 0.6677 |
| Monthly household income 3, median (IQR) | 333.33 (200, 518.33) | 400 (250, 591.67) | 310.67 (184.17, 500) | <0.0001 |
| Use of earphones, n (%) | 186 (5.20%) | 90 (7.13%) | 96 (4.15%) | 0.0002 |
| Duration of earphone use (min), median (IQR) | ||||
| Total | 0 (0, 0) | 0 (0, 0) | 0 (0, 0) | 0.0002 |
| User of earphones | 60 (30, 60) | 30 (20, 60) | 60 (30, 90) | 0.0617 |
| Occupational exposure to noise, n (%) | 587 (16.42%) | 186 (14.74%) | 401 (17.34%) | 0.0473 |
| Duration of occupational exposure to noise (months), median (IQR) | ||||
| Total | 0 (0, 0) | 0 (0, 0) | 0 (0, 0) | 0.0448 |
| Occupational exposure to noise | 96 (36, 216) | 96 (36, 216) | 108 (36, 216) | 0.8920 |
| Sleep duration (hours), n (%) | 0.0295 | |||
| <6 | 504 (14.1%) | 163 (12.92%) | 341 (14.74%) | |
| 6–7 | 1,039 (29.06%) | 377 (29.87%) | 662 (28.62%) | |
| 7–8 | 1,099 (30.74%) | 418 (33.12%) | 681 (29.44%) | |
| ≥8 | 933 (26.1%) | 304 (24.09%) | 629 (27.19%) | |
| High perceived stress, n (%) | 770 (21.54%) | 274 (21.71%) | 496 (21.44%) | 0.8648 |
| Exposure to indoor second-hand smoke | ||||
| At work, n (%) | 1162 (32.5%) | 418 (33.12%) | 744 (32.17%) | 0.5753 |
| At home, n (%) | 349 (9.76%) | 123 (9.75%) | 226 (9.77%) | >0.9999 |
| Current smoking, n (%) | 653 (18.27%) | 203 (16.09%) | 450 (19.46%) | 0.0128 |
| Heavy drinking 4, n (%) | 665 (18.60%) | 193 (15.29%) | 472 (20.41%) | 0.0002 |
| Difficulties in controlling alcohol use, n (%) | 294 (8.22%) | 87 (6.89%) | 207 (8.95%) | 0.0354 |
| Having drinking-related problem in life, n (%) | 165 (4.62%) | 56 (4.44%) | 109 (4.71%) | 0.7393 |
| Menopause (females) | <0.0001 | |||
| Yes | 1209 (55.97%) | 372 (48.37%) | 837 (60.17%) | |
| No | 951 (44.03%) | 397 (51.63%) | 554 (39.83%) | |
| Hypertension, n (%) | 683 (19.10%) | 196 (15.53%) | 487 (21.05%) | <0.0001 |
| Diabetes mellitus, n (%) | 247 (6.91%) | 60 (4.75%) | 187 (8.08%) | 0.0001 |
| Anemia, n (%) | 284 (7.94%) | 115 (9.11%) | 169 (7.31%) | 0.0606 |
| Kidney failure, n (%) | 13 (0.36%) | 6 (0.48%) | 7 (0.30%) | 0.4004 |
| Thyroid disorder, n (%) | 85 (2.38%) | 35 (2.77%) | 50 (2.16%) | 0.2527 |
| Dyslipidemia, n (%) | 331 (9.26%) | 87 (6.89%) | 244 (10.55%) | 0.0003 |
| n = 3575 | Univariate Analysis | Multivariate Analysis | |||
|---|---|---|---|---|---|
| OR (95% CI) | p-Value | OR (95% CI) | p-Value | ||
| Hearing loss (unilateral or bilateral) | 1170: 2405 | 0.651 (0.560, 0.757) | <0.0001 | 0.829 (0.701, 0.980) 1 | 0.0285 |
| Hearing loss (bilateral) | 638: 2937 | 0.602 (0.497, 0.729) | <0.0001 | 0.791 (0.641, 0.976) 1 | 0.0287 |
| High-tone hearing loss | 2122: 1453 | 0.610 (0.531, 0.701) | <0.0001 | 0.777 (0.661, 0.912) 1 | 0.0021 |
| Tinnitus | 811: 2764 | 0.866 (0.734, 1.023) | 0.0902 | 0.911 (0.767, 1.081) 2 | 0.2847 |
| Tinnitus-related annoyance | 259: 3316 | 0.776 (0.590, 1.021) | 0.0705 | 0.886 (0.668, 1.176) 2 | 0.4036 |
| Multivariable Analysis | |||
|---|---|---|---|
| Consumption Per Week (Reference = No) | OR (95% CI) | p-Value | |
| Hearing loss (unilateral or bilateral) | Chocolate | 0.835 (0.703, 0.992) 1 | 0.0406 |
| Cookie | 0.989 (0.973, 1.006) 1 | 0.1912 | |
| Ice cream | 1.055 (0.967, 1.151) 1 | 0.2308 | |
| Cake | 0.975 (0.861, 1.105) 1 | 0.6931 | |
| Hearing loss (bilateral) | Chocolate | 0.766 (0.617, 0.951) 1 | 0.0156 |
| Cookie | 0.989 (0.968, 1.010) 1 | 0.3043 | |
| Ice cream | 1.146 (1.039, 1.265) 1 | 0.0063 | |
| Cake | 1.041 (0.901, 1.203) 1 | 0.5826 | |
| Tinnitus | Chocolate | 0.920 (0.772, 1.097) 2 | 0.3536 |
| Cookie | 1.001 (0.985, 1.018) 2 | 0.8901 | |
| Ice cream | 1.009 (0.922, 1.104) 2 | 0.8478 | |
| Cake | 0.932 (0.814, 1.067) 2 | 0.3081 | |
| Tinnitus-related annoyance | Chocolate | 0.876 (0.655, 1.172) 2 | 0.3735 |
| Cookie | 0.987 (0.956, 1.020) 2 | 0.4431 | |
| Ice cream | 1.120 (0.990, 1.267) 2 | 0.0721 | |
| Cake | 0.973 (0.787, 1.204) 2 | 0.8028 | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, S.-Y.; Jung, G.; Jang, M.-j.; Suh, M.-W.; Lee, J.h.; Oh, S.-H.; Park, M.K. Association of Chocolate Consumption with Hearing Loss and Tinnitus in Middle-Aged People Based on the Korean National Health and Nutrition Examination Survey 2012–2013. Nutrients 2019, 11, 746. https://doi.org/10.3390/nu11040746
Lee S-Y, Jung G, Jang M-j, Suh M-W, Lee Jh, Oh S-H, Park MK. Association of Chocolate Consumption with Hearing Loss and Tinnitus in Middle-Aged People Based on the Korean National Health and Nutrition Examination Survey 2012–2013. Nutrients. 2019; 11(4):746. https://doi.org/10.3390/nu11040746
Chicago/Turabian StyleLee, Sang-Yeon, Gucheol Jung, Myoung-jin Jang, Myung-Whan Suh, Jun ho Lee, Seung-Ha Oh, and Moo Kyun Park. 2019. "Association of Chocolate Consumption with Hearing Loss and Tinnitus in Middle-Aged People Based on the Korean National Health and Nutrition Examination Survey 2012–2013" Nutrients 11, no. 4: 746. https://doi.org/10.3390/nu11040746
APA StyleLee, S.-Y., Jung, G., Jang, M.-j., Suh, M.-W., Lee, J. h., Oh, S.-H., & Park, M. K. (2019). Association of Chocolate Consumption with Hearing Loss and Tinnitus in Middle-Aged People Based on the Korean National Health and Nutrition Examination Survey 2012–2013. Nutrients, 11(4), 746. https://doi.org/10.3390/nu11040746





