Comparison of Pro-Regenerative Effects of Carbohydrates and Protein Administrated by Shake and Non-Macro-Nutrient Matched Food Items on the Skeletal Muscle after Acute Endurance Exercise
Abstract
1. Introduction
2. Methods
2.1. Participants
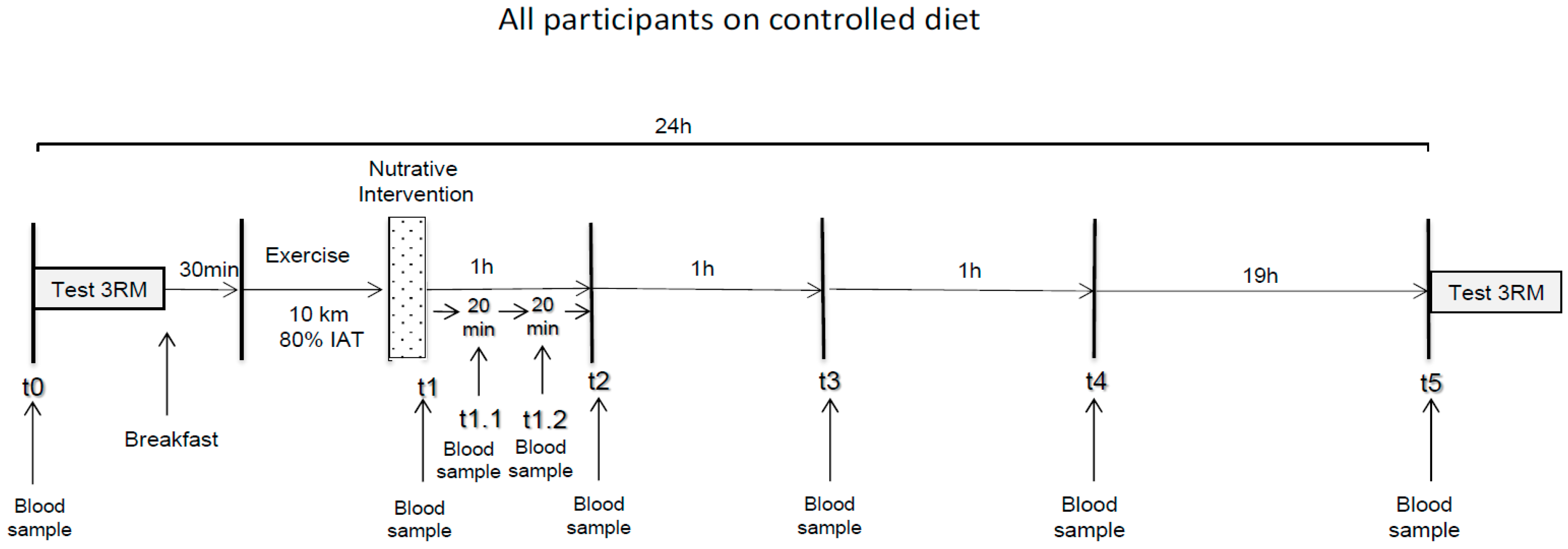
2.2. Experimental Procedure
3. Measurements
3.1. Determination of Serum Glucose and Serum Insulin
3.2. Determination of Serum Cortisol
3.3. Hematology
3.4. Skeletal Muscle Creatine Kinase (CK mm) and Myoglobin (Myo)
3.5. Serum Cytokine Levels
3.6. Leg Strength—3-RM Back Squat
3.7. Statistical Analyses
4. Results
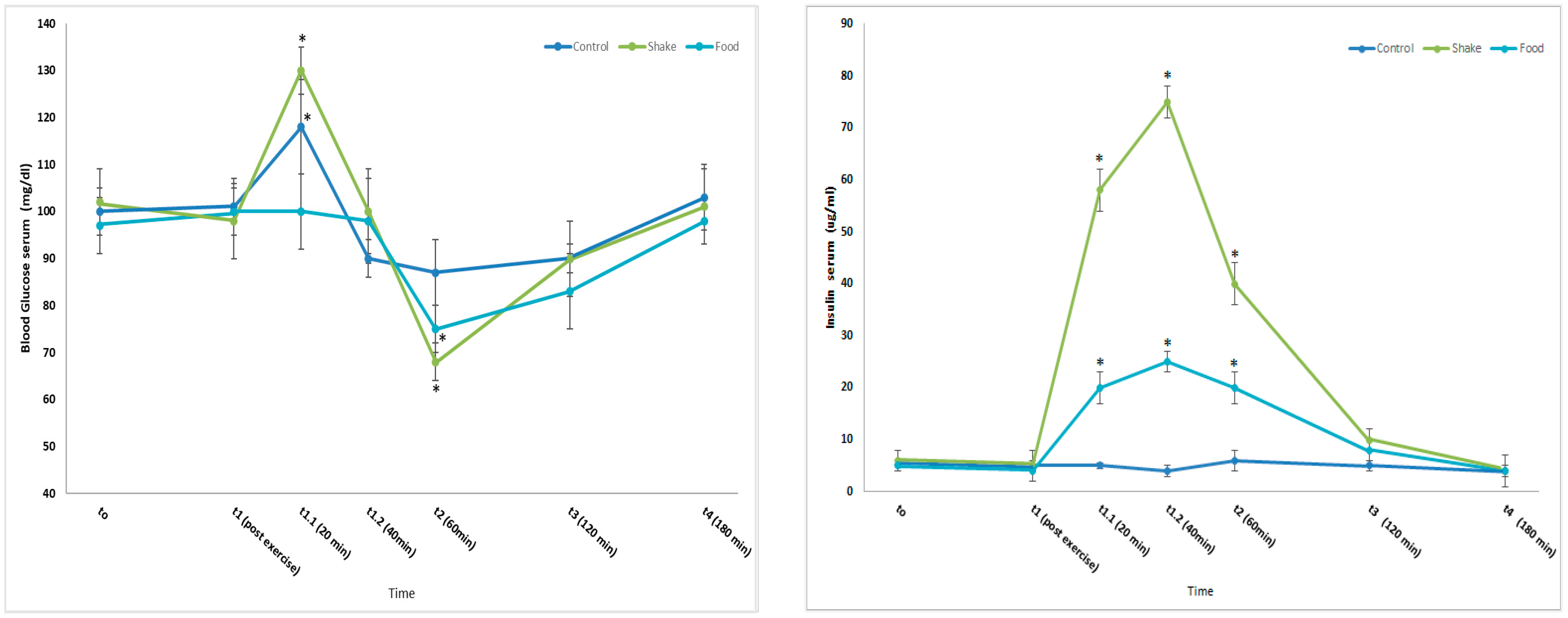
4.1. Effects of Exercise and Protein/Carbohydrate on Blood Glucose and Insulin Concentrations
4.2. Effects of Exercise and Protein/Carbohydrate on Hematopoietic Parameters
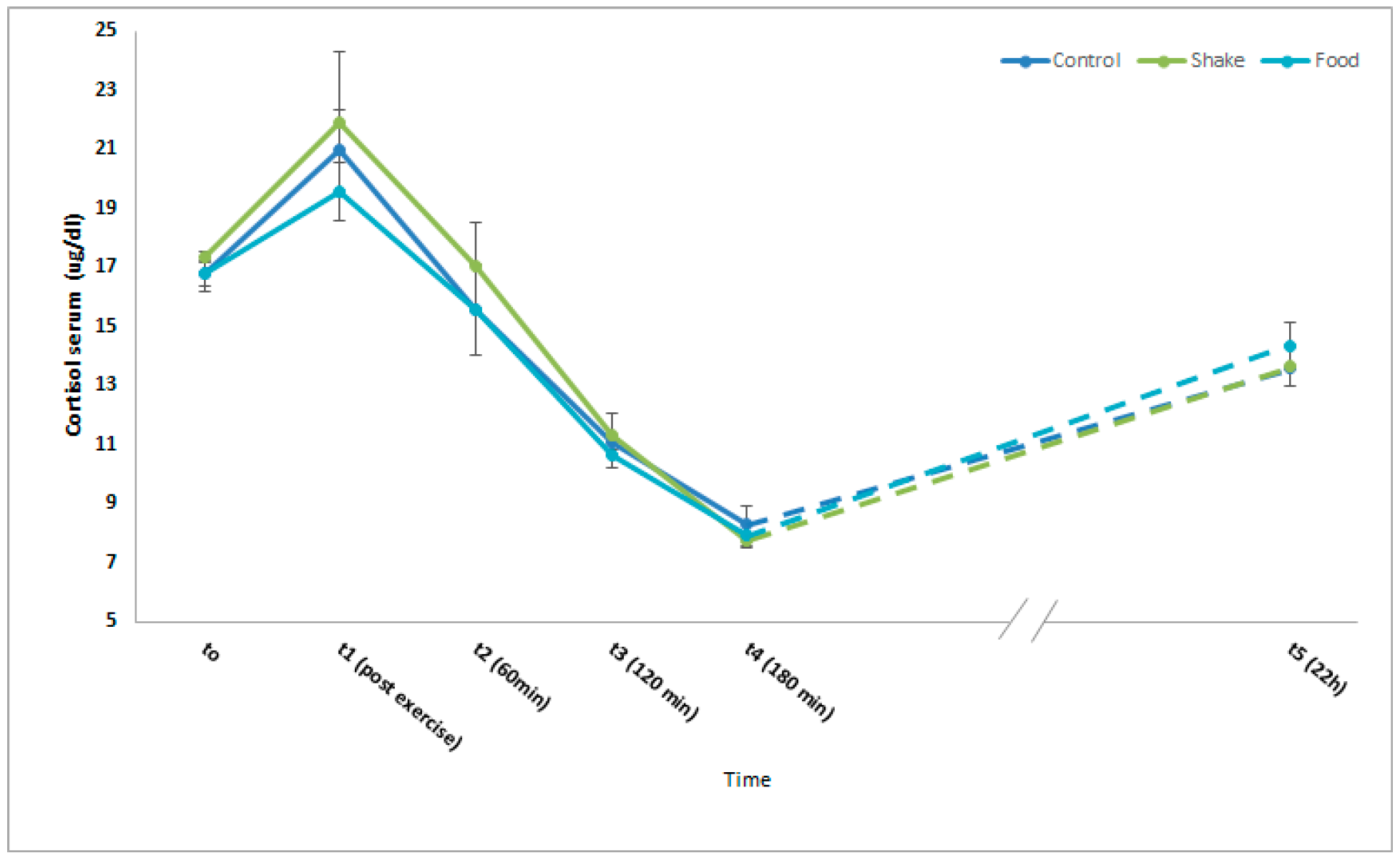
4.3. Effects of Exercise and Protein/Carbohydrate on Blood Cortisol Levels
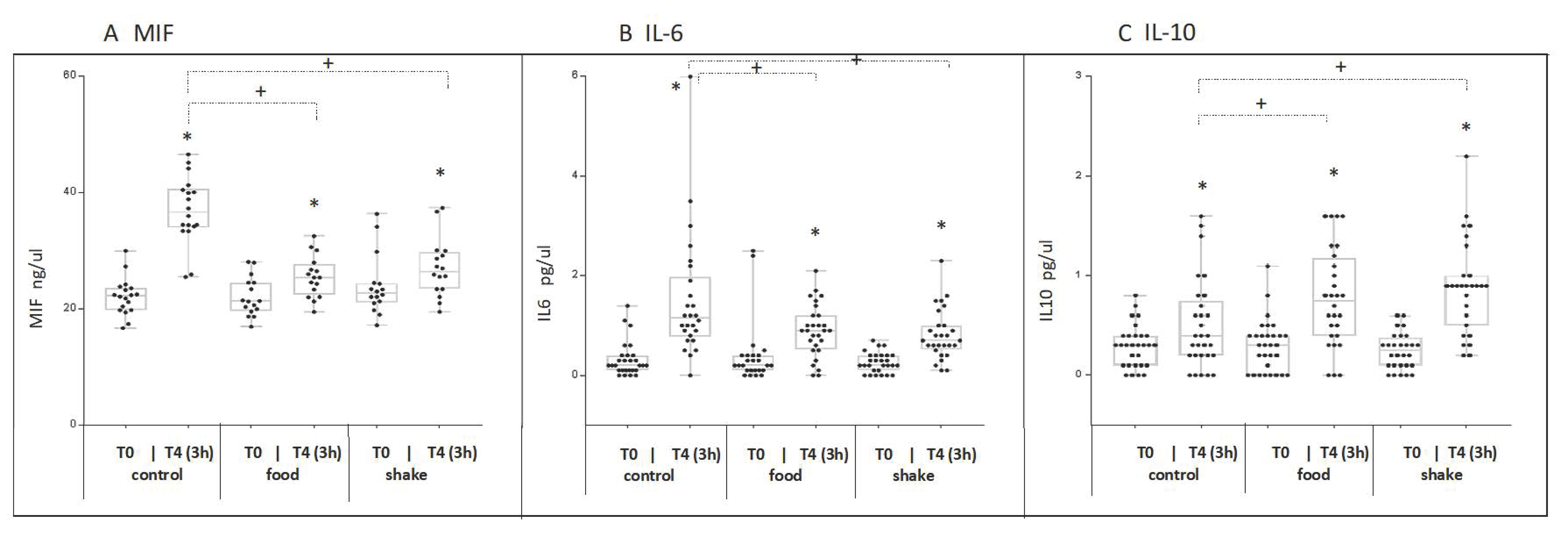
4.4. Effects of Exercise and Protein/Carbohydrate on Markers of Inflammation
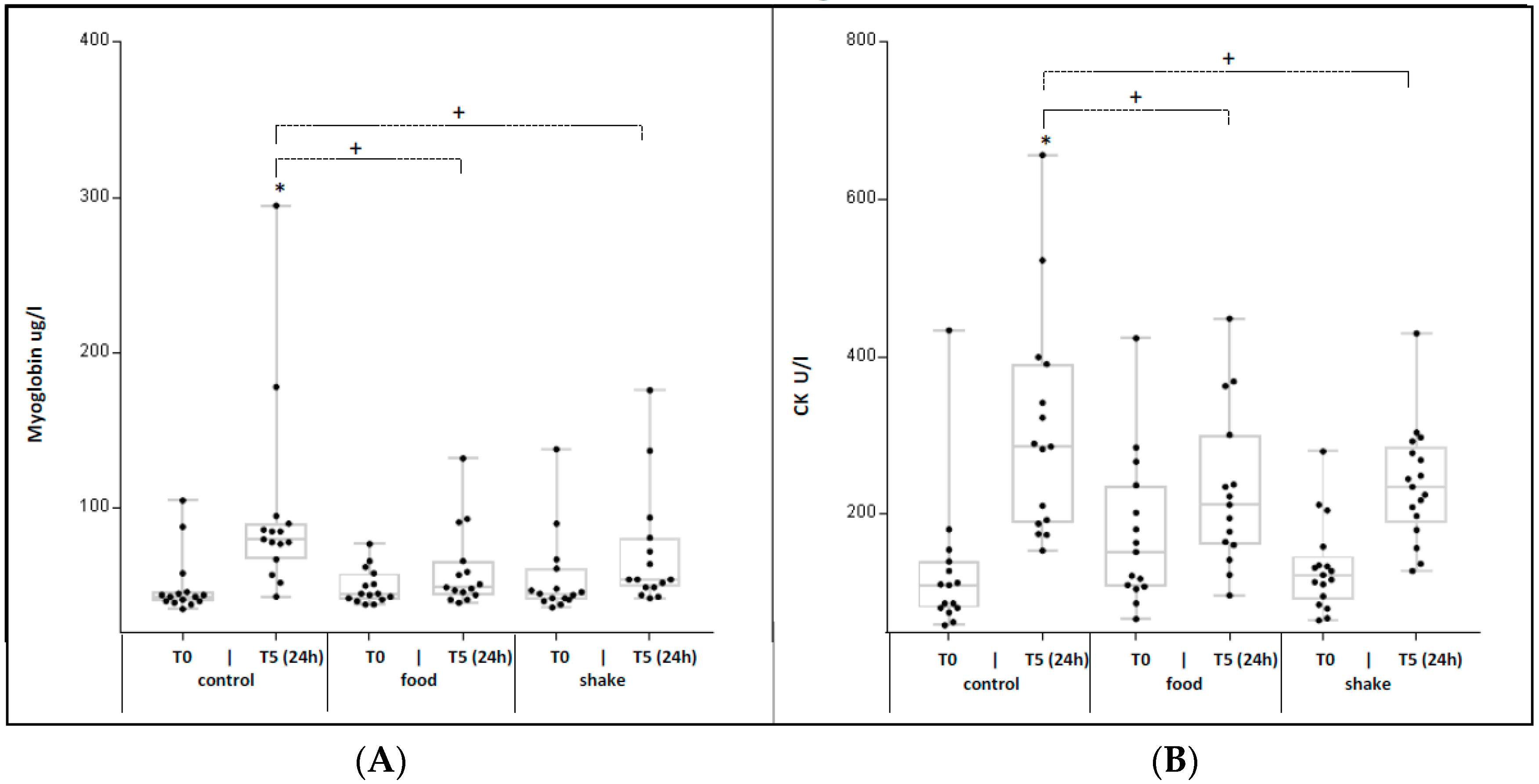
4.5. Effects of Exercise and Protein/Carbohydrate on Serum Markers for Skeletal Muscle Damage
4.6. Effects of Exercise and Protein/Carbohydrate on Leg Strength as a Functional Marker for Skeletal Muscle Regeneration
5. Discussion
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ANS | anaerobic threshold |
| CK | Creatine kinase |
| CKmm | Creatine kinase skeletal muscle |
| Myo | Myoglobin |
| IL 10 | Interleukin 10 |
| IL 6 | Interleukin 6 |
| MIF | Macrophage migration inhibitor factor |
| SD | standard deviations |
| SEM | standard error of the mean |
| TNF alpha | Tumor necrosis receptor alpha |
| km | kilometer |
| IGF 1 | insulin like growth factor 1 |
| mTOR | mechanistic Target of Rapamycin |
References
- Maughan, R.J.; Depiesse, F.; Geyer, H. The use of dietary supplements by athletes international association of athletics federations. J. Sports Sci. 2007, 25, 103–113. [Google Scholar] [CrossRef]
- Reidy, P.T.; Rasmussen, B.B. Role of Ingested amino acids and protein in the promotion of resistance exercise-induced muscle protein anabolism. J. Nutr. 2016, 146, 155–183. [Google Scholar] [CrossRef]
- Bonaldo, P.; Sandri, M. Cellular and molecular mechanisms of muscle atrophy. Dis. Models Mech. 2013, 6, 25–39. [Google Scholar] [CrossRef]
- Armstrong, R.B.; Warren, G.L.; Warren, J.A. Mechanisms of exercise-induced muscle fibre injury. Sports Med. 1991, 12, 184–207. [Google Scholar] [CrossRef]
- Trappe, T.A.; White, F.; Lambert, C.P.; Cesar, D.; Hellerstein, M.; Evans, W.J. Effect of ibuprofen and acetaminophen on postexercise muscle protein synthesis. Am. J. Physiol. Endocrinol. Metab. 2002, 282, 551–556. [Google Scholar] [CrossRef]
- Koopman, R.; Wagenmakers, A.J.; Manders, R.J.; Zorenc, A.H.; Senden, J.M.; Gorselink, M.; Keizer, H.A.; van Loon, L.J. Combined ingestion of protein and free leucine with carbohydrate increases postexercise muscle protein synthesis in vivo in male subjects. Am. J. Physiol. Endocrinol. Metab. 2005, 288, 645–653. [Google Scholar] [CrossRef]
- Mackley, A.L.; Rasmussen, L.K.; Kadi, F.; Schjerling, P.; Helmark, I.C.; Ponsot, E.; Aagaard, P.; Durigan, J.L.; Kjaer, M. Activation of satellite cells and the regeneration of human skeletal muscle are expedited by ingestion of nonsteroidal anti-inflammatory medication. FASEB J. 2016, 30, 2266–2281. [Google Scholar] [CrossRef]
- Phillips, S.M.; Tipton, K.D.; Aarsland, A.; Wolf, S.E.; Wolfe, R.R. Mixed muscle protein synthesis and breakdown after resistance exercise in humans. Am. J. Physiol. 1997, 273, 9–107. [Google Scholar] [CrossRef]
- Kitajima, Y.; Tashiro, Y.; Suzuki, N.; Warita, H.; Kato, M.; Tateyama, M.; Ando, R.; Izumi, R.; Yamazaki, M.; Abe, M.; et al. Proteasome dysfunction induces muscle growth defects and protein aggregation. J. Cell Sci. 2014, 127, 5204–5217. [Google Scholar] [CrossRef]
- Sandri, M. Signaling in muscle atrophy and hypertrophy. Physiology 2008, 23, 160–170. [Google Scholar] [CrossRef]
- Tang, J.E.; Moore, D.R.; Kujbida, G.W.; Tarnopolsky, M.A.; Phillips, S.M. Ingestion of whey hydrolysate, casein, or soy protein isolate: Effects on mixed muscle protein synthesis at rest and following resistance exercise in young men. J. Appl. Physiol. 2009, 107, 987–992. [Google Scholar] [CrossRef]
- Symons, T.B.; Sheffield-Moore, M.; Wolfe, R.R.; Paddon-Jones, D. A moderate serving of high-quality protein maximally stimulates skeletal muscle protein synthesis in young and elderly subjects. J. Am. Diet. Assoc. 2009, 109, 1582–1586. [Google Scholar] [CrossRef]
- Markus, C.R.; Olivier, B.; de Haan, E.H. Whey protein rich in α-lactalbumin increases the ratio of plasma tryptophan to the sum of the other large neutral amino acids and improves cognitive performance in stress-vulnerable subjects. Am. J. Clin. Nutr. 2002, 75, 1051–1056. [Google Scholar] [CrossRef]
- White, J.P.; Wilson, J.M.; Austin, K.G.; Greer, B.K.; St John, N.; Panton, L.B. Effect of carbohydrate-protein supplement timing on acute exercise-induced muscle damage. J. Int. Soc. Sports Nutr. 2008, 5, 5. [Google Scholar] [CrossRef]
- Buckley, J.D.; Thomson, R.L.; Coates, A.M.; Howe, P.R.; Denichilo, M.O.; Rowney, M.K. Supplementation with a whey protein hydrolysate enhances recovery of muscle force-generating capacity following eccentric exercise. J. Sci. Med. Sport 2010, 13, 178–181. [Google Scholar] [CrossRef]
- Trommelen, J.; Betz, M.W.; van Loon, L.J.C. The muscle protein synthetic response to meal ingestion following resistance-type exercise. Sports Med. 2019, 49, 185–197. [Google Scholar] [CrossRef]
- Manninen, A.H. Hyperinsulinaemia, hyperaminoacidaemia and postexercise exercise: Influence on performance and recovery. Int. J. Sport Nutr. Exerc. Metab. 2006, 17, 87–103. [Google Scholar]
- Saunders, M.J.; Kane, M.D.; Todd, M.K. Effects of a carbohydrate-protein beverage on cycling endurance and muscle damage. Med. Sci. Sports Exerc. 2004, 36, 1233–1238. [Google Scholar] [CrossRef]
- Kerksick, C.; Harvey, T.; Stout, J.; Campbell, B.; Wilborn, C.; Kreider, R.; Antonio, J. International society of sports nutrition position stand: Nutrient timing. J. Int. Soc. Sports Nutr. 2008, 5, 17. [Google Scholar] [CrossRef]
- Valentine, R.J.; Saunders, M.J.; Todd, M.K.; Laurent, T.G. Influence of carbohydrate-protein beverage on cycling endurance and indices of muscle disruption. Int. J. Sport Nutr. Exerc. Metab. 2008, 18, 363–378. [Google Scholar] [CrossRef]
- Hill, K.M.; Stathis, C.G.; Grinfeld, E.; Hayes, A.; McAinch, A.J. Coingestion of carbohydrate and whey protein isolates enhance PGC-1α mRNA expression: A randomised, single blind, cross over study. J. Int. Soc. Sports Nutr. 2013, 10, 8. [Google Scholar] [CrossRef]
- Staples, A.W.; Burd, N.A.; West, D.W.; Currie, K.D.; Atherton, P.J.; Moore, D.R.; Rennie, M.J.; Macdonald, M.J.; Baker, S.K.; Phillips, S.M. Carbohydrate does not augment exercise-induced protein accretion versus protein alone. Med. Sci. Sports Exerc. 2011, 43, 1154–1161. [Google Scholar] [CrossRef] [PubMed]
- van Loon, L.J.; Saris, W.H.; Verhagen, H.; Wagenmakers, A.J. Plasma insulin responses after ingestion of different amino acid or protein mixtures with carbohydrate. Am. J. Clin. Nutr. 2000, 72, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Behrends, C.; Sowa, M.E.; Gygi, S.P.; Harper, J.W. Network organization of the human autophagy system. Nature 2010, 466, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Anthony, J.C.; Lang, C.H.; Crozier, S.J.; Anthony, T.G.; MacLean, D.A.; Kimball, S.R.; Jefferson, L.S. Contribution of insulin to the translational control of protein synthesis in skeletal muscle by leucine. Am. J. Physiol. Endocrinol. Metab. 2002, 282, 1092–1101. [Google Scholar] [CrossRef]
- Fujita, S.; Rasmussen, B.B.; Cadenas, J.G.; Grady, J.J.; Volpi, E. Effect of insulin on human skeletal muscle protein synthesis is modulated by insulin-induced changes in muscle blood flow and amino acid availability. Am. J. Physiol. Endocrinol. Metab. 2006, 291, 745–754. [Google Scholar] [CrossRef] [PubMed]
- Ivy, J.L.; Goforth, H.W., Jr.; Damon, B.M.; McCauley, T.R.; Parsons, E.C.; Price, T.B. Early postexercise muscle glycogen recovery is enhanced with a carbohydrate-protein supplement. J. Appl. Physiol. 2002, 93, 1337–1344. [Google Scholar] [CrossRef] [PubMed]
- Diel, P.; Le Viet, D.; Humm, J.; Huss, J.; Oderkerk, T.; Simon, W.; Geisler, S. Effects of a nutritive administration of carbohydrates and protein by foodstuffs on skeletal muscle inflammation and damage after acute endurance exercise. J. Nutr. Health Food Sci. 2017, 5, 17. [Google Scholar] [CrossRef]
- Mader, A.; Liesen, H.; Heck, H.; Phillipi, H.; Rost, R.; Schürch, P. Zur beurteilung der sportartspezifischen ausdauerleistungsfähigkeit im labor. Sportarzt Sportmed. 1976, 27, 80–112. [Google Scholar]
- Nicholson, R.M.; Seivert, G.G. Indices of lactate threshold and their relationship with 10-km running velocity. Med. Sci. Sports Exerc. 2001, 33, 339–342. [Google Scholar] [CrossRef] [PubMed]
- Todd, M.; Haywood, K.M.; Roberton, M.A.; Getchell, N. National Strength & Conditioning Association, SA’s Guide to Tests and Assessments; Human Kinetics: Champaign, IL, USA, 2012. [Google Scholar]
- Johannsen, N.M.; Swift, D.L.; Johnson, W.D.; Dixit, V.D.; Earnest, C.P.; Blair, S.N.; Church, T.S. Effect of different doses of aerobic exercise on total white blood cell (WBC) and WBC subfraction number in postmenopausal women: Results from DREW. PLoS ONE 2012, 7, E31319. [Google Scholar] [CrossRef]
- Lima-Silva, A.E.; Pires, F.O.; Lira, F.S.; Casarini, D.; Kiss, M.A. Low carbohydrate diet affects the oxygen uptake on-kinetics and rating of perceived exertion in high intensity exercise. Psychophysiology 2011, 48, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Adams, P. The impact of brief high-intensity exercise on blood glucose levels. Diabetes Metab. Syndr. Obes. 2013, 6, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Ang, M.; Müller, A.S.; Wagenlehner, F.; Pilatz, A.; Linn, T. Combining protein and carbohydrate increases postprandial insulin levels but does not improve glucose response in patients with type 2 diabetes. Metabolism 2012, 61, 1696–1702. [Google Scholar] [CrossRef]
- Beiter, T.; Hoene, M.; Prenzler, F.; Mooren, F.C.; Steinacker, J.M.; Weigert, C.; Nieß, A.M.; Munz, B. Exercise, skeletal muscle and inflammation: ARE-binding proteins as key regulators in inflammatory and adaptive networks. Exerc. Immunol. Rev. 2015, 21, 42–57. [Google Scholar]
- Walsh, N.P.; Gleeson, M.; Pyne, D.B.; Nieman, D.C.; Dhabhar, F.S.; Shephard, R.J.; Oliver, S.J.; Bermon, S.; Kajeniene, A. Position statement part two: Maintaining immune health. Exerc. Immunol. Rev. 2011, 17, 64–103. [Google Scholar] [PubMed]
- Petersen, A.M.; Pedersen, B.K. The role of IL-6 in mediating the anti-inflammatory effects of exercise. J. Physiol. Pharmacol. 2006, 57, 43–51. [Google Scholar]
- Velders, M.; Schleipen, B.; Fritzemeier, K.H.; Zierau, O.; Diel, P. Selective estrogen receptor-β activation stimulates skeletal muscle growth and regeneration. FASEB J. 2012, 26, 1909–1920. [Google Scholar] [CrossRef] [PubMed]
- Klein, G.L. The effect of glucocorticoids on bone and muscle. Osteoporos. Sarcopenia. 2015, 1, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Burnley, E.C.D.; Olson, A.N.; Sharp, R.L.; Baier, S.M.; Alekel, D.L. Impact of protein supplements on muscle recovery after exercis—induced muscle soreness. J. Exerc. Sci. Fit. 2010, 8, 89–96. [Google Scholar] [CrossRef]
- Outlaw, J.J.; Wilborn, C.D.; Smith-Ryan, A.E.; Hayward, S.E.; Urbina, S.L.; Taylor, L.W. Effects of a pre-and post-workout protein-carbohydrate supplement in trained crossfit individuals. SpringerPlus 2014, 3, 369. [Google Scholar] [CrossRef] [PubMed]
- Millard-Stafford, M.; Warren, G.L.; Thomas, L.M.; Doyle, J.A.; Snow, T.; Hitchcock, K. Recovery from run training: Efficacy of a carbohydrate-protein beverage? Int. J. Sport Nutr. Exerc. Metab. 2005, 15, 610–624. [Google Scholar] [CrossRef]
- Seifert, J.G.; Kipp, R.W.; Amann, M.; Gazal, O. Muscle damage, fluid ingestion, and energy supplementation during recreational alpine skiing. Int. J. Sport Nutr. Exerc. Metab. 2005, 15, 528–536. [Google Scholar] [CrossRef] [PubMed]
- Saunders, M.J. Coingestion of carbohydrate-protein during endurance muscle anabolism: The search for the optimal recovery drink. Br. J. Sports Med. 2007, 40, 900–905. [Google Scholar]
- Westerblad, H.; Allen, D.G.; Bruton, J.D.; Andrade, F.H.; Lännergren, J. Mechanisms underlying the reduction of isometric force in skeletal muscle fatigue. Acta Physiol. Scand. 1998, 162, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Nocella, M.; Colombini, B.; Benelli, G.; Cecchi, G.; Bagni, M.A.; Bruton, J. Force decline during fatigue is due to both a decrease in the force per individual cross-bridge and the number of cross-bridges. J. Physiol. 2011, 589, 3371–3381. [Google Scholar] [CrossRef] [PubMed]
- Lamb, G.D.; Westerblad, H. Acute effects of reactive oxygen and nitrogen species on the contractile function of skeletal muscle. J. Physiol. 2011, 598, 2119–2127. [Google Scholar] [CrossRef] [PubMed]
- Meyer, D.G.; Kushner, M.A.; Brent, L.J.; Schoenfeld, J.B.; Hugentobler, J.; Lloyd, S.R.; Vermeil, A.; Chu, A.D.; Harbin, J.; McGill, M.S. The back squat: A proposed assessment of functional deficits and technical factors that limit performance. Strength Cond. J. 2014, 36, 4–27. [Google Scholar] [CrossRef] [PubMed]
- Baird, M.F.; Graham, S.M.; Baker, J.S.; Bickerstaff, G.F. Creatine-kinase- and exercise-related muscle damage implications for muscle performance and recovery. J. Nutr Metab. 2012, 2012, 960363. [Google Scholar] [CrossRef]
| Carbohydrates (g) | Protein (g) | Fat (g) | Calories (kcal) | |
|---|---|---|---|---|
| 30 g Cornflakes | 25.2 | 2.1 | 0.3 | 111.9 |
| 250 mL milk (1.5% fat) | 12.0 | 8.5 | 3.8 | 116.2 |
| One banana (100 g) | 27.4 | 1.3 | 0.4 | 118.4 |
| Sum | 64.6 | 11.9 | 4.5 | 346.5 |
| Carbohydrates (g) | Protein (g) | Fat (g) | Calories (kcal) | ||
|---|---|---|---|---|---|
| Food | 76 g white bread and 100 g sour milk cheese | 35.3 | 36.1 | 3.5 | 321 |
| Shake | 45 g Glucose 52 g Whey protein | 45 | 52 | 0 | 386 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Isenmann, E.; Blume, F.; Bizjak, D.A.; Hundsdörfer, V.; Pagano, S.; Schibrowski, S.; Simon, W.; Schmandra, L.; Diel, P. Comparison of Pro-Regenerative Effects of Carbohydrates and Protein Administrated by Shake and Non-Macro-Nutrient Matched Food Items on the Skeletal Muscle after Acute Endurance Exercise. Nutrients 2019, 11, 744. https://doi.org/10.3390/nu11040744
Isenmann E, Blume F, Bizjak DA, Hundsdörfer V, Pagano S, Schibrowski S, Simon W, Schmandra L, Diel P. Comparison of Pro-Regenerative Effects of Carbohydrates and Protein Administrated by Shake and Non-Macro-Nutrient Matched Food Items on the Skeletal Muscle after Acute Endurance Exercise. Nutrients. 2019; 11(4):744. https://doi.org/10.3390/nu11040744
Chicago/Turabian StyleIsenmann, Eduard, Franziska Blume, Daniel A. Bizjak, Vera Hundsdörfer, Sarah Pagano, Sebastian Schibrowski, Werner Simon, Lukas Schmandra, and Patrick Diel. 2019. "Comparison of Pro-Regenerative Effects of Carbohydrates and Protein Administrated by Shake and Non-Macro-Nutrient Matched Food Items on the Skeletal Muscle after Acute Endurance Exercise" Nutrients 11, no. 4: 744. https://doi.org/10.3390/nu11040744
APA StyleIsenmann, E., Blume, F., Bizjak, D. A., Hundsdörfer, V., Pagano, S., Schibrowski, S., Simon, W., Schmandra, L., & Diel, P. (2019). Comparison of Pro-Regenerative Effects of Carbohydrates and Protein Administrated by Shake and Non-Macro-Nutrient Matched Food Items on the Skeletal Muscle after Acute Endurance Exercise. Nutrients, 11(4), 744. https://doi.org/10.3390/nu11040744






