Palatability of Goat’s versus Cow’s Milk: Insights from the Analysis of Eating Behavior and Gene Expression in the Appetite-Relevant Brain Circuit in Laboratory Animal Models
Abstract
:1. Introduction
2. Materials and Methods
2.1. Animals
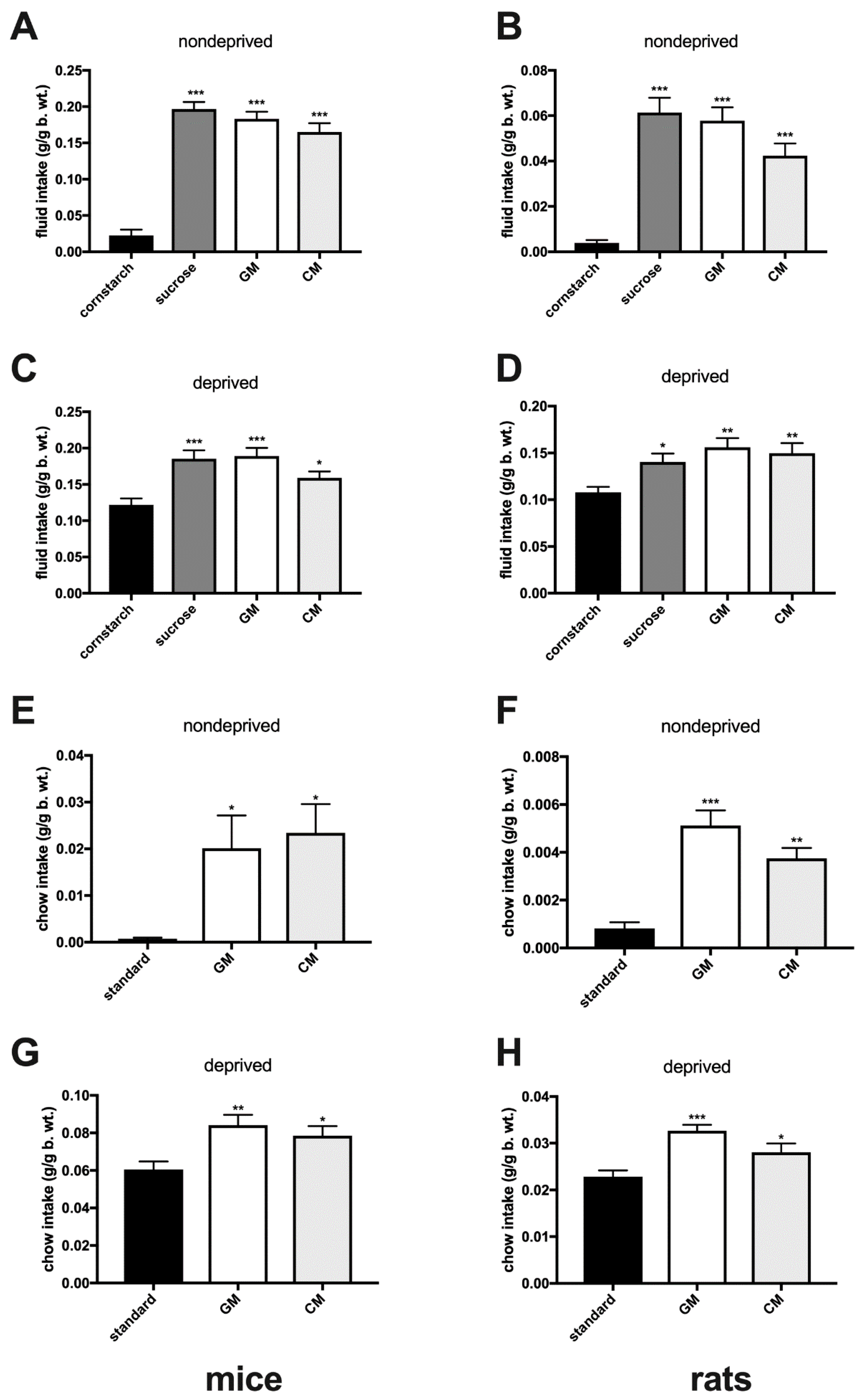
2.2. Episodic Intake of Individually Presented GM and CM in Sated Adult Mice and Rats
2.3. Energy Deprivation-Induced Intake of Individually Presented GM and CM in Mice and Rats
2.4. Episodic Intake of Individually Presented GM- and CM-Enriched Chow in Sated Adult Mice and Rats
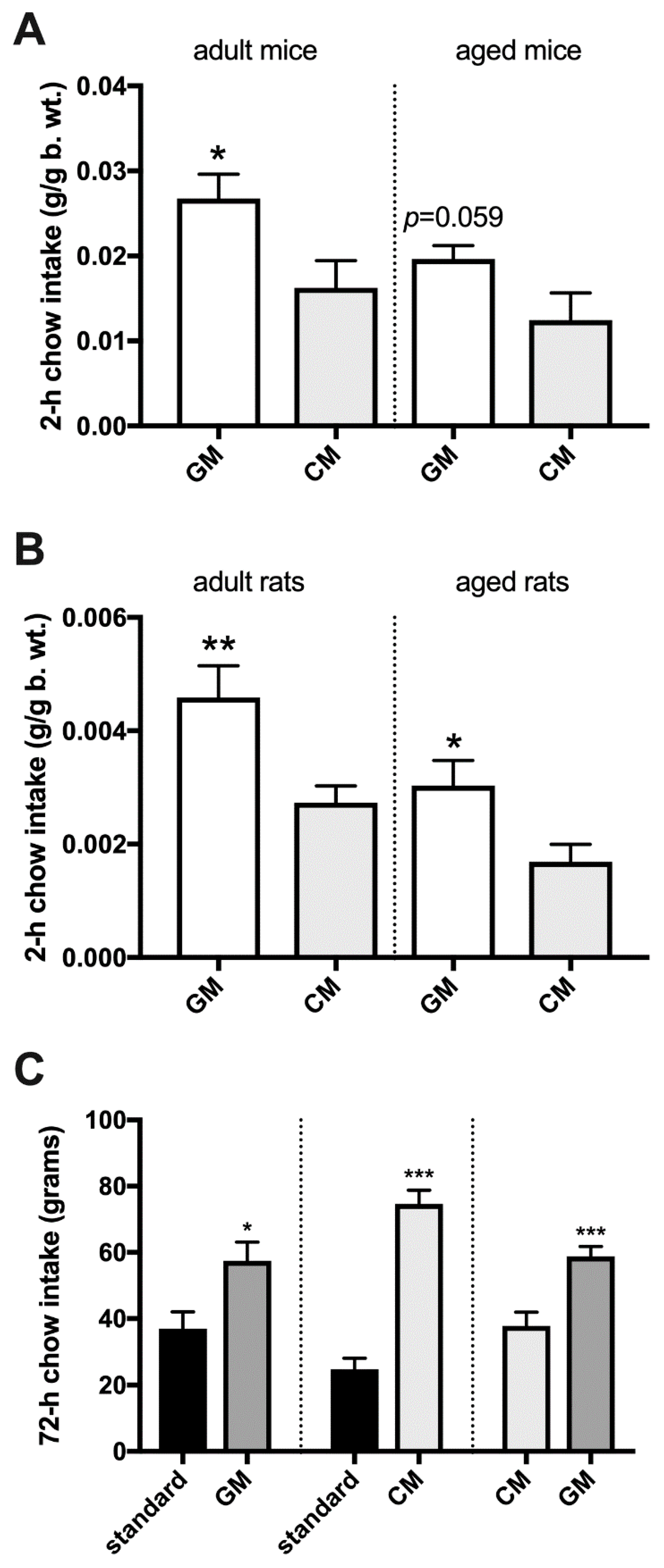
2.5. Energy Deprivation-Induced Intake of Individually Presented GM- and CM-Enriched Chow in Sated Adult Mice and Rats
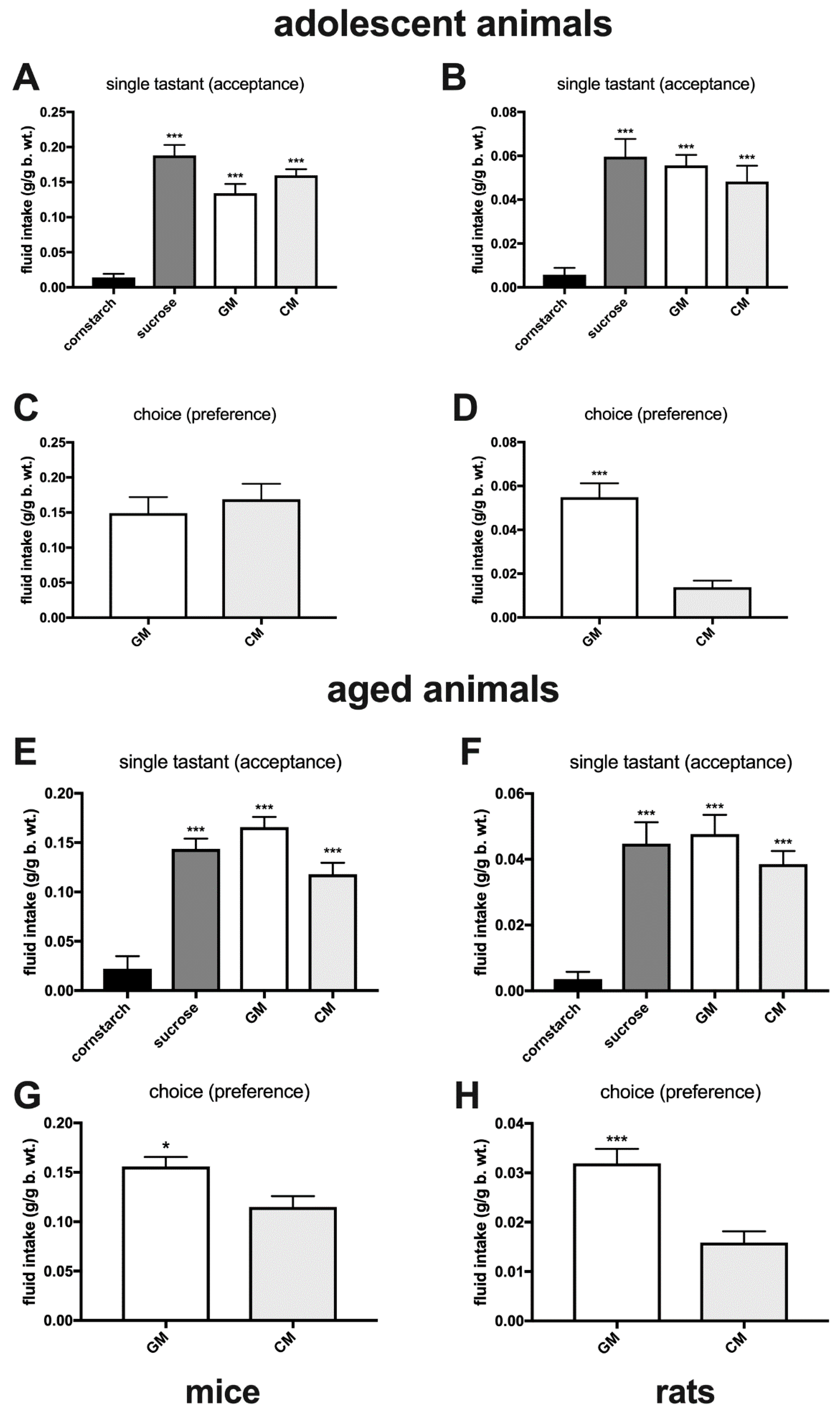
2.6. Episodic Intake of Individually Presented GM and CM in Sated Adolescent and Aged Rodents
2.7. Episodic Intake of GM and CM Presented Simultaneously in Sated Adolescent, Adult, and Aged Rodents
2.8. Episodic Intake of GM- and CM-Enriched Chow Presented Simultaneously in Sated Adult and Aged Rodents
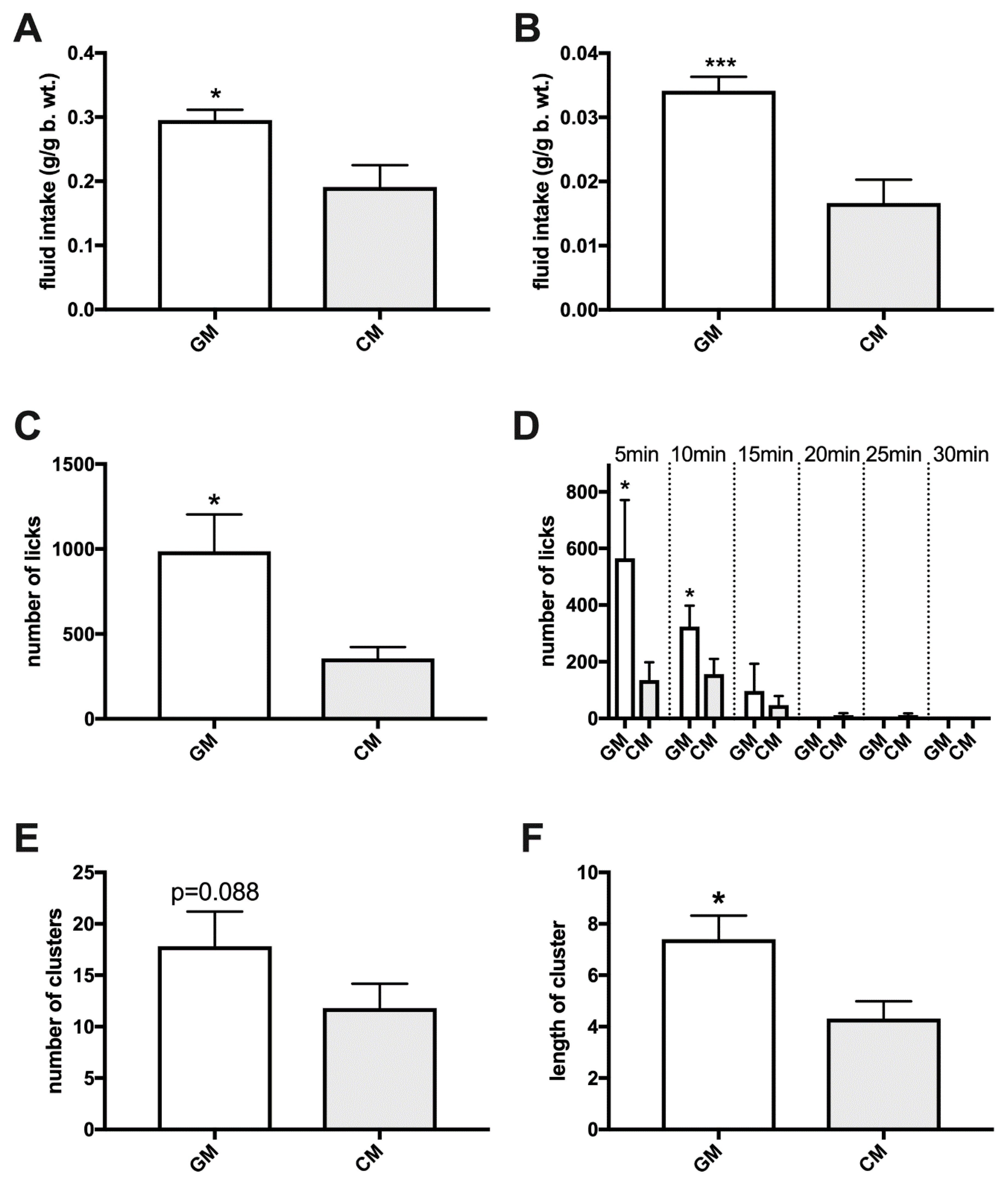
2.9. Lickometer-Assessed Preference for Simultaneously Presented GM and CM in Sated Adult Rats
2.10. 72-h Cumulative Intake of Simultaneously Presented CM- and GM-Enriched Chow in Adult Rats
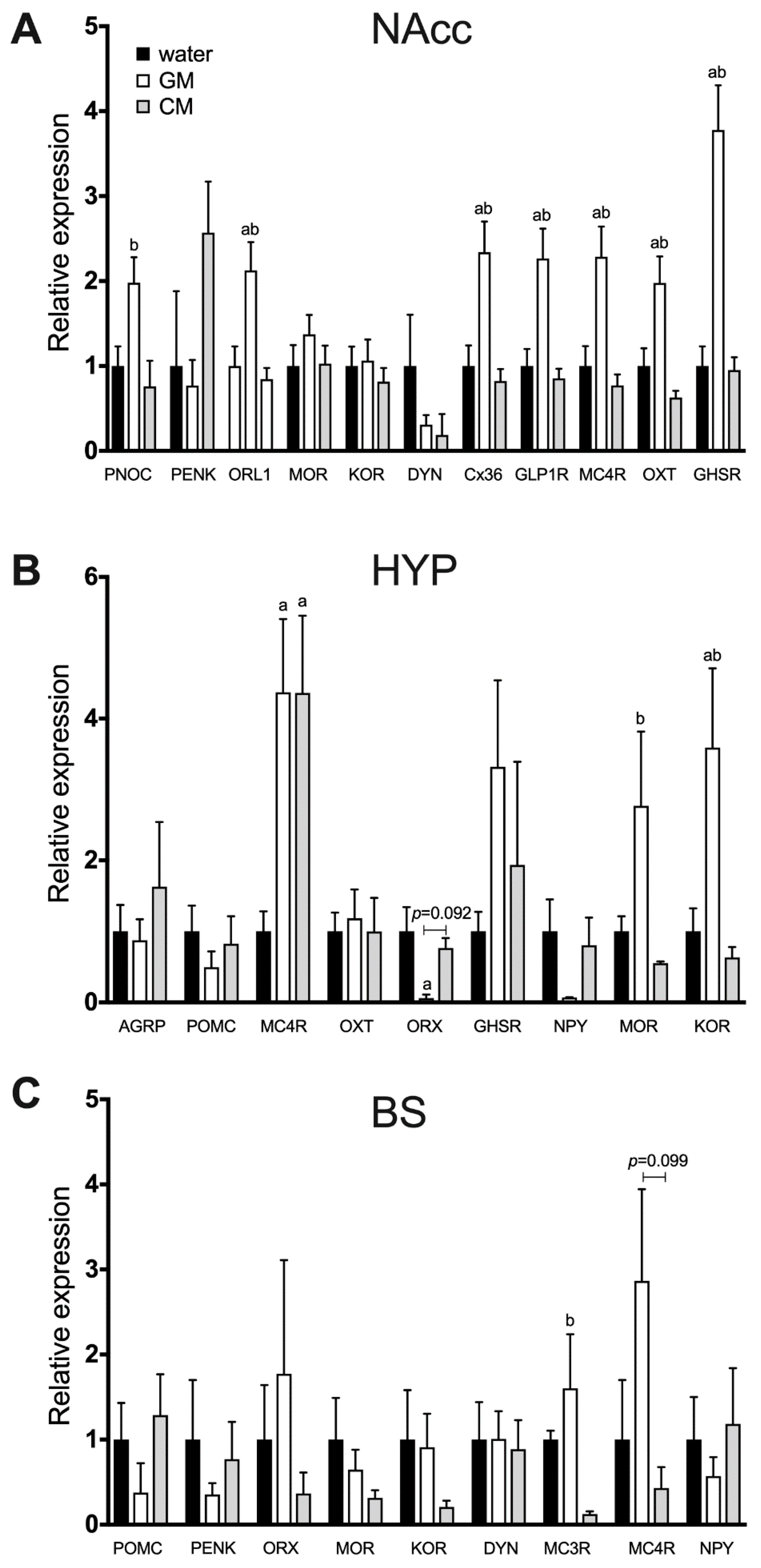
2.11. Effect of 24-h CM vs. GM Consumption on Feeding-Related Gene Expression in the Brain Circuit
2.12. Data Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Drewnowski, A.; Specter, S.E. Poverty and obesity: The role of energy density and energy costs. Am. J. Clin. Nutr. 2004, 79, 6–16. [Google Scholar] [CrossRef] [PubMed]
- Huth, P.J.; Fulgoni, V.L.; Keast, D.R.; Park, K.; Auestad, N. Major food sources of calories, added sugars, and saturated fat and their contribution to essential nutrient intakes in the U.S. diet: Data from the National Health and Nutrition Examination Survey (2003–2006). Nutr. J. 2013, 12, 116. [Google Scholar] [CrossRef] [PubMed]
- Drewnowski, A. The contribution of milk and milk products to micronutrient density and affordability of the U.S. diet. J. Am. Coll. Nutr. 2011, 30 (Suppl. 1), 422S–428S. [Google Scholar] [CrossRef]
- Vissers, P.A.; Streppel, M.T.; Feskens, E.J.; de Groot, L.C. The contribution of dairy products to micronutrient intake in the Netherlands. J. Am. Coll. Nutr. 2011, 30 (Suppl. 1), 415S–421S. [Google Scholar] [CrossRef]
- Feskanich, D.; Willett, W.C.; Colditz, G.A. Calcium, vitamin D, milk consumption, and hip fractures: A prospective study among postmenopausal women. Am. J. Clin. Nutr. 2003, 77, 504–511. [Google Scholar] [CrossRef]
- Chevalley, T.; Bonjour, J.P.; Ferrari, S.; Rizzoli, R. High-protein intake enhances the positive impact of physical activity on BMC in prepubertal boys. J. Bone Miner. Res. 2008, 23, 131–142. [Google Scholar] [CrossRef] [PubMed]
- Haenlein, G.F.W. Goat milk in human nutrition. Small Rumin. Res. 2007, 51, 155–163. [Google Scholar] [CrossRef]
- Bellioni-Businco, B.; Paganelli, R.; Lucenti, P.; Giampietro, P.G.; Perborn, H.; Businco, L. Allergenicity of goat’s milk in children with cow’s milk allergy. J. Allergy Clin. Immunol. 1999, 103, 1191–1194. [Google Scholar] [CrossRef]
- Barrionuevo, M.; Lopez Aliaga, I.; Alferez, M.J.; Mesa, E.; Nestares, T.; Campos, M.S. Beneficial effect of goat milk on bioavailability of copper, zinc and selenium in rats. J. Physiol. Biochem. 2003, 59, 111–118. [Google Scholar] [CrossRef]
- Barrionuevo, M.; Alferez, M.J.; Lopez, A.I.; Sanz, S.M.; Campos, M.S. Beneficial effect of goat milk on nutritive utilization of iron and copper in malabsorption syndrome. J. Dairy Sci. 2002, 85, 657–664. [Google Scholar] [CrossRef]
- Díaz-Castro, J.; López-Frías, M.R.; Campos, M.S.; López-Frías, M.; Alférez, M.J.; Nestares, T.; Ojeda, M.L.; López-Aliaga, I. Severe nutritional iron-deficiency anaemia has a negative effect on some bone turnover biomarkers in rats. Eur. J. Nutr. 2012, 51, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Castro, J.; Lopez-Frias, M.R.; Campos, M.S.; Lopez-Frias, M.; Alférez, M.J.; Nestares, T.; Ortega, E.; Lopez-Aliaga, I. Goat milk during iron repletion improves bone turnover impaired by severe iron deficiency. J. Dairy Sci. 2011, 94, 2752–2761. [Google Scholar] [CrossRef] [PubMed]
- Silanikove, N.; Leitner, G.; Merin, U.; Prosser, C.G. Recent advances in exploiting goat’s milk: Quality, safety and production aspects. Small Rumin. Res. 2010, 89, 110–124. [Google Scholar] [CrossRef]
- Gosnell, B.A.; Levine, A.S. Reward systems and food intake: Role of opioids. Int. J. Obes. 2009, 33 (Suppl. 2), S54–S58. [Google Scholar] [CrossRef] [PubMed]
- Olszewski, P.K.; Alsio, J.; Schioth, H.B.; Levine, A.S. Opioids as facilitators of feeding: Can any food be rewarding? Physiol. Behav. 2011, 104, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Olszewski, P.K.; Cedernaes, J.; Olsson, F.; Levine, A.S.; Schioth, H.B. Analysis of the network of feeding neuroregulators using the Allen Brain Atlas. Neurosci. Biobehav. Rev. 2008, 32, 945–956. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agustí, A.; García-Pardo, M.P.; López-Almela, I.; Campillo, I.; Maes, M.; Romaní-Pérez, M.; Sanz, Y. Interplay Between the Gut-Brain Axis, Obesity and Cognitive Function. Front. Neurosci. 2018, 12, 155. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, G.J. Roles for gut vagal sensory signals in determining energy availability and energy expenditure. Brain Res. 2018, 1693, 151–153. [Google Scholar] [CrossRef]
- De Kloet, A.D.; Herman, J.P. Fat-brain connections: Adipocyte glucocorticoid control of stress and metabolism. Front. Neuroendocrinol. 2018, 48, 50–57. [Google Scholar] [CrossRef]
- Olszewski, P.K.; Shaw, T.J.; Grace, M.K.; Höglund, C.E.; Fredriksson, R.; Schiöth, H.B.; Levine, A.S. Complexity of neural mechanisms underlying overconsumption of sugar in scheduled feeding: Involvement of opioids, orexin, oxytocin and NPY. Peptides 2009, 30, 226–233. [Google Scholar] [CrossRef]
- Zink, A.N.; Perez-Leighton, C.E.; Kotz, C.M. The orexin neuropeptide system: Physical activity and hypothalamic function throughout the aging process. Front. Syst. Neurosci. 2014, 8, 211. [Google Scholar] [CrossRef] [PubMed]
- Gosnell, B.A.; Levine, A.S.; Morley, J.E. The effects of aging on opioid modulation of feeding in rats. Life Sci. 1983, 32, 2793–2799. [Google Scholar] [CrossRef]
- Morley, J.E. Pathophysiology of the anorexia of aging. Curr. Opin. Clin. Nutr. Metab. Care 2013, 16, 27–32. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van de Heijning, B.J.; Kegler, D.; Schipper, L.; Voogd, E.; Oosting, A.; van der Beek, E.M. Acute and Chronic Effects of Dietary Lactose in Adult Rats Are not Explained by Residual Intestinal Lactase Activity. Nutrients 2015, 7, 5542–5555. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McCutcheon, J.E.; Marinelli, M. Age matters. Eur. J. Neurosci. 2009, 29, 997–1014. [Google Scholar] [CrossRef] [PubMed]
- Herisson, F.M.; Brooks, L.L.; Waas, J.R.; Levine, A.S.; Olszewski, P.K. Functional relationship between oxytocin and appetite for carbohydrates versus saccharin. Neuroreport 2014, 25, 909–914. [Google Scholar] [CrossRef]
- Herisson, F.M.; Waas, J.R.; Fredriksson, R.; Schioth, H.B.; Levine, A.S.; Olszewski, P.K. Oxytocin Acting in the Nucleus Accumbens Core Decreases Food Intake. J. Neuroendocrinol. 2016, 28. [Google Scholar] [CrossRef] [PubMed]
- Olszewski, P.K.; Klockars, A.; Olszewska, A.M.; Fredriksson, R.; Schioth, H.B.; Levine, A.S. Molecular, immunohistochemical, and pharmacological evidence of oxytocin’s role as inhibitor of carbohydrate but not fat intake. Endocrinology 2010, 151, 4736–4744. [Google Scholar] [CrossRef] [PubMed]
- Bonacchi, K.B.; Ackroff, K.; Touzani, K.; Bodnar, R.J.; Sclafani, A. Opioid mediation of starch and sugar preference in the rat. Pharmacol. Biochem. Behav. 2010, 96, 507–514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Levine, A.S.; Kotz, C.M.; Gosnell, B.A. Sugars: Hedonic aspects, neuroregulation, and energy balance. Am. J. Clin. Nutr. 2003, 78, 834S–842S. [Google Scholar] [CrossRef]
- Martire, S.I.; Holmes, N.; Westbrook, R.F.; Morris, M.J. Altered feeding patterns in rats exposed to a palatable cafeteria diet: Increased snacking and its implications for development of obesity. PLoS ONE 2013, 8, e60407. [Google Scholar] [CrossRef] [PubMed]
- Martire, S.I.; Maniam, J.; South, T.; Holmes, N.; Westbrook, R.F.; Morris, M.J. Extended exposure to a palatable cafeteria diet alters gene expression in brain regions implicated in reward, and withdrawal from this diet alters gene expression in brain regions associated with stress. Behav. Brain Res. 2014, 265, 132–141. [Google Scholar] [CrossRef]
- Drewnowski, A. Energy density, palatability, and satiety: Implications for weight control. Nutr. Rev. 1998, 56, 347–353. [Google Scholar] [CrossRef] [PubMed]
- Moran, T.H.; Ladenheim, E.E. Physiologic and Neural Controls of Eating. Gastroenterol. Clin. N. Am. 2016, 45, 581–599. [Google Scholar] [CrossRef]
- Landi, F.; Calvani, R.; Tosato, M.; Martone, A.; Ortolani, E.; Savera, G.; Sisto, A.; Marzetti, E. Anorexia of Aging: Risk Factors, Consequences, and Potential Treatments. Nutrients 2016, 8, 69. [Google Scholar] [CrossRef] [PubMed]
- Tenk, J.; Rostás, I.; Füredi, N.; Mikó, A.; Solymár, M.; Soós, S.; Gaszner, B.; Feller, D.; Székely, M.; Pétervári, E.; et al. Age-related changes in central effects of corticotropin-releasing factor (CRF) suggest a role for this mediator in aging anorexia and cachexia. Geroscience 2017, 39, 61–72. [Google Scholar] [CrossRef] [Green Version]
- Tomm, R.J.; Tse, M.T.; Tobiansky, D.J.; Schweitzer, H.R.; Soma, K.K.; Floresco, S.B. Effects of aging on executive functioning and mesocorticolimbic dopamine markers in male Fischer 344 x brown Norway rats. Neurobiol. Aging 2018, 72, 134–146. [Google Scholar] [CrossRef]
- Inui-Yamamoto, C.; Yamamoto, T.; Ueda, K.; Nakatsuka, M.; Kumabe, S.; Inui, T.; Iwai, Y. Taste preference changes throughout different life stages in male rats. PLoS ONE 2017, 12, e0181650. [Google Scholar] [CrossRef]
- Sakai, M.; Kazui, H.; Shigenobu, K.; Komori, K.; Ikeda, M.; Nishikawa, T. Gustatory Dysfunction as an Early Symptom of Semantic Dementia. Dement. Geriatr. Cogn. Dis. Extra 2017, 7, 395–405. [Google Scholar] [CrossRef] [Green Version]
- Shin, Y.K.; Cong, W.N.; Cai, H.; Kim, W.; Maudsley, S.; Egan, J.M.; Martin, B. Age-related changes in mouse taste bud morphology, hormone expression, and taste responsivity. J. Gerontol. A Biol. Sci. Med. Sci. 2012, 67, 336–344. [Google Scholar] [CrossRef]
- Naneix, F.; Darlot, F.; Coutureau, E.; Cador, M. Long-lasting deficits in hedonic and nucleus accumbens reactivity to sweet rewards by sugar overconsumption during adolescence. Eur. J. Neurosci. 2016, 43, 671–680. [Google Scholar] [CrossRef]
- Glass, M.J.; Grace, M.K.; Cleary, J.P.; Billington, C.J.; Levine, A.S. Naloxone’s effect on meal microstructure of sucrose and cornstarch diets. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2001, 281, R1605–R1612. [Google Scholar] [CrossRef] [PubMed]
- Higgs, S.; Cooper, S.J. Evidence for early opioid modulation of licking responses to sucrose and intralipid: A microstructural analysis in the rat. Psychopharmacology 1998, 139, 342–355. [Google Scholar] [CrossRef] [PubMed]
- D’Aquila, P.S. Dopamine on D2-like receptors “reboosts” dopamine D1-like receptor-mediated behavioural activation in rats licking for sucrose. Neuropharmacology 2010, 58, 1085–1096. [Google Scholar] [CrossRef] [PubMed]
- Dwyer, D.M. EPS Prize Lecture. Licking and liking: The assessment of hedonic responses in rodents. Q. J. Exp. Psychol. 2012, 65, 371–394. [Google Scholar] [CrossRef]
- Mendez, I.A.; Ostlund, S.B.; Maidment, N.T.; Murphy, N.P. Involvement of Endogenous Enkephalins and beta-Endorphin in Feeding and Diet-Induced Obesity. Neuropsychopharmacology 2015, 40, 2103–2112. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.D.; Smith, G.P. Analysis of the microstructure of the rhythmic tongue movements of rats ingesting maltose and sucrose solutions. Behav. Neurosci. 1992, 106, 217–228. [Google Scholar] [CrossRef] [PubMed]
- Ostlund, S.B.; Kosheleff, A.; Maidment, N.T.; Murphy, N.P. Decreased consumption of sweet fluids in mu opioid receptor knockout mice: A microstructural analysis of licking behavior. Psychopharmacology 2013, 229, 105–113. [Google Scholar] [CrossRef] [PubMed]
- Mendez, I.A.; Maidment, N.T.; Murphy, N.P. Parsing the hedonic and motivational influences of nociceptin on feeding using licking microstructure analysis in mice. Behav. Pharmacol. 2016, 27, 516–527. [Google Scholar] [CrossRef] [Green Version]
- Rubio-Martín, E.; García-Escobar, E.; Ruiz de Adana, M.S.; Lima-Rubio, F.; Peláez, L.; Caracuel, A.M.; Bermúdez-Silva, F.J.; Soriguer, F.; Rojo-Martínez, G.; Olveira, G. Comparison of the Effects of Goat Dairy and Cow Dairy Based Breakfasts on Satiety, Appetite Hormones, and Metabolic Profile. Nutrients 2017, 9, 877. [Google Scholar] [CrossRef]
- Park, Y.W.; Juarez, M.; Ramos, M.; Haenlein, G.F.W. Physico-chemical characteristics of goat and sheep milk. Small Rumin. Res. 2007, 68, 88–113. [Google Scholar] [CrossRef] [Green Version]
- Klockars, A.; Levine, A.S.; Olszewski, P.K. Hypothalamic Integration of the Endocrine Signaling Related to Food Intake. Curr. Top. Behav. Neurosci. 2018. [Google Scholar] [CrossRef]
- Levine, A.S.; Jewett, D.C.; Cleary, J.P.; Kotz, C.M.; Billington, C.J. Our journey with neuropeptide Y: Effects on ingestive behaviors and energy expenditure. Peptides 2004, 25, 505–510. [Google Scholar] [CrossRef]
- Nixon, J.P.; Kotz, C.M.; Novak, C.M.; Billington, C.J.; Teske, J.A. Neuropeptides controlling energy balance: Orexins and neuromedins. Handb. Exp. Pharmacol. 2012, 209, 77–109. [Google Scholar]
- Wirth, M.M.; Olszewski, P.K.; Yu, C.; Levine, A.S.; Giraudo, S.Q. Paraventricular hypothalamic alpha-melanocyte-stimulating hormone and MTII reduce feeding without causing aversive effects. Peptides 2001, 22, 129–134. [Google Scholar] [CrossRef]
- Girardet, C.; Butler, A.A. Neural melanocortin receptors in obesity and related metabolic disorders. Biochim. Biophys. Acta 2014, 1842, 482–494. [Google Scholar] [CrossRef] [PubMed]
- Olszewski, P.K.; Klockars, A.; Levine, A.S. Oxytocin: A Conditional Anorexigen whose Effects on Appetite Depend on the Physiological, Behavioural and Social Contexts. J. Neuroendocrinol. 2016, 28. [Google Scholar] [CrossRef] [PubMed]
- Gosnell, B.A.; Levine, A.S.; Morley, J.E. The stimulation of food intake by selective agonists of mu, kappa and delta opioid receptors. Life Sci. 1986, 38, 1081–1088. [Google Scholar] [CrossRef]
- Giraudo, S.Q.; Grace, M.K.; Welch, C.C.; Billington, C.J.; Levine, A.S. Naloxone’s anorectic effect is dependent upon the relative palatability of food. Pharmacol. Biochem. Behav. 1993, 46, 917–921. [Google Scholar] [CrossRef]
- Steffensen, S.C.; Bradley, K.D.; Hansen, D.M.; Wilcox, J.D.; Wilcox, R.S.; Allison, D.W.; Merrill, C.B.; Edwards, J.G. The role of connexin-36 gap junctions in alcohol intoxication and consumption. Synapse 2011, 65, 695–707. [Google Scholar] [CrossRef] [PubMed]
- Skibicka, K.P.; Shirazi, R.H.; Rabasa-Papio, C.; Alvarez-Crespo, M.; Neuber, C.; Vogel, H.; Dickson, S.L. Divergent circuitry underlying food reward and intake effects of ghrelin: Dopaminergic VTA-accumbens projection mediates ghrelin’s effect on food reward but not food intake. Neuropharmacology 2013, 73, 274–283. [Google Scholar] [CrossRef] [PubMed]
- Kanoski, S.E.; Hayes, M.R.; Skibicka, K.P. GLP-1 and weight loss: Unraveling the diverse neural circuitry. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2016, 310, R885–R895. [Google Scholar] [CrossRef] [PubMed]
| Sample | Protein | Fat | Carbohydrate (Lactose) | Ash | Moisture |
|---|---|---|---|---|---|
| CM | 37.1 | 1.1 | 51 | 6.5 | 4.3 |
| GM | 36.1 | 0.9 | 49.9 | 9.5 | 3.6 |
| Base Skim Milk Powder Composition (1% Milk Fat) | Goat Skim Milk Powder (SMP) | Cow SMP | ||
|---|---|---|---|---|
| % | g/kg | % | g/kg | |
| Protein | 37.8 | 120 | 37.1 | 120 |
| Fat | 1.7 | 5.40 | 0.86 | 2.78 |
| Ash | 9.4 | 29.84 | 7.3 | 23.61 |
| Moisture | 4.1 | 13.02 | 4.2 | 13.58 |
| Lactose | 47 | 149.21 | 50.54 | 163.47 |
| Total | 100 | 317.46 | 100 | 323.45 |
| Goat SMP | Cow SMP | |||
| Skim Milk Chow—Ingredient | g/kg | g/kg | ||
| Goat SMP—to supply 12% protein | 317.46 | |||
| Cow SMP—to supply 12% protein | 323.45 | |||
| Vitamin mix | 50.00 | 50.00 | ||
| Salt mix | 50.00 | 50.00 | ||
| Corn oil (added to make 5% fat) | 44.60 | 47.22 | ||
| Starch | 447.15 | 452.80 | ||
| Lactose (added to make 16.5%) | 15.79 | 1.53 | ||
| Cellulose | 75.00 | 75.00 | ||
| Moisture | ||||
| 1000.00 | 1000.00 | |||
| Target Dietary Content | ||||
| Protein | 12% | 12% | ||
| Fat | 5% | 5% | ||
| Lactose | 16.50% | 16.50% | ||
| Fiber | 7.50% | 7.50% | ||
| Starch | 44.7% | 45.3% | ||
| Calories | ||||
| Protein (4 cal/g) | 48 | 48 | ||
| Fat (9 cal/g) | 45 | 45 | ||
| Lactose (4 cal/g) | 66 | 66 | ||
| Fiber (2 cal/g) | 15 | 15 | ||
| Starch (4 cal/g) | 179 | 181 | ||
| Calories/100 g | 353 | 355 | ||
| % calories | ||||
| Protein | 14% | 14% | ||
| Fat | 13% | 13% | ||
| Lactose | 19% | 19% | ||
| Fiber | 4% | 4% | ||
| Starch | 51% | 51% | ||
| Skim Milk Powder (1% Milk Fat) | Goat SMP | Cow SMP | ||
| Ingredients | g/kg | g/kg | ||
| Goat SMP | 317.46 | |||
| Cow SMP | 323.45 | |||
| Vitamin mix | 50.00 | 50.00 | ||
| Salt mix | 50.00 | 50.00 | ||
| Corn oil | 44.60 | 47.22 | ||
| Starch | 447.15 | 452.80 | ||
| Lactose | 15.79 | 1.53 | ||
| Cellulose | 75.00 | 75.00 | ||
| Moisture | ||||
| 1000.00 | 1000.00 | |||
| Target Dietary Content | ||||
| Protein | 12% | 12% | ||
| Fat | 5% | 5% | ||
| Lactose | 16.50% | 16.50% | ||
| Fiber | 7.50% | 7.50% | ||
| Starch | 44.7% | 45.3% | ||
| Calories | ||||
| Protein (4 cal/g) | 48 | 48 | ||
| Fat (9 cal/g) | 45 | 45 | ||
| Lactose (4 cal/g) | 66 | 66 | ||
| Fiber (2 cal/g) | 15 | 15 | ||
| Starch (4 cal/g) | 179 | 181 | ||
| Calories/100 g | 353 | 355 | ||
| % calories | ||||
| Protein | 14% | 14% | ||
| Fat | 13% | 13% | ||
| Lactose | 19% | 19% | ||
| Fiber | 4% | 4% | ||
| Starch | 51% | 51% | ||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Klockars, A.; Wood, E.L.; Gartner, S.N.; McColl, L.K.; Levine, A.S.; Carpenter, E.A.; Prosser, C.G.; Olszewski, P.K. Palatability of Goat’s versus Cow’s Milk: Insights from the Analysis of Eating Behavior and Gene Expression in the Appetite-Relevant Brain Circuit in Laboratory Animal Models. Nutrients 2019, 11, 720. https://doi.org/10.3390/nu11040720
Klockars A, Wood EL, Gartner SN, McColl LK, Levine AS, Carpenter EA, Prosser CG, Olszewski PK. Palatability of Goat’s versus Cow’s Milk: Insights from the Analysis of Eating Behavior and Gene Expression in the Appetite-Relevant Brain Circuit in Laboratory Animal Models. Nutrients. 2019; 11(4):720. https://doi.org/10.3390/nu11040720
Chicago/Turabian StyleKlockars, Anica, Erin L. Wood, Sarah N. Gartner, Laura K. McColl, Allen S. Levine, Elizabeth A. Carpenter, Colin G. Prosser, and Pawel K. Olszewski. 2019. "Palatability of Goat’s versus Cow’s Milk: Insights from the Analysis of Eating Behavior and Gene Expression in the Appetite-Relevant Brain Circuit in Laboratory Animal Models" Nutrients 11, no. 4: 720. https://doi.org/10.3390/nu11040720
APA StyleKlockars, A., Wood, E. L., Gartner, S. N., McColl, L. K., Levine, A. S., Carpenter, E. A., Prosser, C. G., & Olszewski, P. K. (2019). Palatability of Goat’s versus Cow’s Milk: Insights from the Analysis of Eating Behavior and Gene Expression in the Appetite-Relevant Brain Circuit in Laboratory Animal Models. Nutrients, 11(4), 720. https://doi.org/10.3390/nu11040720






