Vitamin D Status, Calcium Intake and Risk of Developing Type 2 Diabetes: An Unresolved Issue
Abstract
1. Introduction
2. Methods
3. Vitamin D and Type 2 Diabetes (T2D)
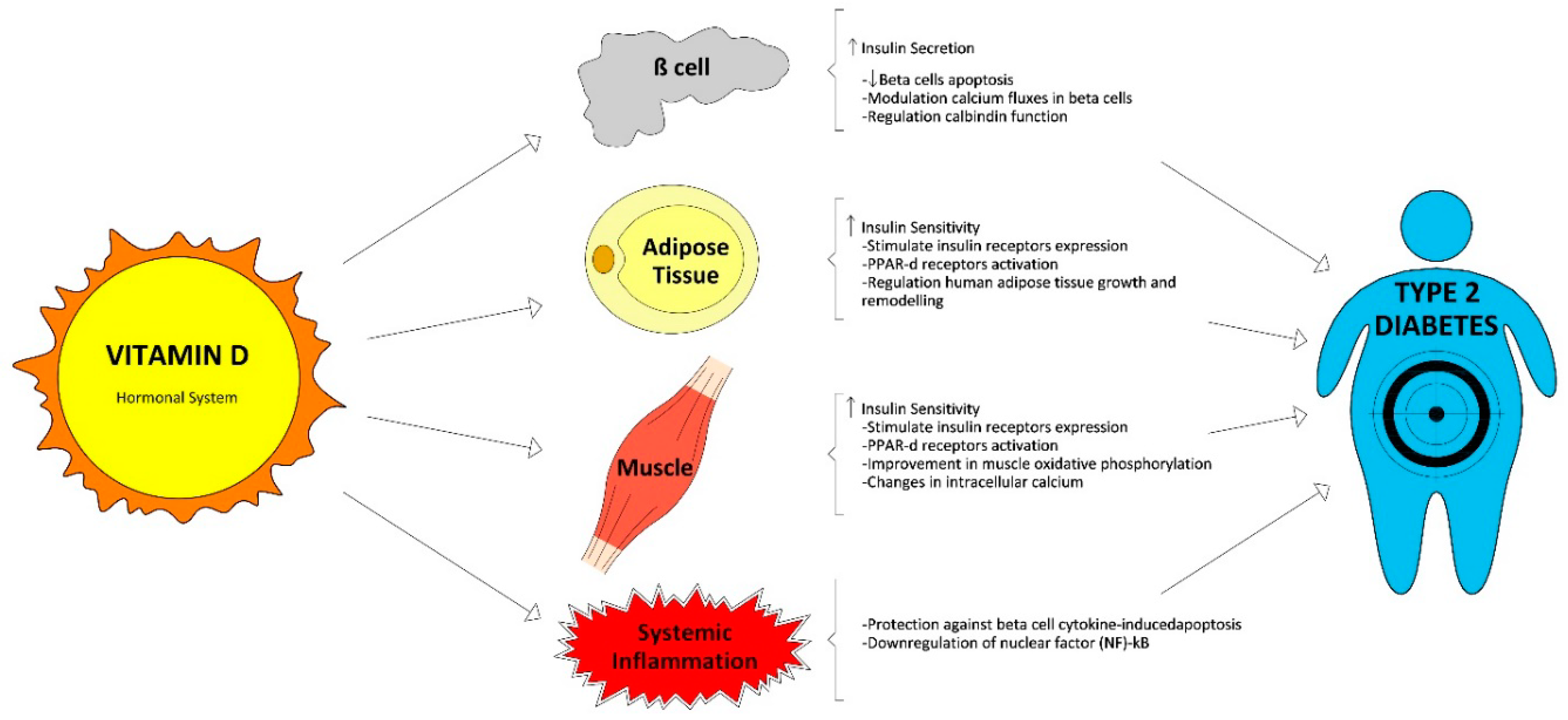
3.1. Vitamin D Physiology and Glucose Homeostasis
3.2. Vitamin D Status and Its Relationship with T2D in Cross-Sectional and Longitudinal Studies
3.3. Vitamin D Supplementation and Risk of T2D: Randomized Trials and Meta-Analysis
3.4. New Thresholds for the Relationship between Vitamin D and T2D
4. Calcium Intake and T2D
4.1. Mechanistic Studies
4.2. Dairy Intake and T2D Risk
4.3. Observational Studies
4.4. Systematic Reviews and Meta-Analyses
5. Nutritional Recommendations for T2D Prevention
6. Unsolved Questions
7. Conclusions and Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- International Diabetes Federation. IDF Diabetes Atlas, 8th ed.; International Diabetes Federation: Brussels, Belgium, 2017; ISBN 978-2-930229-81-2. [Google Scholar]
- Pittas, A.G.; Nelson, J.; Mitri, J.; Hillmann, W.; Garganta, C.; Nathan, D.M.; Hu, F.B.; Dawson-Hughes, B.; Diabetes Prevention Program Research Group. Plasma 25-hydroxyvitamin D and progression to diabetes in patients at risk for diabetes: An ancillary analysis in the Diabetes Prevention Program. Diabetes Care 2012, 35, 565–573. [Google Scholar] [CrossRef] [PubMed]
- Maddaloni, E.; Cavallari, I.; Napoli, N.; Conte, C. Vitamin D and Diabetes Mellitus. Front. Horm. Res. 2018, 50, 161–176. [Google Scholar] [PubMed]
- Ford, E.S.; Ajani, U.A.; McGuire, L.C.; Liu, S. Concentrations of serum vitamin D and the metabolic syndrome among U.S. adults. Diabetes Care 2005, 28, 1228–1230. [Google Scholar] [CrossRef]
- Chiu, K.C.; Chu, A.; Go, V.L.W.; Saad, M.F. Hypovitaminosis D is associated with insulin resistance and β cell dysfunction. Am. J. Clin. Nutr. 2004, 79, 820–825. [Google Scholar] [CrossRef] [PubMed]
- Lips, P.; Eekhoff, M.; van Schoor, N.; Oosterwerff, M.; de Jongh, R.; Krul-Poel, Y.; Simsek, S. Vitamin D and type 2 diabetes. J. Steroid Biochem. Mol. Biol. 2017, 173, 280–285. [Google Scholar] [CrossRef]
- Kalergis, M.; Leung Yinko, S.S.L.; Nedelcu, R. Dairy products and prevention of type 2 diabetes: Implications for research and practice. Front. Endocrinol. 2013, 4, 90. [Google Scholar] [CrossRef] [PubMed]
- Talaei, M.; Pan, A.; Yuan, J.M.; Koh, W.P. Dairy intake and risk of type 2 diabetes. Clin. Nutr. 2018, 37, 712–718. [Google Scholar] [CrossRef]
- Kim, K.N.; Oh, S.Y.; Hong, Y.C. Associations of serum calcium levels and dietary calcium intake with incident type 2 diabetes over 10 years: The Korean Genome and Epidemiology Study (KoGES). Diabetol. Metab. Syndr. 2018, 10, 50. [Google Scholar] [CrossRef]
- Cade, C.; Norman, A.W. Rapid normalization/stimulation by 1,25- dihydroxyvitamin d3 of insulin secretion and glucose tolerance in the vitamin d-deficient rat. Endocrinology 1987, 120, 1490–1497. [Google Scholar] [CrossRef]
- Zeitz, U.; Weber, K.; Soegiarto, D.W.; Wolf, E.; Balling, R.; Erben, R.G. Impaired insulin secretory capacity in mice lacking a functional vitamin D receptor. FASEB J. 2003, 17, 509–511. [Google Scholar] [CrossRef]
- Bouillon, R.; Carmeliet, G.; Verlinden, L.; Van Etten, E.; Verstuyf, A.; Luderer, H.F.; Lieben, L.; Mathieu, C.; Demay, M. Vitamin D and human health: Lessons from vitamin D receptor null mice. Endocr. Rev. 2008, 29, 726–776. [Google Scholar] [CrossRef] [PubMed]
- Bland, R.; Markovic, D.; Hills, C.E.; Hughes, S.V.; Chan, S.L.F.; Squires, P.E.; Hewison, M. Expression of 25-hydroxyvitamin D3-1alpha-hydroxylase in pancreatic islets. J. Steroid Biochem. Mol. Biol. 2004, 89–90, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Reusch, J.E.; Begum, N.; Sussman, K.E.; Draznin, B. Regulation of GLUT-4 phosphorylation by intracellular calcium in adipocytes. Endocrinology 1991, 129, 3269–3273. [Google Scholar] [CrossRef] [PubMed]
- Leung, P.S. The Potential Protective Action of Vitamin D in Hepatic Insulin Resistance and Pancreatic Islet Dysfunction in Type 2 Diabetes Mellitus. Nutrients 2016, 8, 147. [Google Scholar] [CrossRef] [PubMed]
- Wright, D.C.; Hucker, K.A.; Holloszy, J.O.; Han, D.H. Ca2+ and AMPK both mediate stimulation of glucose transport by muscle contractions. Diabetes 2004, 53, 330–335. [Google Scholar] [CrossRef]
- Draznin, B. Cytosolic calcium and insulin resistance. Am. J. Kidney Dis. 1993, 21, 32–38. [Google Scholar] [CrossRef]
- Nimitphong, H.; Holick, M.F.; Fried, S.K.; Lee, M.-J. 25-hydroxyvitamin D(3) and 1,25-dihydroxyvitamin D(3) promote the differentiation of human subcutaneous preadipocytes. PLoS ONE 2012, 7, e52171. [Google Scholar] [CrossRef]
- Blum, M.; Dolnikowski, G.; Seyoum, E.; Harris, S.S.; Booth, S.L.; Peterson, J.; Saltzman, E.; Dawson-Hughes, B. Vitamin D(3) in fat tissue. Endocrine 2008, 33, 90–94. [Google Scholar] [CrossRef]
- Wamberg, L.; Christiansen, T.; Paulsen, S.K.; Fisker, S.; Rask, P.; Rejnmark, L.; Richelsen, B.; Pedersen, S.B. Expression of vitamin D-metabolizing enzymes in human adipose tissue—The effect of obesity and diet-induced weight loss. Int. J. Obes. 2013, 37, 651–657. [Google Scholar] [CrossRef]
- Hyppönen, E.; Boucher, B.J. Adiposity, vitamin D requirements, and clinical implications for obesity-related metabolic abnormalities. Nutr. Rev. 2018, 76, 678–692. [Google Scholar] [CrossRef]
- Chun, R.F.; Liu, P.T.; Modlin, R.L.; Adams, J.S.; Hewison, M. Impact of vitamin D on immune function: lessons learned from genome-wide analysis. Front. Physiol. 2014, 5, 151. [Google Scholar] [CrossRef] [PubMed]
- Christakos, S.; Liu, Y. Biological actions and mechanism of action of calbindin in the process of apoptosis. J. Steroid Biochem. Mol. Biol. 2004, 89–90, 401–404. [Google Scholar] [CrossRef] [PubMed]
- Salum, E.; Kals, J.; Kampus, P.; Salum, T.; Zilmer, K.; Aunapuu, M.; Arend, A.; Eha, J.; Zilmer, M. Vitamin D reduces deposition of advanced glycation end-products in the aortic wall and systemic oxidative stress in diabetic rats. Diabetes Res. Clin. Pract. 2013, 100, 243–249. [Google Scholar] [CrossRef] [PubMed]
- Forouhi, N.G.; Luan, J.; Cooper, A.; Boucher, B.J.; Wareham, N.J. Baseline serum 25-hydroxy vitamin d is predictive of future glycemic status and insulin resistance: The Medical Research Council Ely Prospective Study 1990-2000. Diabetes 2008, 57, 2619–2625. [Google Scholar] [CrossRef] [PubMed]
- Kayaniyil, S.; Vieth, R.; Retnakaran, R.; Knight, J.A.; Qi, Y.; Gerstein, H.C.; Perkins, B.A.; Harris, S.B.; Zinman, B.; Hanley, A.J. Association of vitamin D with insulin resistance and beta-cell dysfunction in subjects at risk for type 2 diabetes. Diabetes Care 2010, 33, 1379–1381. [Google Scholar] [CrossRef] [PubMed]
- Maki, K.C.; Fulgoni, V.L., 3rd; Keast, D.R.; Rains, T.M.; Park, K.M.; Rubin, M.R. Vitamin D intake and status are associated with lower prevalence of metabolic syndrome in U.S. adults: National Health and Nutrition Examination Surveys 2003–2006. Metab. Syndr. Relat. Disord. 2012, 10, 363–372. [Google Scholar] [CrossRef]
- Gannage-Yared, M.-H.; Chedid, R.; Khalife, S.; Azzi, E.; Zoghbi, F.; Halaby, G. Vitamin D in relation to metabolic risk factors, insulin sensitivity and adiponectin in a young Middle-Eastern population. Eur. J. Endocrinol. 2009, 160, 965–971. [Google Scholar] [CrossRef]
- Scragg, R.; Holdaway, I.; Singh, V.; Metcalf, P.; Baker, J.; Dryson, E. Serum 25-hydroxyvitamin D3 levels decreased in impaired glucose tolerance and diabetes mellitus. Diabetes Res. Clin. Pract. 1995, 27, 181–188. [Google Scholar] [CrossRef]
- Olson, M.L.; Maalouf, N.M.; Oden, J.D.; White, P.C.; Hutchison, M.R. Vitamin D deficiency in obese children and its relationship to glucose homeostasis. J. Clin. Endocrinol. Metab. 2012, 97, 279–285. [Google Scholar] [CrossRef]
- Parikh, S.; Guo, D.-H.; Pollock, N.K.; Petty, K.; Bhagatwala, J.; Gutin, B.; Houk, C.; Zhu, H.; Dong, Y. Circulating 25-hydroxyvitamin D concentrations are correlated with cardiometabolic risk among American black and white adolescents living in a year-round sunny climate. Diabetes Care 2012, 35, 1133–1138. [Google Scholar] [CrossRef]
- Kayaniyil, S.; Retnakaran, R.; Harris, S.B.; Vieth, R.; Knight, J.A.; Gerstein, H.C.; Perkins, B.A.; Zinman, B.; Hanley, A.J. Prospective associations of vitamin D with beta-cell function and glycemia: The PROspective Metabolism and ISlet cell Evaluation (PROMISE) cohort study. Diabetes 2011, 60, 2947–2953. [Google Scholar] [CrossRef] [PubMed]
- Forouhi, N.G.; Ye, Z.; Rickard, A.P.; Khaw, K.T.; Luben, R.; Langenberg, C.; Wareham, N.J. Circulating 25-hydroxyvitamin D concentration and the risk of type 2 diabetes: Results from the European Prospective Investigation into Cancer (EPIC)-Norfolk cohort and updated meta-analysis of prospective studies. Diabetologia 2012, 55, 2173–2182. [Google Scholar] [CrossRef] [PubMed]
- Afzal, S.; Bojesen, S.E.; Nordestgaard, B.G. Low 25-hydroxyvitamin D and risk of type 2 diabetes: A prospective cohort study and metaanalysis. Clin. Chem. 2013, 59, 381–391. [Google Scholar] [CrossRef]
- Song, Y.; Wang, L.; Pittas, A.G.; Del Gobbo, L.C.; Zhang, C.; Manson, J.E.; Hu, F.B. Blood 25-hydroxy vitamin D levels and incident type 2 diabetes: A meta-analysis of prospective studies. Diabetes Care 2013, 36, 1422–1428. [Google Scholar] [CrossRef]
- Park, S.K.; Garland, C.F.; Gorham, E.D.; BuDoff, L.; Barrett-Connor, E. Plasma 25-hydroxyvitamin D concentration and risk of type 2 diabetes and pre-diabetes: 12-year cohort study. PLoS ONE 2018, 13, e0193070. [Google Scholar] [CrossRef]
- Avila-Rubio, V.; Garcia-Fontana, B.; Novo-Rodriguez, C.; Cantero-Hinojosa, J.; Reyes-Garcia, R.; Munoz-Torres, M. Higher Levels of Serum 25-Hydroxyvitamin D Are Related to Improved Glucose Homeostasis in Women with Postmenopausal Osteoporosis. J. Women’s Health 2018, 27, 1007–1015. [Google Scholar] [CrossRef] [PubMed]
- Zheng, J.-S.; Imamura, F.; Sharp, S.J.; van der Schouw, Y.T.; Sluijs, I.; Gundersen, T.E.; Ardanaz, E.; Boeing, H.; Bonet, C.; Gómez, J.H.; et al. Association of plasma vitamin D metabolites with incident type 2 diabetes: EPIC-InterAct case-cohort study. J. Clin. Endocrinol. Metab. 2019, 104, 1293–1303. [Google Scholar] [CrossRef]
- Heath, A.K.; Williamson, E.J.; Hodge, A.M.; Ebeling, P.R.; Eyles, D.W.; Kvaskoff, D.; O’Dea, K.; Giles, G.G.; English, D.R. Vitamin D status and the risk of type 2 diabetes: The Melbourne Collaborative Cohort Study. Diabetes Res. Clin. Pract. 2018, 149, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Sollid, S.T.; Hutchinson, M.Y.S.; Fuskevag, O.M.; Figenschau, Y.; Joakimsen, R.M.; Schirmer, H.; Njolstad, I.; Svartberg, J.; Kamycheva, E.; Jorde, R. No effect of high-dose vitamin D supplementation on glycemic status or cardiovascular risk factors in subjects with prediabetes. Diabetes Care 2014, 37, 2123–2131. [Google Scholar] [CrossRef]
- Forouhi, N.G.; Menon, R.K.; Sharp, S.J.; Mannan, N.; Timms, P.M.; Martineau, A.R.; Rickard, A.P.; Boucher, B.J.; Chowdhury, T.A.; Griffiths, C.J.; et al. Effects of vitamin D2 or D3 supplementation on glycaemic control and cardiometabolic risk among people at risk of type 2 diabetes: Results of a randomized double-blind placebo-controlled trial. Diabetes Obes. Metab. 2016, 18, 392–400. [Google Scholar] [CrossRef]
- Griffin, S.J.; Little, P.S.; Hales, C.N.; Kinmonth, A.L.; Wareham, N.J. Diabetes risk score: Towards earlier detection of type 2 diabetes in general practice. Diabetes. Metab. Res. Rev. 2000, 16, 164–171. [Google Scholar] [CrossRef]
- Davidson, M.B.; Duran, P.; Lee, M.L.; Friedman, T.C. High-dose vitamin D supplementation in people with prediabetes and hypovitaminosis D. Diabetes Care 2013, 36, 260–266. [Google Scholar] [CrossRef] [PubMed]
- Gagnon, C.; Daly, R.M.; Carpentier, A.; Lu, Z.X.; Shore-Lorenti, C.; Sikaris, K.; Jean, S.; Ebeling, P.R. Effects of combined calcium and vitamin D supplementation on insulin secretion, insulin sensitivity and beta-cell function in multi-ethnic vitamin D-deficient adults at risk for type 2 diabetes: A pilot randomized, placebo-controlled trial. PLoS ONE 2014, 9, e109607. [Google Scholar] [CrossRef] [PubMed]
- Oosterwerff, M.M.; Eekhoff, E.M.; Van Schoor, N.M.; Boeke, A.J.P.; Nanayakkara, P.; Meijnen, R.; Knol, D.L.; Kramer, M.H.; Lips, P. Effect of moderate-dose vitamin D supplementation on insulin sensitivity in vitamin D-deficient non-Western immigrants in the Netherlands: A randomized placebo-controlled trial. Am. J. Clin. Nutr. 2014, 100, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Salehpour, A.; Shidfar, F.; Hosseinpanah, F.; Vafa, M.; Razaghi, M.; Amiri, F. Does vitamin D3 supplementation improve glucose homeostasis in overweight or obese women? A double-blind, randomized, placebo-controlled clinical trial. Diabet. Med. 2013, 30, 1477–1481. [Google Scholar] [CrossRef] [PubMed]
- Tuomainen, T.-P.; Virtanen, J.K.; Voutilainen, S.; Nurmi, T.; Mursu, J.; de Mello, V.D.F.; Schwab, U.; Hakumaki, M.; Pulkki, K.; Uusitupa, M. Glucose Metabolism Effects of Vitamin D in Prediabetes: The VitDmet Randomized Placebo-Controlled Supplementation Study. J. Diabetes Res. 2015, 2015, 672653. [Google Scholar] [CrossRef] [PubMed]
- Wagner, H.; Alvarsson, M.; Mannheimer, B.; Degerblad, M.; Ostenson, C.-G. No Effect of High-Dose Vitamin D Treatment on beta-Cell Function, Insulin Sensitivity, or Glucose Homeostasis in Subjects With Abnormal Glucose Tolerance: A Randomized Clinical Trial. Diabetes Care 2016, 39, 345–352. [Google Scholar] [CrossRef]
- Von Hurst, P.R.; Stonehouse, W.; Coad, J. Vitamin D supplementation reduces insulin resistance in South Asian women living in New Zealand who are insulin resistant and vitamin D deficient—A randomised, placebo-controlled trial. Br. J. Nutr. 2010, 103, 549. [Google Scholar] [CrossRef]
- Mitri, J.; Dawson-Hughes, B.; Hu, F.B.; Pittas, A.G. Effects of vitamin D and calcium supplementation on pancreatic beta cell function, insulin sensitivity, and glycemia in adults at high risk of diabetes: The Calcium and Vitamin D for Diabetes Mellitus (CaDDM) randomized controlled trial. Am. J. Clin. Nutr. 2011, 94, 486–494. [Google Scholar] [CrossRef]
- Manson, J.E.; Cook, N.R.; Lee, I.-M.; Christen, W.; Bassuk, S.S.; Mora, S.; Gibson, H.; Gordon, D.; Copeland, T.; D’Agostino, D.; et al. Vitamin D Supplements and Prevention of Cancer and Cardiovascular Disease. N. Engl. J. Med. 2018, 380, 33–44. [Google Scholar] [CrossRef]
- Rafiq, S.; Jeppesen, P.B. Body mass index, vitamin d, and type 2 diabetes: A systematic review and meta-analysis. Nutrients 2018, 10, 1182. [Google Scholar] [CrossRef]
- Tang, H.; Li, D.; Li, Y.; Zhang, X.; Song, Y.; Li, X. Effects of Vitamin D Supplementation on Glucose and Insulin Homeostasis and Incident Diabetes among Nondiabetic Adults: A Meta-Analysis of Randomized Controlled Trials. Int. J. Endocrinol. 2018, 2018, 7908764. [Google Scholar] [CrossRef] [PubMed]
- LeBlanc, E.S.; Pratley, R.E.; Dawson-Hughes, B.; Staten, M.A.; Sheehan, P.R.; Lewis, M.R.; Peters, A.; Kim, S.H.; Chatterjee, R.; Aroda, V.R.; et al. Baseline Characteristics of the Vitamin D and Type 2 Diabetes (D2d) Study: A Contemporary Prediabetes Cohort That Will Inform Diabetes Prevention Efforts. Diabetes Care 2018, 41, 1590–1599. [Google Scholar] [CrossRef] [PubMed]
- Belenchia, A.M.; Tosh, A.K.; Hillman, L.S.; Peterson, C.A. Correcting vitamin D insufficiency improves insulin sensitivity in obese adolescents: A randomized controlled trial. Am. J. Clin. Nutr. 2013, 97, 774–781. [Google Scholar] [CrossRef] [PubMed]
- Jorde, R.; Sneve, M.; Torjesen, P.; Figenschau, Y. No improvement in cardiovascular risk factors in overweight and obese subjects after supplementation with vitamin D3 for 1 year. J. Intern. Med. 2010, 267, 462–472. [Google Scholar] [CrossRef] [PubMed]
- Pittas, A.G.; Lau, J.; Hu, F.B.; Dawson-Hughes, B. The Role of Vitamin D and Calcium in Type 2 Diabetes. A Systematic Review and Meta-Analysis. J. Clin. Endocrinol. Metab. 2007, 92, 2017–2029. [Google Scholar] [CrossRef]
- Ojuka, E.O. Role of calcium and AMP kinase in the regulation of mitochondrial biogenesis and GLUT4 levels in muscle. Proc. Nutr. Soc. 2004, 63, 275–278. [Google Scholar] [CrossRef] [PubMed]
- Milner, R.D.G.; Hales, C.N. The role of calcium and magnesium in insulin secretion from rabbit pancreas studied in vitro. Diabetologia 1967, 3, 47–49. [Google Scholar] [CrossRef]
- Zemel, M.B. Nutritional and endocrine modulation of intracellular calcium: Implications in obesity, insulin resistance and hypertension. Mol. Cell. Biochem. 1998, 188, 129–136. [Google Scholar] [CrossRef]
- Dunlop, T.W.; Väisänen, S.; Frank, C.; Molnár, F.; Sinkkonen, L.; Carlberg, C. The Human Peroxisome Proliferator-activated Receptor δ Gene is a Primary Target of 1α,25-Dihydroxyvitamin D3 and its Nuclear Receptor. J. Mol. Biol. 2005, 349, 248–260. [Google Scholar] [CrossRef]
- Mozaffarian, D.; Cao, H.; King, I.B.; Lemaitre, R.N.; Song, X.; Siscovick, D.S.; Hotamisligil, G.S. Trans-Palmitoleic Acid, Metabolic Risk Factors, and New-Onset Diabetes in U.S. Adults. Ann. Intern. Med. 2010, 153, 790. [Google Scholar] [CrossRef] [PubMed]
- Mozaffarian, D.; de Oliveira Otto, M.C.; Lemaitre, R.N.; Fretts, A.M.; Hotamisligil, G.; Tsai, M.Y.; Siscovick, D.S.; Nettleton, J.A. trans-Palmitoleic acid, other dairy fat biomarkers, and incident diabetes: The Multi-Ethnic Study of Atherosclerosis (MESA). Am. J. Clin. Nutr. 2013, 97, 854–861. [Google Scholar] [CrossRef] [PubMed]
- Kratz, M.; Baars, T.; Guyenet, S. The relationship between high-fat dairy consumption and obesity, cardiovascular, and metabolic disease. Eur. J. Nutr. 2013, 52, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Alhazmi, A.; Stojanovski, E.; McEvoy, M.; Garg, M.L. Macronutrient Intakes and Development of Type 2 Diabetes: A Systematic Review and Meta-Analysis of Cohort Studies. J. Am. Coll. Nutr. 2012, 31, 243–258. [Google Scholar] [CrossRef] [PubMed]
- Givens, D.I.; Beswick, A.D.; Fehily, A.M.; Pickering, J.E.; Gallacher, J. The Survival Advantage of Milk and Dairy Consumption: An Overview of Evidence from Cohort Studies of Vascular Diseases, Diabetes and Cancer AU—Elwood, Peter C. J. Am. Coll. Nutr. 2008, 27, 723S–734S. [Google Scholar]
- Tong, X.; Dong, J.-Y.; Wu, Z.-W.; Li, W.; Qin, L.-Q. Dairy consumption and risk of type 2 diabetes mellitus: A meta-analysis of cohort studies. Eur. J. Clin. Nutr. 2011, 65, 1027. [Google Scholar] [CrossRef]
- Soedamah-Muthu, S.S.; Masset, G.; Verberne, L.; Geleijnse, J.M.; Brunner, E.J. Consumption of dairy products and associations with incident diabetes, CHD and mortality in the Whitehall II study. Br. J. Nutr. 2013, 109, 718–726. [Google Scholar] [CrossRef] [PubMed]
- Struijk, E.A.; Heraclides, A.; Witte, D.R.; Soedamah-Muthu, S.S.; Geleijnse, J.M.; Toft, U.; Lau, C.J. Dairy product intake in relation to glucose regulation indices and risk of type 2 diabetes. Nutr. Metab. Cardiovasc. Dis. 2013, 23, 822–828. [Google Scholar] [CrossRef]
- Fumeron, F.; Lamri, A.; Abi Khalil, C.; Jaziri, R.; Porchay-Baldérelli, I.; Lantieri, O.; Vol, S.; Balkau, B.; Marre, M. Dairy Consumption and the Incidence of Hyperglycemia and the Metabolic Syndrome. Diabetes Care 2011, 34, 813–817. [Google Scholar] [CrossRef] [PubMed]
- Sluijs, I.; Forouhi, N.G.; Beulens, J.W.J.; van der Schouw, Y.T.; Agnoli, C.; Arriola, L.; Balkau, B.; Barricarte, A.; Boeing, H.; Bueno-de-Mesquita, H.B.; et al. The amount and type of dairy product intake and incident type 2 diabetes: Results from the EPIC-InterAct Study. Am. J. Clin. Nutr. 2012, 96, 382–390. [Google Scholar]
- Pittas, A.G.; Dawson-Hughes, B.; Li, T.; Van Dam, R.M.; Willett, W.C.; Manson, J.E.; Hu, F.B. Vitamin D and calcium intake in relation to type 2 diabetes in women. Diabetes Care 2006, 29, 650–656. [Google Scholar] [CrossRef] [PubMed]
- Van Dam, R.M.; Hu, F.B.; Rosenberg, L.; Krishnan, S.; Palmer, J.R. Dietary calcium and magnesium, major food sources, and risk of type 2 diabetes in U.S. black women. Diabetes Care 2006, 29, 2238–2243. [Google Scholar] [CrossRef] [PubMed]
- Villegas, R.; Gao, Y.-T.; Dai, Q.; Yang, G.; Cai, H.; Li, H.; Zheng, W.; Shu, X.O. Dietary calcium and magnesium intakes and the risk of type 2 diabetes: The Shanghai Women’s Health Study. Am. J. Clin. Nutr. 2009, 89, 1059–1067. [Google Scholar] [CrossRef]
- Kirii, K.; Mizoue, T.; Iso, H.; Takahashi, Y.; Kato, M.; Inoue, M.; Noda, M.; Tsugane, S. Calcium, vitamin D and dairy intake in relation to type 2 diabetes risk in a Japanese cohort. Diabetologia 2009, 52, 2542–2550. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.M.; Woo, H.W.; Kim, M.K.; Lee, Y.-H.; Shin, D.H.; Shin, M.-H.; Choi, B.Y. Dietary total, animal, vegetable calcium and type 2 diabetes incidence among Korean adults: The Korean Multi-Rural Communities Cohort (MRCohort). Nutr. Metab. Cardiovasc. Dis. 2017, 27, 1152–1164. [Google Scholar] [CrossRef] [PubMed]
- Gagnon, C.; Lu, Z.X.; Magliano, D.J.; Dunstan, D.W.; Shaw, J.E.; Zimmet, P.Z.; Sikaris, K.; Grantham, N.; Ebeling, P.R.; Daly, R.M. Serum 25-hydroxyvitamin D, calcium intake, and risk of type 2 diabetes after 5 years: Results from a national, population-based prospective study (the Australian diabetes, obesity and lifestyle study). Diabetes Care 2011, 34, 1133–1138. [Google Scholar] [CrossRef]
- Reyes-Garcia, R.; Mendoza, N.; Palacios, S.; Salas, N.; Quesada-Charneco, M.; Garcia-Martin, A.; Fonolla, J.; Lara-Villoslada, F.; Muñoz-Torres, M. Effects of Daily Intake of Calcium and Vitamin D-Enriched Milk in Healthy Postmenopausal Women: A Randomized, Controlled, Double-Blind Nutritional Study. J. Women’s Heal. 2018, 27, 561–568. [Google Scholar] [CrossRef]
- Grantham, N.M.; Magliano, D.J.; Hodge, A.; Jowett, J.; Meikle, P.; Shaw, J.E. The association between dairy food intake and the incidence of diabetes in Australia: The Australian Diabetes Obesity and Lifestyle Study (AusDiab). Public Health Nutr. 2013, 16, 339–345. [Google Scholar] [CrossRef]
- Malik, V.S.; Sun, Q.; van Dam, R.M.; Rimm, E.B.; Willett, W.C.; Rosner, B.; Hu, F.B. Adolescent dairy product consumption and risk of type 2 diabetes in middle-aged women1–3. Am. J. Clin. Nutr. 2011, 94, 854–861. [Google Scholar] [CrossRef]
- Dehghan, M.; Mente, A.; Rangarajan, S.; Sheridan, P.; Mohan, V.; Iqbal, R.; Gupta, R.; Lear, S.; Wentzel-Viljoen, E.; Avezum, A.; et al. Association of dairy intake with cardiovascular disease and mortality in 21 countries from five continents (PURE): A prospective cohort study. Lancet 2018, 392, 2288–2297. [Google Scholar] [CrossRef]
- Rideout, T.C.; Marinangeli, C.P.F.; Martin, H.; Browne, R.W.; Rempel, C.B. Consumption of low-fat dairy foods for 6 months improves insulin resistance without adversely affecting lipids or bodyweight in healthy adults: A randomized free-living cross-over study. Nutr. J. 2013, 12, 56. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Norat, T.; Romundstad, P.; Vatten, L.J. Dairy products and the risk of type 2 diabetes: A systematic review and dose-response meta-analysis of cohort studies. Am. J. Clin. Nutr. 2013, 98, 1066–1083. [Google Scholar] [CrossRef] [PubMed]
- Krebs-Smith, S.M.; Guenther, P.M.; Subar, A.F.; Kirkpatrick, S.I.; Dodd, K.W. Americans do not meet federal dietary recommendations. J. Nutr. 2010, 140, 1832–1838. [Google Scholar] [CrossRef]
- Liu, A.-D.; Zhang, B.; DU, W.-W.; Wang, H.-J.; Su, C.; Zhai, F.-Y. Milk consumption and its changing trend of Chinese adult aged 18–44 in nine provinces (autonomous region) from 1991 to 2006. Zhonghua Yu Fang Yi Xue Za Zhi 2011, 45, 304–309. [Google Scholar] [PubMed]
- NIH Consensus conference. Optimal calcium intake. NIH Consensus Development Panel on Optimal Calcium Intake. JAMA 1994, 272, 1942–1948. [Google Scholar] [CrossRef]
- Ross, A.C.; Taylor, C.L.; Yaktine, A.L.; Del Valle, H.B. (Eds.) Dietary Reference Intakes for Calcium and Vitamin D; National Academies Press: Washington, DC, USA, 2012; Volume 130, ISBN 978-0-309-16394-1. [Google Scholar]
- Rosen, C.J.; Abrams, S.A.; Aloia, J.F.; Brannon, P.M.; Clinton, S.K.; Durazo-Arvizu, R.A.; Gallagher, J.C.; Gallo, R.L.; Jones, G.; Kovacs, C.S.; et al. IOM Committee Members Respond to Endocrine Society Vitamin D Guideline. J. Clin. Endocrinol. Metab. 2012, 97, 1146–1152. [Google Scholar] [CrossRef] [PubMed]
- Scragg, R. Emerging Evidence of Thresholds for Beneficial Effects from Vitamin D Supplementation. Nutrients 2018, 10, 561. [Google Scholar] [CrossRef] [PubMed]
| Study, Year | Country | Population Characteristics (Mean age/age range (years)) | Type of Treatment | N | Duration (months) | Main Outcomes |
|---|---|---|---|---|---|---|
| LeBlanc et al. D2d Research Group [54] 2018 | US | Prediabetes (59) | 4000 IU D3/day vs. placebo | 2423 | 36 | Not yet published |
| Forouhi et al. [41] 2016 | UK | Prediabetes IFG or IGT or positive diabetes risk score (53) | 100,000 IU D2/month or 100,000 IU D3/month vs. placebo | 340 | 4 | No effect on HbA1c Improve pulse wave velocity (arterial stiffness) |
| Wagner et al. [48] 2016 | Sweden | Prediabetes or diet-controlled T2D (67.3) | 30,000 IU D3/week vs. placebo | 44 | 2 | No difference in insulin secretion/sensitivity, beta cell function and glucose tolerance |
| Tuomainen et al. [47] 2015 | Finland | Prediabetes (65.7) | 40 μg/day D3 or 80 μg/day D3 vs. placebo | 68 | 5 | No difference in glucose homeostasis indicators |
| Gagnon et al. [44] 2014 | Australia | 25(OH)D ≤ 22ng/ml at risk of T2D (54) | 1200 mg calcium carbonate and 2000–6000 IU D3 day to target vs. placebo | 95 | 6 | No difference in insulin secretion/sensitivity and beta cell function. A post-hoc analysis (only prediabetes patients) showed a significant beneficial effect on insulin sensitivity |
| Sollid et al. [40] 2014 | Norway | Prediabetes IFG or IGT (62.1) | 20,000 IU D3/week vs. placebo | 511 | 12 | No difference in insulin secretion/sensitivity or glucose metabolism, blood pressure or lipid status |
| Oosterwerff et al. [45] 2014 | Netherlands | Overweight, vitamin D deficient subjects with prediabetes (20–65) | Calcium carbonate 500 mg (all) and 1200 IU D3/day vs. placebo | 130 | 4 | No difference in insulin sensitivity or in beta cell function. A post hoc analysis (without diabetes patients at baseline), showed a significant increase in the insulinogenic index when 25(OH)D ≥ 60 nmol/L |
| Salehpour et al. [46] 2013 | Iran | Healthy, overweight/obese women (38) | 25 μg D3/daily vs. placebo | 77 | 4 | No effect in glycemic indices (glucose, insulin, HbA1c and HOMA-IR) |
| Belenchia et al. [55] 2013 | US | Obese adolescents (14.1) | 4000 IU D3/day vs. placebo | 35 | 6 | Significant effect in fasting insulin, HOMA-IR and leptin-to-adiponectin ratio |
| Davidson et al. [43] 2013 | US | Prediabetes and hypovitaminosis D (52) | D3 to target serum 25OHD level of 65–90 ng/mL vs. placebo | 109 | 12 | No difference on insulin secretion/sensitivity or the development of diabetes or returning to normal glucose tolerance |
| Mitri et al. [50] 2011 | US | Prediabetes. At risk for T2D (57) | 2000 IU D3/daily vs. calcium carbonate 800 mg/day | 92 | 4 | Significant effect in beta cell function and improvement in insulin secretion |
| von Hurst et al. [49] 2010 | New Zealand | Insulin resistance, At risk for T2D 25(OH)D < 20 ng/mL (23–68) | 4000 IU D3/day vs. placebo | 81 | 6.5 | No difference in FPG, HOMA2%B; C-peptide Significant effect on HOMA IR and insulin |
| Jorde et al. [56] 2010 | Norway | Overweight/Obese; At risk for T2D (21–70) | 500 mg calcium/day plus D3, 40,000 IU/week or D3 20,000 IU/week | 438 | 48 | No difference in HbA1c, FPG, 2hs PG and HOMA-IR |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muñoz-Garach, A.; García-Fontana, B.; Muñoz-Torres, M. Vitamin D Status, Calcium Intake and Risk of Developing Type 2 Diabetes: An Unresolved Issue. Nutrients 2019, 11, 642. https://doi.org/10.3390/nu11030642
Muñoz-Garach A, García-Fontana B, Muñoz-Torres M. Vitamin D Status, Calcium Intake and Risk of Developing Type 2 Diabetes: An Unresolved Issue. Nutrients. 2019; 11(3):642. https://doi.org/10.3390/nu11030642
Chicago/Turabian StyleMuñoz-Garach, Araceli, Beatriz García-Fontana, and Manuel Muñoz-Torres. 2019. "Vitamin D Status, Calcium Intake and Risk of Developing Type 2 Diabetes: An Unresolved Issue" Nutrients 11, no. 3: 642. https://doi.org/10.3390/nu11030642
APA StyleMuñoz-Garach, A., García-Fontana, B., & Muñoz-Torres, M. (2019). Vitamin D Status, Calcium Intake and Risk of Developing Type 2 Diabetes: An Unresolved Issue. Nutrients, 11(3), 642. https://doi.org/10.3390/nu11030642






