Dietary Polyphenols Targeting Arterial Stiffness: Interplay of Contributing Mechanisms and Gut Microbiome-Related Metabolism
Abstract
1. Introduction
2. Arterial Stiffness
3. Factors Contributing to Arterial Stiffness
4. Dietary Polyphenols
5. Bioavailability of Polyphenols
6. Polyphenols and the Intestinal Microbiome
6.1. Polyphenol Metabolism by the Gut Microbiota
6.2. Polyphenols Modulate Intestinal Microbiota Composition
6.3. Intestinal Microbial Metabolism with Impact on Cardiovascular Health
7. Epidemiological Studies with AS Assessment
8. Interventional Human Studies with AS Assessment
8.1. Cocoa, Coffee, Tea, and Their Isolated Polyphenols
8.2. Fruit, Wine, and Their Isolated Polyphenols
8.3. Soy and Isoflavonoids
8.4. Miscellaneous Dietary Polyphenols
| Dietary Intervention/Polyphenol | Study Design | Health Status | Effects | References |
|---|---|---|---|---|
| Flavonoid-rich dark chocolate (single dose of 100 g) | 17 young volunteers; randomized, single-blind, sham procedure-controlled, cross-over design | Healthy | ↑ resting and hyperemic brachial artery diameter; ↑ FMD; ↓ Aix; No change in PWV | [114] |
| Cocoa (0, 80, 200, 500, and 800 mg cocoa flavonoids/day/10 g cocoa in five periods of 1 week) | 20 volunteers; randomized, double-blind, controlled, cross-over design | Healthy | ↑ FMD; ↓ PWV; ↓ BP; ↓ pulse pressure; ↓ ET-1 | [115] |
| Flavanol-rich dark chocolate vs. flavanol-free white chocolate (100 g/day for 3 days) | 12 volunteers | Healthy | Dark chocolate ingestion improved flow-mediated dilation (p = 0.03), wave reflections, endothelin-1 and 8-iso-PGF(2α) in contrast to white chocolate effects | [116] |
| Flavanol-rich dark chocolate vs. flavanol-free white chocolate, (100 g/day for 15 days) | 19 volunteers (11 M); cross-over design | Hypertensive patients with IGT | ↓ systolic and diastolic BP; ↑ FMD; ↑ insulin sensitivity; | [117] |
| Flavonoid-rich vs. flavonoid-poor dark chocolate | 32 volunteers (16 M); sleep deprivation, randomized double-blind crossover design | Healthy | flavanol-rich chocolate promote: ↓ BP; ↓ pulse pressure; ↑ FMD; mitigated the increase in pulse-wave velocity | [118] |
| Cocoa flavanol-containing (450 mg) drink vs. cocoa flavanol-free control drink (twice a day for 14 days) | 22 young (M) and 20 elderly (M) volunteers; randomized, controlled, double-masked, parallel-group dietary intervention trial | Healthy | ↑ FMD in both groups; ↓ pulse wave velocity; ↓ total peripheral resistance-, ↑ arteriolar and microvascular vasodilator capacity; ↓ Aix in elderly | [119] |
| Cocoa beverage (960 mg total polyphenols; 480 mg flavanols) | 18 volunteers; randomized, double-blind, crossover study | T2D | ↓ large artery elasticity | [120] |
| Dark chocolate (37 g/day) and a sugar-free cocoa beverage (total cocoa 22 g/day, total flavanols 814 mg/d, 4 weeks) | 30 middle-aged volunteers (15 M); randomized, placebo-controlled, cross-over study | Overweight | ↑ basal diameter and peak diameter of the brachial artery and basal blood flow volume; ↓ Aix in only women | [121] |
| (2)-Epicatechin (100 mg/d), quercetin-3-glucoside (160 mg/d) or placebo (capsules for 4 weeks) | 37 volunteers; a randomized, double-blind, placebo-controlled, crossover trial | Healthy | no effect on FMD, arterial stiffness | [122] |
| Dark chocolate (70 g, 150 mg epicatechin) and pure epicatechin capsules (2 × 50 mg epicatechin) with 75 g white chocolate | 20 (M) volunteers; randomized crossover study | Healthy | dark chocolate and epicatechin significantly ↑ FMD; ↓ Aix | [123] |
| Chlorogenic acid (450 mg or 900 mg) vs. 200 mg (−)-epicatechin | 16 volunteers; cross-over study | Healthy | no effect on BP; no significant effect on peak FMD response; ↑ post-ischemic FMD response | [124] |
| Coffee polyphenol extract (355 mg chlorogenic acids) | 19 (M) volunteers; randomized, acute, crossover, intervention study | Healthy | ↑ secretion of Glucagon-like peptide 1; ↑ postprandial hyperglycemia; ↑ FMD | [125] |
| Black tea (0, 100, 200, 400, and 800 mg tea flavonoids/day in 5 periods of 1 week) | 19 (M) volunteers | Healthy | ↑ FMD; ↓ blood pressure; ↓ stiffness index | [126] |
| Green tea (9 g/day for 4 weeks) | 55 (31 M) volunteers; randomized, cross-over | T2D | No effect on brachial-ankle PWV; No effect on inflammatory markers | [127] |
| Green tea (836 mg catechins, acute) | 20 volunteers; 2 different catechol-O-methyltransferase genotypes | Healthy | ↓ digital volume pulse stiffness index (SI) in GG subjects; ↑ BP and insulin response in GG subjects | [128] |
| Dietary Intervention | Study Design | Health Status | Effects | References |
|---|---|---|---|---|
| Apple with skin (acute and 4 weeks) | 30 volunteers; randomized, controlled, cross-over | Healthy | ↑ FMD | [129] |
| Red (anthocyanin-rich) or blond (anthocyanin-poor) orange juice (1 liter, acute) | 18 volunteers (9 M); Randomized, cross-over design | Healthy | ↓ Aix after red orange juice | [130] |
| Grapefruit juice (340 mL/day (210 mg naringenin glycosides), for 6 months) | 48 postmenopausal women; double-blind, randomized, controlled, cross-over | Healthy | ↓ carotid-femoral PWV | [131] |
| Orange juice or hesperidin supplement (acute intake; both 320 mg hesperidin) | 16 fasted volunteers (M) | Healthy | no effect on endothelial function; no effect on arterial stiffness; no effect on BP | [132] |
| Pomegranate extract-containing drink (<50 mg pomegranate polyphenols per 237 mL) | 19 young volunteers (M); randomized, controlled, crossover | Healthy | no effect on digital volume pulse-stiffness index | [133] |
| Pomegranate juice (330 mL/day for 4 weeks) | 51 adults volunteers (16 M) | Healthy | no effect on PWV; ↓ systolic and diastolic BP; ↓ mean arterial pressure | [134] |
| Mango fruit preparation Careless™ (single dose of 100 mg or 300 mg) | 10 volunteers (F); randomized, double-blind, crossover pilot study | Healthy | ↑ coetaneous blood flow; No effect on endothelial function | [135] |
| Cranberry juice cocktail (500 mL/day (27% juice) for 4 weeks) | 35 volunteers (M); double-blind, cross-over | Healthy | no effect on Aix; ↓ in Aix significant within-group in abdominally obese M | [136] |
| Cranberry juice (54% juice, 835 mg total polyphenols, and 94 mg anthocyanins, for 4 weeks) | 15 volunteers; acute pilot study 44 volunteers; chronic placebo-controlled crossover | Coronary heart disease | ↑ brachial artery FMD and digital pulse amplitude tonometry ratio in the pilot study; ↓ carotid-femoral PWV for chronic treatment | [137] |
| Blueberry (300 g of blueberry) | 16 smokers (M) 3-armed randomized-controlled | Healthy | ↓ peripheral arterial dysfunction; no differences in digital augmentation index | [138,139] |
| Blueberry (300 g of blueberry) | 24 volunteers (M) (12 non-smokers and 12 smokers) | Healthy | ↓ peripheral arterial dysfunction (reactive hyperemia index); no change in digital augmentation index dAix | [140] |
| Blueberry powder (22 g freeze-dried, for 8 weeks) | 48 postmenopausal (F); randomized, double-blind, placebo-controlled | Pre- and stage 1-hypertension | ↓ systolic and diastolic BP; ↓ brachial-ankle PWV | [141] |
| Strawberry powder (40 g freeze-dried) | 30 overweight or obese adults (17 M) | Healthy | no effect on vascular function | [142] |
| Blackcurrant extract (low sugar fruit drinks containing 150, 300, and 600 mg of total anthocyanins, acute) | 14 (M) and 9 postmenopausal (F); randomized, double-blind, cross-over | Healthy | no effect on arterial stiffness; no effect on 8-isoprostane F2α | [143] |
| Black raspberry (750 mg/day, acute, and 12 weeks) | 26 and 39 volunteers, respectively | Metabolic syndrome | ↓ augmentation index acutely; ↑ brachial artery FMD after 12 weeks of treatment | [144,145] |
| Concord grape juice (7 mL/kg/day, 70-kg person consumed 490 mL/day; 965 mg total polyphenols and 327 kcal, 2-weeks) | 26 healthy smokers (10 M); randomized, placebo-controlled, double-blind, cross-over | Healthy | ↑ values of FMD and PWV | [146] |
| Grape seed extract (150 mg twice daily, 6 weeks) | 29 middle-aged (15 M); single-center, randomized, two-arm, double-blinded, placebo-controlled | Pre- hypertension | ↓ systolic and diastolic BP; no significant changes in FMD | [147] |
| Grape-wine extract (capsules MegaNatural™ combined with Provinols™, 4 weeks) | 60 volunteers; double-blind, placebo-controlled, crossover | Mildly Hypertensive, untreated | ↓ 24-h ambulatory systolic and diastolic BPs; no effect on FMD | [148] |
| Red wine (400 mL, ~13% (v/v) alcohol, 6 weeks) | 45 postmenopausal women; randomized parallel-arm | Hypercholesterolemia | ↓ Aix; no effect on central hemodynamic parameters | [149] |
| Resveratrol (100 mg tablet, oligo-stilbene 27.97 mg/100 mg/day, 12 weeks) | 25 volunteers (15 M); double-blind, randomized, placebo-controlled | T2D | ↓ systolic BP; ↓ cardio-ankle vascular index | [150] |
| Resveratrol (resVida™; 6 capsules, 30, 90, and 270 mg, single dose) | 19 volunteers (14 M); double-blind, placebo-controlled | Overweight/obese/post-menopausal untreated borderline hypertension | ↑ FMD response | [151] |
| Resveratrol (Resvida, 75 mg capsule/day, 6 weeks) | 28 obese volunteers (12M); randomized, double-blind, placebo-controlled crossover | Healthy | ↑ FMD response; no effect on BP and arterial compliance | [152] |
| Dietary Intervention | Study Design | Health Status | Effects | References |
|---|---|---|---|---|
| Isoflavone, red clover-extracted (500-mg tablets, 2 × 40 mg of isoflavones/day, 6 weeks) | 80 volunteers (46 M); randomized, double-blind, cross-over, placebo-controlled | Healthy | improved arterial stiffness; ↑ systemic arterial compliance; ↓ total peripheral resistance; ↓ central PWV | [153] |
| Isoflavone (50 mg/day, as black soybean tea, 2 months) | 55 volunteers (F); smokers and nonsmokers | Healthy | ↓ cardio-ankle vascular index in premenopausal; no effect in postmenopausal; no effect on BP and brachial-ankle PWV | [154] |
| Isoflavone-containing soya protein isolate (50 g/d soya protein, 6 weeks) | 20 volunteers (9 M); randomized, placebo-controlled, cross-over | Moderately elevated brachial BP | ↓ brachial diastolic BP; no effect on Aix and PWV | [155] |
| Flavonoid-enriched chocolate (split dose of 27 g/day (850 mg flavan-3-ols (90 mg epicatechin)) + 100 mg isoflavones (aglycone equivalents)/day, 1 year) | 93 postmenopausal volunteers; double-blind, parallel-design, placebo-controlled | T2D | no change in intima-media thickness of the common carotid artery Aix or BP improved pulse pressure variability; equol producers had larger ↓ in diastolic BP, mean arterial pressure, and PWV | [156] |
| Soy germ pasta (80-g serving/day, naturally enriched in isoflavone aglycons, 4 weeks) | 62 volunteers (25 M); randomized, controlled, parallel study | Hypercholesterolemia | improved arterial stiffness; the best effect in equol producers | [157] |
| Soy germ pasta (one serving/day of (31–33 mg) total isoflavones), 8 weeks) | 26 volunteers (13 M); randomized, controlled, double-blind, crossover | T2D | improved arterial stiffness; ↓ systolic and diastolic BP | [158] |
| Isoflavone capsule (80 mg aglycone equivalents of daidzein and genistein, a SoyLife extract (40%) with a typical soy germ ratio of genistein:daidzein: glycitein (15:50:35), acute) | 28 volunteers; equol producer phenotype (14 M), double-blind, placebo-controlled crossover | Healthy | improved carotid-femoral PWV in equol producers; no vascular effects | [54] |
| Soy nuts snack (70 g of soy nuts: 101 mg of aglycone equivalents (55 mg of genistein, 42 mg of daidzein, and 4 mg of glycitein), 4 weeks) | 17 volunteers (12 postmenopausal F, 5 M) | Metabolic syndrome | improved arterial stiffness (Aix) | [159] |
| Dietary Intervention | Study Design | Health Status | Effects | References |
|---|---|---|---|---|
| Olive leaf extract (51 mg oleuropein; 10 mg hydroxytyrosol, acute) | 18 volunteers (9 M); randomized, double-blind, placebo-controlled, cross-over | Healthy | ↓ digital volume pulse-stiffness index; ↓ ex vivo IL-8 production | [160] |
| Red yeast rice and olive fruit extract (9, 32 mg hydroxytyrosol) | 50 volunteers; randomized, double-blind, placebo-controlled | Metabolic syndrome | ↓ SBP and DBP; ↓ LDL and oxidized LDL; ↓ lipoprotein-associated phospholipase A2 | [161,162] |
| Olive fruit extract (50 mg and 100 mg hydroxytytosol) | 36 volunteers; 11-day, double-blind, placebo-controlled | Risk for arterial stiffness | ↓ Cardio-Ankle Vascular Index | [163] |
| Polyphenols (250 mL beverage: 361 mg of (poly)phenols, 120 mg vitamin C; twice/day, 4 weeks) | 20 volunteers (10 M); a randomized, double-blind, placebo-controlled design | Healthy | No effect on the cutaneous vascular response; No effect on PWV | [164] |
| Curcumin capsules (250 mg of curcuminoids, 3 capsules/twice a day, 6 months) | 107 volunteers (50 M); randomized, double-blinded, placebo-controlled | T2D | ↓ PWV | [165] |
| Curcumin (25 mg of highly absorptive curcumin dispersed with colloidal nanoparticles, 6 pills/day, 8 weeks) | 32 sedentary postmenopausal women (F) | Healthy | ↓ FMD | [166] |
| Walnut-enriched ad libitum diet (56 g of shelled, unroasted English walnuts/day, 8 weeks) | 46 volunteers (18 M); randomized, controlled, single-blind, crossover clinical | Overweight | ↓ FMD, beneficial trends in systolic BP reduction | [167] |
| Onion skin extract (162 mg/day quercetin, 6 weeks) | 70 volunteers (35 M); double-blinded, placebo-controlled cross-over | Healthy | ↓ 24 h systolic BP in the subgroup of hypertensives | [168] |
| Lemon balm extract (3.3 g of lemon balm leaves extracted in 200 mL of hot water, once daily, 4 weeks) | 28 Japanese volunteers (14 M); an open-label, parallel-group comparative | Healthy | ↓ in brachial-ankle PWV | [169] |
9. Impact on Mechanisms Contributing to AS
9.1. Vascular Effects
9.2. Oxidant Status
9.3. Anti-Inflammatory Activity
9.4. Antiglycation/AGEs
9.5. Autophagy
10. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| AP-1 | Activator protein 1 |
| AMPK | adenosine monophosphate-activated protein kinase |
| AGEs | advanced glycation end-products |
| ACE | angiotensin converting enzyme |
| Ang II | angiotensin II |
| AT-1 | angiotensin II receptor type 1 |
| ARE | antioxidant response element protein |
| Aix | aortic augmentation index |
| AS | arterial stiffness |
| AhR | aryl hydrocarbon receptor |
| CV | cardiovascular |
| CAT | catalase |
| COMT | catechol-O-methyltransferase |
| COX-2 | cyclooxygenase 2 |
| CRP | C-reactive protein |
| DBP | diastolic blood pressure |
| eNOS | endothelial nitric oxide synthase |
| E-selectin | endothelial selectin |
| ET-1 | endothelin-1 |
| EGCG | epigallocatechin gallate |
| EFSA | European Food Safety Authority |
| ERK | extracellular signal-regulated kinases |
| FMD | flow-mediated dilation |
| GSL | glutamyl-cysteine ligase |
| GPx | glutathione peroxidase |
| GR | glutathione reductase |
| GST | glutathione-S-transferase |
| Hmox | heme oxygenase |
| Hmox-1 | heme oxygenase 1 |
| HDL | high-density lipoprotein |
| HUVECs | human umbilical vein endothelial cells |
| HOCl | hypochlorous acid |
| IGF-1 | insulin-like growth factor 1 |
| ICAM-1 | intercellular adhesion molecule-1 |
| IFN-γ | interferon |
| IL-6 | interleukin 6 |
| LV | left ventricular |
| LPS | lipopolysaccharide |
| LOX-1 | lectin-like oxidized low-density lipoprotein receptor 1 |
| LOX | lysyl oxidase |
| MMP | matrix metalloproteinase |
| MAPK | mitogen-activated protein kinase |
| MCP | monocyte chemoattractant protein |
| l-NAME | N(ω)-nitro-l-arginine methyl ester |
| Nrf2 | NFE2-related factor 2 |
| NADPH | nicotinamide adenine dinucleotide phosphate |
| NO | nitric oxide |
| NF-κB | nuclear factor kappa B |
| oxLDL | oxidized low-density lipoprotein |
| PONs | paraoxonase |
| PPAR-γ | peroxisome proliferator-activated receptor γ |
| PI3K | phosphoinositide 3-kinase |
| PKC | protein kinase C |
| PWV | pulse wave velocity |
| NLRP3 | pyrin-domain containing 3 |
| RNS | reactive nitrogen species |
| ROS | reactive oxygen species |
| RAGE | receptor of AGE |
| RAAS | renin-angiotensin-aldosterone system |
| SCFA | short-chain fatty acids |
| SIRT | sirtuin |
| sRAGE | soluble receptors for AGE |
| SOD | superoxide dismutase |
| SBP | systolic blood pressure |
| TBARS | thiobarbituric acid reactive substances |
| TRX | thioredoxin |
| TLR | toll-like receptor |
| TG | triglyceride |
| TMAO | trimethylamine-N-oxide |
| TNF | tumor necrosis factor |
| T2D | type 2 diabetes |
| VCAM-1 | vascular cell adhesion molecule-1 |
| VSMCs | vascular smooth muscle cells |
| XO | xanthine oxidase |
References
- Hamilton, P.K.; Lockhart, C.J.; Quinn, C.E.; McVeigh, G.E. Arterial stiffness: Clinical relevance, measurement and treatment. Clin. Sci. (Lond.) 2007, 113, 157–170. [Google Scholar] [CrossRef] [PubMed]
- Laurent, S.; Cockcroft, J.; Van Bortel, L.; Boutouyrie, P.; Giannattasio, C.; Hayoz, D.; Pannier, B.; Vlachopoulos, C.; Wilkinson, I.; Struijker-Boudier, H.; et al. Expert consensus document on arterial stiffness: Methodological issues and clinical applications. Eur. Heart J. 2006, 27, 2588–2605. [Google Scholar] [CrossRef] [PubMed]
- Della Corte, V.; Tuttolomondo, A.; Pecoraro, R.; Di Raimondo, D.; Vassallo, V.; Pinto, A. Inflammation, Endothelial Dysfunction and Arterial Stiffness as Therapeutic Targets in Cardiovascular Medicine. Curr. Pharm. Des. 2016, 22, 4658–4668. [Google Scholar] [CrossRef] [PubMed]
- Palombo, C.; Kozakova, M. Arterial stiffness, atherosclerosis and cardiovascular risk: Pathophysiologic mechanisms and emerging clinical indications. Vascul. Pharmacol. 2016, 77, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Lyle, A.N.; Raaz, U. Killing Me Unsoftly: Causes and Mechanisms of Arterial Stiffness. Arterioscler. Thromb. Vasc. Biol. 2017, 37, e1–e11. [Google Scholar] [CrossRef] [PubMed]
- O’Rourke, M.F.; Hashimoto, J. Mechanical factors in arterial aging: A clinical perspective. J. Am. Coll. Cardiol. 2007, 50, 1–13. [Google Scholar] [CrossRef]
- Van Bortel, L. Arterial stiffness: From surrogate marker to therapeutic target. Artery Res. 2016, 14, 10–14. [Google Scholar] [CrossRef]
- Cecelja, M.; Chowienczyk, P. Molecular Mechanisms of Arterial Stiffening. Pulse 2016, 4, 43–48. [Google Scholar] [CrossRef]
- Quinn, U.; Tomlinson, L.A.; Cockcroft, J.R. Arterial stiffness. JRSM Cardiovasc. Dis. 2012, 1. [Google Scholar] [CrossRef]
- Avolio, A. Arterial Stiffness. Pulse 2013, 1, 14–28. [Google Scholar] [CrossRef]
- Sell, D.R.; Monnier, V.M. Molecular Basis of Arterial Stiffening: Role of Glycation—A Mini-Review. Gerontology 2012, 58, 227–237. [Google Scholar] [CrossRef] [PubMed]
- Smulyan, H.; Mookherjee, S.; Safar, M.E. The two faces of hypertension: Role of aortic stiffness. J. Am. Soc. Hypertens. 2016, 10, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Vlassopoulos, A.; Lean, M.E.J.; Combet, E. Oxidative stress, protein glycation and nutrition—Interactions relevant to health and disease throughout the lifecycle. Proc. Nutr. Soc. 2014, 73, 430–438. [Google Scholar] [CrossRef] [PubMed]
- Papaioannou, T.G.; Karatzi, K.; Psaltopoulou, T.; Tousoulis, D. Arterial ageing: Major nutritional and life-style effects. Ageing Res. Rev. 2017, 37, 162–163. [Google Scholar] [CrossRef] [PubMed]
- Lilamand, M.; Kelaiditi, E.; Guyonnet, S.; Antonelli Incalzi, R.; Raynaud-Simon, A.; Vellas, B.; Cesari, M. Flavonoids and arterial stiffness: Promising perspectives. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 698–704. [Google Scholar] [CrossRef] [PubMed]
- Li, P.; Wang, L.; Liu, C. Overweightness, obesity and arterial stiffness in healthy subjects: A systematic review and meta-analysis of literature studies. Postgrad. Med. 2017, 129, 224–230. [Google Scholar] [CrossRef]
- Wu, C.F.; Liu, P.Y.; Wu, T.J.; Hung, Y.; Yang, S.P.; Lin, G.M. Therapeutic modification of arterial stiffness: An update and comprehensive review. World J. Cardiol. 2015, 7, 742–753. [Google Scholar] [CrossRef]
- Sasaki, Y.; Ikeda, Y.; Iwabayashi, M.; Akasaki, Y.; Ohishi, M. The Impact of Autophagy on Cardiovascular Senescence and Diseases. Int. Heart J. 2017, 58, 666–673. [Google Scholar] [CrossRef]
- LaRocca, T.J.; Martens, C.R.; Seals, D.R. Nutrition and other lifestyle influences on arterial aging. Ageing Res. Rev. 2017, 39, 106–119. [Google Scholar] [CrossRef]
- Mozos, I.; Stoian, D.; Luca, C.T. Crosstalk between Vitamins A, B12, D, K, C, and E Status and Arterial Stiffness. Dis. Markers 2017. [Google Scholar] [CrossRef]
- Daiber, A.; Steven, S.; Weber, A.; Shuvaev, V.V.; Muzykantov, V.R.; Laher, I.; Li, H.; Lamas, S.; Münzel, T. Targeting vascular (endothelial) dysfunction. Br. J. Pharmacol. 2017, 174, 1591–1619. [Google Scholar] [CrossRef] [PubMed]
- Sharman, J.E.; Boutouyrie, P.; Laurent, S. Arterial (Aortic) Stiffness in Patients with Resistant Hypertension: From Assessment to Treatment. Curr. Hypertens. Rep. 2017, 19. [Google Scholar] [CrossRef] [PubMed]
- Santilli, F.; D’Ardes, D.; Davi, G. Oxidative stress in chronic vascular disease: From prediction to prevention. Vasc. Pharmacol. 2015, 74, 23–37. [Google Scholar] [CrossRef] [PubMed]
- Mozos, I.; Luca, C.T. Crosstalk between Oxidative and Nitrosative Stress and Arterial Stiffness. Curr. Vasc. Pharmacol. 2017, 15, 446–456. [Google Scholar] [CrossRef] [PubMed]
- Mozos, I.; Malainer, C.; Horbanczuk, J.; Gug, C.; Stoian, D.; Luca, C.T.; Atanasov, A.G. Inflammatory Markers for Arterial Stiffness in Cardiovascular Diseases. Front. Immunol. 2017, 8, 16. [Google Scholar] [CrossRef] [PubMed]
- Zanoli, L.; Rastelli, S.; Inserra, G.; Castellino, P. Arterial structure and function in inflammatory bowel disease. World J. Gastroenterol. 2015, 21, 11304–11311. [Google Scholar] [CrossRef] [PubMed]
- De Meyer, G.R.Y.; Grootaert, M.O.J.; Michiels, C.F.; Kurdi, A.; Schrijvers, D.M.; Martinet, W. Autophagy in Vascular Disease. Circ. Res. 2015, 116, 468–479. [Google Scholar] [CrossRef]
- Perrotta, I.; Aquila, S. The role of oxidative stress and autophagy in atherosclerosis. Oxid. Med. Cell. Longev. 2015, 2015, 130315. [Google Scholar] [CrossRef]
- Fang, C.; Gu, L.; Smerin, D.; Mao, S.; Xiong, X. The Interrelation between Reactive Oxygen Species and Autophagy in Neurological Disorders. Oxid. Med. Cell. Longev. 2017, 2017, 8495160. [Google Scholar] [CrossRef]
- Costa, C.; Tsatsakis, A.; Mamoulakis, C.; Teodoro, M.; Briguglio, G.; Caruso, E.; Tsoukalas, D.; Margina, D.; Dardiotis, E.; Kouretas, D.; et al. Current evidence on the effect of dietary polyphenols intake on chronic diseases. Food Chem. Toxicol. 2017, 110, 286–299. [Google Scholar] [CrossRef]
- Manach, C.; Scalbert, A.; Morand, C.; Remesy, C.; Jimenez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef] [PubMed]
- Tresserra-Rimbau, A.; Rimm, E.B.; Medina-Remon, A.; Martinez-Gonzalez, M.A.; de la Torre, R.; Corella, D.; Salas-Salvado, J.; Gomez-Gracia, E.; Lapetra, J.; Aros, F.; et al. Inverse association between habitual polyphenol intake and incidence of cardiovascular events in the PREDIMED study. Nutr. Metab. Carbiovasc. Dis. 2014, 24, 639–647. [Google Scholar] [CrossRef] [PubMed]
- Ludovici, V.; Barthelmes, J.; Nagele, M.P.; Enseleit, F.; Ferri, C.; Flammer, A.J.; Ruschitzka, F.; Sudano, I. Cocoa, Blood Pressure, and Vascular Function. Front. Nutr. 2017, 4, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Rienks, J.; Barbaresko, J.; Nöthlings, U. Association of Polyphenol Biomarkers with Cardiovascular Disease and Mortality Risk: A Systematic Review and Meta-Analysis of Observational Studies. Nutrients 2017, 9, 415. [Google Scholar] [CrossRef] [PubMed]
- Pinto, P.; Santos, C.N. Worldwide (poly)phenol intake: Assessment methods and identified gaps. Eur. J. Nutr. 2017, 56, 1393–1408. [Google Scholar] [CrossRef] [PubMed]
- Gil-Cardoso, K.; Gines, I.; Pinent, M.; Ardevol, A.; Blay, M.; Terra, X. Effects of flavonoids on intestinal inflammation, barrier integrity and changes in gut microbiota during diet-induced obesity. Nutr. Res. Rev. 2016, 29, 234–248. [Google Scholar] [CrossRef] [PubMed]
- Goszcz, K.; Duthie, G.G.; Stewart, D.; Leslie, S.J.; Megson, I.L. Bioactive polyphenols and cardiovascular disease: Chemical antagonists, pharmacological agents or xenobiotics that drive an adaptive response? Br. J. Pharmacol. 2017, 174, 1209–1225. [Google Scholar] [CrossRef]
- Zhang, H.; Tsao, R. Dietary polyphenols, oxidative stress and antioxidant and anti-inflammatory effects. Curr. Opin. Food Sci. 2016, 8, 33–42. [Google Scholar] [CrossRef]
- Milenkovic, D.; Morand, C.; Cassidy, A.; Konic-Ristic, A.; Tomas-Barberan, F.; Ordovas, J.M.; Kroon, P.; De Caterina, R.; Rodriguez-Mateos, A. Interindividual Variability in Biomarkers of Cardiometabolic Health after Consumption of Major Plant-Food Bioactive Compounds and the Determinants Involved. Adv. Nutr. 2017, 8, 558–570. [Google Scholar]
- Menezes, R.; Rodriguez-Mateos, A.; Kaltsatou, A.; Gonzalez-Sarrias, A.; Greyling, A.; Giannaki, C.; Andres-Lacueva, C.; Milenkovic, D.; Gibney, E.R.; Dumont, J.; et al. Impact of Flavonols on Cardiometabolic Biomarkers: A Meta-Analysis of Randomized Controlled Human Trials to Explore the Role of Inter-Individual Variability. Nutrients 2017, 9. [Google Scholar] [CrossRef]
- Almeida, A.F.; Borge, G.I.A.; Piskula, M.; Tudose, A.; Tudoreanu, L.; Valentová, K.; Williamson, G.; Santos, C.N. Bioavailability of Quercetin in Humans with a Focus on Interindividual Variation. Compr. Rev. Food Sci. Food Saf. 2018, 17, 714–731. [Google Scholar] [CrossRef]
- Figueira, I.; Menezes, R.; Macedo, D.; Costa, I.; Dos Santos, C.N. Polyphenols Beyond Barriers: A Glimpse into the Brain. Curr. Neuropharmacol. 2017, 15, 562–594. [Google Scholar] [CrossRef] [PubMed]
- Cardona, F.; Andres-Lacueva, C.; Tulipani, S.; Tinahones, F.J.; Queipo-Ortuno, M.I. Benefits of polyphenols on gut microbiota and implications in human health. J. Nutr. Biochem. 2013, 24, 1415–1422. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, L.A.; Barrow, A. Metabolism of flavonoid compounds in germ-free rats. Biochem. J. 1972, 130, 1161–1162. [Google Scholar] [CrossRef] [PubMed]
- Valdes, L.; Cuervo, A.; Salazar, N.; Ruas-Madiedo, P.; Gueimonde, M.; Gonzalez, S. The relationship between phenolic compounds from diet and microbiota: Impact on human health. Food Funct. 2015, 6, 2424–2439. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.K.; Chang, H.W.; Yan, D.; Lee, K.M.; Ucmak, D.; Wong, K.; Abrouk, M.; Farahnik, B.; Nakamura, M.; Zhu, T.H.; et al. Influence of diet on the gut microbiome and implications for human health. J. Transl. Med. 2017, 15, 17. [Google Scholar] [CrossRef]
- Sheflin, A.M.; Melby, C.L.; Carbonero, F.; Weir, T.L. Linking dietary patterns with gut microbial composition and function. Gut Microbes 2017, 8, 113–129. [Google Scholar] [CrossRef]
- Rowland, I.; Gibson, G.; Heinken, A.; Scott, K.; Swann, J.; Thiele, I.; Tuohy, K. Gut microbiota functions: Metabolism of nutrients and other food components. Eur. J. Nutr. 2018, 57, 1–24. [Google Scholar] [CrossRef]
- Wiese, S.; Esatbeyoglu, T.; Winterhalter, P.; Kruse, H.P.; Winkler, S.; Bub, A.; Kulling, S.E. Comparative biokinetics and metabolism of pure monomeric, dimeric, and polymeric flavan-3-ols: A randomized cross-over study in humans. Mol. Nutr. Food Res. 2015, 59, 610–621. [Google Scholar] [CrossRef]
- Ottaviani, J.I.; Heiss, C.; Spencer, J.P.E.; Kelm, M.; Schroeter, H. Recommending flavanols and procyanidins for cardiovascular health: Revisited. Mol. Asp. Med. 2018, 61, 63–75. [Google Scholar] [CrossRef]
- Trost, K.; Ulaszewska, M.M.; Stanstrup, J.; Albanese, D.; De Filippo, C.; Tuohy, K.M.; Natella, F.; Scaccini, C.; Mattivi, F. Host: Microbiome co-metabolic processing of dietary polyphenols—An acute, single blinded, cross-over study with different doses of apple polyphenols in healthy subjects. Food Res. Int. 2018, 112, 108–128. [Google Scholar] [CrossRef] [PubMed]
- Landete, J.M.; Arques, J.; Medina, M.; Gaya, P.; de Las Rivas, B.; Munoz, R. Bioactivation of Phytoestrogens: Intestinal Bacteria and Health. Crit. Rev. Food Sci. Nutr. 2016, 56, 1826–1843. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Guo, K.; Chen, C.; Wang, P.; Zhang, B.; Zhou, Q.; Mei, F.; Su, Y. Soya isoflavone consumption in relation to carotid intima-media thickness in Chinese equol excretors aged 40–65 years. Br. J. Nutr. 2012, 108, 1698–1704. [Google Scholar] [CrossRef] [PubMed]
- Hazim, S.; Curtis, P.J.; Schar, M.Y.; Ostertag, L.M.; Kay, C.D.; Minihane, A.M.; Cassidy, A. Acute benefits of the microbial-derived isoflavone metabolite equol on arterial stiffness in men prospectively recruited according to equol producer phenotype: A double-blind randomized controlled trial. Am. J. Clin. Nutr. 2016, 103, 694–702. [Google Scholar] [CrossRef] [PubMed]
- Clavel, T.; Desmarchelier, C.; Haller, D.; Gerard, P.; Rohn, S.; Lepage, P.; Daniel, H. Intestinal microbiota in metabolic diseases: From bacterial community structure and functions to species of pathophysiological relevance. Gut Microbes 2014, 5, 544–551. [Google Scholar] [CrossRef] [PubMed]
- Braune, A.; Blaut, M. Bacterial species involved in the conversion of dietary flavonoids in the human gut. Gut Microbes 2016, 7, 216–234. [Google Scholar] [CrossRef]
- Karas, D.; Ulrichova, J.; Valentova, K. Galloylation of polyphenols alters their biological activity. Food Chem. Toxicol. 2017, 105, 223–240. [Google Scholar] [CrossRef]
- Rodriguez-Mateos, A.; Cifuentes-Gomez, T.; Gonzalez-Salvador, I.; Ottaviani, J.I.; Schroeter, H.; Kelm, M.; Heiss, C.; Spencer, J.P. Influence of age on the absorption, metabolism, and excretion of cocoa flavanols in healthy subjects. Mol. Nutr. Food Res. 2015, 59, 1504–1512. [Google Scholar] [CrossRef]
- Tomas-Barberan, F.A.; Gonzalez-Sarrias, A.; Garcia-Villalba, R.; Nunez-Sanchez, M.A.; Selma, M.V.; Garcia-Conesa, M.T.; Espin, J.C. Urolithins, the rescue of “old” metabolites to understand a “new” concept: Metabotypes as a nexus among phenolic metabolism, microbiota dysbiosis, and host health status. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef]
- Espin, J.C.; Gonzalez-Sarrias, A.; Tomas-Barberan, F.A. The gut microbiota: A key factor in the therapeutic effects of (poly) phenols. Biochem. Pharmacol. 2017, 139, 82–93. [Google Scholar] [CrossRef]
- Cortes-Martin, A.; Garcia-Villalba, R.; Gonzalez-Sarrias, A.; Romo-Vaquero, M.; Loria-Kohen, V.; Ramirez-de-Molina, A.; Tomas-Barberan, F.A.; Selma, M.V.; Espin, J.C. The gut microbiota urolithin metabotypes revisited: The human metabolism of ellagic acid is mainly determined by aging. Food Funct. 2018, 9, 4100–4106. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; Wedick, N.M.; Pan, A.; Townsend, M.K.; Cassidy, A.; Franke, A.A.; Rimm, E.B.; Hu, F.B.; van Dam, R.M. Gut microbiota metabolites of dietary lignans and risk of type 2 diabetes: A prospective investigation in two cohorts of U.S. women. Diabetes Care 2014, 37, 1287–1295. [Google Scholar] [CrossRef] [PubMed]
- Vanharanta, M.; Voutilainen, S.; Rissanen, T.H.; Adlercreutz, H.; Salonen, J.T. Risk of cardiovascular disease-related and all-cause death according to serum concentrations of enterolactone: Kuopio Ischaemic Heart Disease Risk Factor Study. Arch. Intern. Med. 2003, 163, 1099–1104. [Google Scholar] [CrossRef] [PubMed]
- van der Schouw, Y.T.; Pijpe, A.; Lebrun, C.E.; Bots, M.L.; Peeters, P.H.; van Staveren, W.A.; Lamberts, S.W.; Grobbee, D.E. Higher usual dietary intake of phytoestrogens is associated with lower aortic stiffness in postmenopausal women. Arterioscler. Thromb. Vasc. Biol. 2002, 22, 1316–1322. [Google Scholar] [CrossRef] [PubMed]
- Morling, J.R.; Yeoh, S.E.; Kolbach, D.N. Rutosides for treatment of post-thrombotic syndrome. Cochrane Database Syst. Rev. 2015, 4, CD005625. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Zapata, M.J.; Vernooij, R.W.; Uriona Tuma, S.M.; Stein, A.T.; Moreno, R.M.; Vargas, E.; Capella, D.; Bonfill Cosp, X. Phlebotonics for venous insufficiency. Cochrane Database Syst. Rev. 2016, 4, CD003229. [Google Scholar] [CrossRef] [PubMed]
- Williamson, G.; Clifford, M.N. Colonic metabolites of berry polyphenols: The missing link to biological activity? Br. J. Nutr. 2010, 104 (Suppl. 3), S48–S66. [Google Scholar] [CrossRef]
- Amaretti, A.; Raimondi, S.; Leonardi, A.; Quartieri, A.; Rossi, M. Hydrolysis of the rutinose-conjugates flavonoids rutin and hesperidin by the gut microbiota and bifidobacteria. Nutrients 2015, 7, 2788–2800. [Google Scholar] [CrossRef]
- Slamova, K.; Kapesova, J.; Valentova, K. “Sweet Flavonoids”: Glycosidase-Catalyzed Modifications. Int. J. Mol. Sci. 2018, 19. [Google Scholar] [CrossRef]
- Najmanova, I.; Pourova, J.; Voprsalova, M.; Pilarova, V.; Semecky, V.; Novakova, L.; Mladenka, P. Flavonoid metabolite 3-(3-hydroxyphenyl)propionic acid formed by human microflora decreases arterial blood pressure in rats. Mol. Nutr. Food Res. 2016, 60, 981–991. [Google Scholar] [CrossRef]
- Pourova, J.; Najmanova, I.; Voprsalova, M.; Migkos, T.; Pilarova, V.; Applova, L.; Novakova, L.; Mladenka, P. Two flavonoid metabolites, 3,4-dihydroxyphenylacetic acid and 4-methylcatechol, relax arteries ex vivo and decrease blood pressure in vivo. Vascul. Pharmacol. 2018, 111, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Khymenets, O.; Urpi-Sarda, M.; Tulipani, S.; Garcia-Aloy, M.; Monagas, M.; Mora-Cubillos, X.; Llorach, R.; Andres-Lacueva, C. Cocoa Polyphenols and Inflammatory Markers of Cardiovascular Disease. Nutrients 2014, 6, 844–880. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.; Sun, J.H.; Chen, P.; Lakshman, S.; Molokin, A.; Harnly, J.M.; Vinyard, B.T.; Urban, J.F.; Davis, C.D.; Solano-Aguilar, G. Flavanol-Enriched Cocoa Powder Alters the Intestinal Microbiota, Tissue and Fluid Metabolite Profiles, and Intestinal Gene Expression in Pigs. J. Nutr. 2016, 146, 673–680. [Google Scholar] [CrossRef] [PubMed]
- Massot-Cladera, M.; Perez-Berezo, T.; Franch, A.; Castell, M.; Perez-Cano, F.J. Cocoa modulatory effect on rat faecal microbiota and colonic crosstalk. Arch. Biochem. Biophys. 2012, 527, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Seo, K.H.; Kim, D.H.; Jeong, D.; Yokoyama, W.; Kim, H. Chardonnay grape seed flour supplemented diets alter intestinal microbiota in diet-induced obese mice. J. Food Biochem. 2017, 41, 9. [Google Scholar] [CrossRef]
- Chacar, S.; Itani, T.; Hajal, J.; Saliba, Y.; Louka, N.; Faivre, J.F.; Maroun, R.; Fares, N. The Impact of Long-Term Intake of Phenolic Compounds-Rich Grape Pomace on Rat Gut Microbiota. J. Food Sci. 2018, 83, 246–251. [Google Scholar] [CrossRef] [PubMed]
- Nash, V.; Ranadheera, C.S.; Georgousopoulou, E.N.; Mellor, D.D.; Panagiotakos, D.B.; McKune, A.J.; Kellett, J.; Naumovski, N. The effects of grape and red wine polyphenols on gut microbiota—A systematic review. Food Res. Int. 2018, 113, 277–287. [Google Scholar] [CrossRef] [PubMed]
- Cueva, C.; Gil-Sanchez, I.; Ayuda-Duran, B.; Gonzalez-Manzano, S.; Gonzalez-Paramas, A.M.; Santos-Buelga, C.; Bartolome, B.; Moreno-Arribas, M.V. An Integrated View of the Effects of Wine Polyphenols and Their Relevant Metabolites on Gut and Host Health. Molecules 2017, 22, 15. [Google Scholar] [CrossRef]
- Yuan, X.J.; Long, Y.; Ji, Z.H.; Gao, J.; Fu, T.; Yan, M.; Zhang, L.; Su, H.X.; Zhang, W.L.; Wen, X.H.; et al. Green Tea Liquid Consumption Alters the Human Intestinal and Oral Microbiome. Mol. Nutr. Food Res. 2018, 62, 15. [Google Scholar] [CrossRef]
- Cheng, M.; Zhang, X.; Miao, Y.J.; Cao, J.X.; Wu, Z.F.; Weng, P.F. The modulatory effect of (−)-epigallocatechin 3-O-(3-O-methyl) gallate (EGCG3″Me) on intestinal microbiota of high fat diet-induced obesity mice model. Food Res. Int. 2017, 92, 9–16. [Google Scholar] [CrossRef]
- Pavlidou, E.; Giaginis, C.; Fasoulas, A.; Petridis, D. Clinical Evaluation of the Effect of Blueberries Consumption on Chronic Diseases, Illness Prevention and Health Promotion. Nat. Prod. J. 2018, 8, 45–53. [Google Scholar] [CrossRef]
- Prieto, I.; Hidalgo, M.; Segarra, A.B.; Martinez-Rodriguez, A.M.; Cobo, A.; Ramirez, M.; Abriouel, H.; Galvez, A.; Martinez-Canamero, M. Influence of a diet enriched with virgin olive oil or butter on mouse gut microbiota and its correlation to physiological and biochemical parameters related to metabolic syndrome. PLoS ONE 2018, 13, 20. [Google Scholar] [CrossRef] [PubMed]
- Tomas-Barberan, F.A.; Selma, M.V.; Espin, J.C. Interactions of gut microbiota with dietary polyphenols and consequences to human health. Curr. Opin. Clin. Nutr. Metab. Care 2016, 19, 471–476. [Google Scholar] [CrossRef] [PubMed]
- Stevens, J.F.; Maier, C.S. The chemistry of gut microbial metabolism of polyphenols. Phytochem. Rev. 2016, 15, 425–444. [Google Scholar] [CrossRef] [PubMed]
- Janeiro, M.H.; Ramirez, M.J.; Milagro, F.I.; Martinez, J.A.; Solas, M. Implication of Trimethylamine N-Oxide (TMAO) in Disease: Potential Biomarker or New Therapeutic Target. Nutrients 2018, 10, 1398. [Google Scholar] [CrossRef] [PubMed]
- Velasquez, M.T.; Ramezani, A.; Manal, A.; Raj, D.S. Trimethylamine N-Oxide: The Good, the Bad and the Unknown. Toxins 2016, 8, 11. [Google Scholar] [CrossRef] [PubMed]
- Li, T.J.; Chen, Y.L.; Gua, C.J.; Li, X.D. Elevated Circulating Trimethylamine N-Oxide Levels Contribute to Endothelial Dysfunction in Aged Rats through Vascular Inflammation and Oxidative Stress. Front. Physiol. 2017, 8, 350. [Google Scholar] [CrossRef] [PubMed]
- Lyu, M.; Wang, Y.F.; Fan, G.W.; Wang, X.Y.; Xu, S.Y.; Zhu, Y. Balancing Herbal Medicine and Functional Food for Prevention and Treatment of Cardiometabolic Diseases through Modulating Gut Microbiota. Front. Microbiol. 2017, 8, 21. [Google Scholar] [CrossRef]
- Battson, M.L.; Lee, D.M.; Jarrell, D.K.; Hou, S.; Ecton, K.E.; Weir, T.L.; Gentile, C.L. Suppression of gut dysbiosis reverses Western diet-induced vascular dysfunction. Am. J. Physiol. Endocrinol. Metab. 2018, 314, E468–E477. [Google Scholar] [CrossRef]
- Chen, M.L.; Yi, L.; Zhang, Y.; Zhou, X.; Ran, L.; Yang, J.N.; Zhu, J.D.; Zhang, Q.Y.; Mi, M.T. Resveratrol Attenuates Trimethylamine-N-Oxide (TMAO)-Induced Atherosclerosis by Regulating TMAO Synthesis and Bile Acid Metabolism via Remodeling of the Gut Microbiota. mBio 2016, 7, 14. [Google Scholar] [CrossRef]
- Bresciani, L.; Dall’Asta, M.; Favari, C.; Calani, L.; Del Rio, D.; Brighenti, F. An in vitro exploratory study of dietary strategies based on polyphenol-rich beverages, fruit juices and oils to control trimethylamine production in the colon. Food Funct. 2018, 9, 6470–6483. [Google Scholar] [CrossRef] [PubMed]
- Menni, C.; Lin, C.H.; Cecelja, M.; Mangino, M.; Matey-Hernandez, M.L.; Keehn, L.; Mohney, R.P.; Steves, C.J.; Spector, T.D.; Kuo, C.F.; et al. Gut microbial diversity is associated with lower arterial stiffness in women. Eur. Heart J. 2018, 39, 2390–2397. [Google Scholar] [CrossRef] [PubMed]
- Laurent, S.; Bruno, R.M. Gut microbiome composition, a third player in the inflammation–arterial stiffness relationship. Eur. Heart J. 2018, 39, 2398–2400. [Google Scholar] [CrossRef] [PubMed]
- Cross, T.W.L.; Zidon, T.M.; Welly, R.J.; Park, Y.M.; Britton, S.L.; Koch, L.G.; Rottinghaus, G.E.; de Godoy, M.R.C.; Padilla, J.; Swanson, K.S.; et al. Soy Improves Cardiometabolic Health and Cecal Microbiota in Female Low-Fit Rats. Sci. Rep. 2017, 7, 15. [Google Scholar] [CrossRef] [PubMed]
- Sies, H.; Hollman, P.C.H.; Grune, T.; Stahl, W.; Biesalski, H.K.; Williamson, G. Protection by Flavanol-Rich Foods Against Vascular Dysfunction and Oxidative Damage: 27th Hohenheim Consensus Conference. Adv. Nutr. 2012, 3, 217–221. [Google Scholar] [CrossRef] [PubMed]
- Feliciano, R.P.; Istas, G.; Heiss, C.; Rodriguez-Mateos, A. Plasma and Urinary Phenolic Profiles after Acute and Repetitive Intake of Wild Blueberry. Molecules 2016, 21, 15. [Google Scholar] [CrossRef] [PubMed]
- Mansuri, M.L.; Parihar, P.; Solanki, I.; Parihar, M.S. Flavonoids in modulation of cell survival signalling pathways. Genes Nutr. 2014, 9. [Google Scholar] [CrossRef]
- Wang, J.N.; Guo, Z.H.; Fu, Y.X.; Wu, Z.Y.; Huang, C.; Zheng, C.L.; Shar, P.A.; Wang, Z.Z.; Xiao, W.; Wang, Y.H. Weak-binding molecules are not drugs?-toward a systematic strategy for finding effective weak-binding drugs. Brief. Bioinform. 2017, 18, 321–332. [Google Scholar] [CrossRef]
- Ottaviani, J.I.; Fong, R.; Kimball, J.; Ensunsa, J.L.; Britten, A.; Lucarelli, D.; Luben, R.; Grace, P.B.; Mawson, D.H.; Tym, A.; et al. Evaluation at scale of microbiome-derived metabolites as biomarker of flavan-3-ol intake in epidemiological studies. Sci. Rep. 2018, 8, 11. [Google Scholar] [CrossRef]
- Jennings, A.; Welch, A.A.; Fairweather-Tait, S.J.; Kay, C.; Minihane, A.M.; Chowienczyk, P.; Jiang, B.Y.; Cecelja, M.; Spector, T.; Macgregor, A.; et al. Higher anthocyanin intake is associated with lower arterial stiffness and central blood pressure in women. Am. J. Clin. Nutr. 2012, 96, 781–788. [Google Scholar] [CrossRef]
- Vlachopoulos, C.V.; Alexopoulos, N.A.; Aznaouridis, K.A.; Ioakeimidis, N.C.; Dima, I.A.; Dagre, A.; Vasiliadou, C.; Stefanadi, E.C.; Stefanadis, C.I. Relation of habitual cocoa consumption to aortic stiffness and wave reflections, and to central hemodynamics in healthy individuals. Am. J. Cardiol. 2007, 99, 1473–1475. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, S.; Dixit, M. Role of Polyphenols and Other Phytochemicals on Molecular Signaling. Oxid. Med. Cell. Longev. 2015, 2015, 504253. [Google Scholar] [CrossRef] [PubMed]
- Vlachopoulos, C.; Alexopoulos, N.; Stefanadis, C. Effects of nutrition on arterial rigidity and reflected waves. Sang. Thromb. Vaiss. 2007, 19, 479–486. [Google Scholar]
- Arranz, S.; Valderas-Martinez, P.; Chiva-Blanch, G.; Casas, R.; Urpi-Sarda, M.; Lamuela-Raventos, R.M.; Estruch, R. Cardioprotective effects of cocoa: Clinical evidence from randomized clinical intervention trials in humans. Mol. Nutr. Food. Res. 2013, 57, 936–947. [Google Scholar] [CrossRef] [PubMed]
- Sansone, R.; Ottaviani, J.I.; Rodriguez-Mateos, A.; Heinen, Y.; Noske, D.; Spencer, J.P.; Crozier, A.; Merx, M.W.; Kelm, M.; Schroeter, H.; et al. Methylxanthines enhance the effects of cocoa flavanols on cardiovascular function: Randomized, double-masked controlled studies. Am. J. Clin. Nutr. 2017, 105, 352–360. [Google Scholar] [CrossRef] [PubMed]
- Macready, A.L.; George, T.W.; Chong, M.F.; Alimbetov, D.S.; Jin, Y.; Vidal, A.; Spencer, J.P.; Kennedy, O.B.; Tuohy, K.M.; Minihane, A.M.; et al. Flavonoid-rich fruit and vegetables improve microvascular reactivity and inflammatory status in men at risk of cardiovascular disease—FLAVURS: A randomized controlled trial. Am. J. Clin. Nutr. 2014, 99, 479–489. [Google Scholar] [CrossRef] [PubMed]
- Grassi, D.; Draijer, R.; Desideri, G.; Mulder, T.; Ferri, C. Black Tea Lowers Blood Pressure and Wave Reflections in Fasted and Postprandial Conditions in Hypertensive Patients: A Randomised Study. Nutrients 2015, 7, 1037–1051. [Google Scholar] [CrossRef] [PubMed]
- Ray, S.; Miglio, C.; Eden, T.; Del Rio, D. Assessment of vascular and endothelial dysfunction in nutritional studies. Nutr. Metab. Carbiovasc. Dis. 2014, 24, 940–946. [Google Scholar] [CrossRef]
- Vendrame, S.; Del Bo, C.; Ciappellano, S.; Riso, P.; Klimis-Zacas, D. Berry Fruit Consumption and Metabolic Syndrome. Antioxidants 2016, 5, 34. [Google Scholar] [CrossRef]
- Rodriguez-Mateos, A.; Heiss, C.; Borges, G.; Crozier, A. Berry (poly)phenols and cardiovascular health. J. Agric. Food Chem. 2014, 62, 3842–3851. [Google Scholar] [CrossRef]
- Blumberg, J.B.; Vita, J.A.; Chen, C.Y.O. Concord Grape Juice Polyphenols and Cardiovascular Risk Factors: Dose-Response Relationships. Nutrients 2015, 7, 10032–10052. [Google Scholar] [CrossRef] [PubMed]
- Pase, M.P.; Grima, N.A.; Sarris, J. The effects of dietary and nutrient interventions on arterial stiffness: A systematic review. Am. J. Clin. Nutr. 2011, 93, 446–454. [Google Scholar] [CrossRef] [PubMed]
- Nestel, P.; Fujii, A.; Zhang, L. An isoflavone metabolite reduces arterial stiffness and blood pressure in overweight men and postmenopausal women. Atherosclerosis 2007, 192, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Vlachopoulos, C.; Aznaouridis, K.; Alexopoulos, N.; Economou, E.; Andreadou, I.; Stefanadis, C. Effect of dark chocolate on arterial function in healthy individuals. Am. J. Hypertens. 2005, 18, 785–791. [Google Scholar] [CrossRef] [PubMed]
- Grassi, D.; Desideri, G.; Necozione, S.; di Giosia, P.; Barnabei, R.; Allegaert, L.; Bernaert, H.; Ferri, C. Cocoa consumption dose-dependently improves flow-mediated dilation and arterial stiffness decreasing blood pressure in healthy individuals. J. Hypertens. 2015, 33, 294–303. [Google Scholar] [CrossRef]
- Grassi, D.; Desideri, G.; Necozione, S.; Ruggieri, F.; Blumberg, J.B.; Stornello, M.; Ferri, C. Protective effects of flavanol-rich dark chocolate on endothelial function and wave reflection during acute hyperglycemia. Hypertension 2012, 60, 827–832. [Google Scholar] [CrossRef]
- Grassi, D.; Desideri, G.; Necozione, S.; Lippi, C.; Casale, R.; Properzi, G.; Blumberg, J.B.; Ferri, C. Blood pressure is reduced and insulin sensitivity increased in glucose-intolerant, hypertensive subjects after 15 days of consuming high-polyphenol dark chocolate. J. Nutr. 2008, 138, 1671–1676. [Google Scholar] [CrossRef]
- Grassi, D.; Socci, V.; Tempesta, D.; Ferri, C.; De Gennaro, L.; Desideri, G.; Ferrara, M. Flavanol-rich chocolate acutely improves arterial function and working memory performance counteracting the effects of sleep deprivation in healthy individuals. J. Hypertens. 2016, 34, 1298–1308. [Google Scholar] [CrossRef]
- Heiss, C.; Sansone, R.; Karimi, H.; Krabbe, M.; Schuler, D.; Rodriguez-Mateos, A.; Kraemer, T.; Cortese-Krott, M.M.; Kuhnle, G.G.; Spencer, J.P.; et al. Impact of cocoa flavanol intake on age-dependent vascular stiffness in healthy men: A randomized, controlled, double-masked trial. Age 2015, 37, 9794. [Google Scholar] [CrossRef]
- Basu, A.; Betts, N.M.; Leyva, M.J.; Fu, D.; Aston, C.E.; Lyons, T.J. Acute Cocoa Supplementation Increases Postprandial HDL Cholesterol and Insulin in Obese Adults with Type 2 Diabetes after Consumption of a High-Fat Breakfast. J. Nutr. 2015, 145, 2325–2332. [Google Scholar] [CrossRef]
- West, S.G.; McIntyre, M.D.; Piotrowski, M.J.; Poupin, N.; Miller, D.L.; Preston, A.G.; Wagner, P.; Groves, L.F.; Skulas-Ray, A.C. Effects of dark chocolate and cocoa consumption on endothelial function and arterial stiffness in overweight adults. Br. J. Nutr. 2014, 111, 653–661. [Google Scholar] [CrossRef] [PubMed]
- Dower, J.I.; Geleijnse, J.M.; Gijsbers, L.; Zock, P.L.; Kromhout, D.; Hollman, P.C. Effects of the pure flavonoids epicatechin and quercetin on vascular function and cardiometabolic health: A randomized, double-blind, placebo-controlled, crossover trial. Am. J. Clin. Nutr. 2015, 101, 914–921. [Google Scholar] [CrossRef] [PubMed]
- Dower, J.I.; Geleijnse, J.M.; Kroon, P.A.; Philo, M.; Mensink, M.; Kromhout, D.; Hollman, P.C. Does epicatechin contribute to the acute vascular function effects of dark chocolate? A randomized, crossover study. Mol. Nutr. Food Res. 2016, 60, 2379–2386. [Google Scholar] [CrossRef] [PubMed]
- Ward, N.C.; Hodgson, J.M.; Woodman, R.J.; Zimmermann, D.; Poquet, L.; Leveques, A.; Actis-Goretta, L.; Puddey, I.B.; Croft, K.D. Acute effects of chlorogenic acids on endothelial function and blood pressure in healthy men and women. Food Funct. 2016, 7, 2197–2203. [Google Scholar] [CrossRef] [PubMed]
- Jokura, H.; Watanabe, I.; Umeda, M.; Hase, T.; Shimotoyodome, A. Coffee polyphenol consumption improves postprandial hyperglycemia associated with impaired vascular endothelial function in healthy male adults. Nutr. Res. 2015, 35, 873–881. [Google Scholar] [CrossRef] [PubMed]
- Grassi, D.; Mulder, T.P.; Draijer, R.; Desideri, G.; Molhuizen, H.O.; Ferri, C. Black tea consumption dose-dependently improves flow-mediated dilation in healthy males. J. Hypertens. 2009, 27, 774–781. [Google Scholar] [CrossRef] [PubMed]
- Ryu, O.H.; Lee, J.; Lee, K.W.; Kim, H.Y.; Seo, J.A.; Kim, S.G.; Kim, N.H.; Baik, S.H.; Choi, D.S.; Choi, K.M. Effects of green tea consumption on inflammation, insulin resistance and pulse wave velocity in type 2 diabetes patients. Diabetes Res. Clin. Pract. 2006, 71, 356–358. [Google Scholar] [CrossRef] [PubMed]
- Miller, R.J.; Jackson, K.G.; Dadd, T.; Mayes, A.E.; Brown, A.L.; Minihane, A.M. The impact of the catechol-O-methyltransferase genotype on the acute responsiveness of vascular reactivity to a green tea extract. Br. J. Nutr. 2011, 105, 1138–1144. [Google Scholar] [CrossRef] [PubMed]
- Bondonno, N.P.; Bondonno, C.P.; Blekkenhorst, L.C.; Considine, M.J.; Maghzal, G.; Stocker, R.; Woodman, R.J.; Ward, N.C.; Hodgson, J.M.; Croft, K.D. Flavonoid-Rich Apple Improves Endothelial Function in Individuals at Risk for Cardiovascular Disease: A Randomised Controlled Clinical Trial. Mol. Nutr. Food Res. 2018, 62. [Google Scholar] [CrossRef]
- Cerletti, C.; Gianfagna, F.; Tamburrelli, C.; De Curtis, A.; D’Imperio, M.; Coletta, W.; Giordano, L.; Lorenzet, R.; Rapisarda, P.; Reforgiato Recupero, G.; et al. Orange juice intake during a fatty meal consumption reduces the postprandial low-grade inflammatory response in healthy subjects. Thromb. Res. 2015, 135, 255–259. [Google Scholar] [CrossRef]
- Habauzit, V.; Verny, M.A.; Milenkovic, D.; Barber-Chamoux, N.; Mazur, A.; Dubray, C.; Morand, C. Flavanones protect from arterial stiffness in postmenopausal women consuming grapefruit juice for 6 mo: A randomized, controlled, crossover trial. Am. J. Clin. Nutr. 2015, 102, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Schaer, M.Y.; Curtis, P.J.; Hazim, S.; Ostertag, L.M.; Kay, C.D.; Potter, J.F.; Cassidy, A. Orange juice-derived flavanone and phenolic metabolites do not acutely affect cardiovascular risk biomarkers: A randomized, placebo-controlled, crossover trial in men at moderate risk of cardiovascular disease. Am. J. Clin. Nutr. 2015, 101, 931–938. [Google Scholar] [CrossRef] [PubMed]
- Mathew, A.S.; Capel-Williams, G.M.; Berry, S.E.; Hall, W.L. Acute effects of pomegranate extract on postprandial lipaemia, vascular function and blood pressure. Plant Foods Hum. Nutr. 2012, 67, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Lynn, A.; Hamadeh, H.; Leung, W.C.; Russell, J.M.; Barker, M.E. Effects of Pomegranate Juice Supplementation on Pulse Wave Velocity and Blood Pressure in Healthy Young and Middle-aged Men and Women. Plant Foods Hum. Nutr. 2012, 67, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Gerstgrasser, A.; Rochter, S.; Dressler, D.; Schon, C.; Reule, C.; Buchwald-Werner, S. In vitro Activation of eNOS by Mangifera indica (Careless (TM)) and Determination of an Effective Dosage in a Randomized, Double-Blind, Human Pilot Study on Microcirculation. Planta Med. 2016, 82, 298–304. [Google Scholar] [CrossRef] [PubMed]
- Ruel, G.; Lapointe, A.; Pomerleau, S.; Couture, P.; Lemieux, S.; Lamarche, B.; Couillard, C. Evidence that cranberry juice may improve augmentation index in overweight men. Nutr. Res. 2013, 33, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Dohadwala, M.M.; Holbrook, M.; Hamburg, N.M.; Shenouda, S.M.; Chung, W.B.; Titas, M.; Kluge, M.A.; Wang, N.; Palmisano, J.; Milbury, P.E.; et al. Effects of cranberry juice consumption on vascular function in patients with coronary artery disease. Am. J. Clin. Nutr. 2011, 93, 934–940. [Google Scholar] [CrossRef]
- Del Bo, C.; Porrini, M.; Fracassetti, D.; Campolo, J.; Klimis-Zacas, D.; Riso, P. A single serving of blueberry (V. corymbosum) modulates peripheral arterial dysfunction induced by acute cigarette smoking in young volunteers: A randomized-controlled trial. Food Funct. 2014, 5, 3107–3116. [Google Scholar] [CrossRef]
- Del Bo, C.; Porrini, M.; Campolo, J.; Parolini, M.; Lanti, C.; Klimis-Zacas, D.; Riso, P. A single blueberry (Vaccinium corymbosum) portion does not affect markers of antioxidant defence and oxidative stress in healthy volunteers following cigarette smoking. Mutagenesis 2016, 31, 215–224. [Google Scholar] [CrossRef]
- Del Bo, C.; Deon, V.; Campolo, J.; Lanti, C.; Parolini, M.; Porrini, M.; Klimis-Zacas, D.; Riso, P. A serving of blueberry (V. corymbosum) acutely improves peripheral arterial dysfunction in young smokers and non-smokers: Two randomized, controlled, crossover pilot studies. Food Funct. 2017, 8, 4108–4117. [Google Scholar] [CrossRef]
- Johnson, S.A.; Figueroa, A.; Navaei, N.; Wong, A.; Kalfon, R.; Ormsbee, L.T.; Feresin, R.G.; Elam, M.L.; Hooshmand, S.; Payton, M.E.; et al. Daily Blueberry Consumption Improves Blood Pressure and Arterial Stiffness in Postmenopausal Women with Pre- and Stage 1-Hypertension: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. J. Acad. Nutr. Diet. 2015, 115, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Richter, C.K.; Skulas-Ray, A.C.; Gaugler, T.L.; Lambert, J.D.; Proctor, D.N.; Kris-Etherton, P.M. Incorporating freeze-dried strawberry powder into a high-fat meal does not alter postprandial vascular function or blood markers of cardiovascular disease risk: A randomized controlled trial. Am. J. Clin. Nutr. 2017, 105, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Castro-Acosta, M.L.; Smith, L.; Miller, R.J.; McCarthy, D.I.; Farrimond, J.A.; Hall, W.L. Drinks containing anthocyanin-rich blackcurrant extract decrease postprandial blood glucose, insulin and incretin concentrations. J. Nutr. Biochem. 2016, 38, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Jeong, H.S.; Hong, S.J.; Lee, T.B.; Kwon, J.W.; Jeong, J.T.; Joo, H.J.; Park, J.H.; Ahn, C.M.; Yu, C.W.; Lim, D.S. Effects of Black Raspberry on Lipid Profiles and Vascular Endothelial Function in Patients with Metabolic Syndrome. Phytother. Res. 2014, 28, 1492–1498. [Google Scholar] [CrossRef] [PubMed]
- Jeong, H.S.; Kim, S.; Hong, S.J.; Choi, S.C.; Choi, J.H.; Kim, J.H.; Park, C.Y.; Cho, J.Y.; Lee, T.B.; Kwon, J.W.; et al. Black Raspberry Extract Increased Circulating Endothelial Progenitor Cells and Improved Arterial Stiffness in Patients with Metabolic Syndrome: A Randomized Controlled Trial. J. Med. Food 2016, 19, 346–352. [Google Scholar] [CrossRef] [PubMed]
- Siasos, G.; Tousoulis, D.; Kokkou, E.; Oikonomou, E.; Kollia, M.E.; Verveniotis, A.; Gouliopoulos, N.; Zisimos, K.; Plastiras, A.; Maniatis, K.; et al. Favorable Effects of Concord Grape Juice on Endothelial Function and Arterial Stiffness in Healthy Smokers. Am. J. Hypertens. 2014, 27, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Park, E.; Edirisinghe, I.; Choy, Y.Y.; Waterhouse, A.; Burton-Freeman, B. Effects of grape seed extract beverage on blood pressure and metabolic indices in individuals with pre-hypertension: A randomised, double-blinded, two-arm, parallel, placebo-controlled trial. Br. J. Nutr. 2016, 115, 226–238. [Google Scholar] [CrossRef]
- Draijer, R.; de Graaf, Y.; Slettenaar, M.; de Groot, E.; Wright, C.I. Consumption of a polyphenol-rich grape-wine extract lowers ambulatory blood pressure in mildly hypertensive subjects. Nutrients 2015, 7, 3138–3153. [Google Scholar] [CrossRef]
- Naissides, M.; Pal, S.; Mamo, J.C.; James, A.P.; Dhaliwal, S. The effect of chronic consumption of red wine polyphenols on vascular function in postmenopausal women. Eur. J. Clin. Nutr. 2006, 60, 740–745. [Google Scholar] [CrossRef]
- Imamura, H.; Yamaguchi, T.; Nagayama, D.; Saiki, A.; Shirai, K.; Tatsuno, I. Resveratrol Ameliorates Arterial Stiffness Assessed by Cardio-Ankle Vascular Index in Patients with Type 2 Diabetes Mellitus. Int. Heart J. 2017, 58, 577–583. [Google Scholar] [CrossRef]
- Wong, R.H.X.; Howe, P.R.C.; Buckley, J.D.; Coates, A.M.; Kunz, I.; Berry, N.M. Acute resveratrol supplementation improves flow-mediated dilatation in overweight/obese individuals with mildly elevated blood pressure. Nutr. Metab. Carbiovasc. Dis. 2011, 21, 851–856. [Google Scholar] [CrossRef] [PubMed]
- Wong, R.H.X.; Berry, N.M.; Coates, A.M.; Buckley, J.D.; Bryan, J.; Kunz, I.; Howe, P.R.C. Chronic resveratrol consumption improves brachial flow-mediated dilatation in healthy obese adults. J. Hypertens. 2013, 31, 1819–1827. [Google Scholar] [CrossRef] [PubMed]
- Teede, H.J.; McGrath, B.P.; DeSilva, L.; Cehun, M.; Fassoulakis, A.; Nestel, P.J. Isoflavones reduce arterial stiffness: A placebo-controlled study in men and postmenopausal women. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 1066–1071. [Google Scholar] [CrossRef] [PubMed]
- Hoshida, S.; Miki, T.; Nakagawa, T.; Shinoda, Y.; Inoshiro, N.; Terada, K.; Adachi, T. Different effects of isoflavones on vascular function in premenopausal and postmenopausal smokers and nonsmokers: NYMPH study. Heart Vessels 2011, 26, 590–595. [Google Scholar] [CrossRef] [PubMed]
- Richter, C.K.; Skulas-Ray, A.C.; Fleming, J.A.; Link, C.J.; Mukherjea, R.; Krul, E.S.; Kris-Etherton, P.M. Effects of isoflavone-containing soya protein on ex vivo cholesterol efflux, vascular function and blood markers of CVD risk in adults with moderately elevated blood pressure: A dose-response randomised controlled trial. Br. J. Nutr. 2017, 117, 1403–1413. [Google Scholar] [CrossRef] [PubMed]
- Curtis, P.J.; Potter, J.; Kroon, P.A.; Wilson, P.; Dhatariya, K.; Sampson, M.; Cassidy, A. Vascular function and atherosclerosis progression after 1 y of flavonoid intake in statin-treated postmenopausal women with type 2 diabetes: A double-blind randomized controlled trial. Am. J. Clin. Nutr. 2013, 97, 936–942. [Google Scholar] [CrossRef] [PubMed]
- Clerici, C.; Setchell, K.D.; Battezzati, P.M.; Pirro, M.; Giuliano, V.; Asciutti, S.; Castellani, D.; Nardi, E.; Sabatino, G.; Orlandi, S.; et al. Pasta naturally enriched with isoflavone aglycons from soy germ reduces serum lipids and improves markers of cardiovascular risk. J. Nutr. 2007, 137, 2270–2278. [Google Scholar] [CrossRef] [PubMed]
- Clerici, C.; Nardi, E.; Battezzati, P.M.; Asciutti, S.; Castellani, D.; Corazzi, N.; Giuliano, V.; Gizzi, S.; Perriello, G.; Di Matteo, G.; et al. Novel soy germ pasta improves endothelial function, blood pressure, and oxidative stress in patients with type 2 diabetes. Diabetes Care 2011, 34, 1946–1948. [Google Scholar] [CrossRef]
- Reverri, E.J.; LaSalle, C.D.; Franke, A.A.; Steinberg, F.M. Soy provides modest benefits on endothelial function without affecting inflammatory biomarkers in adults at cardiometabolic risk. Mol. Nutr. Food Res. 2015, 59, 323–333. [Google Scholar] [CrossRef]
- Lockyer, S.; Corona, G.; Yaqoob, P.; Spencer, J.P.; Rowland, I. Secoiridoids delivered as olive leaf extract induce acute improvements in human vascular function and reduction of an inflammatory cytokine: A randomised, double-blind, placebo-controlled, cross-over trial. Br. J. Nutr. 2015, 114, 75–83. [Google Scholar] [CrossRef]
- Verhoeven, V.; Van der Auwera, A.; Van Gaal, L.; Remmen, R.; Apers, S.; Stalpaert, M.; Wens, J.; Hermans, N. Can red yeast rice and olive extract improve lipid profile and cardiovascular risk in metabolic syndrome?: A double blind, placebo controlled randomized trial. BMC Complement. Altern. Med. 2015, 15, 8. [Google Scholar] [CrossRef] [PubMed]
- Hermans, N.; Van der Auwera, A.; Breynaert, A.; Verlaet, A.; De Bruyne, T.; Van Gaal, L.; Pieters, L.; Verhoeven, V. A red yeast rice-olive extract supplement reduces biomarkers of oxidative stress, OxLDL and Lp-PLA(2), in subjects with metabolic syndrome: A randomised, double-blind, placebo-controlled trial. Trials 2017, 18, 8. [Google Scholar] [CrossRef] [PubMed]
- Pais, P.; Rull, S.; Villar, A. Impact of a proprietary standardized olive fruit extract (Proliva (R)) on CAVI assessments in subjects with arterial stiffness risk. Planta Med. 2016, 82, 2. [Google Scholar] [CrossRef]
- Mullan, A.; Delles, C.; Ferrell, W.; Mullen, W.; Edwards, C.A.; McColl, J.H.; Roberts, S.A.; Lean, M.E.; Sattar, N. Effects of a beverage rich in (poly)phenols on established and novel risk markers for vascular disease in medically uncomplicated overweight or obese subjects: A four week randomized placebo-controlled trial. Atherosclerosis 2016, 246, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Chuengsamarn, S.; Rattanamongkolgul, S.; Phonrat, B.; Tungtrongchitr, R.; Jirawatnotai, S. Reduction of atherogenic risk in patients with type 2 diabetes by curcuminoid extract: A randomized controlled trial. J. Nutr. Biochem. 2014, 25, 144–150. [Google Scholar] [CrossRef] [PubMed]
- Akazawa, N.; Choi, Y.; Miyaki, A.; Tanabe, Y.; Sugawara, J.; Ajisaka, R.; Maeda, S. Curcumin ingestion and exercise training improve vascular endothelial function in postmenopausal women. Nutr. Res. 2012, 32, 795–799. [Google Scholar] [CrossRef] [PubMed]
- Katz, D.L.; Davidhi, A.; Ma, Y.Y.; Kavak, Y.; Bifulco, L.; Njike, V.Y. Effects of Walnuts on Endothelial Function in Overweight Adults with Visceral Obesity: A Randomized, Controlled, Crossover Trial. J. Am. Coll. Nutr. 2012, 31, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Bruell, V.; Burak, C.; Stoffel-Wagner, B.; Wolffram, S.; Nickenig, G.; Mueller, C.; Langguth, P.; Alteheld, B.; Fimmers, R.; Naaf, S.; et al. Effects of a quercetin-rich onion skin extract on 24 h ambulatory blood pressure and endothelial function in overweight-to-obese patients with (pre-) hypertension: A randomised double-blinded placebo-controlled cross-over trial. Br. J. Nutr. 2015, 114, 1263–1277. [Google Scholar] [CrossRef]
- Yui, S.; Fujiwara, S.; Harada, K.; Motoike-Hamura, M.; Sakai, M.; Matsubara, S.; Miyazaki, K. Beneficial Effects of Lemon Balm Leaf Extract on In Vitro Glycation of Proteins, Arterial Stiffness, and Skin Elasticity in Healthy Adults. J. Nutr. Sci. Vitaminol. 2017, 63, 59–68. [Google Scholar] [CrossRef]
- Siti, H.N.; Kamisah, Y.; Kamsiah, J. The role of oxidative stress, antioxidants and vascular inflammation in cardiovascular disease (a review). Vasc. Pharmacol. 2015, 71, 40–56. [Google Scholar] [CrossRef]
- Katz, D.L.; Doughty, K.; Ali, A. Cocoa and Chocolate in Human Health and Disease. Antioxid. Redox Signal. 2011, 15, 2779–2811. [Google Scholar] [CrossRef] [PubMed]
- Croft, K.D. Dietary polyphenols: Antioxidants or not? Arch. Biochem. Biophys. 2016, 595, 120–124. [Google Scholar] [CrossRef] [PubMed]
- Latham, L.S.; Hensen, Z.K.; Minor, D.S. Chocolate—Guilty pleasure or healthy supplement? J. Clin. Hypertens. 2014, 16, 101–106. [Google Scholar] [CrossRef]
- Ferri, C.; Desideri, G.; Ferri, L.; Proietti, I.; Di Agostino, S.; Martella, L.; Mai, F.; Di Giosia, P.; Grassi, D. Cocoa, Blood Pressure, and Cardiovascular Health. J. Agric. Food Chem. 2015, 63, 9901–9909. [Google Scholar] [CrossRef] [PubMed]
- Ibero-Baraibar, I.; Suarez, M.; Arola-Arnal, A.; Zulet, M.A.; Martinez, J.A. Cocoa extract intake for 4 weeks reduces postprandial systolic blood pressure response of obese subjects, even after following an energy-restricted diet. Food Nutr. Res. 2016, 60. [Google Scholar] [CrossRef] [PubMed]
- Hugel, H.M.; Jackson, N.; May, B.; Zhang, A.L.; Xue, C.C. Polyphenol protection and treatment of hypertension. Phytomedicine 2016, 23, 220–231. [Google Scholar] [CrossRef] [PubMed]
- Persson, I.A.L.; Josefsson, M.; Persson, K.; Andersson, R.G.G. Tea flavanols inhibit angiotensin-converting enzyme activity and increase nitric oxide production in human endothelial cells. J. Pharm. Pharmacol. 2006, 58, 1139–1144. [Google Scholar] [CrossRef]
- Li, X.; Dai, Y.N.; Yan, S.J.; Shi, Y.L.; Li, J.X.; Liu, J.L.; Cha, L.; Mu, J.J. Resveratrol lowers blood pressure in spontaneously hypertensive rats via calcium-dependent endothelial NO production. Clin. Exp. Hypertens. 2016, 38, 287–293. [Google Scholar] [CrossRef]
- Taguchi, K.; Hida, M.; Hasegawa, M.; Matsumoto, T.; Kobayashi, T. Dietary polyphenol morin rescues endothelial dysfunction in a diabetic mouse model by activating the Akt/eNOS pathway. Mol. Nutr. Food Res. 2016, 60, 580–588. [Google Scholar] [CrossRef]
- Kim, H.S.; Quon, M.J.; Kim, J.A. New insights into the mechanisms of polyphenols beyond antioxidant properties; lessons from the green tea polyphenol, epigallocatechin 3-gallate. Redox Biol. 2014, 2, 187–195. [Google Scholar] [CrossRef]
- Smoliga, J.M.; Baur, J.A.; Hausenblas, H.A. Resveratrol and health—A comprehensive review of human clinical trials. Mol. Nutr. Food Res. 2011, 55, 1129–1141. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.I.; Acosta, C.; Anderson, C.M.; Anderson, H.D. Peripheral and Cerebral Resistance Arteries in the Spontaneously Hypertensive Heart Failure Rat: Effects of Stilbenoid Polyphenols. Molecules 2017, 22, 380. [Google Scholar] [CrossRef] [PubMed]
- Behbahani, J.; Thandapilly, S.J.; Louis, X.L.; Huang, Y.S.; Shao, Z.J.; Kopilas, M.A.; Wojciechowski, P.; Netticadan, T.; Anderson, H.D. Resveratrol and Small Artery Compliance and Remodeling in the Spontaneously Hypertensive Rat. Am. J. Hypertens. 2010, 23, 1273–1278. [Google Scholar] [CrossRef] [PubMed]
- Thandapilly, S.J.; LeMaistre, J.L.; Louis, X.L.; Anderson, C.M.; Netticadan, T.; Anderson, H.D. Vascular and Cardiac Effects of Grape Powder in the Spontaneously Hypertensive Rat. Am. J. Hypertens. 2012, 25, 1070–1076. [Google Scholar] [CrossRef] [PubMed]
- Pons, Z.; Margalef, M.; Bravo, F.I.; Arola-Arnal, A.; Muguerza, B. Grape seed flavanols decrease blood pressure via Sirt-1 and confer a vasoprotective pattern in rats. J. Funct. Foods 2016, 24, 164–172. [Google Scholar] [CrossRef]
- Fleenor, B.S.; Sindler, A.L.; Marvi, N.K.; Howell, K.L.; Zigler, M.L.; Yoshizawa, M.; Seals, D.R. Curcumin ameliorates arterial dysfunction and oxidative stress with aging. Exp. Gerontol. 2013, 48, 269–276. [Google Scholar] [CrossRef]
- Spigoni, V.; Mena, P.; Cito, M.; Fantuzzi, F.; Bonadonna, R.C.; Brighenti, F.; Dei Cas, A.; Del Rio, D. Effects on Nitric Oxide Production of Urolithins, Gut-Derived Ellagitannin Metabolites, in Human Aortic Endothelial Cells. Molecules 2016, 21, 1009. [Google Scholar] [CrossRef]
- Rowlands, D.J.; Chapple, S.; Siow, R.C.; Mann, G.E. Equol-stimulated mitochondrial reactive oxygen species activate endothelial nitric oxide synthase and redox signaling in endothelial cells: Roles for F-actin and GPR30. Hypertension 2011, 57, 833–840. [Google Scholar] [CrossRef]
- Cheng, C.; Wang, X.; Weakley, S.M.; Kougias, P.; Lin, P.H.; Yao, Q.; Chen, C. The soybean isoflavonoid equol blocks ritonavir-induced endothelial dysfunction in porcine pulmonary arteries and human pulmonary artery endothelial cells. J. Nutr. 2010, 140, 12–17. [Google Scholar] [CrossRef]
- Bonacasa, B.; Siow, R.C.; Mann, G.E. Impact of dietary soy isoflavones in pregnancy on fetal programming of endothelial function in offspring. Microcirculation 2011, 18, 270–285. [Google Scholar] [CrossRef]
- Egea, J.; Fabregat, I.; Frapart, Y.M.; Ghezzi, P.; Gorlach, A.; Kietzmann, T.; Kubaichuk, K.; Knaus, U.G.; Lopez, M.G.; Olaso-Gonzalez, G.; et al. European contribution to the study of ROS: A summary of the findings and prospects for the future from the COST action BM1203 (EU-ROS). Redox Biol. 2017, 13, 94–162. [Google Scholar] [CrossRef] [PubMed]
- Amiot, M.J.; Riva, C.; Vinet, A. Effects of dietary polyphenols on metabolic syndrome features in humans: A systematic review. Obes. Rev. 2016, 17, 573–586. [Google Scholar] [CrossRef] [PubMed]
- Jiang, R.J.; Hodgson, J.M.; Mas, E.; Croft, K.D.; Ward, N.C. Chlorogenic acid improves ex vivo vessel function and protects endothelial cells against HOCl-induced oxidative damage, via increased production of nitric oxide and induction of Hmox-1. J. Nutr. Biochem. 2016, 27, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Zenkov, N.K.; Chechushkov, A.V.; Kozhin, P.M.; Kandalintseva, N.V.; Martinovich, G.G.; Menshchikova, E.B. Plant phenols and autophagy. Biochemistry 2016, 81, 297–314. [Google Scholar] [CrossRef] [PubMed]
- Forman, H.J.; Davies, K.J.A.; Ursini, F. How Do Nutritional Antioxidants Really Work: Nucleophilic Tone and Para-Hormesis Versus Free Radical Scavenging in vivo. Free Radic. Biol. Med. 2014, 74, 307. [Google Scholar] [CrossRef]
- Mann, G.E.; Bonacasa, B.; Ishii, T.; Siow, R.C. Targeting the redox sensitive Nrf2-Keap1 defense pathway in cardiovascular disease: Protection afforded by dietary isoflavones. Curr. Opin. Pharmacol. 2009, 9, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Liang, X.; Shi, L.; Wang, L.; Chen, J.; Kang, C.; Zhu, J.; Mi, M. Estrogen receptor and PI3K/Akt signaling pathway involvement in S-(-)equol-induced activation of Nrf2/ARE in endothelial cells. PLoS ONE 2013, 8, e79075. [Google Scholar] [CrossRef]
- Chapple, S.; Rowlands, D.J.; Siow, R.; Mann, G. The Estrogenic Compound Equol Affords Cardiovascular Protection via Activation of Nrf2 Antioxidant Defenses. Free Radic. Biol. Med. 2012, 53, S157. [Google Scholar] [CrossRef]
- Martin, M.A.; Ramos, S. Cocoa polyphenols in oxidative stress: Potential health implications. J. Funct. Foods 2016, 27, 570–588. [Google Scholar] [CrossRef]
- Kuntz, S.; Kunz, C.; Herrmann, J.; Borsch, C.H.; Abel, G.; Frohling, B.; Dietrich, H.; Rudloff, S. Anthocyanins from fruit juices improve the antioxidant status of healthy young female volunteers without affecting anti-inflammatory parameters: Results from the randomised, double-blind, placebo-controlled, cross-over ANTHONIA (ANTHOcyanins in Nutrition Investigation Alliance) study. Br. J. Nutr. 2014, 112, 925–936. [Google Scholar] [CrossRef]
- Kivela, A.M.; Kansanen, E.; Jyrkkanen, H.K.; Nurmi, T.; Yla-Herttuala, S.; Levonen, A.L. Enterolactone induces heme oxygenase-1 expression through nuclear factor-E2-related factor 2 activation in endothelial cells. J. Nutr. 2008, 138, 1263–1268. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.X.; Xiao, L.; Yuan, Y.; Luo, X.Q.; Jiang, M.L.; Ni, J.H.; Wang, N.P. Procyanidin B2 inhibits NLRP3 inflammasome activation in human vascular endothelial cells. Biochem. Pharmacol. 2014, 92, 599–606. [Google Scholar] [CrossRef] [PubMed]
- Misawa, T.; Saitoh, T.; Kozaki, T.; Park, S.; Takahama, M.; Akira, S. Resveratrol inhibits the acetylated alpha-tubulin-mediated assembly of the NLRP3-inflammasome. Int. Immunol. 2015, 27, 425–434. [Google Scholar] [CrossRef] [PubMed]
- Shi, A.M.; Shi, H.T.; Wang, Y.; Liu, X.; Cheng, Y.; Li, H.; Zhao, H.L.; Wang, S.N.; Dong, L. Activation of Nrf2 pathway and inhibition of NLRP3 inflammasome activation contribute to the protective effect of chlorogenic acid on acute liver injury. Int. Immunopharmacol. 2018, 54, 125–130. [Google Scholar] [CrossRef] [PubMed]
- El-Bassossy, H.M.; Hassan, N.A.; Mahmoud, M.F.; Fahmy, A. Baicalein protects against hypertension associated with diabetes: Effect on vascular reactivity and stiffness. Phytomedicine 2014, 21, 1742–1745. [Google Scholar] [CrossRef] [PubMed]
- Kunnumakkara, A.B.; Bordoloi, D.; Padmavathi, G.; Monisha, J.; Roy, N.K.; Prasad, S.; Aggarwal, B.B. Curcumin, the golden nutraceutical: Multitargeting for multiple chronic diseases. Br. J. Pharmacol. 2017, 174, 1325–1348. [Google Scholar] [CrossRef] [PubMed]
- Warner, E.F.; Zhang, Q.Z.; Raheem, K.S.; O’Hagan, D.; O’Connell, M.A.; Kay, C.D. Common Phenolic Metabolites of Flavonoids, but Not Their Unmetabolized Precursors, Reduce the Secretion of Vascular Cellular Adhesion Molecules by Human Endothelial Cells(1-3). J. Nutr. 2016, 146, 465–473. [Google Scholar] [CrossRef] [PubMed]
- Han, Q.A.; Yan, C.; Wang, L.; Li, G.; Xu, Y.; Xia, X. Urolithin A attenuates ox-LDL-induced endothelial dysfunction partly by modulating microRNA-27 and ERK/PPAR-gamma pathway. Mol. Nutr. Food Res. 2016, 60, 1933–1943. [Google Scholar] [CrossRef]
- Fry, J.L.; Al Sayah, L.; Weisbrod, R.M.; Van Roy, I.; Weng, X.; Cohen, R.A.; Bachschmid, M.M.; Seta, F. Vascular Smooth Muscle Sirtuin-1 Protects Against Diet-Induced Aortic Stiffness. Hypertension 2016, 68, 775–784. [Google Scholar] [CrossRef]
- Pusparini; Dharma, R.; Suyatna, F.D.; Mansyur, M.; Hidajat, A. Effect of soy isoflavone supplementation on vascular endothelial function and oxidative stress in postmenopausal women: A community randomized controlled trial. Asia Pac. J. Clin. Nutr. 2013, 22, 357–364. [Google Scholar] [CrossRef]
- Subedi, L.; Ji, E.; Shin, D.; Jin, J.; Yeo, J.H.; Kim, S.Y. Equol, a Dietary Daidzein Gut Metabolite Attenuates Microglial Activation and Potentiates Neuroprotection In Vitro. Nutrients 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Yuan, C.; Wang, G.; Luo, J.; Ma, H.; Xu, L.; Mu, Y.; Li, Y.; Seeram, N.P.; Huang, X.; et al. Urolithins Attenuate LPS-Induced Neuroinflammation in BV2Microglia via MAPK, Akt, and NF-kappaB Signaling Pathways. J. Agric. Food Chem. 2018, 66, 571–580. [Google Scholar] [CrossRef] [PubMed]
- Bo, S.; Ciccone, G.; Castiglione, A.; Gambino, R.; De Michieli, F.; Villois, P.; Durazzo, M.; Cavallo-Perin, P.; Cassader, M. Anti-Inflammatory and Antioxidant Effects of Resveratrol in Healthy Smokers A Randomized, Double-Blind, Placebo-Controlled, Cross-Over Trial. Curr. Med. Chem. 2013, 20, 1323–1331. [Google Scholar] [CrossRef] [PubMed]
- Rangel-Huerta, O.D.; Pastor-Villaescusa, B.; Aguilera, C.M.; Gil, A. A Systematic Review of the Efficacy of Bioactive Compounds in Cardiovascular Disease: Phenolic Compounds. Nutrients 2015, 7, 5177–5216. [Google Scholar] [CrossRef] [PubMed]
- Chiva-Blanch, G.; Urpi-Sarda, M.; Llorach, R.; Rotches-Ribalta, M.; Guillen, M.; Casas, R.; Arranz, S.; Valderas-Martinez, P.; Portoles, O.; Corella, D. Differential effects of polyphenols and alcohol of red wine on the expression of adhesion molecules and inflammatory cytokines related to atherosclerosis: A randomized clinical trial. Am. J. Clin. Nutr. 2012, 95, 1506. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.Y.; Wang, P.Y.; He, K.; Qin, L.Q. Effect of soy isoflavones on circulating C-reactive protein in postmenopausal women: Meta-analysis of randomized controlled trials. Menopause J. N. Am. Menopause Soc. 2011, 18, 1256–1262. [Google Scholar] [CrossRef] [PubMed]
- Nicastro, H.L.; Mondul, A.M.; Rohrmann, S.; Platz, E.A. Associations between urinary soy isoflavonoids and two inflammatory markers in adults in the United States in 2005-2008. Cancer Causes Control 2013, 24, 1185–1196. [Google Scholar] [CrossRef]
- Ren, G.Y.; Chen, C.Y.; Chen, G.C.; Chen, W.G.; Pan, A.; Pan, C.W.; Zhang, Y.H.; Qin, L.Q.; Chen, L.H. Effect of Flaxseed Intervention on Inflammatory Marker C-Reactive Protein: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients 2016, 8, 12. [Google Scholar] [CrossRef]
- Reger, M.K.; Zollinger, T.W.; Liu, Z.; Jones, J.; Zhang, J. Association between Urinary Phytoestrogens and C-reactive Protein in the Continuous National Health and Nutrition Examination Survey. J. Am. Coll. Nutr. 2017, 36, 434–441. [Google Scholar] [CrossRef]
- Frankenfeld, C.L. Cardiometabolic risk factors are associated with high urinary enterolactone concentration, independent of urinary enterodiol concentration and dietary fiber intake in adults. J. Nutr. 2014, 144, 1445–1453. [Google Scholar] [CrossRef]
- Sahebkar, A.; Gurban, C.; Serban, A.; Andrica, F.; Serban, M.C. Effects of supplementation with pomegranate juice on plasma C-reactive protein concentrations: A systematic review and meta-analysis of randomized controlled trials. Phytomedicine 2016, 23, 1095–1102. [Google Scholar] [CrossRef] [PubMed]
- Scoditti, E.; Capurso, C.; Capurso, A.; Massaro, M. Vascular effects of the Mediterranean diet-Part II: Role of omega-3 fatty acids and olive oil polyphenols. Vasc. Pharmacol. 2014, 63, 127–134. [Google Scholar] [CrossRef] [PubMed]
- Konstantinidou, V.; Covas, M.I.; Munoz-Aguayo, D.; Khymenets, O.; de la Torre, R.; Saez, G.; Tormos, M.D.; Toledo, E.; Marti, A.; Ruiz-Gutierrez, V.; et al. In vivo nutrigenomic effects of virgin olive oil polyphenols within the frame of the Mediterranean diet: A randomized controlled trial. FASEB J. 2010, 24, 2546–2557. [Google Scholar] [CrossRef] [PubMed]
- Calabriso, N.; Massaro, M.; Scoditti, E.; Pellegrino, M.; Ingrosso, I.; Giovinazzo, G.; Carluccio, M.A. Red Grape Skin Polyphenols Blunt Matrix Metalloproteinase-2 and-9 Activity and Expression in Cell Models of Vascular Inflammation: Protective Role in Degenerative and Inflammatory Diseases. Molecules 2016, 21, 18. [Google Scholar] [CrossRef] [PubMed]
- Yahfoufi, N.; Alsadi, N.; Jambi, M.; Matar, C. The Immunomodulatory and Anti-Inflammatory Role of Polyphenols. Nutrients 2018, 10, 1618. [Google Scholar] [CrossRef] [PubMed]
- Khangholi, S.; Majid, F.A.A.; Berwary, N.J.A.; Ahmad, F.; Aziz, R.B. The Mechanisms of Inhibition of Advanced Glycation End Products Formation through Polyphenols in Hyperglycemic Condition. Planta Med. 2016, 82, 32–45. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.X.; Chen, X.Q. Structures Required of Polyphenols for Inhibiting Advanced Glycation end Products Formation. Curr. Drug Metab. 2013, 14, 414–431. [Google Scholar] [CrossRef] [PubMed]
- Kumagai, Y.; Nakatani, S.; Onodera, H.; Nagatomo, A.; Nishida, N.; Matsuura, Y.; Kobata, K.; Wada, M. Anti-Glycation Effects of Pomegranate (Punica granatum L.) Fruit Extract and Its Components in Vivo and in Vitro. J. Agric. Food Chem. 2015, 63, 7760–7764. [Google Scholar] [CrossRef] [PubMed]
- Zhuang, Y.L.; Ma, Q.Y.; Guo, Y.; Sun, L.P. Purification and identification of rambutan (Nephelium lappaceum) peel phenolics with evaluation of antioxidant and antiglycation activities in vitro. Int. J. Food Sci. Technol. 2017, 52, 1810–1819. [Google Scholar] [CrossRef]
- Ma, H.; Liu, W.X.; Frost, L.; Kirschenbaum, L.J.; Dain, J.A.; Seeram, N.P. Glucitol-core containing gallotannins inhibit the formation of advanced glycation end-products mediated by their antioxidant potential. Food Funct. 2016, 7, 2213–2222. [Google Scholar] [CrossRef]
- Bhuiyan, M.N.I.; Mitsuhashi, S.; Sigetomi, K.; Ubukata, M. Quercetin inhibits advanced glycation end product formation via chelating metal ions, trapping methylglyoxal, and trapping reactive oxygen species. Biosci. Biotechnol. Biochem. 2017, 81, 882–890. [Google Scholar] [CrossRef] [PubMed]
- Yeh, W.J.; Hsia, S.M.; Lee, W.H.; Wu, C.H. Polyphenols with antiglycation activity and mechanisms of action: A review of recent findings. J. Food Drug Anal. 2017, 25, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Navarro, M.; Morales, F.J. Evaluation of an olive leaf extract as a natural source of antiglycative compounds. Food Res. Int. 2017, 92, 56–63. [Google Scholar] [CrossRef] [PubMed]
- Navarro, M.; Morales, F.J.; Ramos, S. Olive leaf extract concentrated in hydroxytyrosol attenuates protein carbonylation and the formation of advanced glycation end products in a hepatic cell line (HepG2). Food Funct. 2017, 8, 944–953. [Google Scholar] [CrossRef] [PubMed]
- Zhang, P.W.; Tian, C.; Xu, F.Y.; Chen, Z.; Burnside, R.; Yi, W.J.; Xiang, S.Y.; Xie, X.; Wu, N.N.; Yang, H.; et al. Green Tea Polyphenols Alleviate Autophagy Inhibition Induced by High Glucose in Endothelial Cells. Biomed. Environ. Sci. 2016, 29, 524–528. [Google Scholar] [CrossRef] [PubMed]
- Holczer, M.; Besze, B.; Zambo, V.; Csala, M.; Banhegyi, G.; Kapuy, O. Epigallocatechin-3-Gallate (EGCG) Promotes Autophagy-Dependent Survival via Influencing the Balance of mTOR-AMPK Pathways upon Endoplasmic Reticulum Stress. Oxid. Med. Cell. Longev. 2018, 2018, 6721530. [Google Scholar] [CrossRef]
- Kim, H.S.; Montana, V.; Jang, H.J.; Parpura, V.; Kim, J.A. Epigallocatechin Gallate (EGCG) Stimulates Autophagy in Vascular Endothelial Cells a potential role for reducing lipid accumulation. J. Biol. Chem. 2013, 288, 22693–22705. [Google Scholar] [CrossRef]
- Wang, W.; Jing, T.; Yang, X.; He, Y.; Wang, B.; Xiao, Y.; Shang, C.; Zhang, J.; Lin, R. Hydroxytyrosol regulates the autophagy of vascular adventitial fibroblasts through the SIRT1-mediated signaling pathway. Can. J. Physiol. Pharmacol. 2018, 96, 88–96. [Google Scholar] [CrossRef]
- Yang, X.; Jing, T.; Li, Y.; He, Y.; Zhang, W.; Wang, B.; Xiao, Y.; Wang, W.; Zhang, J.; Wei, J.; et al. Hydroxytyrosol Attenuates LPS-Induced Acute Lung Injury in Mice by Regulating Autophagy and Sirtuin Expression. Curr. Mol. Med. 2017, 17, 149–159. [Google Scholar] [CrossRef]
- Cetrullo, S.; D’Adamo, S.; Guidotti, S.; Borzi, R.M.; Flamigni, F. Hydroxytyrosol prevents chondrocyte death under oxidative stress by inducing autophagy through sirtuin 1-dependent and -independent mechanisms. Biochim. Biophys. Acta 2016, 1860, 1181–1191. [Google Scholar] [CrossRef]
- Rigacci, S.; Miceli, C.; Nediani, C.; Berti, A.; Cascella, R.; Pantano, D.; Nardiello, P.; Luccarini, I.; Casamenti, F.; Stefani, M. Oleuropein aglycone induces autophagy via the AMPK/mTOR signalling pathway: A mechanistic insight. Oncotarget 2015, 6, 35344–35357. [Google Scholar] [CrossRef] [PubMed]
- Boakye, Y.D.; Groyer, L.; Heiss, E.H. An increased autophagic flux contributes to the anti-inflammatory potential of urolithin A in macrophages. Biochim. Biophys. Acta Gen. Subj. 2018, 1862, 61–70. [Google Scholar] [CrossRef] [PubMed]
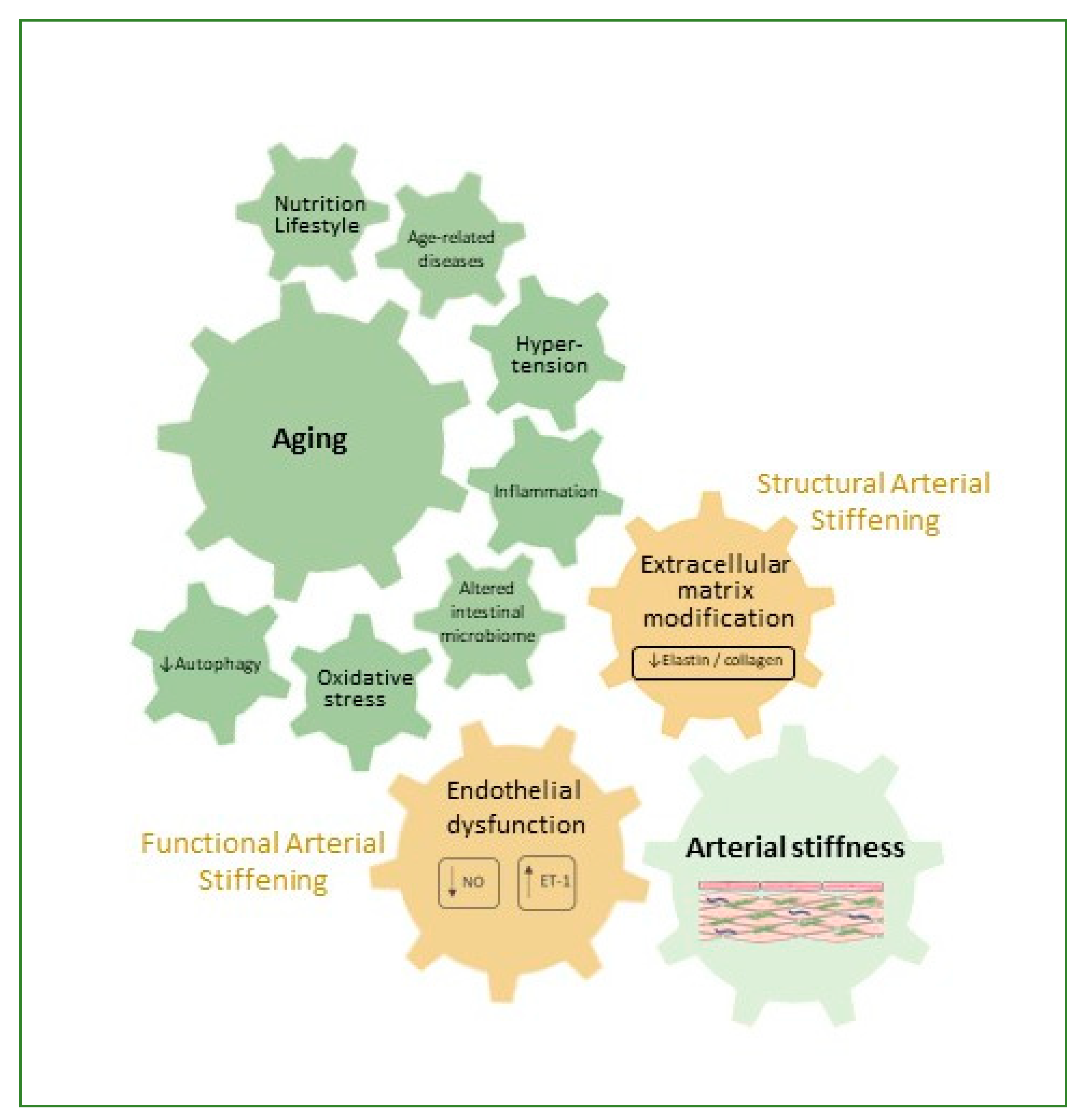
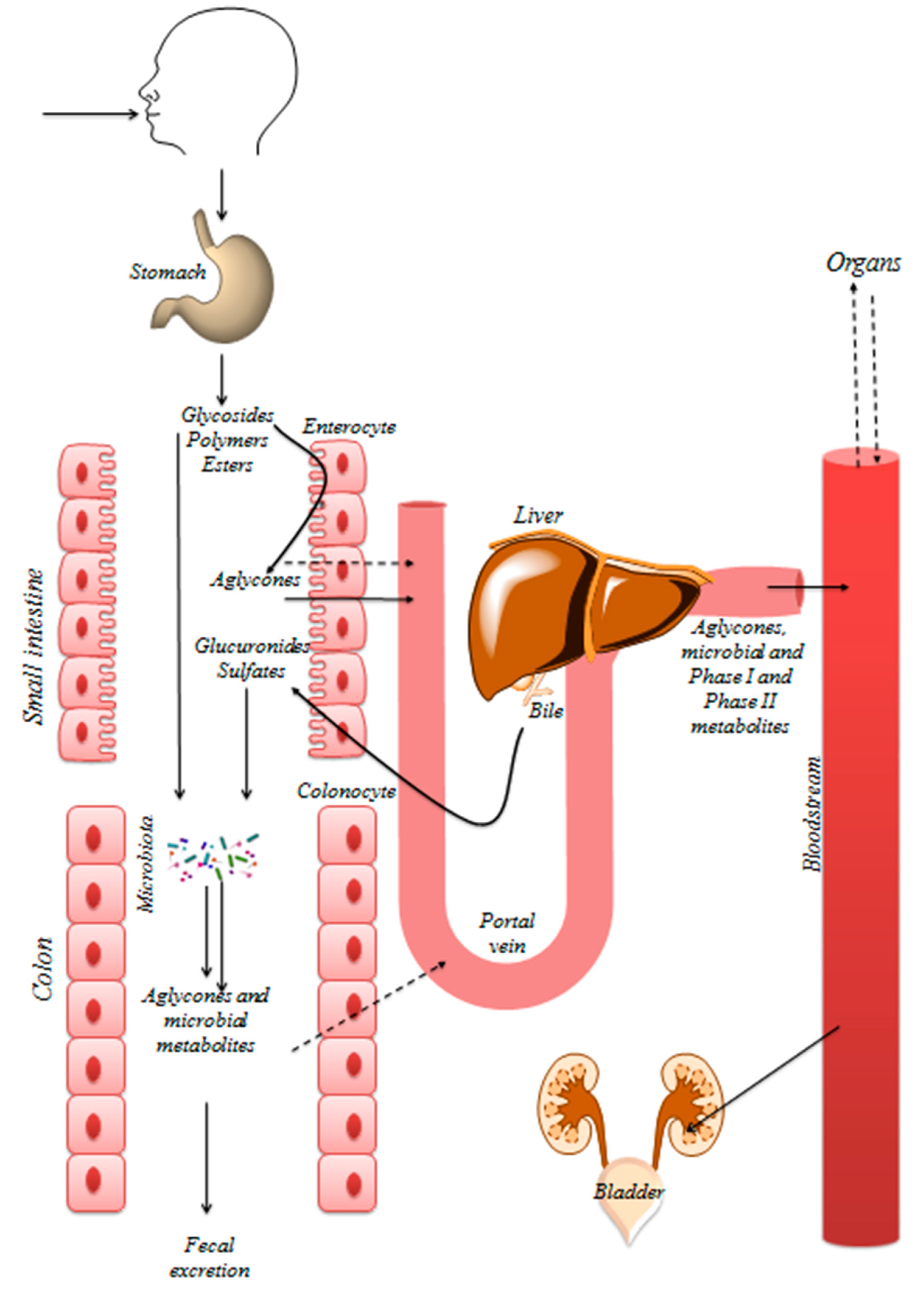
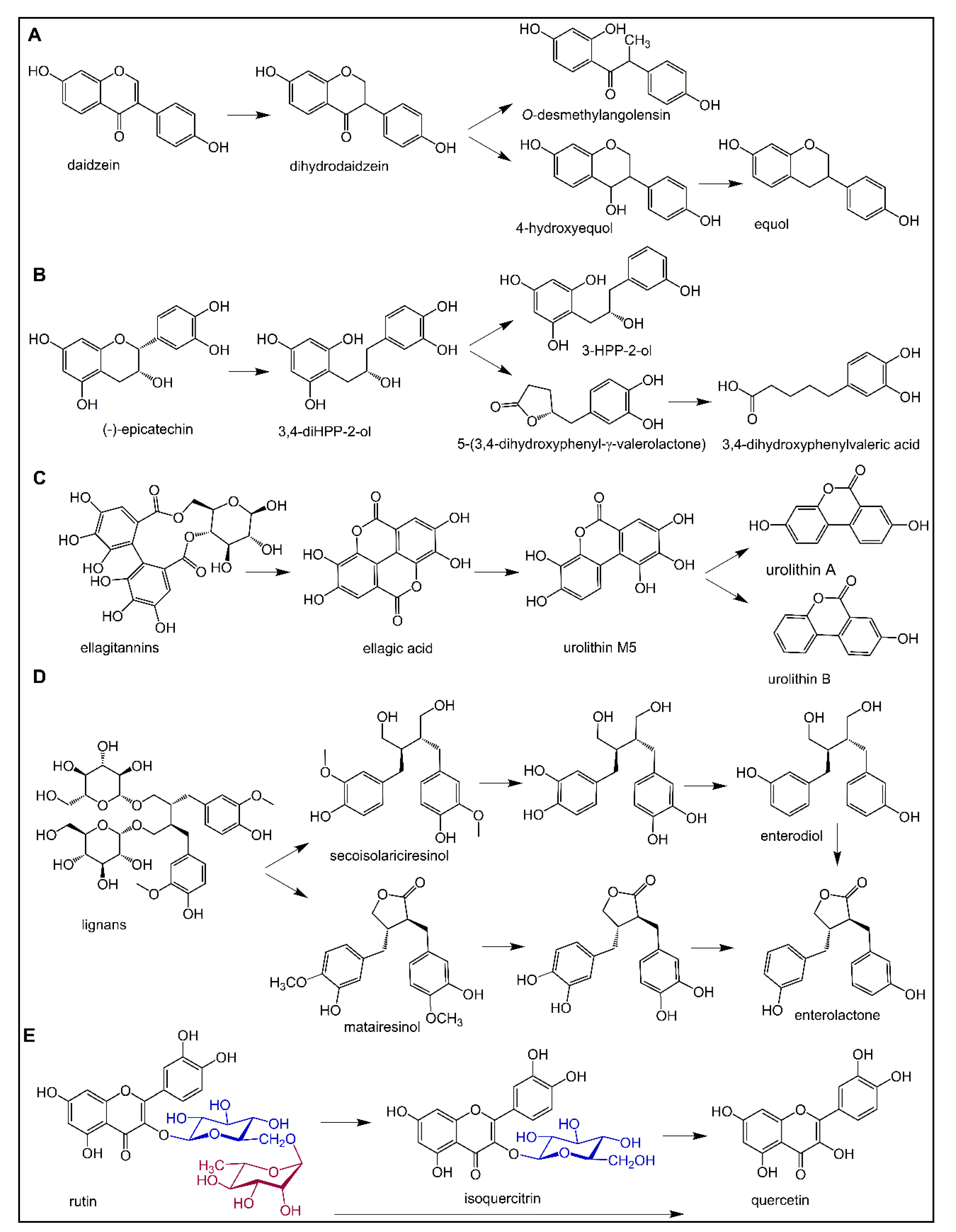
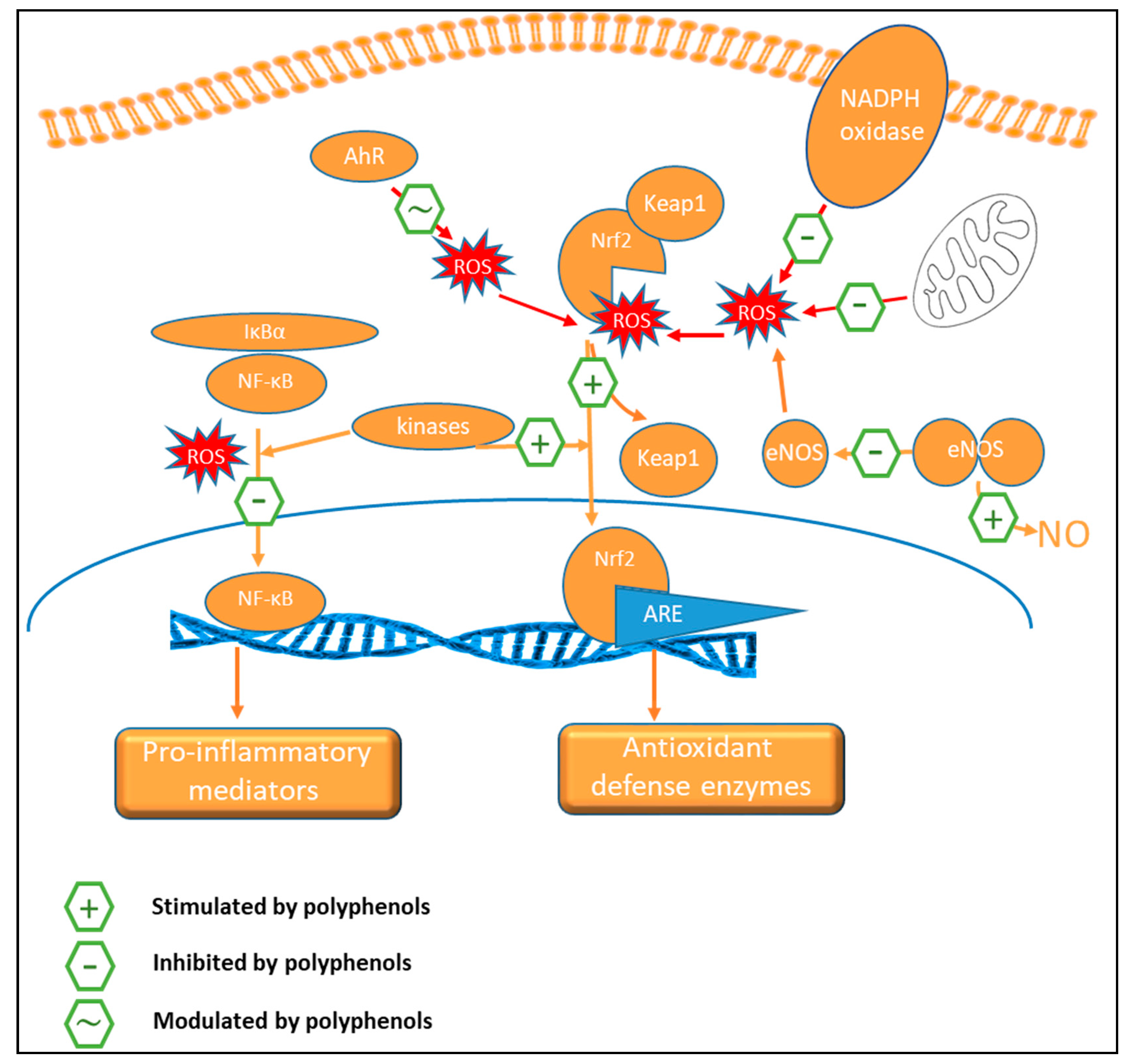
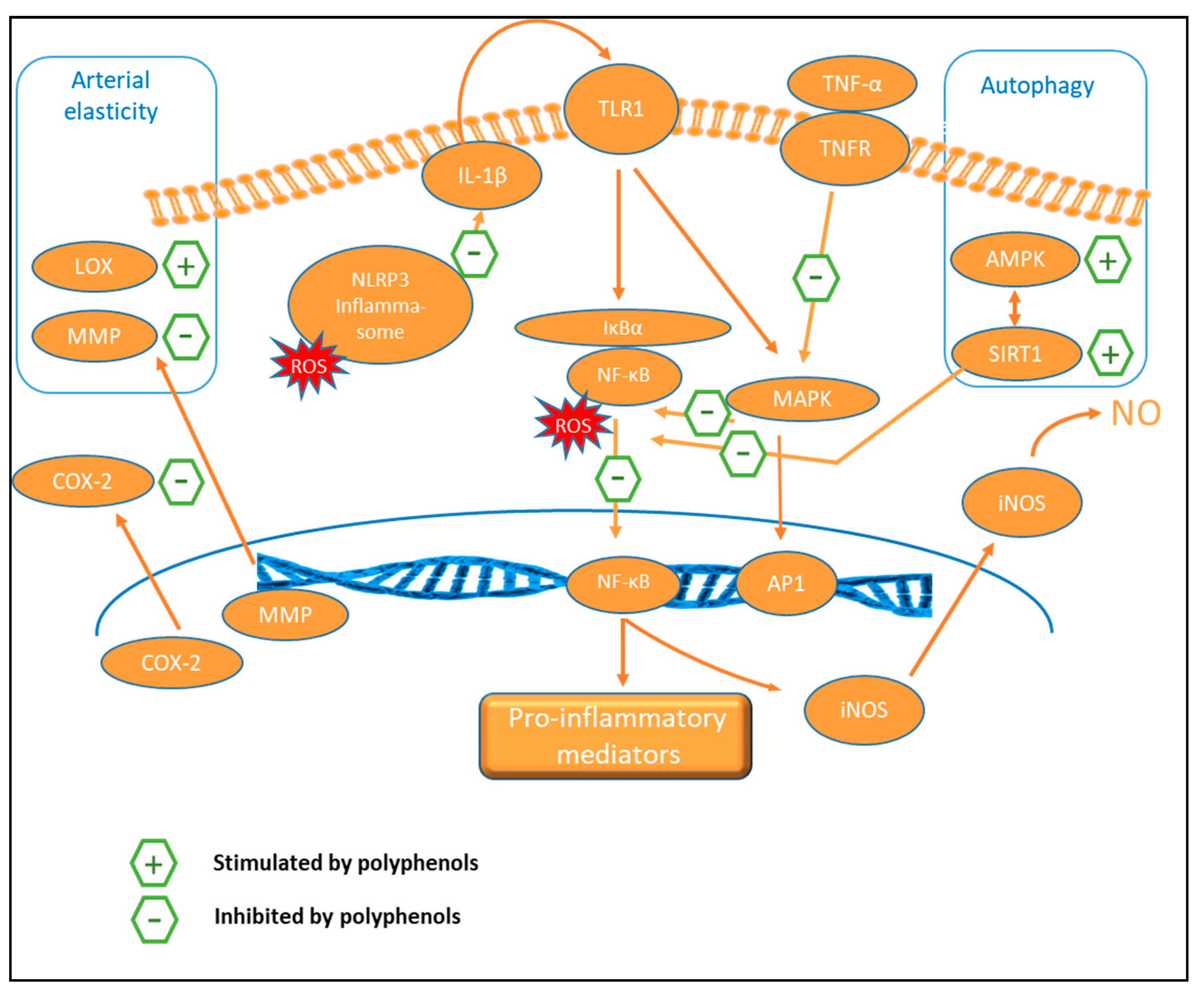
| Dietary Polyphenol Class | Subclass | Compounds (Examples) | Dietary Sources (Examples) |
|---|---|---|---|
| Phenolic acids | Chlorogenic acid, caffeic acid, gallic acid, ferulic acid | coffee, berries, kiwi, apple, cherry | |
| Phenolic alcohols | Hydroxytyrosol | olive | |
| Stilbenes | Resveratrol | grapes, wine | |
| Lignans | Secolariciresinol | linseed | |
| Flavonoids | |||
| Isoflavones | Genistein, daidzein | soy, miso | |
| Flavones | Luteolin, apigenin | celery, parsley, capsicum pepper | |
| Flavanones | Hesperetin, naringenin | oranges, grapefruit, lemon | |
| Flavonols | Quercetin, kaempferol, myricetin | onion, leek, broccoli, berries | |
| Flavanols | (Epi)catechins, (epi)gallocatechins, epigallocatechin gallate | grapes, wine, cocoa, apricots, beans, green tea | |
| Anthocyanins | Delphinidin, cyanidin, malvidin | berries, aubergine, black grapes, rhubarb, red wine | |
| Tannins | Condensed tannins | Procyanidins | cocoa, chocolate, apples, grapes |
| Hydrolyzable tannins | Gallotannins, ellagitannins | mango, pomegranate |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
De Bruyne, T.; Steenput, B.; Roth, L.; De Meyer, G.R.Y.; Santos, C.N.d.; Valentová, K.; Dambrova, M.; Hermans, N. Dietary Polyphenols Targeting Arterial Stiffness: Interplay of Contributing Mechanisms and Gut Microbiome-Related Metabolism. Nutrients 2019, 11, 578. https://doi.org/10.3390/nu11030578
De Bruyne T, Steenput B, Roth L, De Meyer GRY, Santos CNd, Valentová K, Dambrova M, Hermans N. Dietary Polyphenols Targeting Arterial Stiffness: Interplay of Contributing Mechanisms and Gut Microbiome-Related Metabolism. Nutrients. 2019; 11(3):578. https://doi.org/10.3390/nu11030578
Chicago/Turabian StyleDe Bruyne, Tess, Bieke Steenput, Lynn Roth, Guido R. Y. De Meyer, Claudia Nunes dos Santos, Kateřina Valentová, Maija Dambrova, and Nina Hermans. 2019. "Dietary Polyphenols Targeting Arterial Stiffness: Interplay of Contributing Mechanisms and Gut Microbiome-Related Metabolism" Nutrients 11, no. 3: 578. https://doi.org/10.3390/nu11030578
APA StyleDe Bruyne, T., Steenput, B., Roth, L., De Meyer, G. R. Y., Santos, C. N. d., Valentová, K., Dambrova, M., & Hermans, N. (2019). Dietary Polyphenols Targeting Arterial Stiffness: Interplay of Contributing Mechanisms and Gut Microbiome-Related Metabolism. Nutrients, 11(3), 578. https://doi.org/10.3390/nu11030578







