Beverage Intake and Drinking Patterns—Clues to Support Older People Living in Long-Term Care to Drink Well: DRIE and FISE Studies
Abstract
1. Introduction
2. Materials and Methods
2.1. DRIE
2.2. FISE
2.3. Data Analysis
3. Results
3.1. Study Flow and Participant Characteristics
3.2. Drinks Intake and Patterns
3.2.1. Hydration and Drinks Intake
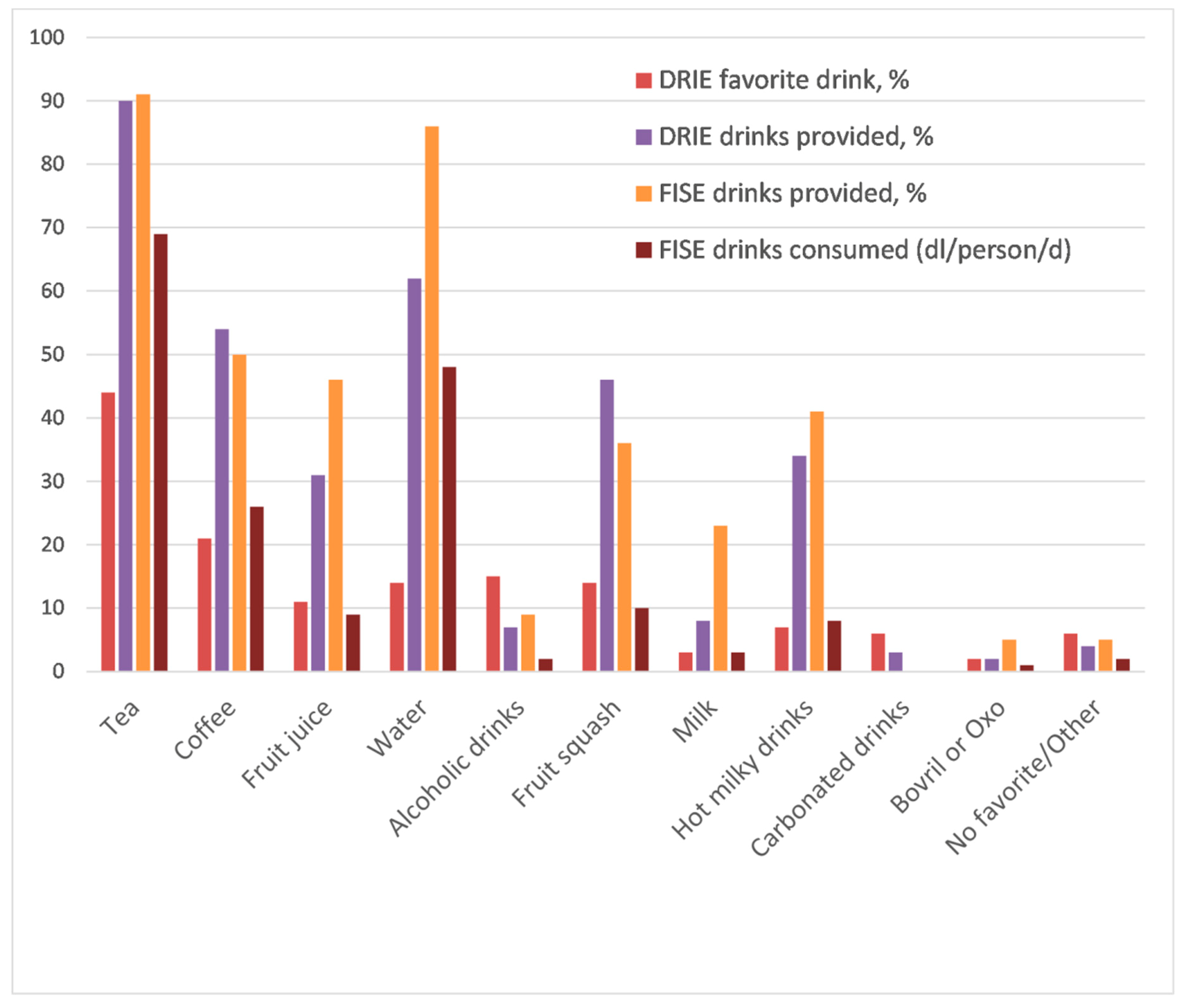
3.2.2. Types of Drinks Enjoyed by Residents and Provided by Long-Term Care Facilities
3.2.3. Proportions of Drinks Consumed
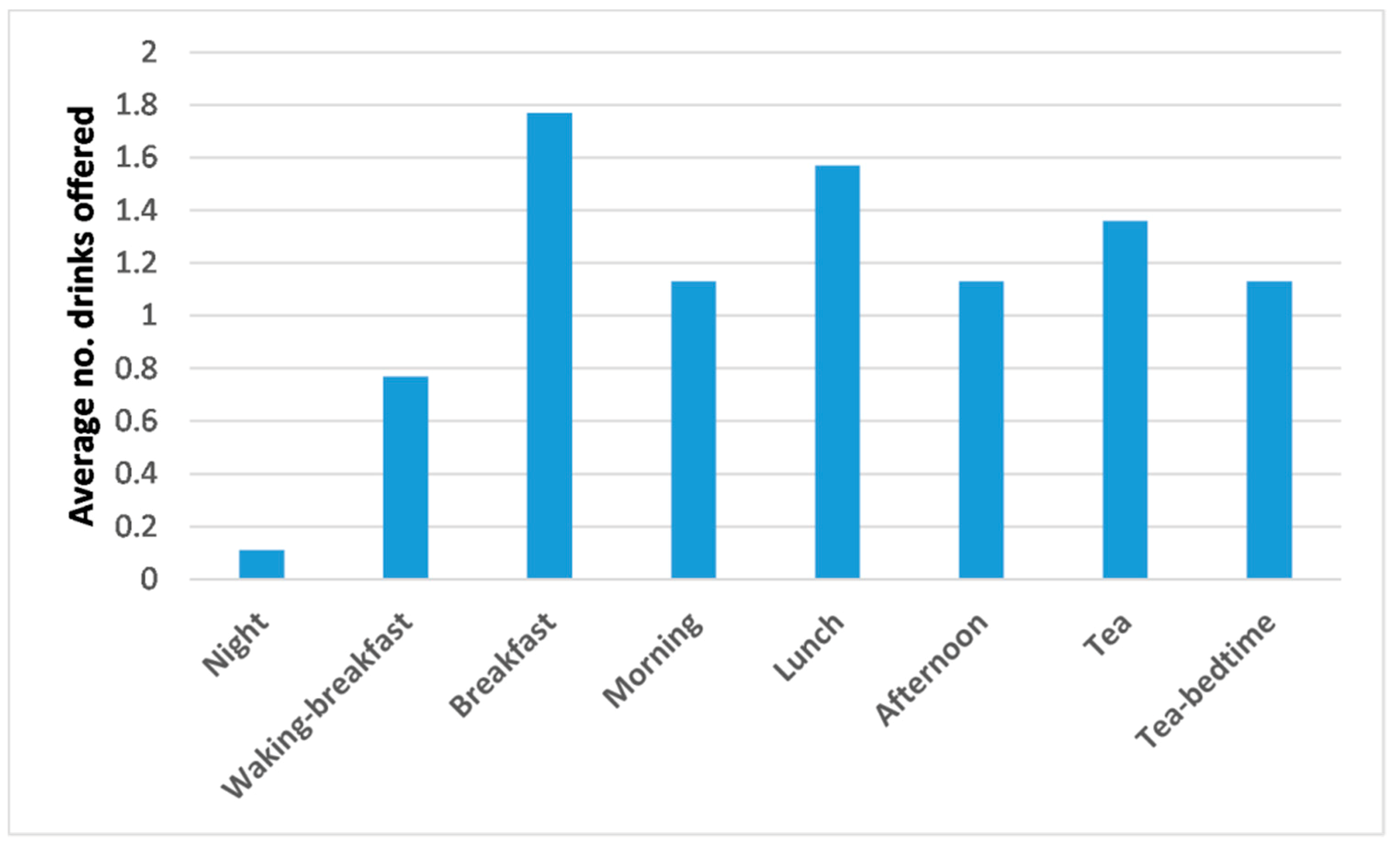
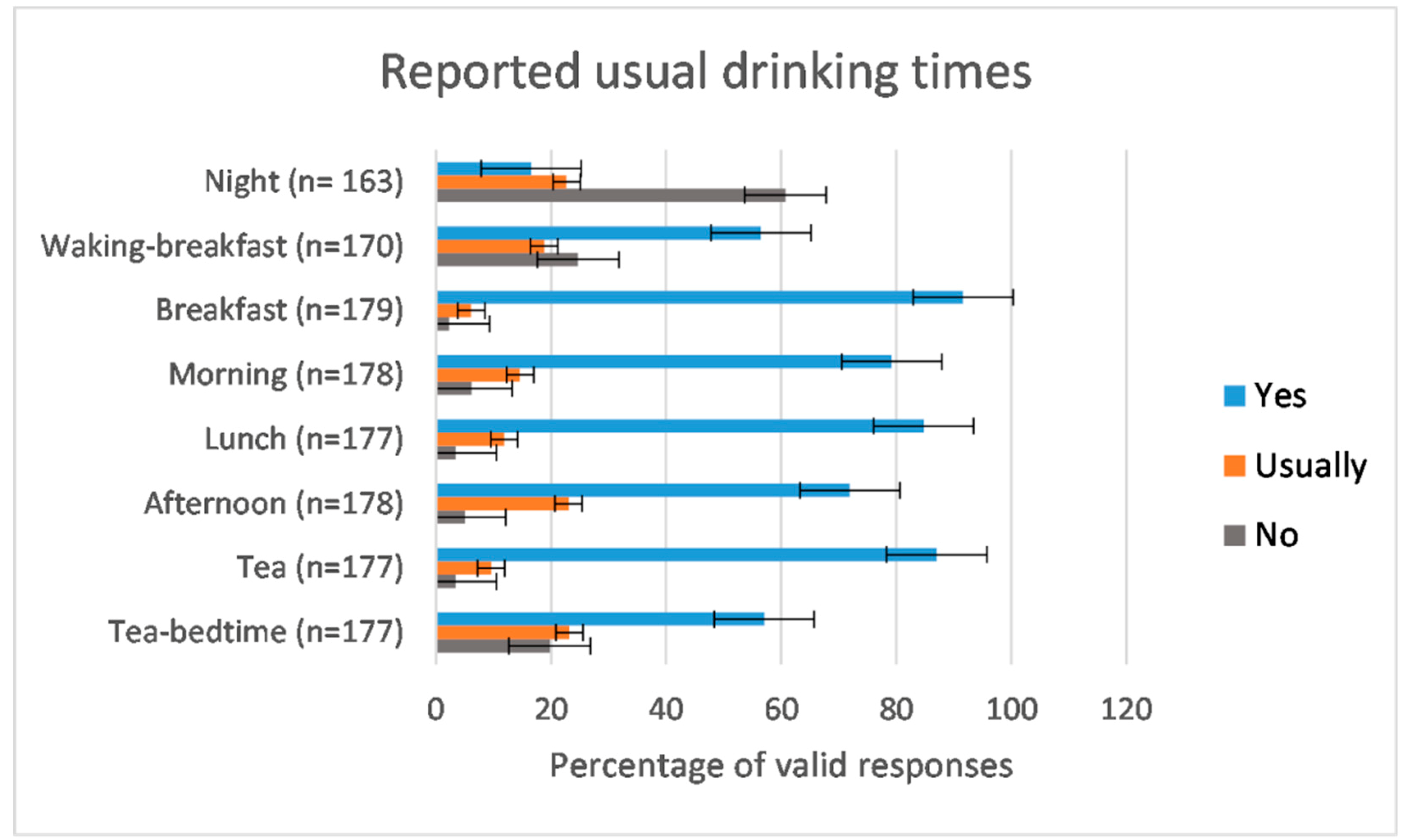
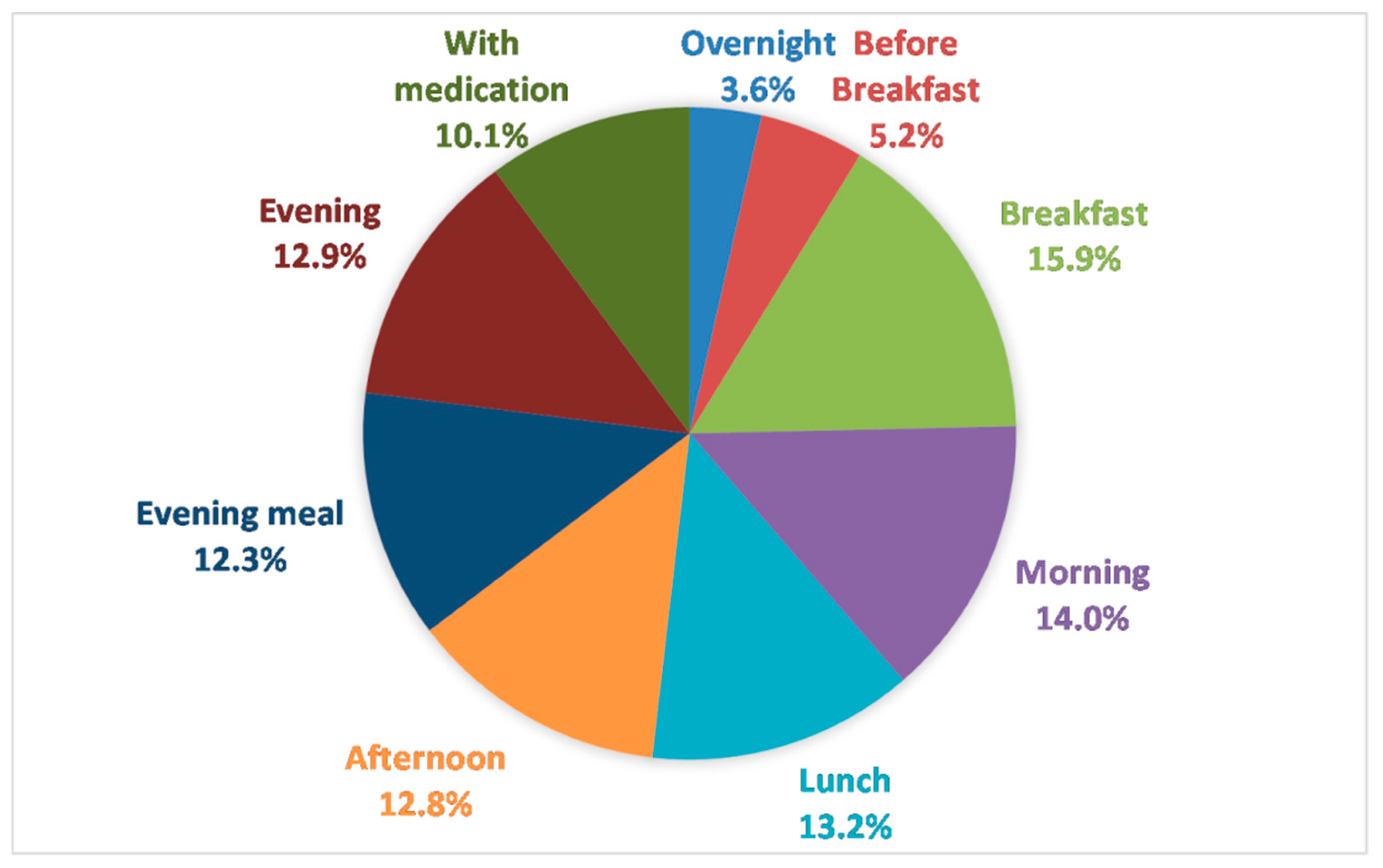
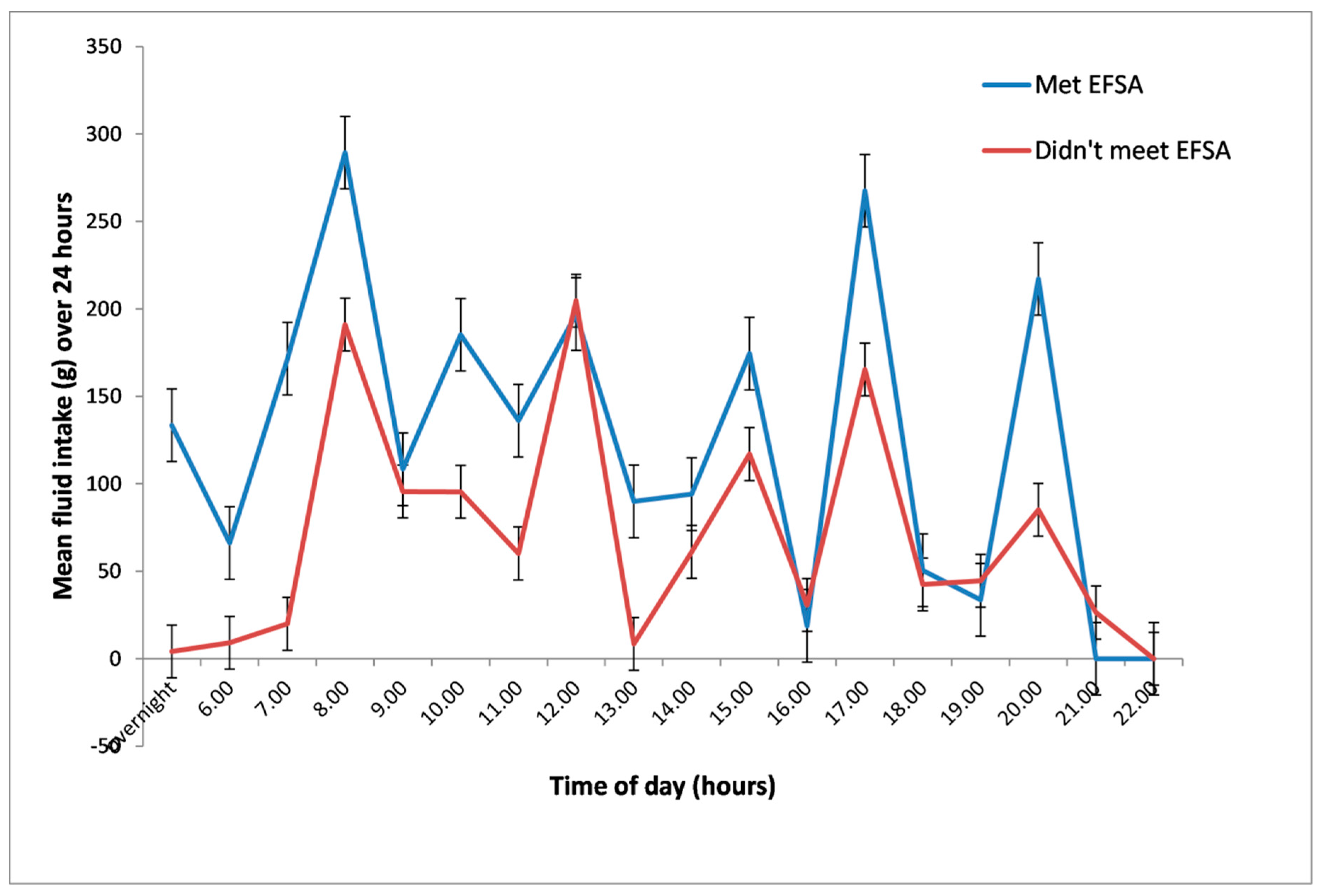
3.2.4. Numbers and Timing of Drinks—Routines
3.2.5. Variety of Drinks
3.2.6. Thirst and Knowledge of Hydration Status
3.2.7. Drinks Outside Routine Provision
3.2.8. Reasons for Cutting Down on Drinks
4. Discussion
5. Conclusions
- Offering drinks more often through the day is likely to increase fluid intake. Earlier in the day can be more helpful than later, as evening drinks may be resisted. Don’t rely on residents helping themselves to drinks, or requesting them—residents who don’t, will drink too little.
- Regular drinks provision is vital, so care home staff should know the importance of not missing drinks rounds and that all residents must be offered drinks during rounds.
- Drinks handed to residents need to provide enough fluid to meet minimum requirements. Where small cups are used, more frequent drinks are needed to ensure adequate fluid.
- Promoting more fluid with medications helps to increase fluid intake, makes swallowing pills easier and reduces the side effects from some medications [47].
- Improving continence support and ease of access to toilets is likely to improve drinking.
- Personal preference is key, so noting and adapting to residents’ preferences for types and presentation of drinks is vital.
- Offering hot milky drinks, fruit juice and alcohol more frequently may improve drinking and enjoyment (as these drinks are often completely consumed, and are as hydrating as water, coffee and tea).
- Asking all residents whether they sometimes drink less than they would like to and if so, why? Individualizing care to address these factors may support drinking.
- Thirst, or lack of it, is not a good guide to whether older adults are drinking enough.
- All residents of long-term care are at risk of dehydration, but focusing particular drinking support on those with cognitive deficits and diabetes will support hydration [3].
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bunn, D.; Hooper, L.; Welch, A. Dehydration and Malnutrition in Residential Care: Recommendations for Strategies for Improving Practice Derived from a Scoping Review of Existing Policies and Guidelines. Geriatrics 2018, 3, 77. [Google Scholar] [CrossRef]
- Hooper, L. Why, Oh Why Are So Many Older Adults Not Drinking Enough Fluid? J. Acad. Nutr. Diet. 2016, 116, 774–778. [Google Scholar] [CrossRef] [PubMed]
- Hooper, L.; Bunn, D.K.; Downing, A.; Jimoh, F.O.; Groves, J.; Free, C.; Cowap, V.; Potter, J.F.; Hunter, P.R.; Shepstone, L. Which Frail Older People Are Dehydrated? The UK DRIE Study. J. Gerontol. A Biol. Sci. Med. Sci. 2016, 71, 1341–1347. [Google Scholar] [CrossRef] [PubMed]
- Marra, M.V.; Simmons, S.F.; Shotwell, M.S.; Hudson, A.; Hollingsworth, E.K.; Long, E.; Kuertz, B.; Silver, H.J. Elevated Serum Osmolality and Total Water Deficit Indicate Impaired Hydration Status in Residents of Long-Term Care Facilities Regardless of Low or High Body Mass Index. J. Acad. Nutr. Diet. 2016, 116, 828–836. [Google Scholar] [CrossRef] [PubMed]
- Abdelhamid, A.; Bunn, D.; Copley, M.; Cowap, V.; Dickinson, A.; Gray, L.; Howe, A.; Killett, A.; Lee, J.; Li, F.; et al. Effectiveness of interventions to directly support food and drink intake in people with dementia: systematic review and meta-analysis. BMC Geriatr. 2016, 16, 26. [Google Scholar] [CrossRef] [PubMed]
- Bunn, D.; Jimoh, F.; Wilsher, S.H.; Hooper, L. Increasing fluid intake and reducing dehydration risk in older people living in long-term care: A systematic review. J. Am. Med. Dir. Assoc. 2015, 16, 101–113. [Google Scholar] [CrossRef] [PubMed]
- Bunn, D.K.; Abdelhamid, A.; Copley, M.; Cowap, V.; Dickinson, A.; Howe, A.; Killett, A.; Poland, F.; Potter, J.F.; Richardson, K.; et al. Effectiveness of interventions to indirectly support food and drink intake in people with dementia: Eating and Drinking Well IN dementiA (EDWINA) systematic review. BMC geriatr. 2016, 16, 89. [Google Scholar] [CrossRef] [PubMed]
- Bhalla, A.; Sankaralingam, S.; Dundas, R.; Swaminathan, R.; Wolfe, C.; Rudd, A.G. Influence of raised plasma osmolality on clinical outcome after acute stroke. Stroke 2000, 31, 2043–2048. [Google Scholar] [CrossRef] [PubMed]
- Stookey, J.D.; Purser, J.L.; Pieper, C.F.; Cohen, H.J. Plasma hypertonicity: another marker of frailty? J. Am. Geriatr. Soc. 2004, 52, 1313–1320. [Google Scholar] [CrossRef]
- Stookey, J.D.; Pieper, C.F.; Cohen, H.J. Is the prevalence of dehydration among community-dwelling older adults really low? Informing current debate over the fluid recommendation for adults aged 70+years. Public Health Nutr. 2007, 8, 1275–1285. [Google Scholar] [CrossRef]
- Wachtel, T.J.; Tetu-Mouradjian, L.M.; Goldman, D.L.; Ellis, S.E.; O’Sullivan, P.S. Hyperosmolarity and acidosis in diabetes mellitus: A three-year experience in Rhode Island. J. Gen. Intern. Med. 1991, 6, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Szafara, K.L.; Kruse, R.L.; Mehr, D.R.; Ribbe, M.W.; van der Steen, J.T. Mortality following nursing home-acquired lower respiratory infection: LRI severity, antibiotic treatment, and water intake. J. Am. Med. Dir. Assoc. 2012, 13, 376–383. [Google Scholar] [CrossRef] [PubMed]
- Cheuvront, S.N.; Ely, B.R.; Kenefick, R.W.; Sawka, M.N. Biological variation and diagnostic accuracy of dehydration assessment markers. Am. J. Clin. Nutr. 2010, 92, 565–573. [Google Scholar] [CrossRef] [PubMed]
- Thomas, D.R.; Cote, T.R.; Lawhorne, L.; Levenson, S.A.; Rubenstein, L.Z.; Smith, D.A.; Stefanacci, R.G.; Tangalos, E.G.; Morley, J.E. Understanding Clinical Dehydration and Its Treatment. J. Am. Med. Dir. Assoc. 2008, 9, 292–301. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine. Dietary Reference Intakes for Water, Potassium, Sodium, Chloride, and Sulfate. Panel on Dietary Reference Intakes for Electrolytes and Water, Standing Committee on the Scientific Evaluation of Dietary Reference Intakes; National Academies Press: Washington, DC, USA, 2005. [Google Scholar]
- Leiper, J.B.; Seonaid Primrose, C.; Primrose, W.R.; Phillimore, J.; Maughan, R.J. A comparison of water turnover in older people in community and institutional settings. J. Nutr. Health Aging 2005, 9, 189–193. [Google Scholar] [PubMed]
- Hooper, L.; Bunn, D.; Jimoh, F.O.; Fairweather-Tait, S.J. Water-loss dehydration and aging. Mech. Ageing Dev. 2014. [CrossRef]
- Nordic Nutrition Recommendations 2012: Integrating Nutrition and Physical Activity. Available online: https://www.norden.org/en/publication/nordic-nutrition-recommendations-2012-0 (accessed on 4 February 2019).
- EFSA Panel on Dietetic Products Nutrition and Allergies (NDA). Scientific Opinion on Dietary Reference Values for water. EFSA J. 2010, 8, 1459. [Google Scholar] [CrossRef]
- Centers for Medicare & Medicaid Services. Long-Term Care Facility Resident Assessment Instrument 3.0 User’s Manual Version 1.13. Available online: https://www.cms.gov/Medicare/Quality-Initiatives-Patient-Assessment-Instruments/NursinghomeQualityInits/MDS30RAIManual.html (accessed on 4 February 2019).
- Volkert, D.; Beck, A.M.; Cederholm, T.; Cruz-Jentoft, A.; Goisser, S.; Hooper, L.; Kiesswetter, E.; Maggio, M.; Raynaud-Simon, A.; Sieber, C.C.; et al. ESPEN guideline on clinical nutrition and hydration in geriatrics. Clin. Nutr. 2018, 18, 18. [Google Scholar] [CrossRef]
- Hooper, L.; Bunn, D.K.; Abdelhamid, A.; Gillings, R.; Jennings, A.; Maas, K.; Millar, S.; Twomlow, E.; Hunter, P.R.; Shepstone, L.; et al. Water-loss (intracellular) dehydration assessed using urinary tests: How well do they work? Diagnostic accuracy in older people. Am. J. Clin. Nutr. 2016, 104, 121–131. [Google Scholar] [CrossRef]
- Siervo, M.; Bunn, D.; Prado, C.M.; Hooper, L. Accuracy of prediction equations for serum osmolarity in frail older people with and without diabetes. Am. J. Clin. Nutr. 2014, 100, 867–876. [Google Scholar] [CrossRef]
- Jimoh, F.O.; Bunn, D.; Hooper, L. Assessment of a Self-Reported Drinks Diary for the Estimation of Drinks Intake by Care Home Residents: Fluid Intake Study in the Elderly (FISE). J. Nutr. Health Aging 2015, 19, 491–496. [Google Scholar] [CrossRef] [PubMed]
- Drinks Diary 2015. Available online: https://www.uea.ac.uk/medicine/research/research-evidence-studies/drinks-diary (accessed on 20 December 2018).
- Bunn, D.; Hooper, L. Signs and symptoms of low-intake dehydration do not work in older care home residents: DRIE diagnostic accuracy study. J. Am. Med. Dir. Assoc. 2018, in press. [Google Scholar]
- Folstein, M.F.; Folstein, S.E.; McHugh, P.R. “Mini-mental state”: A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975, 12, 189–198. [Google Scholar] [CrossRef]
- Shapiro, S.S.; Wilk, M.B. An Analysis of Variance Test for Normality (Complete Samples). Biometrika 1965, 52, 591–611. [Google Scholar] [CrossRef]
- Thompson, F.E. Dietary Assessment Methodology, 2nd ed.; Coulston, A., Boushey, C., Ferruzzi, M.E., Eds.; Academic Press: San Diego, CA, USA, 2008. [Google Scholar]
- De Vries, J.H.M.; de Groot, L.C.P.G.M.; van Staveren, W.A. Dietary assessment in elderly people: Experiences gained from studies in the Netherlands. Eur. J. Clin. Nutr. 2009, 63, S69. [Google Scholar] [CrossRef] [PubMed]
- Hedrick, V.; Myers, E.; Zoellner, J.; Duffey, K.; Davy, B. Validation of a Rapid Method to Assess Habitual Beverage Intake Patterns. Nutrients 2018, 10, 83. [Google Scholar] [CrossRef] [PubMed]
- Maughan, R.J.; Watson, P.; Cordery, P.A.; Walsh, N.P.; Oliver, S.J.; Dolci, A.; Rodriguez-Sanchez, N.; Galloway, S.D. A randomized trial to assess the potential of different beverages to affect hydration status: Development of a beverage hydration index. Am. J. Clin. Nutr. 2016, 103, 717–723. [Google Scholar] [CrossRef]
- Gaspar, P.M. Comparison of four standards for determining adequate water intake of nursing home residents. Res. Theory Nurs. Pract. 2011, 25, 11–22. [Google Scholar] [CrossRef]
- Holben, D.H.; Hassell, J.T.; Williams, J.L.; Helle, B. Fluid Intake Compared with Established Standards and Symptoms of Dehydration among Elderly Residents of a Long-Term-Care Facility. J. Am. Diet. Assoc. 1999, 99, 1447–1450. [Google Scholar] [CrossRef]
- Adams, F. How much do elders drink? Geriatric Nursing 1988, 9, 218–221. [Google Scholar] [CrossRef]
- Armstrong-Esther, C.A.; Browne, K.D.; Armstrong-Esther, D.C.; Sander, L. The institutionalized elderly: Dry to the bone! Int. J. Nurs. Stud. 1996, 33, 619–628. [Google Scholar] [CrossRef]
- Gaspar, P.; Finsaas, K.; Finsaas, S.; Gaspar, P.; Holmes, M.; Scherb, C.; Bestram, M. What is the Hydration Level of Assisted Living Memory Care (ALMC) Residents? J. Am. Med. Dir. Assoc. 2017, 18. [Google Scholar] [CrossRef]
- Haveman-Nies, A.; de Groot, L.C.; Van Staveren, W.A. Fluid intake of elderly Europeans. J. Nutr. Health Aging 1997, 1, 151–155. [Google Scholar] [PubMed]
- Drewnowski, A.; Rehm, C.D.; Constant, F. Water and beverage consumption among adults in the United States: Cross-sectional study using data from NHANES 2005–2010. BMC Public Health 2013, 13, 1068. [Google Scholar] [CrossRef] [PubMed]
- Mistura, L.; D’Addezio, L.; Turrini, A. Beverage Consumption Habits in Italian Population: Association with Total Water Intake and Energy Intake. Nutrients 2016, 8, 674. [Google Scholar] [CrossRef] [PubMed]
- McKiernan, F.; Houchins, J.A.; Mattes, R.D. Relationships between human thirst, hunger, drinking, and feeding. Physiol. Behav. 2008, 94, 700–708. [Google Scholar] [CrossRef]
- Spangler, P.F.; Risley, T.R.; Bilyew, D.D. The management of dehydration and incontinence in nonambulatory geriatric patients. J. Appl. Behav. Anal. 1984, 17, 397–401. [Google Scholar] [CrossRef]
- Zembrzuski, C.D. Oral fluid intake and the effect on postural blood pressure and falls in skilled nursing facility residents. Ph.D. Thesis, New York University, New York, NY, USA, 2006. [Google Scholar]
- Gibson, S.; Shirreffs, S.M. Beverage consumption habits “24/7” among British adults: Association with total water intake and energy intake. Nutr. J. 2013, 12, 9. [Google Scholar] [CrossRef]
- Finch, S.; Doyle, W.; Lowe, C.; Bates, C.J.; Prentice, A.; Smithers, G.; Clarke, P.C. National Diet & Nutrition Survey: People Aged 65 Years and Older; HMSO: Norwich, UK, 1998.
- Wu, S.-J.; Wang, H.-H.; Yeh, S.-H.; Wang, Y.-H.; Yang, Y.-M. Hydration status of nursing home residents in Taiwan: A cross-sectional study. J. Adv. Nurs. 2011, 67, 583–590. [Google Scholar] [CrossRef]
- Are You Taking the Right Amount of Water With Your Medicine? Available online: https://safemedicationuse.ca/newsletter/newsletter_water.html (accessed on 20 December 2018).

| Characteristic, n (%) Unless Otherwise Labelled | DRIE Participants n = 188 | FISE Participants | ||
|---|---|---|---|---|
| All FISE Participants, n = 22 | Low Drinks Intake (<EFSA std), n = 10 | Good Drinks Intake (≥EFSA std), n = 12 | ||
| Age (years), mean ±SD (range) | 85.7 ± 7.8 (65 to 105) | 86.8 ± 8.5 (68 to 100) | 81.9 ± 9.1 (68 to 93) | 90.8 ± 5.7 (82 to 100) |
| Female | 124 (66%) | 16 (73%) | 7 (70%) | 9 (75%) |
| BMI (kg/m2), mean ± SD | 25.8 ± 5.6 | 24.7 ± 4.4 | 25.1 ± 5.5 | 24.4 ± 3.5 |
| Underweight, <20 | 32 (17%) | 4 (18%) | 2 (20%) | 2 (17%) |
| Normal, 20–24.9 | 57 (30%) | 8 (36%) | 4 (40%) | 4 (33%) |
| Overweight, 25–29.9 | 63 (34%) | 7 (32%) | 1 (10%) | 6 (50%) |
| Obese, ≥30 | 36 (19%) | 3 (14%) | 3 (30%) | 0 (0%) |
| MMSE score, mean ± SD | 21.8 ± 5.7 (n = 180) | 25.0 ± 4.4 | 22.2 ± 5.0 | 27.0 ± 2.3 |
| Normal cognitive function (≥24) | 83 (46%) | 16 (73%) | 5 (50%) | 11 (92%) |
| Cognitive impairment (<24) | 97 (54%) | 6 (27%) | 5 (50%) | 1 (8%) |
| Barthel Index (BI) score, mean ± SD | 67.4 (26.1) | 79.1 ± 23.6 | 71.0 ± 30.1 | 85.8 ± 14.7 |
| BI score ≥67 | 109 (58%) | 18 (82%) | 7 (70%) | 11 (92%) |
| BI score <67 | 79 (42%) | 4 (18%) | 3 (30%) | 1 (8%) |
| Serum osmolality * (mOsm/kg), mean ± SD | 293.4 ± 8.1 | 292.4 ± 9.6 | 292.5 ± 11.4 | 292.3 ± 8.4 |
| Dehydrated (>300 mOsm/kg) | 38 (20%) | 4 (18%) | 2 (20%) | 2 (17%) |
| Impending or current dehydration (≥295 mOsm/kg) | 90 (48%) | 9 (41%) | 4 (40%) | 5 (42%) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jimoh, O.F.; Brown, T.J.; Bunn, D.; Hooper, L. Beverage Intake and Drinking Patterns—Clues to Support Older People Living in Long-Term Care to Drink Well: DRIE and FISE Studies. Nutrients 2019, 11, 447. https://doi.org/10.3390/nu11020447
Jimoh OF, Brown TJ, Bunn D, Hooper L. Beverage Intake and Drinking Patterns—Clues to Support Older People Living in Long-Term Care to Drink Well: DRIE and FISE Studies. Nutrients. 2019; 11(2):447. https://doi.org/10.3390/nu11020447
Chicago/Turabian StyleJimoh, Oluseyi F., Tracey J. Brown, Diane Bunn, and Lee Hooper. 2019. "Beverage Intake and Drinking Patterns—Clues to Support Older People Living in Long-Term Care to Drink Well: DRIE and FISE Studies" Nutrients 11, no. 2: 447. https://doi.org/10.3390/nu11020447
APA StyleJimoh, O. F., Brown, T. J., Bunn, D., & Hooper, L. (2019). Beverage Intake and Drinking Patterns—Clues to Support Older People Living in Long-Term Care to Drink Well: DRIE and FISE Studies. Nutrients, 11(2), 447. https://doi.org/10.3390/nu11020447






