25-Hydroxyvitamin D Measurement in Human Hair: Results from a Proof-of-Concept study
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Hair Collection, Washing and Sample Preparation
2.3. Blood Sample Collection
2.4. 25(OH)D Assessment
2.5. Approximation of 25(OH)D Concentration in Hair
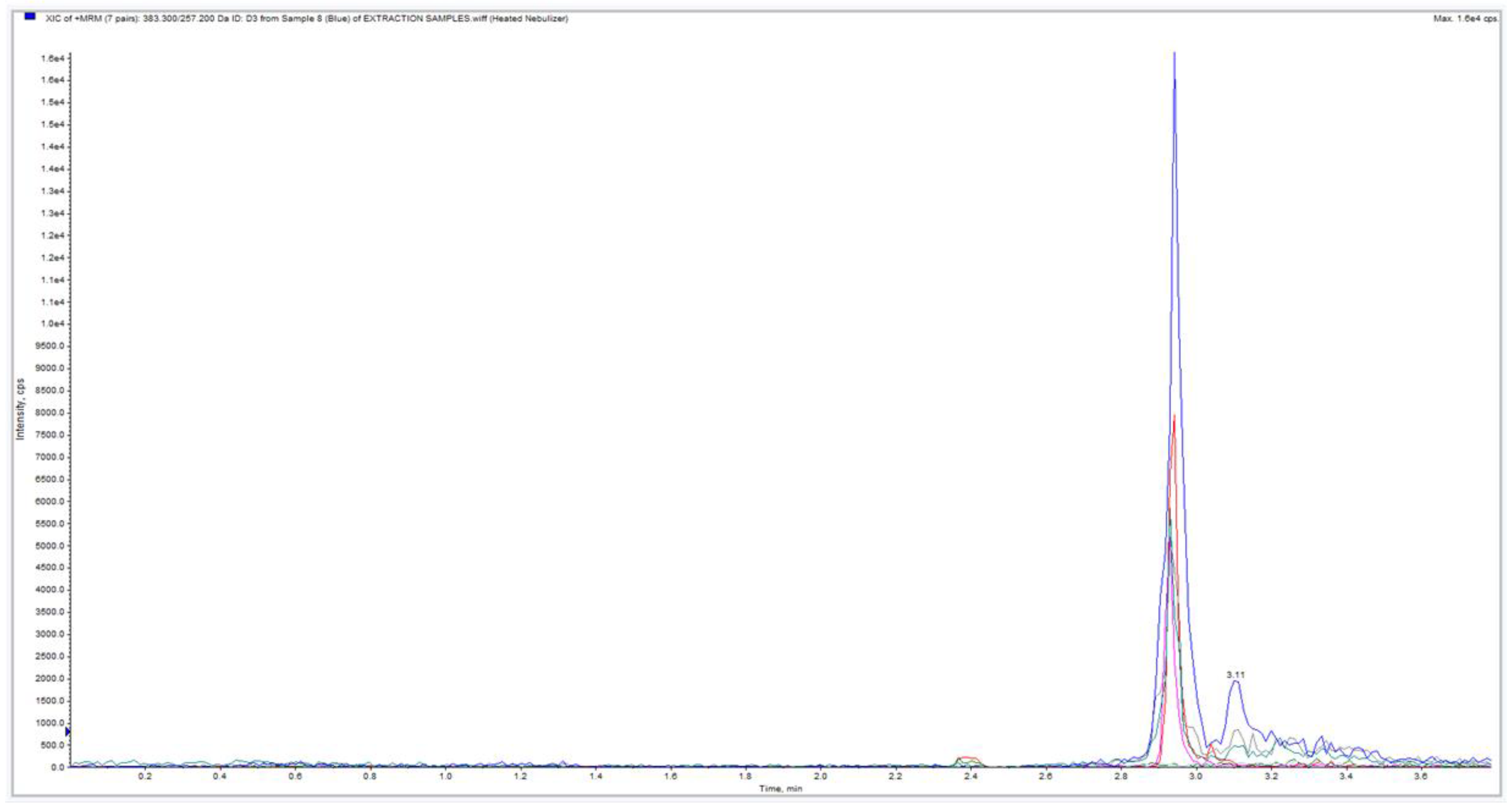
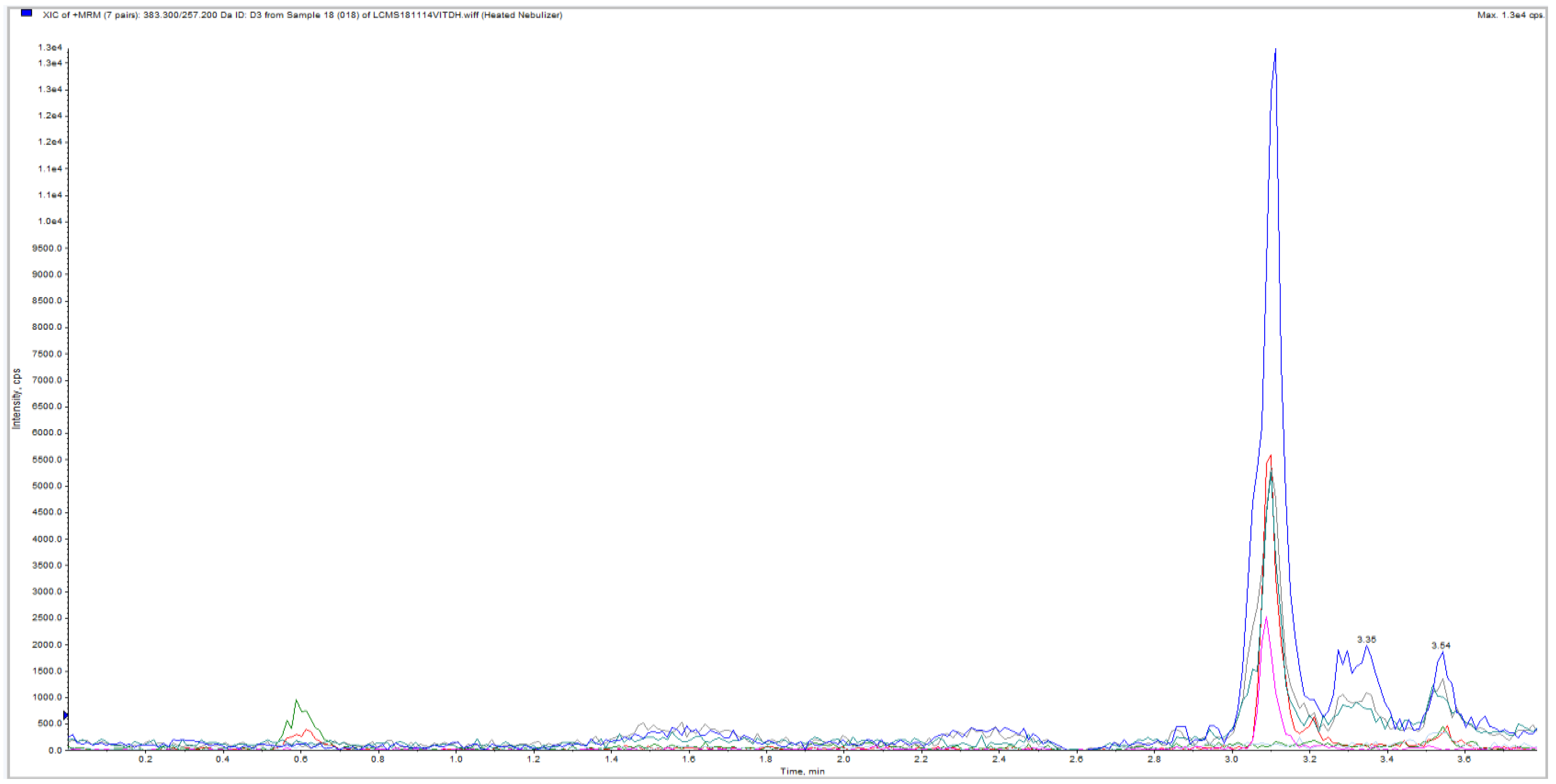
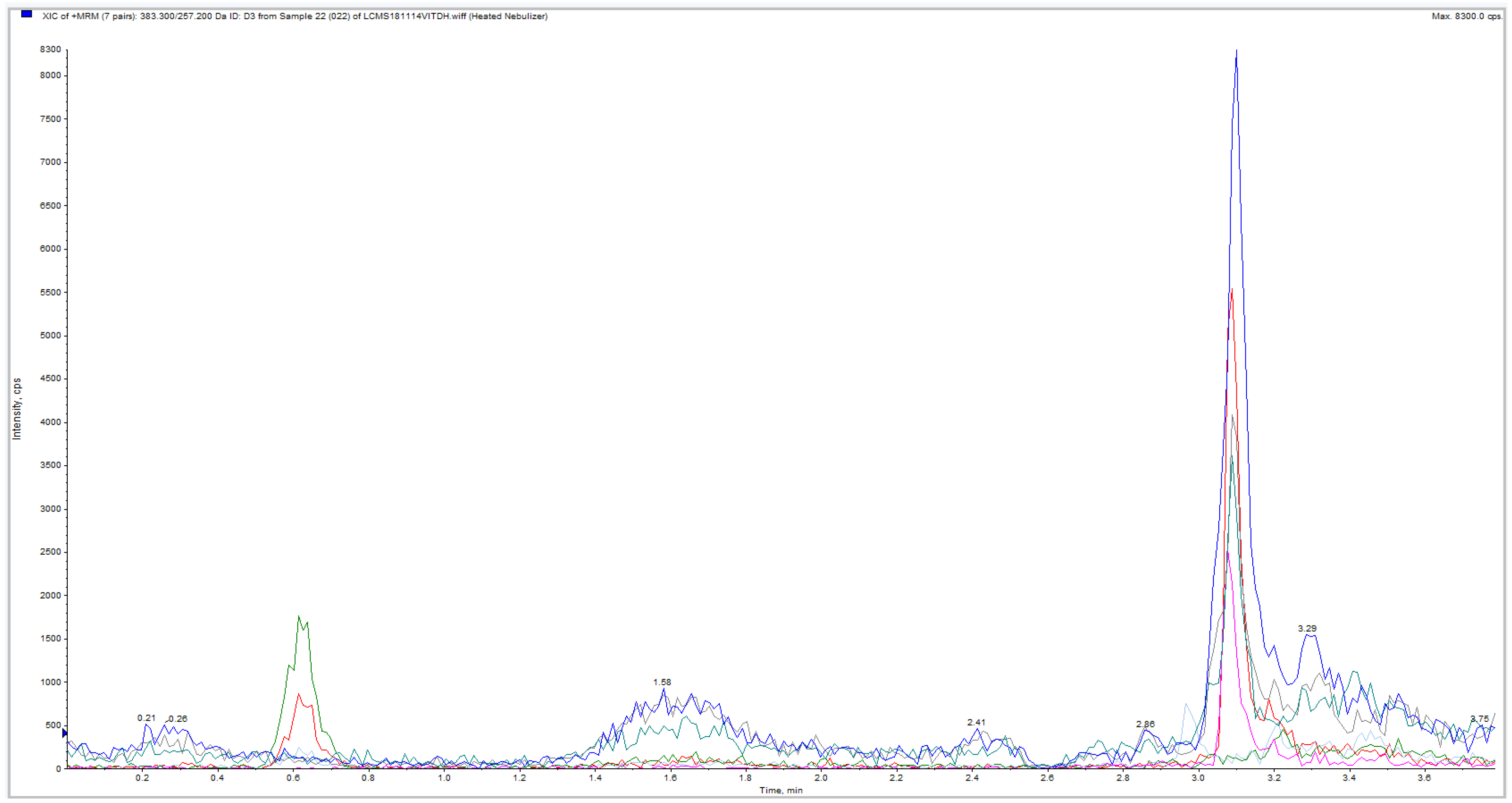
3. Results
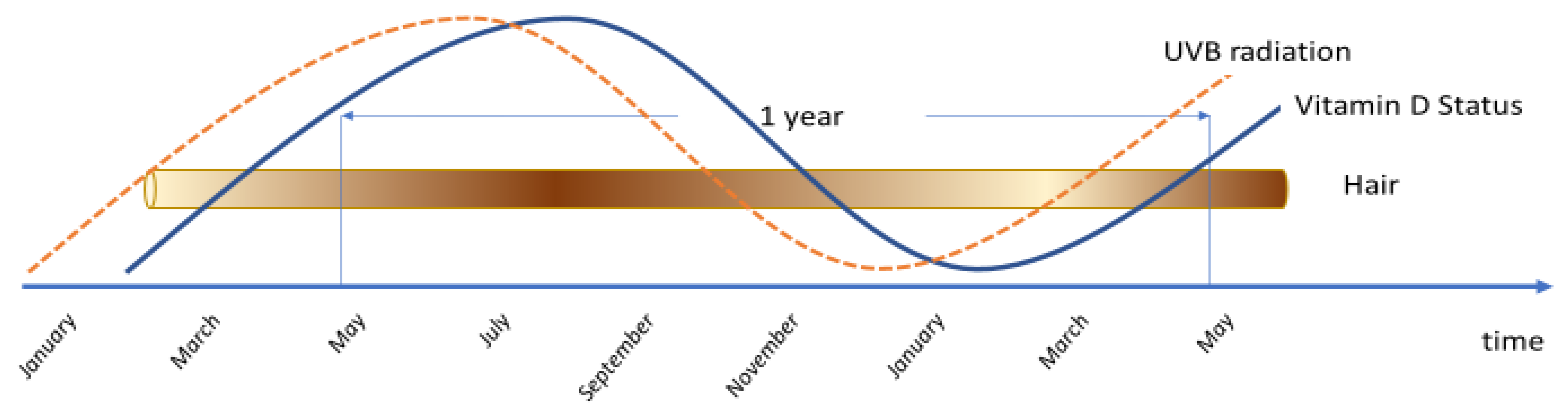
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| LC-MS/MS | Liquid Chromatography Tandem Mass Spectrometry |
| 25OHD | 25-hydroxyvitamin D |
References
- Theodoratou, E.; Tzoulaki, I.; Zgaga, L.; Ioannidis, J.P.A. Vitamin D and multiple health outcomes: Umbrella review of systematic reviews and meta-analyses of observational studies and randomised trials. BMJ 2014, 348, g2035. [Google Scholar] [CrossRef] [PubMed]
- Laird, E.; McNulty, H.; Ward, M.; Hoey, L.; McSorley, E.; Wallace, J.M.; Carson, E.; Molloy, A.M.; Healy, M.; Casey, M.C.; et al. Vitamin D deficiency is associated with inflammation in older Irish adults. J. Clin. Endocrinol. Metab. 2014, 99, 1807–1815. [Google Scholar] [CrossRef] [PubMed]
- Kelly, D.; Theodoratou, E.; Farrington, S.M.; Fraser, R.; Campbell, H.; Dunlop, M.G.; Zgaga, L. The contributions of adjusted ambient ultraviolet B radiation at place of residence and other determinants to serum 25-hydroxyvitamin D concentrations. Br. J. Dermatol. 2016, 174, 1068–1078. [Google Scholar] [CrossRef] [PubMed]
- O’Sullivan, F.; Laird, E.; Kelly, D.; van Geffen, J.; van Weele, M.; McNulty, H.; Hoey, L.; Healy, M.; McCarroll, K.; Cunningham, C.; et al. Ambient UVB Dose and Sun Enjoyment Are Important Predictors of Vitamin D Status in an Older Population. J. Nutr. 2017, 147, 858–868. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Vitamin D: Importance in the prevention of cancers, type 1 diabetes, heart disease, and osteoporosis. Am. J. Clin. Nutr. 2004, 79, 362–371. [Google Scholar] [CrossRef]
- Zgaga, L.; Theodoratou, E.; Farrington, S.M.; Agakov, F.; Tenesa, A.; Walker, M.; Knox, S.; Wallace, A.M.; Cetnarskyj, R.; McNeill, G.; et al. Diet, environmental factors, and lifestyle underlie the high prevalence of vitamin D deficiency in healthy adults in Scotland, and supplementation reduces the proportion that are severely deficient. J. Nutr. 2011, 141, 1535–1542. [Google Scholar] [CrossRef]
- Harnack, L.J.; Steffen, L.; Zhou, X.; Luepker, R.V. Trends in vitamin D intake from food sources among adults in the Minneapolis-St Paul, MN, metropolitan area, 1980–1982 through 2007–2009. Am. Diet. Assoc. 2011, 111, 1329–1334. [Google Scholar] [CrossRef] [PubMed]
- Holden, J.M.; Lemar, L.E.; Exler, J. Vitamin D in foods: Development of the US Department of Agriculture database. Am. J. Clin. Nutr. 2008, 87, 1092S–1096S. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef]
- Knox, S.; Harris, J.; Calton, L.; Wallace, A.M. A simple automated solid-phase extraction procedure for measurement of 25-hydroxyvitamin D3 and D2 by liquid chromatography-tandem mass spectrometry. Ann. Clin. Biochem. 2009, 46, 226–230. [Google Scholar] [CrossRef]
- Shah, S.; Chiang, C.; Lu, Z.; Bui, M.; Zebaze, R.; Seeman, E. Serum 25-Hydroxyvitamin D Insufficiency in Search of a Bone Disease. J. Clin. Endocrinol. Metab. 2017, 102, 2321–2328. [Google Scholar] [CrossRef] [PubMed]
- Wallace, A.M.; Gibson, S.; de la Hunty, A.; Lamberg-Allardt, C.; Ashwell, M. Measurement of 25-hydroxyvitamin D in the clinical laboratory: Current procedures, performance characteristics and limitations. Steroids 2010, 75, 477–488. [Google Scholar] [CrossRef] [PubMed]
- Barroso, M.; Gallardo, E. Assessing cocaine abuse using LC-MS/MS measurements in biological specimens. Bioanalysis 2015, 7, 1497–1525. [Google Scholar] [CrossRef] [PubMed]
- Russell, E.; Koren, G.; Rieder, M.; Van Uum, S. Hair cortisol as a biological marker of chronic stress: Current status, future directions and unanswered questions. Psychoneuroendocrinology 2012, 37, 589–601. [Google Scholar] [CrossRef] [PubMed]
- Wolff, K.; Farrell, M.; Marsden, J.; Monteiro, M.G.; Ali, R.; Welch, S.; Strang, J. A review of biological indicators of illicit drug use, practical considerations and clinical usefulness. Addiction 1999, 94, 1279–1298. [Google Scholar] [CrossRef] [PubMed]
- Wright, K.D.; Hickman, R.; Laudenslager, M.L. Hair cortisol analysis: A promising biomarker of HPA activation in older adults. Gerontologist 2015, 55 (Suppl. 1), S140–S145. [Google Scholar] [CrossRef] [PubMed]
- Cooper, G.A.; Kronstrand, R.; Kintz, P. Society of Hair Testing. Society of Hair Testing guidelines for drug testing in hair. Forensic. Sci. Int. 2012, 218, 20–24. [Google Scholar] [CrossRef]
- Gao, W.; Kirschbaum, C.; Grass, J.; Stalder, T. LC-MS based analysis of endogenous steroid hormones in human hair. J. Steroid Biochem. Mol. Biol. 2016, 162, 92–99. [Google Scholar] [CrossRef]
- Gaudl, A.; Kratzsch, J.; Bae, Y.J.; Kiess, W.; Thiery, J.; Ceglarek, U. Liquid chromatography quadrupole linear ion trap mass spectrometry for quantitative steroid hormone analysis in plasma, urine, saliva and hair. J. Chromatogr. A 2016, 1464, 64–71. [Google Scholar] [CrossRef]
- Thomson, S.; Koren, G.; Fraser, L.A.; Rieder, M.; Friedman, T.C.; Van Uum, S.H. Hair analysis provides a historical record of cortisol levels in Cushing’s syndrome. Exp. Clin. Endocrinol. Diabetes 2010, 118, 133–138. [Google Scholar] [CrossRef]
- Hassanien, A.E.; Tharwat, A.; Own, H.S. Computational model for vitamin D deficiency using hair mineral analysis. Comput. Biol. Chem. 2017, 70, 198–210. [Google Scholar] [CrossRef] [PubMed]
- Jeruszka-Bielak, M.; Brzozowska, A. Relationship between nutritional habits and hair calcium levels in young women. Biol. Trace Elem. Res. 2011, 144, 63–76. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wacker, M.; Holick, M.F. Sunlight and Vitamin D: A global perspective for health. Dermatoendocrinol 2013, 5, 51–108. [Google Scholar] [CrossRef] [PubMed]
- Tridico, S.R.; Koch, S.; Michaud, A.; Thomson, G.; Kirkbride, K.P.; Bunce, M. Interpreting biological degradative processes acting on mammalian hair in the living and the dead: Which ones are taphonomic? Proc. Biol. Sci. 2014, 281, 20141755. [Google Scholar] [CrossRef]
- Yuen, A.W.; Jablonski, N.G. Vitamin D: In the evolution of human skin colour. Med. Hypotheses 2010, 74, 39–44. [Google Scholar] [CrossRef]
- Handel, I.; Watt, K.A.; Pilkington, J.G.; Pemberton, J.M.; Macrae, A.; Scott, P.; McNeilly, T.N.; Berry, J.L.; Clements, D.N.; Nussey, D.H.; et al. Vitamin D status predicts reproductive fitness in a wild sheep population. Sci. Rep. 2016, 6, 18986. [Google Scholar] [CrossRef]
- Lutsey, P.L.; Eckfeldt, J.H.; Ogagarue, E.R.; Folsom, A.R.; Michos, E.D.; Gross, M. The 25-hydroxyvitamin D3 C-3 epimer: Distribution, correlates, and reclassification of 25-hydroxyvitamin D status in the population-based Atherosclerosis Risk in Communities Study (ARIC). Clin. Chim. Acta 2015, 442, 75–81. [Google Scholar] [CrossRef]
| Subject | Gender | Age | Hair Color | BMI | Circulating 25OHD (nmol/L) |
|---|---|---|---|---|---|
| 1 | Female | 37 | Brown | 22.5 | 72 |
| 2 | Male | 64 | Grey | 21.5 | 76 |
| 3 | Male | 33 | Ginger | 24.9 | 78 |
| Sample ID | Subject | Sample | Segment | Weight (mg) | LC-MS/MS | Approximated Sample Conc. (pg/mg) |
|---|---|---|---|---|---|---|
| 1 | 1 (L.Z.) | hair | 1 (root) | 27 | ND | ND |
| 2 | 2 | 27 | ND | ND | ||
| 3 | 3 | 29 | + | 20.3 | ||
| 4 | 4 | 25 | ND | ND | ||
| 5 | 5 | 23 | ND | ND | ||
| 6 | 6 | 20 | ND | ND | ||
| 7 | 7 | 24 | + | 16.2 | ||
| 8 | 8 | 26 | + | 13.6 | ||
| 9 | 9 | 30 | + | 30.9 | ||
| 10 | 10 | 40 | ND | ND | ||
| 11 | 11 (end) | 44 | + | 11.9 | ||
| 12 | 2 (M.H.) | hair | 1 (root) | 35 | + | 433 |
| 13 | 2 | 35 | + | 26.5 | ||
| 14 | 3 | 30 | + | 315 | ||
| 15 | 4 (end) | 20 | + | 911 | ||
| 16 | 3 (E.L.) | beard | 1 | 35 | + | 231 |
| 17 | Blank | - | 0 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zgaga, L.; Laird, E.; Healy, M. 25-Hydroxyvitamin D Measurement in Human Hair: Results from a Proof-of-Concept study. Nutrients 2019, 11, 423. https://doi.org/10.3390/nu11020423
Zgaga L, Laird E, Healy M. 25-Hydroxyvitamin D Measurement in Human Hair: Results from a Proof-of-Concept study. Nutrients. 2019; 11(2):423. https://doi.org/10.3390/nu11020423
Chicago/Turabian StyleZgaga, Lina, Eamon Laird, and Martin Healy. 2019. "25-Hydroxyvitamin D Measurement in Human Hair: Results from a Proof-of-Concept study" Nutrients 11, no. 2: 423. https://doi.org/10.3390/nu11020423
APA StyleZgaga, L., Laird, E., & Healy, M. (2019). 25-Hydroxyvitamin D Measurement in Human Hair: Results from a Proof-of-Concept study. Nutrients, 11(2), 423. https://doi.org/10.3390/nu11020423







