Blueberries Improve Pain, Gait Performance, and Inflammation in Individuals with Symptomatic Knee Osteoarthritis
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Recruiting, Inclusion/Exclusion Criteria
2.3. Intervention
2.4. Pain and Gait Assessment
2.5. Biochemical Variables
2.6. Statistical Analysis
3. Results
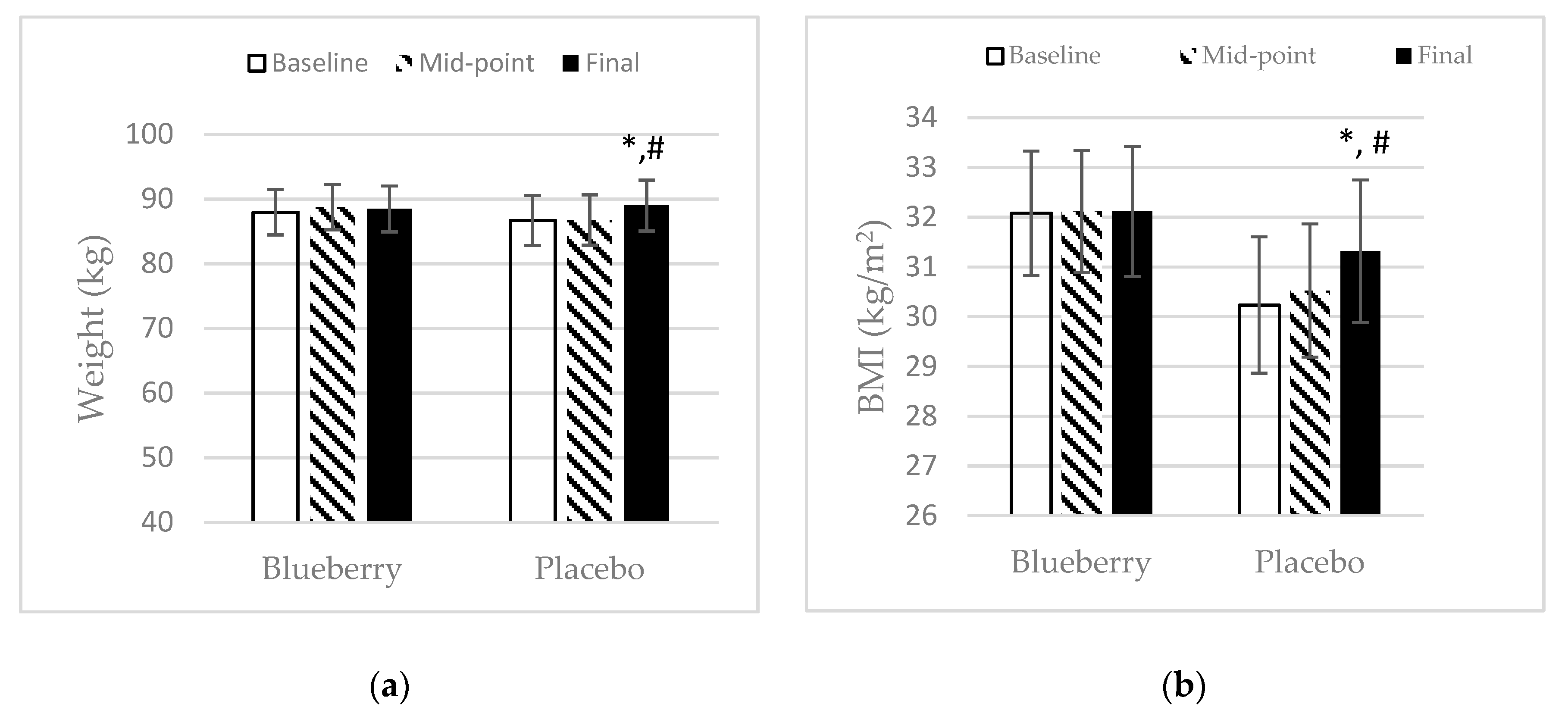
3.1. Demographics
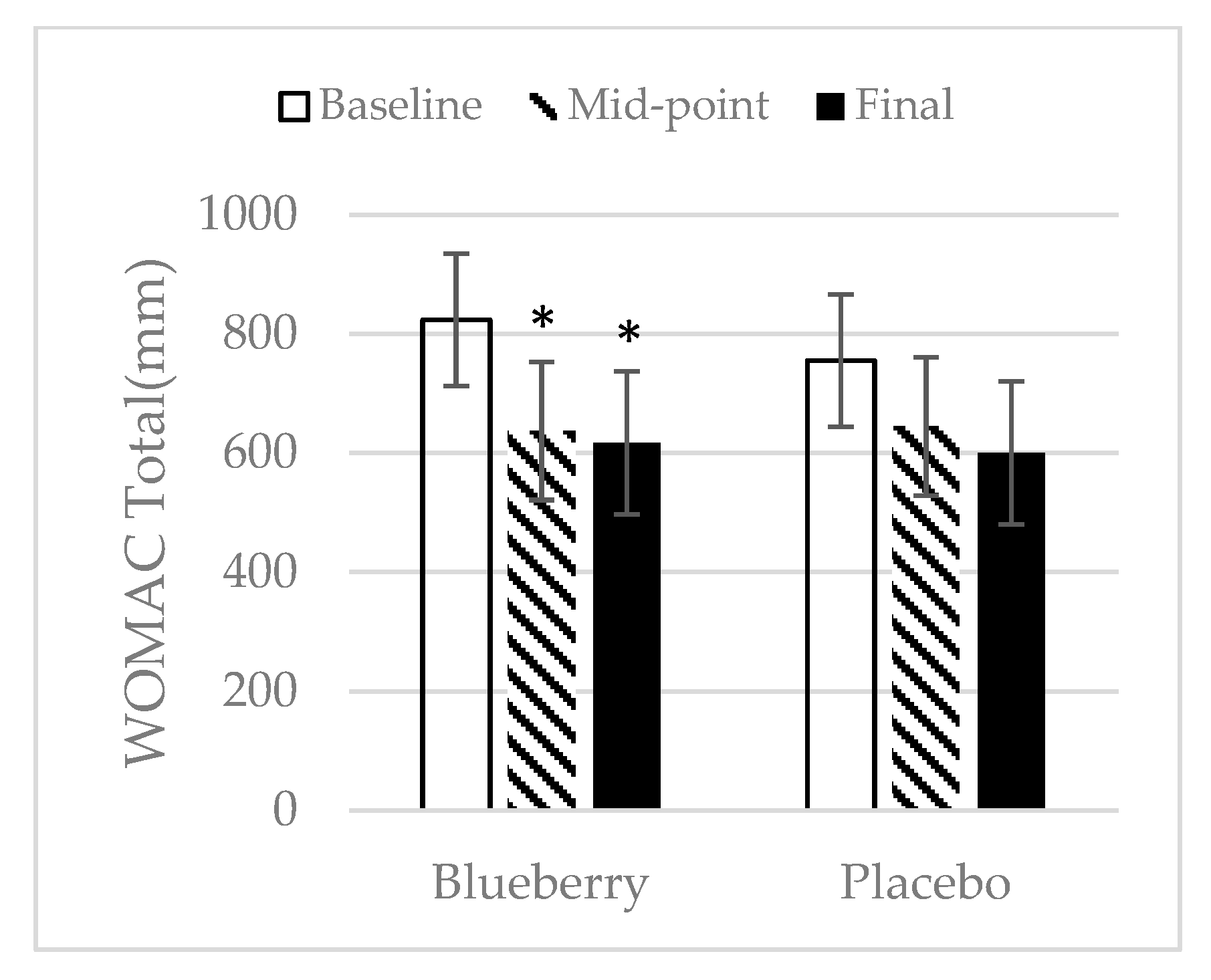
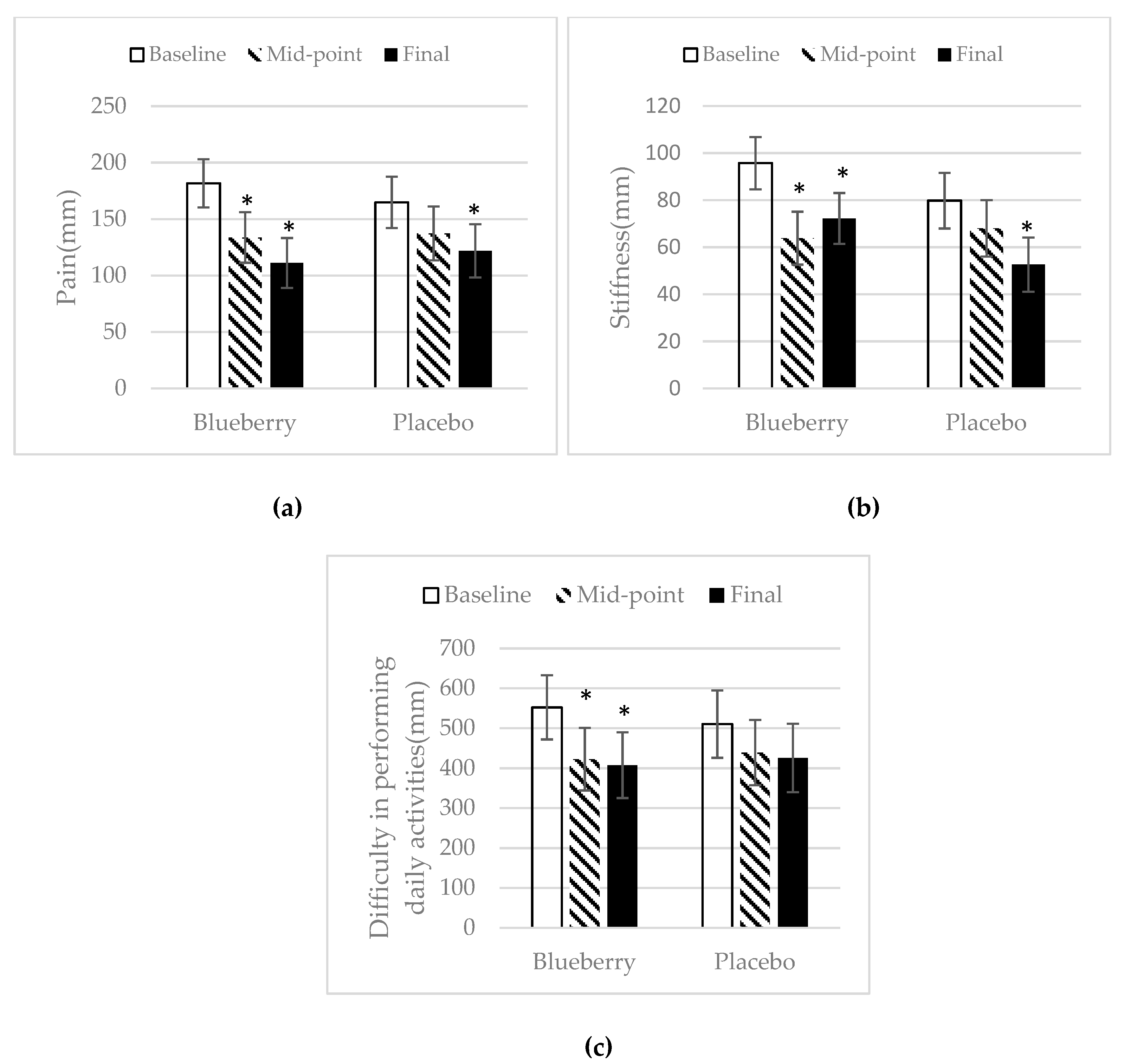
3.2. WOMAC
3.3. Gait Performance
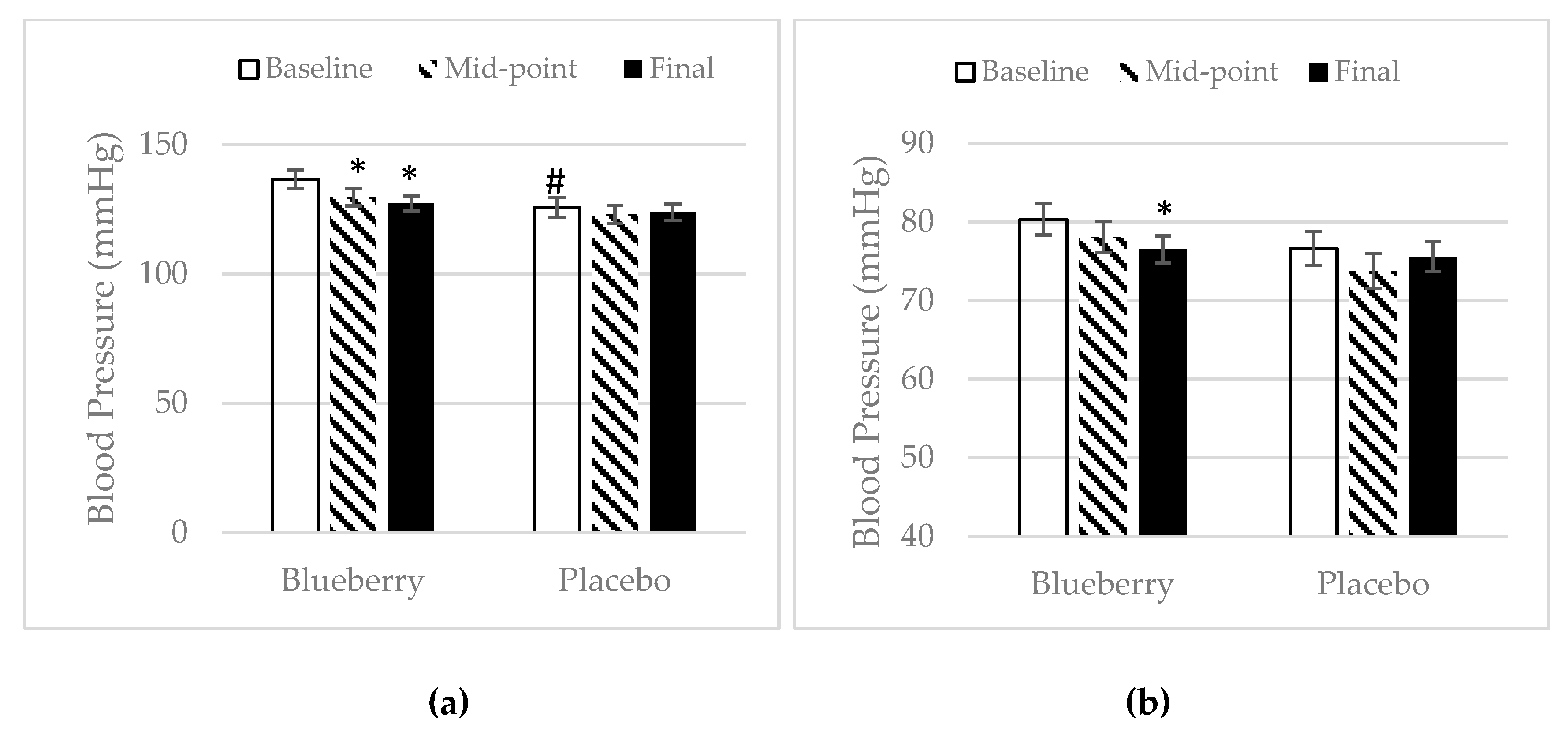
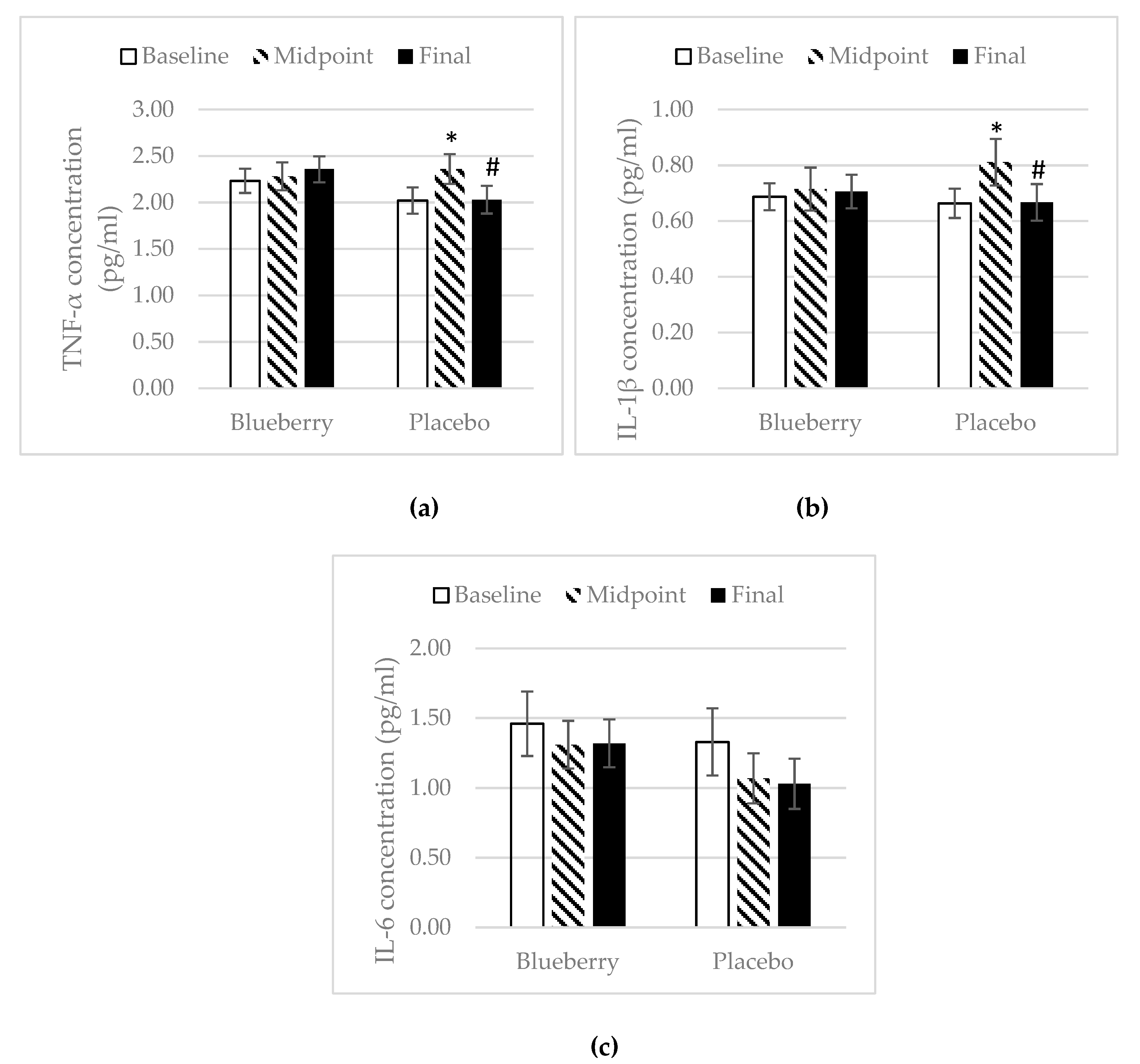
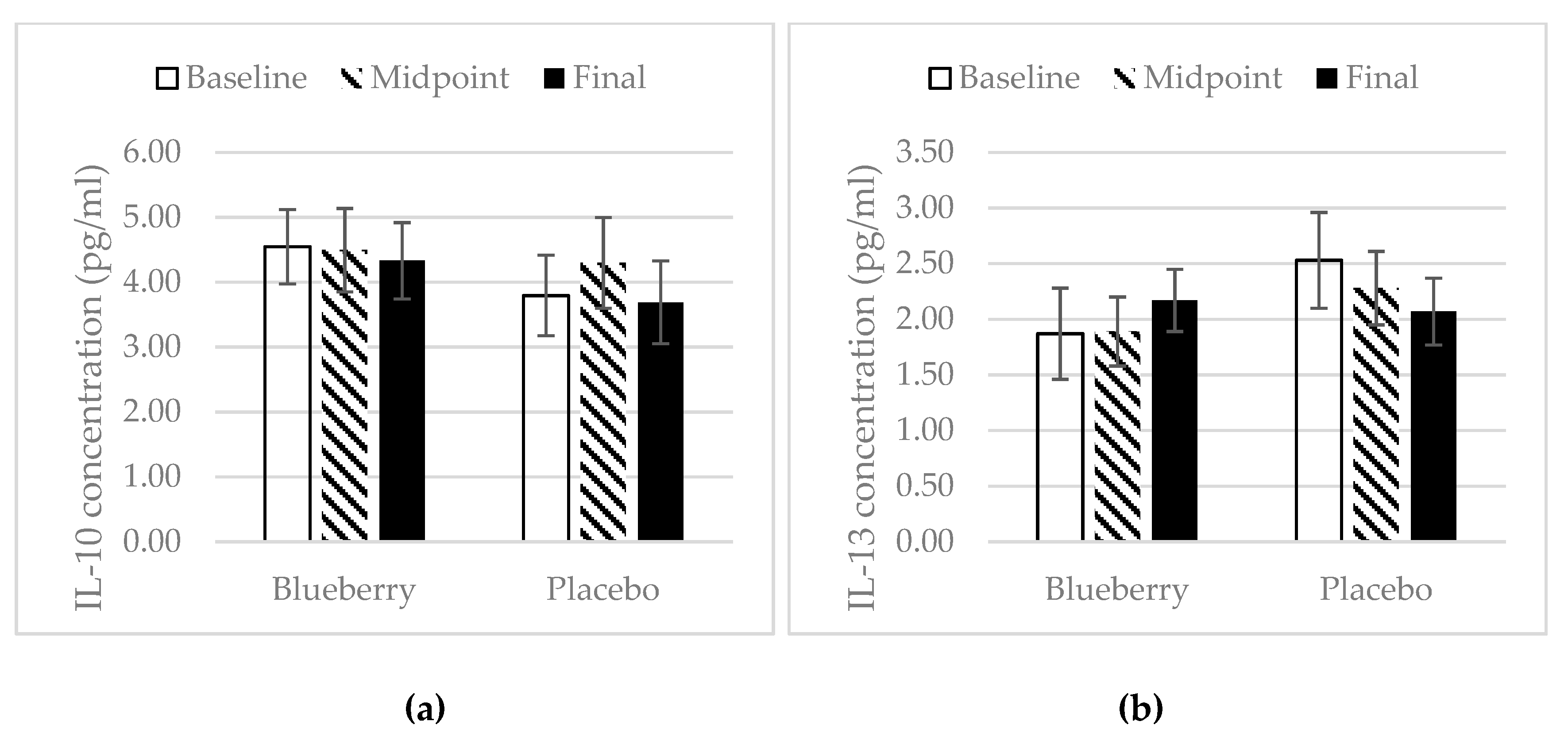
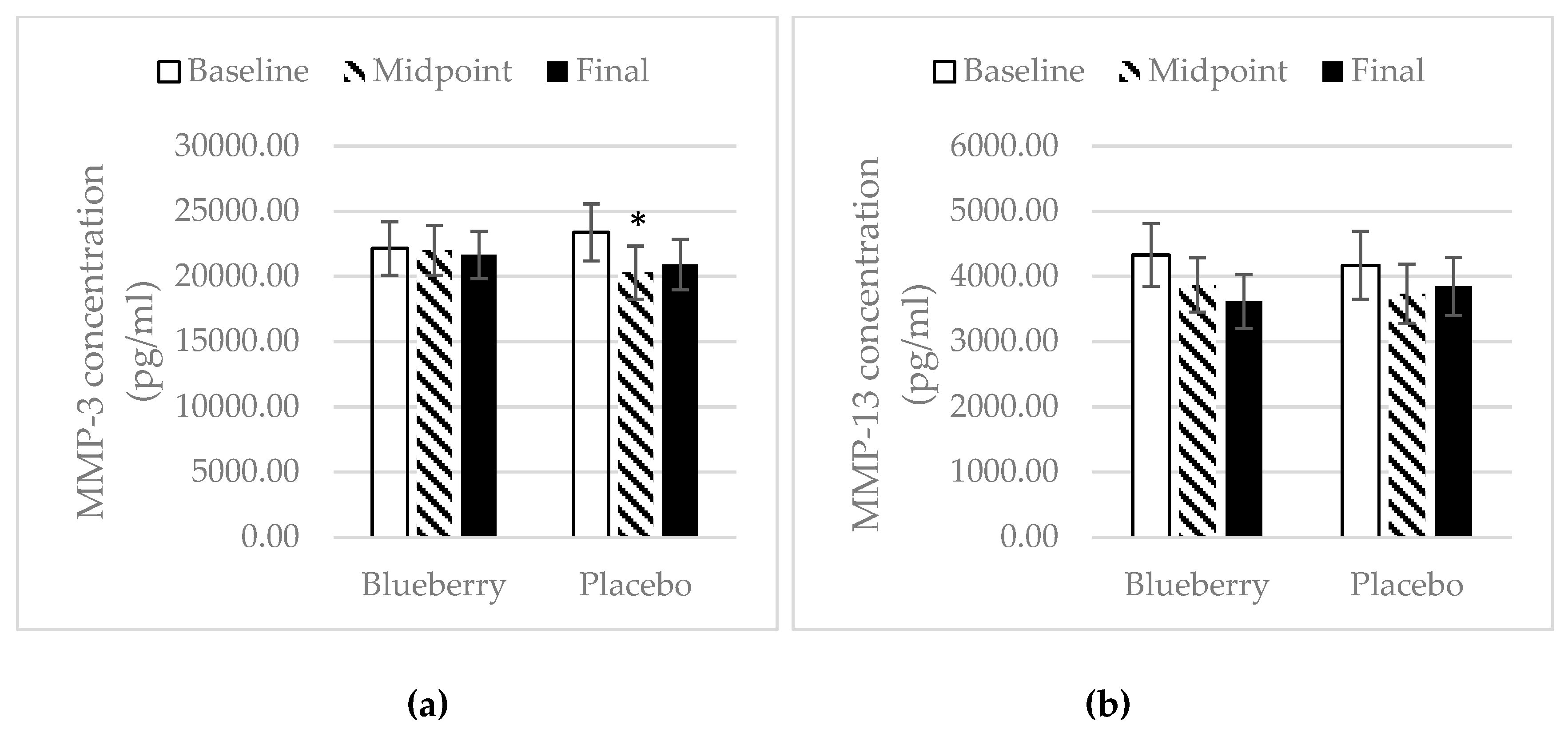
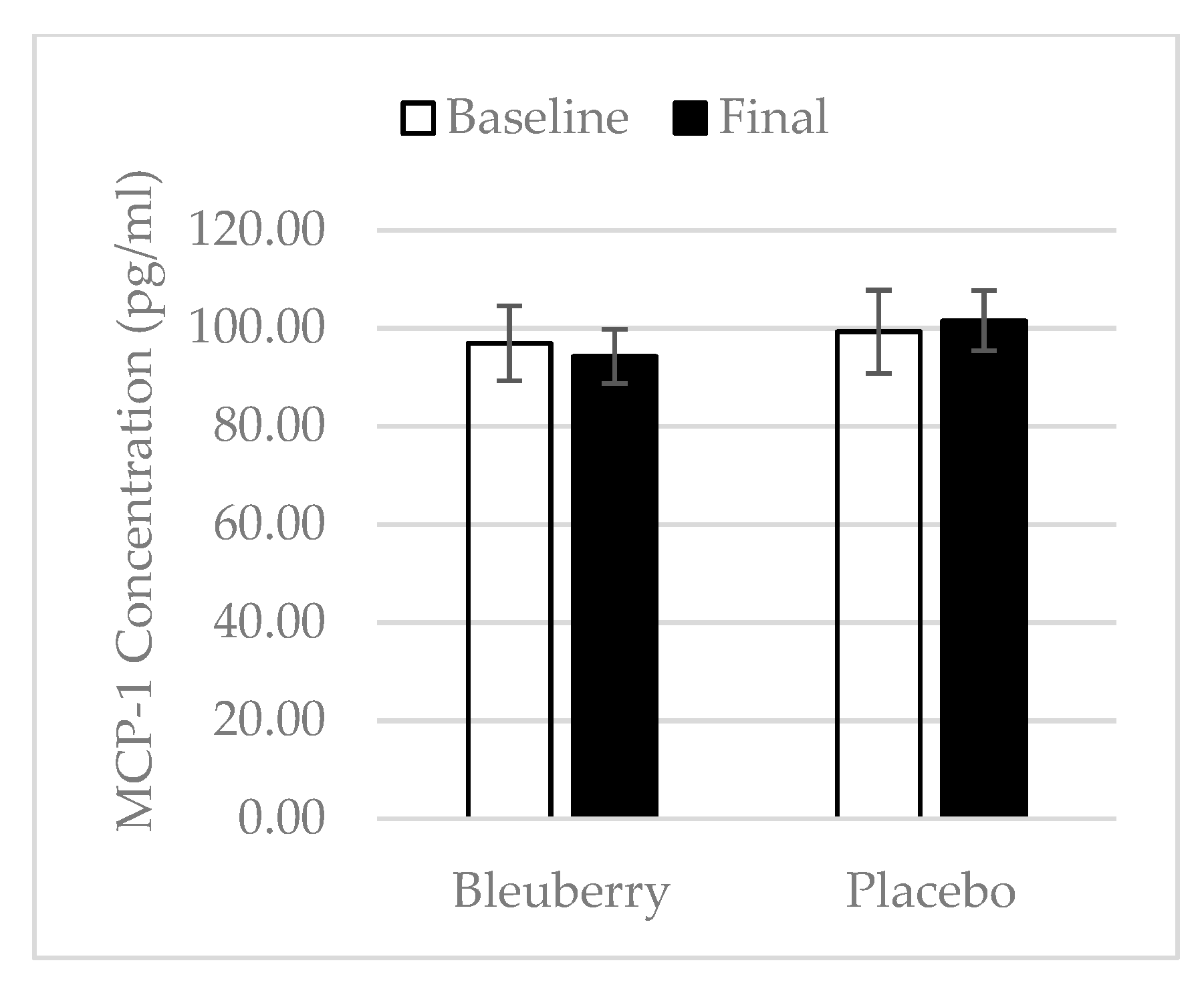
3.4. Inflammation
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Felson, D.T.; Lawrence, R.C.; Dieppe, P.A.; Hirsch, R.; Helmick, C.G.; Jordan, J.M.; Kington, R.S.; Lane, N.E.; Nevitt, M.C.; Zhang, Y.; et al. Osteoarthritis: New insights. Part 1: The disease and its risk factors. Ann. Intern. Med. 2000, 133, 635–646. [Google Scholar] [CrossRef] [PubMed]
- Escott-Stump, S. Osteoarthritis and Degrative Joint Disease. In Nutrition and Diagnosis-Related Care, 7th ed.; Lippincott Williams & Wilkins, a Wolters Kluwer Business: Baltimore, MA, USA, 2012; p. 658. ISBN 978-1-60831-017-3. [Google Scholar]
- Stefanik, J.J.; Gross, K.D.; Guermazi, A.; Felson, D.T.; Roemer, F.W.; Niu, J.; Lynch, J.A.; Segal, N.A.; Lewis, C.E.; Lewis, C.L.; et al. Relation of Step Length to Magnetic Resonance Imaging-Detected Structural Damage in the Patellofemoral Joint: The Multicenter Osteoarthritis Study. Arthr. Care Res. (Hoboken) 2016, 68, 776–783. [Google Scholar] [CrossRef]
- Barbour, K.E. Vital Signs: Prevalence of Doctor-Diagnosed Arthritis and Arthritis-Attributable Activity Limitation-United States, 2013–2015. MMWR Morb. Mortal Wkly. Rep. 2017, 66, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Suri, P.; Morgenroth, D.C.; Hunter, D.J. Epidemiology of osteoarthritis and associated comorbidities. PM R 2012, 4, S10–S19. [Google Scholar] [CrossRef]
- Lee, K.M.; Chung, C.Y.; Sung, K.H.; Lee, S.Y.; Won, S.H.; Kim, T.G.; Choi, Y.; Kwon, S.S.; Kim, Y.H.; Park, M.S. Risk Factors for Osteoarthritis and Contributing Factors to Current Arthritic Pain in South Korean Older Adults. Yonsei Med. J. 2015, 56, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Bannuru, R.R.; Schmid, C.H.; Kent, D.M.; Vaysbrot, E.E.; Wong, J.B.; McAlindon, T.E. Comparative effectiveness of pharmacologic interventions for knee osteoarthritis: A systematic review and network meta-analysis. Ann. Intern. Med. 2015, 162, 46–54. [Google Scholar] [CrossRef]
- Nelson, A.E.; Allen, K.D.; Golightly, Y.M.; Goode, A.P.; Jordan, J.M. A systematic review of recommendations and guidelines for the management of osteoarthritis: The chronic osteoarthritis management initiative of the U.S. bone and joint initiative. Semin. Arthritis Rheum. 2014, 43, 701–712. [Google Scholar] [CrossRef] [PubMed]
- Bjarnason, I.; Hayllar, J.; MacPherson, A.J.; Russell, A.S. Side effects of nonsteroidal anti-inflammatory drugs on the small and large intestine in humans. Gastroenterology 1993, 104, 1832–1847. [Google Scholar] [CrossRef]
- Borer, J.S.; Simon, L.S. Cardiovascular and gastrointestinal effects of COX-2 inhibitors and NSAIDs: Achieving a balance. Arthritis Res. Ther. 2005, 7, S14–S22. [Google Scholar] [CrossRef]
- Pandey, K.B.; Rizvi, S.I. Plant polyphenols as dietary antioxidants in human health and disease. Oxid. Med. Cell Longev. 2009, 2, 270–278. [Google Scholar] [CrossRef]
- Clifford, M.N. Chlorogenic acids and other cinnamates—Nature, occurrence and dietary burden. J. Sci. Food Agric. 1999, 79, 362–372. [Google Scholar] [CrossRef]
- Dave, M.; Attur, M.; Palmer, G.; Al-Mussawir, H.E.; Kennish, L.; Patel, J.; Abramson, S.B. The antioxidant resveratrol protects against chondrocyte apoptosis via effects on mitochondrial polarization and ATP production. Arthritis Rheum. 2008, 58, 2786–2797. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.S.; Tseng, C.Y.; Lee, C.H.; Su, S.L.; Lee, H.S. Effects of (−)-epigallocatechin-3-gallate on cyclooxygenase 2, PGE2, and IL-8 expression induced by IL-1β in human synovial fibroblasts. Rheumatol. Int. 2009, 30, 1197–1203. [Google Scholar] [CrossRef] [PubMed]
- Csaki, C.; Mobasheri, A.; Shakibaei, M. Synergistic chondroprotective effects of curcumin and resveratrol in human articular chondrocytes: Inhibition of IL-1beta-induced NF-kappaB-mediated inflammation and apoptosis. Arthritis Res. Ther. 2009, 11, R165. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Wang, N.; Hafeez, B.B.; Cheruvu, V.K.; Haqqi, T.M. Punica granatum L. extract inhibits IL-1beta-induced expression of matrix metalloproteinases by inhibiting the activation of MAP kinases and NF-kappaB in human chondrocytes in vitro. J. Nutr. 2005, 135, 2096–2102. [Google Scholar] [CrossRef] [PubMed]
- Rasheed, Z.; Akhtar, N.; Haqqi, T.M. Pomegranate extract inhibits the interleukin-1β-induced activation of MKK-3, p38α-MAPK and transcription factor RUNX-2 in human osteoarthritis chondrocytes. Arthritis Res. Ther. 2010, 12, R195. [Google Scholar] [CrossRef]
- Wang, J.; Gao, J.S.; Chen, J.W.; Li, F.; Tian, J. Effect of resveratrol on cartilage protection and apoptosis inhibition in experimental osteoarthritis of rabbit. Rheumatol. Int. 2012, 32, 1541–1548. [Google Scholar] [CrossRef] [PubMed]
- Belcaro, G.; Cesarone, M.R.; Dugall, M.; Pellegrini, L.; Ledda, A.; Grossi, M.G.; Togni, S.; Appendino, G. Efficacy and safety of Meriva®, a curcumin-phosphatidylcholine complex, during extended administration in osteoarthritis patients. Altern. Med. Rev. 2010, 15, 337–344. [Google Scholar]
- Panahi, Y.; Hosseini, M.S.; Khalili, N.; Naimi, E.; Soflaei, S.S.; Majeed, M.; Sahebkar, A. Effects of supplementation with curcumin on serum adipokine concentrations: A randomized controlled trial. Nutrition 2016, 32, 1116–1122. [Google Scholar] [CrossRef]
- Ayral, X.; Pickering, E.H.; Woodworth, T.G.; Mackillop, N.; Dougados, M. Synovitis: A Potential Predictive Factor of Structural Progression of Medial Tibiofemoral Knee Osteoarthritis—Results of a 1 Year Longitudinal Arthroscopic Study in 422 Patients. Osteoarthr. Cartil. 2005, 13, 361–367. [Google Scholar] [CrossRef]
- Krasnokutsky, S.; Belitskaya-Lévy, I.; Bencardino, J.; Samuels, J.; Attur, M.; Regatte, R.; Rosenthal, P.; Greenberg, J.; Schweitzer, M.; Abramson, S.B.; et al. Quantitative MRI Evidence of Synovial Proliferation Is Associated with Radiographic Severity of Knee Osteoarthritis. Arthritis Rheum. 2011, 63, 2983–2991. [Google Scholar] [CrossRef]
- Doss, F.; Menard, J.; Hauschild, M.; Kreutzer, H.-J.; Mittlmeier, T.; Müller-Steinhardt, M.; Müller, B. Elevated IL-6 Levels in the Synovial Fluid of Osteoarthritis Patients Stem from Plasma Cells. Scand. J. Rheumatol. 2007, 36, 136–139. [Google Scholar] [CrossRef]
- Mabey, T.; Honsawek, S. Cytokines as Biochemical Markers for Knee Osteoarthritis. World J. Orthop. 2015, 6, 95–105. [Google Scholar] [CrossRef] [PubMed]
- Rose, B.J.; Kooyman, D.L. A Tale of Two Joints: The Role of Matrix Metalloproteases in Cartilage Biology. Dis. Mark. 2016. [Google Scholar] [CrossRef] [PubMed]
- Sokolove, J.; Lepus, C.M. Role of Inflammation in the Pathogenesis of Osteoarthritis: Latest Findings and Interpretations. Ther. Adv. Musculoskelet. Dis. 2013, 5, 77–94. [Google Scholar] [CrossRef] [PubMed]
- Pozgan, U.; Caglic, D.; Rozman, B.; Nagase, H.; Turk, V.; Turk, B. Expression and Activity Profiling of Selected Cysteine Cathepsins and Matrix Metalloproteinases in Synovial Fluids from Patients with Rheumatoid Arthritis and Osteoarthritis. Biol. Chem. 2010, 391, 571–579. [Google Scholar] [CrossRef]
- Jansen, N.W.D.; Roosendaal, G.; Hooiveld, M.J.J.; Bijlsma, J.W.J.; Roon, J.A.G.V.; Theobald, M.; Lafeber, F.P.J.G. Interleukin-10 Protects against Blood-Induced Joint Damage. Br. J. Haematol. 2008, 142, 953–961. [Google Scholar] [CrossRef]
- Hart, P.H.; Ahern, M.J.; Smith, M.D.; Finlay-Jones, J.J. Regulatory Effects of IL-13 on Synovial Fluid Macrophages and Blood Monocytes from Patients with Inflammatory Arthritis. Clin. Exp. Immunol. 1995, 99, 331–337. [Google Scholar] [CrossRef]
- USDA Special Interest Databases on Flavonoids: USDA ARS. Available online: https://www.ars.usda.gov/northeast-area/beltsville-md-bhnrc/beltsville-human-nutrition-research-center/nutrient-data-laboratory/docs/usda-special-interest-databases-on-flavonoids/ (accessed on 7 October 2018).
- Jones, D.R. Blueberry polyphenols and their effects on bone and joint. Adv. Food Technol. Nutr. Sci. Open J. 2016, 2, S27–S33. [Google Scholar] [CrossRef]
- Trzeciakiewicz, A.; Habauzit, V.; Horcajada, M.N. When nutrition interacts with osteoblast function: Molecular mechanisms of polyphenols. Nutr. Res. Rev. 2009, 22, 68–81. [Google Scholar] [CrossRef]
- Horcajada, M.N.; Offord, E. Naturally plant-derived compounds: Role in bone anabolism. Curr. Mol. Pharmacol. 2012, 5, 205–218. [Google Scholar] [CrossRef]
- McAnulty, L.S.; Nieman, D.C.; Dumke, C.L.; Shooter, L.A.; Henson, D.A.; Utter, A.C.; Milne, G.; McAnulty, S.R. Effect of blueberry ingestion on natural killer cell counts, oxidative stress, and inflammation prior to and after 2.5 h of running. Appl. Physiol. Nutr. Metab. 2011, 36, 976–984. [Google Scholar] [CrossRef]
- McLeay, Y.; Barnes, M.J.; Mundel, T.; Hurst, S.M.; Hurst, R.D.; Stannard, S.R. Effect of New Zealand blueberry consumption on recovery from eccentric exercise-induced muscle damage. J. Int. Soc. Sports Nutr. 2012, 9, 19. [Google Scholar] [CrossRef] [PubMed]
- Schrager, M.A.; Hilton, J.; Gould, R.; Kelly, V.E. Effects of blueberry supplementation on measures of functional mobility in older adults. Appl. Physiol. Nutr. Metab. 2015, 40, 543–549. [Google Scholar] [CrossRef] [PubMed]
- U.S. Highbush Blueberry Council E-Newsletter. Available online: http://www.blueberrytech.org/berrylatest/july-aug-2016.html (accessed on 4 January 2019).
- Salaffi, F.; Leardini, G.; Canesi, B.; Mannoni, A.; Fioravanti, A.; Caporali, R.; Lapadula, G.; Punzi, L. GOnorthrosis and Quality Of Life Assessment (GOQOLA). Reliability and validity of the Western Ontario and McMaster Universities (WOMAC) Osteoarthritis Index in Italian patients with osteoarthritis of the knee. Osteoarthr. Cartil. 2003, 11, 551–560. [Google Scholar] [CrossRef]
- DeCaria, J.E.; Montero-Odasso, M.; Wolfe, D.; Chesworth, B.M.; Petrella, R.J. The effect of intra-articular hyaluronic acid treatment on gait velocity in older knee osteoarthritis patients: A randomized, controlled study. Arch. Gerontol. Geriatr. 2012, 55, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Angeloni, C.; Pirola, L.; Vauzour, D.; Maraldi, T. Dietary Polyphenols and Their Effects on Cell Biochemistry and Pathophysiology. Oxid. Med. Cell. Longev. 2012. [Google Scholar] [CrossRef] [PubMed]
- Leong, D.J.; Choudhury, M.; Hanstein, R.; Hirsh, D.M.; Kim, S.J.; Majeska, R.J.; Schaffler, M.B.; Hardin, J.A.; Spray, D.C.; Goldring, M.B.; et al. Green Tea Polyphenol Treatment Is Chondroprotective, Anti-Inflammatory and Palliative in a Mouse Post-Traumatic Osteoarthritis Model. Arthritis Res. Ther. 2014, 16, 508. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, B.B.; Harikumar, K.B. Potential Therapeutic Effects of Curcumin, the Anti-Inflammatory Agent, against Neurodegenerative, Cardiovascular, Pulmonary, Metabolic, Autoimmune and Neoplastic Diseases. Int. J. Biochem. Cell Biol. 2009, 41, 40–59. [Google Scholar] [CrossRef]
- Ryan, D.J.; Islam, P.; Freeman, S.L.; Paine, S.W. The Anti-Inflammatory Effects of Resveratrol in Equine Osteoarthritis Using the Cartilage Explant Model. Equine Vet. J. 2017, 49, 13–16. [Google Scholar] [CrossRef]
- Oben, J.; Enonchong, E.; Kothari, S.; Chambliss, W.; Garrison, R.; Dolnick, D. Phellodendron and Citrus Extracts Benefit Joint Health in Osteoarthritis Patients: A Pilot, Double-Blind, Placebo-Controlled Study. Nutr. J. 2009, 8, 38. [Google Scholar] [CrossRef]
- Ghoochani, N.; Karandish, M.; Mowla, K.; Haghighizadeh, M.H.; Jalali, M.T. The effect of pomegranate juice on clinical signs, matrix metalloproteinases and antioxidant status in patients with knee osteoarthritis. J. Sci. Food Agric. 2016, 96, 4377–4381. [Google Scholar] [CrossRef] [PubMed]
- Schumacher, H.R.; Pullman-Mooar, S.; Gupta, S.R.; Dinnella, J.E.; Kim, R.; McHugh, M.P. Randomized double-andoblind crossover study of the efficacy of a tart cherry juice blend in treatment of osteoarthritis (OA) of the knee. Osteoarthr. Cartil. 2013, 21, 1035–1041. [Google Scholar] [CrossRef]
- Elbaz, A.; Mor, A.; Segal, G.; Debi, R.; Shazar, N.; Herman, A. Novel classification of knee osteoarthritis severity based on spatiotemporal gait analysis. Osteoarthr. Cartil. 2014, 22, 457–463. [Google Scholar] [CrossRef]
- Debi, R.; Mor, A.; Segal, G.; Segal, O.; Agar, G.; Debbi, E.; Halperin, N.; Haim, A.; Elbaz, A. Correlation between single limb support phase and self-evaluation questionnaires in knee osteoarthritis populations. Disabil. Rehabil. 2011, 33, 1103–1109. [Google Scholar] [CrossRef] [PubMed]
- Javaheri, B.; Poulet, B.; Aljazzar, A.; de Souza, R.; Piles, M.; Hopkinson, M.; Shervill, E.; Pollard, A.; Chan, B.; Chang, Y.-M.; et al. Stable Sulforaphane Protects against Gait Anomalies and Modifies Bone Microarchitecture in the Spontaneous STR/Ort Model of Osteoarthritis. Bone 2017, 103, 308–317. [Google Scholar] [CrossRef] [PubMed]
- Innes, J.F.; Fuller, C.J.; Grover, E.R.; Kelly, A.L.; Burn, J.F. Randomised, Double-Blind, Placebo-Controlled Parallel Group Study of P54FP for the Treatment of Dogs with Osteoarthritis. Vet. Rec. 2003, 152, 457–460. [Google Scholar] [CrossRef]
- Yuankun, X.; Yan, K.; Bin, W.; Jian-Hao, L. Monocyte chemoattractant protein 1 induced chondrocytes degeneration and cartilage degradation in osteoarthritis. Osteoar. Cartil. 2016, 24, S140–S141. [Google Scholar] [CrossRef]
- Shakibaei, M.; Mobasheri, A.; Buhrmann, C. Curcumin synergizes with resveratrol to stimulate the MAPK signaling pathway in human articular chondrocytes in vitro. Genes Nutr. 2011, 6, 171–179. [Google Scholar] [CrossRef]
- Joseph, S.V.; Edirisinghe, I.; Burton-Freeman, B.M. Fruit Polyphenols: A Review of Anti-inflammatory Effects in Humans. Crit. Rev. Food Sci. Nutr. 2016, 56, 419–444. [Google Scholar] [CrossRef]
- Crouvezier, S.; Powell, B.; Keir, D.; Yaqoob, P. The effects of phenolic components of tea on the production of pro- and anti-inflammatory cytokines by human leukocytes in vitro. Cytokine 2001, 13, 280–286. [Google Scholar] [CrossRef]
- Karlsen, A.; Retterstøl, L.; Laake, P.; Paur, I.; Bøhn, S.K.; Sandvik, L.; Blomhoff, R. Anthocyanins inhibit nuclear factor-kappaB activation in monocytes and reduce plasma concentrations of pro-inflammatory mediators in healthy adults. J. Nutr. 2007, 137, 1951–1954. [Google Scholar] [CrossRef] [PubMed]
- Panickar, K.S. Effects of dietary polyphenols on neuroregulatory factors and pathways that mediate food intake and energy regulation in obesity. Mol. Nutr. Food Res. 2013, 57, 34–47. [Google Scholar] [CrossRef] [PubMed]
- Molan, A.L.; Lila, M.A.; Mawson, J. Satiety in rats following blueberry extract consumption induced by appetite-suppressing mechanisms unrelated to in vitro or in vivo antioxidant capacity. Food Chem. 2008. Available online: http://agris.fao.org/agris-search/search.do?recordID=US201300851909 (accessed on 7 October 2018). [CrossRef]
- Johnson, S.A.; Figueroa, A.; Navaei, N.; Wong, A.; Kalfon, R.; Ormsbee, L.T.; Feresin, R.G.; Elam, M.L.; Hooshmand, S.; Payton, M.E.; et al. Daily Blueberry Consumption Improves Blood Pressure and Arterial Stiffness in Postmenopausal Women with Pre- and Stage 1-Hypertension: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. J. Acad. Nutr. Diet. 2015, 115, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Ascherio, A.; Rimm, E.B.; Giovannucci, E.L.; Spiegelman, D.; Stampfer, M.; Willett, W.C. Dietary fat and risk of coronary heart disease in men: Cohort follow up study in the United States. BMJ 1996, 313, 84–90. [Google Scholar] [CrossRef]
- Reshef, N.; Hayari, Y.; Goren, C.; Boaz, M.; Madar, Z.; Knobler, H. Antihypertensive effect of sweetie fruit in patients with stage I hypertension. Am. J. Hypertens. 2005, 18, 1360–1363. [Google Scholar] [CrossRef]
- Naruszewicz, M.; Laniewska, I.; Millo, B.; Dłuzniewski, M. Combination therapy of statin with flavonoids rich extract from chokeberry fruits enhanced reduction in cardiovascular risk markers in patients after myocardial infraction (MI). Atherosclerosis 2007, 194, e179–e184. [Google Scholar] [CrossRef]
| Baseline | Midpoint | Final | ||
|---|---|---|---|---|
| Blueberry | Male (n) | 9 | 9 | 9 |
| Female (n) | 24 | 19 | 18 | |
| Total (n) | 33 | 28 | 27 | |
| Avg Age | 57.7 ± 1.8 | 57.5 ± 1.9 | 56.4 ± 1.9 | |
| Avg BMI | 32.1 ± 1.3 | 32.1 ± 1.2 | 32.1 ± 1.3 | |
| Placebo | Male (n) | 7 | 5 | 5 |
| Female (n) | 23 | 18 | 17 | |
| Total (n) | 30 | 23 | 22 | |
| Avg Age | 55.3 ± 1.5 | 54.5 ± 1.7 | 54.6 ± 1.8 | |
| Avg BMI | 30.2 ± 1.4 | 30.5 ± 1.3 | 31.3 ± 1.4 | |
| Total | Male (n) | 16 | 14 | 14 |
| Female (n) | 47 | 37 | 35 | |
| Total (n) | 63 | 51 | 49 | |
| Avg Age | 56.5 ± 1.2 | 56.2 ± 1.3 | 55.6 | |
| Avg BMI | 31.9 ± 0.8 | 31.6 ± 0.9 | 32.2 ± 0.9 | |
| Drop Rate (%) | NA | 19% | 22% | |
| Blueberry | Placebo | |||||
|---|---|---|---|---|---|---|
| Gait Parameters | Baseline | Midpoint | Final | Baseline | Midpoint | Final |
| Normal cadence (steps/min) | 103.7 ± 2.4 | 111.3 ± 2.1 * | 114.2 ± 2.46 * | 108.4 ± 2.7 | 113.9 ± 2.3 * | 117.2 ± 2.7 * |
| Fast cadence (steps/min) | 144.0 ± 4.7 | 146.4 ± 3.8 | 146.0 ± 2.5 | 152.1 ± 5.2 | 149.3 ± 4.2 | 156.0 ± 4.7 # |
| Normal velocity (cm/s) | 96.3 ± 4.12 | 108.1 ± 3.8 * | 115.3 ± 4.4 *,# | 105.3 ± 4.6 | 114.9 ± 4.1 * | 119.0 ± 4.9 * |
| Fast velocity (cm/s) | 163.8 ± 8.2 | 165.2 ± 6.7 | 163.4 ± 7.0 | 176.7 ± 9.1 | 170.7 ± 7.4 | 179.8 ± 7.7 # |
| Left step length normal (cm) | 55.1 ± 1.6 | 58.0 ± 1.5 * | 60.0 ± 1.5 *,# | 58.1 ± 1.8 | 60.2 ± 1.6 * | 60.8 ± 1.7 * |
| Left step length fast (cm) | 67.5 ± 2.0 | 67.2 ± 1.9 | 66.9 ± 1.8 | 69.1 ± 2.3 | 68.3 ± 2.2 | 68.7 ± 2.0 |
| Right step length normal (cm) | 54.9 ± 1.6 | 58.1 ± 1.5 * | 60.4 ± 1.6 *,# | 58.1 ± 1.8 | 60.8 ± 1.7 * | 60.7 ± 1.8 * |
| Right step length fast (cm) | 67.2 ± 2.01 | 67.3 ± 1.8 | 66.5 ± 1.9 | 70.0 ± 2.3 | 69.3 ± 2.0 | 69.6 ± 2.1 |
| Left stride length normal (cm) | 110.2 ± 3.1 | 116.3 ± 2.9 * | 120.7 ± 3.0 *,# | 116.4 ± 3.5 | 121.0 ± 3.2 * | 121.7 ± 3.4 * |
| Left stride length fast (cm) | 134.9 ± 4.0 | 134.7 ± 3.6 | 133.6 ± 3.6 | 139.5 ± 4.4 | 137.4 ± 4.0 | 138.3 ± 4.0 |
| Right stride length normal (cm) | 110.2 ± 3.2 | 116.4 ± 2.9 * | 120.6 ± 3.1 *,# | 116.3 ± 3.5 | 121.1 ± 3.2 * | 121.6 ± 3.4 *,# |
| Right stride length fast (cm) | 134.8 ± 4.0 | 134.8 ± 3.7 | 133.7 ± 3.7 | 139.5 ± 4.5 | 137.8 ± 4.1 | 138.9 ± 4.1 |
| Left single support normal (%) | 35.3 ± 0.6 | 36.3 ± 0.5 * | 37.0 ± 0.6 *,# | 36.8 ± 0.6 | 37.6 ± 0.5 * | 37.6 ± 0.5 * |
| Left single support fast (%) | 40.4 ± 0.6 | 40.1 ± 0.6 | 40.1 ± 0.6 | 40.8 ± 0.7 | 40.3 ± 0.6 | 41.1 ± 0.6 # |
| Right single support normal (%) | 35.3 ± 0.5 | 36.6 ± 0.4 * | 37.1 ± 0.4 * | 36.9 ± 0.6 | 37.4 ± 0.4 | 37.6 ± 0.5 |
| Right single support fast (%) | 40.1 ± 0.6 | 40.4 ± 0.6 | 40.1 ± 0.58 | 41.0 ± 0.7 | 40.7 ± 0.6 | 40.8 ± 0.6 |
| Left double support normal (%) | 29.5 ± 1.0 | 27.2 ± 0.8 * | 26.1 ± 0.8 *,# | 26.6 ± 1.1 | 25.2 ± 0.9 * | 25.3 ± 0.9 |
| Left double support fast (%) | 20.0 ± 1.2 | 19.3 ± 1.1 | 19.7 ± 1.1 | 18.1 ± 1.3 | 19.1 ± 1.2 | 17.7 ± 1.3 # |
| Right double support normal (%) | 29.5 ± 1.0 | 27.3 ± 0.8 * | 26.1 ± 0.8 *,# | 26.5 ± 1.1 | 25.3 ± 0.9 * | 25.3 ± 0.9 |
| Right double support fast (%) | 19.8 ± 1.2 | 19.4 ± 1.1 | 19.8 ± 1.1 | 18.3 ± 1.33 | 19.2 ± 1.2 | 18.9 ± 1.2 # |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Du, C.; Smith, A.; Avalos, M.; South, S.; Crabtree, K.; Wang, W.; Kwon, Y.-H.; Vijayagopal, P.; Juma, S. Blueberries Improve Pain, Gait Performance, and Inflammation in Individuals with Symptomatic Knee Osteoarthritis. Nutrients 2019, 11, 290. https://doi.org/10.3390/nu11020290
Du C, Smith A, Avalos M, South S, Crabtree K, Wang W, Kwon Y-H, Vijayagopal P, Juma S. Blueberries Improve Pain, Gait Performance, and Inflammation in Individuals with Symptomatic Knee Osteoarthritis. Nutrients. 2019; 11(2):290. https://doi.org/10.3390/nu11020290
Chicago/Turabian StyleDu, Chen, Amy Smith, Marco Avalos, Sanique South, Keith Crabtree, Wanyi Wang, Young-Hoo Kwon, Parakat Vijayagopal, and Shanil Juma. 2019. "Blueberries Improve Pain, Gait Performance, and Inflammation in Individuals with Symptomatic Knee Osteoarthritis" Nutrients 11, no. 2: 290. https://doi.org/10.3390/nu11020290
APA StyleDu, C., Smith, A., Avalos, M., South, S., Crabtree, K., Wang, W., Kwon, Y.-H., Vijayagopal, P., & Juma, S. (2019). Blueberries Improve Pain, Gait Performance, and Inflammation in Individuals with Symptomatic Knee Osteoarthritis. Nutrients, 11(2), 290. https://doi.org/10.3390/nu11020290






