Daily Green Tea Infusions in Hypercalciuric Renal Stone Patients: No Evidence for Increased Stone Risk Factors or Oxalate-Dependent Stones
Abstract
1. Introduction
2. Material and Methods
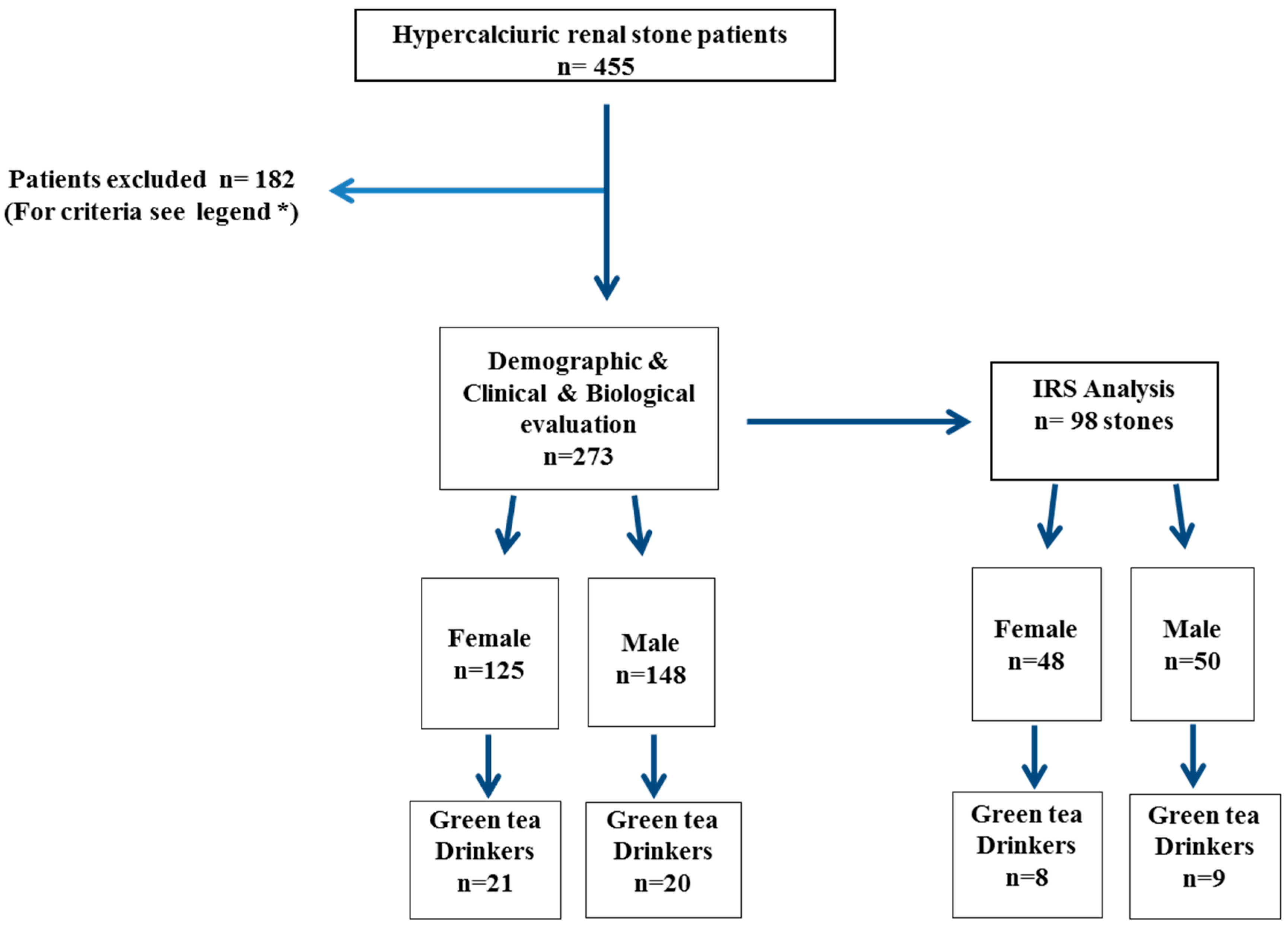
2.1. Population
2.2. Statistical Analyses
3. Results
3.1. Demographic and Clinical Data
3.2. Diet, Metabolic, and Urinary Stone Risk Factors
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| BMI | body mass index |
| COM | calcium oxalate monohydrate |
| COD | calcium oxalate dihydrate |
| CaOx | Calcium oxalate |
| EGC | epigallocatechin |
| IRS analysis | infrared spectroscopy analysis |
| MAP | mean arterial pressure |
| NS | not significant |
| SWL | shock wave lithotripsy |
| URS | flexible ureteroscopy |
| RSS | relative super saturation |
References
- Daudon, M.; Knebelmann, B. Epidemiology of urolithiasis. Rev. Prat. 2011, 61, 372–378. [Google Scholar]
- Mahdavi, R.; Lofti Yagin, N.; Liebman, M.; Nikniaz, Z. Effect of different brewing times on soluble oxalate content of loose-packed black teas and tea bags. Urolithiasis 2013, 41, 15–19. [Google Scholar] [CrossRef]
- Hönow, R.; Gu, K.L.R.; Hesse, A.; Siener, R. Oxalate content of green tea of different origin, quality, preparation and time of harvest. Urol. Res. 2010, 38, 377–381. [Google Scholar] [CrossRef]
- Charrier, M.J.; Savage, G.P.; Vanhanen, L. Oxalate content and calcium binding capacity of tea and herbal teas. Asia Pac. J. Clin. Nutr. 2002, 11, 298–301. [Google Scholar] [CrossRef]
- Lotito, S.B.; Frei, B. Consumption of flavonoid-rich foods and increased plasma antioxidant capacity in humans: Cause, consequence, or epiphenomenon? Free Radic. Biol. Med. 2006, 41, 1727–1746. [Google Scholar] [CrossRef]
- Jin, Y.; Jin, C.H.; Row, K.H. Separation of catechin compounds from different teas. Biotechnol. J. 2006, 1, 209–213. [Google Scholar] [CrossRef]
- Neveu, V.; Perez-Jiménez, J.; Vos, F.; Crespy, V.; Du Chaffaut, L.; Mennen, L.; Knox, C.; Eisner, R.; Cruz, J.; Wishart, D.; et al. Phenol-Explorer: An online comprehensive database on polyphenol contents in foods. Database (Oxford) 2010. [Google Scholar] [CrossRef]
- Pérez-Jiménez, J.; Neveu, V.; Vos, F.; Scalbert, A. Systematic analysis of the content of 502 polyphenols in 452 foods and beverages: An application of the phenol-explorer database. J. Agric. Food Chem. 2010, 58, 4959–4969. [Google Scholar] [CrossRef]
- Lambert, J.D.; Sang, S.; Yang, C.S. Biotransformation of green tea polyphenols and the biological activities of those metabolites. Mol. Pharm. 2007, 4, 819–825. [Google Scholar] [CrossRef]
- Sang, S.; Lambert, J.D.; Ho, C.T.; Yang, C.S. The chemistry and biotransformation of tea constituents. Pharmacol. Res. 2011, 64, 87–99. [Google Scholar] [CrossRef]
- Warden, B.A.; Smith, L.S.; Beecher, G.R.; Balentine, D.A.; Clevidence, B.A. Catechins are bioavailable in men and women drinking black tea throughout the day. J. Nutr. 2001, 131, 1731–1737. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.S.; Chen, L.; Lee, M.J.; Balentine, D.; Kuo, M.C.; Schantz, S.P. Blood and urine levels of tea catechins after ingestion of different amounts of green tea by human volunteers. Cancer Epidemiol. Biomark. Prev. 1998, 7, 351–354. [Google Scholar]
- Lee, M.J.; Maliakal, P.; Chen, L.; Meng, X.; Bondoc, F.Y.; Prabhu, S.; Lambert, G.; Mohr, S.; Yang, C.S. Pharmacokinetics of tea catechins after ingestion of green tea and (−)-epigallocatechin-3-gallate by humans: Formation of different metabolites and individual variability. Cancer Epidemiol. Biomark. Prev. 2002, 11, 1025–1032. [Google Scholar]
- Lee, M.J.; Wang, Z.Y.; Li, H.; Chen, L.; Sun, Y.; Gobbo, S.; Balentine, D.A.; Yang, C.S. Analysis of plasma and urinary tea polyphenols in human subjects. Cancer Epidemiol. Biomark. Prev. 1995, 4, 393–399. [Google Scholar]
- Henning, S.M.; Niu, Y.; Lee, N.H.; Thames, G.D.; Minutti, R.R.; Wang, H.; Go, V.L.W.; Heber, D. Bioavailability and antioxidant activity of tea flavanols after consumption of green tea, black tea, or a green tea extract supplement. Am. J. Clin. Nutr. 2004, 80, 1558–1564. [Google Scholar] [CrossRef] [PubMed]
- Chow, H.H.; Cai, Y.; Alberts, D.S.; Hakim, I.; Dorr, R.; Shahi, F.; Crowell, J.A.; Yang, C.S.; Hara, Y. Phase I pharmacokinetic study of tea polyphenols following single-dose administration of epigallocatechin gallate and polyphenon E. Cancer Epidemiol. Biomark. Prev. 2001, 10, 53–58. [Google Scholar]
- Chow, H.H.; Hakim, I.A.; Vining, D.R.; Crowell, J.A.; Ranger-Moore, J.; Chew, W.M.; Celaya, C.A.; Rodney, S.R.; Hara, Y.; Alberts, D.S. Effects of dosing condition on the oral bioavailability of green tea catechins after single-dose administration of polyphenon E in healthy individuals. Clin. Cancer Res. 2005, 11, 4627–4633. [Google Scholar] [CrossRef] [PubMed]
- Van Amelsvoort, J.M.; Van Hof, K.H.; Mathot, J.N.J.J.; Mulder, T.P.J.; Wiersma, A.; Tijburg, L.B.M. Plasma concentrations of individual tea catechins after a single oral dose in humans. Xenobiotica 2001, 31, 891–901. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Meng, X.; Winnik, B.; Lee, M.J.; Lu, H.; Sheng, S.; Buckley, B.; Yang, C.S. Analysis of urinary metabolites of tea catechins by liquid chromatography/electrospray ionization mass spectrometry. Chem. Res. Toxicol. 2001, 14, 702–707. [Google Scholar] [CrossRef]
- Baba, S.; Osakabe, N.; Natsume, M.; Muto, Y.; Takizawa, T.; Terao, J. In vivo comparison of the bioavailability of (+)-catechin, (−)-epicatechin and their mixture in orally administered rats. J. Nutr. 2001, 131, 2885–2891. [Google Scholar] [CrossRef]
- Itoh, Y.; Yasui, T.; Okada, A.; Tozawa, K.; Hayashi, Y.; Kohri, K. Preventive effects of green tea on renal stone formation and the role of oxidative stress in nephrolithiasis. J. Urol. 2005, 173, 271–275. [Google Scholar] [CrossRef] [PubMed]
- Grases, F.; Prieto, R.M.; Gomila, I.; Sanchis, P.; Costa-Bauzá, A. Phytotherapy and renal stones: The role of antioxidants. A pilot study in Wistar rats. Urol. Res. 2009, 37, 35–40. [Google Scholar] [CrossRef] [PubMed]
- Jeong, B.C.; Kim, B.S.; Kim, J.I.; Kim, H.H. Effects of green tea on urinary stone formation: An in vivo and in vitro study. J. Endourol. 2006, 20, 356–361. [Google Scholar] [CrossRef] [PubMed]
- Werness, P.G.; Brown, C.M.; Smith, L.H.; Finlayson, B. EQUIL2: A BASIC computer program for the calculation of urinary saturation. J. Urol. 1985, 134, 1242–1244. [Google Scholar] [CrossRef]
- Tiselius, H.G. Aspects on estimation of the risk of calcium oxalate crystallization in urine. Urol. Int. 1991, 47, 255–259. [Google Scholar] [CrossRef] [PubMed]
- Daudon, M.; Bader, C.A.; Jungers, P. Urinary calculi: Review of classification methods and correlations with etiology. Scanning Microsc. 1993, 7, 1081–1104. [Google Scholar] [PubMed]
- Shu, X.; Cai, H.; Xiang, Y.B.; Li, H.; Lipworth, L.; Miller, N.L.; Zheng, W.; Shu, X.-O.; Hsi, R. Green tea intake and risk of incident kidney stones: Prospective cohort studies in middle-aged and elderly Chinese individuals. Int. J. Urol. 2018. [Google Scholar] [CrossRef]
- Shen, C.L.; Chyu, M.C.; Cao, J.J.; Yeh, J.K. Green tea polyphenols improve bone microarchitecture in high-fat-diet-induced obese female rats through suppressing bone formation and erosion. J. Med. Food. 2013, 16, 421–427. [Google Scholar] [CrossRef]
- Shen, C.L.; Chyu, M.C.; Wang, J.S. Tea and bone health: Steps forward in translational nutrition. Am. J. Clin. Nutr. 2013, 98, 1694S–1699S. [Google Scholar] [CrossRef]
- Chen, Z.; Wang, C.; Zhou, H.; Sang, L.; Li, X. Modulation of calcium oxalate crystallization by commonly consumed green tea. CrystEngComm 2010, 12, 845–852. [Google Scholar] [CrossRef]
- Daudon, M.; Doré, J.C.; Jungers, P.; Lacour, B. Changes in stone composition according to age and gender of patients: A multivariate epidemiological approach. Urol. Res. 2004, 32, 241–247. [Google Scholar] [CrossRef] [PubMed]
| >Population | Female (n = 125) | Male (n = 148) | ||||
|---|---|---|---|---|---|---|
| Non-Drinkers (n = 102) | Green Tea (n = 21) | p | Non-Drinkers (n = 122) | Green Tea (n = 20) | p | |
| Age (years) | 46 (34–59) | 42 (33–53) | NS | 48 (38–58) | 44 (37–57) | NS |
| BMI (kg/m2) | 24.9 (21.4–29.4) | 23.5 (21.2–25.6) | NS | 25.7 (23.2–29.0) | 25.8 (24.5–29.6) | NS |
| MAP (mmHg) | 83.3 (76.7–92.5) | 81.7 (76.7–93.4) | NS | 90.0 (80.0–96.7) | 86.7 (82.5–93.3) | NS |
| Hypertension | 37% | 33% | NS | 47% | 50% | NS |
| Dyslipidemia | 22% | 9% | NS | 26% | 15% | NS |
| Diabetes | 4% | 9% | NS | 8% | 0% | NS |
| Age first stone (years) | 27.0 (19.0–41.0) | 30.0 (18.7–41.2) | NS | 29.5 (21.0–41.5) | 27.5 (22.5–39.5) | NS |
| SWL (% of patients) | 37.3% | 33.3% | NS | 47.5% | 50.0% | NS |
| URS (% of patients) | 45.1% | 47.6% | NS | 51.6% | 40.0% | NS |
| Population | Female (n = 125) | Male (n = 148) | ||||
|---|---|---|---|---|---|---|
| Non-Drinkers (n = 102) | Green Tea (n = 21) | p | Non-Drinkers (n = 122) | Green Tea (n = 20) | p | |
| Fluid intake ≥ 2 L/day (%) | 11.2% | 50.0% | <0.0001 | 30.3% | 22.2% | 0.47 |
| Blood | ||||||
| Sodium (mmol/L) | 139 (138–140) | 139 (138–140) | 0.84 | 139 (138–140) | 140 (138–140) | 0.35 |
| Potassium (mmol/L) | 4.0 (3.8–4.3) | 4.1 (3.8–4.3) | 0.37 | 4.0 (3.9–4.3) | 4.1 (3.9–4.3) | 0.39 |
| Fasting glucose (mmol/L) | 5.5 (5.1–6.3) | 5.1 (5.06–5.5) | 0.06 | 5.4 (5.0–5.9) | 5.8 (5.5–6.4) | 0.008 |
| tCO2 (mmol/L) | 27.5 (26.0–29.0) | 27.9 (25.1–29.7) | 0.70 | 27.6 (26.3–29.7) | 27.6 (25.3–29.6) | 0.78 |
| Creatinine clearance (mL/min) | 126 (95–145) | 109 (84–138) | 0.16 | 121 (97–153) | 118 (101–157) | 0.94 |
| Ionized calcium (mmol/L) | 1.18 (1.15–1.21) | 1.19 (1.16–1.21) | 0.43 | 1.18 (1.15–1.21) | 1.17 (1.14–1.21) | 0.82 |
| PTH (pg/mL) | 35 (26–49) | 31 (25–40) | 0.31 | 35 (27–49) | 40 (33–55) | 0.11 |
| 25 OH vitamin D (pg/mL) | 24 (17–34) | 25 (19–37) | 0.52 | 25 (16–37) | 24 (20–30) | 0.95 |
| 1-25 (OH)2 vitamin D (ng/mL) | 66 (55–85) | 79 (54–90) | 0.5 | 66 (52–84) | 58 (57–87) | 0.48 |
| BALP (UI/L) | 13.5 (10.3–17.0) | 12.3 (10.3–15.1) | 0.36 | 13.5 (10.0–17.0) | 10.6 (9.5–13.0) | 0.06 |
| Deoxypyridin (mmol/mmol creat) | 5.7 (4.5–8.1) | 6.6 (4.1–9.7) | 0.7 | 5.4 (4.2–7.5) | 5.2 (3.9–6.4) | 0.29 |
| Urine | ||||||
| Diuresis (mL/day) | 1880 (1460–2582) | 1908 (1757–2368) | 0.89 | 1865 (1433–2337) | 1836 (1231–2301) | 0.62 |
| Calcium (mmol/day) | 6.2 (4.6–8.1) | 7.0 (5.5–10.0) | 0.08 | 5.7 (4.0–8.5) | 6.3 (4.5–8.1) | 0.95 |
| Oxalate (mmol/day) | 0.32 (0.24–0.42) | 0.27 (0.32–0.34) | 0.09 | 0.30 (0.19–0.41) | 0.29 (0.21–0.43) | 0.77 |
| Urate (mmol/day) | 3.5 (2.6–4.7) | 3.2 (2.7–3.7) | 0.33 | 3.5 (2.9–4.5) | 3.8 (3.0–4.7) | 0.51 |
| Citrate (mmol/day) | 2.4 (1.1–3.4) | 2.0 (1.7–3.4) | 0.98 | 2.5 (1.5–3.5) | 2.2 (0.4–2.7) | 0.22 |
| Fasting pH | 6.33 (5.68–6.66) | 6.2 (6.0–6.6) | 0.83 | 6.18 (5.62–6.61) | 5.74 (5.32–6.32) | 0.06 |
| Sodium (mmol/day) | 113 (84–157) | 121 (84–149) | 0.25 | 127 (95–173) | 126 (90–146) | 0.4 |
| Ammonium (mmol/day) | 35 (26–45) | 30 (26–45) | 0.28 | 35 (25–48) | 44 (31–54) | 0.39 |
| Magnesium (mmol/day) | 4.1 (3.2–5.0) | 5.0 (3.3–7.0) | 0.08 | 4.4 (3.2–5.9) | 4.2 (2.7–4.9) | 0.48 |
| Population | Female | Male | ||||
|---|---|---|---|---|---|---|
| Non-Drinkers | Green Tea | p-Value | Non-Drinkers | Green Tea | p Value | |
| AP CaOx index | 0.74 (0.39–1.30) | 0.71 (0.28–1.31) | 0.42 | 0.67 (0.37–1.10) | 0.76 (0.41–1.28) | 0.73 |
| Br RSS | 1.3 (0.3–2.5) | 1.0 (0.1–1.7) | 0.27 | 0.9 (0.3–1.9) | 0.6 (0.2–1.3) | 0.39 |
| UA RSS | 0.54 (0.25–1.81) | 0.75 (0.19–1.48) | 0.95 | 1.08 (0.41–2.26) | 1.99 (0.33–3.96) | 0.47 |
| CaOx RSS | 5.6 (3.7–8.6) | 3.7 (2.0–8.3) | 0.16 | 5.3(3.2–7.7) | 6.0 (3.7–8.7) | 0.64 |
| Ca.Ox | 0.53 (0.26–1.03) | 0.59 (0.23–1.15) | 0.67 | 0.47 (0.25–0.89) | 0.57 (0.30–0.91) | 0.57 |
| Ratio Ca/Ox | 19 (12.5–30) | 26 (20.5–40.5) | 0.01 | 19 (12–34) | 18.5 (12–28.5) | 0.65 |
| Ionic Strength | 0.08 (0.06–0.12) | 0.09 (0.07–0.12) | 0.72 | 0.09 (0.05–0.12) | 0.07 (0.075–0.105) | 0.43 |
| Population | Female | Male | ||||
|---|---|---|---|---|---|---|
| Non-Drinkers (n = 40) | Green Tea (n = 8) | p Value | Non-Drinkers (n = 41) | Green Tea (n = 9) | p Value | |
| Major COM component (%) | 42 | 0 | 0.036 | 33 | 44 | 0.99 |
| Major COD component (%) | 26 | 50 | 0.23 | 41 | 55 | 0.72 |
| Carbapatite major component (%) | 16 | 12.5 | 0.99 | 7 | 0 | 0.99 |
| Type Ia or Ib (%) | 23 | 0 | 0.32 | 18 | 11 | 0.99 |
| Type IIa or IIb (%) | 50 | 62 | 0.7 | 64 | 78 | 0.69 |
| Type IVa (%) | 10 | 12 | 0.99 | 8 | 0 | 0.99 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rode, J.; Bazin, D.; Dessombz, A.; Benzerara, Y.; Letavernier, E.; Tabibzadeh, N.; Hoznek, A.; Tligui, M.; Traxer, O.; Daudon, M.; et al. Daily Green Tea Infusions in Hypercalciuric Renal Stone Patients: No Evidence for Increased Stone Risk Factors or Oxalate-Dependent Stones. Nutrients 2019, 11, 256. https://doi.org/10.3390/nu11020256
Rode J, Bazin D, Dessombz A, Benzerara Y, Letavernier E, Tabibzadeh N, Hoznek A, Tligui M, Traxer O, Daudon M, et al. Daily Green Tea Infusions in Hypercalciuric Renal Stone Patients: No Evidence for Increased Stone Risk Factors or Oxalate-Dependent Stones. Nutrients. 2019; 11(2):256. https://doi.org/10.3390/nu11020256
Chicago/Turabian StyleRode, Julie, Dominique Bazin, Arnaud Dessombz, Yahia Benzerara, Emmanuel Letavernier, Nahid Tabibzadeh, Andras Hoznek, Mohamed Tligui, Olivier Traxer, Michel Daudon, and et al. 2019. "Daily Green Tea Infusions in Hypercalciuric Renal Stone Patients: No Evidence for Increased Stone Risk Factors or Oxalate-Dependent Stones" Nutrients 11, no. 2: 256. https://doi.org/10.3390/nu11020256
APA StyleRode, J., Bazin, D., Dessombz, A., Benzerara, Y., Letavernier, E., Tabibzadeh, N., Hoznek, A., Tligui, M., Traxer, O., Daudon, M., & Haymann, J.-P. (2019). Daily Green Tea Infusions in Hypercalciuric Renal Stone Patients: No Evidence for Increased Stone Risk Factors or Oxalate-Dependent Stones. Nutrients, 11(2), 256. https://doi.org/10.3390/nu11020256






