Rice Endosperm Protein Administration to Juvenile Mice Regulates Gut Microbiota and Suppresses the Development of High-Fat Diet-Induced Obesity and Related Disorders in Adulthood
Abstract
1. Introduction
2. Materials and Methods
2.1. Preparation of REP and Experimental Diets
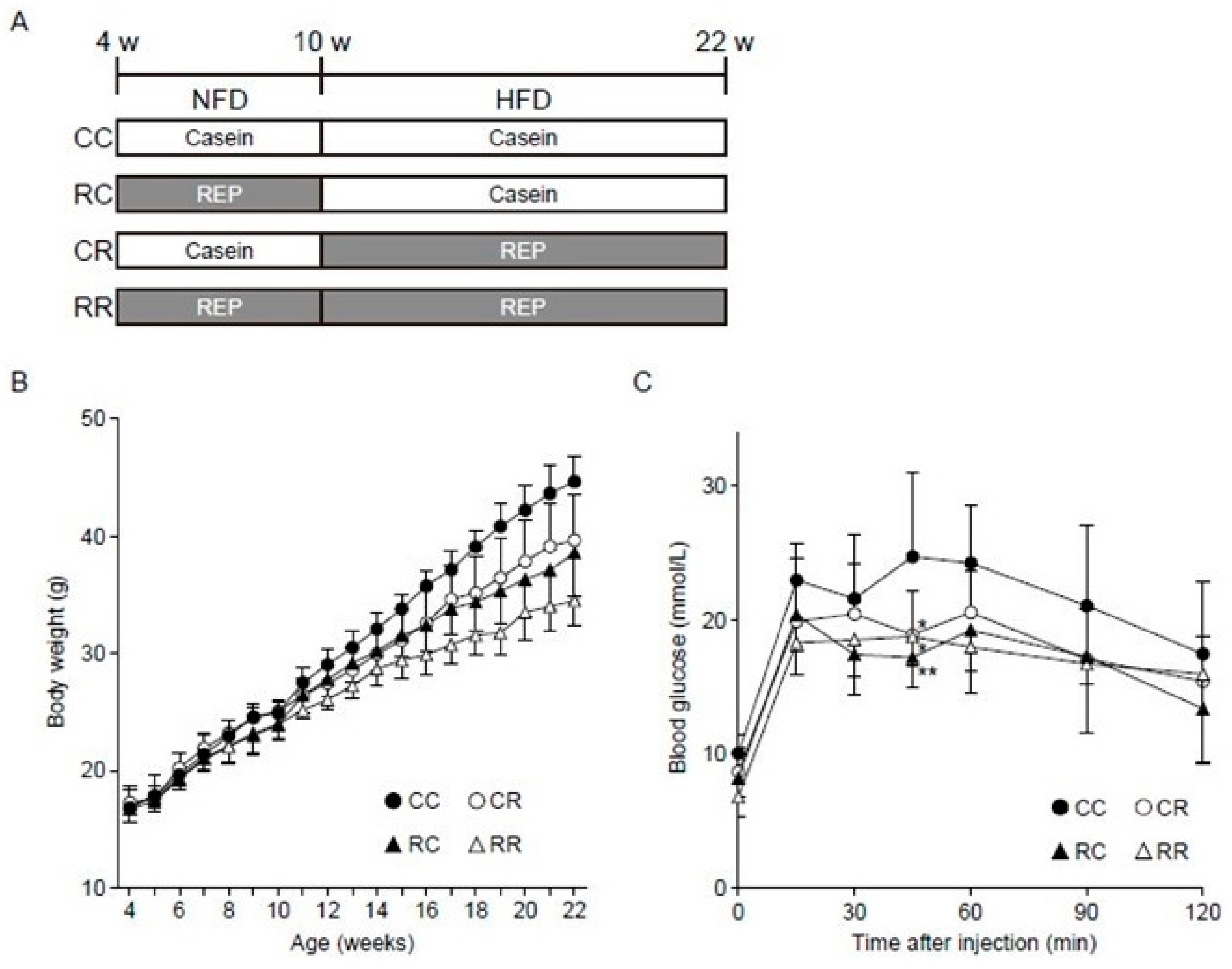
2.2. Animal Experiments
2.3. Blood and Urine Analysis
2.4. Cytokine Quantification in Serum and Tissues
2.5. Analysis of Kidney Morphology
2.6. Analysis of Liver Morphology and TG Content
2.7. Metabolic Assessment
2.8. Determination of Fecal Total Lipid and Bile Acid Levels
2.9. 16S rRNA Analysis of Fecal Samples and Bioinformatics Analysis
2.10. OGTT
2.11. Measurement of the Antimicrobial Activity of Peptides Derived from Casein and REP
2.12. Statistical Analysis
2.13. Data Availability
3. Results
3.1. REP Administration during the Juvenile Period Suppresses the Development of Obesity and Related Disorders in Adulthood
3.2. Administration of REP during the Juvenile Period Lowers Inflammation Markers in Adulthood
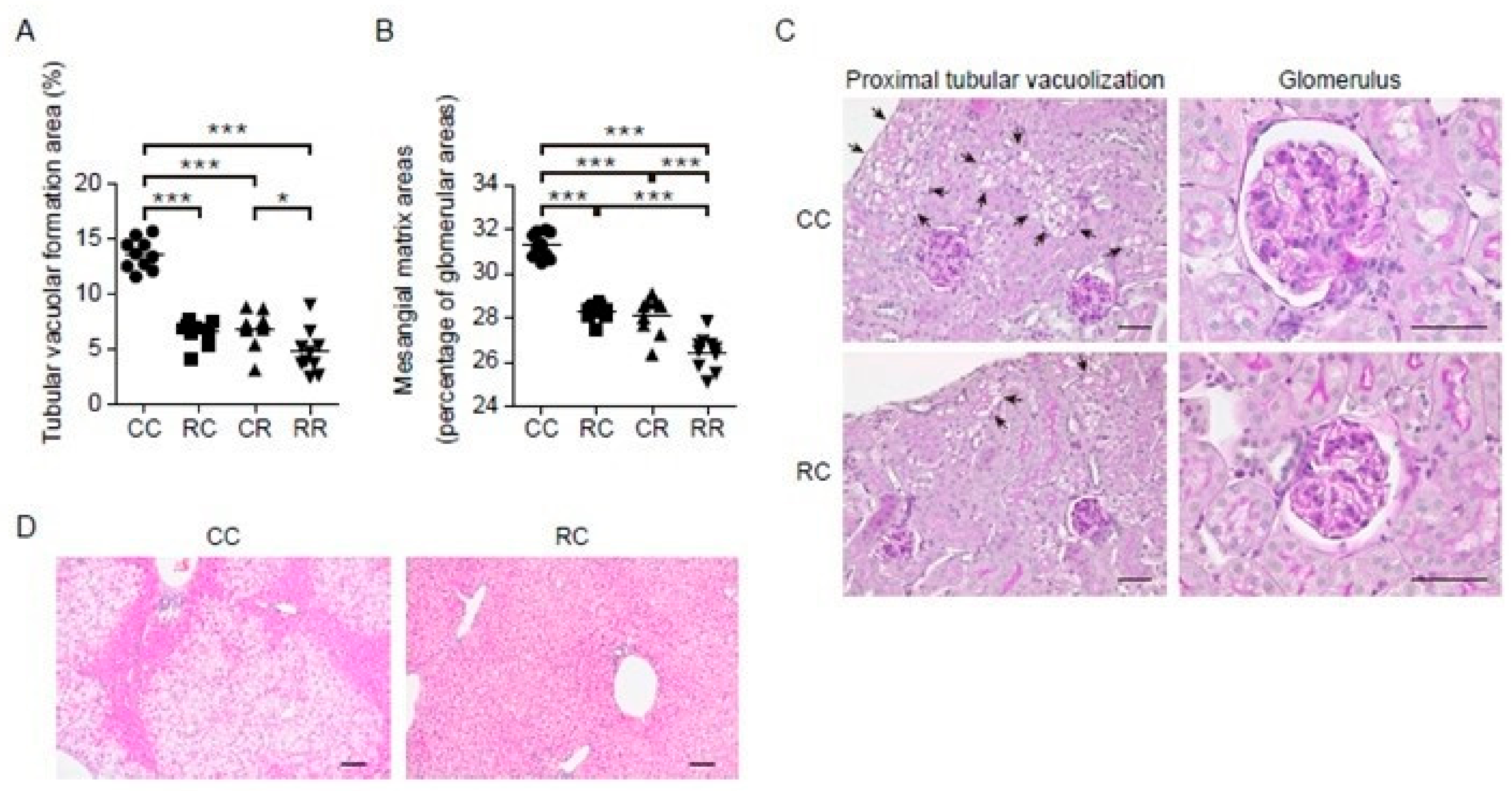
3.3. Administration of REP during the Juvenile Period Suppresses the Development of HFD-Induced Kidney and Liver Disease in Adulthood
3.4. No Differences in Energy Consumption among the CC, RC, CR, and RR Groups
3.5. Administration of REP Regulates Lipid and Total Bile Acid Excretion in Feces
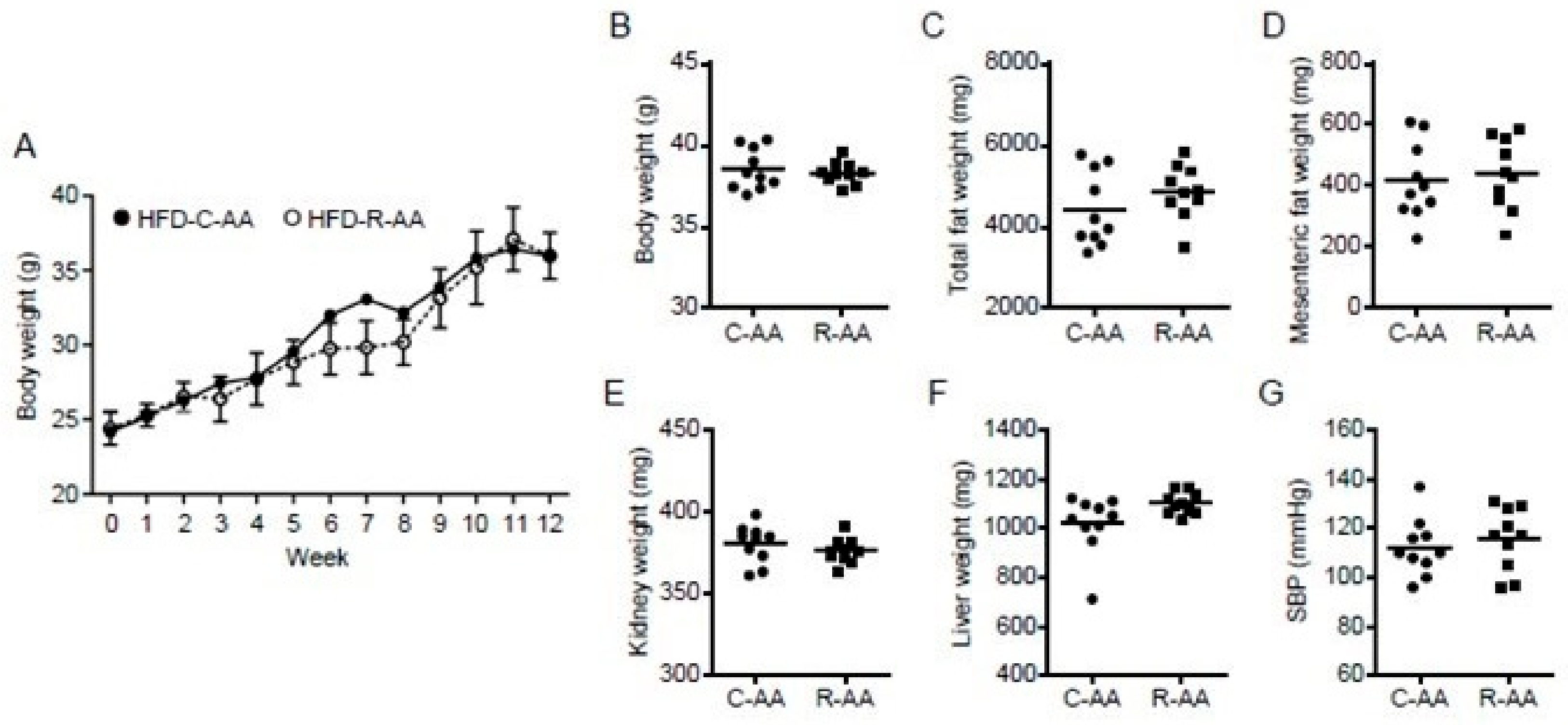
3.6. Differences in the Amino Acid Composition of REP and Casein Are not Associated with the Inhibitory Effect of REP on HFD-Induced Obesity
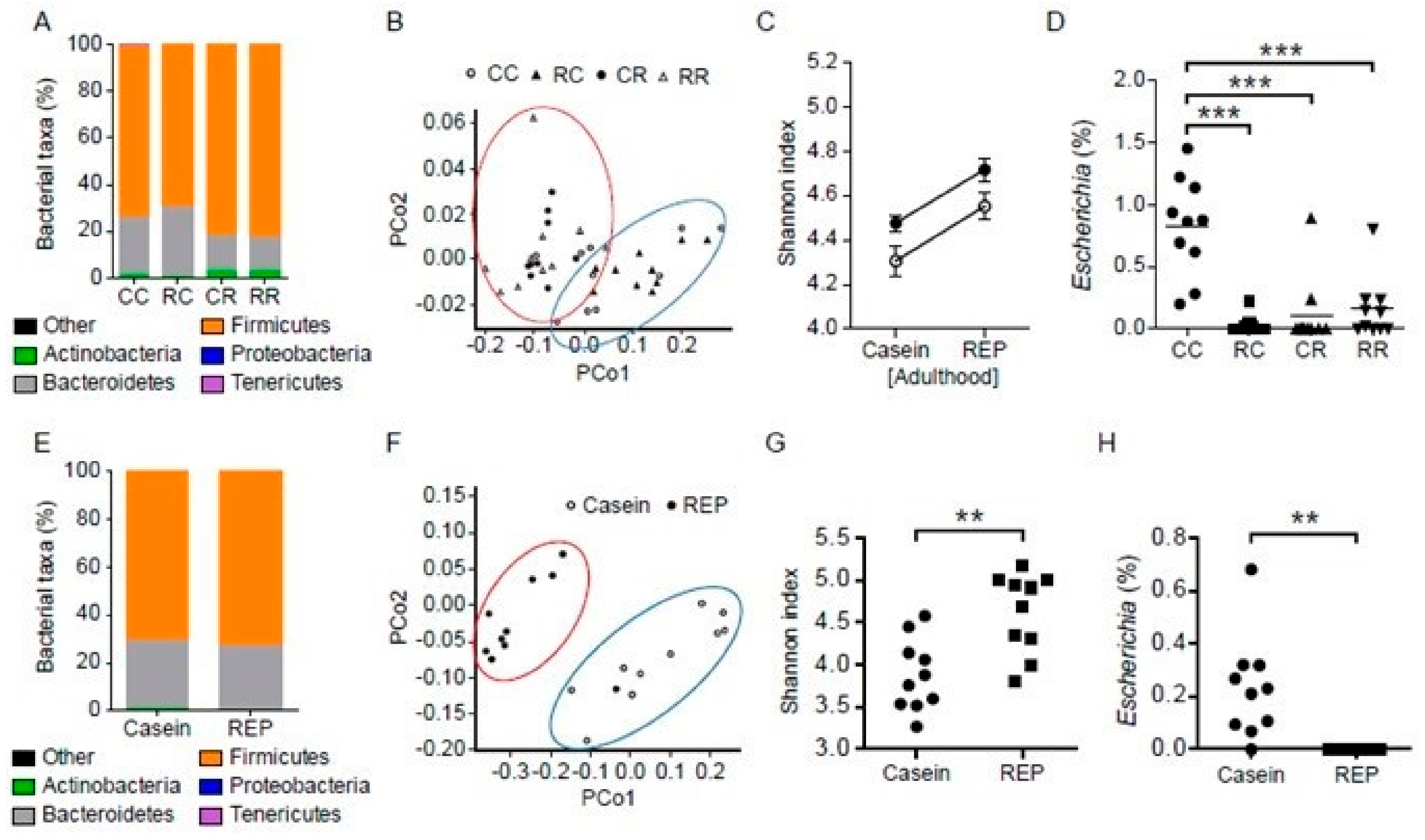
3.7. Administration of REP during the Juvenile Period Suppresses the Development of HFD-Induced Dysbiosis of the Gut Microbiota in Adulthood
3.8. REP-Derived Peptides Show Antibacterial Activity
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kopelman, P.G. Obesity as a medical problem. Nature 2000, 404, 635–643. [Google Scholar] [CrossRef]
- Kaur, J. A comprehensive review on metabolic syndrome. Cardiol. Res. Pr. 2014, 2014, 943162. [Google Scholar] [CrossRef]
- Luk, A.O.; So, W.Y.; Ma, R.C.; Kong, A.P.; Ozaki, R.; Ng, V.S.; Yu, L.W.; Lau, W.W.; Yang, X.; Chow, F.C.; et al. Metabolic syndrome predicts new onset of chronic kidney disease in 5829 patients with type 2 diabetes: A 5-year prospective analysis of the Hong Kong Diabetes Registry. Diabetes Care 2008, 31, 2357–2361. [Google Scholar] [CrossRef]
- Cawley, J.; Meyerhoefer, C. The medical care costs of obesity: An instrumental variables approach. J. Health Econ. 2012, 31, 219–230. [Google Scholar] [CrossRef]
- Lloyd, L.J.; Langley-Evans, S.C.; McMullen, S. Childhood obesity and adult cardiovascular disease risk: A systematic review. Int. J. Obes. 2010, 34, 18–28. [Google Scholar] [CrossRef]
- The, N.S.; Suchindran, C.; North, K.E.; Popkin, B.M.; Gordon-Larsen, P. Association of adolescent obesity with risk of severe obesity in adulthood. JAMA 2010, 304, 2042–2047. [Google Scholar] [CrossRef]
- Laitinen, T.T.; Nuotio, J.; Juonala, M.; Niinikoski, H.; Rovio, S.; Viikari, J.S.A.; Rönnemaa, T.; Magnussen, C.G.; Jokinen, E.; Lagström, H.; et al. Success in Achieving the Targets of the 20-Year Infancy-Onset Dietary Intervention: Association With Insulin Sensitivity and Serum Lipids. Diabetes Care 2018, 41, 2236–2244. [Google Scholar] [CrossRef]
- Jääskeläinen, P.; Magnussen, C.G.; Pahkala, K.; Mikkilä, V.; Kähönen, M.; Sabin, M.A.; Fogelholm, M.; Hutri-Kähönen, N.; Taittonen, L.; Telama, R.; et al. Childhood nutrition in predicting metabolic syndrome in adults: The cardiovascular risk in Young Finns Study. Diabetes Care 2012, 35, 1937–1943. [Google Scholar] [CrossRef]
- Quick, V.; Wall, M.; Larson, N.; Haines, J.; Neumark-Sztainer, D. Personal, behavioral and socio-environmental predictors of overweight incidence in young adults: 10-yr longitudinal findings. Int. J. Behav. Nutr. Phys. Act. 2013, 10, 37. [Google Scholar] [CrossRef]
- Ministry of Health, Labour and Welfare, Japan. The National Health and Nutrition Survey in Japan; MHLW: Tokyo, Japan, 2012. (In Japanese)
- St-Onge, M.P.; Keller, K.L.; Heymsfield, S.B. Changes in childhood food consumption patterns: A cause for concern in light of increasing body weights. Am. J. Clin. Nutr. 2003, 78, 1068–1073. [Google Scholar] [CrossRef]
- Pingali, P. Westernization of Asian diets and the transformation of food systems: Implications for research and policy. Food Policy 2007, 32, 281–298. [Google Scholar] [CrossRef]
- Kumagai, T.; Kawamura, H.; Fuse, T.; Watanabe, T.; Saito, Y.; Masumura, T.; Watanabe, R.; Kadowaki, M. Production of rice protein by alkaline extraction improves its digestibility. J. Nutr. Sci. Vitam. 2006, 52, 467–472. [Google Scholar] [CrossRef]
- Kumagai, T.; Watanabe, R.; Saito, M.; Watanabe, T.; Kubota, M.; Kadowaki, M. Superiority of alkali-extracted rice protein in bioavailability to starch degraded rice protein comes from digestion of prolamin in growing rats. J. Nutr. Sci. Vitam. 2009, 55, 170–177. [Google Scholar] [CrossRef][Green Version]
- Tanaka, K.; Sugimoto, T.; Ogawa, M.; Kasai, Z. Isolation and Characterization of Two Types of Protein Bodies in the Rice Endosperm. Agric. Biol. Chem. 1980, 44, 1633–1639. [Google Scholar] [CrossRef]
- Yamagata, H.; Sugimoto, T.; Tanaka, K.; Kasai, Z. Biosynthesis of storage proteins in developing rice seeds. Plant Physiol. 1982, 70, 1094–1100. [Google Scholar] [CrossRef]
- Morita, T.; Ohhashi, A.; Kasaoka, S.; Ikai, M.; Kiriyama, S. Rice protein isolates produced by the two different methods lower serum cholesterol concentration in rats compared with casein. J. Sci. Food Agric. 1996, 71, 415–424. [Google Scholar] [CrossRef]
- Yang, L.; Kumagai, T.; Kawamura, H.; Watanabe, T.; Kubota, M.; Fujimura, S.; Watanabe, R.; Kadowaki, M. Effects of rice proteins from two cultivars, Koshihikari and Shunyo, on cholesterol and triglyceride metabolism in growing and adult rats. Biosci. Biotechnol. Biochem. 2007, 71, 694–703. [Google Scholar] [CrossRef]
- Um, M.Y.; Ahn, J.; Jung, C.H.; Ha, T.Y. Cholesterol-lowering Effect of Rice Protein by Enhancing Fecal Excretion of Lipids in Rats. Prev. Nutr. Food Sci. 2013, 18, 210–213. [Google Scholar] [CrossRef][Green Version]
- Kubota, M.; Watanabe, R.; Yamaguchi, M.; Hosojima, M.; Saito, A.; Fujii, M.; Fujimura, S.; Kadowaki, M. Rice endosperm protein slows progression of fatty liver and diabetic nephropathy in Zucker diabetic fatty rats. Br. J. Nutr. 2016, 116, 1326–1335. [Google Scholar] [CrossRef]
- Araújo, J.R.; Tomas, J.; Brenner, C.; Sansonetti, P.J. Impact of high-fat diet on the intestinal microbiota and small intestinal physiology before and after the onset of obesity. Biochimie 2017, 141, 97–106. [Google Scholar] [CrossRef]
- Shin, N.R.; Whon, T.W.; Bae, J.W. Proteobacteria: Microbial signature of dysbiosis in gut microbiota. Trends Biotechnol. 2015, 33, 496–503. [Google Scholar] [CrossRef] [PubMed]
- Sonnenburg, J.L.; Bäckhed, F. Diet-microbiota interactions as moderators of human metabolism. Nature 2016, 535, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Miller, L.; Houghton, J.A. The micro-Kjeldahl determination of the nitrogen content of amino acids and proteins. J. Biol. Chem. 1945, 159, 373–380. [Google Scholar]
- Dutta, S.; Sengupta, P. Men and mice: Relating their ages. Life Sci. 2016, 152, 244–248. [Google Scholar] [CrossRef] [PubMed]
- Kuwahara, S.; Hosojima, M.; Kaneko, R.; Aoki, H.; Nakano, D.; Sasagawa, T.; Kabasawa, H.; Kaseda, R.; Yasukawa, R.; Ishikawa, T.; et al. Megalin-Mediated Tubuloglomerular Alterations in High-Fat Diet-Induced Kidney Disease. J. Am. Soc. Nephrol. 2016, 27, 1996–2008. [Google Scholar] [CrossRef]
- Rabot, S.; Membrez, M.; Bruneau, A.; Gérard, P.; Harach, T.; Moser, M.; Raymond, F.; Mansourian, R.; Chou, C.J. Germ-free C57BL/6J mice are resistant to high-fat-diet-induced insulin resistance and have altered cholesterol metabolism. FASEB J. 2010, 24, 4948–4959. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Sloane-Stanley, G.H. A simple method for the isolation and purification of total lipids from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar]
- Zhang, J.C.; Yao, W.; Dong, C.; Yang, C.; Ren, Q.; Ma, M.; Hashimoto, K. Blockade of interleukin-6 receptor in the periphery promotes rapid and sustained antidepressant actions: A possible role of gut-microbiota-brain axis. Transl. Psychiatry 2017, 7, e1138. [Google Scholar] [CrossRef]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Peña, A.G.; Goodrich, J.K.; Gordon, J.I.; et al. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef]
- Wang, Q.; Garrity, G.M.; Tiedje, J.M.; Cole, J.R. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 2007, 73, 5261–5267. [Google Scholar] [CrossRef]
- Cole, J.R.; Wang, Q.; Fish, J.A.; Chai, B.; McGarrell, D.M.; Sun, Y.; Brown, C.T.; Porras-Alfaro, A.; Kuske, C.R.; Tiedje, J.M. Ribosomal Database Project: Data and tools for high throughput rRNA analysis. Nucleic Acids Res. 2014, 42, D633–D642. [Google Scholar] [CrossRef] [PubMed]
- Warwick, R.M.; Clarke, K.R. Practical measures of marine biodiversity based on relatedness of species. Oceanogr. Mar. Biol. 2001, 39, 207–231. [Google Scholar]
- Nakano, T.; Simatani, M.; Murakami, Y.; Sato, N.; Idota, T. Digestibility and Absorption of Enzymatically Hydrolyzed Whey Protein. J. Jpn. Soc. Nutr. Food Sci. 1994, 47, 195–201. [Google Scholar] [CrossRef]
- Taniguchi, M.; Kawabe, J.; Toyoda, R.; Namae, T.; Ochiai, A.; Saitoh, E.; Tanaka, T. Cationic peptides from peptic hydrolysates of rice endosperm protein exhibit antimicrobial, LPS-neutralizing, and angiogenic activities. Peptides 2017, 97, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, M.; Kameda, M.; Namae, T.; Ochiai, A.; Saitoh, E.; Tanaka, T. Identification and characterization of multifunctional cationic peptides derived from peptic hydrolysates of rice bran protein. J. Funct. Foods 2017, 34, 287–296. [Google Scholar] [CrossRef]
- Chen, X.; Devaraj, S. Gut Microbiome in Obesity, Metabolic Syndrome, and Diabetes. Curr. Diabetes Rep. 2018, 18, 129. [Google Scholar] [CrossRef]
- Yamamoto, T.; Takabatake, Y.; Takahashi, A.; Kimura, T.; Namba, T.; Matsuda, J.; Minami, S.; Kaimori, J.Y.; Matsui, I.; Matsusaka, T.; et al. High-Fat Diet-Induced Lysosomal Dysfunction and Impaired Autophagic Flux Contribute to Lipotoxicity in the Kidney. J. Am. Soc. Nephrol. 2017, 28, 1534–1551. [Google Scholar] [CrossRef]
- Hu, T.; Jacobs, D.R.; Larson, N.I.; Cutler, G.J.; Laska, M.N.; Neumark-Sztainer, D. Higher Diet Quality in Adolescence and Dietary Improvements Are Related to Less Weight Gain during the Transition from Adolescence to Adulthood. J. Pediatr. 2016, 178, 188–193. [Google Scholar] [CrossRef]
- Yatsunenko, T.; Rey, F.E.; Manary, M.J.; Trehan, I.; Dominguez-Bello, M.G.; Contreras, M.; Magris, M.; Hidalgo, G.; Baldassano, R.N.; Anokhin, A.P.; et al. Human gut microbiome viewed across age and geography. Nature 2012, 486, 222–227. [Google Scholar] [CrossRef]
- Willson, K.; Situ, C. Systematic review on effects of diet on gut microbiota in relation to metabolic syndromes. J. Clin. Nutr. Metab. 2017, 1, 1–12. [Google Scholar]
- Cani, P.D.; Amar, J.; Iglesias, M.A.; Poggi, M.; Knauf, C.; Bastelica, D.; Neyrinck, A.M.; Fava, F.; Tuohy, K.M.; Chabo, C.; et al. Metabolic endotoxemia initiates obesity and insulin resistance. Diabetes 2007, 56, 1761–1772. [Google Scholar] [CrossRef] [PubMed]
- Qin, J.; Li, Y.; Cai, Z.; Li, S.; Zhu, J.; Zhang, F.; Liang, S.; Zhang, W.; Guan, Y.; Shen, D.; et al. A metagenome-wide association study of gut microbiota in type 2 diabetes. Nature 2012, 490, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Jazayeri, O.; Daghighi, S.M.; Rezaee, F. Lifestyle alters GUT-bacteria function: Linking immune response and host. Best Pr. Res. Clin. Gastroenterol. 2017, 31, 625–635. [Google Scholar] [CrossRef] [PubMed]
- Hildebrandt, M.A.; Hoffmann, C.; Sherrill-Mix, S.A.; Keilbaugh, S.A.; Hamady, M.; Chen, Y.Y.; Knight, R.; Ahima, R.S.; Bushman, F.; Wu, G.D. High-fat diet determines the composition of the murine gut microbiome independently of obesity. Gastroenterology 2009, 137, 1716–1724. [Google Scholar] [CrossRef] [PubMed]
- Delzenne, N.M.; Cani, P.D. Interaction between obesity and the gut microbiota: Relevance in nutrition. Annu. Rev. Nutr. 2011, 31, 15–31. [Google Scholar] [CrossRef] [PubMed]
- Sato, J.; Kanazawa, A.; Ikeda, F.; Yoshihara, T.; Goto, H.; Abe, H.; Komiya, K.; Kawaguchi, M.; Shimizu, T.; Ogihara, T.; et al. Gut Dysbiosis and Detection of “Live Gut Bacteria” in Blood of Japanese Patients With Type 2 Diabetes. Diabetes Care 2014, 37, 2343–2350. [Google Scholar] [CrossRef]
- Mahmoodpoor, F.; Rahbar Saadat, Y.; Barzegari, A.; Ardalan, M.; Zununi Vahed, S. The impact of gut microbiota on kidney function and pathogenesis. Biomed. Pharm. 2017, 93, 412–419. [Google Scholar] [CrossRef]
- Hoek, K.S.; Milne, J.M.; Grieve, P.A.; Dionysius, D.A.; Smith, R. Antibacterial activity in bovine lactoferrin-derived peptides. Antimicrob. Agents Chemother. 1997, 41, 54–59. [Google Scholar] [CrossRef]
- Choi, S.K.; Adachi, M.; Utsumi, S. Improved bile acid-binding ability of soybean glycinin A1a polypeptide by the introduction of a bile acid-binding peptide (VAWWMY). Biosci. Biotechnol. Biochem. 2004, 68, 1980–1983. [Google Scholar] [CrossRef]
- Stanley, M.M.; Paul, D.; Gacke, D.; Murphy, J. Effects of cholestyramine, metamucil, and cellulose on fecal bile salt excretion in man. Gastroenterology 1973, 65, 889–894. [Google Scholar] [CrossRef]
- Van Bennekum, A.M.; Nguyen, D.V.; Schulthess, G.; Hauser, H.; Phillips, M.C. Mechanisms of cholesterol-lowering effects of dietary insoluble fibres: Relationships with intestinal and hepatic cholesterol parameters. Br. J. Nutr. 2005, 94, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Hosojima, M.; Shimada, H.; Obi, Y.; Kuwahara, S.; Kaseda, R.; Kabasawa, H.; Kondo, H.; Fujii, M.; Watanabe, R.; Suzuki, Y.; et al. A Randomized, Double-Blind, Crossover Pilot Trial of Rice Endosperm Protein Supplementation in Maintenance Hemodialysis Patients. Sci. Rep. 2017, 7, 18003. [Google Scholar] [CrossRef] [PubMed]
- Hosojima, M.; Kaseda, R.; Kondo, H.; Fujii, M.; Kubota, M.; Watanabe, R.; Tanabe, N.; Kadowaki, M.; Suzuki, Y.; Saito, A. Beneficial effects of rice endosperm protein intake in Japanese men with risk factors for metabolic syndrome: A randomized, crossover clinical trial. BMC Nutr. 2016, 2, 25. [Google Scholar] [CrossRef]
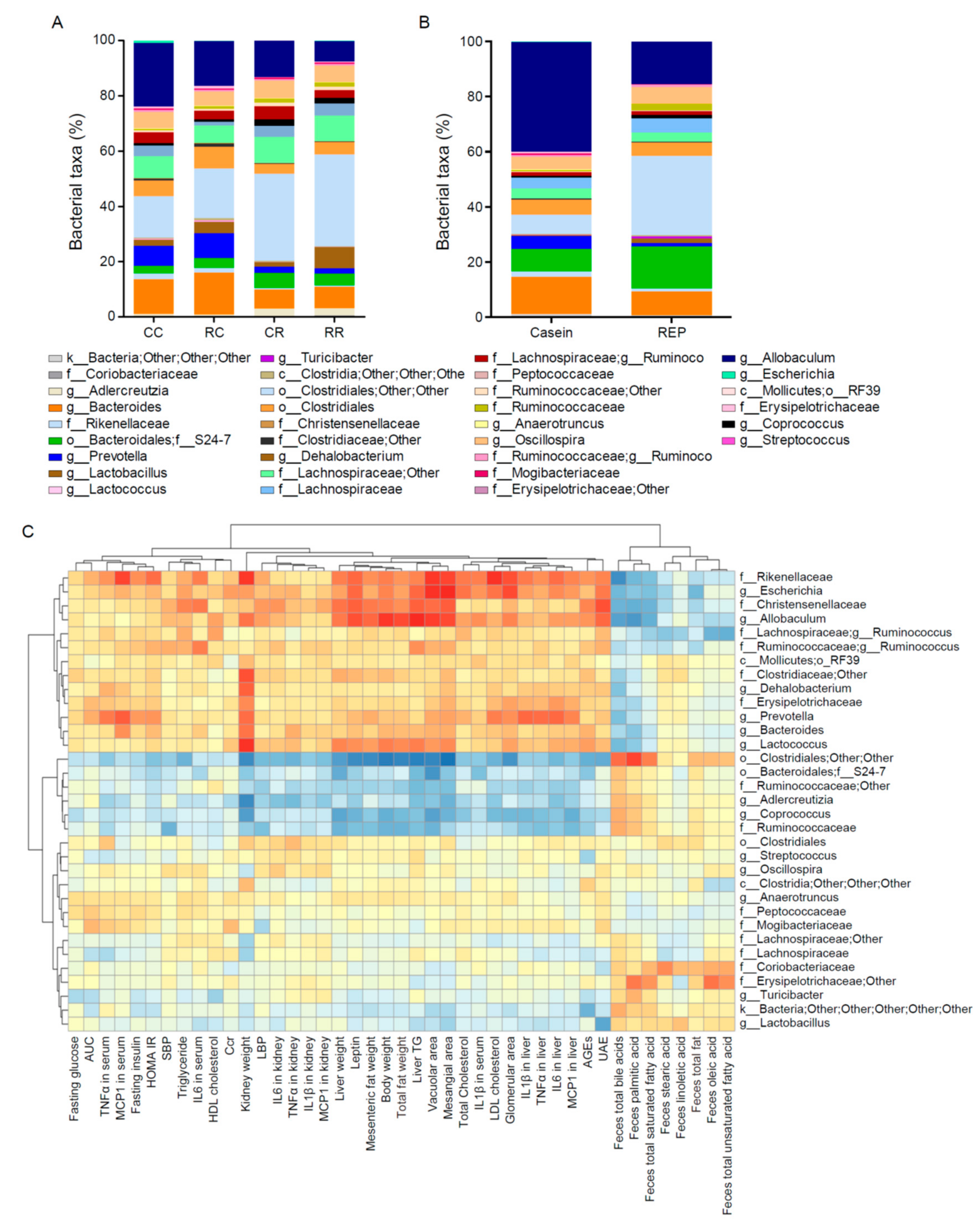
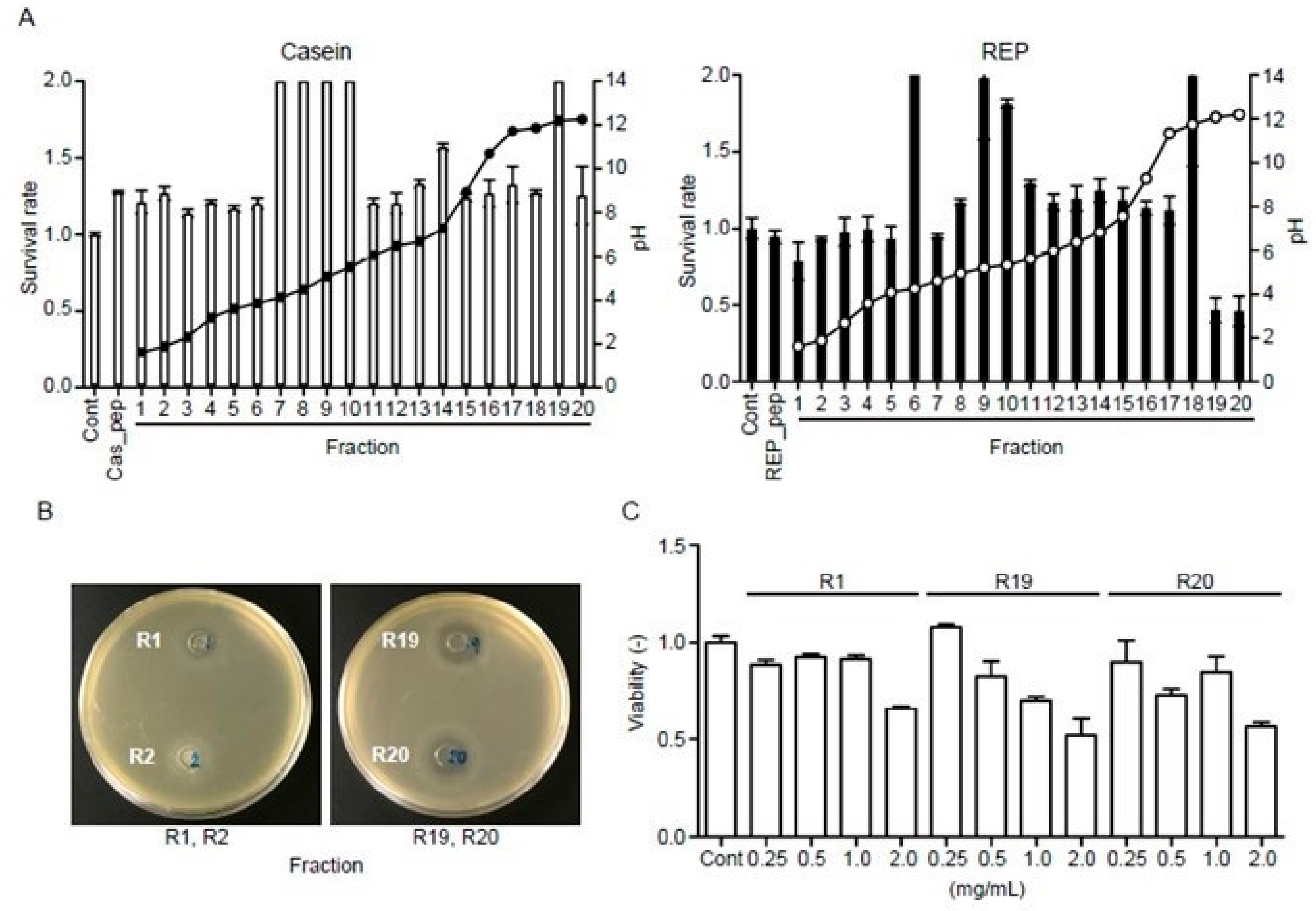
| Parameter | Group | P Value | |||||
|---|---|---|---|---|---|---|---|
| CC | RC | CR | RR | Juvenile- Period Protein Source | Adulthood Protein Source | Interaction | |
| Body weight (g) | 44.6 ± 2.2 | 39.3 ± 3.7 | 39.5 ± 3.7 | 34.5 ± 2.1 | <0.001 | <0.001 | 0.829 |
| SBP (mmHg) | 117.0 ± 6.5 | 106.5 ± 10.0 | 105.1 ± 7.8 | 104.5 ± 7.7 | 0.037 | 0.010 | 0.062 |
| Total cholesterol (mmol/L) | 3.34 ± 0.56 | 2.73 ± 0.31 | 2.69 ± 0.22 | 2.60 ± 0.40 | 0.008 | 0.003 | 0.045 |
| Triglyceride (mmol/L) | 0.37 ± 0.13 | 0.24 ± 0.10 | 0.19 ± 0.08 | 0.15 ± 0.07 | 0.010 | <0.001 | 0.145 |
| Fasting glucose (mmol/L) | 10.0 ± 1.4 | 8.2 ± 1.5 | 8.7 ± 1.8 | 6.8 ± 1.6 | 0.001 | 0.012 | 0.986 |
| Fasting insulin (pmol/L) | 0.31 ± 0.11 | 0.20 ± 0.07 | 0.22 ± 0.11 | 0.15 ± 0.02 | 0.006 | 0.027 | 0.498 |
| AUC (102 mmol/L·min) | 14.1 ± 5.2 | 10.5 ± 2.4 | 11.2 ± 2.4 | 9.9 ± 2.4 | 0.038 | 0.121 | 0.307 |
| HOMA-IR | 2.29 ± 0.85 | 1.21 ± 0.49 | 1.45 ± 0.88 | 0.77 ± 0.23 | 0.001 | 0.008 | 0.391 |
| AGEs (µg/mL) | 4.05 ± 0.63 | 3.19 ± 0.85 | 3.57 ± 0.79 | 2.65 ± 0.92 | 0.001 | 0.055 | 0.910 |
| Total fat weight (g) | 5.8 ± 0.4 | 4.7 ± 1.0 | 4.7 ± 1.1 | 3.2 ± 0.6 | <0.001 | <0.001 | 0.428 |
| Mesenteric fat weight (mg) | 772.1 ± 193.0 | 544.7 ± 164.6 | 504.4 ± 150.9 | 291.8 ± 63.4 | <0.001 | <0.001 | 0.878 |
| Parameter | NFD-C | NFD-R | P Value |
|---|---|---|---|
| Body weight (g) | 23.3 ± 1.1 | 22.9 ± 1.0 | 0.353 |
| Total fat weight (mg) | 761.0 ± 146.4 | 814.5 ± 162.4 | 0.449 |
| Mesenteric fat weight (mg) | 159.6 ± 35.7 | 158.2 ± 41.7 | 0.934 |
| Lean body weight (g) | 22.6 ± 1.1 | 22.1 ± 0.9 | 0.273 |
| Total cholesterol (mmol/L) | 3.0 ± 0.2 | 2.2 ± 0.5 | <0.001 |
| Triglyceride (mmol/L) | 0.59 ± 0.20 | 0.42 ± 0.23 | 0.098 |
| Glucose (mmol/L) | 6.8 ± 1.3 | 6.6 ± 0.8 | 0.700 |
| Parameter | Group | P Value | |||||
|---|---|---|---|---|---|---|---|
| CC | RC | CR | RR | Juvenile- Period Protein Source | Adulthood Protein Source | Interaction | |
| Serum | |||||||
| LBP (µg/mL) | 10.5 ± 1.3 | 8.6 ± 1.1 | 8.6 ± 1.8 | 7.7 ± 1.4 | 0.006 | 0.004 | 0.270 |
| Leptin (nmol/L) | 3.18 ± 0.46 | 2.19 ± 0.40 | 2.07 ± 0.57 | 1.19 ± 0.46 | <0.001 | <0.001 | 0.697 |
| IL-1β (pg/mL) | 973.2 ± 344.2 | 625.1 ± 169.0 | 684.5 ± 229.2 | 599.1 ± 157.9 | 0.006 | 0.043 | 0.088 |
| IL-6 (pg/mL) | 114.1 ± 34.0 | 88.2 ± 14.7 | 89.7 ± 12.0 | 85.9 ± 12.5 | 0.028 | 0.046 | 0.095 |
| TNF-α (pg/mL) | 171.6 ± 139.3 | 73.5 ± 102.3 | 111.4 ± 139.3 | 40.5 ± 29.0 | 0.022 | 0.197 | 0.703 |
| MCP-1 (pg/mL) | 1041.8 ± 1218.7 | 274.7 ± 363.0 | 516.8 ± 628.7 | 222.5 ± 118.6 | 0.024 | 0.208 | 0.301 |
| Kidney | |||||||
| IL-1β (pg/g tissue) | 827.8 ± 164.1 | 698.5 ± 125.2 | 755.7 ± 119.2 | 705.3 ± 134.5 | 0.045 | 0.455 | 0.368 |
| IL-6 (pg/g tissue) | 251.4 ± 53.4 | 210.8 ± 53.3 | 216.8 ± 27.7 | 190.1 ± 24.6 | 0.016 | 0.044 | 0.606 |
| TNF-α (pg/g tissue) | 155.4 ± 26.9 | 131.3 ± 27.3 | 144.6 ± 26.5 | 127.7 ± 23.9 | 0.018 | 0.392 | 0.668 |
| MCP-1 (pg/g tissue) | 1698.3 ± 297.0 | 1387.9 ± 288.2 | 1487.5 ± 198.3 | 1404.7 ± 255.4 | 0.023 | 0.250 | 0.179 |
| Liver | |||||||
| IL-1β (pg/g tissue) | 567.0 ± 211.7 | 400.7 ± 95.9 | 440.6 ± 134.7 | 363.0 ± 93.8 | 0.010 | 0.077 | 0.331 |
| IL-6 (pg/g tissue) | 322.8 ± 147.8 | 216.0 ± 24.7 | 249.3 ± 93.8 | 173.8 ± 44.7 | 0.003 | 0.053 | 0.591 |
| TNF-α (pg/g tissue) | 122.1 ± 38.4 | 88.6 ± 12.4 | 98.3 ± 35.7 | 81.1 ± 18.4 | 0.008 | 0.091 | 0.372 |
| MCP-1 (pg/g tissue) | 372.9 ± 88.5 | 284.0 ± 58.2 | 317.9 ± 91.8 | 260.6 ± 60.3 | 0.005 | 0.113 | 0.517 |
| Parameter | Group | P Value | |||||
|---|---|---|---|---|---|---|---|
| CC | RC | CR | RR | Juvenile- Period Protein Source | Adulthood Protein Source | Interaction | |
| Kidney weight (mg) | 411.2 ± 23.0 | 382.6 ± 12.1 | 413.1 ± 22.0 | 375.0 ± 16.8 | <0.001 | 0.634 | 0.429 |
| UAE (µg/day) | 239.2 ± 39.4 | 182.7 ± 38.9 | 155.4 ± 57.1 | 96.6 ± 44.4 | <0.001 | <0.001 | 0.939 |
| CCr (µL/s) | 6.6 ± 1.4 | 5.5 ± 1.5 | 5.5 ± 1.3 | 5.0 ± 1.8 | 0.106 | 0.116 | 0.556 |
| Tubular vacuolar formation area (%) | 13.6 ± 1.4 | 6.6 ± 1.1 | 6.9 ± 1.6 | 4.8 ± 2.0 | <0.001 | <0.001 | <0.001 |
| Glomerular area (µm2) | 4113 ± 249 | 3630 ± 245 | 3603 ± 246 | 3381 ± 165 | <0.001 | <0.001 | 0.080 |
| Mesangial matrix area (%) | 31.3 ± 0.6 | 28.3 ± 0.4 | 28.1 ± 0.8 | 26.4 ± 0.8 | <0.001 | <0.001 | 0.011 |
| Liver weight (mg) | 1753.2 ± 284.0 | 1327.6 ± 136.3 | 1432.8 ± 233.3 | 1274.6 ± 124.6 | <0.001 | 0.007 | 0.047 |
| Liver TG (mg/liver) | 25.5 ± 3.8 | 15.5 ± 4.3 | 16.5 ± 4.5 | 11.6 ± 3.9 | <0.001 | <0.001 | 0.056 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Higuchi, Y.; Hosojima, M.; Kabasawa, H.; Kuwahara, S.; Goto, S.; Toba, K.; Kaseda, R.; Tanaka, T.; Kitamura, N.; Takihara, H.; et al. Rice Endosperm Protein Administration to Juvenile Mice Regulates Gut Microbiota and Suppresses the Development of High-Fat Diet-Induced Obesity and Related Disorders in Adulthood. Nutrients 2019, 11, 2919. https://doi.org/10.3390/nu11122919
Higuchi Y, Hosojima M, Kabasawa H, Kuwahara S, Goto S, Toba K, Kaseda R, Tanaka T, Kitamura N, Takihara H, et al. Rice Endosperm Protein Administration to Juvenile Mice Regulates Gut Microbiota and Suppresses the Development of High-Fat Diet-Induced Obesity and Related Disorders in Adulthood. Nutrients. 2019; 11(12):2919. https://doi.org/10.3390/nu11122919
Chicago/Turabian StyleHiguchi, Yuki, Michihiro Hosojima, Hideyuki Kabasawa, Shoji Kuwahara, Sawako Goto, Koji Toba, Ryohei Kaseda, Takahiro Tanaka, Nobutaka Kitamura, Hayato Takihara, and et al. 2019. "Rice Endosperm Protein Administration to Juvenile Mice Regulates Gut Microbiota and Suppresses the Development of High-Fat Diet-Induced Obesity and Related Disorders in Adulthood" Nutrients 11, no. 12: 2919. https://doi.org/10.3390/nu11122919
APA StyleHiguchi, Y., Hosojima, M., Kabasawa, H., Kuwahara, S., Goto, S., Toba, K., Kaseda, R., Tanaka, T., Kitamura, N., Takihara, H., Okuda, S., Taniguchi, M., Arao, H., Narita, I., & Saito, A. (2019). Rice Endosperm Protein Administration to Juvenile Mice Regulates Gut Microbiota and Suppresses the Development of High-Fat Diet-Induced Obesity and Related Disorders in Adulthood. Nutrients, 11(12), 2919. https://doi.org/10.3390/nu11122919





