Nutritional Gaps and Supplementation in the First 1000 Days
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Methods
2.2. Selected Literature
3. Why Nutrition Matters: Maternal Physiological Changes and the Role of Nutrition
- The first 1000 days of life represents the time from pregnancy through the child’s second birthday.
- Mothers undergo major physiological changes to maintain pregnancy and prepare for a healthy labor and delivery; these changes begin after conception and affect all organ systems’ development, but especially the fetal cardiovascular, endocrine, gastrointestinal, hematological, respiratory and skeletal systems.
- Optimal nutrition status during pregnancy is critical, as it impacts early embryonic development, organogenesis and neural development.
- Nutrients such as the carotenoids (lutein + zeaxanthin), choline, folate, iodine, iron, omega-3 fatty acids and vitamin D play critical roles during fetal development.
3.1. Optimal Nutrition to Help Sustain A Healthy Pregnancy and Critical Periods of Development
3.1.1. Carotenoids
3.1.2. Choline
3.1.3. Folate
3.1.4. Iodine
3.1.5. Iron
3.1.6. Omega-3 Fatty Acids
3.1.7. Vitamin D
- Pregnancy is a period of increased nutrient demands, when optimal nutrition is critical for maturing, proliferating, and differentiating cells throughout the fetus.
- Carotenoids play a key role in brain, eye and nervous system development.
- Choline fuels cell growth and proliferation, as well as nervous and cognitive system development.
- Adequate intakes of folate prior to and during pregnancy may help to prevent neural tube defects. Folate also plays a key role in DNA synthesis and amino acid metabolism.
- Iodine helps produce thyroid hormones, that are transferred to the fetus early in life.
- Iron is a major component of hemoglobin, a protein that allows red bloods cells to transport oxygen throughout the body.
- Omega-3 fatty acids are crucial for the development of the nervous system and eye, and overall fetal growth.
- Vitamin D supports the skeletal system, helps to regulate calcium levels by increasing calcium absorption, and may negate adverse pregnancy outcomes including preeclampsia, SGA, preterm birth and gestational diabetes mellitus.
- Ensuring that women of childbearing age receive optimal nutrition should be a priority for health professionals.
3.2. The Postpartum Period: Feeding Baby
- AAP and AND recommend exclusive breastfeeding for 6 months, with continuation for ≥1 year, or as desired by mother.
- Health professionals should be aware of the differences between breast milk and formula to help families make healthy feeding plans for their infants.
- Lactating mothers have increased nutrient needs, and they may be deficient in iron, zinc, and vitamin D.
- While breast milk is the gold standard for feeding, breastfeeding or formula feeding can be the primary source of nutrition for the growing child.
4. Nutrition for the Growing Child, 0–24 Months
- By 6 months of age, infant iron and zinc stores are depleted, therefore foods that contain these nutrients should be preferentially offered.
- From 6 months onward, growth and development continue to be rapid, and nutrients such as protein, calcium and vitamin D are required for accretion of skeletal mass and to help prevent nutritional rickets.
- Omega-3 fatty acids, specifically DHA, are required for continued brain and eye development.
- Carotenoids, such as lutein and zeaxanthin, continue to play key roles in eye and neural development.
5. Nutrient Deficiency During the First 1000 Days
5.1. Maternal and Fetal Nutrient Deficiency
5.1.1. B-Vitamins
5.1.2. Calcium
5.1.3. Carotenoids
5.1.4. Choline
5.1.5. Magnesium
5.1.6. Omega-3 Fatty Acids
5.1.7. Trace Minerals
Zinc
Copper
Iodine
Iron
5.1.8. Vitamin A
5.1.9. Vitamin D
- During the gestational period, expectant mothers can become deficient in various vitamins and minerals, due to the physiological demands of pregnancy.
- Vitamin and mineral deficiencies during pregnancy include folate (especially during the first trimester), vitamin D, iodine, iron, magnesium, choline, carotenoids, and omega-3 fatty acids.
- Deficiency of fat-soluble vitamins including A and D, put the expectant mother at risk for night blindness and the increased risk for adverse pregnancy outcomes, such as preeclampsia or SGA.
- There is a wide range of intakes for many nutrients across the U.S. population, including iron, vitamin B6, folate, vitamin B12, lutein, and zeaxanthin, suggesting pregnant women can benefit from the guidance of health professionals to promote optimal intakes and prevent nutrient excesses.
5.2. Maternal Postnatal Deficiency
5.3. Nutrient Deficiencies in Infants and Toddlers, 0–24 Months
5.3.1. B-Vitamins
5.3.2. Carotenoids
5.3.3. Choline
5.3.4. Magnesium
5.3.5. Omega-3 Fatty Acids
5.3.6. Trace Minerals
Zinc
Copper
Iodine
Iron
5.3.7. Vitamin A
5.3.8. Vitamin D
- Little is known about the prevalence of nutrient deficiencies for infants and toddlers in the United States, making it difficult for healthcare providers to give appropriate dietary guidance to families.
- Nutrient deficiencies in infants and toddlers can have long-term consequences for growth and development as a child and may impact health and wellness as an adult.
- Iodine has not been included in large-scale dietary intake surveys, and little is known about the prevalence or impact of deficiency.
- Intake of nutrients is variable in infants and toddlers and poorly understood, but data suggest children 0–24 months may not be getting enough vitamin D, choline, omega-3 fatty acids, iodine, or iron.
6. Supplementation During the First 1000 Days
6.1. Maternal and Fetal Supplementation
6.1.1. B-Vitamins
Vitamin B6
Vitamin B9 (Folate)
Vitamin B12
6.1.2. Carotenoids
6.1.3. Choline
6.1.4. Magnesium
6.1.5. Omega-3 Fatty Acids
6.1.6. Trace Minerals
Zinc
Copper
Iodine
Iron
6.1.7. Vitamin A
6.1.8. Vitamin D
- Supplementation of fat-soluble vitamins, A and D, during pregnancy may influence maternal and fetal health, especially in areas with a high risk of deficiency.
- Among the trace minerals, zinc and iron supplementation during pregnancy have been extensively studied. Oral zinc supplementation at a dose of 5–50 mg/day shows some benefit for the mother, however more research is needed to understand its effects in baby.
- Iron supplementation can reduce the risk of poor birth outcomes in anemic women.
- Among the B vitamins, the most is known about the benefits of folate (vitamin B9). A recent meta-analysis showed folate supplementation can reduce the risk of LBW and SGA
- Supplementation with omega-3 fatty acids during pregnancy has several benefits for the mother and developing child. These include decreased risk for preterm birth, and a decrease in childhood allergies.
- Choline supplementation during pregnancy may help to optimize cognitive development in the fetus.
6.2. Maternal Postnatal Supplementation
6.2.1. Vitamin A
6.2.2. Vitamin D
6.3. Supplementation During Breastfeeding
6.3.1. Calcium
6.3.2. Carotenoids
6.3.3. Choline
6.3.4. Iodine
6.3.5. Omega-3 Fatty Acids
6.3.6. Vitamin A
6.3.7. Vitamin B12
6.3.8. Vitamin D
- Very little information has been reported in the literature on maternal postnatal health or the potential benefits of supplementation. There is very little information for healthcare providers to help guide mothers in their post-birth recovery or their nutritional transition to motherhood.
- Breast milk lacks key nutrients, including iron, iodine, folic acid and vitamin D, and may have low levels of omega-3 fatty acids. Women who are exclusively breastfeeding their infants should consider taking a supplement to achieve replete status.
6.4. Infants and Toddlers Supplementation, 0–24 Months
6.4.1. Carotenoids
6.4.2. Omega-3 Fatty Acids
6.4.3. Trace Minerals
Zinc
Iron
6.4.4. Vitamin A
6.4.5. Vitamin D
- Clinical trials of nutrient supplementation show that recommended intakes for vitamins and minerals in infants and toddlers can be benefited from further review, including vitamin D, omega-3 fatty acids, iron, and carotenoids.
- The benefits of supplementation for copper, iodine, vitamin B6, folate, vitamin B12, magnesium and choline are poorly understood in infants and toddlers, 0–2 years old.
- Carotenoids are currently used in infant formula, with no adverse health reports.
- For deficient children, nutrient supplementation can impact long-term growth and development, and may influence health and wellness into adulthood.
7. The Role of the Microbiome in Pregnancy, the Postnatal Period, and the Growing Child
- The gut microbiota changes during pregnancy and continues to evolve during the postpartum phase. Changes in microbial diversity and composition have been noted from the first to third trimesters.
- The mother’s fecal, vaginal, oral and skin microbiota have a direct impact on the infant’s microbiota
- Bacterial colonization of the infant’s gastrointestinal tract is influenced by mode of delivery (vaginal vs. caesarean)
- By the end of the first year of life, gut microbiota begins to converge towards an “adult like” profile. Microbiome changes continue through childhood.
8. Supplement Safety
9. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Schwarzenberg, S.J.; Georgieff, M.K. Advocacy for Improving Nutrition in the First 1000 Days to Support Childhood Development and Adult Health. Pediatrics 2018, 141. [Google Scholar] [CrossRef] [PubMed]
- Meo, S.A.; Hassain, A. Metabolic Physiology in Pregnancy. J. Pak. Med Assoc. 2016, 66, S8–S10. [Google Scholar] [PubMed]
- Soma-Pillay, P.; Nelson-Piercy, C.; Tolppanen, H.; Mebazaa, A. Physiological changes in pregnancy. Cardiovasc. J. Afr. 2016, 27, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Shin, G.H.; Toto, E.L.; Schey, R. Pregnancy and postpartum bowel changes: Constipation and fecal incontinence. Am. J. Gastroenterol. 2015, 110, 521–529. [Google Scholar] [CrossRef]
- Lowensohn, R.I.; Stadler, D.D.; Naze, C. Current Concepts of Maternal Nutrition. Obstet. Gynecol. Surv. 2016, 71, 413–426. [Google Scholar] [CrossRef]
- Ackerman, S. Discovering the Brain; National Academies Press (US): Washington, DC, USA, 1992.
- Trumbo, P.; Schlicker, S.; Yates, A.A.; Poos, M. Dietary reference intakes for energy, carbohdrate, fiber, fat, fatty acids, cholesterol, protein and amino acids. J. Acad. Nutr. Diet. 2002, 102, 1621. [Google Scholar]
- Institute of Medicine (US) Standing Committee on the Scientific Evaluation of Dietary Reference Intakes. Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D, and Fluoride; National Academies Press (US): Washington, DC, USA, 1997. [Google Scholar]
- Institute of Medicine (US) Standing Committee on the Scientific Evaluation of Dietary Reference Intakes. Dietary Reference Intakes for Thiamin, Riboflavin, Niacin, Vitamin B6, Folate, Vitamin B12, Pantothenic Acid, Biotin, and Choline; National Academies Press (US): Washington, DC, USA, 1998. [Google Scholar]
- Monsen, E.R. Dietary reference intakes for the antioxidant nutrients: Vitamin C, vitamin E, selenium, and carotenoids. J. Acad. Nutr. Diet. 2000, 100, 637. [Google Scholar]
- Trumbo, P.; Yates, A.A.; Schlicker, S.; Poos, M. Dietary reference intakes: Vitamin A, vitamin K, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium, and zinc. J. Acad. Nutr. Diet. 2001, 101, 294. [Google Scholar]
- Del Valle, H.B.; Yaktine, A.L.; Taylor, C.L.; Ross, A.C. Dietary Reference Intakes for Calcium and Vitamin D.; National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
- Bone, R.; Landrum, J.; Fernandez, L.; Tarsis, S. Analysis of the macular pigment by HPLC: Retinal distribution and age study. Investig. Ophthalmol. Vis. Sci. 1988, 29, 843–849. [Google Scholar]
- Handelman, G.J.; Dratz, E.; Reay, C.; Van Kuijk, J. Carotenoids in the human macula and whole retina. Investig. Ophthalmol. Vis. Sci. 1988, 29, 850–855. [Google Scholar]
- Panova, I.; Tatikolov, A.; Sukhikh, G. Correlation between the content of albumin and carotenoids in human vitreous body during prenatal development. Bull. Exp. Biol. Med. 2007, 144, 681–683. [Google Scholar] [CrossRef] [PubMed]
- Oostenbrug, G.; Mensink, R.; Van Houwelingen, A.; Al, M.; Hornstra, G. Pregnancy-induced hypertension: Maternal and neonatal plasma lipid-soluble antioxidant levels and its relationship with fatty acid unsaturation. Eur. J. Clin. Nutr. 1998, 52, 754. [Google Scholar] [CrossRef] [PubMed]
- Horton, D.K.; Adetona, O.; Aguilar-Villalobos, M.; Cassidy, B.E.; Pfeiffer, C.M.; Schleicher, R.L.; Caldwell, K.L.; Needham, L.L.; Rathbun, S.L.; Vena, J.E. Changes in the concentrations of biochemical indicators of diet and nutritional status of pregnant women across pregnancy trimesters in Trujillo, Peru, 2004–2005. Nutr. J. 2013, 12, 80. [Google Scholar] [CrossRef] [PubMed]
- Picone, S.; Ritieni, A.; Fabiano, A.; Troise, A.D.; Graziani, G.; Paolillo, P.; Volti, G.L.; D’Orazio, N.; Galvano, F.; Gazzolo, D. Arterial cord blood lutein levels in preterm and term healthy newborns are sex and gestational age dependent. Clin. Biochem. 2012, 45, 1558–1563. [Google Scholar] [CrossRef] [PubMed]
- Picone, S.; Ritieni, A.; Fabiano, A.; Graziani, G.; Paolillo, P.; Livolti, G.; Galvano, F.; Gazzolo, D. Lutein levels in arterial cord blood correlate with neuroprotein activin A in healthy preterm and term newborns. Clin. Biochem. 2018, 52, 80–84. [Google Scholar] [CrossRef]
- Yeum, K.-J.; Ferland, G.; Patry, J.; Russell, R.M. Relationship of plasma carotenoids, retinol and tocopherols in mothers and newborn infants. J. Am. Coll. Nutr. 1998, 17, 442–447. [Google Scholar] [CrossRef]
- Kiely, M.; Cogan, P.; Kearney, P.; Morrissey, P. Concentrations of tocopherols and carotenoids in maternal and cord blood plasma. Eur. J. Clin. Nutr. 1999, 53, 711. [Google Scholar] [CrossRef]
- Thoene, M.; Anderson-Berry, A.; Van Ormer, M.; Furtado, J.; Soliman, G.A.; Goldner, W.; Hanson, C. Quantification of Lutein + Zeaxanthin Presence in Human Placenta and Correlations with Blood Levels and Maternal Dietary Intake. Nutrients 2019, 11, 134. [Google Scholar] [CrossRef]
- Crabtree, D.V.; Ojima, I.; Geng, X.; Adler, A.J. Tubulins in the primate retina: Evidence that xanthophylls may be endogenous ligands for the paclitaxel-binding site. Bioorg. Med. Chem. 2001, 9, 1967–1976. [Google Scholar] [CrossRef]
- Stahl, W.; Sies, H. The role of carotenoids and retinoids in gap junctional communication. J. Int. Vitaminol. Nutr. 1998, 68, 354–359. [Google Scholar]
- Lindbergh, C.A.; Mewborn, C.M.; Hammond, B.R.; Renzi-Hammond, L.M.; Curran-Celentano, J.M.; Miller, L.S. Relationship of lutein and zeaxanthin levels to neurocognitive functioning: An fMRI study of older adults. J. Int. Neuropsychol. Soc. 2017, 23, 11–22. [Google Scholar] [CrossRef]
- Gruszecki, W.I.; Strzałka, K. Carotenoids as modulators of lipid membrane physical properties. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2005, 1740, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Zeisel, S.H. Choline: Critical role during fetal development and dietary requirements in adults. Annu. Rev. Nutr. 2006, 26, 229–250. [Google Scholar] [CrossRef]
- Radziejewska, A.; Chmurzynska, A. Folate and choline absorption and uptake: Their role in fetal development. Biochimie 2019, 158, 10–19. [Google Scholar] [CrossRef]
- Wallace, T.C.; Blusztajn, J.K.; Caudill, M.A.; Klatt, K.C.; Natker, E.; Zeisel, S.H.; Zelman, K.M. Choline: The Underconsumed and Underappreciated Essential Nutrient. Nutr. Today 2018, 53, 240–253. [Google Scholar] [CrossRef] [PubMed]
- US Department of Agriculture, Agricultural Research Service. Nutrient Intakes from Food and Beverages: Mean Amounts Consumed per Individual, by Gender and Age, What We Eat in America, NHANES 2015–2016; US Department of Agriculture, Agricultural Research Service: Maryland, MD, USA, 2018. [Google Scholar]
- Elmadfa, I.; Meyer, A.L. Vitamins for the first 1000 days: Preparing for life. J. Int. De Vitaminol. Nutr. 2012, 82, 342–347. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, M.B. The role of iodine in human growth and development. Semin. Cell Dev. Biol. 2011, 22, 645–652. [Google Scholar] [CrossRef] [PubMed]
- Chittimoju, S.B.; Pearce, E.N. Iodine Deficiency and Supplementation in Pregnancy. Clin. Obstet. Gynecol. 2019, 62, 330–338. [Google Scholar] [CrossRef]
- Diemert, A.; Lezius, S.; Pagenkemper, M.; Hansen, G.; Drozdowska, A.; Hecher, K.; Arck, P.; Zyriax, B.C. Maternal nutrition, inadequate gestational weight gain and birth weight: Results from a prospective birth cohort. BMC Pregnancy Childbirth 2016, 16, 224. [Google Scholar] [CrossRef]
- Beard, J.L. Why iron deficiency is important in infant development. J. Nutr. 2008, 138, 2534–2536. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wu, Y.; Li, T.; Wang, X.; Zhu, C. Iron Metabolism and Brain Development in Premature Infants. Front. Physiol. 2019, 10, 463. [Google Scholar] [CrossRef] [PubMed]
- Sachdev, H.; Gera, T.; Nestel, P. Effect of iron supplementation on mental and motor development in children: Systematic review of randomised controlled trials. Public Health Nutr. 2005, 8, 117–132. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, J.A.; Bell, S.J.; Guan, Y.; Yu, Y.H. Folic Acid supplementation and pregnancy: More than just neural tube defect prevention. Rev. Obstet. Gynecol. 2011, 4, 52–59. [Google Scholar] [PubMed]
- Baker, E.J.; Miles, E.A.; Burdge, G.C.; Yaqoob, P.; Calder, P.C. Metabolism and functional effects of plant-derived omega-3 fatty acids in humans. Prog. Lipid Res. 2016, 64, 30–56. [Google Scholar] [CrossRef]
- Ruyle, M.; Connor, W.E.; Anderson, G.J.; Lowensohn, R.I. Placental transfer of essential fatty acids in humans: Venous-arterial difference for docosahexaenoic acid in fetal umbilical erythrocytes. Proc. Natl. Acad. Sci. USA 1990, 87, 7902–7906. [Google Scholar] [CrossRef] [PubMed]
- Clandinin, M.T.; Chappell, J.E.; Leong, S.; Heim, T.; Swyer, P.R.; Chance, G.W. Extrauterine fatty acid accretion in infant brain: Implications for fatty acid requirements. Early Hum. Dev. 1980, 4, 131–138. [Google Scholar] [CrossRef]
- Martinez, M. Tissue levels of polyunsaturated fatty acids during early human development. J. Pediatr. 1992, 120, S129–138. [Google Scholar] [CrossRef]
- Crawford, M.A.; Hassam, A.G.; Williams, G. Essential fatty acids and fetal brain growth. Lancet 1976, 1, 452–453. [Google Scholar] [CrossRef]
- Makrides, M.; Simmer, K.; Goggin, M.; Gibson, R. Erythrocyte docosahexaenoic acid correlates with the visual response of healthy, term infants. Pediatr. Res. 1993, 33, 425. [Google Scholar]
- Elagizi, A.; Lavie, C.J.; Marshall, K.; DiNicolantonio, J.J.; O’Keefe, J.H.; Milani, R.V. Omega-3 Polyunsaturated Fatty Acids and Cardiovascular Health: A Comprehensive Review. Prog. Cardiovasc. Dis. 2018, 61, 76–85. [Google Scholar] [CrossRef] [Green Version]
- Vidakovic, A.J.; Jaddoe, V.W.; Voortman, T.; Demmelmair, H.; Koletzko, B.; Gaillard, R. Maternal plasma polyunsaturated fatty acid levels during pregnancy and childhood lipid and insulin levels. Nutr. Metab. Cardiovasc. Dis. 2017, 27, 78–85. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vidakovic, A.J.; Gishti, O.; Voortman, T.; Felix, J.F.; Williams, M.A.; Hofman, A.; Demmelmair, H.; Koletzko, B.; Tiemeier, H.; Jaddoe, V.W. Maternal plasma PUFA concentrations during pregnancy and childhood adiposity: The Generation R Study. Am. J. Clin. Nutr. 2016, 103, 1017–1025. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Dijk, S.J.; Zhou, J.; Peters, T.J.; Buckley, M.; Sutcliffe, B.; Oytam, Y.; Gibson, R.A.; McPhee, A.; Yelland, L.N.; Makrides, M.; et al. Effect of prenatal DHA supplementation on the infant epigenome: Results from a randomized controlled trial. Clin. Epigenetics 2016, 8, 114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wagner, C.L.; Taylor, S.N.; Dawodu, A.; Johnson, D.D.; Hollis, B.W. Vitamin D and its role during pregnancy in attaining optimal health of mother and fetus. Nutrients 2012, 4, 208–230. [Google Scholar] [CrossRef] [Green Version]
- Agarwal, S.; Kovilam, O.; Agrawal, D.K. Vitamin D and its impact on maternal-fetal outcomes in pregnancy: A critical review. Crit. Rev. Food Sci. Nutr. 2018, 58, 755–769. [Google Scholar] [CrossRef]
- Santamaria, C.; Bi, W.G.; Leduc, L.; Tabatabaei, N.; Jantchou, P.; Luo, Z.C.; Audibert, F.; Nuyt, A.M.; Wei, S.Q. Prenatal vitamin D status and offspring’s growth, adiposity and metabolic health: A systematic review and meta-analysis. Br. J. Nutr. 2018, 119, 310–319. [Google Scholar] [CrossRef]
- Eidelman, A.I. Breastfeeding and the use of human milk: An analysis of the American Academy of Pediatrics 2012 Breastfeeding Policy Statement. Breastfeed. Med. Off. J. Acad. Breastfeed. Med. 2012, 7, 323–324. [Google Scholar] [CrossRef]
- Lessen, R.; Kavanagh, K. Position of the academy of nutrition and dietetics: Promoting and supporting breastfeeding. J. Acad. Nutr. Diet. 2015, 115, 444–449. [Google Scholar] [CrossRef]
- Fewtrell, M.; Bronsky, J.; Campoy, C.; Domellof, M.; Embleton, N.; Fidler Mis, N.; Hojsak, I.; Hulst, J.M.; Indrio, F.; Lapillonne, A.; et al. Complementary Feeding: A Position Paper by the European Society for Paediatric Gastroenterology, Hepatology, and Nutrition (ESPGHAN) Committee on Nutrition. J. Pediatr. Gastroenterol. Nutr. 2017, 64, 119–132. [Google Scholar] [CrossRef]
- Butte, N.F.; Lopez-Alarcon, M.G.; Garza, C. Nutrient Adequacy of Exclusive Breastfeeding for the Term Infant during the First Six Months of Life; WHO: Geneva, Switzerland, 2002. [Google Scholar]
- Critch, J.N.; Canadian Pediatric Society; Nutrition and Gastroenterology Committee. Nutrition for healthy term infants: Recommendations from birth to six months. Can. J. Diet. Pract. Res. 2012, 73, 204. [Google Scholar] [CrossRef] [Green Version]
- Pope, J.; Nizielski, S.; McCook, A. Nutrition for a Changing World; WH Freeman: New York, NY, USA, 2016. [Google Scholar]
- Andreas, N.J.; Kampmann, B.; Mehring Le-Doare, K. Human breast milk: A review on its composition and bioactivity. Early Hum. Dev. 2015, 91, 629–635. [Google Scholar] [CrossRef]
- Butte, N.F.; Hopkinson, J.M.; Wong, W.W.; Smith, E.O.B.; Ellis, K.J. Body composition during the first 2 years of life: An updated reference. Pediatr. Res. 2000, 47, 578. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Minns, L.M.; Kerling, E.H.; Neely, M.R.; Sullivan, D.K.; Wampler, J.L.; Harris, C.L.; Berseth, C.L.; Carlson, S.E. Toddler formula supplemented with docosahexaenoic acid (DHA) improves DHA status and respiratory health in a randomized, double-blind, controlled trial of US children less than 3 years of age. Prostaglandins Leukot. Essent. Fat. Acids 2010, 82, 287–293. [Google Scholar] [CrossRef] [PubMed]
- Buss, C.; Davis, E.P.; Shahbaba, B.; Pruessner, J.C.; Head, K.; Sandman, C.A. Maternal cortisol over the course of pregnancy and subsequent child amygdala and hippocampus volumes and affective problems. Proc. Natl. Acad. Sci. USA 2012, 109, E1312–E1319. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bale, T.L.; Baram, T.Z.; Brown, A.S.; Goldstein, J.M.; Insel, T.R.; McCarthy, M.M.; Nemeroff, C.B.; Reyes, T.M.; Simerly, R.B.; Susser, E.S. Early life programming and neurodevelopmental disorders. Biol. Psychiatry 2010, 68, 314–319. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Picciano, M.F. Pregnancy and lactation: Physiological adjustments, nutritional requirements and the role of dietary supplements. J. Nutr. 2003, 133, 1997S–2002S. [Google Scholar] [CrossRef] [PubMed]
- Pietrobelli, A.; Agosti, M. Nutrition in the first 1000 days: Ten practices to minimize obesity emerging from published science. Int. J. Environ. Res. Public Health 2017, 14, 1491. [Google Scholar] [CrossRef] [Green Version]
- Cusick, S.; Georgieff, M.; Rao, R. Approaches for reducing the risk of early-life iron deficiency-induced brain dysfunction in children. Nutrients 2018, 10, 227. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Arth, A.; Kancherla, V.; Pachon, H.; Zimmerman, S.; Johnson, Q.; Oakley, G.P., Jr. A 2015 global update on folic acid-preventable spina bifida and anencephaly. Birth Defects Res. Part A Clin. Mol. Teratol. 2016, 106, 520–529. [Google Scholar] [CrossRef]
- Lumley, J.; Watson, L.; Watson, M.; Bower, C. Periconceptional supplementation with folate and/or multivitamins for preventing neural tube defects. Cochrane Database Syst. Rev. 2001, Cd001056. [Google Scholar] [CrossRef]
- Wolff, T.; Witkop, C.T.; Miller, T.; Syed, S.B. U.S. Preventive Services Task Force Evidence Syntheses, formerly Systematic Evidence Reviews. In Folic Acid Supplementation for the Prevention of Neural Tube Defects: An Update of the Evidence for the U.S. Preventive Services Task Force; Agency for Healthcare Research and Quality (US): Rockville, MD, USA, 2009. [Google Scholar]
- Wald, N.J.; Morris, J.K.; Blakemore, C. Public health failure in the prevention of neural tube defects: Time to abandon the tolerable upper intake level of folate. Public Health Rev. 2018, 39, 2. [Google Scholar] [CrossRef] [PubMed]
- Gernand, A.D.; Schulze, K.J.; Stewart, C.P.; West, K.P., Jr.; Christian, P. Micronutrient deficiencies in pregnancy worldwide: Health effects and prevention. Nat. Rev. Endocrinol. 2016, 12, 274–289. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bailey, R.; Pac, S.; Fulgoni, V., III; Reidy, K. Total Usual Dietary Intakes of Pregnant Women in the United States, 2001–2014 (FS08-01-19); Oxford University Press: Oxford, UK, 2019. [Google Scholar]
- US Department of Health and Human Services. Food Standards: Amendment of standards of identity for enriched grain products to require addition of folic acid. Fed. Regist. 1996, 61, 8781–8797. [Google Scholar]
- Goonewardene, M.; Shehata, M.; Hamad, A. Anaemia in pregnancy. Best Pract. Res. Clin. Obstet. Gynaecol. 2012, 26, 3–24. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, C.S. Calcium and bone metabolism disorders during pregnancy and lactation. Endocrinol. Metab. Clin. 2011, 40, 795–826. [Google Scholar] [CrossRef]
- Djagbletey, R.; Darkwa, E.O.; deGraft-Johnson, P.K.; Sottie, D.A.Y.; Essuman, R.; Aryee, G.; Aniteye, E. Serum Calcium and Magnesium Levels in Normal Ghanaian Pregnant Women: A Comparative Cross-Sectional Study. Open Access Maced. J. Med Sci. 2018, 6, 2006. [Google Scholar] [CrossRef] [Green Version]
- Milman, N.; Paszkowski, T.; Cetin, I.; Castelo-Branco, C. Supplementation during pregnancy: Beliefs and science. Gynecol. Endocrinol. 2016, 32, 509–516. [Google Scholar] [CrossRef]
- World Health Organization. WHO Recommendation: Calcium Supplementation during Pregnancy for the Prevention of Pre-Eclampsia and its Complications; World Health Organization: Geneva, Switzerland, 2018. [Google Scholar]
- Cohen, J.M.; Kramer, M.S.; Platt, R.W.; Basso, O.; Evans, R.W.; Kahn, S.R. The association between maternal antioxidant levels in midpregnancy and preeclampsia. Am. J. Obstet. Gynecol. 2015, 213, 695.e1–695.e13. [Google Scholar] [CrossRef]
- Cohen, J.M.; Kahn, S.R.; Platt, R.W.; Basso, O.; Evans, R.W.; Kramer, M.S. Small-for-gestational-age birth and maternal plasma antioxidant levels in mid-gestation: A nested case-control study. BJOG Int. J. Obstet. Gynaecol. 2015, 122, 1313–1321. [Google Scholar] [CrossRef]
- Hollenbeck, C.B. The importance of being choline. J. Am. Diet. Assoc. 2010, 110, 1162–1165. [Google Scholar] [CrossRef]
- Brunst, K.J.; Wright, R.O.; DiGioia, K.; Enlow, M.B.; Fernandez, H.; Wright, R.J.; Kannan, S. Racial/ethnic and sociodemographic factors associated with micronutrient intakes and inadequacies among pregnant women in an urban US population. Public Health Nutr. 2014, 17, 1960–1970. [Google Scholar] [CrossRef] [Green Version]
- Caudill, M.A. Pre- and postnatal health: Evidence of increased choline needs. J. Am. Diet. Assoc. 2010, 110, 1198–1206. [Google Scholar] [CrossRef]
- Wallace, T.C.; Fulgoni, V.L. Usual Choline Intakes Are Associated with Egg and Protein Food Consumption in the United States. Nutrients 2017. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Freedman, R.; Hunter, S.K.; Law, A.J.; Wagner, B.D.; D’Alessandro, A.; Christians, U.; Noonan, K.; Wyrwa, A.; Hoffman, M.C. Higher Gestational Choline Levels in Maternal Infection Are Protective for Infant Brain Development. J. Pediatr. 2019, 208, 198–206.e192. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.T.; Dyer, R.A.; King, D.J.; Richardson, K.J.; Innis, S.M. Early second trimester maternal plasma choline and betaine are related to measures of early cognitive development in term infants. PLoS ONE 2012, 7, e43448. [Google Scholar] [CrossRef] [Green Version]
- Mills, J.L.; Fan, R.; Brody, L.C.; Liu, A.; Ueland, P.M.; Wang, Y.; Kirke, P.N.; Shane, B.; Molloy, A.M. Maternal choline concentrations during pregnancy and choline-related genetic variants as risk factors for neural tube defects. Am. J. Clin. Nutr. 2014, 100, 1069–1074. [Google Scholar] [CrossRef]
- Shaw, G.M.; Finnell, R.H.; Blom, H.J.; Carmichael, S.L.; Vollset, S.E.; Yang, W.; Ueland, P.M. Choline and risk of neural tube defects in a folate-fortified population. Epidemiology 2009, 20, 714–719. [Google Scholar] [CrossRef] [Green Version]
- Hung, J.; Abratte, C.M.; Wang, W.; Li, R.; Moriarty, D.J.; Caudill, M.A. Ethnicity and folate influence choline status in young women consuming controlled nutrient intakes. J. Am. Coll. Nutr. 2008, 27, 253–259. [Google Scholar] [CrossRef]
- Wu, B.T.; Innis, S.M.; Mulder, K.A.; Dyer, R.A.; King, D.J. Low plasma vitamin B-12 is associated with a lower pregnancy-associated rise in plasma free choline in Canadian pregnant women and lower postnatal growth rates in their male infants. Am. J. Clin. Nutr. 2013, 98, 1209–1217. [Google Scholar] [CrossRef] [Green Version]
- Schwalfenberg, G.K.; Genuis, S.J. The Importance of Magnesium in Clinical Healthcare. Scientifica 2017, 4179326. [Google Scholar] [CrossRef]
- Bailey, R.L.; Fulgoni, V.L., 3rd; Keast, D.R.; Dwyer, J.T. Dietary supplement use is associated with higher intakes of minerals from food sources. Am. J. Clin. Nutr. 2011, 94, 1376–1381. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mouw, D.R.; Latessa, R.A.; Hickner, J. Clinical inquiries. What are the causes of hypomagnesemia? J. Fam. Pract. 2005, 54, 174–176. [Google Scholar] [PubMed]
- Black, R.E. Micronutrients in pregnancy. Br. J. Nutr. 2001, 85, S193–S197. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seelig, M. Magnesium deficiency in the pathogenesis of disease: Early roots of cardiovascular, skeletal, and renal abnormalities.; Springer Science & Business Media: Boston, MA, USA, 2012. [Google Scholar]
- Stark, K.D.; Van Elswyk, M.E.; Higgins, M.R.; Weatherford, C.A.; Salem, N., Jr. Global survey of the omega-3 fatty acids, docosahexaenoic acid and eicosapentaenoic acid in the blood stream of healthy adults. Prog. Lipid Res. 2016, 63, 132–152. [Google Scholar] [CrossRef] [PubMed]
- Thompson, M.; Hein, N.; Hanson, C.; Smith, L.M.; Anderson-Berry, A.; Richter, C.K.; Nordgren, T.M. Omega-3 fatty acid intake by age, gender, and pregnancy status in the United States: National health and nutrition examination survey 2003–2014. Nutrients 2019, 11, 117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Z.; Fulgoni, V.L.; Kris-Etherton, P.M.; Mitmesser, S.H. Dietary Intakes of EPA and DHA Omega-3 Fatty Acids among US Childbearing-Age and Pregnant Women: An Analysis of NHANES 2001–2014. Nutrients 2018, 10, 416. [Google Scholar] [CrossRef] [Green Version]
- Foreman-van Drongelen, M.M.; Zeijdner, E.E.; van Houwelingen, A.C.; Kester, A.D.; Al, M.D.; Hasaart, T.H.; Hornstra, G. Essential fatty acid status measured in umbilical vessel walls of infants born after a multiple pregnancy. Early Hum. Dev. 1996, 46, 205–215. [Google Scholar] [CrossRef]
- Ackland, M.L.; Michalczyk, A.A. Zinc and infant nutrition. Arch. Biochem. Biophys. 2016, 611, 51–57. [Google Scholar] [CrossRef]
- Keen, C.L.; Uriu-Hare, J.Y.; Hawk, S.N.; Jankowski, M.A.; Daston, G.P.; Kwik-Uribe, C.L.; Rucker, R.B. Effect of copper deficiency on prenatal development and pregnancy outcome. Am. J. Clin. Nutr. 1998, 67, 1003s–1011s. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blumfield, M.L.; Hure, A.J.; Macdonald-Wicks, L.; Smith, R.; Collins, C.E. A systematic review and meta-analysis of micronutrient intakes during pregnancy in developed countries. Nutr. Rev. 2013, 71, 118–132. [Google Scholar] [CrossRef]
- Corbo, M.D.; Lam, J. Zinc deficiency and its management in the pediatric population: A literature review and proposed etiologic classification. J. Am. Acad. Dermatol. 2013, 69, 616–624.e611. [Google Scholar] [CrossRef]
- Erdman, J.W., Jr.; MacDonald, I.A.; Zeisel, S.H. Present Knowledge in Nutrition; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Azizi, F.; Smyth, P. Breastfeeding and maternal and infant iodine nutrition. Clin. Endocrinol. 2009, 70, 803–809. [Google Scholar] [CrossRef] [PubMed]
- Caldwell, K.L.; Makhmudov, A.; Ely, E.; Jones, R.L.; Wang, R.Y. Iodine status of the US population, National Health and Nutrition Examination Survey, 2005–2006 and 2007–2008. Thyroid 2011, 21, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Caldwell, K.L.; Pan, Y.; Mortensen, M.E.; Makhmudov, A.; Merrill, L.; Moye, J. Iodine status in pregnant women in the National Children’s Study and in U.S. women (15–44 years), National Health and Nutrition Examination Survey 2005–2010. Thyroid 2013, 23, 927–937. [Google Scholar] [CrossRef]
- Zimmermann, M.B. Iodine deficiency in pregnancy and the effects of maternal iodine supplementation on the offspring: A review. Am. J. Clin. Nutr. 2009, 89, 668s–672s. [Google Scholar] [CrossRef] [Green Version]
- Mei, Z.; Cogswell, M.E.; Looker, A.C.; Pfeiffer, C.M.; Cusick, S.E.; Lacher, D.A.; Grummer-Strawn, L.M. Assessment of iron status in US pregnant women from the National Health and Nutrition Examination Survey (NHANES), 1999–2006. Am. J. Clin. Nutr. 2011, 93, 1312–1320. [Google Scholar] [CrossRef]
- Di Renzo, G.C.; Spano, F.; Giardina, I.; Brillo, E.; Clerici, G.; Roura, L.C. Iron deficiency anemia in pregnancy. Women’s Health 2015, 11, 891–900. [Google Scholar] [CrossRef]
- WHO. Global Prevalence of Vitamin A Deficiency in Populations at risk 1995–2005: WHO Global Database on Vitamin A Deficiency; WHO, Ed.; WHO: Geneva, Switzerland, 2009. [Google Scholar]
- De Oliveira, M.R. Vitamin A and retinoids as mitochondrial toxicants. Oxidative Med. Cell. Longev. 2015. [Google Scholar] [CrossRef]
- McCauley, M.E.; van den Broek, N.; Dou, L.; Othman, M. Vitamin A supplementation during pregnancy for maternal and newborn outcomes. Cochrane Database Syst. Rev. 2015, Cd008666. [Google Scholar] [CrossRef]
- Heyden, E.L.; Wimalawansa, S.J. Vitamin D: Effects on human reproduction, pregnancy, and fetal well-being. J. Steroid Biochem. Mol. Biol. 2018, 180, 41–50. [Google Scholar] [CrossRef]
- Hollis, B.W.; Wagner, C.L.; Howard, C.R.; Ebeling, M.; Shary, J.R.; Smith, P.G.; Taylor, S.N.; Morella, K.; Lawrence, R.A.; Hulsey, T.C. Maternal Versus Infant Vitamin D Supplementation During Lactation: A Randomized Controlled Trial. Pediatrics 2015, 136, 625–634. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saraf, R.; Morton, S.M.; Camargo, C.A., Jr.; Grant, C.C. Global summary of maternal and newborn vitamin D status—A systematic review. Matern. Child Nutr. 2016, 12, 647–668. [Google Scholar] [CrossRef] [PubMed]
- Lerchbaum, E.; Obermayer-Pietsch, B. Vitamin D and fertility: A systematic review. Eur. J. Endocrinol. 2012, 166, 765–778. [Google Scholar] [CrossRef]
- Von Websky, K.; Hasan, A.A.; Reichetzeder, C.; Tsuprykov, O.; Hocher, B. Impact of vitamin D on pregnancy-related disorders and on offspring outcome. J. Steroid Biochem. Mol. Biol. 2018, 180, 51–64. [Google Scholar] [CrossRef]
- Dovnik, A.; Mujezinović, F. The association of vitamin D levels with common pregnancy complications. Nutrients 2018, 10, 867. [Google Scholar] [CrossRef] [Green Version]
- Brummelte, S.; Galea, L.A. Postpartum depression: Etiology, treatment and consequences for maternal care. Horm. Behav. 2016, 77, 153–166. [Google Scholar] [CrossRef]
- McKinney, J.; Keyser, L.; Clinton, S.; Pagliano, C. ACOG Committee Opinion No. 736: Optimizing Postpartum Care. Obstet. Gynecol. 2018, 132, 784–785. [Google Scholar] [CrossRef]
- Tully, K.P.; Stuebe, A.M.; Verbiest, S.B. The fourth trimester: A critical transition period with unmet maternal health needs. Am. J. Obstet. Gynecol. 2017, 217, 37–41. [Google Scholar] [CrossRef]
- Bjørke Monsen, A.L.; Ueland, P.M. Homocysteine and methylmalonic acid in diagnosis and risk assessment from infancy to adolescence. Am. J. Clin. Nutr. 2003, 78, 7–21. [Google Scholar] [CrossRef] [Green Version]
- Ahluwalia, N.; Herrick, K.A.; Rossen, L.M.; Rhodes, D.; Kit, B.; Moshfegh, A.; Dodd, K.W. Usual nutrient intakes of US infants and toddlers generally meet or exceed Dietary Reference Intakes: Findings from NHANES 2009–2012. Am. J. Clin. Nutr. 2016, 104, 1167–1174. [Google Scholar] [CrossRef] [Green Version]
- Bailey, R.L.; Catellier, D.J.; Jun, S.; Dwyer, J.T.; Jacquier, E.F.; Anater, A.S.; Eldridge, A.L. Total Usual Nutrient Intakes of US Children (Under 48 Months): Findings from the Feeding Infants and Toddlers Study (FITS) 2016. J. Nutr. 2018, 148, 1557S–1566S. [Google Scholar] [CrossRef] [PubMed]
- Davis, K.E.; Li, X.; Adams-Huet, B.; Sandon, L. Infant feeding practices and dietary consumption of US infants and toddlers: National Health and Nutrition Examination Survey (NHANES) 2003–2012. Public Health Nutr. 2018, 21, 711–720. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Honzik, T.; Adamovicova, M.; Smolka, V.; Magner, M.; Hruba, E.; Zeman, J. Clinical presentation and metabolic consequences in 40 breastfed infants with nutritional vitamin B12 deficiency--what have we learned? Eur. J. Paediatr. Neurol. Off. J. Eur. Paediatr. Neurol. Soc. 2010, 14, 488–495. [Google Scholar] [CrossRef] [PubMed]
- Bone, R.A.; Landrum, J.T.; Tarsis, S.L. Preliminary identification of the human macular pigment. Vision Res 1985, 25, 1531–1535. [Google Scholar] [CrossRef]
- Teller, D.Y. First glances: The vision of infants. the Friedenwald lecture. Investig. Ophthalmol. Vis. Sci. 1997, 38, 2183–2203. [Google Scholar]
- Zimmer, J.P.; Hammond, B.R., Jr. Possible influences of lutein and zeaxanthin on the developing retina. Clin. Ophthalmol. 2007, 1, 25–35. [Google Scholar]
- Lien, E.L.; Hammond, B.R. Nutritional influences on visual development and function. Prog. Retin. Eye Res. 2011. [Google Scholar] [CrossRef]
- Hendrickson, A.; Possin, D.; Vajzovic, L.; Toth, C.A. Histologic development of the human fovea from midgestation to maturity. Am. J. Ophthalmol. 2012, 154, 767–778.e762. [Google Scholar] [CrossRef] [Green Version]
- Bernstein, P.S.; Sharifzadeh, M.; Liu, A.; Ermakov, I.; Nelson, K.; Sheng, X.; Panish, C.; Carlstrom, B.; Hoffman, R.O.; Gellermann, W. Blue-light reflectance imaging of macular pigment in infants and children. Investig. Ophthalmol. Vis. Sci. 2013, 54, 4034–4040. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Banks, M.S.; Bennett, P.J. Optical and photoreceptor immaturities limit the spatial and chromatic vision of human neonates. J. Opt. Soc. Am. A 1988, 5, 2059–2079. [Google Scholar] [CrossRef] [Green Version]
- Hammond, B.R., Jr. Possible role for dietary lutein and zeaxanthin in visual development. Nutr. Rev. 2008, 66, 695–702. [Google Scholar] [CrossRef] [PubMed]
- Hendrickson, A.E.; Yuodelis, C. The morphological development of the human fovea. Ophthalmology 1984, 91, 603–612. [Google Scholar] [CrossRef]
- Snodderly, D.M.; Auran, J.D.; Delori, F.C. The macular pigment. II. Spatial distribution in primate retinas. Investig. Ophthalmol. Vis. Sci. 1984, 25, 674–685. [Google Scholar]
- Snodderly, D.M.; Brown, P.K.; Delori, F.C.; Auran, J.D. The macular pigment. I. Absorbance spectra, localization, and discrimination from other yellow pigments in primate retinas. Investig. Ophthalmol. Vis. Sci. 1984, 25, 660–673. [Google Scholar]
- Li, B.; Ahmed, F.; Bernstein, P.S. Studies on the singlet oxygen scavenging mechanism of human macular pigment. Arch. Biochem. Biophys. 2010, 504, 56–60. [Google Scholar] [CrossRef] [Green Version]
- Mohn, E.S.; Erdman, J.W., Jr.; Kuchan, M.J.; Neuringer, M.; Johnson, E.J. Lutein accumulates in subcellular membranes of brain regions in adult rhesus macaques: Relationship to DHA oxidation products. PLoS ONE 2017, 12, e0186767. [Google Scholar] [CrossRef] [Green Version]
- Li, S.Y.; Fung, F.K.; Fu, Z.J.; Wong, D.; Chan, H.H.; Lo, A.C. Anti-inflammatory effects of lutein in retinal ischemic/hypoxic injury: In vivo and in vitro studies. Investig. Ophthalmol. Vis. Sci. 2012, 53, 5976–5984. [Google Scholar] [CrossRef] [Green Version]
- Sasaki, M.; Ozawa, Y.; Kurihara, T.; Noda, K.; Imamura, Y.; Kobayashi, S.; Ishida, S.; Tsubota, K. Neuroprotective effect of an antioxidant, lutein, during retinal inflammation. Investig. Ophthalmol. Vis. Sci. 2009, 50, 1433–1439. [Google Scholar] [CrossRef] [Green Version]
- Ilcol, Y.O.; Ozbek, R.; Hamurtekin, E.; Ulus, I.H. Choline status in newborns, infants, children, breast-feeding women, breast-fed infants and human breast milk. J. Nutr. Biochem. 2005, 16, 489–499. [Google Scholar] [CrossRef]
- Meck, W.H.; Williams, C.L.; Cermak, J.M.; Blusztajn, J.K. Developmental periods of choline sensitivity provide an ontogenetic mechanism for regulating memory capacity and age-related dementia. Front. Integr. Neurosci. 2007, 1, 7. [Google Scholar] [CrossRef] [Green Version]
- Grimes, C.; Szymlek-Gay, E.; Campbell, K.; Nicklas, T. Food sources of total energy and nutrients among US infants and toddlers: National Health and Nutrition Examination Survey 2005–2012. Nutrients 2015, 7, 6797–6836. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crawford, M.A. Essential fatty acids and neurodevelopmental disorder. Adv. Exp. Med. Biol. 1992, 318, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Forsyth, S.; Gautier, S.; Salem, N., Jr. Estimated dietary intakes of arachidonic acid and docosahexaenoic acid in infants and young children living in developing countries. Ann. Nutr. Metab. 2016, 69, 64–74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ozden, T.A.; Gokcay, G.; Cantez, M.S.; Durmaz, O.; Issever, H.; Omer, B.; Saner, G. Copper, zinc and iron levels in infants and their mothers during the first year of life: A prospective study. BMC Pediatr. 2015, 15, 157. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Au, L.E.; Gurzo, K.; Paolicelli, C.; Whaley, S.E.; Weinfield, N.S.; Ritchie, L.D. Diet Quality of US Infants and Toddlers 7–24 Months Old in the WIC Infant and Toddler Feeding Practices Study-2. J. Nutr. 2018, 148, 1786–1793. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hamner, H.C.; Perrine, C.G.; Scanlon, K.S. Usual Intake of Key Minerals among Children in the Second Year of Life, NHANES 2003–2012. Nutrients 2016, 8, 468. [Google Scholar] [CrossRef] [Green Version]
- Terrin, G.; Berni Canani, R.; Di Chiara, M.; Pietravalle, A.; Aleandri, V.; Conte, F.; De Curtis, M. Zinc in Early Life: A Key Element in the Fetus and Preterm Neonate. Nutrients 2015, 7, 10427–10446. [Google Scholar] [CrossRef]
- Krebs, N.F. Update on zinc deficiency and excess in clinical pediatric practice. Ann. Nutr. Metab. 2013, 62 (Suppl. 1), 19–29. [Google Scholar] [CrossRef]
- Cetin, I.; Berti, C.; Calabrese, S. Role of micronutrients in the periconceptional period. Hum. Reprod. Update 2009, 16, 80–95. [Google Scholar] [CrossRef] [Green Version]
- Shazia, Q.; Mohammad, Z.H.; Rahman, T.; Shekhar, H.U. Correlation of oxidative stress with serum trace element levels and antioxidant enzyme status in Beta thalassemia major patients: A review of the literature. Anemia 2012, 2012, 270923. [Google Scholar] [CrossRef] [Green Version]
- Pehrsson, P.R.; Patterson, K.Y.; Spungen, J.H.; Wirtz, M.S.; Andrews, K.W.; Dwyer, J.T.; Swanson, C.A. Iodine in food- and dietary supplement-composition databases. Am. J. Clin. Nutr. 2016, 104 (Suppl. 3), 868s–876s. [Google Scholar] [CrossRef] [Green Version]
- Fallah, R.; Du, L.; Braverman, L.E.; He, X.; Segura-Harrison, M.; Yeh, M.W.; Pearce, E.N.; Chiu, H.K.; Mittelman, S.D.; Leung, A.M. Iodine Nutrition in Weaning Infants in the United States. Thyroid 2019, 29, 573–576. [Google Scholar] [CrossRef]
- Melse-Boonstra, A.; Jaiswal, N. Iodine deficiency in pregnancy, infancy and childhood and its consequences for brain development. Best Pract. Res. Clin. Endocrinol. Metab. 2010, 24, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Pearce, E.N.; Leung, A.M.; Blount, B.C.; Bazrafshan, H.R.; He, X.; Pino, S.; Valentin-Blasini, L.; Braverman, L.E. Breast milk iodine and perchlorate concentrations in lactating Boston-area women. J. Clin. Endocrinol. Metab. 2007, 92, 1673–1677. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trumpff, C.; De Schepper, J.; Tafforeau, J.; Van Oyen, H.; Vanderfaeillie, J.; Vandevijvere, S. Mild iodine deficiency in pregnancy in Europe and its consequences for cognitive and psychomotor development of children: A review. J. Trace Elem. Med. Biol. 2013, 27, 174–183. [Google Scholar] [CrossRef]
- Angermayr, L.; Clar, C. Iodine supplementation for preventing iodine deficiency disorders in children. Cochrane Database Syst. Rev. 2004, Cd003819. [Google Scholar] [CrossRef] [PubMed]
- Velasco, I.; Bath, S.C.; Rayman, M.P. Iodine as Essential Nutrient during the First 1000 Days of Life. Nutrients 2018, 10, 290. [Google Scholar] [CrossRef] [Green Version]
- Gordon, R.C.; Rose, M.C.; Skeaff, S.A.; Gray, A.R.; Morgan, K.M.; Ruffman, T. Iodine supplementation improves cognition in mildly iodine-deficient children. Am. J. Clin. Nutr. 2009, 90, 1264–1271. [Google Scholar] [CrossRef]
- Brotanek, J.M.; Gosz, J.; Weitzman, M.; Flores, G. Iron deficiency in early childhood in the United States: Risk factors and racial/ethnic disparities. Pediatrics 2007, 120, 568–575. [Google Scholar] [CrossRef] [Green Version]
- Baker, R.D.; Greer, F.R. Diagnosis and prevention of iron deficiency and iron-deficiency anemia in infants and young children (0-3 years of age). Pediatrics 2010, 126, 1040–1050. [Google Scholar] [CrossRef] [Green Version]
- Bartleman, J.J.M. Infant and child nutrition. Medicine 2019, 47, 195–198. [Google Scholar] [CrossRef]
- Ahluwalia, N. Nutrition Monitoring of Children Aged Birth to 24 Mo (B-24): Data Collection and Findings from the NHANES. Adv. Nutr. 2019. [Google Scholar] [CrossRef] [Green Version]
- Vinkhuyzen, A.A.E.; Eyles, D.W.; Burne, T.H.; Blanken, L.M.E.; Kruithof, C.J.; Verhulst, F.; Jaddoe, V.W.; Tiemeier, H.; McGrath, J.J. Prevalence and predictors of vitamin D deficiency based on maternal mid-gestation and neonatal cord bloods: The Generation R Study. J. Steroid Biochem. Mol. Biol. 2016, 164, 161–167. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.M.; Smith, J.R.; Philipp, B.L.; Chen, T.C.; Mathieu, J.; Holick, M.F. Vitamin D deficiency in a healthy group of mothers and newborn infants. Clin. Pediatr. 2007, 46, 42–44. [Google Scholar] [CrossRef]
- Feldman-Winter, L.B.; Schanler, R.J.; O’Connor, K.G.; Lawrence, R.A. Pediatricians and the promotion and support of breastfeeding. Arch. Pediatr. Adolesc. Med. 2008, 162, 1142–1149. [Google Scholar] [CrossRef] [Green Version]
- Salam, R.A.; Zuberi, N.F.; Bhutta, Z.A. Pyridoxine (vitamin B6) supplementation during pregnancy or labour for maternal and neonatal outcomes. Cochrane Database Syst. Rev. 2015, Cd000179. [Google Scholar] [CrossRef]
- Haider, B.A.; Bhutta, Z.A. Multiple-micronutrient supplementation for women during pregnancy. Cochrane Database Syst. Rev. 2017, 4, Cd004905. [Google Scholar] [CrossRef]
- De-Regil, L.M.; Fernandez-Gaxiola, A.C.; Dowswell, T.; Pena-Rosas, J.P. Effects and safety of periconceptional folate supplementation for preventing birth defects. Cochrane Database Syst. Rev. 2010, Cd007950. [Google Scholar] [CrossRef]
- Ashokkumar, B.; Mohammed, Z.M.; Vaziri, N.D.; Said, H.M. Effect of folate oversupplementation on folate uptake by human intestinal and renal epithelial cells. Am. J. Clin. Nutr. 2007, 86, 159–166. [Google Scholar] [CrossRef] [Green Version]
- Duggan, C.; Srinivasan, K.; Thomas, T.; Samuel, T.; Rajendran, R.; Muthayya, S.; Finkelstein, J.L.; Lukose, A.; Fawzi, W.; Allen, L.H.; et al. Vitamin B-12 supplementation during pregnancy and early lactation increases maternal, breast milk, and infant measures of vitamin B-12 status. J. Nutr. 2014, 144, 758–764. [Google Scholar] [CrossRef] [Green Version]
- Siddiqua, T.J.; Ahmad, S.M.; Ahsan, K.B.; Rashid, M.; Roy, A.; Rahman, S.M.; Shahab-Ferdows, S.; Hampel, D.; Ahmed, T.; Allen, L.H.; et al. Vitamin B12 supplementation during pregnancy and postpartum improves B12 status of both mothers and infants but vaccine response in mothers only: A randomized clinical trial in Bangladesh. Eur. J. Nutr. 2016, 55, 281–293. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, K.; Thomas, T.; Kapanee, A.R.; Ramthal, A.; Bellinger, D.C.; Bosch, R.J.; Kurpad, A.V.; Duggan, C. Effects of maternal vitamin B12 supplementation on early infant neurocognitive outcomes: A randomized controlled clinical trial. Matern. Child Nutr. 2017, 13. [Google Scholar] [CrossRef] [PubMed]
- Lorenzoni, F.; Giampietri, M.; Ferri, G.; Lunardi, S.; Madrigali, V.; Battini, L.; Boldrini, A.; Ghirri, P. Lutein administration to pregnant women with gestational diabetes mellitus is associated to a decrease of oxidative stress in newborns. Gynecol. Endocrinol. 2013. [Google Scholar] [CrossRef] [PubMed]
- Oostenbrug, G.S.; Mensink, R.P.; Al, M.D.; van Houwelingen, A.C.; Hornstra, G. Maternal and neonatal plasma antioxidant levels in normal pregnancy, and the relationship with fatty acid unsaturation. Br. J. Nutr. 1998, 80, 67–73. [Google Scholar] [CrossRef] [Green Version]
- Cena, H.; Castellazzi, A.M.; Pietri, A.; Roggi, C.; Turconi, G. Lutein concentration in human milk during early lactation and its relationship with dietary lutein intake. Public Health Nutr. 2009, 12, 1878–1884. [Google Scholar] [CrossRef] [Green Version]
- Johnson, E.J.; Maras, J.E.; Rasmussen, H.M.; Tucker, K.L. Intake of lutein and zeaxanthin differ with age, sex, and ethnicity. J. Am. Diet. Assoc. 2010, 110, 1357–1362. [Google Scholar] [CrossRef]
- Donaldson, M.S. A carotenoid health index based on plasma carotenoids and health outcomes. Nutrients 2011, 3, 1003–1022. [Google Scholar] [CrossRef] [Green Version]
- Cheatham, C.L.; Goldman, B.D.; Fischer, L.M.; da Costa, K.A.; Reznick, J.S.; Zeisel, S.H. Phosphatidylcholine supplementation in pregnant women consuming moderate-choline diets does not enhance infant cognitive function: A randomized, double-blind, placebo-controlled trial. Am. J. Clin. Nutr. 2012, 96, 1465–1472. [Google Scholar] [CrossRef]
- Caudill, M.A.; Strupp, B.J.; Muscalu, L.; Nevins, J.E.H.; Canfield, R.L. Maternal choline supplementation during the third trimester of pregnancy improves infant information processing speed: A randomized, double-blind, controlled feeding study. Faseb J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2018, 32, 2172–2180. [Google Scholar] [CrossRef] [Green Version]
- Ross, R.G.; Hunter, S.K.; McCarthy, L.; Beuler, J.; Hutchison, A.K.; Wagner, B.D.; Leonard, S.; Stevens, K.E.; Freedman, R. Perinatal choline effects on neonatal pathophysiology related to later schizophrenia risk. Am. J. Psychiatry 2013, 170, 290–298. [Google Scholar] [CrossRef]
- Boeke, C.E.; Gillman, M.W.; Hughes, M.D.; Rifas-Shiman, S.L.; Villamor, E.; Oken, E. Choline intake during pregnancy and child cognition at age 7 years. Am. J. Epidemiol. 2012, 177, 1338–1347. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rylander, R. Treatment with Magnesium in Pregnancy. Aims Public Health 2015, 2, 804–809. [Google Scholar] [CrossRef] [PubMed]
- Makrides, M.; Crosby, D.D.; Bain, E.; Crowther, C.A. Magnesium supplementation in pregnancy. Cochrane Database Syst. Rev. 2014, Cd000937. [Google Scholar] [CrossRef] [PubMed]
- Asemi, Z.; Karamali, M.; Jamilian, M.; Foroozanfard, F.; Bahmani, F.; Heidarzadeh, Z.; Benisi-Kohansal, S.; Surkan, P.J.; Esmaillzadeh, A. Magnesium supplementation affects metabolic status and pregnancy outcomes in gestational diabetes: A randomized, double-blind, placebo-controlled trial. Am. J. Clin. Nutr. 2015, 102, 222–229. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koletzko, B.; Lien, E.; Agostoni, C.; Bohles, H.; Campoy, C.; Cetin, I.; Decsi, T.; Dudenhausen, J.W.; Dupont, C.; Forsyth, S.; et al. The roles of long-chain polyunsaturated fatty acids in pregnancy, lactation and infancy: Review of current knowledge and consensus recommendations. J. Perinat. Med. 2008, 36, 5–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Collins, C.T.; Gibson, R.A.; McPhee, A.J.; Makrides, M. The role of long chain polyunsaturated fatty acids in perinatal nutrition. Semin. Perinatol. 2019, 151156. [Google Scholar] [CrossRef] [PubMed]
- Olsen, S.F.; Secher, N.J. Low consumption of seafood in early pregnancy as a risk factor for preterm delivery: Prospective cohort study. BMJ 2002, 324, 447. [Google Scholar] [CrossRef] [Green Version]
- Olsen, S.F.; Halldorsson, T.I.; Thorne-Lyman, A.L.; Strom, M.; Gortz, S.; Granstrom, C.; Nielsen, P.H.; Wohlfahrt, J.; Lykke, J.A.; Langhoff-Roos, J.; et al. Plasma Concentrations of Long Chain N-3 Fatty Acids in Early and Mid-Pregnancy and Risk of Early Preterm Birth. EBioMedicine 2018, 35, 325–333. [Google Scholar] [CrossRef] [Green Version]
- Makrides, M.; Gibson, R.A.; McPhee, A.J.; Yelland, L.; Quinlivan, J.; Ryan, P. Effect of DHA supplementation during pregnancy on maternal depression and neurodevelopment of young children: A randomized controlled trial. Jama 2010, 304, 1675–1683. [Google Scholar] [CrossRef] [Green Version]
- Carlson, S.E.; Colombo, J.; Gajewski, B.J.; Gustafson, K.M.; Mundy, D.; Yeast, J.; Georgieff, M.K.; Markley, L.A.; Kerling, E.H.; Shaddy, D.J. DHA supplementation and pregnancy outcomes. Am. J. Clin. Nutr. 2013, 97, 808–815. [Google Scholar] [CrossRef] [Green Version]
- Smuts, C.M.; Huang, M.; Mundy, D.; Plasse, T.; Major, S.; Carlson, S.E. A randomized trial of docosahexaenoic acid supplementation during the third trimester of pregnancy. Obstet. Gynecol. 2003, 101, 469–479. [Google Scholar] [CrossRef] [PubMed]
- Palmer, D.J.; Sullivan, T.; Gold, M.S.; Prescott, S.L.; Heddle, R.; Gibson, R.A.; Makrides, M. Effect of n-3 long chain polyunsaturated fatty acid supplementation in pregnancy on infants’ allergies in first year of life: Randomised controlled trial. BMJ 2012, 344, e184. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stratakis, N.; Roumeliotaki, T.; Oken, E.; Ballester, F.; Barros, H.; Basterrechea, M.; Cordier, S.; de Groot, R.; den Dekker, H.T.; Duijts, L.; et al. Fish and seafood consumption during pregnancy and the risk of asthma and allergic rhinitis in childhood: A pooled analysis of 18 European and US birth cohorts. Int. J. Epidemiol. 2017, 46, 1465–1477. [Google Scholar] [CrossRef] [PubMed]
- Hess, S.Y.; King, J.C. Effects of maternal zinc supplementation on pregnancy and lactation outcomes. Food Nutr. Bull. 2009, 30, S60–78. [Google Scholar] [CrossRef]
- Chaffee, B.W.; King, J.C. Effect of zinc supplementation on pregnancy and infant outcomes: A systematic review. Paediatr. Perinat. Epidemiol. 2012, 26 (Suppl. 1), 118–137. [Google Scholar] [CrossRef] [Green Version]
- Ota, E.; Mori, R.; Middleton, P.; Tobe-Gai, R.; Mahomed, K.; Miyazaki, C.; Bhutta, Z.A. Zinc supplementation for improving pregnancy and infant outcome. Cochrane Database Syst. Rev. 2015, Cd000230. [Google Scholar] [CrossRef]
- Karamali, M.; Heidarzadeh, Z.; Seifati, S.M.; Samimi, M.; Tabassi, Z.; Hajijafari, M.; Asemi, Z.; Esmaillzadeh, A. Zinc supplementation and the effects on metabolic status in gestational diabetes: A randomized, double-blind, placebo-controlled trial. J. Diabetes Complicat. 2015, 29, 1314–1319. [Google Scholar] [CrossRef]
- Blumberg, J.; Frei, B.; Fulgoni, V.; Weaver, C.; Zeisel, S. Contribution of dietary supplements to nutritional adequacy in various adult age groups. Nutrients 2017, 9, 1325. [Google Scholar] [CrossRef] [Green Version]
- Wilson, R.L.; Bianco-Miotto, T.; Leemaqz, S.Y.; Grzeskowiak, L.E.; Dekker, G.A.; Roberts, C.T. Early pregnancy maternal trace mineral status and the association with adverse pregnancy outcome in a cohort of Australian women. J. Trace Elem. Med. Biol. 2018, 46, 103–109. [Google Scholar] [CrossRef]
- Pretell, E.; Artidoro, C. Impairment of mental development by iodine deficiency and its correlation. A retrospective view of studies in Peru, In: The Damaged Brain of Iodine Deficiency: Cognitive, Behavioral, Neuromotor, Educative Aspects; Cognizant Communication Corp: Putnam Valley, NY, USA, 1994. [Google Scholar]
- Pretell, E.A.; Torres, T.; Zenteno, V.; Cornejo, M. Prophylaxis of endemic goiter with iodized oil in rural Peru. Adv. Exp. Med. Biol. 1972, 30, 246–265. [Google Scholar]
- Zhou, S.J.; Anderson, A.J.; Gibson, R.A.; Makrides, M. Effect of iodine supplementation in pregnancy on child development and other clinical outcomes: A systematic review of randomized controlled trials. Am. J. Clin. Nutr. 2013, 98, 1241–1254. [Google Scholar] [CrossRef] [PubMed]
- Taylor, P.N.; Okosieme, O.E.; Murphy, R.; Hales, C.; Chiusano, E.; Maina, A.; Joomun, M.; Bestwick, J.P.; Smyth, P.; Paradice, R.; et al. Maternal perchlorate levels in women with borderline thyroid function during pregnancy and the cognitive development of their offspring: Data from the Controlled Antenatal Thyroid Study. J. Clin. Endocrinol. Metab. 2014, 99, 4291–4298. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention. Recommendations to prevent and control iron deficiency in the United States. MMWR RR 1998, 47, 1–29. [Google Scholar]
- Siu, A.L. Screening for Iron Deficiency Anemia in Young Children: USPSTF Recommendation Statement. Pediatrics 2015, 136, 746–752. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pena-Rosas, J.P.; De-Regil, L.M.; Dowswell, T.; Viteri, F.E. Daily oral iron supplementation during pregnancy. Cochrane Database Syst. Rev. 2012, 12, Cd004736. [Google Scholar] [CrossRef] [PubMed]
- Banhidy, F.; Acs, N.; Puho, E.H.; Czeizel, A.E. Iron deficiency anemia: Pregnancy outcomes with or without iron supplementation. Nutrition 2011, 27, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Aranda, N.; Ribot, B.; Garcia, E.; Viteri, F.E.; Arija, V. Pre-pregnancy iron reserves, iron supplementation during pregnancy, and birth weight. Early Hum. Dev. 2011, 87, 791–797. [Google Scholar] [CrossRef]
- Chatterjee, R.; Shand, A.; Nassar, N.; Walls, M.; Khambalia, A.Z. Iron supplement use in pregnancy—Are the right women taking the right amount? Clin. Nutr. 2016, 35, 741–747. [Google Scholar] [CrossRef]
- Cantor, A.G.; Bougatsos, C.; McDonagh, M. Routine Iron Supplementation and Screening for Iron Deficiency Anemia in Pregnancy. Ann. Intern. Med. 2015, 163, 400. [Google Scholar] [CrossRef]
- Cruz, S.; da Cruz, S.P.; Ramalho, A. Impact of Vitamin A Supplementation on Pregnant Women and on Women Who Have Just Given Birth: A Systematic Review. J. Am. Coll. Nutr. 2018, 37, 243–250. [Google Scholar] [CrossRef]
- Christian, P.; Labrique, A.B.; Ali, H.; Richman, M.J.; Wu, L.; Rashid, M.; West, K.P., Jr. Maternal vitamin A and beta-carotene supplementation and risk of bacterial vaginosis: A randomized controlled trial in rural Bangladesh. Am. J. Clin. Nutr. 2011, 94, 1643–1649. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Neves, P.A.; Saunders, C.; Barros, D.C.; Ramalho, A. Vitamin A supplementation in Brazilian pregnant and postpartum women: A systematic review. Rev. Bras. De Epidemiol. Braz. J. Epidemiol. 2015, 18, 824–836. [Google Scholar] [CrossRef] [Green Version]
- West, K.P., Jr.; Christian, P.; Labrique, A.B.; Rashid, M.; Shamim, A.A.; Klemm, R.D.; Massie, A.B.; Mehra, S.; Schulze, K.J.; Ali, H.; et al. Effects of vitamin A or beta carotene supplementation on pregnancy-related mortality and infant mortality in rural Bangladesh: A cluster randomized trial. Jama 2011, 305, 1986–1995. [Google Scholar] [CrossRef] [PubMed]
- Oliveira-Menegozzo, J.M.; Bergamaschi, D.P.; Middleton, P.; East, C.E. Vitamin A supplementation for postpartum women. Cochrane Database Syst. Rev. 2010. [Google Scholar] [CrossRef] [Green Version]
- Thorne-Lyman, A.L.; Fawzi, W.W. Vitamin A and carotenoids during pregnancy and maternal, neonatal and infant health outcomes: A systematic review and meta-analysis. Paediatr. Perinat. Epidemiol. 2012, 26 (Suppl 1), 36–54. [Google Scholar] [CrossRef]
- Roth, D.E.; Leung, M.; Mesfin, E.; Qamar, H.; Watterworth, J.; Papp, E. Vitamin D supplementation during pregnancy: State of the evidence from a systematic review of randomised trials. BMJ 2017, 359, 5237. [Google Scholar] [CrossRef] [Green Version]
- Fu, Z.M.; Ma, Z.Z.; Liu, G.J.; Wang, L.L.; Guo, Y. Vitamins supplementation affects the onset of preeclampsia. J. Formos. Med Assoc. Taiwan Yi Zhi 2018, 117, 6–13. [Google Scholar] [CrossRef]
- De-Regil, L.M.; Palacios, C.; Lombardo, L.K.; Pena-Rosas, J.P. Vitamin D supplementation for women during pregnancy. Cochrane Database Syst. Rev. 2016, 1. [Google Scholar] [CrossRef]
- Asemi, Z.; Hashemi, T.; Karamali, M.; Samimi, M.; Esmaillzadeh, A. Effects of vitamin D supplementation on glucose metabolism, lipid concentrations, inflammation, and oxidative stress in gestational diabetes: A double-blind randomized controlled clinical trial. Am. J. Clin. Nutr. 2013, 98, 1425–1432. [Google Scholar] [CrossRef] [Green Version]
- Tchum, S.K.; Tanumihardjo, S.A.; Newton, S.; de Benoist, B.; Owusu-Agyei, S.; Arthur, F.K.; Tetteh, A. Evaluation of vitamin A supplementation regimens in Ghanaian postpartum mothers with the use of the modified-relative-dose-response test. Am. J. Clin. Nutr. 2006, 84, 1344–1349. [Google Scholar] [CrossRef]
- Fernandes, T.F.; Figueiroa, J.N.; Grande de Arruda, I.K.; Diniz Ada, S. Effect on infant illness of maternal supplementation with 400 000 IU vs 200 000 IU of vitamin A. Pediatrics 2012, 129, e960–966. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernandes, T.F.; Andreto, L.M.; Vieira, C.S.; de Arruda, I.K.; Diniz Ada, S. Serum retinol concentrations in mothers and newborns at delivery in a public maternity hospital in Recife, northeast Brazil. J. HealthPopul. Nutr. 2014, 32, 28–35. [Google Scholar]
- Gould, J.F.; Makrides, M.; Colombo, J.; Smithers, L.G. Randomized controlled trial of maternal omega-3 long-chain PUFA supplementation during pregnancy and early childhood development of attention, working memory, and inhibitory control. Am. J. Clin. Nutr. 2014, 99, 851–859. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gould, J.F.; Anderson, A.J.; Yelland, L.N.; Smithers, L.G.; Skeaff, C.M.; Zhou, S.J.; Gibson, R.A.; Makrides, M. Association of cord blood vitamin D with early childhood growth and neurodevelopment. J. Paediatr. Child Health 2017, 53, 75–83. [Google Scholar] [CrossRef]
- Diogenes, M.E.; Bezerra, F.F.; Rezende, E.P.; Taveira, M.F.; Pinhal, I.; Donangelo, C.M. Effect of calcium plus vitamin D supplementation during pregnancy in Brazilian adolescent mothers: A randomized, placebo-controlled trial. Am. J. Clin. Nutr. 2013, 98, 82–91. [Google Scholar] [CrossRef] [Green Version]
- Ares Segura, S.; Arena Ansotegui, J.; Diaz-Gomez, N.M. The importance of maternal nutrition during breastfeeding: Do breastfeeding mothers need nutritional supplements? An. Pediatr. 2016, 84, 347.e1–347.e7. [Google Scholar] [CrossRef]
- Chierici, R.; Saccomandi, D.; Vigi, V. Dietary supplements for the lactating mother: Influence on the trace element content of milk. Acta Paediatr. 1999, 88, 7–13. [Google Scholar] [CrossRef]
- Bae, Y.J.; Kratzsch, J. Vitamin D and calcium in the human breast milk. Best Pract. Res. Clin. Endocrinol. Metab. 2018, 32, 39–45. [Google Scholar] [CrossRef]
- Kovacs, C.S. Maternal vitamin D deficiency: Fetal and neonatal implications. Semin. Fetal Neonatal Med. 2013, 18, 129–135. [Google Scholar] [CrossRef]
- Bolzetta, F.; Veronese, N.; De Rui, M.; Berton, L.; Carraro, S.; Pizzato, S.; Girotti, G.; De Ronch, I.; Manzato, E.; Coin, A. Duration of breastfeeding as a risk factor for vertebral fractures. Bone 2014, 68, 41–45. [Google Scholar] [CrossRef]
- Jarjou, L.M.; Sawo, Y.; Goldberg, G.R.; Laskey, M.A.; Cole, T.J.; Prentice, A. Unexpected long-term effects of calcium supplementation in pregnancy on maternal bone outcomes in women with a low calcium intake: A follow-up study. Am. J. Clin. Nutr. 2013, 98, 723–730. [Google Scholar] [CrossRef] [Green Version]
- Patton, S.; Canfield, L.M.; Huston, G.E.; Ferris, A.M.; Jensen, R.G. Carotenoids of human colostrum. Lipids 1990, 25, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Schweigert, F.J.; Bathe, K.; Chen, F.; Buscher, U.; Dudenhausen, J.W. Effect of the stage of lactation in humans on carotenoid levels in milk, blood plasma and plasma lipoprotein fractions. Eur. J. Nutr. 2004, 43, 39–44. [Google Scholar] [CrossRef] [PubMed]
- Rios, J.J.; Xavier, A.A.O.; Diaz-Salido, E.; Arenilla-Velez, I.; Jaren-Galan, M.; Garrido-Fernandez, J.; Aguayo-Maldonado, J.; Perez-Galvez, A. Xanthophyll esters are found in human colostrum. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xavier, A.A.O.; Díaz-Salido, E.; Arenilla-Vélez, I.; Aguayo-Maldonado, J.; Garrido-Fernández, J.; Fontecha, J.; Sánchez-García, A.; Pérez-Gálvez, A. Carotenoid Content in Human Colostrum is Associated to Preterm/Full-Term Birth Condition. Nutrients 2018, 10, 1654. [Google Scholar] [CrossRef] [Green Version]
- Canfield, L.M.; Clandinin, M.T.; Davies, D.P.; Fernandez, M.C.; Jackson, J.; Hawkes, J.; Goldman, W.J.; Pramuk, K.; Reyes, H.; Sablan, B.; et al. Multinational study of major breast milk carotenoids of healthy mothers. Eur. J. Nutr. 2003, 42, 133–141. [Google Scholar]
- Canfield, L.M.; Giuliano, A.R.; Neilson, E.M.; Blashil, B.M.; Graver, E.J.; Yap, H.H. Kinetics of the response of milk and serum beta-carotene to daily beta-carotene supplementation in healthy, lactating women. Am. J. Clin. Nutr. 1998, 67, 276–283. [Google Scholar] [CrossRef] [PubMed]
- Canfield, L.M.; Taren, D.L.; Kaminsky, R.G.; Mahal, Z. Short-term beta-carotene supplementation of lactating mothers consuming diets low in vitamin A. J. Nutr. Biochem. 1999, 10, 532–538. [Google Scholar] [CrossRef]
- Jackson, J.G.; Zimmer, J.P. Lutein and zeaxanthin in human milk independently and significantly differ among women from Japan, Mexico, and the United Kingdom. Nutr. Res. 2007, 27, 449–453. [Google Scholar] [CrossRef]
- Lietz, G.; Mulokozi, G.; Henry, J.C.; Tomkins, A.M. Xanthophyll and hydrocarbon carotenoid patterns differ in plasma and breast milk of women supplemented with red palm oil during pregnancy and lactation. J. Nutr. 2006, 136, 1821–1827. [Google Scholar] [CrossRef] [Green Version]
- Zielinska, M.A.; Hamulka, J.; Grabowicz-Chadrzynska, I.; Brys, J.; Wesolowska, A. Association between Breastmilk LC PUFA, Carotenoids and Psychomotor Development of Exclusively Breastfed Infants. Int. J. Env. Res. Public Health 2019, 16, 1144. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Macias, C.; Schweigert, F.J. Changes in the concentration of carotenoids, vitamin A, alpha-tocopherol and total lipids in human milk throughout early lactation. Ann. Nutr. Metab. 2001, 45, 82–85. [Google Scholar] [CrossRef] [PubMed]
- Lipkie, T.E.; Morrow, A.L.; Jouni, Z.E.; McMahon, R.J.; Ferruzzi, M.G. Longitudinal Survey of Carotenoids in Human Milk from Urban Cohorts in China, Mexico, and the USA. PLoS ONE 2015, 10, e0127729. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giuliano, A.R.; Neilson, E.M.; Yap, H.-H.; Baier, M.; Canfield, L.M. Quantitation of and inter/intra-individual variability in major carotenoids of mature human milk. J. Nutr. Biochem. 1994, 5, 551–556. [Google Scholar] [CrossRef]
- De Azeredo, V.B.; Trugo, N.M. Retinol, carotenoids, and tocopherols in the milk of lactating adolescents and relationships with plasma concentrations. Nutrition 2008, 24, 133–139. [Google Scholar] [CrossRef]
- Jackson, J.G.; Lien, E.L.; White, S.J.; Bruns, N.J.; Kuhlman, C.F. Major carotenoids in mature human milk: longitudinal and diurnal patterns. Nutr Biochem 1998, 9, 2–7. [Google Scholar] [CrossRef]
- Haftel, L.; Berkovich, Z.; Reifen, R. Elevated milk beta-carotene and lycopene after carrot and tomato paste supplementation. Nutrition 2015, 31, 443–445. [Google Scholar] [CrossRef]
- Nagayama, J.; Noda, K.; Uchikawa, T.; Maruyama, I.; Shimomura, H.; Miyahara, M. Effect of maternal Chlorella supplementation on carotenoid concentration in breast milk at early lactation. Int. J. Food Sci. Nutr. 2014, 65, 573–576. [Google Scholar] [CrossRef]
- Sherry, C.L.; Oliver, J.S.; Renzi, L.M.; Marriage, B.J. Lutein supplementation increases breast milk and plasma lutein concentrations in lactating women and infant plasma concentrations but does not affect other carotenoids. J. Nutr. 2014, 144, 1256–1263. [Google Scholar] [CrossRef] [Green Version]
- Bouhouch, R.R.; Bouhouch, S.; Cherkaoui, M.; Aboussad, A.; Stinca, S.; Haldimann, M.; Andersson, M.; Zimmermann, M.B. Direct iodine supplementation of infants versus supplementation of their breastfeeding mothers: A double-blind, randomised, placebo-controlled trial. Lancet Diabetes Endocrinol. 2014, 2, 197–209. [Google Scholar] [CrossRef]
- Makrides, M.; Neumann, M.A.; Gibson, R.A. Effect of maternal docosahexaenoic acid (DHA) supplementation on breast milk composition. Eur. J. Clin. Nutr. 1996, 50, 352–357. [Google Scholar] [PubMed]
- Brenna, J.T.; Varamini, B.; Jensen, R.G.; Diersen-Schade, D.A.; Boettcher, J.A.; Arterburn, L.M. Docosahexaenoic and arachidonic acid concentrations in human breast milk worldwide. Am. J. Clin. Nutr. 2007, 85, 1457–1464. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gibson, R.A.; Neumann, M.; Makrides, M. Effect of increasing breast milk docosahexaenoic acid on plasma and erythrocyte phospholipid fatty acids and neural indices of exclusively breast fed infants. Eur. J. Clin. Nutr. 1997, 51, 578. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Urrutia-Pereira, M.; Solé, D. Vitamin D deficiency in pregnancy and its impact on the fetus, the newborn and in childhood. Rev. Paul. De Pediatr. 2015, 33, 104–113. [Google Scholar] [CrossRef] [Green Version]
- Roth, D.E.; Gernand, A.D.; Al Mahmud, A. Vitamin D Supplementation in Pregnancy and Lactation and Infant Growth. New Engl. J. Med. 2018, 379, 1881. [Google Scholar] [CrossRef] [PubMed]
- Bettler, J.; Zimmer, J.P.; Neuringer, M.; DeRusso, P.A. Serum lutein concentrations in healthy term infants fed human milk or infant formula with lutein. Eur. J. Nutr. 2010, 49, 45–51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kon, I.Y.; Gmoshinskaya, M.V.; Safronova, A.I.; Alarcon, P.; Vandenplas, Y. Growth and Tolerance Assessment of a Lutein-fortified Infant Formula. Pediatr. Gastroenterol. Hepatol. Nutr. 2014, 17, 104–111. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mackey, A.D.; Albrecht, D.; Oliver, J.; Williams, T.; Long, A.C.; Price, P.T. Plasma carotenoid concentrations of infants are increased by feeding a milk-based infant formula supplemented with carotenoids. J. Sci. Food. Agric. 2013, 93, 1945–1952. [Google Scholar] [CrossRef]
- Manzoni, P.; Guardione, R.; Bonetti, P.; Priolo, C.; Maestri, A.; Mansoldo, C.; Mostert, M.; Anselmetti, G.; Sardei, D.; Bellettato, M.; et al. Lutein and Zeaxanthin Supplementation in Preterm Very Low-Birth-Weight Neonates in Neonatal Intensive Care Units: A Multicenter Randomized Controlled Trial. Am. J. Perinatol. 2012. [Google Scholar] [CrossRef] [Green Version]
- Perrone, S.; Longini, M.; Marzocchi, B.; Picardi, A.; Bellieni, C.V.; Proietti, F.; Rodriguez, A.; Turrisi, G.; Buonocore, G. Effects of Lutein on Oxidative Stress in the Term Newborn: A Pilot Study. Neonatology 2010, 97, 36–40. [Google Scholar] [CrossRef]
- Perrone, S.; Tei, M.; Longini, M.; Santacroce, A.; Turrisi, G.; Proietti, F.; Felici, C.; Picardi, A.; Bazzini, F.; Vasarri, P.; et al. Lipid and protein oxidation in newborn infants after lutein administration. Oxidative Med. Cell. Longev. 2014, 2014, 781454. [Google Scholar] [CrossRef] [Green Version]
- Rubin, L.P.; Chan, G.M.; Barrett-Reis, B.M.; Fulton, A.B.; Hansen, R.M.; Ashmeade, T.L.; Oliver, J.S.; Mackey, A.D.; Dimmit, R.A.; Hartmann, E.E.; et al. Effect of carotenoid supplementation on plasma carotenoids, inflammation and visual development in preterm infants. J. Perinatol. 2012, 32, 418–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kabali, H.; Irigoyen, M.; Nunez-Davis, R.; Budacki, J.; Mohanty, S.; Leister, K.; Bonner, R. Exposure and use of mobile media devices by young children. Pediatrics 2015, 136, 1044–1050. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stringham, J.; Stringham, N.; O’Brien, K. Macular carotenoid supplementation improves visual performance, sleep quality, and adverse physical symptoms in those with high screen time exposure. Foods 2017, 6, 47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malinow, M.R.; Feeney-Burns, L.; Peterson, L.H.; Klein, M.L.; Neuringer, M. Diet-related macular anomalies in monkeys. Investig. Ophthalmol. Vis. Sci. 1980, 19, 857–863. [Google Scholar]
- Leung, I.Y.; Sandstrom, M.M.; Zucker, C.L.; Neuringer, M.; Snodderly, D.M. Nutritional manipulation of primate retinas, II: Effects of age, n-3 fatty acids, lutein, and zeaxanthin on retinal pigment epithelium. Investig. Ophthalmol. Vis. Sci. 2004, 45, 3244–3256. [Google Scholar] [CrossRef]
- Feeney-Burns, L.; Neuringer, M.; Gao, C.L. Macular pathology in monkeys fed semipurified diets. Prog. Clin. Biol. Res. 1989, 314, 601–622. [Google Scholar]
- Vishwanathan, R.; Kuchan, M.J.; Sen, S.; Johnson, E.J. Lutein and preterm infants with decreased concentrations of brain carotenoids. J. Pediatric Gastroenterol. Nutr. 2014, 59, 659–665. [Google Scholar] [CrossRef]
- Johnson, E.J.; Vishwanathan, R.; Johnson, M.A.; Hausman, D.B.; Davey, A.; Scott, T.M.; Green, R.C.; Miller, L.S.; Gearing, M.; Woodard, J.; et al. Relationship between Serum and Brain Carotenoids, alpha-Tocopherol, and Retinol Concentrations and Cognitive Performance in the Oldest Old from the Georgia Centenarian Study. J. Aging Res. 2013, 2013, 951786. [Google Scholar] [CrossRef] [Green Version]
- Lieblein-Boff, J.C.; Johnson, E.J.; Kennedy, A.D.; Lai, C.S.; Kuchan, M.J. Exploratory Metabolomic Analyses Reveal Compounds Correlated with Lutein Concentration in Frontal Cortex, Hippocampus, and Occipital Cortex of Human Infant Brain. PLoS ONE 2015, 10, e0136904. [Google Scholar] [CrossRef] [Green Version]
- Cheatham, C.L.; Sheppard, K.W. Synergistic Effects of Human Milk Nutrients in the Support of Infant Recognition Memory: An Observational Study. Nutrients 2015, 7, 9079–9095. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carlson, S.E.; Werkman, S.H.; Tolley, E.A. Effect of long-chain n-3 fatty acid supplementation on visual acuity and growth of preterm infants with and without bronchopulmonary dysplasia. Am. J. Clin. Nutr. 1996, 63, 687–697. [Google Scholar] [CrossRef] [PubMed]
- Birch, E.E.; Hoffman, D.R.; Uauy, R.; Birch, D.G.; Prestidge, C. Visual acuity and the essentiality of docosahexaenoic acid and arachidonic acid in the diet of term infants. Pediatr. Res. 1998, 44, 201–209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Birch, E.E.; Hoffman, D.R.; Castaneda, Y.S.; Fawcett, S.L.; Birch, D.G.; Uauy, R.D. A randomized controlled trial of long-chain polyunsaturated fatty acid supplementation of formula in term infants after weaning at 6 wk of age. Am. J. Clin. Nutr. 2002, 75, 570–580. [Google Scholar] [CrossRef] [Green Version]
- Birch, E.E.; Garfield, S.; Hoffman, D.R.; Uauy, R.; Birch, D.G. A randomized controlled trial of early dietary supply of long-chain polyunsaturated fatty acids and mental development in term infants. Dev. Med. Child Neurol. 2000, 42, 174–181. [Google Scholar] [CrossRef]
- Smithers, L.G.; Gibson, R.A.; McPhee, A.; Makrides, M. Higher dose of docosahexaenoic acid in the neonatal period improves visual acuity of preterm infants: Results of a randomized controlled trial. Am. J. Clin. Nutr. 2008, 88, 1049–1056. [Google Scholar] [CrossRef] [Green Version]
- Smithers, L.G.; Gibson, R.A.; Makrides, M. Maternal supplementation with docosahexaenoic acid during pregnancy does not affect early visual development in the infant: A randomized controlled trial. Am. J. Clin. Nutr. 2011, 93, 1293–1299. [Google Scholar] [CrossRef] [Green Version]
- Makrides, M.; Gibson, R.A.; McPhee, A.J.; Collins, C.T.; Davis, P.G.; Doyle, L.W.; Simmer, K.; Colditz, P.B.; Morris, S.; Smithers, L.G.; et al. Neurodevelopmental outcomes of preterm infants fed high-dose docosahexaenoic acid: A randomized controlled trial. Jama 2009, 301, 175–182. [Google Scholar] [CrossRef] [Green Version]
- Colombo, J.; Carlson, S.E.; Cheatham, C.L.; Shaddy, D.J.; Kerling, E.H.; Thodosoff, J.M.; Gustafson, K.M.; Brez, C. Long-term effects of LCPUFA supplementation on childhood cognitive outcomes. Am. J. Clin. Nutr. 2013, 98, 403–412. [Google Scholar] [CrossRef] [Green Version]
- Manley, B.J.; Makrides, M.; Collins, C.T.; McPhee, A.J.; Gibson, R.A.; Ryan, P.; Sullivan, T.R.; Davis, P.G. High-dose docosahexaenoic acid supplementation of preterm infants: Respiratory and allergy outcomes. Pediatrics 2011, 128, e71–e77. [Google Scholar] [CrossRef]
- Foiles, A.M.; Kerling, E.H.; Wick, J.A.; Scalabrin, D.M.; Colombo, J.; Carlson, S.E. Formula with long-chain polyunsaturated fatty acids reduces incidence of allergy in early childhood. Pediatr. Allergy Immunol. 2016, 27, 156–161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Birch, E.E.; Khoury, J.C.; Berseth, C.L.; Castaneda, Y.S.; Couch, J.M.; Bean, J.; Tamer, R.; Harris, C.L.; Mitmesser, S.H.; Scalabrin, D.M. The impact of early nutrition on incidence of allergic manifestations and common respiratory illnesses in children. J. Pediatr. 2010, 156, 902–906.e901. [Google Scholar] [CrossRef] [PubMed]
- Liberato, S.C.; Singh, G.; Mulholland, K. Zinc supplementation in young children: A review of the literature focusing on diarrhoea prevention and treatment. Clin. Nutr. 2015, 34, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.H.; Peerson, J.M.; Baker, S.K.; Hess, S.Y. Preventive zinc supplementation among infants, preschoolers, and older prepubertal children. Food Nutr. Bull. 2009, 30, S12–S40. [Google Scholar] [CrossRef] [PubMed]
- Liu, E.; Pimpin, L.; Shulkin, M.; Kranz, S.; Duggan, C.P.; Mozaffarian, D.; Fawzi, W.W. Effect of Zinc Supplementation on Growth Outcomes in Children under 5 Years of Age. Nutrients 2018, 10, 377. [Google Scholar] [CrossRef] [Green Version]
- Cho, J.M.; Kim, J.Y.; Yang, H.R. Effects of oral zinc supplementation on zinc status and catch-up growth during the first 2 years of life in children with non-organic failure to thrive born preterm and at term. Pediatrics Neonatol. 2019, 60, 201–209. [Google Scholar] [CrossRef] [Green Version]
- Dobe, M.; Garg, P.; Bhalla, G. Fortification as an effective strategy to bridge iron gaps during complementary feeding. Clin. Epidemiol. Glob. Health 2018, 168–171. [Google Scholar] [CrossRef]
- Domellof, M.; Braegger, C.; Campoy, C.; Colomb, V.; Decsi, T.; Fewtrell, M.; Hojsak, I.; Mihatsch, W.; Molgaard, C.; Shamir, R.; et al. Iron requirements of infants and toddlers. J. Pediatr. Gastroenterol. Nutr. 2014, 58, 119–129. [Google Scholar] [CrossRef]
- Szajewska, H.; Ruszczynski, M.; Chmielewska, A. Effects of iron supplementation in nonanemic pregnant women, infants, and young children on the mental performance and psychomotor development of children: A systematic review of randomized controlled trials. Am. J. Clin. Nutr. 2010, 91, 1684–1690. [Google Scholar] [CrossRef] [Green Version]
- Lozoff, B.; Jiang, Y.; Li, X.; Zhou, M.; Richards, B.; Xu, G.; Clark, K.M.; Liang, F.; Kaciroti, N.; Zhao, G.; et al. Low-Dose Iron Supplementation in Infancy Modestly Increases Infant Iron Status at 9 Mo without Decreasing Growth or Increasing Illness in a Randomized Clinical Trial in Rural China. J. Nutr. 2016, 146, 612–621. [Google Scholar] [CrossRef] [Green Version]
- Ziegler, E.E.; Nelson, S.E.; Jeter, J.M. Iron supplementation of breastfed infants from an early age. Am. J. Clin. Nutr. 2009, 89, 525–532. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berglund, S.K.; Chmielewska, A.; Starnberg, J.; Westrup, B.; Hägglöf, B.; Norman, M.; Domellöf, M.J.P.R. Effects of iron supplementation of low-birth-weight infants on cognition and behavior at 7 years: A randomized controlled trial. Pediatr. Res. 2018, 83, 111. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pongcharoen, T.; DiGirolamo, A.M.; Ramakrishnan, U.; Winichagoon, P.; Flores, R.; Martorell, R. Long-term effects of iron and zinc supplementation during infancy on cognitive function at 9 y of age in northeast Thai children: A follow-up study. Am. J. Clin. Nutr. 2011, 93, 636–643. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Checkley, W.; West, K.P., Jr.; Wise, R.A.; Baldwin, M.R.; Wu, L.; LeClerq, S.C.; Christian, P.; Katz, J.; Tielsch, J.M.; Khatry, S.; et al. Maternal vitamin A supplementation and lung function in offspring. New Engl. J. Med. 2010, 362, 1784–1794. [Google Scholar] [CrossRef] [Green Version]
- Malaba, L.C.; Iliff, P.J.; Nathoo, K.J.; Marinda, E.; Moulton, L.H.; Zijenah, L.S.; Zvandasara, P.; Ward, B.J.; Humphrey, J.H. Effect of postpartum maternal or neonatal vitamin A supplementation on infant mortality among infants born to HIV-negative mothers in Zimbabwe. Am. J. Clin. Nutr. 2005, 81, 454–460. [Google Scholar] [CrossRef] [Green Version]
- Wagner, C.L.; Greer, F.R. Prevention of rickets and vitamin D deficiency in infants, children, and adolescents. Pediatrics 2008, 122, 1142–1152. [Google Scholar] [CrossRef] [Green Version]
- Trilok-Kumar, G.; Kaur, M.; Rehman, A.M.; Arora, H.; Rajput, M.M.; Chugh, R.; Kurpad, A.; Sachdev, H.S.; Filteau, S. Effects of vitamin D supplementation in infancy on growth, bone parameters, body composition and gross motor development at age 3–6 years: Follow-up of a randomized controlled trial. Int. J. Epidemiol. 2015, 44, 894–905. [Google Scholar] [CrossRef]
- Gallo, S.; Hazell, T.; Vanstone, C.A.; Agellon, S.; Jones, G.; L’Abbe, M.; Rodd, C.; Weiler, H.A. Vitamin D supplementation in breastfed infants from Montreal, Canada: 25-hydroxyvitamin D and bone health effects from a follow-up study at 3 years of age. Osteoporos. Int. 2016, 27, 2459–2466. [Google Scholar] [CrossRef]
- Gallo, S.; Comeau, K.; Vanstone, C.; Agellon, S.; Sharma, A.; Jones, G.; L’Abbe, M.; Khamessan, A.; Rodd, C.; Weiler, H. Effect of different dosages of oral vitamin D supplementation on vitamin D status in healthy, breastfed infants: A randomized trial. Jama 2013, 309, 1785–1792. [Google Scholar] [CrossRef] [Green Version]
- Holmlund-Suila, E.; Viljakainen, H.; Hytinantti, T.; Lamberg-Allardt, C.; Andersson, S.; Makitie, O. High-dose vitamin d intervention in infants--effects on vitamin d status, calcium homeostasis, and bone strength. J. Clin. Endocrinol. Metab. 2012, 97, 4139–4147. [Google Scholar] [CrossRef] [Green Version]
- Perrine, C.G.; Sharma, A.J.; Jefferds, M.E.; Serdula, M.K.; Scanlon, K.S. Adherence to vitamin D recommendations among US infants. Pediatrics 2010, 125, 627–632. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dawodu, A.; Zalla, L.; Woo, J.G.; Herbers, P.M.; Davidson, B.S.; Heubi, J.E.; Morrow, A.L.J.M. Heightened attention to supplementation is needed to improve the vitamin D status of breastfeeding mothers and infants when sunshine exposure is restricted. Matern. Child Nutr. 2014, 10, 383–397. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Butel, M.J.; Waligora-Dupriet, A.J.; Wydau-Dematteis, S. The developing gut microbiota and its consequences for health. J. Dev. Orig. Health Dis. 2018, 9, 590–597. [Google Scholar] [CrossRef] [PubMed]
- Collado, M.C.; Rautava, S.; Aakko, J.; Isolauri, E.; Salminen, S. Human gut colonisation may be initiated in utero by distinct microbial communities in the placenta and amniotic fluid. Sci. Rep. 2016, 6, 23129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jimenez, E.; Fernandez, L.; Marin, M.L.; Martin, R.; Odriozola, J.M.; Nueno-Palop, C.; Narbad, A.; Olivares, M.; Xaus, J.; Rodriguez, J.M. Isolation of commensal bacteria from umbilical cord blood of healthy neonates born by cesarean section. Curr. Microbiol. 2005, 51, 270–274. [Google Scholar] [CrossRef]
- Stinson, L.F.; Keelan, J.A.; Payne, M.S. Characterization of the bacterial microbiome in first-pass meconium using propidium monoazide (PMA) to exclude nonviable bacterial DNA. Lett. Appl. Microbiol. 2019, 68, 378–385. [Google Scholar] [CrossRef]
- Koren, O.; Goodrich, J.K.; Cullender, T.C.; Spor, A.; Laitinen, K.; Backhed, H.K.; Gonzalez, A.; Werner, J.J.; Angenent, L.T.; Knight, R.; et al. Host remodeling of the gut microbiome and metabolic changes during pregnancy. Cell 2012, 150, 470–480. [Google Scholar] [CrossRef] [Green Version]
- Crusell, M.K.W.; Hansen, T.H.; Nielsen, T.; Allin, K.H.; Ruhlemann, M.C.; Damm, P.; Vestergaard, H.; Rorbye, C.; Jorgensen, N.R.; Christiansen, O.B.; et al. Gestational diabetes is associated with change in the gut microbiota composition in third trimester of pregnancy and postpartum. Microbiome 2018, 6, 89. [Google Scholar] [CrossRef]
- Jost, T.; Lacroix, C.; Braegger, C.; Chassard, C. Stability of the maternal gut microbiota during late pregnancy and early lactation. Curr. Microbiol. 2014, 68, 419–427. [Google Scholar] [CrossRef] [Green Version]
- Hermansson, H.; Kumar, H.; Collado, M.C.; Salminen, S.; Isolauri, E.; Rautava, S. Breast Milk Microbiota Is Shaped by Mode of Delivery and Intrapartum Antibiotic Exposure. Front. Nutr. 2019, 6, 4. [Google Scholar] [CrossRef] [Green Version]
- Kumar, H.; du Toit, E.; Kulkarni, A.; Aakko, J.; Linderborg, K.M.; Zhang, Y.; Nicol, M.P.; Isolauri, E.; Yang, B.; Collado, M.C.; et al. Distinct Patterns in Human Milk Microbiota and Fatty Acid Profiles Across Specific Geographic Locations. Front. Microbiol. 2016, 7, 1619. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robertson, R.C.; Manges, A.R.; Finlay, B.B.; Prendergast, A.J. The Human Microbiome and Child Growth—First 1000 Days and Beyond. Trends Microbiol. 2019, 27, 131–147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferretti, P.; Pasolli, E.; Tett, A.; Asnicar, F.; Gorfer, V.; Fedi, S.; Armanini, F.; Truong, D.T.; Manara, S.; Zolfo, M.; et al. Mother-to-Infant Microbial Transmission from Different Body Sites Shapes the Developing Infant Gut Microbiome. Cell Host Microbe 2018, 24, 133–145.e135. [Google Scholar] [CrossRef]
- Tamburini, S.; Shen, N.; Wu, H.C.; Clemente, J.C. The microbiome in early life: Implications for health outcomes. Nat. Med. 2016, 22, 713–722. [Google Scholar] [CrossRef] [PubMed]
- Backhed, F.; Roswall, J.; Peng, Y.; Feng, Q.; Jia, H.; Kovatcheva-Datchary, P.; Li, Y.; Xia, Y.; Xie, H.; Zhong, H.; et al. Dynamics and Stabilization of the Human Gut Microbiome during the First Year of Life. Cell Host Microbe 2015, 17, 852. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bokulich, N.A.; Chung, J.; Battaglia, T.; Henderson, N.; Jay, M.; Li, H.; Lieber, A.D.; Wu, F.; Perez-Perez, G.I.; Chen, Y.; et al. Antibiotics, birth mode, and diet shape microbiome maturation during early life. Sci. Transl. Med. 2016, 8, 343ra82. [Google Scholar] [CrossRef] [Green Version]
- Hollister, E.B.; Riehle, K.; Luna, R.A.; Weidler, E.M.; Rubio-Gonzales, M.; Mistretta, T.A.; Raza, S.; Doddapaneni, H.V.; Metcalf, G.A.; Muzny, D.M.; et al. Structure and function of the healthy pre-adolescent pediatric gut microbiome. Microbiome 2015, 3, 36. [Google Scholar] [CrossRef] [Green Version]
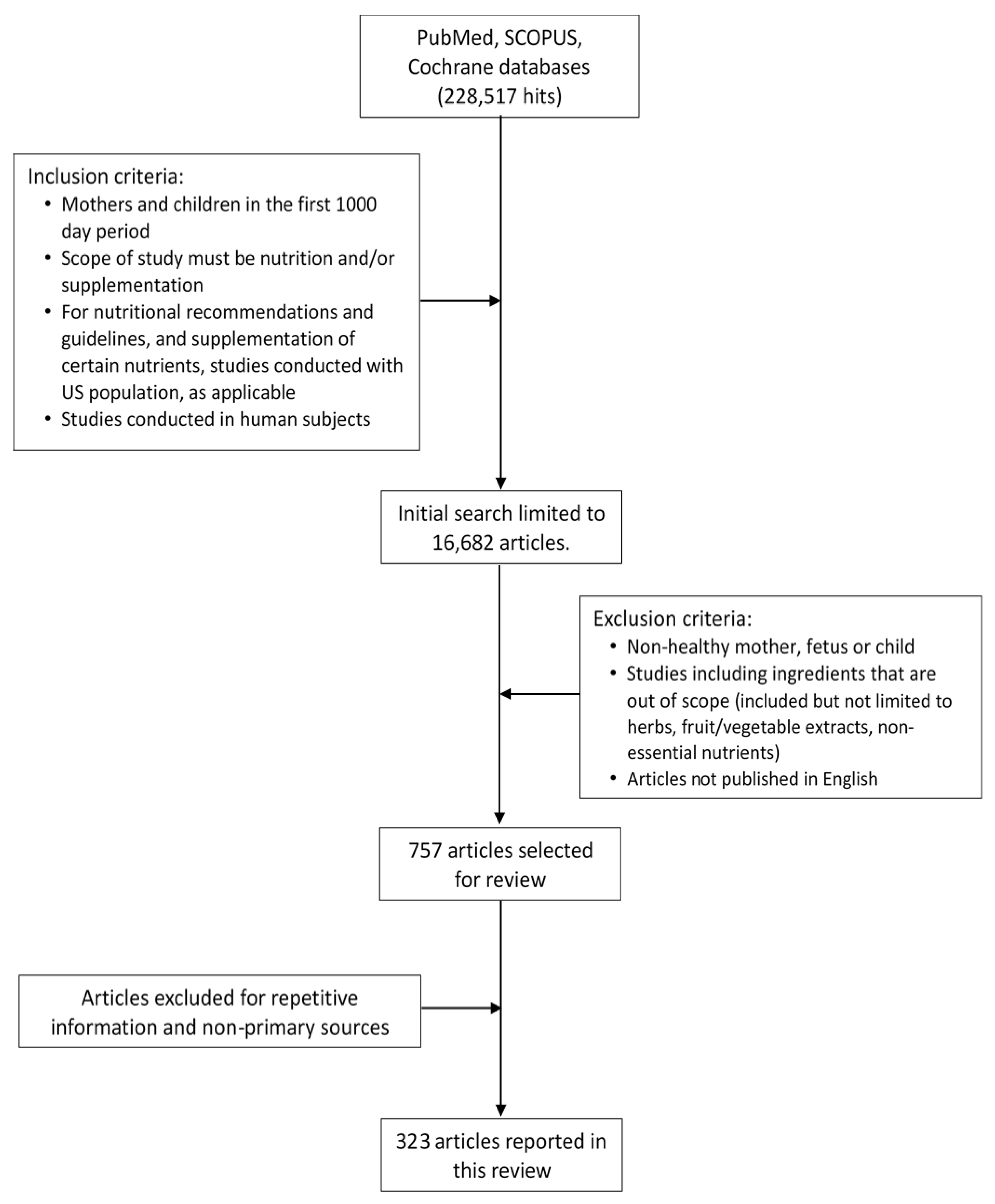
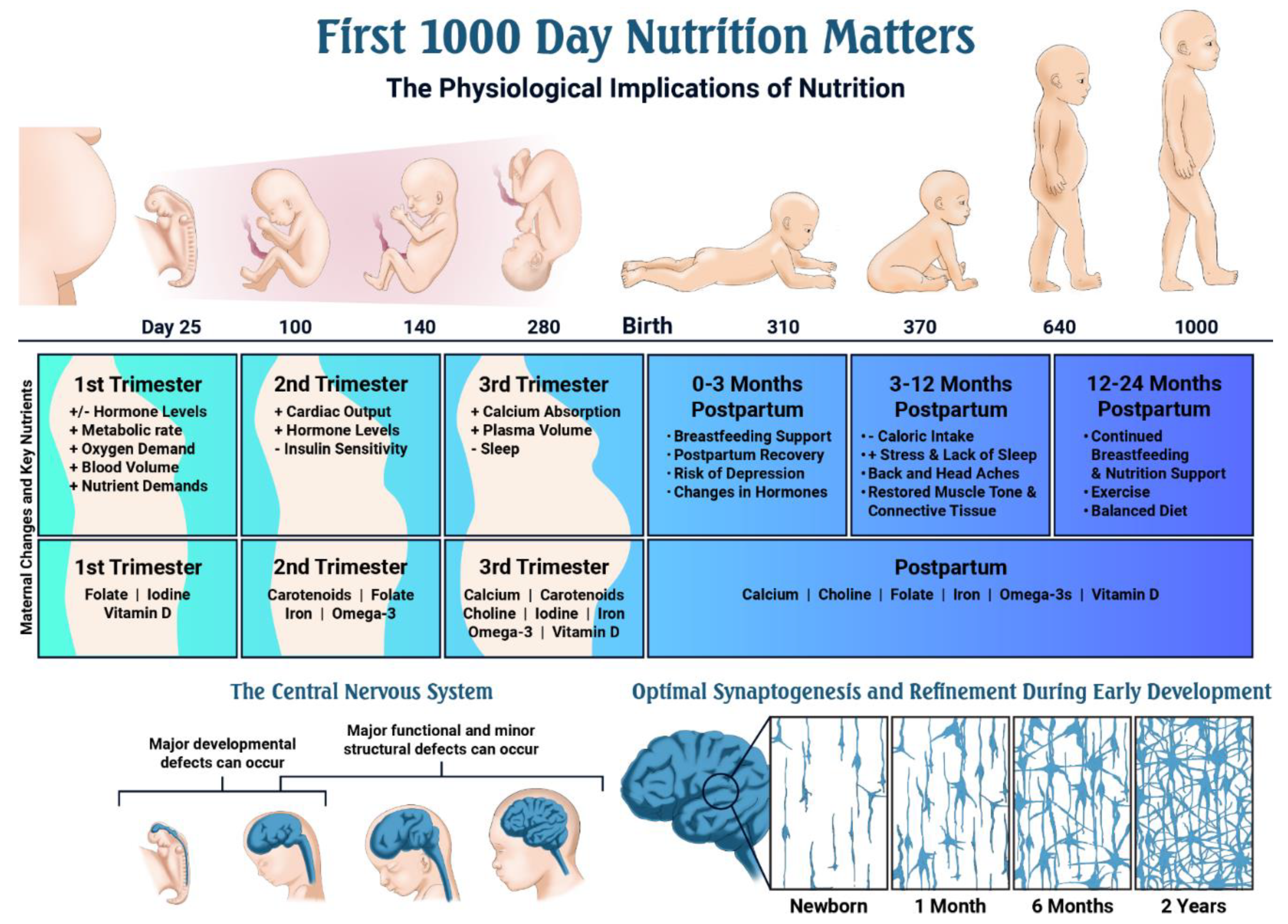
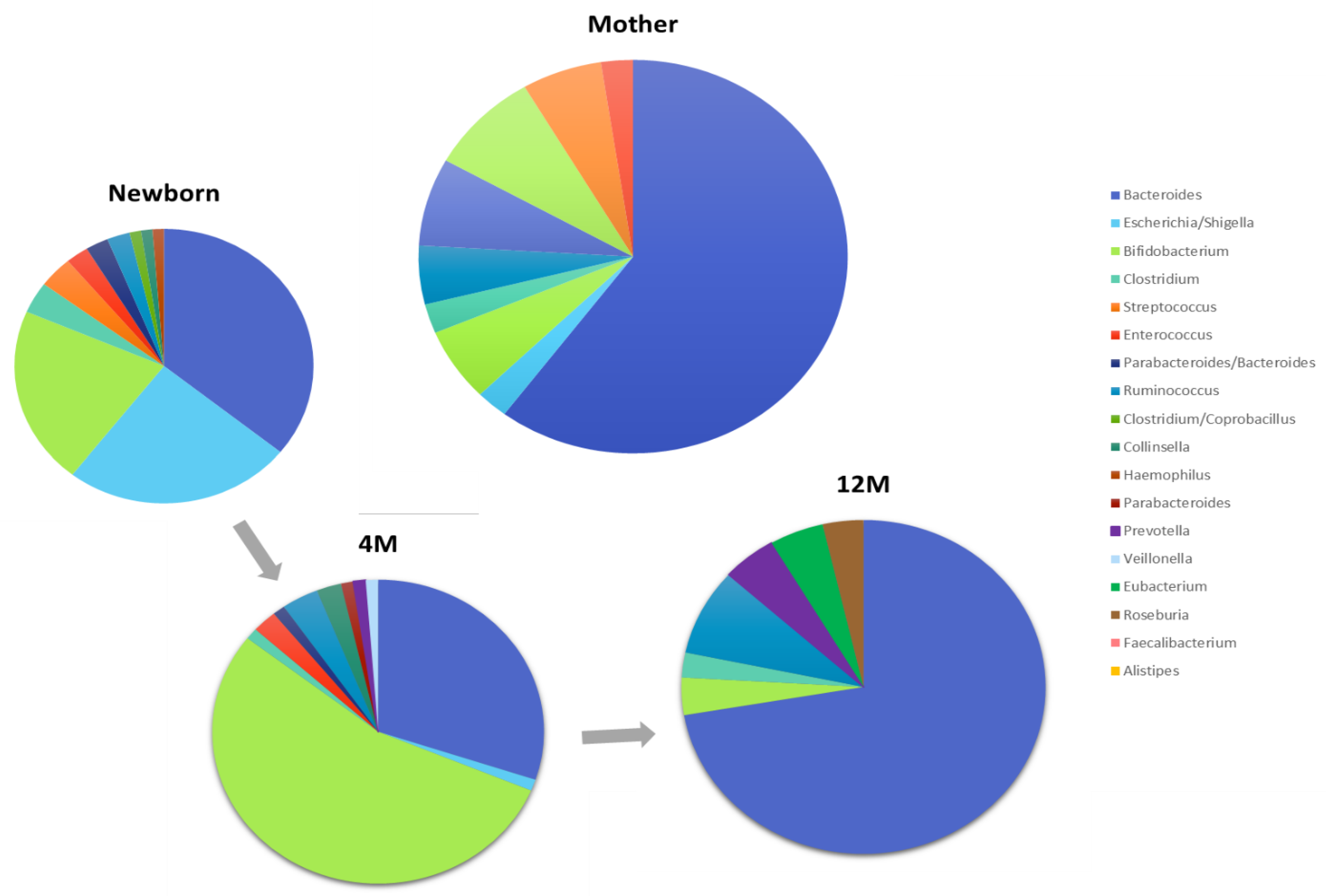
| Organ System | Description of Changes with Pregnancy |
|---|---|
| Cardiovascular | Cardiac output increases 20% by 8 weeks gestation and maternal heart rate increases by 10–20 bpm Blood pressure decreases in the first and second trimesters, but increases during the third. Cardiac output increases during labor (15% in stage 1 labor, 50% in stage 2 labor, with uterine contractions leading to an auto-transfusion of blood (300–500 mL) back into circulation) |
| Urinary | Renal vasodilation occurs and renal plasma flow and glomerular filtration rate (GFR) increases compared to non-pregnant levels, by 40%–65% and 50%–85%, respectively As GFR increases, serum creatinine and urea concentrations decrease to 44.2 μmol/L and 3.2 mmol/L Increases in renal blood flow cause an overall increase in renal size by 1–1.5 cm, reaching the maximal size by mid-pregnancy Kidney, pelvis, and calyceal systems dilate due to mechanical compressive forces on the ureters Alterations in tubular handling of wastes and nutrients occur Less effective reabsorption of glucose and variability in glucose excretion |
| Endocrine | Levels of human chorionic gonadotropin (hCG), estrogen, and progesterone peak during pregnancy and may cause increased nausea and vomiting Electrolyte imbalances, dehydration, weight loss, or vitamin and mineral deficiencies may occur in severe cases of nausea and excessive vomiting (hyperemesis gravidum) |
| Gastrointestinal | Mechanical changes in the alimentary tract occur due to the growing uterus; the stomach is displaced upwards leading to increased intra-gastric pressure Other gastrointestinal problems may also occur during pregnancy, including heartburn, constipation and incontinence |
| Hematological | Plasma volume increases by 50% by week 34 of gestation and is proportional to the birthweight of the baby Hemoglobin, hematocrit, and red blood cell count decrease Requirements for iron, folate, and vitamin B12 increase to accommodate hemoglobin synthesis by the mother and the growing fetus Production of clotting factors (VIII, IX and X) increases to prepare for hemostasis following delivery |
| Respiratory | Demand for oxygen increases due to a 15% increase in metabolic rate and a 20% increased consumption of oxygen Feelings of breathlessness without hypoxia are common in the third trimester, but may occur anytime during pregnancy |
| Skeletal | Maternal bone turnover is low in the first trimester, but increases during the third trimester when fetal calcium needs are highest |
| Nutrient | DRI (Pregnancy) DRI (Lactation) | DRI (Non-Pregnancy) | Examples of Common Dietary Sources (Listed Alphabetically) |
|---|---|---|---|
| Carbohydrate | 175 g/day 210 g/day | 130 g/day | Fruits, legumes, low-fat dairy products, vegetables (starch and non-starchy), whole grains |
| Total Fiber | 28 g/day * 29 g/day * | 25 g/day * | Fruits, legumes, vegetables, whole grains |
| Protein | 71 g/day 71 g/day | 46 g/day | Animal sources: Beef, chicken, dairy products, eggs, pork, seafood, turkey Plant sources: Legumes, nuts, quinoa, seeds, soy |
| Total Fat | No DRI, 20%–35% of total calories suggested | Limit saturated and trans-fat, increase consumption of polyunsaturated fatty acids | |
| Linoleic Acid (Omega-6) | 13 g/day * 13 g/day * | 12 g/day * | Nuts, seeds, vegetable oils (including soybean, safflower and corn oil) |
| alpha-Linolenic Acid (Omega-3) | 1.4 g/day * 1.3 g/day * | 1.1 g/day * | Fatty fish, oils (including flax seed). Smaller amounts found in poultry, meats and eggs |
| Vitamin A | 770 μg RAE/day 1300 μg RAE/day | 700 μg RAE/day | Apricots, broccoli, carrots, fortified milk and eggs, kale, mangoes, margarine, sweet potatoes |
| Vitamin C | 85 mg/day 120 mg/day | 75 mg/day | Citrus fruits, kiwifruit, strawberries, vegetables (red pepper, green pepper, broccoli, Brussels sprouts, cabbage) |
| Vitamin D | 600 IU/day 600 IU/day | 600 IU/day | Eggs, fatty fish, fortified foods such as orange juice and milk |
| Vitamin E | 15 mg/day 19 mg/day | 15 mg/day | Nuts, plant-based oils, seeds |
| Vitamin K | 90 μg/day * 90 μg/day * | 90 μg/day * | Broccoli, green beans, kale, peas, spinach, vegetable oils (canola, soy) |
| Vitamin B6 | 1.9 mg/day 2.0 mg/day | 1.3 mg/day | Fish, meat, poultry and whole grains including oats |
| Vitamin B12 | 2.6 μg/day 2.8 μg/day | 2.4 μg/day | Dairy products, eggs, meat, poultry, seafood |
| Choline | 450 mg/day * 550 mg/day * | 425 mg/day * | Beef and chicken, eggs (with yolk), mushrooms, salmon, wheat germ |
| Folate | 600 μg/day 500 μg/day | 400 μg/day | Beans, dark green vegetables (including spinach and asparagus), fortified cereals, fortified juices (including orange juice), nuts |
| Calcium | 1000 mg/day 1000 mg/day | 1000 mg/day | Cheese, green leafy vegetables (including broccoli, kale, and cabbage), milk, yogurt |
| Iodine | 220 μg/day 290 μg/day | 150 μg/day | Dairy products, fish, iodized salt, seaweed |
| Iron | 27 mg/day 9 mg/day | 18 mg/day (19–50 years.) 8 mg/day (51 years+) | Heme sources: Fish, meat, poultry, seafood Non-heme sources: Fortified cereals, nuts, seeds, spinach |
| Zinc | 11 mg/day 12 mg/day | 8 mg/day | Nuts, legumes, meat, seeds, shellfish |
| Lutein | --† --† | --† | Cooked leafy greens (including spinach and kale), egg yolks |
| Zeaxanthin | --† --† | --† | Corn, yellow and orange peppers |
| Nutrient | Infants 0–6 Months | Infants 7–12 Months | Children 1–3 Years |
|---|---|---|---|
| Carbohydrate | 60 g/day * | 95 g/day * | 130 g/day |
| Total Fiber | ND | ND | 19 g/day * |
| Protein | 9.1 g/day * | 11.0 g/day | 13 g/day |
| Total Fat | 31 g/day * | 30 g/day * | ND |
| Linoleic Acid (Omega-6) | 4.4 g/day * | 4.6 g/day * | 7.0 g/day * |
| Alpha-Linolenic Acid (Omega-3) | 0.5 g/day * | 0.5 g/day * | 0.7 g/day * |
| Vitamin A | 400 μg RAE/day * | 500 μg RAE/day * | 300 μg RAE/day |
| Vitamin C | 40 mg/day * | 50 mg/day * | 15 mg/day |
| Vitamin D | 400 IU/day * | 400 IU/day * | 600 IU/day |
| Vitamin E | 4 mg/day * | 5 mg/day * | 6 mg/day |
| Vitamin K | 2.0 μg/day * | 2.5 μg/day * | 30 μg/day * |
| Vitamin B6 | 0.1 mg/day * | 0.3 mg/day * | 0.5 mg/day |
| Vitamin B12 | 0.4 μg/day * | 0.5 μg/day * | 0.9 μg/day |
| Choline | 125 mg/day * | 150 mg/day * | 200 mg/day * |
| Folate | 65 μg/day * | 80 μg/day * | 150 μg/day |
| Calcium | 200 mg/day * | 260 mg/day * | 700 mg/day |
| Iodine | 110 μg/day * | 130 μg/day * | 90 μg/day |
| Iron | 0.27 mg/day * | 11 mg/day | 7 mg/day |
| Zinc | 2 mg/day * | 3 mg/day | 3 mg/day |
| Lutein | --† | --† | --† |
| Zeaxanthin | --† | --† | --† |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beluska-Turkan, K.; Korczak, R.; Hartell, B.; Moskal, K.; Maukonen, J.; Alexander, D.E.; Salem, N.; Harkness, L.; Ayad, W.; Szaro, J.; et al. Nutritional Gaps and Supplementation in the First 1000 Days. Nutrients 2019, 11, 2891. https://doi.org/10.3390/nu11122891
Beluska-Turkan K, Korczak R, Hartell B, Moskal K, Maukonen J, Alexander DE, Salem N, Harkness L, Ayad W, Szaro J, et al. Nutritional Gaps and Supplementation in the First 1000 Days. Nutrients. 2019; 11(12):2891. https://doi.org/10.3390/nu11122891
Chicago/Turabian StyleBeluska-Turkan, Katrina, Renee Korczak, Beth Hartell, Kristin Moskal, Johanna Maukonen, Diane E. Alexander, Norman Salem, Laura Harkness, Wafaa Ayad, Jacalyn Szaro, and et al. 2019. "Nutritional Gaps and Supplementation in the First 1000 Days" Nutrients 11, no. 12: 2891. https://doi.org/10.3390/nu11122891
APA StyleBeluska-Turkan, K., Korczak, R., Hartell, B., Moskal, K., Maukonen, J., Alexander, D. E., Salem, N., Harkness, L., Ayad, W., Szaro, J., Zhang, K., & Siriwardhana, N. (2019). Nutritional Gaps and Supplementation in the First 1000 Days. Nutrients, 11(12), 2891. https://doi.org/10.3390/nu11122891




