Deriving the Dietary Approaches to Stop Hypertension (DASH) Score in Women from Seven Pregnancy Cohorts from the European ALPHABET Consortium
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Populations
2.2. Dietary Data Collection and Treatment
2.3. DASH Score Creation
2.3.1. Food Group Classification and Item Selections
2.3.2. Scoring Method
2.4. Statistical Analyses
3. Results
3.1. DASH Creation Choices—Food Components and Items Retained
3.2. DASH Scores and Intakes of DASH Food Groups
4. Discussion
4.1. Harmonisation Process Choices
4.2. Daily Frequency Consumption
4.3. Dietary Data Utilisation
5. Conclusions
Supplementary Materials
Ethical Approvals
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Godfrey, K.M.; Costello, P.M.; Lillycrop, K.A. The developmental environment, epigenetic biomarkers and long-term health. J. Dev. Orig. Health Dis. 2015, 6, 399–406. [Google Scholar] [CrossRef]
- Haugen, A.C.; Schug, T.T.; Collman, G.; Heindel, J.J. Evolution of DOHaD: The impact of environmental health sciences. J. Dev. Orig. Health Dis. 2015, 6, 55–64. [Google Scholar] [CrossRef]
- Procter, S.B.; Campbell, C.G. Position of the Academy of Nutrition and Dietetics: Nutrition and lifestyle for a healthy pregnancy outcome. J. Acad. Nutr. Diet. 2014, 114, 1099–1103. [Google Scholar] [CrossRef] [PubMed]
- Devereux, G.; Turner, S.W.; Craig, L.C.; McNeill, G.; Martindale, S.; Harbour, P.J.; Helms, P.J.; Seaton, A. Low maternal vitamin E intake during pregnancy is associated with asthma in 5-year-old children. Am. J. Respir. Crit. Care Med. 2006, 174, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Knudsen, V.K.; Orozova-Bekkevold, I.M.; Mikkelsen, T.B.; Wolff, S.; Olsen, S.F. Major dietary patterns in pregnancy and fetal growth. Eur. J. Clin. Nutr. 2008, 62, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Abu-Saad, K.; Fraser, D. Maternal nutrition and birth outcomes. Epidemiol. Rev. 2010, 32, 5–25. [Google Scholar] [CrossRef]
- Gernand, A.D.; Schulze, K.J.; Stewart, C.P.; West, K.P.; Christian, P. Micronutrient deficiencies in pregnancy worldwide: Health effects and prevention. Nat. Rev. Endocrinol. 2016, 12, 274–289. [Google Scholar] [CrossRef]
- Shapiro, A.L.; Kaar, J.L.; Crume, T.L.; Starling, A.P.; Siega-Riz, A.M.; Ringham, B.M.; Glueck, D.H.; Norris, J.M.; Barbour, L.A.; Friedman, J.E.; et al. Maternal diet quality in pregnancy and neonatal adiposity: The Healthy Start Study. Int. J. Obes. 2016, 40, 1056–1062. [Google Scholar] [CrossRef]
- Borge, T.C.; Aase, H.; Brantsæter, A.L.; Biele, G. The importance of maternal diet quality during pregnancy on cognitive and behavioural outcomes in children: A systematic review and meta-analysis. BMJ Open. 2017, 7, e016777. [Google Scholar] [CrossRef]
- Fung, T.T.; Chiuve, S.E.; McCullough, M.L.; Rexrode, K.M.; Logroscino, G.; Hu, F.B. Adherence to a DASH-style diet and risk of coronary heart disease and stroke in women. Arch. Intern. Med. 2008, 168, 713–720. [Google Scholar] [CrossRef]
- Hinderliter, A.L.; Babyak, M.A.; Sherwood, A.; Blumenthal, J.A. The DASH diet and insulin sensitivity. Curr. Hypertens. Rep. 2011, 13, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Harrington, J.M.; Fitzgerald, A.P.; Kearney, P.M.; McCarthy, V.J.; Madden, J.; Browne, G.; Dolan, E.; Perry, I.J. DASH diet score and distribution of blood pressure in middle-aged men and women. Am. J. Hypertens. 2013, 26, 1311–1320. [Google Scholar] [CrossRef] [PubMed]
- Jones-McLean, E.; Hu, J.; Greene-Finestone, L.S.; de Groh, M. A DASH dietary pattern and the risk of colorectal cancer in Canadian adults. Health Promot. Chronic Dis. Prev. Can. 2015, 35, 12–20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berz, J.P.B.; Singer, M.R.; Guo, X.; Daniels, S.R.; Moore, L.L. Use of a DASH Food Group Score to Predict Excess Weight Gain in Adolescent Girls in the National Growth and Health Study. Arch Pediatr. Adolesc. Med. 2011, 165, 540–546. [Google Scholar] [CrossRef] [Green Version]
- Siervo, M.; Lara, J.; Chowdhury, S.; Ashor, A.; Oggioni, C.; Mathers, J.C. Effects of the Dietary Approach to Stop Hypertension (DASH) diet on cardiovascular risk factors: A systematic review and meta-analysis. Br. J. Nutr. 2015, 113, 1–15. [Google Scholar] [CrossRef]
- Asemi, Z.; Samimi, M.; Tabassi, Z.; Sabihi, S.; Esmaillzadeh, A. A randomized controlled clinical trial investigating the effect of DASH diet on insulin resistance, inflammation, and oxidative stress in gestational diabetes. Nutrition 2013, 29, 619–624. [Google Scholar] [CrossRef]
- Jiang, F.; Li, Y.; Xu, P.; Li, J.; Chen, X.; Yu, H.; Gao, B.; Xu, B.; Li, X.; Chen, W. The efficacy of the Dietary Approaches to Stop Hypertension diet with respect to improving pregnancy outcomes in women with hypertensive disorders. J. Hum. Nutr. Diet. 2019. [Google Scholar] [CrossRef]
- Fulay, A.P.; Rifas-Shiman, S.L.; Oken, E.; Perng, W. Associations of the dietary approaches to stop hypertension (DASH) diet with pregnancy complications in Project Viva. Eur. J. Clin. Nutr. 2018, 72, 1385. [Google Scholar] [CrossRef]
- Phillips, C.M.; Chen, L.W.; Heude, B.; Bernard, J.Y.; Harvey, N.C.; Duijts, L.; Mensink-Bout, S.M.; Polanska, K.; Mancano, G.; Suderman, M.; et al. Dietary Inflammatory Index and Non-Communicable Disease Risk: A Narrative Review. Nutrients 2019, 11, 1873. [Google Scholar] [CrossRef]
- Mendez, M.A.; Kogevinas, M. A comparative analysis of dietary intakes during pregnancy in Europe: A planned pooled analysis of birth cohort studies. Am. J. Clin. Nutr. 2011, 94 (Suppl. 6), 1993S–1999S. [Google Scholar] [CrossRef]
- Olsen, S.F.; Eva Birgisdottir, B.; Halldorsson, T.I.; Brantsæter, A.L.; Haugen, M.; Torjusen, H.; Petersen, S.B.; Strøm, M.; Meltzer, H.M. Possibilities and considerations when merging dietary data from the world’s two largest pregnancy cohorts: The Danish National Birth Cohort and the Norwegian Mother and Child Cohort Study. Acta Obstet. Gynecol. Scand. 2014, 93, 1131–1140. [Google Scholar] [CrossRef] [PubMed]
- Fraser, A.; Macdonald-Wallis, C.; Tilling, K.; Boyd, A.; Golding, J.; Davey Smith, G.; Henderson, J.; Macleod, J.; Molloy, L.; Ness, A.; et al. Cohort Profile: The Avon Longitudinal Study of Parents and Children: ALSPAC mothers cohort. Int. J. Epidemiol. 2013, 42, 97–110. [Google Scholar] [CrossRef] [PubMed]
- Boyd, A.; Golding, J.; Macleod, J.; Lawlor, D.A.; Fraser, A.; Henderson, J.; Molloy, L.; Ness, A.; Ring, S.; Davey Smith, G. Cohort Profile: The ‘Children of the 90s’—The index offspring of the Avon Longitudinal Study of Parents and Children. Int. J. Epidemiol. 2013, 42, 111–127. [Google Scholar] [CrossRef] [PubMed]
- Heude, B.; Forhan, A.; Slama, R.; Douhaud, L.; Bedel, S.; Saurel-Cubizolles, M.J.; Hankard, R.; Thiebaugeorges, O.; De Agostini, M.; Annesi-Maesano, I.; et al. Cohort Profile: The EDEN mother-child cohort on the prenatal and early postnatal determinants of child health and development. Int. J. Epidemiol. 2016, 45, 353–363. [Google Scholar] [CrossRef] [PubMed]
- Kooijman, M.N.; Kruithof, C.J.; van Duijn, C.M.; Duijts, L.; Franco, O.H.; van IJzendoorn, M.H.; de Jongste, J.C.; Klaver, C.C.; van der Lugt, A.; Mackenbach, J.P.; et al. The Generation R Study: Design and cohort update 2017. Eur. J. Epidemiol. 2016, 31, 1243–1264. [Google Scholar] [CrossRef] [PubMed]
- O’Mahony, D.; Fallon, U.B.; Hannon, F.; Kloeckner, K.; Avalos, G.; Murphy, A.W.; Kelleher, C.C. The Lifeways Cross-Generation Study: Design, recruitment and data management considerations. Ir. Med. J. 2007, 100, 3–6. [Google Scholar]
- Kennelly, M.A.; Ainscough, K.; Lindsay, K.; Gibney, E.; Mc Carthy, M.; McAuliffe, F.M. Pregnancy, exercise and nutrition research study with smart phone app support (Pears): Study protocol of a randomized controlled trial. Contemp. Clin. Trials 2016, 46, 92–99. [Google Scholar] [CrossRef]
- Polanska, K.; Hanke, W.; Gromadzinska, J.; Ligocka, D.; Gulczynska, E.; Sobala, W.; Wasowicz, W. Polish mother and child cohort study—Defining the problem, the aim of the study and methodological assumption. Int. J. Occup. Med. Environ. Health 2009, 22, 383–391. [Google Scholar] [CrossRef]
- Walsh, J.M.; McGowan, C.A.; Mahony, R.; Foley, M.E.; McAuliffe, F.M. Low glycaemic index diet in pregnancy to prevent macrosomia (ROLO study): Randomised control trial. BMJ 2012, 345, e5605. [Google Scholar] [CrossRef]
- Inskip, H.M.; Godfrey, K.M.; Robinson, S.M.; Law, C.M.; Barker, D.J.; Cooper, C. Cohort Profile: The Southampton Women’s Survey. Int. J. Epidemiol. 2006, 35, 42–48. [Google Scholar] [CrossRef]
- Deschamps, V.; de Lauzon-Guillain, B.; Lafay, L.; Borys, J.-M.; Charles, M.A.; Romon, M. Reproducibility and relative validity of a food-frequency questionnaire among French adults and adolescents. Eur. J. Clin. Nutr. 2009, 63, 282–291. [Google Scholar] [CrossRef] [PubMed]
- Klipstein-Grobusch, K.D.; Den Breeijen, J.H.; Goldbohm, R.A.; Geleijnse, J.M.; Hofman, A.; Grobbee, D.E.; Witteman, J.C.M. Dietary assessment in the elderly: Validation of a semiquantitative food frequency questionnaire. Eur. J. Clin. Nutr. 1998, 52, 588–596. [Google Scholar] [CrossRef] [PubMed]
- Murrin, C.; Shrivastava, A.; Kelleher, C.C. Maternal macronutrient intake during pregnancy and 5 years postpartum and associations with child weight status aged five. Eur. J. Clin. Nutr. 2013, 67, 670–679. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harrington, J. Validation of a Food Frequency Questionnaire as a Tool for Assessing Nutrient Intake. Masters’ Thesis, National University of Ireland Galway, Galway, Ireland, 1997. [Google Scholar]
- Horan, M.K.; McGowan, C.A.; Doyle, O.; McAuliffe, F.M. Well-being in pregnancy: An examination of the effect of socioeconomic, dietary and lifestyle factors including impact of a low glycaemic index dietary intervention. Eur. J. Clin. Nutr. 2014, 68, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Rogers, I.; Emmett, P. Diet during pregnancy in a population of pregnant women in South West England. ALSPAC Study Team. Avon Longitudinal Study of Pregnancy and Childhood. Eur. J. Clin. Nutr. 1998, 52, 246–250. [Google Scholar] [CrossRef]
- Robinson, S.; Godfrey, K.; Osmond, C.; Cox, V.; Barker, D. Evaluation of a food frequency questionnaire used to assess nutrient intakes in pregnant women. Eur. J. Clin. Nutr. 1996, 50, 302–308. [Google Scholar]
- Miller, P.E.; Cross, A.J.; Subar, A.F.; Krebs-Smith, S.M.; Park, Y.; Powell-Wiley, T.; Hollenbeck, A.; Reedy, J. Comparison of 4 established DASH diet indexes: Examining associations of index scores and colorectal cancer. Am. J. Clin. Nutr. 2013, 98, 794–803. [Google Scholar] [CrossRef]
- Kwan, M.W.M.; Wong, M.C.S.; Wang, H.H.X.; Liu, K.Q.L.; Lee, C.L.S.; Yan, B.P.Y.; Yu, C.M.; Griffiths, S.M. Compliance with the Dietary Approaches to Stop Hypertension (DASH) Diet: A Systematic Review. PLoS ONE 2013, 8, e78412. [Google Scholar] [CrossRef]
- Dixon, L.B.; Subar, A.F.; Peters, U.; Weissfeld, J.L.; Bresalier, R.S.; Risch, A.; Schatzkin, A.; Hayes, R.B. Adherence to the USDA Food Guide, DASH Eating Plan, and Mediterranean dietary pattern reduces risk of colorectal adenoma. J. Nutr. 2007, 137, 2443–2450. [Google Scholar] [CrossRef]
- U.S. Department of Health and Human Services. Your Guide to Lowering Your Blood Pressure with DASH. Report No.: NIH Publication No. 06-4082. 1998 Revised 2006; p. 64. Available online: https://www.nhlbi.nih.gov/files/docs/public/heart/new_dash.pdf (accessed on 15 June 2018).
- Eurocode 2. Eurocode 2 Food Coding System. Available online: http://www.ianunwin.demon.co.uk/eurocode/index.htm (accessed on 19 February 2019).
- Programme National Nutrition Santé. La santé vient en mangeant. Le guide alimentaire pour tous. 2002, p. 130. Available online: http://www.mangerbouger.fr/PNNS/ (accessed on 7 January 2018).
- Programme National Nutrition Santé. La santé vient en mangeant et en bougeant. Le guide nutrition de la grossesse. 2007 Revised 2015. p. 40. Available online: http://www.mangerbouger.fr/PNNS/ (accessed on 7 January 2018).
- Sofi, F.; Macchi, C.; Abbate, R.; Gensini, G.F.; Casini, A. Mediterranean diet and health status: An updated meta-analysis and a proposal for a literature-based adherence score. Public Health Nutr. 2014, 17, 2769–2782. [Google Scholar] [CrossRef]
- Drouin-Chartier, J.P.; Brassard, D.; Tessier-Grenier, M.; Côté, J.A.; Labonté, M.È.; Desroches, S.; Couture, P.; Lamarche, B. Systematic Review of the Association between Dairy Product Consumption and Risk of Cardiovascular-Related Clinical Outcomes. Adv Nutr. 2016, 7, 1026–1040. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beer-Borst, S.; Hercberg, S.; Morabia, A.; Bernstein, M.S.; Galan, P.; Galasso, R.; Giampaoli, S.; McCrum, E.; Panico, S.; Preziosi, P.; et al. Dietary patterns in six european populations: Results from EURALIM, a collaborative European data harmonization and information campaign. Eur. J. Clin. Nutr. 2000, 54, 253–262. [Google Scholar] [CrossRef] [PubMed]
- Irala-Estévez, J.D.; Groth, M.; Johansson, L.; Oltersdorf, U.; Prättälä, R.; Martínez-González, M.A. A systematic review of socio-economic differences in food habits in Europe: Consumption of fruit and vegetables. Eur. J. Clin. Nutr. 2000, 54, 706–714. [Google Scholar] [CrossRef] [PubMed]
- Verger, P.; Ireland, J.; Møller, A.; Abravicius, J.A.; De Henauw, S.; Naska, A. Improvement of comparability of dietary intake assessment using currently available individual food consumption surveys. Eur. J. Clin. Nutr. 2002, 56 (Suppl. 2), S18–S24. [Google Scholar] [CrossRef]
- NutriGen Alliance Investigators; De Souza, R.J.; Zulyniak, M.A.; Desai, D.; Shaikh, M.R.; Campbell, N.C.; Lefebvre, D.L.; Gupta, M.; Wilson, J.; Wahi, G.; et al. Harmonization of Food-Frequency Questionnaires and Dietary Pattern Analysis in 4 Ethnically Diverse Birth Cohorts. J. Nutr. 2016, 146, 2343–2350. [Google Scholar] [Green Version]
- Mellen, P.B.; Gao, S.K.; Vitolins, M.Z.; Goff, D.C. Deteriorating dietary habits among adults with hypertension: DASH dietary accordance, NHANES 1988–1994 and 1999–2004. Arch. Intern. Med. 2008, 168, 308–314. [Google Scholar] [CrossRef]
- Harrington, J.; Fitzgerald, A.P.; Layte, R.; Lutomski, J.; Molcho, M.; Perry, I.J. Sociodemographic, health and lifestyle predictors of poor diets. Public Health Nutr. 2011, 14, 2166–2175. [Google Scholar] [CrossRef] [Green Version]
- Epstein, D.E.; Sherwood, A.; Smith, P.J.; Craighead, L.; Caccia, C.; Lin, P.H.; Babyak, M.A.; Johnson, J.J.; Hinderliter, A.; Blumenthal, J.A. Determinants and Consequences of Adherence to the DASH Diet in African American and White Adults with High Blood Pressure: Results from the ENCORE Trial. J. Acad. Nutr. Diet. 2012, 112, 1763–1773. [Google Scholar] [CrossRef]
- Folsom, A.R.; Parker, E.D.; Harnack, L.J. Degree of concordance with DASH diet guidelines and incidence of hypertension and fatal cardiovascular disease. Am. J. Hypertens. 2007, 20, 225–232. [Google Scholar] [CrossRef]
- Appel, L.J.; Moore, T.J.; Obarzanek, E.; Vollmer, W.M.; Svetkey, L.P.; Sacks, F.M.; Bray, G.A.; Vogt, T.M.; Cutler, J.A.; Windhauser, M.M.; et al. A clinical trial of the effects of dietary patterns on blood pressure. DASH Collaborative Research Group. N. Engl. J. Med. 1997, 336, 1117–1124. [Google Scholar] [CrossRef]
- Sacks, F.M.; Svetkey, L.P.; Vollmer, W.M.; Appel, L.J.; Bray, G.A.; Harsha, D.; Obarzanek, E.; Conlin, P.R.; Miller, E.R.; Simons-Morton, D.G.; et al. Effects on blood pressure of reduced dietary sodium and the Dietary Approaches to Stop Hypertension (DASH) diet. DASH-Sodium Collaborative Research Group. N. Engl. J. Med. 2001, 344, 3–10. [Google Scholar] [CrossRef]
- Trichopoulou, A.; Naska, A.; Costacou, T.; DAFNE III Group. Disparities in food habits across Europe. Proc. Nutr. Soc. 2002, 61, 553–558. [Google Scholar] [CrossRef] [Green Version]
- Micha, R.; Khatibzadeh, S.; Shi, P.; Andrews, K.G.; Engell, R.E.; Mozaffarian, D. Global, regional and national consumption of major food groups in 1990 and 2010: A systematic analysis including 266 country-specific nutrition surveys worldwide. BMJ Open 2015, 5, e008705. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4593162/ (accessed on 19 February 2019). [CrossRef]
- Dietary Guidelines Advisory Committee. Scientific Report of the 2015 Dietary Guidelines Advisory Committee: Advisory Report of the Secretary of Health and Human Services and the Secretary of Agriculture; U.S. Department of Agriculture, Agricultural Research Service: Washington, DC, USA, 2015.
- Public Health England. The Eatwell Guide. 2018. Available online: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/742750/Eatwell_Guide_booklet_2018v4.pdf (accessed on 22 February 2019).
- Agudo, A.; Slimani, N.; Ocke, M.C.; Naska, A.; Miller, A.B.; Kroke, A.; Bamia, C.; Karalis, D.; Vineis, P.; Palli, D.; et al. Consumption of vegetables, fruit and other plant foods in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohorts from 10 European countries. Public Health Nutr. 2002, 5, 1179–1196. [Google Scholar] [CrossRef] [Green Version]
- Agudo, A.; Slimani, N.; Ocke, M.C.; Naska, A.; Miller, A.B.; Kroke, A.; Bamia, C.; Karalis, D.; Vineis, P.; Palli, D.; et al. Meat, eggs, dairy products, and risk of breast cancer in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort. Am. J. Clin. Nutr. 2009, 90, 602–612. [Google Scholar]
- Blumfield, M.L.; Hure, A.J.; MacDonald-Wicks, L.K.; Patterson, A.J.; Smith, R.; Collins, C.E. Disparities exist between National food group recommendations and the dietary intakes of women. BMC Womens Health 2011, 11, 37. [Google Scholar] [CrossRef]
- Dehghan, M.; Martinez, S.; Zhang, X.; Seron, P.; Lanas, F.; Islam, S.; Merchant, A.T. Relative validity of an FFQ to estimate daily food and nutrient intakes for Chilean adults. Public Health Nutr. 2013, 16, 1782–1788. [Google Scholar] [CrossRef]
- Vioque, J.; Navarrete-Muñoz, E.M.; Gimenez-Monzó, D.; García-de-la-Hera, M.; Granado, F.; Young, I.S.; Ramón, R.; Ballester, F.; Murcia, M.; Rebagliato, M.; et al. Reproducibility and validity of a food frequency questionnaire among pregnant women in a Mediterranean area. Nutr. J. 2013, 12, 26. [Google Scholar] [CrossRef]
- Talegawkar, S.A.; Tanaka, T.; Maras, J.E.; Ferrucci, L.; Tucker, K.L. Validation of Nutrient Intake Estimates Derived Using a Semi-Quantitative FFQ against 3 Day Diet Records in the Baltimore Longitudinal Study of Aging. J. Nutr. Health Aging 2015, 19, 994–1002. [Google Scholar] [CrossRef]
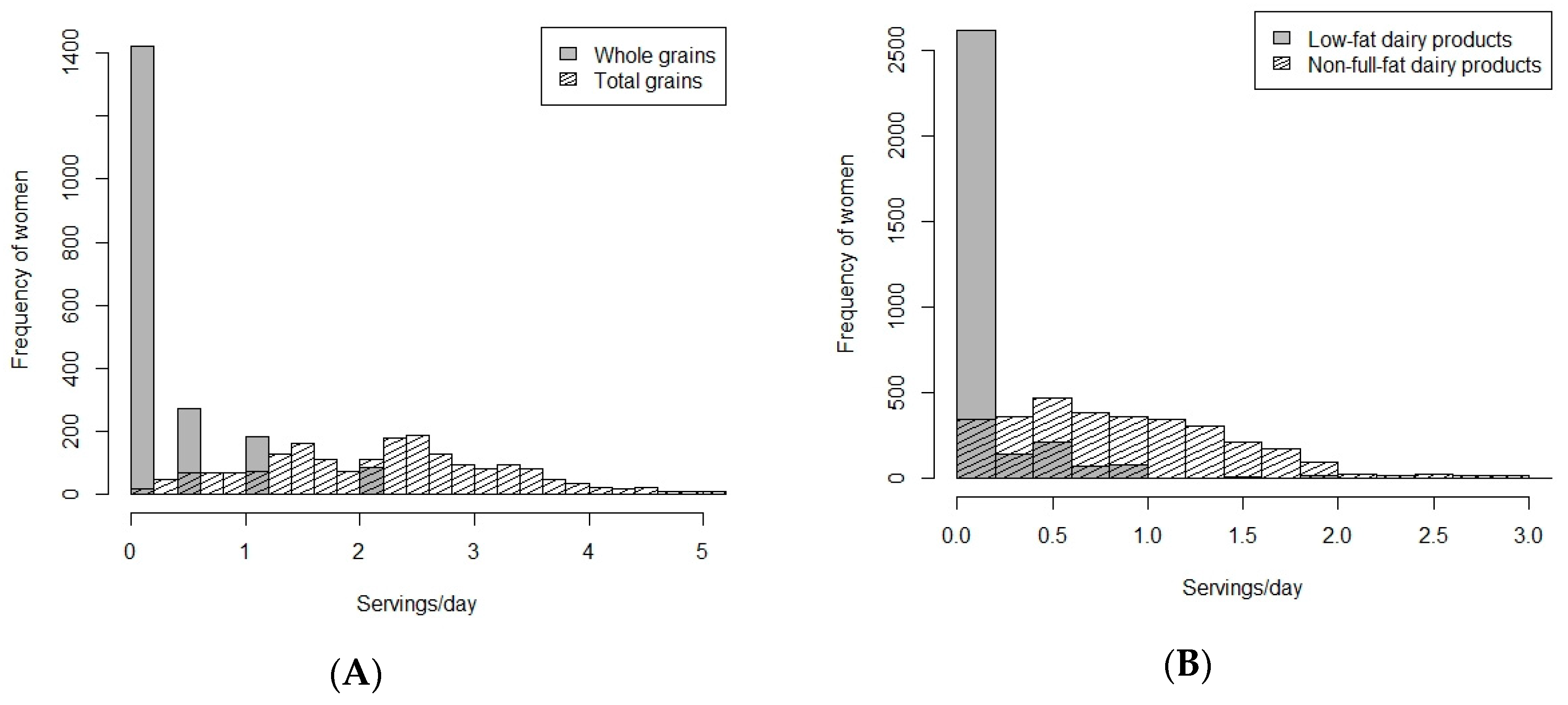
| Cohort | ALSPAC | EDEN | Generation R | Lifeways | REPRO_PL | ROLO | SWS | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Number of Recruited Women | 14,541 | 2002 | 9778 | 1132 | 1451 | 759 | 12,583 | |||
| Women age eligibility | No age limit | 18 and over | No age limit | No age limit | No age limit | 18 and over | 20–34 years | |||
| Study type | Mother-child cohort | Mother-child cohort | Pregnancy-child cohort | Mother-child cohort | Mother-child cohort | Randomised control trial | Pre-pregnancy and pregnancy-child cohort | |||
| Period of inclusion | 1990–1992 | 2003–2006 | 2002–2006 | 2001–2003 | 2007–2011 | 2007–2011 | 1998–2002 | |||
| Location (specific cities) | England (Bristol) | France (Multicentre) | The Netherlands (Rotterdam) | Republic of Ireland (Multicentre) | Poland (Multicentre) | Republic of Ireland (Dublin) | England (Southampton) | |||
| Assessment types | Non-quantitative FFQ | Semi-quantitative FFQ | Semi-quantitative FFQ | Semi-quantitative FFQ | Non-quantitative FFQ | Semi-quantitative FFQ | Non-quantitative FFQ | |||
| Period of FFQ assessment | Around 32 WG | 24–28 WG | Birth | <24 WG | 12–16 WG | 20–24 WG | ≤28 WG | PP | 11 WG | 34 WG |
| FFQ window period | LP | PP | LP | EP | EP | EP | EP | PP | EP | LP |
| Number of women with validated FFQ 1 | 11,965 | 1964 | 1849 | 6402 2 | 1121 | 1314 | 631 | 3156 3 | 2270 | 2649 |
| Mode of FFQ assessment | Self-reported | Self-reported | Self-reported | Self-reported | Self-reported | Self-reported | Nurse administered | |||
| Cohort | ALSPAC | EDEN | Generation R | Lifeways | REPRO_PL | ROLO | SWS | ALPHABET 1 |
|---|---|---|---|---|---|---|---|---|
| FFQ total of food items | 43 | 137 | 293 | 158 | 66 | 158 | 104 | 137.0 |
| FFQ total of food items without alcohol | 43 | 130 | 283 | 154 | 62 | 154 | 99 | 132.1 |
| Total of food items selected for the DASH | 34 | 65 | 136 | 85 | 36 | 85 | 58 | 71.3 |
| % 2 items selected/total food items without alcohol | 79.1% | 50.0% | 48.1% | 55.2% | 58.1% | 55.2% | 58.6% | 57.8% |
| Food components with higher intakes recommended | ||||||||
| Whole grains | 3 | 1 | 13 | 7 | 2 | 7 | 4 | 5.3 |
| Total grains 3 | 7 | 7 | 20 | 14 | 5 | 14 | 8 | 10.7 |
| Vegetables | 5 | 16 | 33 | 24 | 12 | 24 | 16 | 18.6 |
| Fruits | 3 | 12 | 20 | 13 | 10 | 13 | 12 | 11.9 |
| Low-fat dairy products | 2 | 4 | 10 4 | 6 | 0 | 6 | 2 | 4.3 |
| Non-full-fat dairy products 3 | 3 | 6 | 18 4 | 7 | 2 | 7 | 5 | 6.9 |
| Lean meats, poultry, fish | 4 | 9 | 10 | 9 | 13 | 9 | 5 | 8.4 |
| Nuts, seeds, legumes | 7 | 4 | 14 | 5 | 2 | 5 | 2 | 5.6 |
| Food components with lower intakes recommended | ||||||||
| Fats and oils | 5 | 10 | 13 | 15 | 2 | 15 | 11 | 10.1 |
| Sweets and added sugars | 3 | 5 | 9 | 4 | 1 | 4 | 3 | 4.1 |
| Sugar-sweetened beverages | 2 | 3 | 2 | 1 | 0 | 1 | 2 | 1.6 |
| Red and Processed meat | 4 | 12 | 20 | 17 | 4 | 17 | 10 | 12.0 |
| Sodium | Available in grams/day | Available in grams/day | Available in grams/day | Available in grams/day | Available in grams/day | Available in grams/day | Available in grams/day | Available in grams/day |
| Fung’s DASH Components | Original DASH Components | Food Components Selected in ALPHABET | Rationale |
|---|---|---|---|
| Whole grains | Total grains (Additional note: “whole grains are recommended for most grain servings as a good source of fiber and nutrients”) | Total grains |
|
| Vegetables without potatoes | Vegetables | Vegetables (excluding potatoes and condiments) |
|
| Fruits | Fruits | Fruits | Not applicable |
| Low-fat dairy products | Low-fat milks & milk products | Non-full-fat dairy products |
|
| Nuts, seeds, legumes | Nuts, seeds, legumes | Nuts, seeds, legumes | Not applicable |
| Red and processed meats | Lean meat, poultry, fish | Red and processed meats | Not applicable |
| Fats and oils | |||
| Sugar-sweetened beverages | Sweets and added sugars | Sugar-sweetened beverages, sweets, and added sugars |
|
| Sodium | Additional note for reducing salt | Sodium | Not applicable |
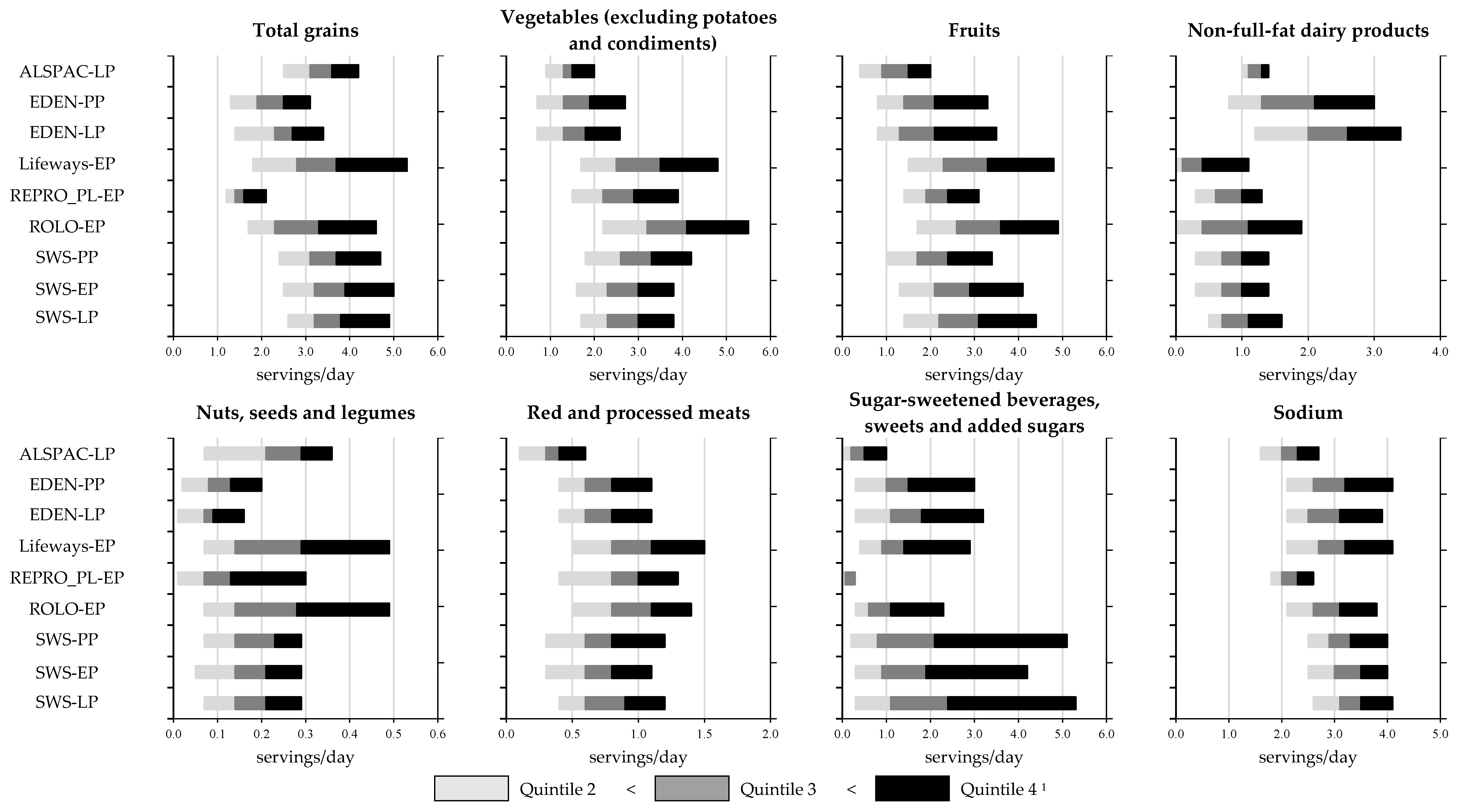
| ALSPAC | EDEN | Generation R | Lifeways | REPRO_PL | ROLO | SWS | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| LP | PP | LP | EP | EP | EP | EP | PP | EP | LP | |||||||||||
| Median[IQR] | Median[IQR] | Median[IQR] | Median[IQR] | Median[IQR] | Median[IQR] | Median[IQR] | Median[IQR] | Median[IQR] | Median[IQR] | |||||||||||
| Total grains | ||||||||||||||||||||
| f/d 1 | 3.4 | [1.4] | 2.3 | [1.5] | 2.5 | [1.7] | - | - | 3.2 | [2.7] | 1.6 | [0.7] | 2.8 | [2.3] | 3.4 | [1.9] | 3.5 | [2.0] | 3.5 | [1.7] |
| g/d 2 | - | - | 142 | [89] | 149 | [91] | 162 | [104] | 162 | [138] | - | - | 183 | [136] | - | - | - | - | - | - |
| Vegetables (excluding potatoes and condiments) | ||||||||||||||||||||
| f/d | 1.5 | [0.9] | 1.6 | [1.6] | 1.5 | [1.4] | - | - | 3.0 | [2.5] | 2.5 | [1.8] | 3.6 | [2.4] | 2.9 | [1.9] | 2.6 | [1.8] | 2.6 | [1.7] |
| g/d | - | - | 176 | [201] | 154 | [167] | 147 | [89] | 269 | [222] | - | - | 332 | [224] | - | - | - | - | - | - |
| Fruits | ||||||||||||||||||||
| f/d | 1.1 | [1.1] | 1.7 | [2.0] | 1.7 | [2.1] | - | - | 2.8 | [2.7] | 2.2 | [1.4] | 3.1 | [2.5] | 2.1 | [2.0] | 2.5 | [2.2] | 2.7 | [2.5] |
| g/d | - | - | 311 | [353] | 293 | [385] | 285 | [264] | 340 | [327] | - | - | 360 | [291] | - | - | - | - | - | - |
| Non-full-fat dairy products | ||||||||||||||||||||
| f/d | 1.3 | [0.3] | 1.6 | [1.5] | 2.2 | [1.8] | - | - | 0.1 | [1.0] | 1.0 | [0.7] | 1.0 | [1.4] | 0.8 | [0.7] | 0.8 | [0.8] | 1.0 | [1.0] |
| g/d | - | - | 303 | [357] | 417 | [403] | 224 | [308] | 18 | [198] | - | - | 250 | [298] | - | - | - | - | - | - |
| Nuts, seeds, legumes | ||||||||||||||||||||
| f/d | 0.3 | [0.3] | 0.1 | [0.1] | 0.1 | [0.1] | - | - | 0.2 | [0.3] | 0.1 | [0.3] | 0.2 | [0.4] | 0.2 | [0.2] | 0.2 | [0.2] | 0.2 | [0.2] |
| g/d | - | - | 13 | [16] | 10 | [17] | 13 | [19] | 19 | [49] | - | - | 19 | [21] | - | - | - | - | - | - |
| Red and processed meats | ||||||||||||||||||||
| f/d | 0.4 | [0.3] | 0.7 | [0.6] | 0.7 | [0.5] | - | - | 0.9 | [0.8] | 1.0 | [0.7] | 0.9 | [0.8] | 0.7 | [0.7] | 0.7 | [0.6] | 0.7 | [0.7] |
| g/d | - | - | 61 | [57] | 60 | [55] | 51 | [44] | 87 | [66] | - | - | 87 | [67] | - | - | - | - | - | - |
| Sugar-sweetened beverages, sweets, and added sugars | ||||||||||||||||||||
| f/d | 0.4 | [0.8] | 1.2 | [1.9] | 1.3 | [2.3] | - | - | 1.1 | [1.9] | 0.3 | [0.2] | 0.9 | [1.4] | 1.2 | [3.8] | 1.2 | [3.1] | 1.7 | [3.8] |
| g/d | - | - | 49 | [153] | 55 | [187] | 67 | [110] | 46 | [101] | - | - | 28 | [66] | - | - | - | - | - | - |
| Sodium | ||||||||||||||||||||
| mg/d | 2161 | [854] | 2871 | [1636] | 2831 | [1461] | 3324 | [1298] | 3001 | [1512] | 2163 | [704] | 2883 | [1381] | 3142 | [1188] | 3246 | [1211] | 3294 | [1157] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aubert, A.M.; Forhan, A.; de Lauzon-Guillain, B.; Chen, L.-W.; Polanska, K.; Hanke, W.; Jankowska, A.; Mensink-Bout, S.M.; Duijts, L.; Suderman, M.; et al. Deriving the Dietary Approaches to Stop Hypertension (DASH) Score in Women from Seven Pregnancy Cohorts from the European ALPHABET Consortium. Nutrients 2019, 11, 2706. https://doi.org/10.3390/nu11112706
Aubert AM, Forhan A, de Lauzon-Guillain B, Chen L-W, Polanska K, Hanke W, Jankowska A, Mensink-Bout SM, Duijts L, Suderman M, et al. Deriving the Dietary Approaches to Stop Hypertension (DASH) Score in Women from Seven Pregnancy Cohorts from the European ALPHABET Consortium. Nutrients. 2019; 11(11):2706. https://doi.org/10.3390/nu11112706
Chicago/Turabian StyleAubert, Adrien M., Anne Forhan, Blandine de Lauzon-Guillain, Ling-Wei Chen, Kinga Polanska, Wojciech Hanke, Agnieszka Jankowska, Sara M. Mensink-Bout, Liesbeth Duijts, Matthew Suderman, and et al. 2019. "Deriving the Dietary Approaches to Stop Hypertension (DASH) Score in Women from Seven Pregnancy Cohorts from the European ALPHABET Consortium" Nutrients 11, no. 11: 2706. https://doi.org/10.3390/nu11112706







