Nutritional and Metabolic Characteristics of UK Adult Phenylketonuria Patients with Varying Dietary Adherence
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Ethics
2.2. Recruitment, Inclusion Criteria, Exclusion Criteria and Study Population
2.2.1. Recruitment
2.2.2. Inclusion Criteria; NCT02915510
2.2.3. Inclusion Criteria; NCT03167697
2.2.4. Exclusion Criteria
2.2.5. Study Population
2.3. Dietary and Nutritional Intake
2.4. Amino Acid Profile
2.5. Statistical Analysis
3. Results
3.1. Dietary and Nutritional Intake
3.1.1. Energy and Macronutrient Intakes
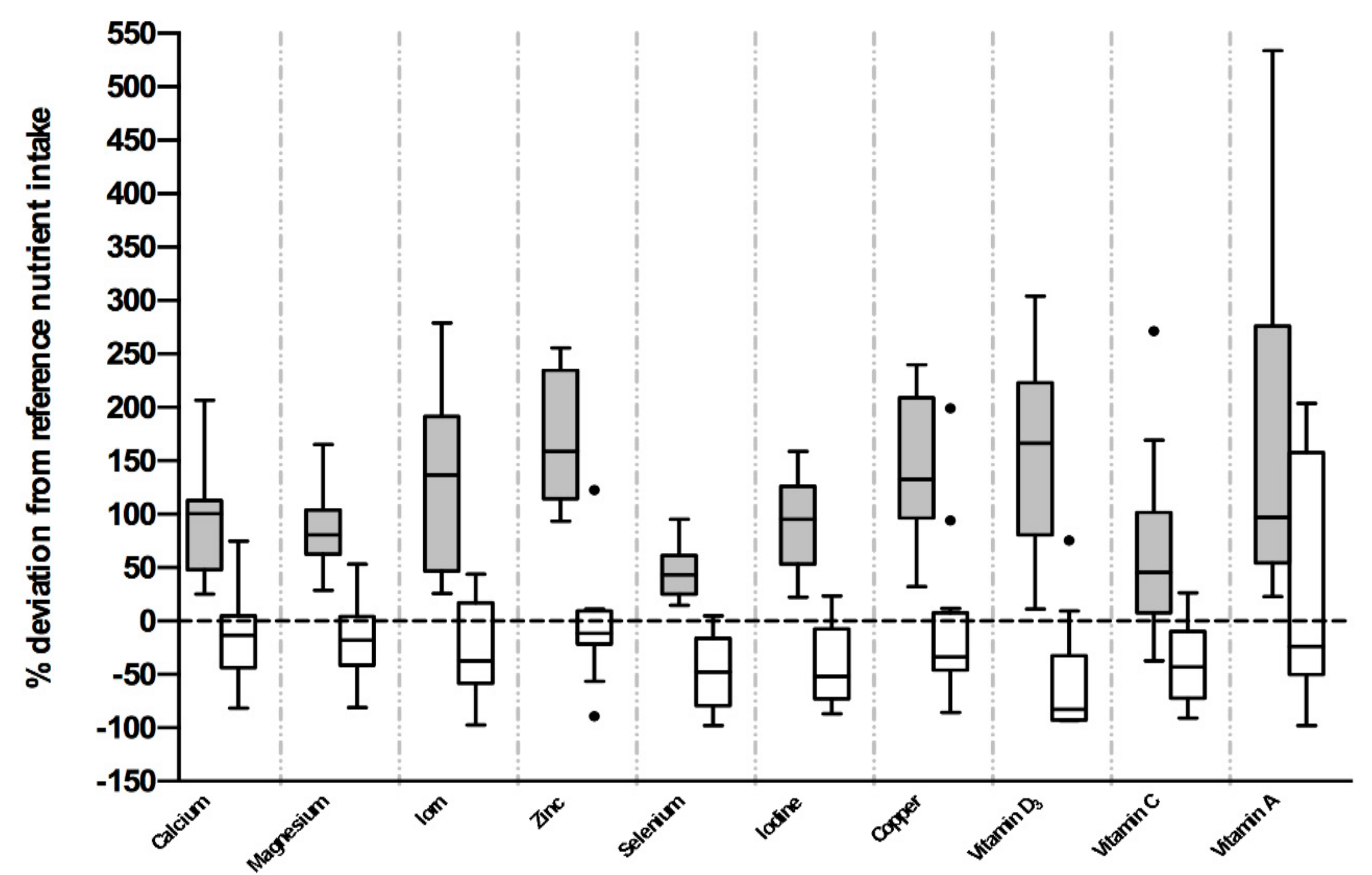
3.1.2. Micronutrient Intakes
3.2. Amino Acid Profile
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- De Groot, M.J.; Hoeksma, M.; Blau, N.; Reijngoud, D.J.; van Spronsen, F.J. Pathogenesis of cognitive dysfunction in phenylketonuria: Review of hypotheses. Mol. Genet. Metab. 2010, 99, S86–S89. [Google Scholar] [CrossRef] [PubMed]
- Blau, N.; van Spronsen, F.J.; Levy, H.L. Phenylketonuria. Lancet 2010, 376, 1417–1427. [Google Scholar] [CrossRef]
- Macdonald, A.; Daly, A.; Davies, P.; Asplin, D.; Hall, S.K.; Rylance, G.; Chakrapani, A. Protein substitutes for PKU: What’s new? J. Inherit. Metab. Dis. 2004, 27, 363–371. [Google Scholar] [CrossRef] [PubMed]
- Van Spronsen, F.J.; de Groot, M.J.; Hoeksma, M.; Reijngoud, D.-J.; van Rijn, M. Large neutral amino acids in the treatment of PKU: From theory to practice. J. Inherit. Metab. Dis. 2010, 33, 671–676. [Google Scholar] [CrossRef] [PubMed]
- Rocha, J.C.; Martel, F. Large neutral amino acids supplementation in phenylketonuric patients. J. Inherit. Metab. Dis. 2009, 32, 472. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, A.; Gokmen-Ozel, H.; van Rijn, M.; Burgard, P. The reality of dietary compliance in the management of phenylketonuria. J. Inherit. Metab. Dis. 2010, 33, 665–670. [Google Scholar] [CrossRef]
- Jurecki, E.R.; Cederbaum, S.; Kopesky, J.; Perry, K.; Rohr, F.; Sanchez-Valle, A.; Viau, K.S.; Sheinin, M.Y.; Cohen-Pfeffer, J.L. Adherence to clinic recommendations among patients with phenylketonuria in the United States. Mol. Genet. Metab. 2017, 120, 190–197. [Google Scholar] [CrossRef]
- Brown, C.S.; Lichter-Konecki, U. Phenylketonuria (PKU): A problem solved? Mol. Genet. Metab. Rep. 2016, 6, 8–12. [Google Scholar] [CrossRef]
- Ford, S.; O’Driscoll, M.; MacDonald, A. Living with Phenylketonuria: Lessons from the PKU community. Mol. Genet. Metab. Rep. 2018, 17, 57–63. [Google Scholar] [CrossRef]
- Walter, J.H.; White, F.J.; Hall, S.K.; MacDonald, A.; Rylance, G.; Boneh, A.; Francis, D.E.; Shortland, G.J.; Schmidt, M.; Vail, A. How practical are recommendations for dietary control in phenylketonuria? Lancet 2002, 360, 55–57. [Google Scholar] [CrossRef]
- Hochuli, M.; Bollhalder, S.; Thierer, C.; Refardt, J.; Gerber, P.; Baumgartner, M.R. Effects of Inadequate Amino Acid Mixture Intake on Nutrient Supply of Adult Patients with Phenylketonuria. Ann. Nutr. Metab. 2017, 71, 129–135. [Google Scholar] [CrossRef] [PubMed]
- Rohde, C.; von Teeffelen-Heithoff, A.; Thiele, A.G.; Arelin, M.; Mütze, U.; Kiener, C.; Gerloff, J.; Baerwald, C.; Schultz, S.; Heller, C.; et al. PKU patients on a relaxed diet may be at risk for micronutrient deficiencies. Eur. J. Clin. Nutr. 2013, 68, 119. [Google Scholar] [CrossRef] [PubMed]
- Infante, J.P.; Huszagh, V.A. Impaired Arachidonic (20:4n-6) and Docosahexaenoic (22:6n-3) Acid Synthesis by Phenylalanine Metabolites as Etiological Factors in the Neuropathology of Phenylketonuria. Mol. Genet. Metab. 2001, 72, 185–198. [Google Scholar] [CrossRef] [PubMed]
- Moseley, K.; Koch, R.; Moser, A.B. Lipid Status and Long-Chain Polyunsaturated Fatty Acid Concentrations in Adults and Adolescents with Phenylketonuria on Phenylalanine-Restricted Diet. J. Inherit. Metab. Dis. 2002, 25, 56–64. [Google Scholar] [CrossRef]
- Rocha, J.C.; MacDonald, A.; Trefz, F. Is overweight an issue in phenylketonuria? Mol. Genet. Metab. 2013, 110, S18–S24. [Google Scholar] [CrossRef]
- Dietary reference values for food energy and nutrients for the United Kingdom. Report of the Panel on Dietary Reference Values of the Committee on Medical Aspects of Food Policy. Rep. Health Soc. Subj. (Lond.) 1991, 41, 1–210.
- Strnadová, K.A.; Holub, M.; Mühl, A.; Heinze, G.; Ratschmann, R.; Mascher, H.; Stöckler-Ipsiroglu, S.; Waldhauser, F.; Votava, F.; Lebl, J.; et al. Long-Term Stability of Amino Acids and Acylcarnitines in Dried Blood Spots. Clin. Chem. 2007, 53, 717–722. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B (Methodol.) 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Van Spronsen, F.J.; van Wegberg, A.M.J.; Ahring, K.; Bélanger-Quintana, A.; Blau, N.; Bosch, A.M.; Burlina, A.; Campistol, J.; Feillet, F.; Giżewska, M.; et al. Key European guidelines for the diagnosis and management of patients with phenylketonuria. Lancet Diabetes Endocrinol. 2017, 5, 743–756. [Google Scholar] [CrossRef]
- Rocha, J.C.; van Spronsen, F.J.; Almeida, M.F.; Soares, G.; Quelhas, D.; Ramos, E.; Guimarães, J.T.; Borges, N. Dietary treatment in phenylketonuria does not lead to increased risk of obesity or metabolic syndrome. Mol. Genet. Metab. 2012, 107, 659–663. [Google Scholar] [CrossRef]
- Azabdaftari, A.; van der Giet, M.; Schuchardt, M.; Hennermann, J.B.; Plöckinger, U.; Querfeld, U. The cardiovascular phenotype of adult patients with phenylketonuria. Orphanet J. Rare Dis. 2019, 14, 213. [Google Scholar] [CrossRef] [PubMed]
- MacLeod, E.L.; Ney, D.M. Nutritional Management of Phenylketonuria. Ann. Nestlé (Engl. Ed.) 2010, 68, 58–69. [Google Scholar] [CrossRef] [PubMed]
- Roberts, C.; Steer, T.; Maplethorpe, N.; Cox, L.; Meadows, S.; Nicholson, S.; Page, P.; Swan, G. National Diet and Nutrition Survey: Results from Years 7 and 8 (Combined) of the Rolling Programme (2014/2015–2015/2016); Public Health England: London, UK, 2018. [Google Scholar]
- Van Wegberg, A.M.J.; MacDonald, A.; Ahring, K.; Bélanger-Quintana, A.; Blau, N.; Bosch, A.M.; Burlina, A.; Campistol, J.; Feillet, F.; Giżewska, M.; et al. The complete European guidelines on phenylketonuria: Diagnosis and treatment. Orphanet J. Rare Dis. 2017, 12, 162. [Google Scholar] [CrossRef] [PubMed]
- Barat, P.; Barthe, N.; Redonnet-Vernhet, I.; Parrot, F. The impact of the control of serum phenylalanine levels on osteopenia in patients with phenylketonuria. Eur. J. Pediatr. 2002, 161, 687–688. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Dueñas, B.; Cambra, F.; Vilaseca, M.; Lambruschini, N.; Campistol, J.; Camacho, J. New approach to osteopenia in phenylketonuric patients. Acta Paediatr. 2002, 91, 899–904. [Google Scholar] [CrossRef]
- Channon, S.; Goodman, G.; Zlotowitz, S.; Mockler, C.; Lee, P.J. Effects of dietary management of phenylketonuria on long-term cognitive outcome. Arch. Dis. Child. 2007, 92, 213–218. [Google Scholar] [CrossRef] [PubMed]
- Romani, C.; Palermo, L.; MacDonald, A.; Limback, E.; Hall, S.K.; Geberhiwot, T. The impact of phenylalanine levels on cognitive outcomes in adults with phenylketonuria: Effects across tasks and developmental stages. Neuropsychology 2017, 31, 242–254. [Google Scholar] [CrossRef]
- Pietz, J.; Kreis, R.; Rupp, A.; Mayatepek, E.; Rating, D.; Boesch, C.; Bremer, H.J. Large neutral amino acids block phenylalanine transport into brain tissue in patients with phenylketonuria. J. Clin. Investig. 1999, 103, 1169–1178. [Google Scholar] [CrossRef]
- Van Vliet, D.; Bruinenberg, V.M.; Mazzola, P.N.; van Faassen, M.H.J.R.; de Blaauw, P.; Kema, I.P.; Heiner-Fokkema, M.R.; van Anholt, R.D.; van der Zee, E.A.; van Spronsen, F.J. Large Neutral Amino Acid Supplementation Exerts Its Effect through Three Synergistic Mechanisms: Proof of Principle in Phenylketonuria Mice. PLoS ONE 2015, 10, e0143833. [Google Scholar] [CrossRef]
- Schindeler, S.; Ghosh-Jerath, S.; Thompson, S.; Rocca, A.; Joy, P.; Kemp, A.; Rae, C.; Green, K.; Wilcken, B.; Christodoulou, J. The effects of large neutral amino acid supplements in PKU: An MRS and neuropsychological study. Mol. Genet. Metab. 2007, 91, 48–54. [Google Scholar] [CrossRef]
- Ashley, J.M.; Bovee, V.H. Dietary intake: Recording and analyzing. In Obesity: Etiology, Assessment, Treatment and Prevention; Andersen, R., Ed.; Human Kinetics: Champaign, IL, USA, 2007; pp. 99–110. [Google Scholar]
- Ahring, K.; Bélanger-Quintana, A.; Dokoupil, K.; Ozel, H.G.; Lammardo, A.M.; MacDonald, A.; Motzfeldt, K.; Nowacka, M.; Robert, M.; van Rijn, M. Dietary management practices in phenylketonuria across European centres. Clin. Nutr. 2009, 28, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Bik-Multanowski, M.; Didycz, B.; Mozrzymas, R.; Nowacka, M.; Kaluzny, L.; Cichy, W.; Schneiberg, B.; Amilkiewicz, J.; Bilar, A.; Gizewska, M.; et al. Quality of life in noncompliant adults with phenylketonuria after resumption of the diet. J. Inherit. Metab. Dis. 2008, 31, 415–418. [Google Scholar] [CrossRef] [PubMed]
- Green, B.; Rahman, Y.; Firman, S.; Adam, S.; Jenkinson, F.; Nicol, C.; Adams, S.; Dawson, C.; Robertson, L.; Dunlop, C.; et al. Improved Eating Behaviour and Nutrient Intake in Noncompliant Patients with Phenylketonuria after Reintroducing a Protein Substitute: Observations from a Multicentre Study. Nutrients 2019, 11, 2035. [Google Scholar] [CrossRef] [PubMed]
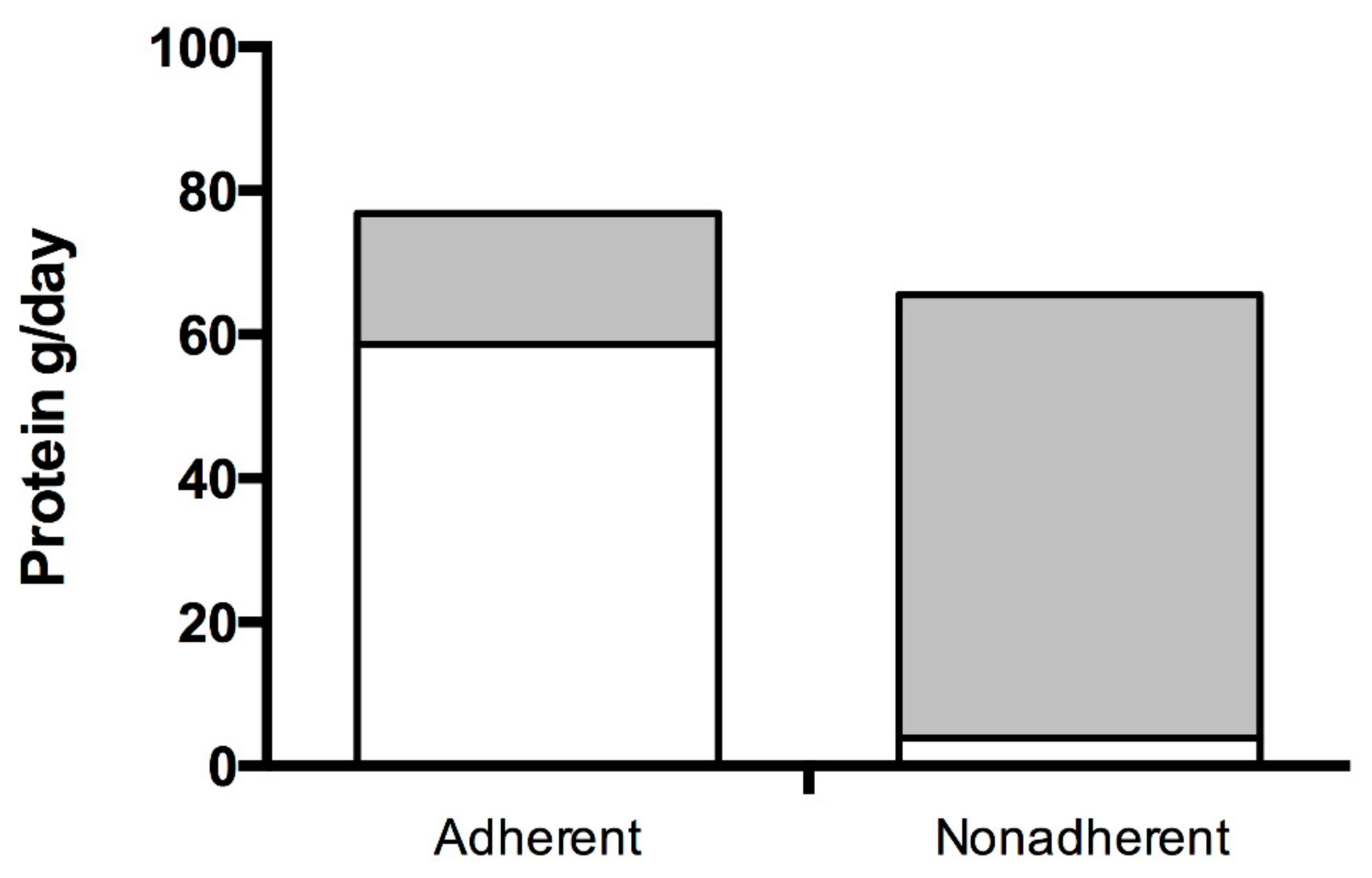
| Adherent (n = 16) | Nonadherent (n = 14) | p Value | |
|---|---|---|---|
| Age, y | 29.5 ± 11.2 | 33.9 ± 8.5 | 0.240 |
| Male: Female, n:n | 7:9 | 5:9 | 0.654 |
| Body weight, kg | 71.1 ± 15.7 | 88.8 ± 20.8 | 0.017 |
| Body mass index, kg/m2 | 24.9 ± 3.8 | 31.8 ± 7.6 | 0.006 |
| Historical Phenylalanine, µmol/L a | 618 ± 292 b | 1050 ± 341 | 0.001 |
| Adherent (n = 15) a | Nonadherent (n = 12) b | Unadjusted p Value | Adjusted q Value | ||
|---|---|---|---|---|---|
| Minerals | |||||
| Sodium | mg | 1673 ± 574 | 1958 ± 834 | 0.304 | 0.330 |
| Potassium | mg | 2983 ± 1152 | 2305± 949 | 0.113 | 0.135 |
| Chloride | mg | 2955 ± 2153 | 2777 ± 1089 | 0.681 | 0.709 |
| Calcium | mg | 1489 ± 337 | 619 ± 319 | <0.001 | <0.001 |
| Phosphorus | mg | 1456 ± 332 | 955 ± 415 | 0.002 | 0.003 |
| Magnesium | mg | 521 ± 123 | 238 ± 98.7 | <0.001 | <0.001 |
| Iron | mg | 26.9 ± 5.61 | 8.61 ± 5.02 | <0.001 | <0.001 |
| Zinc | mg | 22.3 ± 5.52 | 7.26 ± 3.53 | <0.001 | <0.001 |
| Copper | µg | 2.99 ± 0.70 | 1.14 ± 0.94 | 0.003 | 0.004 |
| Manganese | mg | 4.97 ± 1.55 | 4.47 ± 5.75 | 0.081 | 0.101 |
| Selenium | µg | 95.8 ± 16.4 | 34.4 ± 21.9 | <0.001 | <0.001 |
| Iodine | µg | 266 ± 57.0 | 80.5 ± 50.2 | <0.001 | <0.001 |
| Vitamins | |||||
| Vitamin A | µg RE | 1842 ± 1138 | 1162 ± 1494 | 0.018 | 0.025 |
| Vitamin D3 | µg | 25.8 ± 8.96 | 4.16 ± 5.31 | <0.001 | <0.001 |
| Vitamin E | mg α-TE | 21.5 ± 5.71 | 7.6 ± 4.88 | 0.001 | 0.001 |
| Vitamin K1 | µg | 122 ± 38.1 | 34.2 ± 35.6 | <0.001 | 0.001 |
| Thiamin | mg | 2.82 ± 0.77 | 1.37 ± 0.81 | 0.014 | 0.020 |
| Riboflavin | mg | 2.90 ± 0.58 | 1.29 ± 0.57 | 0.001 | <0.001 |
| Niacin | mg | 58.7 ± 12.9 | 31.1 ± 16.5 | <0.001 | <0.001 |
| Pantothenic acid | mg | 10.4 ± 2.24 | 4.64 ± 2.10 | <0.001 | <0.001 |
| Vitamin B6 | mg | 3.55 ± 0.79 | 1.93 ± 1.10 | 0.019 | 0.025 |
| Folate | µg | 203 ± 134 | 231 ± 115 | 0.845 | 0.845 |
| Vitamin B12 | µg | 5.70 ± 1.19 | 4.18 ± 3.97 | 0.223 | 0.253 |
| Biotin | µg | 196 ± 38.8 | 34.8 ± 35.2 | <0.001 | <0.001 |
| Vitamin C | mg | 229 ± 122 | 89.4 ± 65.3 | 0.001 | 0.003 |
| 95% Reference Range µmol/L a | Adherent (n = 16) | Nonadherent (n = 13) | Unadjusted p Value | Adjusted q Value | |
|---|---|---|---|---|---|
| Arginine | 17–91 | 41.3 ± 12.7 | 40.2 ± 16.2 | 0.848 | 0.923 |
| Asparagine | 42–88 | 49.6 ± 9.54 | 51.0 ± 7.35 | 0.673 | 1.121 |
| Aspartic Acid | 26–233 | 112 ± 37.3 | 144 ± 51.7 | 0.225 | 0.643 |
| Citrulline | 16–51 | 28.8 ± 12.6 | 26.5 ± 6.81 | 0.651 | 1.184 |
| Glutamic acid | 97–258 | 155 ± 84.2 | 178 ± 45.7 | 0.127 | 0.423 |
| Glutamine | 209–573 | 306 ± 81.7 | 302 ± 102 | 0.922 | 0.922 |
| Glycine | 207–559 | 344 ± 78.3 | 325 ± 90.7 | 0.556 | 1.112 |
| Histidine | 22–99 | 67.9 ± 24.8 | 74.4 ± 16.5 | 0.429 | 0.953 |
| Isoleucine | 28–96 | 45.3 ± 16.6 | 46.2 ± 13.1 | 0.772 | 0.965 |
| Leucine | 59–162 | 89.0 ± 20.6 | 91.5 ± 19.6 | 0.746 | 1.066 |
| Lysine | 63–220 | 111 ± 27.9 | 104 ± 13.1 | 0.376 | 0.940 |
| Methionine | 10–33 | 19.2 ± 6.25 | 13.9 ± 3.23 | 0.025 | 0.167 |
| Ornithine | 50–210 | 104 ± 99.2 | 83.2 ± 19.0 | 0.777 | 0.914 |
| Phenylalanine | 120–600 b | 464 ± 196 | 861 ± 348 | 0.002 | 0.040 |
| Serine | 79–310 | 181 ± 112 | 171 ± 70.0 | 0.880 | 0.926 |
| Taurine | 124–282 | 172 ± 30.8 | 202 ± 49.5 | 0.053 | 0.212 |
| Threonine | 54–169 | 112 ± 44.8 | 82.8 ± 23.7 | 0.026 | 0.130 |
| Tryptophan | 24–52 | 36.6 ± 6.47 | 30.1 ± 4.27 | 0.004 | 0.040 |
| Tyrosine | 36–99 | 47.8 ± 11.2 | 49.3 ± 13.1 | 0.743 | 1.143 |
| Valine | 105–266 | 162 ± 39.2 | 156 ± 26.9 | 0.764 | 1.019 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Green, B.; Browne, R.; Firman, S.; Hill, M.; Rahman, Y.; Kaalund Hansen, K.; Adam, S.; Skeath, R.; Hallam, P.; Herlihy, I.; et al. Nutritional and Metabolic Characteristics of UK Adult Phenylketonuria Patients with Varying Dietary Adherence. Nutrients 2019, 11, 2459. https://doi.org/10.3390/nu11102459
Green B, Browne R, Firman S, Hill M, Rahman Y, Kaalund Hansen K, Adam S, Skeath R, Hallam P, Herlihy I, et al. Nutritional and Metabolic Characteristics of UK Adult Phenylketonuria Patients with Varying Dietary Adherence. Nutrients. 2019; 11(10):2459. https://doi.org/10.3390/nu11102459
Chicago/Turabian StyleGreen, Benjamin, Robert Browne, Sarah Firman, Melanie Hill, Yusof Rahman, Kit Kaalund Hansen, Sarah Adam, Rachel Skeath, Paula Hallam, Ide Herlihy, and et al. 2019. "Nutritional and Metabolic Characteristics of UK Adult Phenylketonuria Patients with Varying Dietary Adherence" Nutrients 11, no. 10: 2459. https://doi.org/10.3390/nu11102459
APA StyleGreen, B., Browne, R., Firman, S., Hill, M., Rahman, Y., Kaalund Hansen, K., Adam, S., Skeath, R., Hallam, P., Herlihy, I., Jenkinson, F., Nicol, C., Adams, S., Gaff, L., Donald, S., Dawson, C., Robertson, L., Fitzachary, C., Chan, H., ... Stratton, R. (2019). Nutritional and Metabolic Characteristics of UK Adult Phenylketonuria Patients with Varying Dietary Adherence. Nutrients, 11(10), 2459. https://doi.org/10.3390/nu11102459






