Sweet Taste as a Predictor of Dietary Intake: A Systematic Review
Abstract
1. Introduction
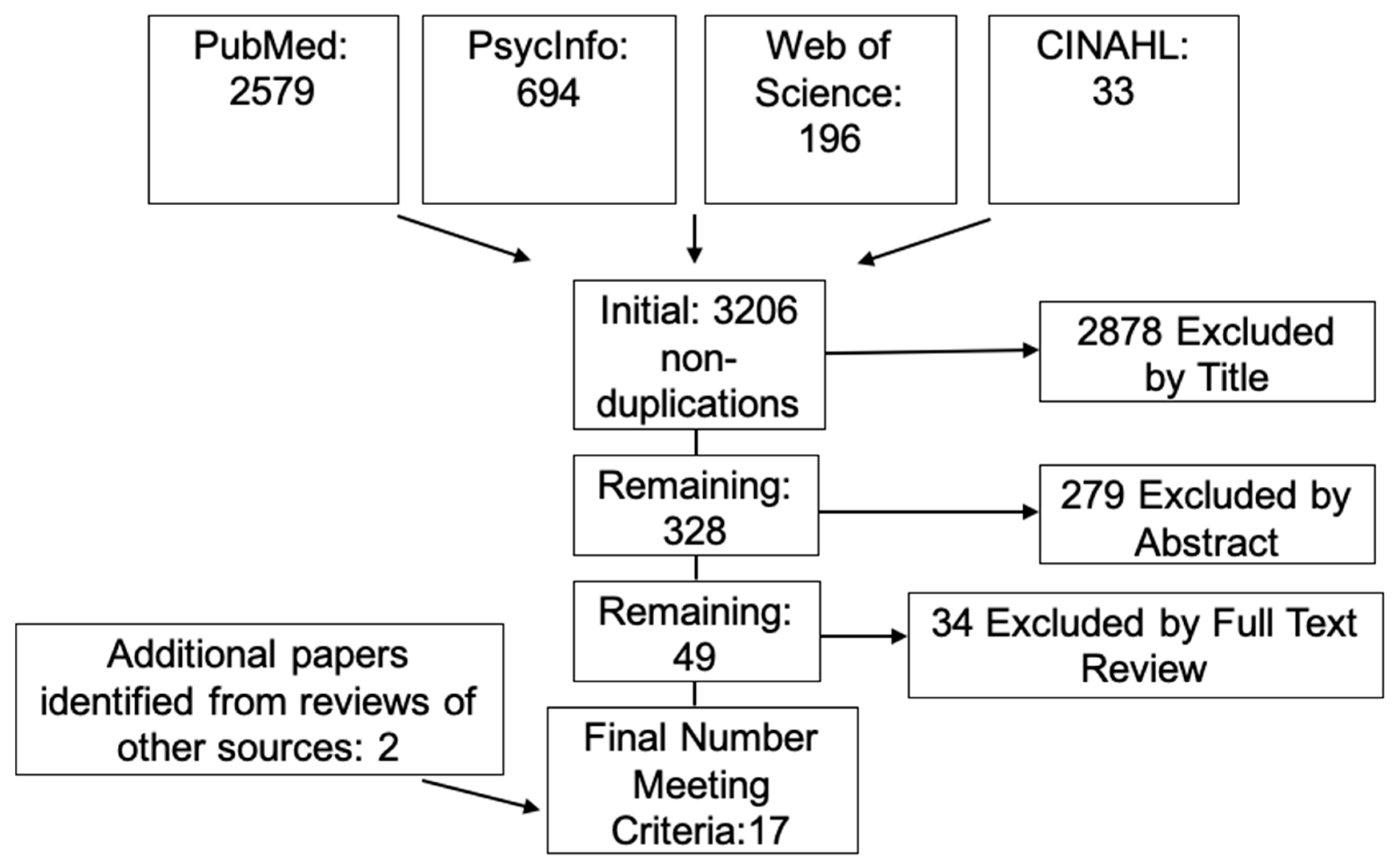
2. Materials and Methods
3. Results
3.1. Sensitivity Testing
3.2. Intensity Testing
3.3. Hedonic Testing
3.3.1. Studies that Determined Sweet Liking Phenotypes
3.3.2. Studies that Did Not Determine Sweet Liking Phenotypes
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Feeney, E.; O’Brien, S.; Scannell, A.; Markey, A.; Gibney, E.R. Genetic variation in taste perception: Does it have a role in healthy eating? Proc. Nutr. Soc. 2011, 70, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Glanz, K.; Basil, M. Why Americans eat what they do: Taste, nutrition, cost, convenience, and weight control concerns. J. Am. Diet. Assoc. 1998, 98, 1118–1126. [Google Scholar] [CrossRef]
- Keast, R.S.J.; Dalton, P.H.; Breslin, P.A.S. Flavor interactions at the sensory level. In Flavor Perception; Taylor, A.J., Roberts, D.D., Eds.; Blackwell Publishing Ltd.: Oxford, UK, 2004; ISBN 978-1-4051-5001-9. [Google Scholar]
- Garcia-Bailo, B.; Toguri, C.; Eny, K.M.; El-Sohemy, A. Genetic variation in taste and its influence on food selection. OMICS J. Integr. Biol. 2009, 13, 69–80. [Google Scholar] [CrossRef]
- Tucker, R.M.; Mattes, R.D. Are free fatty acids effective taste stimuli in humans? Presented at the symposium “The taste for fat: New discoveries on the role of fat in sensory perception, metabolism, sensory pleasure and beyond” held at the Institute of Food Technologists 2011 Annual Meeting, New Orleans, LA., 12 June 2011. J. Food Sci. 2012, 77, S148–S151. [Google Scholar] [PubMed]
- Lawless, H.T.; Heymann, H. Sensory Evaluation of Food: Principles and Practices, 2nd ed.; Springer: New York, NY, USA, 2010. [Google Scholar]
- Leong, C.S.-F.; Forde, C.G.; Tey, S.L.; Henry, C.J. Taste perception and diet in people of Chinese ancestry. Asia Pac. J. Clin. Nutr. 2018, 27, 478. [Google Scholar]
- Weizenbaum, F.; Benson, B.; Solomon, L.; Brehony, K. Relationship among reproductive variables, sucrose taste reactivity and feeding behavior in humans. Physiol. Behav. 1980, 24, 1053–1056. [Google Scholar] [CrossRef]
- Jayasinghe, S.N.; Kruger, R.; Walsh, D.C.; Cao, G.; Rivers, S.; Richter, M.; Breier, B.H. Is sweet taste perception associated with sweet food liking and intake? Nutrients 2017, 9, 750. [Google Scholar] [CrossRef] [PubMed]
- Webb, J.; Bolhuis, D.P.; Cicerale, S.; Hayes, J.E.; Keast, R. The relationships between common measurements of taste function. Chemosens. Percept. 2015, 8, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Landis, B.N.; Welge-Luessen, A.; Bramerson, A.; Bende, M.; Mueller, C.A.; Nordin, S.; Hummel, T. “Taste Strips”—A rapid, lateralized, gustatory bedside identification test based on impregnated filter papers. J. Neurol. 2009, 256, 242–248. [Google Scholar] [CrossRef]
- Green, B.G. Studying taste as a cutaneous sense. Food Qual. Prefer. 2003, 14, 99–109. [Google Scholar] [CrossRef]
- Hummel, T.; Erras, A.; Kobal, G. A test for screening of taste function. Rhinology 1997, 35, 146–148. [Google Scholar] [PubMed]
- Smutzer, G.; Lam, S.; Hastings, L.; Desai, H.; Abarintos, R.A.; Sobel, M.; Sayed, N. A test for measuring gustatory function. Laryngoscope 2008, 118, 1411–1416. [Google Scholar] [CrossRef] [PubMed]
- Duffy, V.B.; Peterson, J.M.; Dinehart, M.E.; Bartoshuk, L.M. Genetic and environmental variation in taste: Associations with sweet intensity, preference, and intake. Top. Clin. Nutr. 2003, 18, 209–220. [Google Scholar] [CrossRef]
- Mattes, R.D. Gustation as a determinant of ingestion: Methodological issues. Am. J. Clin. Nutr. 1985, 41, 672–683. [Google Scholar] [CrossRef] [PubMed]
- Lucas, F.; Bellisle, F. The measurement of food preferences in humans: Do taste-and-spit tests predict consumption? Physiol. Behav. 1987, 39, 739–743. [Google Scholar] [CrossRef]
- Tucker, R.M.; Kaiser, K.A.; Parman, M.A.; George, B.J.; Allison, D.B.; Mattes, R.D. Comparisons of fatty acid taste detection thresholds in people who are lean vs. overweight or obese: A systematic review and meta-analysis. PLoS ONE 2017, 12, e0169583. [Google Scholar] [CrossRef]
- Johnson, R.J.; Segal, M.S.; Sautin, Y.; Nakagawa, T.; Feig, D.I.; Kang, D.-H.; Gersch, M.S.; Benner, S.; Sanchez-Lozada, L.G. Potential role of sugar (fructose) in the epidemic of hypertension, obesity and the metabolic syndrome, diabetes, kidney disease, and cardiovascular disease. Am. J. Clin. Nutr. 2007, 86, 899–906. [Google Scholar]
- Johnson, R.J.; Nakagawa, T.; Sanchez-Lozada, L.G.; Shafiu, M.; Sundaram, S.; Le, M.; Ishimoto, T.; Sautin, Y.Y.; Lanaspa, M.A. Sugar, uric acid, and the etiology of diabetes and obesity. Diabetes 2013, 62, 3307–3315. [Google Scholar] [CrossRef]
- Malik, V.; Hu, F. Sweeteners and risk of obesity and type 2 diabetes: The role of sugar-sweetened beverages. Curr. Diab. Rep. 2012, 12, 195–203. [Google Scholar] [CrossRef]
- Dietary Guidelines Advisory Committee. Scientific Report of the 2015 Dietary Guidelines Advisory Committee; USDA and US Department of Health and Human Services: Washington, DC, USA, 2015.
- Australian National Health and Medical Research Council. Eat for Health: Australian Dietary Guidelines; Australian Government: Canberra, Australia, 2013.
- World Health Organization. Guideline: Sugars Intake for Adults and Children; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- Berridge, K.C.; Robinson, T.E.; Aldridge, J.W. Dissecting components of reward: ‘Liking’, ‘wanting’, and learning. Curr. Opin. Pharmacol. 2009, 9, 65–73. [Google Scholar] [CrossRef]
- Peciña, S. Opioid reward ‘liking’ and ‘wanting’ in the nucleus accumbens. Physiol. Behav. 2008, 94, 675–680. [Google Scholar] [CrossRef] [PubMed]
- Green, E.; Murphy, C. Altered processing of sweet taste in the brain of diet soda drinkers. Physiol. Behav. 2012, 107, 560–567. [Google Scholar] [CrossRef]
- Garneau, N.L.; Nuessle, T.M.; Mendelsberg, B.J.; Shepard, S.; Tucker, R.M. Sweet liker status in children and adults: Consequences for beverage intake in adults. Food Qual. Prefer. 2018, 65, 175–180. [Google Scholar] [CrossRef]
- Low, J.Y.Q.; Lacy, K.E.; McBride, R.; Keast, R.S.J. The association between sweet taste function, anthropometry, and dietary intake in adults. Nutrients 2016, 8, 241. [Google Scholar] [CrossRef] [PubMed]
- Hjartåker, A.; Andersen, L.F.; Lund, E. Comparison of diet measures from a food-frequency questionnaire with measures from repeated 24-hour dietary recalls. The Norwegian Women and Cancer Study. Public Health Nutr. 2007, 10, 1094–1103. [Google Scholar] [CrossRef]
- Smith, S.L.; Ludy, M.-J.; Tucker, R.M. Changes in taste preference and steps taken after sleep curtailment. Physiol. Behav. 2016, 163, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Cordero, E.; Malacara-Hernandez, J.M.; Martinez-Cordero, C. Taste perception in normal and overweight Mexican adults. Appetite 2015, 89, 192–195. [Google Scholar] [CrossRef]
- Han, P.; Keast, R.S.; Roura, E. Salivary leptin and TAS1R2/TAS1R3 polymorphisms are related to sweet taste sensitivity and carbohydrate intake from a buffet meal in healthy young adults. Br. J. Nutr. 2017, 118, 763–770. [Google Scholar] [CrossRef]
- Sartor, F.; Donaldson, L.F.; Markland, D.A.; Loveday, H.; Jackson, M.J.; Kubis, H.P. Taste perception and implicit attitude toward sweet index and soft drink supplementation. Appetite 2011, 57, 237–246. [Google Scholar] [CrossRef]
- Cicerale, S.; Riddell, L.J.; Keast, R.S. The association between perceived sweetness intensity and dietary intake in young adults. J. Food Sci. 2012, 77, H31–H35. [Google Scholar] [CrossRef]
- Stevenson, R.J.; Boakes, R.A.; Oaten, M.J.; Yeomans, M.R.; Mahmut, M.; Francis, H.M. Chemosensory abilities in consumers of a western-style diet. Chem. Senses 2016, 41, 505–513. [Google Scholar] [CrossRef]
- Holt, S.H.A.; Cobiac, L.; Beaumont-Smith, N.E.; Easton, K.; Best, D.J. Dietary habits and the perception and liking of sweetness among Australian and Malaysian students: A cross-cultural study. Food Qual. Prefer. 2000, 11, 299–312. [Google Scholar] [CrossRef]
- Hodge, A.; Patterson, A.J.; Brown, W.J.; Ireland, P.; Giles, G. The Anti Cancer Council of Victoria FFQ: Relative validity of nutrient intakes compared with weighed food records in young to middle-aged women in a study of iron supplementation. Aust. N. Z. J. Public Health 2000, 24, 576–583. [Google Scholar] [CrossRef] [PubMed]
- Drewnowski, A.; Henderson, S.A.; Levine, A.; Hann, C. Taste and food preferences as predictors of dietary practices in young women. Public Health Nutr. 1999, 2, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Turner-McGrievy, G.; Tate, D.F.; Moore, D.; Popkin, B. Taking the bitter with the sweet: Relationship of supertasting and sweet preference with metabolic syndrome and dietary intake. J. Food Sci. 2013, 78, S336–S342. [Google Scholar] [CrossRef] [PubMed]
- Methven, L.; Xiao, C.; Cai, M.; Prescott, J. Rejection thresholds (RjT) of sweet likers and dislikers. Food Qual. Prefer. 2016, 52, 74–80. [Google Scholar] [CrossRef]
- Mattes, R.D.; Mela, D.J. Relationships between and among selected measures of sweet-taste preference and dietary intake. Chem. Senses 1986, 11, 523–539. [Google Scholar] [CrossRef]
- Thompson, F.E.; Byers, T. Dietary assessment resource manual. J. Nutr. 1994, 124, 2245s–2317s. [Google Scholar]
- Bartoshuk, L.M. Methodological problems in psychophysical testing of taste and smell. In Special Senses in Aging: A Current Biological Assessment; Han, S.S., Coons, D.H., Eds.; University of Michigan: Ann Arbor, MI, USA, 1979. [Google Scholar]
- Iatridi, V.; Hayes, J.E.; Yeomans, M.R. Reconsidering the classification of sweet taste liker phenotypes: A methodological review. Food Qual. Prefer. 2018. [Google Scholar] [CrossRef]
- Low, J.Y.Q.; McBride, R.L.; Lacy, K.E.; Keast, R.S.J. Psychophysical evaluation of sweetness functions across multiple sweeteners. Chem. Senses 2017, 42, 111–120. [Google Scholar] [CrossRef]
- Pangborn, R.M. Relative taste intensities of selected sugars and organic acids. J. Food Sci. 1963, 28, 726–733. [Google Scholar] [CrossRef]
- Li, X.; Staszewski, L.; Xu, H.; Durick, K.; Zoller, M.; Adler, E. Human receptors for sweet and umami taste. Proc. Natl. Acad. Sci. USA 2002, 99, 4692–4696. [Google Scholar] [CrossRef]
- Trabulsi, J.; Schoeller, D.A. Evaluation of dietary assessment instruments against doubly labeled water, a biomarker of habitual energy intake. Am. J. Physiol. Endocrinol. Metab. 2001, 281, E891–E899. [Google Scholar] [CrossRef] [PubMed]
- Basiotis, P.P.; Welsh, S.O.; Cronin, F.J.; Kelsay, J.L.; Mertz, W. Number of days of food intake records required to estimate individual and group nutrient intakes with defined confidence. J. Nutr. 1987, 117, 1638–1641. [Google Scholar] [CrossRef] [PubMed]
- Gersovitz, M.; Madden, J.P.; Smiciklas-Wright, H. Validity of the 24-h. dietary recall and seven-day record for group comparisons. J. Am. Diet. Assoc. 1978, 73, 48–55. [Google Scholar]
- Nakamura, Y.; Sanematsu, K.; Ohta, R.; Shirosaki, S.; Koyano, K.; Nonaka, K.; Shigemura, N.; Ninomiya, Y. Diurnal variation of human sweet taste recognition thresholds is correlated with plasma leptin levels. Diabetes 2008, 57, 2661–2665. [Google Scholar] [CrossRef] [PubMed]
- Stevens, J.C.; Cruz, L.A.; Hoffman, J.M.; Patterson, M.Q. Taste sensitivity and aging: High incidence of decline revealed by repeated threshold measures. Chem. Senses 1995, 20, 451–459. [Google Scholar] [CrossRef] [PubMed]
| Author (Year) | Subjects | Taste Test | Sweet Stimuli | Stimuli Concentrations | Dietary Assessment Methods | Key Findings |
|---|---|---|---|---|---|---|
| Mattes (1985) [16] | n = 35 (17 M, 18 F) Age = 18–42 years old | RT | Sucrose | Serial half dilutions of sucrose: 1.2 × 10−5 M to 0.8 M | 7-day diet record with predominant taste recorded | Sweet taste threshold and intensity did not correlate with sweet E, CHO, PRO and fat intake. |
| Martinez-Cordero (2015) [32] | n = 56 (30 M, 26 F) Age = 32.9 ± 7.9 years old | DT | Sucrose Aspartame | Sucrose—14 [ ] from 4.09 × 10−1 M to 1.63 × 102 M Aspartame—14 [ ] from 0.82 × 10−3 M to 3.27 × 10−1 M Both at 0.2 log dilutions per successive solution | 7-day food diaries | Aspartame threshold was negatively associated with E intake (B = −0.003 ± 0.001; p < 0.0009). No association between sucrose threshold and dietary intake. |
| Low (2016) [29] | n = 60 Age = 26.5 ± 1.0 years old | DT; RT | Glucose mono-hydrate Fructose Sucrose Sucralose Erithritol Rebaudio-side A | Varying concentrations for each | Validated FFQ; also assessed consumption of foods and/or beverages sweetened with high-intensity sweeteners | No association between threshold measures and dietary measures. |
| Smith (2016) [31] | n = 51 (9 M, 42 F) Age = 25 ± 8y | DT | Sucrose | 2.1% w/v sucrose Quarter-log step dilutions | 24-hour recall | No association between threshold measures and dietary intake. |
| Han (2017) [33] | n = 30 (16 M, 14 F) Age = 24–34 years old (M), 20–37 years old (F) | Sensi-tivity | Sucrose | 9 mM | Ad libitum intake after soup preload (one sweet, one umami, one no-taste energy control) | Highly-sensitive consumed more non-sweet foods, PRO, %E from PRO, and %E from fat (after non-sweet soup only) (p < 0.05 for all). Highly-sensitive consumed less CHO as %E (p = 0.02). |
| Jayasinghe (2017) [9] | n = 42 (all F) Age = 28 ± 634 years old | DT; RT | Glucose | 15, 30, 45, 60, 90, 120, 150, 180 mM | 4-day weighed food record Sweet food FFQ Sweet beverage liking questionnaire | No association between threshold measures and dietary intake. |
| Author (Year) | Subjects | Taste Test | Sweet Stimuli | Stimuli Concentrations | Dietary Assessment Methods | Key Findings |
|---|---|---|---|---|---|---|
| Mattes (1985) [16] | n = 35 (17 M, 18 F) Age = 18–42 years old | Intensity | Sucrose | 5 concentrations ranging from 0.05 M to 0.80 M | 7-day diet records | No association between intensity measures and dietary intake. |
| Holt (2000) [37] | n = 132, Australian 27 M, 42 F Malaysian 29 M, 34 F; Australian 22.8 ± 4.3 years old Malaysian 21.5 ± 1.2 years old | Intensity | Sucrose | 2, 4, 8, 16 and 32% v/v | Separate FFQ for the Australian and Malaysian participants | No association between intensity measures and dietary intake. |
| Sartor (2011) [34] | n = 12 (7 M, 5 F) Age = 26 ± 6 years old | Intensity | Sucrose | 0, −0.5, −0.75, −1, −1.25, −1.5, −1.75, −2, −2.25, −2.5, −2.75 log(sucrose) mol/L | 14 diet diaries on random days | No association between intensity measures and dietary intake. |
| Cicerale (2012) [35] | n = 85 (89% F) Age = 21 ± 4 years old | Intensity | Sucrose | 200 mM | Food & diet questionnaire Food variety survey 2 × 24-hour food diaries | No association between intensity and any diet measures. |
| Low (2016) [29] | n = 60 Age = 26.5 ± 1.0 years old (SEM) | Intensity | Glucose mono-hydrate Fructose Sucrose Sucralose Erithritol Rebaud-ioside A | Varying concentrations | Validated FFQ; also assessed consumption of foods and/or beverages sweetened with high-intensity sweeteners | Intensity and dietary intake associations varied by sweetener. Rebaudioside A and sucralose intensity ratings were positively associated with mean total E intake (p < 0.01 for both). |
| Stevenson (2016) [36] | n = 87 (38 M, 49 F) Age = 21 ± 3 years old (18–31 years old) | Intensity | Sucrose | 0.03 M and 0.36 M | 26-item Dietary Fat and Sugar questionnaire (DFS) designed to identify variation in saturated fat and added sugar intake | No association between intensity and any diet measures. |
| Jayasinghe (2017) [9] | n = 42 (all F) Age = 28 ± 6 years old | Intensity | Glucose | 125, 250, 500, 1000 mM | 4-day weighed food record Sweet food FFQ Sweet beverage liking questionnaire | Intensity at 250 mM or higher correlated negatively with total E, CHO (starch, total sugar, fructose, glucose) but not sucrose intake (p < 0.05 for all). Intensity also negatively associated with total sweet food intake (p < 0.05 for all). |
| Leong (2018) [7] | n = 100 (50 M, 50 F) Age = 25.7 ± 4.2 years old (M), 25.7 ± 5.1 years old (F) | Intensity | Sucrose | 12.0% w/v | 2 × 24-hour food recalls | No association between intensity and any diet measures. |
| Author (Year) | Subjects | Taste Test | Sweet Stimuli | Stimuli Concentrations | Dietary Assessment Methods | Key Findings |
|---|---|---|---|---|---|---|
| Weizenbaum (1980) [8] | n = 17 (5 M, 12 F) Age = 18.6 y (M), 19.7 years old (F) | Pleasantness | Sucrose | 0.01, 0.023, 0.046, 0.1, 0.23, 0.46, 1.0 M | Ad libitum intake of salted peanuts and candies after testing | No relationship between pleasantness and amount of food consumed. |
| Mattes (1985) [16] | n = 35 (17 M, 18 F) Age = 18–42 years old | Preferred concentration of sweetness | Sucrose | Self-adjusted (dilution) | 7-day diet records | Preferred concentration of sweet solution negatively correlated (r = −0.36, p = 0.04) with CHO intake. |
| Mattes (1986) [42] | n = 25 (all M) Age = 17–34 years old | Preferred concentration of sweetness using an adjustment task | Sucrose | 0M & 1.0 M solutions were provided. Subjects modified the samples until the preferred sweetness was reached. Preferred sweetness levels from both the unsweetened and sweetened baseline stimuli were averaged. | 7-day diet records | Mean preferred concentration was positively correlated with %CHO intake (r = 0.637, p < 0.001). Preferred concentration of the 1.0 M sucrose samples were positively correlated with %CHO intake (r = 0.748, p < 0.001), % sweet calorie intake (r = 0.504, p < 0.001), and frequency of selection of carbohydrate-rich foods (r = 0.532, p < 0.01). |
| Drewnowski (1999) [39] | n = 159 (all F) Age = 27.0 ± 0.7 years old (SEM) | Liking | Sucrose | 5 [ ] ranging from 2% to 32% w/v | 3-day food records; 171-item food preference checklist | No associations between liking and dietary intake measures, but higher hedonic ratings for sucrose were associated with higher ratings for sugar in tea and many sweet desserts. |
| Holt (2000) [37] | n = 132, separated into Australian-born Caucasian and Malaysian born; Australian: 27 M, 42 F Malaysian: 29 M, 34 F; Australian: 22.8 ± 4.3 years old Malaysian: 21.5 ± 1.2 years old | Liking | Sucrose | 2, 4, 8, 16 and 32% v/v | Separate FFQs for the Australian and Malaysian subjects | Refined sugar intake was higher in sweet likers com-pared to dislikers. No other differences were observed. For all participants, positive associations between the preferred level of sucrose and frequency of sweet food consumption, intake of refined sugars, and total sugars were observed (p < 0.05). |
| Sartor (2011) [34] | n = 12 (7 M, 5 F) Age = 26 ± 6 years old | Pleasantness Preference | Sucrose | Pleasantness 11 [ ]: 0, −0.5, −0.75, −1, −1.25, −1.5, −1.75, −2, −2.25, −2.5, −2.75 log(sucrose) M Preference: 10 random presentations of pairs of 0, −0.5, −0.75, −1 and −1.25 log(sucrose) M | 14 diet diaries on random days | No associations between taste measures and dietary intake. |
| Turner-McGrievy (2013) [40] | n = 196 (85% F) Age = 42.6 ± 11.0 years old | Liking | Sucrose | 0.05, 0.10, 0.21, 0.42, and 0.83 M. Participants who liked the 0.83 M sucrose solution the best were classified as sweet likers | 2 × 24-hour food recalls | Those who were sweet likers consumed more E from beverages and less fiber (p < 0.05). |
| Methven (2016) [41] | n = 36 (12 M, 23 F, 1 unknown) Age = 26 years old (median) | Liking | Sucrose | 3%, 6%, 12%, 24%, 36% | FFQ used by EPIC | Intake did not differ between sweet likers and dislikers. |
| Smith (2016) [31] | n = 51 (9 M, 42 F) Age = 25 ± 8 years old | Preferred concentration of sweetness | Sucrose | 2.1% w/v stock solution Quarter-log step dilutions | 24-hour recall | Sweet preference after short-sleep was positively correlated with E intake (r = 0.31, p = 0.043) and CHO intake (r = 0.32, p = 0.34), but not after habitual sleep. |
| Stevenson (2016) [36] | n = 87 (38 M, 49 F) Age = 21 ± 3 years old (18–31 years old) | Liking | Sucrose | 0.03 & 0.36 M | 26-item Dietary Fat and Sugar questionnaire (DFS) designed to identify variation in saturated fat and added sugar intake. | No association between liking and any diet measures. |
| Jayasinghe (2017) [9] | n = 42 (all F) Age = 28 ± 6 years old | Liking | Glucose | 125, 250, 500, 1000 mM | 4-day weighed food record; Sweet food FFQ; Sweet beverage liking questionnaire | Sweet taste liking at 500 mM or higher correlated positively with total E, CHO (total sugar, fructose, glucose) (p < 0.05 for all) but not starch and sucrose intake. |
| Garneau (2018) [28] | n = 418 | Liking | Sucrose | 5 [ ] ranging from 0% w/v to 13.7% w/v | Validated beverage FFQ (BEVQ-15) | Mean E intake from all beverages was higher among likers compared to neutrals (p = 0.004). Total E intake by dislikers did not differ from the other groups. E intake from sugar-sweetened beverages was higher among likers compared to dislikers (p = 0.008). Neutrals did not differ from the other groups. |
| Leong (2018) [7] | n = 100 (50 M, 50 F) Age = 25.7 ± 4.2 years old (M), 25.7 ± 5.1 years old (F) | Liking | Sucrose | 12.0% w/v | 2 × 24-hour food recalls | No association with liking and dietary intake. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tan, S.-Y.; Tucker, R.M. Sweet Taste as a Predictor of Dietary Intake: A Systematic Review. Nutrients 2019, 11, 94. https://doi.org/10.3390/nu11010094
Tan S-Y, Tucker RM. Sweet Taste as a Predictor of Dietary Intake: A Systematic Review. Nutrients. 2019; 11(1):94. https://doi.org/10.3390/nu11010094
Chicago/Turabian StyleTan, Sze-Yen, and Robin M. Tucker. 2019. "Sweet Taste as a Predictor of Dietary Intake: A Systematic Review" Nutrients 11, no. 1: 94. https://doi.org/10.3390/nu11010094
APA StyleTan, S.-Y., & Tucker, R. M. (2019). Sweet Taste as a Predictor of Dietary Intake: A Systematic Review. Nutrients, 11(1), 94. https://doi.org/10.3390/nu11010094






