Dual Anticoagulant/Antiplatelet Activity of Polyphenolic Grape Seeds Extract
Abstract
1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Grape Seed Extract (GSE)
2.3. Blood Donors
2.4. Study Protocol
2.5. Coagulation Assays
2.6. Thromboelastometry
2.7. Measurement of Human Platelet Reactivity
2.8. Estimation of Platelet VASP Phosphorylation
2.9. Statistical Analysis
3. Results
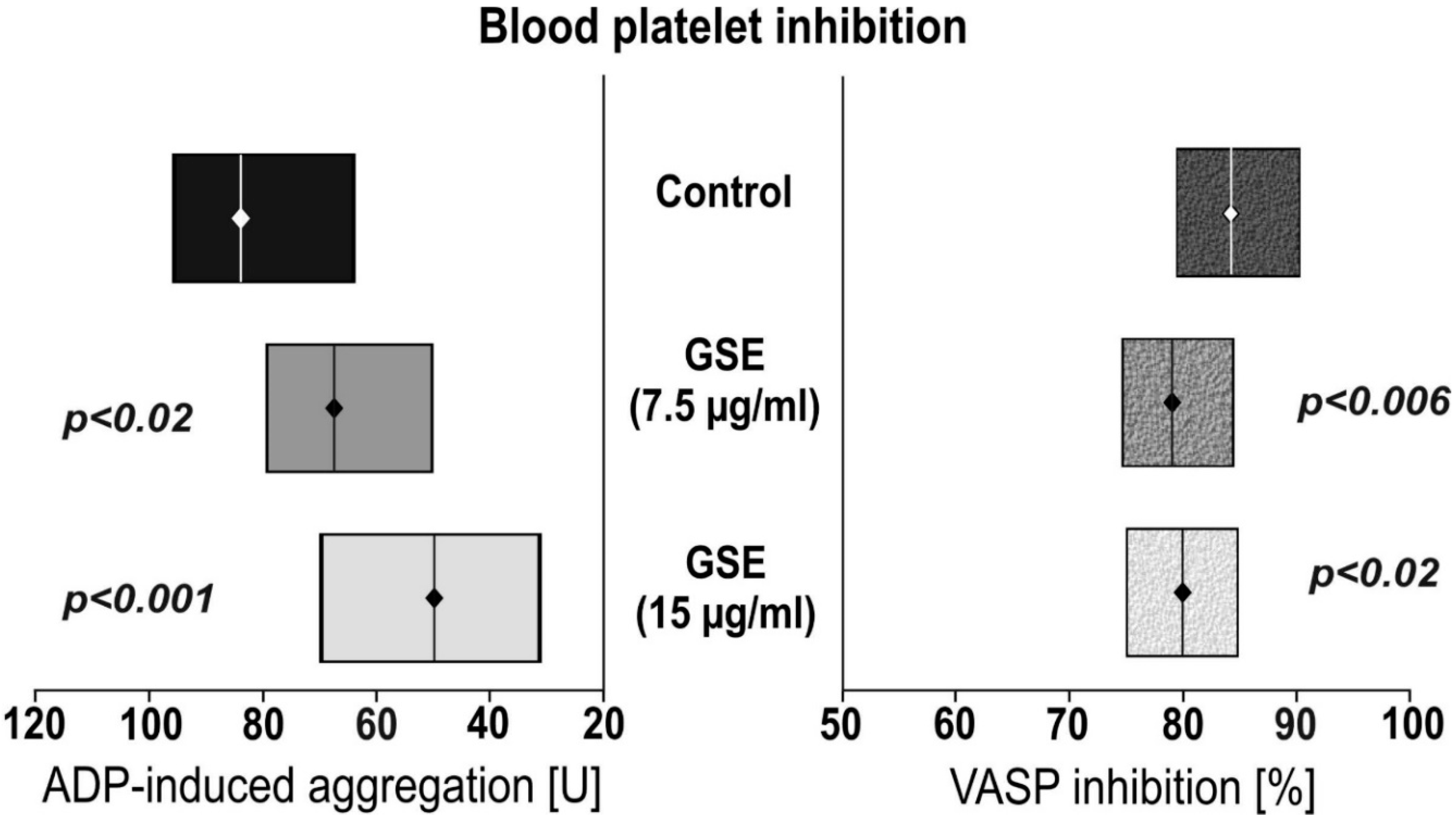
3.1. The Impact of GSE on Platelet Reactivity
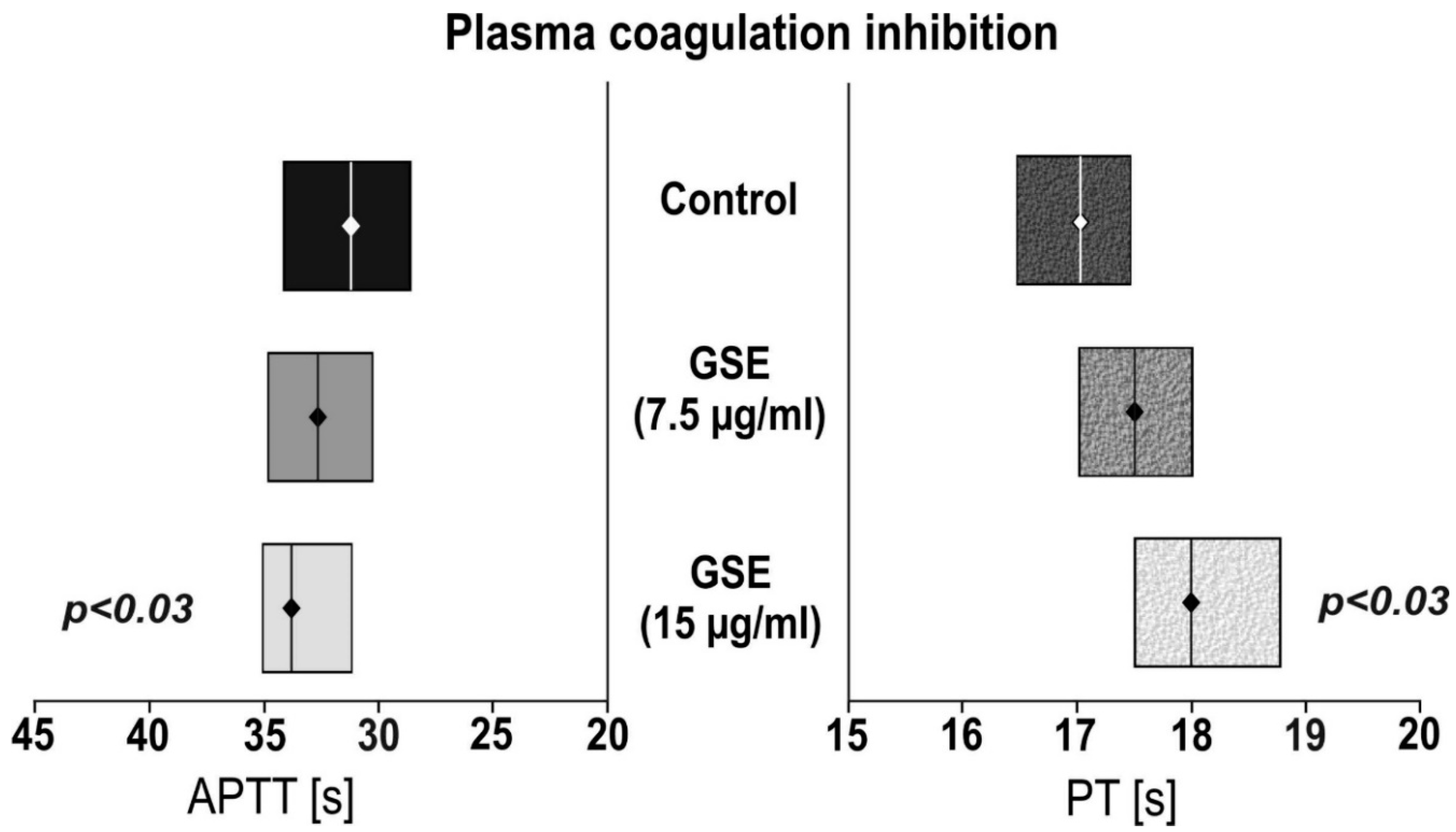
3.2. The Impact of GSE on Plasma Coagulation Tests
3.3. The Impact of GSE on Thromboelastometry Parameters
4. Discussion
Author Contributions
Funding
Conflicts of Interest
References
- Piepoli, M.F. 2016 European Guidelines on cardiovascular disease prevention in clinical practice: The Sixth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of 10 societies and by invited experts). Int. J. Behav. Med. 2017, 24, 321–419. [Google Scholar] [CrossRef]
- Spronk, H.M.H.; Padro, T.; Siland, J.E.; Prochaska, J.H.; Winters, J.; van der Wal, A.C.; Posthuma, J.J.; Lowe, G.; d’Alessandro, E.; Wenzel, P.; et al. Atherothrombosis and Thromboembolism: Position Paper from the Second Maastricht Consensus Conference on Thrombosis. Thromb. Haemost. 2018, 118, 229–250. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, N.; Jain, A.; Mahmoud, A.N.; Bishnoi, R.; Golwala, H.; Karimi, A.; Mojadidi, M.K.; Garg, J.; Gupta, T.; Patel, N.K.; et al. Safety and Efficacy of Dual Versus Triple Antithrombotic Therapy in Patients Undergoing Percutaneous Coronary Intervention. Am. J. Med. 2017, 130, 1280–1289. [Google Scholar] [CrossRef] [PubMed]
- Lam, D.H.; Bell, S.M.; Hira, R.S. Concomitant Use of Antiplatelets and Anticoagulants in Patients with Coronary Heart Disease and Atrial Fibrillation: What Do Recent Clinical Trials Teach Us? Curr. Atheroscler. Rep. 2018, 20, 4. [Google Scholar] [CrossRef] [PubMed]
- Valgimigli, M.; Bueno, H.; Byrne, R.A.; Collet, J.P.; Costa, F.; Jeppsson, A.; Jüni, P.; Kastrati, A.; Kolh, P.; Mauri, L.; et al. 2017 ESC focused update on dual antiplatelet therapy in coronary artery disease developed in collaboration with EACTS: The Task Force for dual antiplatelet therapy in coronary artery disease of the European Society of Cardiology (ESC) and of the European Association for Cardio-Thoracic Surgery (EACTS). Eur. Heart J. 2018, 39, 213–260. [Google Scholar] [CrossRef] [PubMed]
- Keihanian, F.; Saeidinia, A.; Bagheri, R.K.; Johnston, T.P.; Sahebkar, A. Curcumin, hemostasis, thrombosis and coagulation. J. Cell. Physiol. 2017. [Google Scholar] [CrossRef]
- Santhakumar, A.B.; Battino, M.; Alvarez-Suarez, J.M. Dietary polyphenols: Structures, bioavailability and protective effects against atherosclerosis. Food Chem. Toxicol. 2018, 113, 49–65. [Google Scholar] [CrossRef]
- Tomé-Carneiro, J.; Visioli, F. Polyphenol-based nutraceuticals for the prevention and treatment of cardiovascular disease: Review of human evidence. Phytomedicine 2016, 23, 1145–1174. [Google Scholar] [CrossRef]
- Buil-Cosiales, P.; Toledo, E.; Salas-Salvadó, J.; Zazpe, I.; Farràs, M.; Basterra-Gortari, F.J.; Diez-Espino, J.; Estruch, R.; Corella, D.; Ros, E.; et al. Association between dietary fibre intake and fruit, vegetable or whole-grain consumption and the risk of CVD: Results from the PREvención con DIeta MEDiterránea (PREDIMED) trial. Br. J. Nutr. 2016, 116, 534–546. [Google Scholar] [CrossRef]
- Noad, R.L.; Rooney, C.; McCall, D.; Young, I.S.; McCance, D.; McKinley, M.C.; Woodside, J.V.; McKeown, P.P. Beneficial effect of a polyphenol-rich diet on cardiovascular risk: A randomised control trial. Heart 2016, 102, 1371–1379. [Google Scholar] [CrossRef]
- Phang, M.; Lazarus, S.; Wood, L.G.; Garg, M. Diet and thrombosis risk: Nutrients for prevention of thrombotic disease. Semin. Thromb. Hemost. 2011, 37, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Bachmair, E.M.; Ostertag, L.M.; Zhang, X.; de Roos, B. Dietary manipulation of platelet function. Pharmacol. Ther. 2014, 144, 97–113. [Google Scholar] [CrossRef] [PubMed]
- Bertelli, A.A.; Das, D.K. Grapes, wines, resveratrol, and heart health. J. Cardiovasc. Pharmacol. 2009, 54, 468–476. [Google Scholar] [CrossRef] [PubMed]
- Liberale, L.; Bonaventura, A.; Montecucco, F.; Dallegri, F.; Carbone, F. Impact of Red Wine Consumption on Cardiovascular Health. Curr. Med. Chem. 2017. [Google Scholar] [CrossRef] [PubMed]
- Markoski, M.M.; Garavaglia, J.; Oliveira, A.; Olivaes, J.; Marcadenti, A. Molecular Properties of Red Wine Compounds and Cardiometabolic Benefits. Nutr. Metab. Insights 2016, 9, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Rasines-Perea, Z.; Teissedre, P.L. Grape Polyphenols’ Effects in Human Cardiovascular Diseases and Diabetes. Molecules 2017, 22, 68. [Google Scholar] [CrossRef] [PubMed]
- Nassiri-Asl, M.; Hosseinzadeh, H. Review of the Pharmacological Effects of Vitis vinifera (Grape) and its Bioactive Constituents: An Update. Phytother. Res. 2016, 30, 1392–1403. [Google Scholar] [CrossRef]
- Chedea, V.S.; Braicu, C.; Socaciu, C. Antioxidant/prooxidant activity of a polyphenolic grape seed extract. Food Chem. 2010, 121, 132–139. [Google Scholar] [CrossRef]
- Bozan, B.; Tosun, G.; Ozcan, D. Study of polyphenol content in the seeds of red grape (Vitis vinifera L.) varieties cultivated in Turkey and their antiradical activity. Food Chem. 2008, 109, 426–430. [Google Scholar] [CrossRef]
- Jara-Palacios, M.J.; Hernanz, D.; Escudero-Gilete, M.L.; Heredia, F.J. The Use of Grape Seed Byproducts Rich in Flavonoids to Improve the Antioxidant Potential of Red Wines. Molecules 2016, 21, 1526. [Google Scholar] [CrossRef]
- Tong, H.; Song, X.; Sun, X.; Sun, G.; Du, F. Immunomodulatory and antitumor activities of grape seed proanthocyanidins. J. Agric. Food Chem. 2011, 59, 11543–11547. [Google Scholar] [CrossRef] [PubMed]
- Skrzypczak-Jankun, E.; Jankun, J. Theaflavin digallate inactivates plasminogen activator inhibitor: Could tea help in Alzheimer’s disease and obesity? Int. J. Mol. Med. 2010, 26. [Google Scholar] [CrossRef]
- Correia-da-Silva, M.; Sousa, E.; Duarte, B.; Marques, F.; Carvalho, F.; Cunha-Ribeiro, L.M.; Pinto, M.M. Flavonoids with an oligopolysulfated moiety: A new class of anticoagulant agents. J. Med. Chem. 2011, 54, 95–106. [Google Scholar] [CrossRef] [PubMed]
- Bagge, A.; Schott, U.; Kander, T. Effects of naturopathic medicines on Multiplate and ROTEM: A prospective experimental pilot study in healthy volunteers. BMC Complement. Altern. Med. 2016, 16, 64. [Google Scholar] [CrossRef] [PubMed]
- Bijak, M.; Bobrowski, M.; Borowiecka, M.; Podsedek, A.; Golanski, J.; Nowak, P. Anticoagulant effect of polyphenols-rich extracts from black chokeberry and grape seeds. Fitoterapia 2011, 82, 811–817. [Google Scholar] [CrossRef] [PubMed]
- Bijak, M.; Gajak, A.; Nowak, P. Hemostatic and cellular functions of factor XIII. Pol. Merkur. Lekarski 2013, 34, 71–74. [Google Scholar]
- Luzak, B.; Kassassir, H.; Roj, E.; Stanczyk, L.; Watala, C.; Golanski, J. Xanthohumol from hop cones (Humulus lupulus L.) prevents ADP-induced platelet reactivity. Arch. Physiol. Biochem. 2017, 123, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Rywaniak, J.; Luzak, B.; Podsedek, A.; Dudzinska, D.; Rozalski, M.; Watala, C. Comparison of cytotoxic and anti-platelet activities of polyphenolic extracts from Arnica montana flowers and Juglans regia husks. Platelets 2015, 26, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Torres-Urrutia, C.; Guzman, L.; Schmeda-Hirschmann, G.; Moore-Carrasco, R.; Alarcon, M.; Astudillo, L.; Gutierrez, M.; Carrasco, G.; Yuri, J.A.; Aranda, E.; et al. Antiplatelet, anticoagulant, and fibrinolytic activity in vitro of extracts from selected fruits and vegetables. Blood Coagul. Fibrinolysis 2011, 22, 197–205. [Google Scholar] [CrossRef] [PubMed]
- Pawlaczyk, I.; Czerchawski, L.; Kuliczkowski, W.; Karolko, B.; Pilecki, W.; Witkiewicz, W.; Gancarz, R. Anticoagulant and anti-platelet activity of polyphenolic-polysaccharide preparation isolated from the medicinal plant Erigeron canadensis L. Thromb. Res. 2011, 127, 328–340. [Google Scholar] [CrossRef] [PubMed]
- Pawlaczyk, I.; Lewik-Tsirigotis, M.; Capek, P.; Matulova, M.; Sasinkova, V.; Dabrowski, P.; Witkiewicz, W.; Gancarz, R. Effects of extraction condition on structural features and anticoagulant activity of F. vesca L. conjugates. Carbohydr. Polym. 2013, 92, 741–750. [Google Scholar] [CrossRef]
- Ryu, R.; Jung, U.J.; Kim, H.J.; Lee, W.; Bae, J.S.; Park, Y.B.; Choi, M.S. Anticoagulant and Antiplatelet Activities of Artemisia princeps Pampanini and Its Bioactive Components. Prev. Nutr. Food Sci. 2013, 18, 181–187. [Google Scholar] [CrossRef]
- Felix-Silva, J.; Souza, T.; Camara, R.B.; Cabral, B.; Silva-Junior, A.A.; Rebecchi, I.M.; Zucolotto, S.M.; Rocha, H.A.; Fernandes-Pedrosa Mde, F. In vitro anticoagulant and antioxidant activities of Jatropha gossypiifolia L. (Euphorbiaceae) leaves aiming therapeutical applications. BMC Complement. Altern. Med. 2014, 14, 405. [Google Scholar] [CrossRef] [PubMed]
- Cordier, W.; Steenkamp, V. Herbal remedies affecting coagulation: A review. Pharm. Biol. 2012, 50, 443–452. [Google Scholar] [CrossRef] [PubMed]
- Pawlaczyk, I.; Tsirigotis-Maniecka, M.; Czerchawski, L.; Pilecki, W.; Saluk, J.; Wachowicz, B.; Bonarska-Kujawa, D.; Pyrkosz-Biardzka, K.; Kleszczynska, H.; Kuliczkowski, W.; et al. [The plant anticoagulants with perspective to use in treatment of thrombosis]. Przegl. Lek. 2013, 70, 157–161. [Google Scholar]
- Bijak, M.; Saluk, J.; Ponczek, M.B.; Nowak, P. Antithrombin effect of polyphenol-rich extracts from black chokeberry and grape seeds. Phytother. Res. 2013, 27, 71–76. [Google Scholar] [CrossRef]
- Bijak, M.; Kolodziejczyk-Czepas, J.; Ponczek, M.B.; Saluk, J.; Nowak, P. Protective effects of grape seed extract against oxidative and nitrative damage of plasma proteins. Int. J. Biol. Macromol. 2012, 51, 183–187. [Google Scholar] [CrossRef]
- Von Kugelgen, I. Structure, Pharmacology and Roles in Physiology of the P2Y12 Receptor. Adv. Exp. Med. Biol. 2017, 1051, 123–138. [Google Scholar] [CrossRef]
- Bagoly, Z.; Sarkady, F.; Magyar, T.; Kappelmayer, J.; Pongrácz, E.; Csiba, L.; Muszbek, L. Comparison of a new P2Y12 receptor specific platelet aggregation test with other laboratory methods in stroke patients on clopidogrel monotherapy. PLoS ONE 2013, 8, e69417. [Google Scholar] [CrossRef] [PubMed]
- Faggio, C.; Sureda, A.; Morabito, S.; Sanches-Silva, A.; Mocan, A.; Nabavi, S.F.; Nabavi, S.M. Flavonoids and platelet aggregation: A brief review. Eur. J. Pharmacol. 2017, 807, 91–101. [Google Scholar] [CrossRef]
- Offermanns, S. Activation of platelet function through G protein-coupled receptors. Circ. Res. 2006, 99, 1293–1304. [Google Scholar] [CrossRef] [PubMed]
- Kahner, B.N.; Shankar, H.; Murugappan, S.; Prasad, G.L.; Kunapuli, S.P. Nucleotide receptor signaling in platelets. J. Thromb. Haemost. 2006, 4, 2317–2326. [Google Scholar] [CrossRef] [PubMed]
- Boncler, M.; Golanski, J.; Lukasiak, M.; Redzynia, M.; Dastych, J.; Watala, C. A new approach for the assessment of the toxicity of polyphenol-rich compounds with the use of high content screening analysis. PLoS ONE 2017, 12, e0180022. [Google Scholar] [CrossRef]
- Williamson, G.; Manach, C. Bioavailability and bioefficacy of polyphenols in humans. II. Review of 93 intervention studies. Am. J. Clin. Nutr. 2005, 81, 243s–255s. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Scalbert, A.; Morand, C.; Remesy, C.; Jimenez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr. 2004, 79, 727–747. [Google Scholar] [CrossRef] [PubMed]
- Manach, C.; Williamson, G.; Morand, C.; Scalbert, A.; Remesy, C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am. J. Clin. Nutr. 2005, 81, 230S–242S. [Google Scholar] [CrossRef]
| Thromboelastometry Variable | Control | GSE 7.5 µg/mL | GSE 15.0 µg/mL |
|---|---|---|---|
| Clotting Time—CT (s) | 48.5 (43.3; 52.9) | 69.5 * (63.8; 76.0) | 77.1 * (69.3; 88.0) |
| Clotting Formation Time—CFT (s) | 94.3 (87.4; 108.9) | 103.5 (86.5; 131.8) | 120.5 # (98.3; 155.9) |
| Amplitude—A (mm) | 70.5 (58.1; 72.8) | 69.5 (65.0; 72.8) | 67.5 & (61.0; 70.0) |
| Maximum Clot Firmness—MCF (mm) | 58.5 (55.6; 59.9) | 58.0 (64.5; 61.0) | 55.0 @ (52.0; 60.0) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bijak, M.; Sut, A.; Kosiorek, A.; Saluk-Bijak, J.; Golanski, J. Dual Anticoagulant/Antiplatelet Activity of Polyphenolic Grape Seeds Extract. Nutrients 2019, 11, 93. https://doi.org/10.3390/nu11010093
Bijak M, Sut A, Kosiorek A, Saluk-Bijak J, Golanski J. Dual Anticoagulant/Antiplatelet Activity of Polyphenolic Grape Seeds Extract. Nutrients. 2019; 11(1):93. https://doi.org/10.3390/nu11010093
Chicago/Turabian StyleBijak, Michal, Agnieszka Sut, Anna Kosiorek, Joanna Saluk-Bijak, and Jacek Golanski. 2019. "Dual Anticoagulant/Antiplatelet Activity of Polyphenolic Grape Seeds Extract" Nutrients 11, no. 1: 93. https://doi.org/10.3390/nu11010093
APA StyleBijak, M., Sut, A., Kosiorek, A., Saluk-Bijak, J., & Golanski, J. (2019). Dual Anticoagulant/Antiplatelet Activity of Polyphenolic Grape Seeds Extract. Nutrients, 11(1), 93. https://doi.org/10.3390/nu11010093






