Stabilization of Vitamin D in Pea Protein Isolate Nanoemulsions Increases Its Bioefficacy in Rats
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Formation of Pea Protein Nanoaggregates
2.3. Preparation of Nanoemulsion
2.4. Animals
2.5. Blood Sampling and Biochemical Analysis
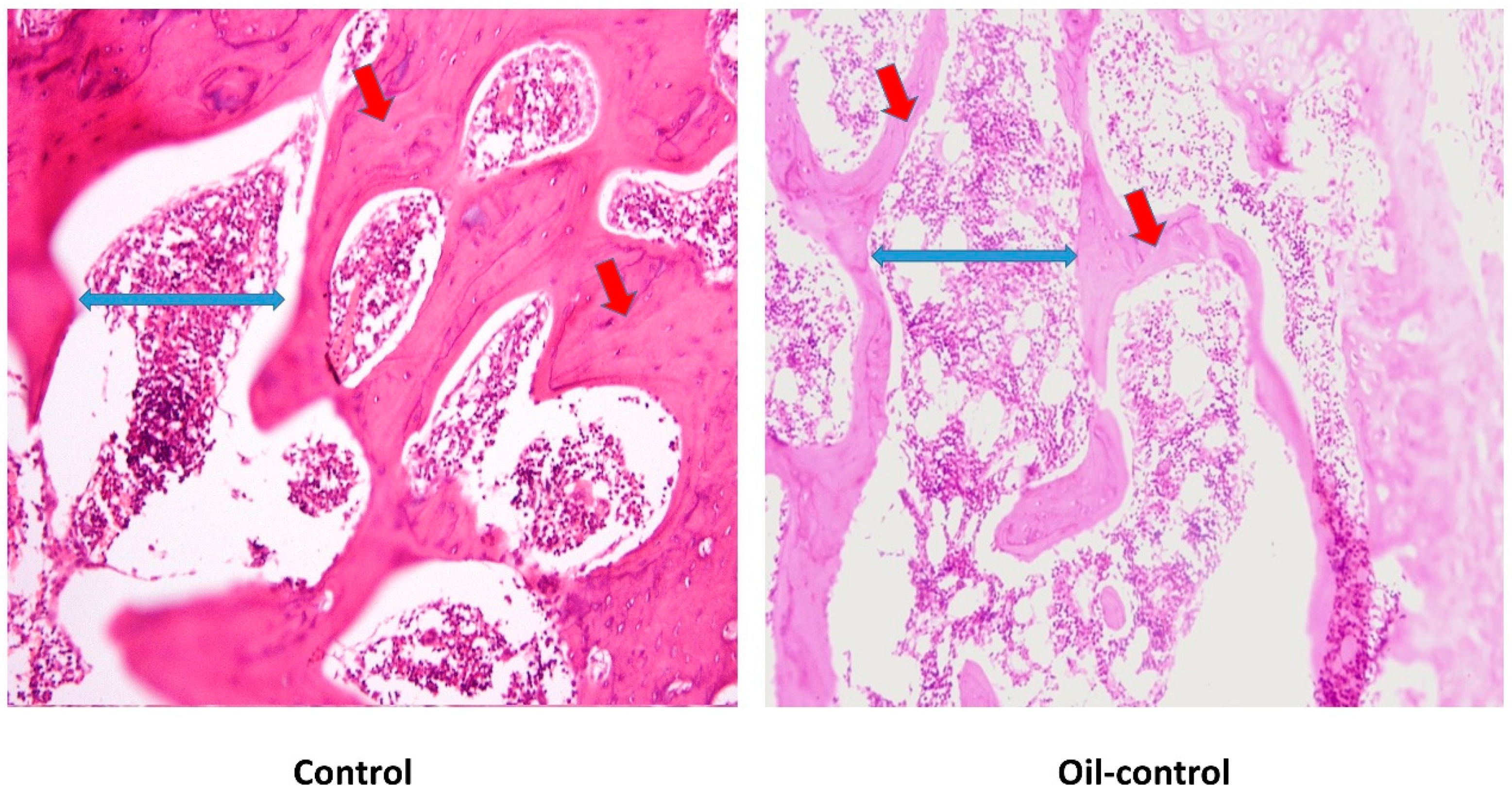
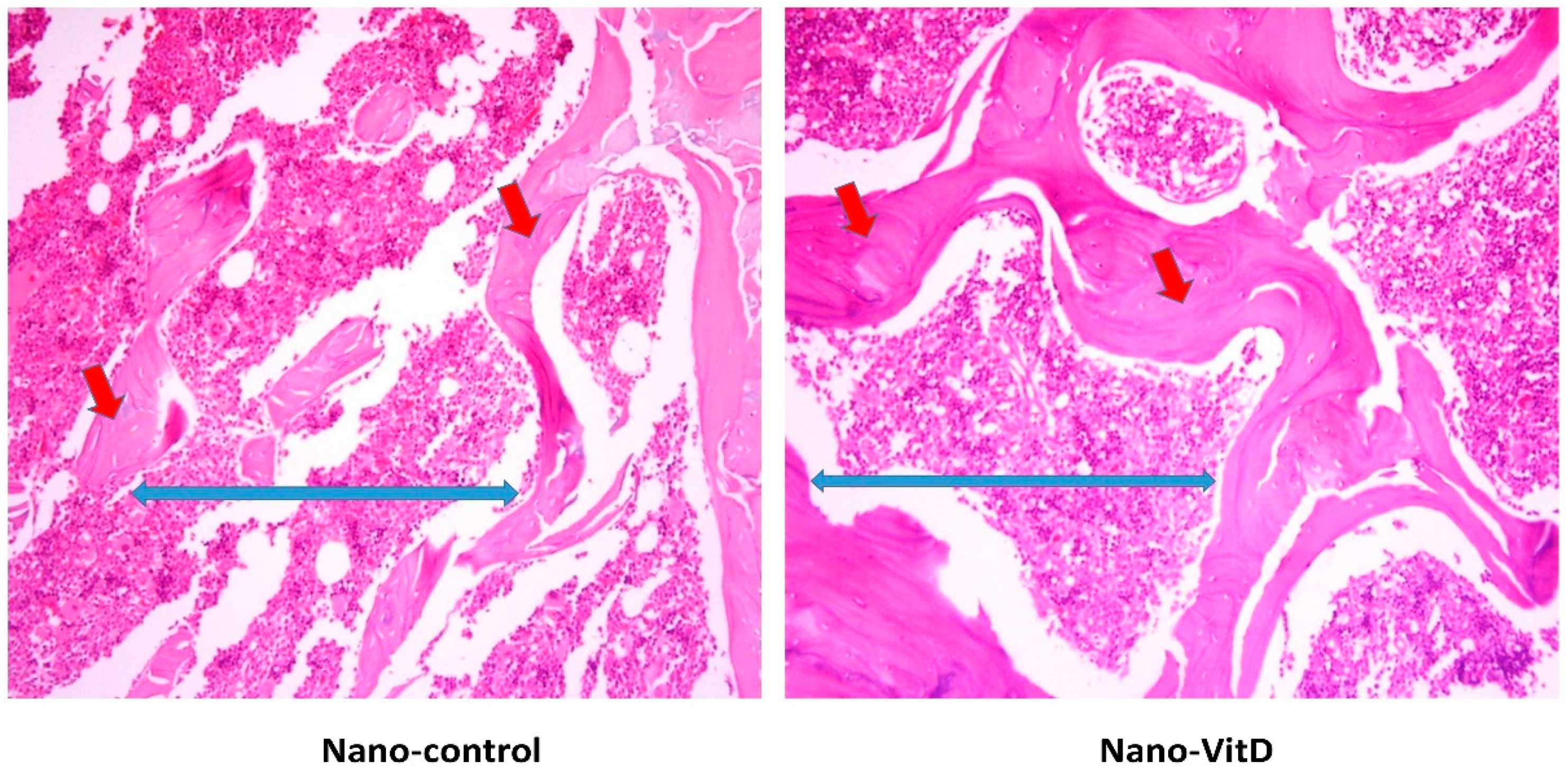
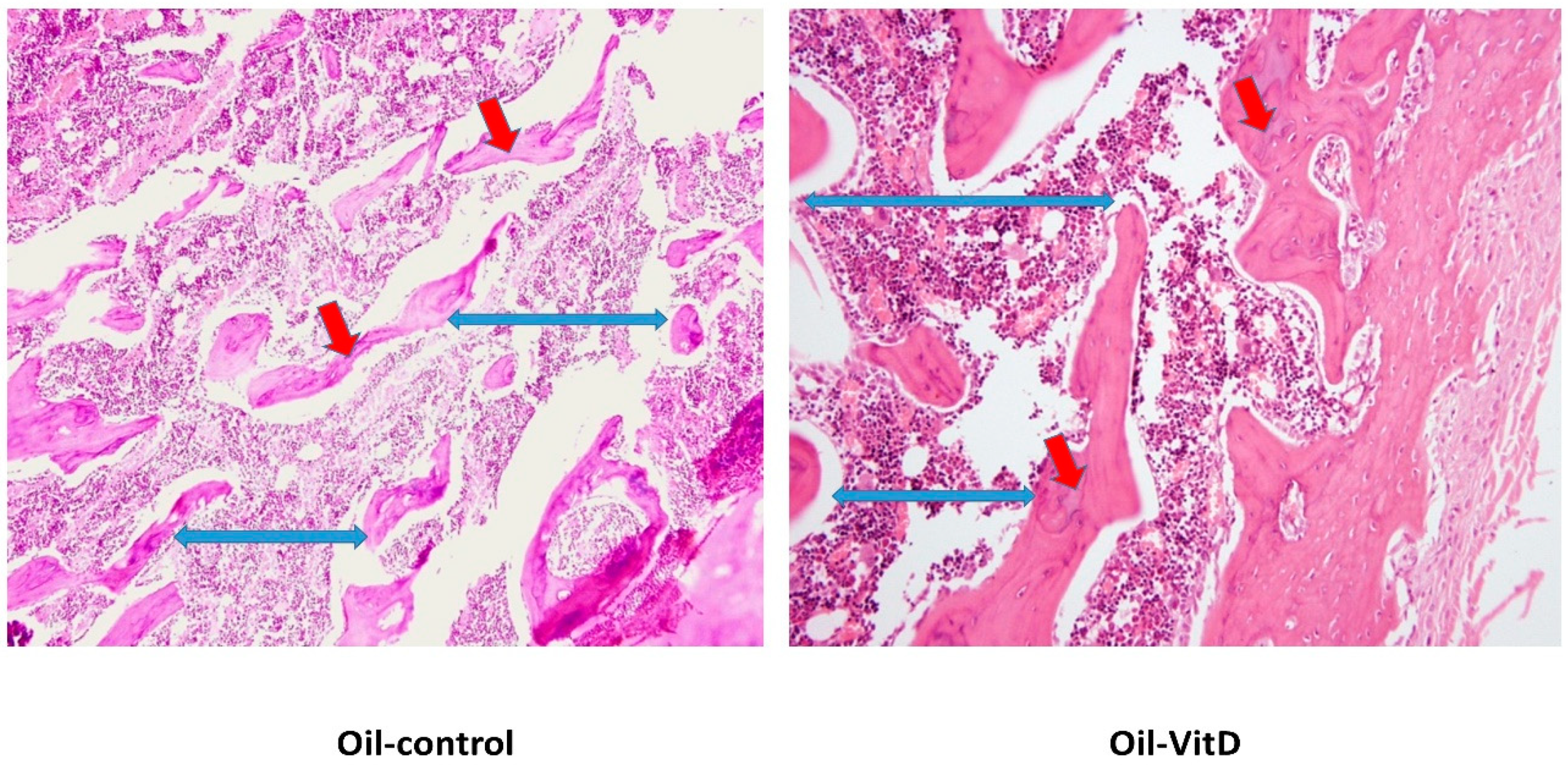
2.6. Histological and Histomorphometric Analysis
2.7. Statistical Analysis
3. Results
3.1. Changes of 25(OH)VitD and Biomarkers of VitD Deficiency Effects on Bone
3.2. Histological Changes after Treatment with VitD Deficient Diets and Therapies
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Roth, D.E.; Abrams, S.A.; Aloia, J.; Bergeron, G.; Bourassa, M.W.; Brown, K.H.; Calvo, M.S.; Cashman, K.D.; Combs, G.; De-Regil, L.M.; et al. Global prevalence and disease burden of vitamin D deficiency: A roadmap for action in low- and middle-income countries. Ann. N. Y. Acad. Sci. 2018, 1430, 44–79. [Google Scholar] [CrossRef] [PubMed]
- Mithal, A.; Wahl, D.A.; Bonjour, J.P.; Burckhardt, P.; Dawson-Hughes, B.; Eisman, J.A.; El-Hajj Fuleihan, G.; Josse, R.G.; Lips, P.; Morales-Torres, J. Global vitamin D status and determinants of hypovitaminosis D. Osteoporos. Int. 2009, 20, 1807–1820. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. High prevalence of vitamin D inadequacy and implications for health. Mayo Clin. Proc. 2006, 81, 353–373. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Chen, W.; Li, D.; Yin, X.; Zhang, X.; Olsen, N.; Zheng, S.G. Vitamin D and Chronic Diseases. Aging Dis. 2017, 8, 346–353. [Google Scholar] [CrossRef] [PubMed]
- Haines, S.T.; Park, S.K. Vitamin D supplementation: What’s known, what to do, and what’s needed. Pharmacotherapy 2012, 32, 354–382. [Google Scholar] [CrossRef] [PubMed]
- Cashman, K.D.; Kiely, M. Vitamin D and Food Fortification. In Vitamin D: Health, Disease and Therapeutics, 4th ed.; Academic Press: London, UK, 2018; Volume 2, pp. 109–127. ISBN 9780128099636. [Google Scholar]
- Park, S.J.; Garcia, C.V.; Shin, G.H.; Kim, J.T. Development of nanostructured lipid carriers for the encapsulation and controlled release of vitamin D3. Food Chem. 2017, 225, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Shu, G.; Khalid, N.; Zhao, Y.; Neves, M.A.; Kobayashi, I.; Nakajima, M. Formulation and stability assessment of ergocalciferol loaded oil-in-water nanoemulsions: Insights of emulsifiers effect on stabilization mechanism. Food Res. Int. 2016, 90, 320–327. [Google Scholar] [CrossRef]
- Ozturk, B.; Argin, S.; Ozilgen, M.; McClements, D.J. Nanoemulsion delivery systems for oil-soluble vitamins: Influence of carrier oil type on lipid digestion and vitamin D3 bioaccessibility. Food Chem. 2015, 187, 499–506. [Google Scholar] [CrossRef]
- Mohammadi, M.; Ghanbarzadeh, B.; Hamishehkar, H. Formulation of nanoliposomal vitamin D3 for potential application in beverage fortification. Adv. Pharm. Bull. 2014, 4, 569–575. [Google Scholar]
- Diarrassouba, F.; Garrait, G.; Remondetto, G.; Alvarez, P.; Beyssac, E.; Subirade, M. Food protein-based microspheres for increased uptake of Vitamin D3. Food Chem. 2015, 173, 1066–1072. [Google Scholar] [CrossRef]
- Hasanvand, E.; Fathi, M.; Bassiri, A. Production and characterization of vitamin D3-loaded starch nanoparticles: Effect of amylose to amylopectin ratio and sonication parameters. J. Food Sci. Technol. 2018, 55, 1314–1324. [Google Scholar] [CrossRef] [PubMed]
- Teng, Z.; Luo, Y.; Wang, Q. Carboxymethyl chitosan-soy protein complex nanoparticles for the encapsulation and controlled release of vitamin D3. Food Chem. 2013, 141, 524–532. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Sharma, U.S. Liposomes in drug delivery: Progress and limitations. Int. J. Pharm. 1997, 154, 123–140. [Google Scholar] [CrossRef]
- McClements, D.J. Encapsulation, protection, and release of hydrophilic active components: Potential and limitations of colloidal delivery systems. Adv. Colloid Interface Sci. 2015, 219, 27–53. [Google Scholar] [CrossRef] [PubMed]
- Jain, K.; Kumar Mehra, N.; Jain, N.K. Nanotechnology in drug delivery: Safety and toxicity issues. Curr. Pharm. Des. 2015, 21, 4252–4261. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Allen, J.C.; Swaisgood, H.E. Binding of Vitamin D and Cholesterol to β-Lactoglobulin. J. Dairy Sci. 1997, 80, 1054–1059. [Google Scholar] [CrossRef]
- Yang, M.C.; Guan, H.H.; Liu, M.Y.; Lin, Y.H.; Yang, J.M.; Chen, W.L.; Chen, C.J.; Mao, S.J.T. Crystal structure of a secondary vitamin D3-binding site of milk β-lactoglobulin. Proteins Struct. Funct. Genet. 2008, 71, 1197–1210. [Google Scholar] [CrossRef]
- Yang, M.C.; Guan, H.H.; Yang, J.M.; Ko, C.N.; Liu, M.Y.; Lin, Y.H.; Huang, Y.C.; Chen, C.J.; Mao, S.J.T. Rational design for crystallization of β-lactoglobulin and vitamin D3 complex: Revealing a secondary binding site. Cryst. Growth Des. 2008, 8, 4268–4276. [Google Scholar] [CrossRef]
- Kohl, E.A.; Schaefer, P.C. Improved high-pressure liquid chromatographic assay of serum 25-hydroxycholecalciferol and 25-hydroxyergocalctferol after reverse-phase sep-pak C18cartridge preparation of sample. J. Liq. Chromatogr. 1981, 4, 2023–2037. [Google Scholar] [CrossRef]
- Kao, P.C.; Heser, D.W. Simultaneous determination of 25-hydroxy- and 1,25-dihydroxyvitamin D from a single sample by dual-cartridge extraction. Clin. Chem. 1984, 30, 56–61. [Google Scholar]
- Foegeding, E.A.; Davis, J.P. Food protein functionality: A comprehensive approach. Food Hydrocoll. 2011, 25, 1853–1864. [Google Scholar] [CrossRef]
- Lam, R.S.H.; Nickerson, M.T. Food proteins: A review on their emulsifying properties using a structure-function approach. Food Chem. 2013, 141, 975–984. [Google Scholar] [CrossRef] [PubMed]
- Beverung, C.J.; Radke, C.J.; Blanch, H.W. Protein adsorption at the oil/water interface: Characterization of adsorption kinetics by dynamic interfacial tension measurements. Biophys. Chem. 1999, 81, 59–80. [Google Scholar] [CrossRef]
- Karaca, A.C.; Low, N.; Nickerson, M. Emulsifying properties of chickpea, faba bean, lentil and pea proteins produced by isoelectric precipitation and salt extraction. Food Res. Int. 2011, 44, 2742–2750. [Google Scholar] [CrossRef]
- Jiang, S.; Ding, J.; Andrade, J.; Rababah, T.M.; Almajwal, A.; Abulmeaty, M.M.; Feng, H. Modifying the physicochemical properties of pea protein by pH-shifting and ultrasound combined treatments. Ultrason. Sonochem. 2017, 38, 835–842. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Yildiz, G.; dos Santos, L.C.; Jiang, S.; Andrade, J.E.; Engeseth, N.J.; Feng, H. Soy protein nano-aggregates with improved functional properties prepared by sequential pH treatment and ultrasonication. Food Hydrocoll. 2016, 55, 200–209. [Google Scholar] [CrossRef]
- Fleet, J.C.; Gliniak, C.; Zhang, Z.; Xue, Y.; Smith, K.B. Serum metabolite profiles and target tissue gene expression define the effect of cholecalciferol intake on calcium metabolism in rats and mice. J. Nutr. 2008, 138, 1114–1120. [Google Scholar] [CrossRef] [PubMed]
- Reeves, P.G.; Nielsen, F.H.; Fahey, G.C. AIN-93 Purified Diets for Laboratory Rodents: Final Report of the American Institute of Nutrition Ad Hoc Writing Committee on the Reformulation of the AIN-76A Rodent Diet. J. Nutr. 1993, 123, 1939–1951. [Google Scholar] [CrossRef]
- Itakura, C.; Yamasaki, K.; Goto, M. Pathology of experimental vitamin D deficiency rickets in growing chickens. II. Parathyroid gland. Avian Pathol. 1978, 7, 515–532. [Google Scholar] [CrossRef]
- Egan, K.P.; Brennan, T.A.; Pignolo, R.J. Bone histomorphometry using free and commonly available software. Histopathology 2012, 61, 1168–1173. [Google Scholar] [CrossRef]
- Holick, M.F. The vitamin D deficiency pandemic: Approaches for diagnosis, treatment and prevention. Rev. Endocr. Metab. Disord. 2017, 18, 153–165. [Google Scholar] [CrossRef] [PubMed]
- Salvia-Trujillo, L.; Martín-Belloso, O.; McClements, D. Excipient Nanoemulsions for Improving Oral Bioavailability of Bioactives. Nanomaterials 2016, 6, 17. [Google Scholar] [CrossRef] [PubMed]
- Kadappan, A.S.; Guo, C.; Gumus, C.E.; Bessey, A.; Wood, R.J.; McClements, D.J.; Liu, Z. The Efficacy of Nanoemulsion-Based Delivery to Improve Vitamin D Absorption: Comparison of In Vitro and In Vivo Studies. Mol. Nutr. Food Res. 2018, 62, 1700836. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.-J.; Kwon, M.; Kim, T.-H.; Kim, K.; Jeong, S.-H.; Chang, H.-R. Pharmacokinetic Characterization of Nano-emulsion Vitamin A, D and E (LaVita) in Rats. Korean J. Environ. Agric. 2011, 30, 196–201. [Google Scholar] [CrossRef]
- Diarrassouba, F.; Garrait, G.; Remondetto, G.; Alvarez, P.; Beyssac, E.; Subirade, M. Improved bioavailability of vitamin D3 using a β-lactoglobulin-based coagulum. Food Chem. 2015, 172, 361–367. [Google Scholar] [CrossRef] [PubMed]
- European Chemical Agency. Regulation (EU) No 528/2012 Concerning the Making Available on the Market and Use of biocidal Products. Assessment Report: Cholecalciferol, PT 14 (Rodenticides). 2018. Available online: https://echa.europa.eu/documents/10162/652777b2-d738-a2e8-7b1d-431fd61ba82f (accessed on 28 December 2018).
- Toromanoff, A.; Ammann, P.; Mosekilde, L.; Thomsen, J.S.; Riond, J.L. Parathyroid hormone increases bone formation and improves mineral balance in vitamin D-deficient female rats. Endocrinology 1997, 138, 2449–2457. [Google Scholar] [CrossRef] [PubMed]
- Khundmiri, S.J.; Murray, R.D.; Lederer, E. PTH and Vitamin D. Compr. Physiol. 2016, 6, 561–601. [Google Scholar] [PubMed]
- Lips, P.; Van Schoor, N.M. The effect of vitamin D on bone and osteoporosis. Best Pract. Res. Clin. Endocrinol. Metab. 2011, 25, 585–591. [Google Scholar] [CrossRef]
- Millán, J.L. The role of phosphatases in the initiation of skeletal mineralization. Calcif. Tissue Int. 2013, 93, 299–306. [Google Scholar] [CrossRef]
- Chuang, L.H.; Tung, Y.C.; Liu, S.Y.; Lee, C.T.; Chen, H.L.; Tsai, W.Y. Nutritional rickets in Taiwanese children: Experiences at a single center. J. Formos. Med. Assoc. 2018, 117, 583–587. [Google Scholar] [CrossRef]
- Bhambri, R.; Naik, V.; Malhotra, N.; Taneja, S.; Rastogi, S.; Ravishanker, U.; Mithal, A. Changes in Bone Mineral Density Following Treatment of Osteomalacia. J. Clin. Densitom. 2006, 9, 120–127. [Google Scholar] [CrossRef] [PubMed]
- Shaheen, S.; Noor, S.S.; Barakzai, Q. Serum alkaline phosphatase screening for vitamin D deficiency states. J. Coll. Phys. Surg. Pak. 2012, 22, 424–427. [Google Scholar]
- Uchida, H.; Kurata, Y.; Hiratsuka, H.; Umemura, T. The effects of a vitamin D—Deficient diet on chronic cadmium exposure in rats. Toxicol. Pathol. 2010, 38, 730–737. [Google Scholar] [CrossRef] [PubMed]
- DeLuca, H.F. Overview of general physiologic features and functions of vitamin D. Am. J. Clin. Nutr. 2004, 80, 1689S–1696S. [Google Scholar] [CrossRef] [PubMed]
- Lips, P. Vitamin D physiology. Prog. Biophys. Mol. Biol. 2006, 92, 4–8. [Google Scholar] [CrossRef] [PubMed]
- Need, A.G.; O’Loughlin, P.D.; Morris, H.A.; Coates, P.S.; Horowitz, M.; Nordin, B.E.C. Vitamin D metabolites and calcium absorption in severe vitamin D deficiency. J. Bone Miner. Res. 2008, 23, 1859–1863. [Google Scholar] [CrossRef] [PubMed]
- Brautbar, N.; Walling, M.W.; Coburn, J.W. Interactions between vitamin D deficiency and phosphorus depletion in the rat. J. Clin. Investig. 1979, 63, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Coburn, J.W.; Massry, S.G. Changes in serum and urinary calcium during phosphate depletion: Studies on mechanisms. J. Clin. Investig. 1970, 49, 1073–1087. [Google Scholar] [CrossRef] [PubMed]
- Clark, I.; Rivera-Cordero, F. Effects of endogenous parathyroid hormone on calcium, magnesium and phosphate metabolism in rats. II. Alterations in dietary phosphate. Endocrinology 1974, 95, 360–369. [Google Scholar] [CrossRef]
- Steele, T.H. Renal resistance to parathyroid hormone during phosphorus deprivation. J. Clin. Investig. 1976, 58, 1461–1464. [Google Scholar] [CrossRef]
- Coburn, K.R. Preliminary investigation of bone change as a result of exposure to reduced atmospheric pressure. Aerosp. Med. 1970, 41, 188–190. [Google Scholar] [PubMed]
- Rader, J.I.; Howard, G.A.; Feist, E.; Turner, R.T.; Baylink, D.J. Bone mineralization and metabolism of3H-25-hydroxyvitamin D3 in thyroparathyroidectomized rats treated with parathyroid extract. Calcif. Tissue Int. 1979, 29, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Khaw, K.; Sneyd, M.; Compston, J. Bone density, parathyroid hormone and 25-hydroxyvitaminD concentrations in middle-aged women. Br. Med. J. 1992, 305, 273–277. [Google Scholar] [CrossRef]
- Bonifácio, B.V.; da Silva, P.B.; Aparecido dos Santos Ramos, M.; Maria Silveira Negri, K.; Maria Bauab, T.; Chorilli, M. Nanotechnology-based drug delivery systems and herbal medicines: A review. Int. J. Nanomedicine 2013, 9, 1–15. [Google Scholar] [PubMed]
- Silva, A.C.; Santos, D.; Ferreira, D.; Lopes, C.M. Lipid-Based Nanocarriers as an Alternative for Oral Delivery of Poorly Water-Soluble Drugs: Peroral and Mucosal Routes. Curr. Med. Chem. 2012, 4495–4510. [Google Scholar] [CrossRef]
- McClements, D.J. Edible lipid nanoparticles: Digestion, absorption, and potential toxicity. Prog. Lipid Res. 2013, 52, 409–423. [Google Scholar] [CrossRef] [PubMed]
- Yen, C.C.; Chen, Y.C.; Wu, M.T.; Wang, C.C.; Wu, Y.T. Nanoemulsion as a strategy for improving the oral bioavailability and anti-inflammatory activity of andrographolide. Int. J. Nanomedicine 2018, 13, 669–680. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Wan, K.; Hu, X.; Zhang, Y.; Yan, Z.; Feng, J.; Zhang, J. Functional nanoemulsion-hybrid lipid nanocarriers enhance the bioavailability and anti-cancer activity of lipophilic diferuloylmethane. Nanotechnology 2016, 27, 085102. [Google Scholar] [CrossRef]
- Cheong, A.M.; Tan, C.P.; Nyam, K.L. Effect of Emulsification Method and Particle Size on the Rate of in vivo Oral Bioavailability of Kenaf (Hibiscus cannabinus L.) Seed Oil. J. Food Sci. 2018, 83, 1964–1969. [Google Scholar] [CrossRef]
- Saratale, R.G.; Lee, H.-S.; Koo, Y.E.; Saratale, G.D.; Kim, Y.J.; Imm, J.Y.; Park, Y. Absorption kinetics of vitamin E nanoemulsion and green tea microstructures by intestinal in situ single perfusion technique in rats. Food Res. Int. 2018, 106, 149–155. [Google Scholar] [CrossRef]
- Belhaj, N.; Dupuis, F.; Arab-Tehrany, E.; Denis, F.M.; Paris, C.; Lartaud, I.; Linder, M. Formulation, characterization and pharmacokinetic studies of coenzyme Q10PUFA’s nanoemulsions. Eur. J. Pharm. Sci. 2012, 47, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Kralova, I.; Sjöblom, J. Surfactants used in food industry: A review. J. Dispers. Sci. Technol. 2009, 30, 1363–1383. [Google Scholar] [CrossRef]
- McClements, D.J.; Rao, J. Food-Grade nanoemulsions: Formulation, fabrication, properties, performance, Biological fate, and Potential Toxicity. Crit. Rev. Food Sci. Nutr. 2011, 51, 285–330. [Google Scholar] [CrossRef] [PubMed]
- Yerramilli, M.; Longmore, N.; Ghosh, S. Stability and Bioavailability of Curcumin in Mixed Sodium Caseinate and Pea Protein Isolate Nanoemulsions. J. Am. Oil Chem. Soc. 2018, 95, 1013–1026. [Google Scholar] [CrossRef]
- Malekzad, H.; Mirshekari, H.; Sahandi Zangabad, P.; Moosavi Basri, S.M.; Baniasadi, F.; Sharifi Aghdam, M.; Karimi, M.; Hamblin, M.R. Plant protein-based hydrophobic fine and ultrafine carrier particles in drug delivery systems. Crit. Rev. Biotechnol. 2018, 38, 47–67. [Google Scholar] [CrossRef]
- Glerup, H. Vitamin D deficiency among immigrants. Ugeskr. Laeger 2000, 162, 6196–6199. [Google Scholar]
- Kauppinen-Mäkelin, R.; Tähtelä, R.; Löyttyniemi, E.; Kärkkäinen, J.; Välimäki, M.J. A high prevalence of hypovitaminosis D in Finnish medical in- and outpatients. J. Intern. Med. 2001, 249, 559–563. [Google Scholar] [CrossRef]
- Thomas, M.K.; Lloyd-Jones, D.M.; Thadhani, R.I.; Shaw, A.C.; Deraska, D.J.; Kitch, B.T.; Vamvakas, E.C.; Dick, I.M.; Prince, R.L.; Finkelstein, J.S. Hypovitaminosis D in Medical Inpatients. N. Engl. J. Med. 1998, 338, 777–783. [Google Scholar] [CrossRef]
- Marshall, I.; Mehta, R.; Ayers, C.; Dhumal, S.; Petrova, A. Prevalence and risk factors for vitamin D insufficiency and deficiency at birth and associated outcome. BMC Pediatr. 2016, 16, 208. [Google Scholar] [CrossRef]
- Kift, R.; Berry, J.L.; Vail, A.; Durkin, M.T.; Rhodes, L.E.; Webb, A.R. Lifestyle factors including less cutaneous sun exposure contribute to starkly lower vitamin D levels in U.K. South Asians compared with the white population. Br. J. Dermatol. 2013, 169, 1272–1278. [Google Scholar] [CrossRef]
- Clemens, T.L.; Henderson, S.L.; Adams, J.S.; Holick, M.F. Incerased skin pigment reduces the capacity of skin to synthesise vitamin D3. Lancet 1982, 319, 74–76. [Google Scholar] [CrossRef]
- Al Jurayyan, N.A.; Mohamed, S.; Al Issa, S.D.; Al Jurayyan, A.N. Rickets and osteomalacia in Saudi children and adolescents attending endocrine clinic, Riyadh, Saudi Arabia. Sudan. J. Paediatr. 2012, 12, 56–63. [Google Scholar] [PubMed]
- Gonnet, M.; Lethuaut, L.; Boury, F. New trends in encapsulation of liposoluble vitamins. J. Control. Release 2010, 146, 276–290. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Calcium and vitamin D. Diagnostics and therapeutics. Clin. Lab. Med. 2000, 20, 569–590. [Google Scholar] [CrossRef]
- Murphy, S.C.; Whited, L.J.; Rosenberry, L.C.; Hammond, B.H.; Bandler, D.K.; Boor, K.J. Fluid milk vitamin fortification compliance in New York State. J. Dairy Sci. 2001, 84, 2813–2820. [Google Scholar] [CrossRef]
- Marshall, E.F. Cholecalciferol: A unique toxicant for rodent control. In Proceedings of the Eleventh Vertebrate Pest Conference, Sacramento, CA, USA, 6–8 March 1984. [Google Scholar]
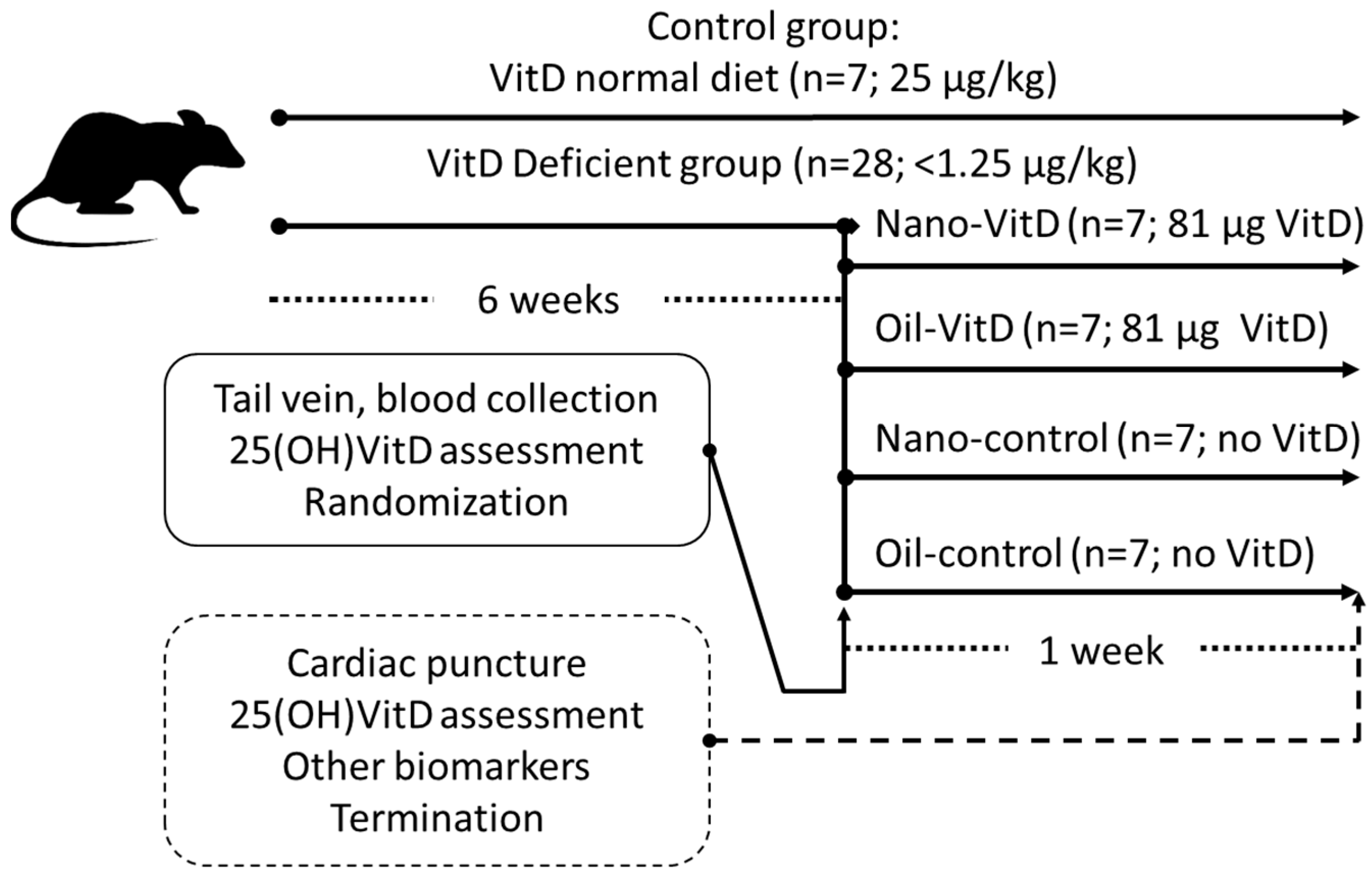
| Body Weight Changes (g) | 25 (OH)VitD (nmol/L) | ||||
|---|---|---|---|---|---|
| Groups 1 | Dietary VitD Intake (IU/kg/day) 1 | Before | After | Before | After |
| Control | 57.78 ± 6.49 a | 247.76 ± 29.47 | 262.28 ± 36.98 * | 31.68 ± 10.40 a | 36.84 ± 9.16 a |
| Nano-control | 1.32 ± 0.11 b | 258.12 ± 21.90 | 270.45 ± 31.72 | 15.38 ± 5.51 b | 15.88 ± 5.77 b |
| Oil-control | 1.30 ± 0.35 b | 225.86 ± 21.56 | 249.48 ± 26.96 | 18.26 ± 6.38 b | 15.59 ± 2.45 b |
| Nano-VitD | 1.46 ± 0.18 b | 239.06 ± 26.10 | 253.11 ± 26.64 | 14.65 ± 1.29 b | 34.37 ± 7.00 b,* |
| Oil-VitD | 1.46 ± 0.12 b | 256.24 ± 25.09 | 266.13 ± 27.53 | 14.33 ± 3.43 b | 14.05 ± 3.08 a |
| Groups 1 | PTH 2 (pg/mL) | Ca (mg/dL) | P (mg/dL) | ALP (U/L) |
|---|---|---|---|---|
| Control | 23.36 ± 12.00 a | 10.24 ± 0.92 a | 3.67 ± 1.13 a | 58.5 ± 11.5 a |
| Nano-control | 37.54 ± 6.61 a | 7.12 ± 1.16 b | 1.38 ± 0.57 b | 196.2 ± 57.7 b |
| Oil-control | 78.93 ± 8.31 b | 6.68 ± 1.92 b | 1.17 ± 0.62 b | 171.0 ± 17.6 b |
| Nano-VitD | 25.22 ± 14.26 a | 9.64 ± 0.60 a | 3.65 ± 0.71 a | 72.4 ± 31.0 a |
| Oil-VitD | 86.05 ± 9.67 b | 5.32 ± 1.28 b | 1.33 ± 0.32 b | 182.6 ± 61.8 b |
| Groups 1 | Osteoid Area (mm2) | Trabecular Separation (mm2) |
|---|---|---|
| Control | 7.67 ± 0.91 a | 1.18 ± 0.51a |
| Nano-control | 4.42 ± 1.49 b | 2.31 ± 1.11 b |
| Oil-control | 4.13 ± 1.06 b | 0.74 ± 0.48 a |
| Nano-VitD | 6.36 ± 1.16 a | 2.55 ± 1.39 b |
| Oil-VitD | 4.41 ± 1.09 b | 2.13 ± 1.18 b |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Almajwal, A.M.; Abulmeaty, M.M.A.; Feng, H.; Alruwaili, N.W.; Dominguez-Uscanga, A.; Andrade, J.E.; Razak, S.; ElSadek, M.F. Stabilization of Vitamin D in Pea Protein Isolate Nanoemulsions Increases Its Bioefficacy in Rats. Nutrients 2019, 11, 75. https://doi.org/10.3390/nu11010075
Almajwal AM, Abulmeaty MMA, Feng H, Alruwaili NW, Dominguez-Uscanga A, Andrade JE, Razak S, ElSadek MF. Stabilization of Vitamin D in Pea Protein Isolate Nanoemulsions Increases Its Bioefficacy in Rats. Nutrients. 2019; 11(1):75. https://doi.org/10.3390/nu11010075
Chicago/Turabian StyleAlmajwal, Ali M., Mahmoud M. A. Abulmeaty, Hao Feng, Nawaf W. Alruwaili, Astrid Dominguez-Uscanga, Juan E. Andrade, Suhail Razak, and Mohamed F. ElSadek. 2019. "Stabilization of Vitamin D in Pea Protein Isolate Nanoemulsions Increases Its Bioefficacy in Rats" Nutrients 11, no. 1: 75. https://doi.org/10.3390/nu11010075
APA StyleAlmajwal, A. M., Abulmeaty, M. M. A., Feng, H., Alruwaili, N. W., Dominguez-Uscanga, A., Andrade, J. E., Razak, S., & ElSadek, M. F. (2019). Stabilization of Vitamin D in Pea Protein Isolate Nanoemulsions Increases Its Bioefficacy in Rats. Nutrients, 11(1), 75. https://doi.org/10.3390/nu11010075






