Satiating Effect of High Protein Diets on Resistance-Trained Individuals in Energy Deficit
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Pre-Intervention Measures
2.3. Dietary Assessment
2.4. Experimental Design and Intervention Measures
2.5. Laboratory Measures
2.6. Test Meal and Satiety Measures
2.7. Post-Laboratory Measures and Support
2.8. Biochemical Analyses
2.9. Statistical Analyses
3. Results
3.1. Nutrition Intake and Body Composition Data
3.2. Dietary Intervention Perceived Satiety Responses
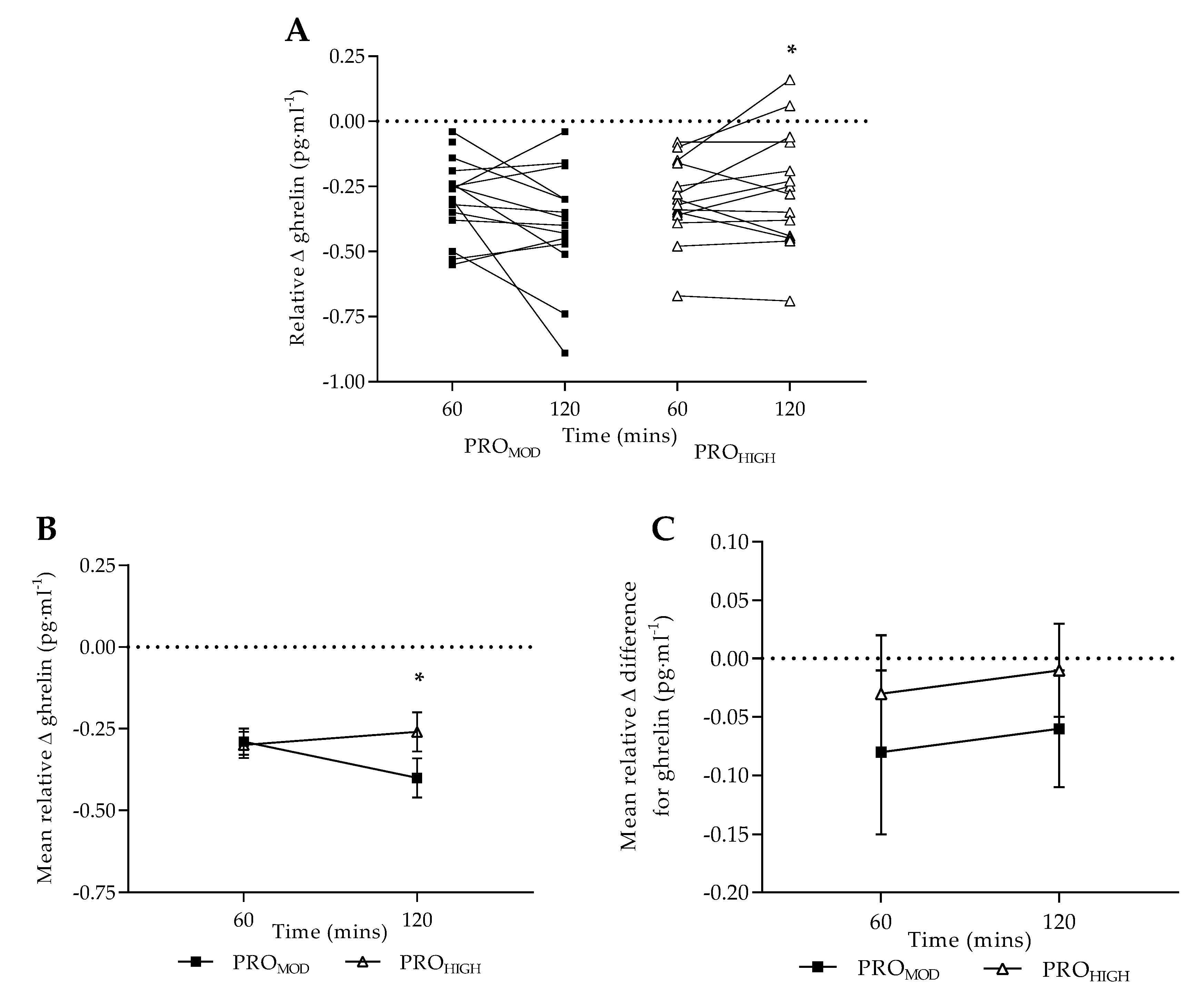
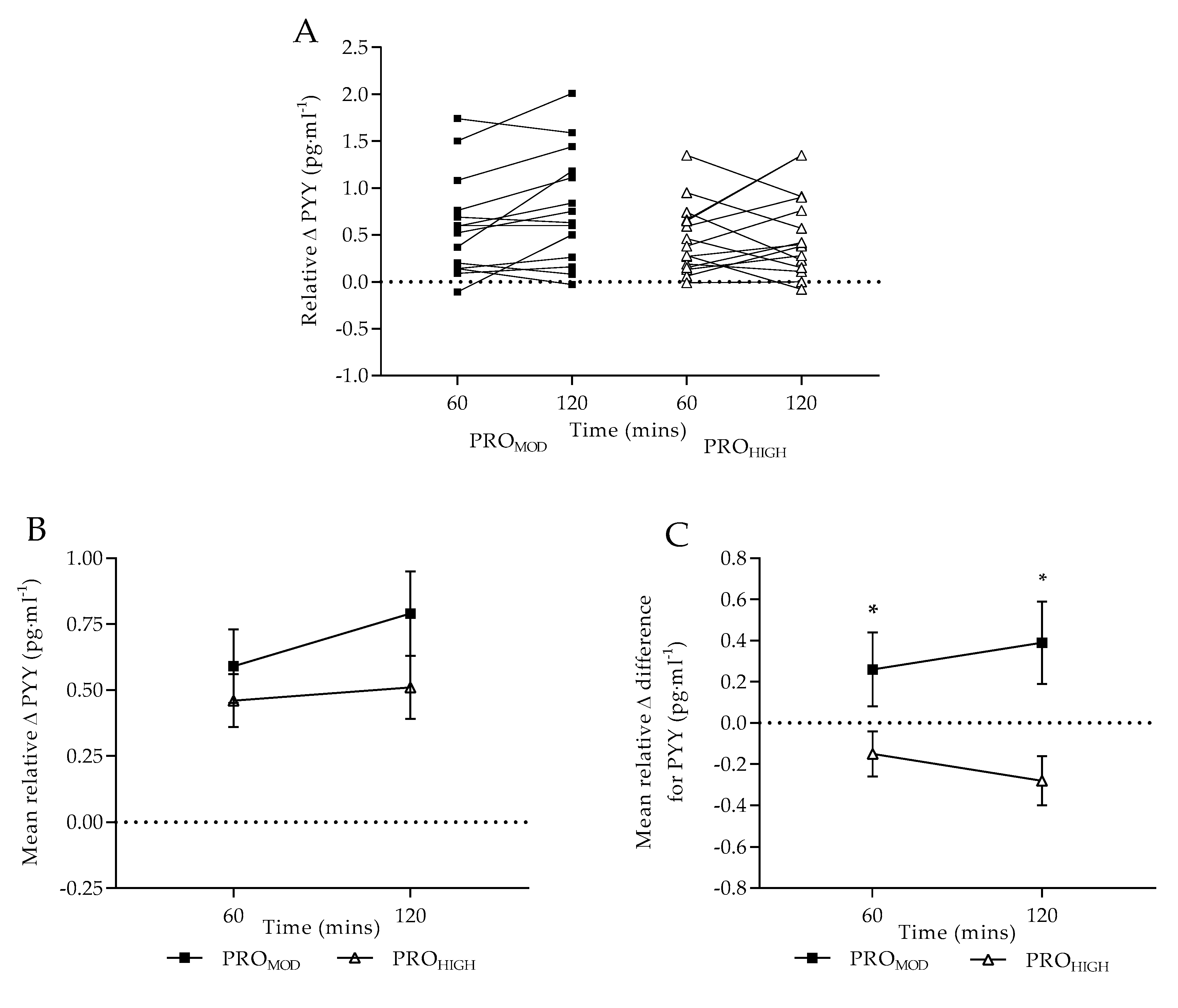
3.3. Test-Meal Satiety Hormone Responses
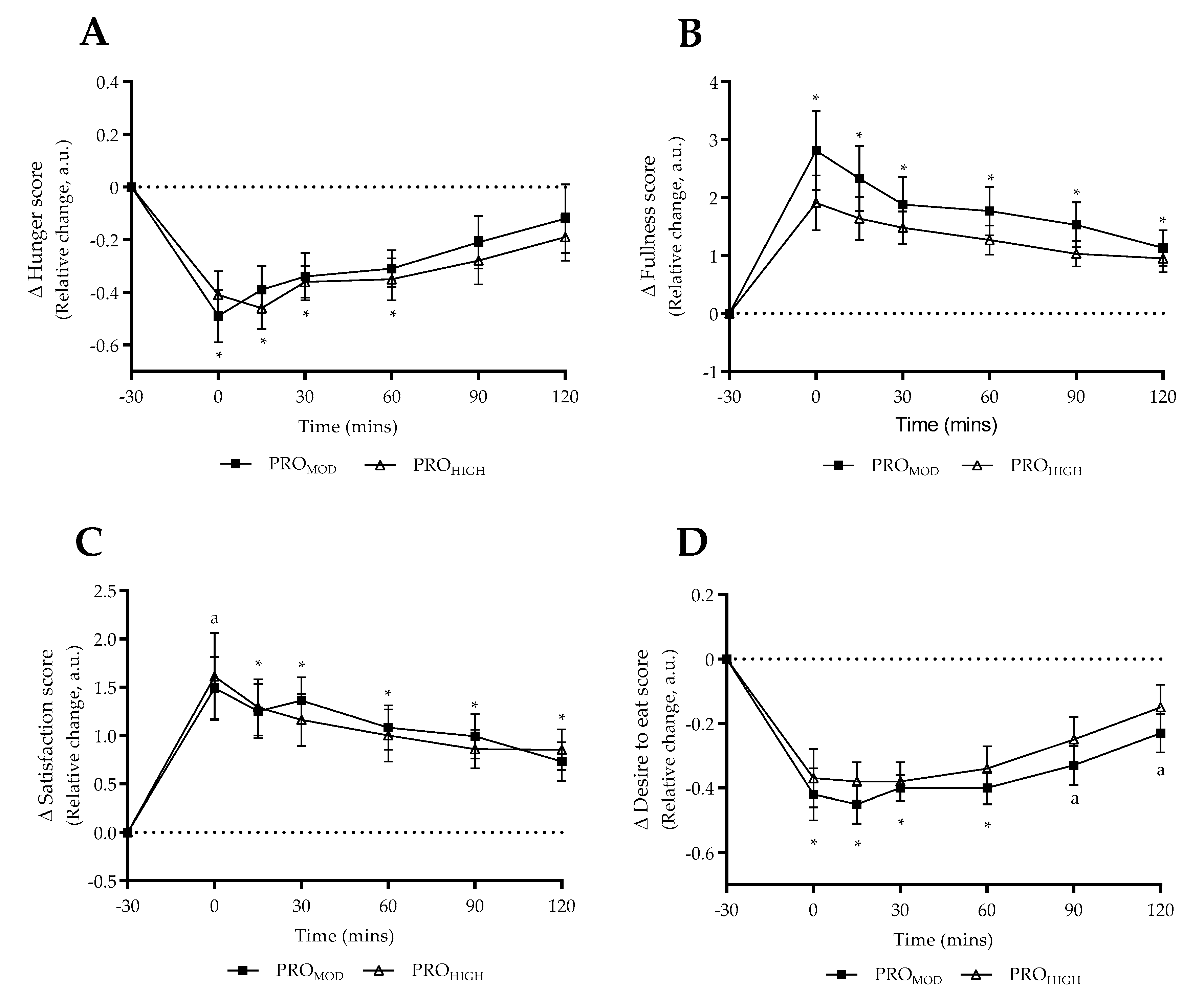
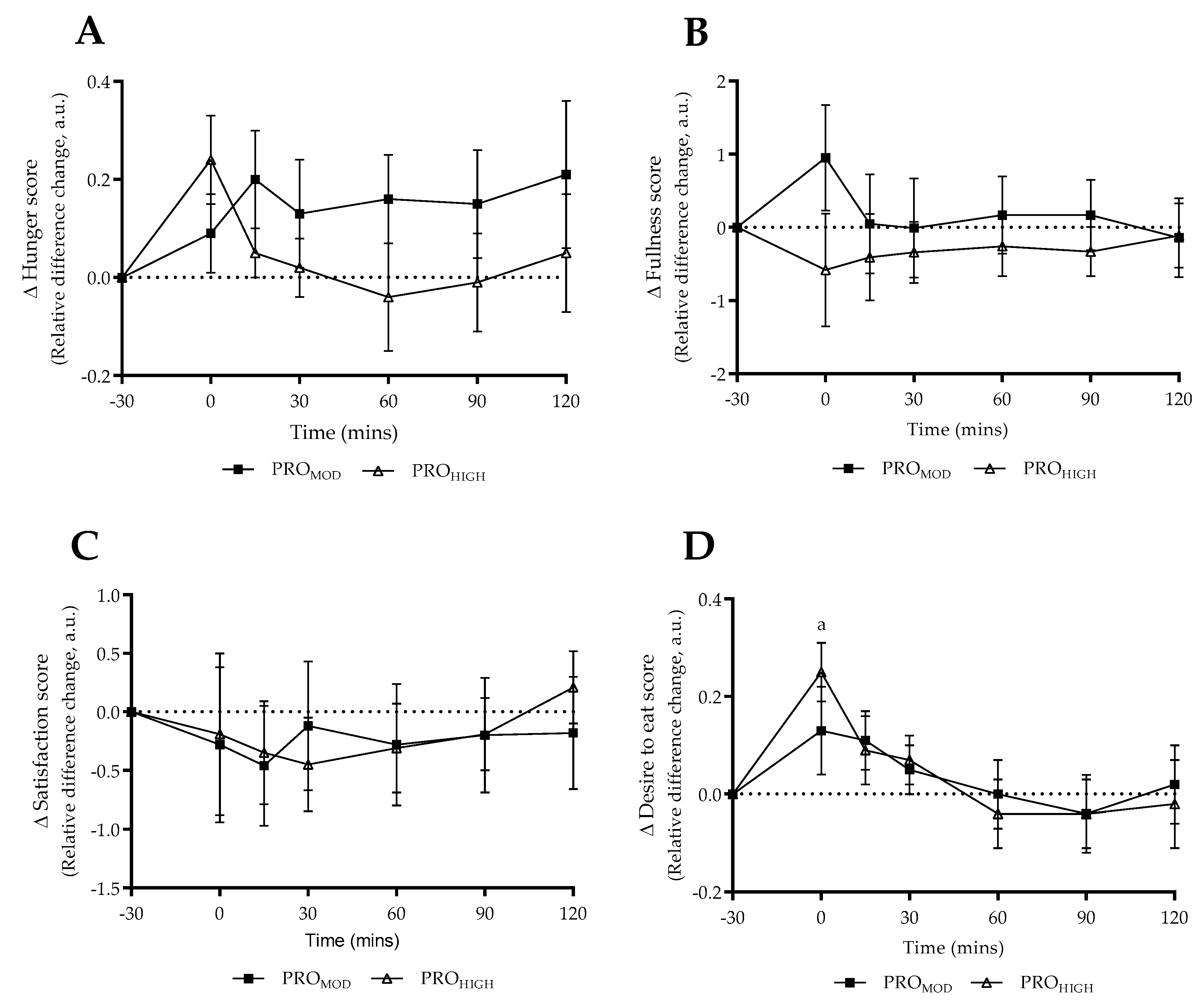
3.4. Test-Meal Satiety Questionnaire Responses
3.5. Ad libitum Intake and Satiety Responses
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Khodaee, M.; Olewinski, L.; Shadgan, B.; Kiningham, R.R. Rapid weight loss in sports with weight classes. Curr. Sports Med. Rep. 2015, 14, 435–441. [Google Scholar] [CrossRef]
- Rossow, L.M.; Fukuda, D.H.; Fahs, C.A.; Loenneke, J.P.; Stout, J.R. Natural bodybuilding competition preparation and recovery: A 12-month case study. Int. J. Sports Physiol. Perform. 2013, 8, 582–592. [Google Scholar] [CrossRef] [PubMed]
- Robinson, S.L.; Lambeth-Mansell, A.; Gillibrand, G.; Smith-Ryan, A.; Bannock, L. A nutrition and conditioning intervention for natural bodybuilding contest preparation: Case study. J. Int. Soc. Sports Nutr. 2015, 12, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Mettler, S.; Mitchell, N.; Tipton, K.D. Increased protein intake reduces lean body mass loss during weight loss in athletes. Med. Sci. Sports Exerc. 2010, 42, 326–337. [Google Scholar] [CrossRef] [PubMed]
- Tarnopolsky, M.A.; Atkinson, S.A.; MacDougall, J.D.; Chesley, A.; Phillips, S.; Schwarcz, H.P. Evaluation of protein requirements for trained strength athletes. J. Appl. Physiol. 1992, 73, 1986–1995. [Google Scholar] [CrossRef] [PubMed]
- Walberg, J.L.; Leidy, M.K.; Sturgill, D.J.; Hinkle, D.E.; Ritchey, S.J.; Sebolt, D.R. Macronutrient content of a hypoenergy diet affects nitrogen retention and muscle function in weight lifters. Int. J. Sports Med. 1988, 9, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Helms, E.R.; Aragon, A.A.; Fitschen, P.J. Evidence-based recommendations for natural bodybuilding contest preparation: Nutrition and supplementation. J. Int. Soc. Sports Nutr. 2014, 11, 20. [Google Scholar] [CrossRef] [PubMed]
- Lemon, P.W.R. Beyond the zone: Protein needs of active individuals. J. Am. Coll. Nutr. 2000, 19, 513S–521S. [Google Scholar] [CrossRef]
- Phillips, S.M. Dietary protein for athletes: From requirements to metabolic advantage. Appl. Physiol. Nutr. Metab. 2006, 31, 647–654. [Google Scholar] [CrossRef]
- Phillips, S.M.; Moore, D.R.; Tang, J.E. A critical examination of dietary protein requirements, benefits, and excesses in athletes. Int. J. Sport Nutr. Exerc. Metab. 2007, 17, 58–76. [Google Scholar] [CrossRef]
- Layman, D.K.; Boileau, R.A.; Erickson, D.J.; Painter, J.E.; Shiue, H.; Sather, C.; Christou, D.D. A reduced ratio of dietary carbohydrate to protein improves body composition and blood lipid profiles during weight loss in adult women. J. Nutr. 2003, 133, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Stiegler, P.; Cunliffe, A. The role of diet and exercise for the maintenance of fat-free mass and resting metabolic rate during weight loss. Sports Med. 2006, 36, 239–262. [Google Scholar] [CrossRef] [PubMed]
- Helms, E.R.; Zinn, C.; Rowlands, D.S.; Brown, S.R. A systematic review of dietary protein during caloric restriction in resistance trained lean athletes: A case for higher intakes. Int. J. Sport Nutr. Exerc. Metab. 2014, 24, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Rand, W.M.; Pellett, P.L.; Young, V.R. Meta-analysis of nitrogen balance studies for estimating protein requirements in healthy adults. Am. J. Clin. Nutr. 2003, 77, 109–127. [Google Scholar] [CrossRef] [PubMed]
- Blachier, F.; Beaumont, M.; Portune, K.J.; Steuer, N.; Lan, A.; Audebert, M.; Khodorova, N.; Andriamihaja, M.; Airinei, G.; Benamouzig, R.; et al. High-protein diets for weight management: Interactions with the intestinal microbiota and consequences for gut health. A position paper by the my new gut study group. Clin. Nutr. 2018. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S.M.; Van Loon, L.J.C. Dietary protein for athletes: From requirements to optimum adaptation. J. Sports Sci. 2011, 29, S29–S38. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S.M. A brief review of higher dietary protein diets in weight loss: A focus on athletes. Sports Med. 2014, 44 (Suppl. 2), S149–S153. [Google Scholar] [CrossRef]
- Spendlove, J.; Mitchell, L.; Gifford, J.; Hackett, D.; Slater, G.; Cobley, S.; O’Connor, H. Dietary intake of competitive bodybuilders. Sport Med. 2015, 45, 1041–1063. [Google Scholar] [CrossRef]
- Dhillon, J.; Craig, B.A.; Leidy, H.J.; Amankwaah, A.F.; Osei-Boadi Anguah, K.; Jacobs, A.; Jones, B.L.; Jones, J.B.; Keeler, C.L.; Keller, C.E.; et al. The effects of increased protein intake on fullness: A meta-analysis and its limitations. J. Acad. Nutr. Diet. 2016, 116, 968–983. [Google Scholar] [CrossRef]
- Weigle, D.S.; Breen, P.A.; Matthys, C.C.; Callahan, H.S.; Meeuws, K.E.; Burden, V.R.; Purnell, J.Q. A high-protein diet induces sustained reductions in appetite, ad libitum caloric intake, and body weight despite compensatory changes in diurnal plasma leptin and ghrelin concentrations. Am. J. Clin. Nutr. 2005, 82, 41–48. [Google Scholar] [CrossRef]
- Martens, E.A.; Lemmens, S.G.; Westerterp-Plantenga, M.S. Protein leverage affects energy intake of high-protein diets in humans. Am. J. Clin. Nutr. 2013, 97, 86–93. [Google Scholar] [CrossRef] [PubMed]
- Martens, E.A.; Tan, S.Y.; Dunlop, M.V.; Mattes, R.D.; Westerterp-Plantenga, M.S. Protein leverage effects of beef protein on energy intake in humans. Am. J. Clin. Nutr. 2014, 99, 1397–1406. [Google Scholar] [CrossRef] [PubMed]
- Helms, E.R.; Zinn, C.; Rowlands, D.S.; Naidoo, R.; Cronin, J. High-protein, low-fat, short-term diet results in less stress and fatigue than moderate-protein moderate-fat diet during weight loss in male weightlifters: A pilot study. Int. J. Sport Nutr. Exerc. Metab. 2015, 25, 163–170. [Google Scholar] [CrossRef] [PubMed]
- DeFreitas, J.M.; Beck, T.W.; Stock, M.S.; Dillon, M.A.; Kasishke, P.R. An examination of the time course of training-induced skeletal muscle hypertrophy. Eur. J. Appl. Physiol. 2011, 111, 2785–2790. [Google Scholar] [CrossRef] [PubMed]
- Brown, N.; Bubeck, D.; Haeufle, D.F.B.; Weickenmeier, J.; Kuhl, E.; Alt, W.; Schmitt, S. Weekly time course of neuro-muscular adaptation to intensive strength training. Front. Physiol. 2017, 8, 329. [Google Scholar] [CrossRef] [PubMed]
- Sale, D.G. Neural adaptation to resistance training. Med. Sci. Sports Exerc. 1988, 20 (Suppl. 5), S135–S145. [Google Scholar] [CrossRef] [PubMed]
- Trappe, S.; Williamson, D.; Godard, M. Maintenance of whole muscle strength and size following resistance training in older men. J. Gerontol. 2002, 57, B138–B143. [Google Scholar] [CrossRef]
- American College of Sports Medicine. American College of Sports Medicine position stand. Progression models in resistance training for healthy adults. Med. Sci. Sports Exerc. 2009, 41, 687–708. [Google Scholar] [CrossRef]
- Utter, A.C.; Kang, J.; Nieman, D.C.; Brown, V.A.; Dumke, C.L.; McAnulty, S.R.; McAnulty, L.S. Carbohydrate supplementation and perceived exertion during resistance exercise. J. Strength Cond. Res. 2005, 19, 939–943. [Google Scholar] [CrossRef]
- Roberts, J.; Zinchenko, A.; Suckling, C.; Smith, L.; Johnstone, J.; Henselmans, M. The short-term effect of high versus moderate protein intake on recovery after strength training in resistance-trained individuals. J. Int. Soc. Sports Nutr. 2017, 14, 44. [Google Scholar] [CrossRef]
- Antonio, J.; Peacock, C.; Ellerbroek, A.; Fromhoff, B.; Silver, T. The effects of consuming a high protein diet (4.4 g/kg/d) on body composition in resistance-trained individuals. J. Int. Soc. Sports Nutr. 2014, 11, 19. [Google Scholar] [CrossRef] [PubMed]
- Antonio, J.; Ellerbroek, A.; Silver, T.; Orris, S.; Scheiner, M.; Gonzalez, A.; Peacock, C. A high protein diet (3.4 g/kg/d) combined with a heavy resistance training program improves body composition in healthy trained men and women—A follow-up investigation. J. Int. Soc. Sports Nutr. 2015, 12, 39. [Google Scholar] [CrossRef] [PubMed]
- Antonio, J.; Ellerbroek, A.; Silver, T.; Vargas, L.; Peacock, C. The effects of a high protein diet on indices of health and body composition—A crossover trial in resistance-trained men. J. Int. Soc. Sports Nutr. 2016, 13, 1–7. [Google Scholar] [CrossRef] [PubMed]
- McArdle, W.; Katch, F.; Katch, V. Exercise Physiology: Energy, Nutrition and Human Performance, 5th ed.; Lippincott Williams and Wilkins: Philadelphia, PA, USA, 2001. [Google Scholar]
- Cunningham, J. Body composition and resting metabolic rate: The myth of feminine metabolism. Am. J. Clin. Nutr. 1982, 36, 721–726. [Google Scholar] [CrossRef] [PubMed]
- Stubbs, R.J.; Hughes, D.A.; Johnstone, A.M.; Rowley, E.; Reid, C.; Elia, M.; Stratton, R.; Delargy, H.; King, N.; Blundell, J.E. The use of visual analogue scales to assess motivation to eat in human subjects: A review of their reliability and validity with an evaluation of new hand-held computerized systems for temporal tracking of appetite ratings. Br. J. Nutr. 2000, 84, 405–415. [Google Scholar] [CrossRef] [PubMed]
- Flint, A.; Raben, A.; Blundell, J.E.; Astrup, A. Reproducibility, power and validity of visual analogue scales in assessment of appetite sensations in single test meal studies. Int. J. Obes. Relat. Metab. Disord. 2000, 24, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Raben, A.; Agerholm-Larsen, L.; Flint, A.; Holst, J.J.; Astrup, A. Meals with similar energy densities but rich in protein, fat, carbohydrate, or alcohol have different effects on energy expenditure and substrate metabolism but not on appetite and energy intake. Am. J. Clin. Nutr. 2003, 77, 91–100. [Google Scholar] [CrossRef]
- Bligh, H.F.; Godsland, I.F.; Frost, G.; Hunter, K.J.; Murray, P.; MacAulay, K.; Hyliands, D.; Talbot, D.C.; Casey, J.; Mulder, T.P.; et al. Plant-rich mixed meals based on Palaeolithic diet principles have a dramatic impact on incretin, peptide YY and satiety response, but show little effect on glucose and insulin homeostasis: An acute-effects randomised study. Br. J. Nutr. 2015, 113, 574–584. [Google Scholar] [CrossRef]
- Blatt, A.D.; Roe, L.S.; Rolls, B.J. Increasing the protein content of meals and its effect on daily energy intake. J. Am. Diet. Assoc. 2011, 111, 290–294. [Google Scholar] [CrossRef]
- Wiessing, K.R.; Xin, L.; Budgett, S.C.; Poppitt, S.D. No evidence of enhanced satiety following whey protein- or sucrose-enriched water beverages: A dose response trial in overweight women. Eur. J. Clin. Nutr. 2015, 69, 1238–1243. [Google Scholar] [CrossRef]
- Ravn, A.M.; Gregersen, N.T.; Christensen, R.; Rasmussen, L.G.; Hels, O.; Belza, A.; Raben, A.; Larsen, T.M.; Toubro, S.; Astrup, A. Thermic effect of a meal and appetite in adults: An individual participant data meta-analysis of meal-test trials. Food Nutr. Res. 2013, 57. [Google Scholar] [CrossRef] [PubMed]
- Phillips, S.M.; Chevalier, S.; Leidy, H.J. Protein “requirements” beyond the RDA: Implications for optimizing health. Appl. Physiol. Nutr. Metab. 2016, 41, 565–572. [Google Scholar] [CrossRef] [PubMed]
- Lemon, P.W.; Tarnopolsky, M.A.; MacDougall, J.D.; Atkinson, S.A. Protein requirements and muscle mass/strength changes during intensive training in novice bodybuilders. J. Appl. Physiol. 1992, 73, 767–775. [Google Scholar] [CrossRef] [PubMed]
- Jakubowicz, D.; Froy, O.; Wainstein, J.; Boaz, M. Meal timing and composition influence ghrelin levels, appetite scores and weight loss maintenance in overweight and obese adults. Steroids 2012, 77, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Barbosa-Silva, M.C.G.; Barros, A.J.D.; Wang, J.; Heymsfield, S.B.; Pierson, R.N. Bioelectrical impedance analysis: Population reference values for phase angle by age and sex. Am. J. Clin. Nutr. 2005, 82, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Norman, K.; Stobäus, N.; Pirlich, M.; Bosy-Westphal, A. Bioelectrical phase angle and impedance vector analysis—Clinical relevance and applicability of impedance parameters. Clin. Nutr. 2012, 31, 854–861. [Google Scholar] [CrossRef] [PubMed]
- Souza, M.F.; Tomeleri, C.M.; Ribeiro, A.S.; Schoenfeld, B.J.; Silva, A.M.; Sardinha, L.B.; Cyrino, E.S. Effect of resistance training on phase angle in older women: A randomized controlled trial. Scand. J. Med. Sci. Sports 2016, 8, 1–9. [Google Scholar] [CrossRef]
- Westerterp-Plantenga, M.S.; Lemmens, S.G.; Westerterp, K.R. Dietary protein—Its role in satiety, energetics, weight loss and health. Br. J. Nutr. 2012, 108, S105–S112. [Google Scholar] [CrossRef]
- Gibbons, C.; Caudwell, P.; Finlayson, G.; Webb, D.L.; Hellström, P.M.; Näslund, E.; Blundell, J.E. Comparison of postprandial profiles of ghrelin, active GLP-1, and total PYY to meals varying in fat and carbohydrate and their association with hunger and the phases of satiety. J. Clin. Endocrinol. Metab. 2013, 98, E847–E855. [Google Scholar] [CrossRef]
- Leidy, H.J.; Ortinau, L.C.; Douglas, S.M.; Hoertel, H.A. Beneficial effects of a higher-protein breakfast on the appetitive, hormonal, and neural signals controlling energy intake regulation in overweight/obese, “breakfast-skipping,” late-adolescent girls. Am. J. Clin. Nutr. 2013, 97, 677–688. [Google Scholar] [CrossRef]
- Doucet, É.; Laviolette, M.; Imbeault, P.; Strychar, I.; Rabasa-Lhoret, R.; Prud’homme, D. Total peptide YY is a correlate of postprandial energy expenditure but not of appetite or energy intake in healthy women. Metab. Clin. Exp. 2008, 57, 1458–1464. [Google Scholar] [CrossRef] [PubMed]
- OConnor, K.L.; Scisco, J.L.; Smith, T.J.; Young, A.J.; Montain, S.J.; Price, L.L.; Lieberman, H.R.; Karl, J.P. Altered appetite-mediating hormone concentrations precede compensatory overeating after severe, short-term energy deprivation in healthy adults. J. Nutr. 2016, 146, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Moran, L.J.; Luscombe-Marsh, N.D.; Noakes, M.; Wittert, G.A.; Keogh, J.B.; Clifton, P.M. The satiating effect of dietary protein is unrelated to postprandial ghrelin secretion. J. Clin. Endocrinol. Metab. 2005, 90, 5205–5211. [Google Scholar] [CrossRef] [PubMed]
- Leidy, H.J.; Armstrong, C.L.; Tang, M.; Mattes, R.D.; Campbell, W.W. The influence of higher protein intake and greater eating frequency on appetite control in overweight and obese men. Obesity 2010, 18, 1725–1732. [Google Scholar] [CrossRef] [PubMed]
- MacKenzie-Shalders, K.L.; Byrne, N.M.; Slater, G.J.; King, N.A. The effect of a whey protein supplement dose on satiety and food intake in resistance training athletes. Appetite 2015, 92, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Cuenca-Sánchez, M.; Navas-Carrillo, D.; Orenes-Piñero, E. Controversies surrounding high-protein diet intake: Satiating effect and kidney and bone health. Adv. Nutr. 2015, 6, 260–266. [Google Scholar] [CrossRef] [PubMed]
- Tan, X.; Cao, X.; Zou, J.; Shen, B.; Zhang, X.; Liu, Z.; Lv, W.; Teng, J.; Dign, X. Indoxyl sulfate, a valuable biomarker in chronic kidney disease and dialysis. Hemodial. Int. 2017, 21, 161–167. [Google Scholar] [CrossRef]
- Vanholder, R.; Schepers, E.; Pletinck, A.; Nagler, E.V.; Glorieux, G. The uremic toxicity of indoxyl sulfate and p-cresyl sulfate: A systematic review. J. Am. Soc. Nephrol. 2014, 25, 1897–1907. [Google Scholar] [CrossRef]
- Russell, W.R.; Gratz, S.W.; Duncan, S.H.; Holtrop, G.; Ince, J.; Scobbie, L.; Duncan, G.; Johnstone, A.M.; Lobley, G.E.; Wallace, R.J.; et al. High-protein, reduced-carbohydrate weight-loss diets promote metabolite profiles likely to be detrimental to colonic health. Am. J. Clin. Nutr. 2011, 93, 1062–1072. [Google Scholar] [CrossRef]
- Long, S.J.; Jeffcoat, A.R.; Millward, D.J. Effect of habitual dietary-protein intake on appetite and satiety. Appetite 2000, 35, 79–88. [Google Scholar] [CrossRef]
- Batterham, R.L.; Heffron, H.; Kapoor, S.; Chivers, J.E.; Chandarana, K.; Herzog, H.; Le Roux, C.W.; Thomas, E.L.; Bell, J.D.; Withers, D.J. Critical role for peptide YY in protein-mediated satiation and body-weight regulation. Cell Metab. 2006, 4, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Scheid, J.L.; De Souza, M.J. The Role of PYY in Eating Behavior and Diet. In Handbook of Behavior, Food and Nutrition; Preedy, V., Watson, R., Martin, C., Eds.; Springer: New York, NY, USA, 2011. [Google Scholar]
- Karra, E.; Chandarana, K.; Batterham, R.L. The role of peptide YY in appetite regulation and obesity. J. Physiol. 2009, 587, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Blom, W.A.; Lluch, A.; Stafleu, A.; Vinoy, S.; Holst, J.J.; Schaafsma, G.; Hendriks, H.F. Effect of a high-protein breakfast on the postprandial ghrelin response. Am. J. Clin. Nutr. 2006, 83, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Campbell, M.D.; Gonzalez, J.T.; Rumbold, P.L.; Walker, M.; Shaw, J.A.; Stevenson, E.J.; West, D.J. Comparison of appetite responses to high- and low-glycemic index postexercise meals under matched insulinemia and fiber in type 1 diabetes. Am. J. Clin. Nutr. 2015, 101, 478–486. [Google Scholar] [CrossRef] [PubMed]
- Clayton, D.J.; James, L.J. The effect of breakfast on appetite regulation, energy balance and exercise performance. Proc. Nutr. Soc. 2016, 75, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Clayton, D.J.; Stensel, D.J.; James, L.J. Effect of breakfast omission on subjective appetite, metabolism, acylated ghrelin and GLP-17-36 during rest and exercise. Nutrition 2016, 32, 179–185. [Google Scholar] [CrossRef]
- Iepsen, E.W.; Lundgren, J.; Holst, J.J.; Madsbad, S.; Torekov, S.S. Successful weight loss maintenance includes long-term increased meal responses of GLP-1 and PYY3-36. Eur. J. Endocrinol. 2016, 174, 775–784. [Google Scholar] [CrossRef]
- Sumithran, P.; Prendergast, L.A.; Delbridge, E.; Purcell, K.; Shulkes, A.; Kriketos, A.; Proietto, J. Long-term persistence of hormonal adaptations to weight loss. N. Engl. J. Med. 2011, 365, 1597–1604. [Google Scholar] [CrossRef]
| Variable | All Participants (n = 16) | Male (n = 9) | Female (n = 7) |
|---|---|---|---|
| Age (years) | 28 ± 2 | 26 ± 2 | 30 ± 3 |
| Height (m) | 1.72 ± 0.03 | 1.79 ± 0.02 | 1.63 ± 0.04 * |
| Body-mass (kg) | 88.83 ± 5.54 | 95.68 ± 5.73 | 80.01 ± 6.21 |
| Body-fat (%) | 21.85 ± 1.82 | 18.33 ± 1.91 | 26.37 ± 2.57 * |
| BIA FM (kg) | 25.52 ± 3.37 | 23.12 ± 3.02 | 28.60 ± 6.82 |
| BIA FM (%) | 27.53 ± 2.46 | 23.40 ± 1.99 | 32.84 ± 4.40 * |
| BIA FFM (kg) | 63.31 ± 3.50 | 72.56 ± 3.05 | 51.41 ± 3.48 * |
| BIA FFM (%) | 72.47 ± 2.46 | 76.60 ± 1.99 | 67.16 ± 4.40 * |
| PA (°) | 8.27 ± 0.16 | 8.71 ± 0.10 | 7.70 ± 0.19 * |
| TBW (%) | 52.26 ± 1.77 | 55.23 ± 1.43 | 48.43 ± 3.17 * |
| ICW (%) | 58.49 ± 0.81 | 59.48 ± 0.48 | 57.23 ± 1.43 |
| ECW (%) | 41.48 ± 0.81 | 40.52 ± 0.84 | 42.71 ± 1.45 |
| Variable | Category | PROMOD | PROHIGH | ||
|---|---|---|---|---|---|
| PRE | POST | PRE | POST | ||
| Energy Intake | (kcal·d−1) | 2359.44 ± 220.84 * | 2057.44 ± 104.47 | 2270.71 ± 150.41 * | 2119.44 ± 122.79 |
| (kcal·kg−1·d−1) | 26.89 ± 1.94 | 23.83 ± 0.93 | 25.53 ± 1.17 | 24.35 ± 0.84 | |
| Protein Intake | (g·d−1) | 158.19 ± 13.55 | 162.94 ± 10.14 | 166.71 ± 18.33 | 256.13 ± 16.64 a,b |
| (g·kg−1·d−1) | 1.80 ± 0.12 | 1.84 ± 0.02 | 1.84 ± 0.15 | 2.89 ± 0.01 a,b | |
| (%EI) | 27.79 ± 1.98 | 31.68 ± 1.33 | 28.79 ± 2.13 | 48.44 ± 1.93 a,b | |
| Carbohydrate Intake | (g·d−1) | 220.75 ± 25.34 | 238.75 ± 13.32 | 221.79 ± 12.07 | 160.88 ± 13.88 a,b |
| (g·kg−1·d−1) | 2.57 ± 0.25 | 2.81 ± 0.18 | 2.55 ± 0.16 | 1.90 ± 0.16 a,b | |
| (%EI) | 38.08 ± 2.52 | 46.55 ± 1.32 a | 40.28 ± 2.45 | 30.32 ± 1.82 a,b | |
| Fat Intake | (g·d−1) | 90.94 ± 11.40 | 46.88 ± 2.62 a | 73.79 ± 7.68 | 47.31 ± 2.80 a |
| (g·kg−1·d−1) | 1.02 ± 0.11 | 0.55 ± 0.03 a | 0.83 ± 0.08 b | 0.55 ± 0.03 a | |
| (%EI) | 14.83 ± 0.77 | 9.12 ± 0.25 a | 12.86 ± 0.90 b | 8.94 ± 0.21 a | |
| Question | PROMOD1–3 | PROMOD5–7 | PROHIGH1–3 | PROHIGH5–7 |
|---|---|---|---|---|
| Hunger | 48.54 ± 3.48 | 51.46 ± 4.89 | 43.33 ± 4.22 | 41.67 ± 3.68 |
| Fullness | 62.29 ± 4.11 | 58.65 ± 4.64 | 64.38 ± 3.81 | 65.52 ± 3.99 |
| Satisfaction | 65.42 ± 4.38 | 58.96 ± 4.51 | 68.65 ± 2.34 | 67.29 ± 4.28 a |
| Desire to eat | 50.94 ± 5.53 | 57.92 ± 5.45 | 45.00 ± 5.13 | 46.15 ± 5.58 |
| Cravings | 46.25 ± 4.96 | 57.60 ± 4.41 b | 40.83 ± 4.59 | 47.19 ± 4.60 |
| Energy | 65.00 ± 2.81 | 67.81 ± 4.54 | 64.48 ± 3.18 | 69.06 ± 4.64 |
| Mood | 72.92 ± 3.35 | 67.92 ± 5.33 | 67.71 ± 3.26 | 71.88 ± 3.89 |
| Training enjoyment | 72.29 ± 4.35 | 68.23 ± 4.73 | 71.67 ± 4.48 | 73.02 ± 4.19 |
| Training motivation | 71.67 ± 4.97 | 66.56 ± 4.68 | 70.21 ± 3.93 | 70.83 ± 5.92 |
| Hormone | Time (mins) | PROMOD | PROHIGH | ||
|---|---|---|---|---|---|
| PRE | POST | PRE | POST | ||
| Ghrelin (pg·mL−1) | 0 | 1125.97 ± 125.85 | 972.81 ± 130.42 | 920.12 ± 143.43 | 1088.17 ± 158.77 |
| 60 | 696.42 ± 96.61 * | 659.73 ± 86.39 * | 672.27 ± 119.29 * | 786.61 ± 117.33 * | |
| 120 | 758.91 ± 129.38 * | 613.57 ± 114.26 *, a | 714.75 ± 136.56 * | 850.60 ± 147.68 * | |
| PYY (pg·mL−1) | 0 | 103.62 ± 10.15 | 84.87 ± 8.94 | 87.94 ± 11.45 | 100.65 ± 9.54 |
| 60 | 130.83 ± 9.09 * | 129.38 ± 10.49 * | 133.84 ± 15.14 * | 141.02 ± 12.23 * | |
| 120 | 137.60 ± 8.71 * | 147.14 ± 11.94 * | 142.11 ± 13.29 * | 143.34 ± 11.98 * | |
| Variable | Category | PROMOD | PROHIGH |
|---|---|---|---|
| Energy Intake | (kcal·d−1) | 2197.81 ± 177.58 | 2348.14 ± 232.46 |
| (kcal·kg−1·d−1) | 25.64 ± 1.62 | 26.52 ± 1.81 | |
| Protein Intake | (g·d−1) | 162.38 ± 11.18 | 165.07 ± 12.89 |
| (g·kg−1·d−1) | 1.85 ± 0.03 | 1.86 ± 0.07 | |
| (%EI) | 30.87 ± 2.13 | 29.40 ± 1.89 | |
| Carbohydrate Intake | (g·d−1) | 214.94 ± 19.61 | 230.61 ± 24.33 |
| (g·kg−1·d−1) | 2.57 ± 0.24 | 2.65 ± 0.23 | |
| (%EI) | 39.14 ± 1.98 | 39.66 ± 1.85 | |
| Fat Intake | (g·d−1) | 72.69 ± 9.71 | 81.29 ± 11.01 |
| (g·kg−1·d−1) | 0.84 ± 0.09 | 0.90 ± 0.09 | |
| (%EI) | 12.64 ± 0.76 | 13.29 ± 0.70 |
| Question Category | PROMOD | PROHIGH | ΔPROMOD | ΔPROHIGH |
|---|---|---|---|---|
| Hunger | 41.67 ± 3.61 | 40.78 ± 3.48 | −9.67 ± 4.68 a | −0.89 ± 2.74 |
| Fullness | 69.00 ± 4.39 | 73.39 ± 3.10 | 9.78 ± 3.74 a | 7.87 ± 4.61 |
| Satisfaction | 73.11 ± 3.80 | 71.56 ± 3.00 | 13.11 ± 4.33 a | 4.26 ± 4.89 |
| Desire to eat | 39.67 ± 2.96 | 38.03 ± 3.62 | −16.78 ± 4.54 a | −8.12 ± 5.22 |
| Cravings | 42.89 ± 4.12 | 46.15 ± 4.23 | −13.00 ± 4.27 a | −1.04 ± 5.08 * |
| Energy | 71.33 ± 3.31 | 71.93 ± 2.91 | 2.99 ± 4.30 | 2.87 ± 3.34 |
| Mood | 76.11 ± 4.03 | 77.55 ± 2.09 | 8.33 ± 3.70 a | 5.68 ± 2.72 a |
| Training enjoyment | 79.35 ± 4.44 | 74.67 ± 4.51 | 9.23 ± 6.57 | 1.67 ± 4.43 |
| Training motivation | 78.85 ± 5.39 | 72.89 ± 4.99 | 8.97 ± 5.08 a | 1.33 ± 3.93 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roberts, J.; Zinchenko, A.; Mahbubani, K.T.; Johnstone, J.; Smith, L.; Merzbach, V.; Blacutt, M.; Banderas, O.; Villasenor, L.; Vårvik, F.T.; et al. Satiating Effect of High Protein Diets on Resistance-Trained Individuals in Energy Deficit. Nutrients 2019, 11, 56. https://doi.org/10.3390/nu11010056
Roberts J, Zinchenko A, Mahbubani KT, Johnstone J, Smith L, Merzbach V, Blacutt M, Banderas O, Villasenor L, Vårvik FT, et al. Satiating Effect of High Protein Diets on Resistance-Trained Individuals in Energy Deficit. Nutrients. 2019; 11(1):56. https://doi.org/10.3390/nu11010056
Chicago/Turabian StyleRoberts, Justin, Anastasia Zinchenko, Krishnaa T. Mahbubani, James Johnstone, Lee Smith, Viviane Merzbach, Miguel Blacutt, Oscar Banderas, Luis Villasenor, Fredrik T. Vårvik, and et al. 2019. "Satiating Effect of High Protein Diets on Resistance-Trained Individuals in Energy Deficit" Nutrients 11, no. 1: 56. https://doi.org/10.3390/nu11010056
APA StyleRoberts, J., Zinchenko, A., Mahbubani, K. T., Johnstone, J., Smith, L., Merzbach, V., Blacutt, M., Banderas, O., Villasenor, L., Vårvik, F. T., & Henselmans, M. (2019). Satiating Effect of High Protein Diets on Resistance-Trained Individuals in Energy Deficit. Nutrients, 11(1), 56. https://doi.org/10.3390/nu11010056









