Interplay between Nutrition and Hearing Loss: State of Art
Abstract
1. Introduction
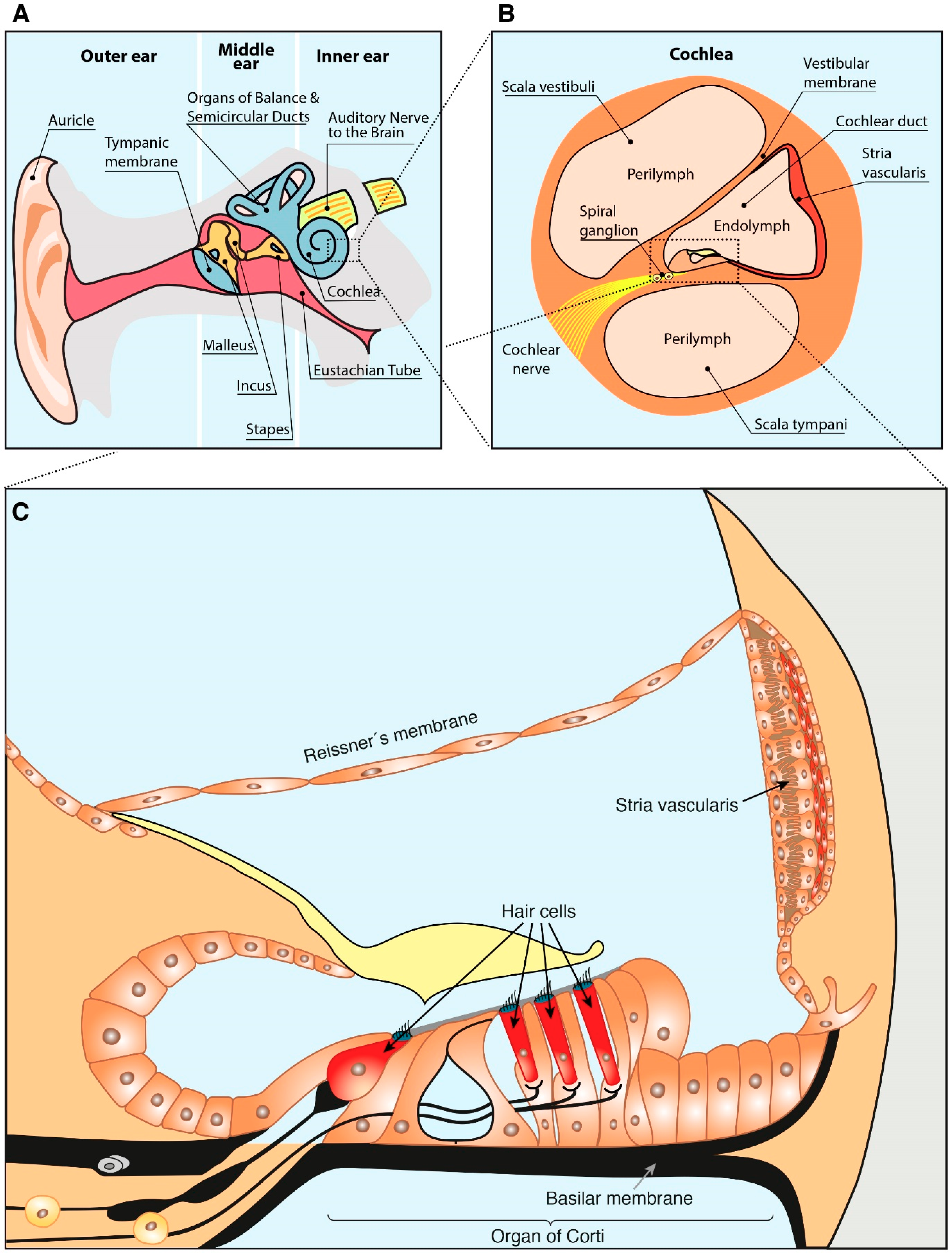
2. Anatomy of the Ear and Hearing Loss
3. Caloric Restriction and Hearing loss
4. Macronutrients and Hearing Loss
5. Micronutrients and Hearing Loss
5.1. Vitamins and Hearing Loss
5.2. Minerals and Hearing Loss
6. Other Dietary Factors and Hearing Loss
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dror, A.A.; Avraham, K.B. Hearing loss: Mechanisms revealed by genetics and cell biology. Annu. Rev. Genet. 2009, 43, 411–437. [Google Scholar] [CrossRef] [PubMed]
- Roth, T.N.; Hanebuth, D.; Probst, R. Prevalence of age-related hearing loss in Europe: A review. Eur. Arch. Otorhinolaryngol. 2011, 268, 1101–1107. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Childhood Hearing Loss: Strategies for Prevention and Care; World Health Organization: Geneva, Switzerland, 2016. [Google Scholar]
- Deafness and Hearing Loss. Available online: http://www.who.int/news-room/fact-sheets/detail/deafness-and-hearing-loss (accessed on 15 October 2018).
- Li-Korotky, H.S. Age-related hearing loss: Quality of care for quality of life. Gerontologist 2012, 52, 265–271. [Google Scholar] [CrossRef]
- TARGEAR NEWSLETTER JUNE 2017: About the Presbycusis. Available online: http://targear.eu/targear-newsletter-june-2017/ (accessed on 22 November 2018).
- Editorial Board. Hearing loss: Time for sound action. The Lancet 2017, 390, 2414. [Google Scholar] [CrossRef]
- Wilson, B.S.; Tucci, D.L.; Merson, M.H.; O’Donoghue, G.M. Global hearing health care: New findings and perspectives. Lancet 2017, 390, 2503–2515. [Google Scholar] [CrossRef]
- Emmett, S.D.; West, K.P., Jr. Nutrition and hearing loss: A neglected cause and global health burden. Am. J. Clin. Nutr. 2015, 102, 987–988. [Google Scholar] [CrossRef] [PubMed]
- Ciorba, A.; Bianchini, C.; Pelucchi, S.; Pastore, A. The impact of hearing loss on the quality of life of elderly adults. Clin. Interv. Aging 2012, 7, 159–163. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, L.L.; Tucci, D.L. Hearing Loss in Adults. N. Engl. J. Med. 2017, 377, 2465–2473. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). Global Costs of Unaddressed Hearing Loss and Cost-Effectiveness of Interventions: A WHO Report; World Health Organization: Geneva, Switzerland, 2017. [Google Scholar]
- Crowson, M.G.; Hertzano, R.; Tucci, D.L. Emerging Therapies for Sensorineural Hearing Loss. Otol. Neurotol. 2017, 38, 792–803. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Multi-Country Assessment of National Capacity to Provide Hearing Care; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar]
- Karli, R.; Gül, A.; Uğur, B. Effect of vitamin B12 deficiency on otoacoustic emissions. Acta Otorhinolaryngol. Ital. 2013, 33, 243–247. [Google Scholar]
- Lasisi, A.O.; Fehintola, F.A.; Yusuf, O.B. Age-related hearing loss, vitamin B12, and folate in the elderly. Otolaryngol. Head Neck Surg. 2010, 143, 826–830. [Google Scholar] [CrossRef] [PubMed]
- Cadoni, G.; Agostino, S.; Scipione, S.; Galli, J. Low serum folate levels: A risk factor for sudden sensorineural hearing loss? Acta Otolaryngol. 2004, 124, 608–611. [Google Scholar] [CrossRef] [PubMed]
- Gok, U.; Halifeoglu, I.; Canatan, H.; Yildiz, M.; Gursu, M.F.; Gur, B. Comparative analysis of serum homocysteine, folic acid and Vitamin B12 levels in patients with noise-induced hearing loss. Auris Nasus Larynx 2004, 31, 19–22. [Google Scholar] [CrossRef] [PubMed]
- Rosen, S.; Olin, P.; Rosen, H.V. Diery prevention of hearing loss. Acta Otolaryngol. 1970, 70, 242–247. [Google Scholar] [CrossRef] [PubMed]
- Dullemeijer, C.; Verhoef, P.; Brouwer, I.A.; Kok, F.J.; Brummer, R.J.; Durga, J. Plasma very long-chain n-3 polyunsaturated fatty acids and age-related hearing loss in older adults. J. Nutr. Health Aging 2010, 14, 347–351. [Google Scholar] [CrossRef]
- Gopinath, B.; Flood, V.M.; Rochtchina, E.; McMahon, C.M.; Mitchell, P. Consumption of omega-3 fatty acids and fish and risk of age-related hearing loss. Am. J. Clin. Nutr. 2010, 92, 416–421. [Google Scholar] [CrossRef]
- Shargorodsky, J.; Curhan, S.G.; Henderson, E.; Eavey, R.; Curhan, G.C. Heavy metals exposure and hearing loss in US adolescents. Arch Otolaryngol. Head Neck Surg. 2011, 137, 1183–1189. [Google Scholar] [CrossRef]
- Magariños, M.; Contreras, J.; Aburto, M.R.; Varela-Nieto, I. Early development of the vertebrate inner ear. Anat. Rec. (Hoboken) 2012, 295, 1775–1790. [Google Scholar] [CrossRef]
- Magariños, M.; Contreras, J.; Varela-Nieto, I. Early development of the vertebrate inner ear. In Development of the Auditory and Vestibular Systems; Romand, R., Varela-NIeto, I., Eds.; Elservier: Amsterdam, The Netherlands, 2014; pp. 1–30. [Google Scholar]
- Sanchez-Calderon, H.; Rodriguez-de la Rosa, L.; Milo, M.; Pichel, J.G.; Holley, M.; Varela-Nieto, I. RNA microarray analysis in prenatal mouse cochlea reveals novel IGF-I target genes: Implication of MEF2 and FOXM1 transcription factors. PLoS ONE 2010, 5, e8699. [Google Scholar] [CrossRef]
- Rivera, T.; Sanz, L.; Camarero, G.; Varela-Nieto, I. Drug delivery to the inner ear: Strategies and their therapeutic implications for sensorineural hearing loss. Curr. Drug Deliv. 2012, 9, 231–242. [Google Scholar] [CrossRef]
- Clark, J.G. Uses and abuses of hearing loss classification. ASHA 1981, 23, 493–500. [Google Scholar] [PubMed]
- Manrique, M.; Ramos, A.; Morera, C.; Cenjor, C.; Lavilla, M.J.; Boleas, M.S.; Cervera-Paz, F.J. Analysis of the cochlear implant as a treatment technique for profound hearing loss in pre and postlocutive patients. Acta Otorrinolaringol. Esp. 2006, 57, 2–23. [Google Scholar] [CrossRef]
- Cediel, R.; Riquelme, R.; Contreras, J.; Díaz, A.; Varela-Nieto, I. Sensorineural hearing loss in insulin-like growth factor I-null mice: A new model of human deafness. Eur. J. Neurosci. 2006, 23, 587–590. [Google Scholar] [CrossRef]
- Jougleux, J.L.; Rioux, F.M.; Church, M.W.; Fiset, S.; Surette, M.E. Mild maternal iron deficiency anemia during pregnancy and lactation in guinea pigs causes abnormal auditory function in the offspring. J. Nutr. 2011, 141, 1390–1395. [Google Scholar] [CrossRef] [PubMed]
- Nandi, S.S.; Dhatrak, S.V. Occupational noise-induced hearing loss in India. Indian J. Occup. Environ. Med. 2008, 12, 53–56. [Google Scholar] [CrossRef] [PubMed]
- Lanvers-Kaminsky, C.; Zehnhoff-Dinnesen, A.A.; Parfitt, R.; Ciarimboli, G. Drug-induced ototoxicity: Mechanisms, Pharmacogenetics, and protective strategies. Clin. Pharmacol. Ther. 2017, 101, 491–500. [Google Scholar] [CrossRef]
- Cruickshanks, K.J.; Tweed, T.S.; Wiley, T.L.; Klein, B.E.; Klein, R.; Chappell, R.; Nondahl, D.M.; Dalton, D.S. The 5-year incidence and progression of hearing loss: The epidemiology of hearing loss study. Arch Otolaryngol. Head Neck Surg. 2003. [Google Scholar] [CrossRef] [PubMed]
- Yamasoba, T.; Lin, F.R.; Someya, S.; Kashio, A.; Sakamoto, T.; Kondo, K. Current concepts in age-related hearing loss: Epidemiology and mechanistic pathways. Hear Res. 2013, 303, 30–38. [Google Scholar] [CrossRef]
- Shearer, A.E.; Hildebrand, M.S.; Sloan, C.M.; Smith, R.J. Deafness in the genomics era. Hear Res. 2011, 282, 1–9. [Google Scholar] [CrossRef]
- Shearer, A.E.; Hildebrand, M.S.; Smith, R.J.H. Hereditary Hearing Loss and Deafness Overview. In Gene Reviews; Adam, M.P., Pagon, R.A., Wallace, S.E., Bean, L.J.H., Stephens, K., Amemiya, A., Eds.; GeneReviews®: Seattle, WA, USA, 1999. [Google Scholar]
- Agrawal, Y.; Platz, E.A.; Niparko, J.K. Prevalence of hearing loss and differences by demographic characteristics among US adults: Data from the National Health and Nutrition Examination Survey, 1999–2004. Arch Intern. Med. 2008, 168, 1522–1530. [Google Scholar] [CrossRef]
- Yang, C.H.; Schrepfer, T.; Schacht, J. Age-related hearing impairment and the triad of acquired hearing loss. Front. Cell. Neurosci. 2015. [Google Scholar] [CrossRef] [PubMed]
- Dawes, P.; Cruickshanks, K.J.; Moore, D.R.; Edmondson-Jones, M.; McCormack, A.; Fortnum, H.; Munro, K.J. Cigarette smoking, passive smoking, alcohol consumption, and hearing loss. J. Assoc. Res. Otolaryngol. 2014, 15, 663–674. [Google Scholar] [CrossRef] [PubMed]
- Fransen, E.; Topsakal, V.; Hendrickx, J.J.; Van Laer, L.; Huyghe, J.R.; Van Eyken, E.; Lemkens, N.; Hannula, S.; Mäki-Torkko, E.; Jensen, M.; et al. Occupational noise, smoking, and a high body mass index are risk factors for age-related hearing impairment and moderate alcohol consumption is protective: A European population-based multicenter study. J. Assoc. Res. Otolaryngol. 2008, 9, 264–276. [Google Scholar] [CrossRef] [PubMed]
- DeCarlo, C.A.; HA, T.; Williams, D.; Dixon, R.A.; MacDonald, S.W. BioAge: Toward a multi-determined, mechanistic account of cognitive aging. Aging Res. Rev. 2014, 18, 95–105. [Google Scholar] [CrossRef] [PubMed]
- López-Otín, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. The hallmarks of aging. Cell 2013, 153, 1194–1217. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.Z.; Yan, D. Aging and hearing loss. J. Pathol. 2007, 211, 188–197. [Google Scholar] [CrossRef] [PubMed]
- Desai, M.; Pratt, L.A.; Lentzner, H.; Robinson, K.N. Trends in vision and hearing among older Americans. Aging Trends 2001, 2, 1–8. [Google Scholar]
- Liberman, M.C. Noise-induced and age-related hearing loss: New perspectives and potential therapies. F1000Research 2017, 6, 927. [Google Scholar] [CrossRef]
- Someya, S.; Yu, W.; Hallows, W.C.; Xu, J.; Vann, J.M.; Leeuwenburgh, C.; Tanokura, M.; Denu, J.M.; Prolla, T.A. Sirt3 mediates reduction of oxidative damage and prevention of age-related hearing loss under caloric restriction. Cell 2010, 143, 802–812. [Google Scholar] [CrossRef]
- Lee, F.S.; Matthews, L.J.; Dubno, J.R.; Mills, J.H. Longitudinal study of pure-tone thresholds in older persons. Ear Hear. 2005, 26, 1–11. [Google Scholar] [CrossRef]
- Le Prell, C.G.; Yamashita, D.; Minami, S.B.; Yamasoba, T.; Miller, J.M. Mechanisms of noise-induced hearing loss indicate multiple methods of prevention. Hear. Res. 2007, 226, 22–43. [Google Scholar] [CrossRef] [PubMed]
- Darrat, I.; Ahmad, N.; Seidman, K.; Seidman, M.D. Auditory research involving antioxidants. Curr. Opin. Otolaryngol. Head Neck Surg. 2007, 15, 358–363. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. World Report on Aging and Health; World Health Organization: Luxembourg, 2015. [Google Scholar]
- Balasubramanian, P.; Howell, P.R.; Anderson, R.M. Aging and Caloric Restriction Research: A Biological Perspective with Translational Potential. EBioMedicine 2017, 21, 37–44. [Google Scholar] [CrossRef] [PubMed]
- O’Flanagan, C.H.; Smith, L.A.; McDonell, S.B.; Hursting, S.D. When less may be more: Calorie restriction and response to cancer therapy. BMC Med. 2017, 15, 106. [Google Scholar] [CrossRef] [PubMed]
- Fontana, L.; Partridge, L.; Longo, V.D. Extending healthy life span--from yeast to humans. Science 2010, 328, 321–326. [Google Scholar] [CrossRef]
- Fujimoto, C.; Yamasoba, T. Oxidative stresses and mitochondrial dysfunction in age-related hearing loss. Oxid. Med. Cell. Longev. 2014, 2014. [Google Scholar] [CrossRef] [PubMed]
- Han, C.; Someya, S. Maintaining good hearing: Calorie restriction, Sirt3, and glutathione. Exp. Gerontol. 2013, 48, 1091–1095. [Google Scholar] [CrossRef]
- Speakman, J.R.; Mitchell, S.E. Caloric restriction. Mol. Aspects Med. 2011, 32, 159–221. [Google Scholar] [CrossRef]
- Mannström, P.; Ulfhake, B.; Kirkegaard, M.; Ulfendahl, M. Dietary restriction reduces age-related degeneration of stria vascularis in the inner ear of the rat. Exp. Gerontol. 2013, 48, 1173–1179. [Google Scholar] [CrossRef]
- Hirschey, M.D.; Shimazu, T.; Goetzman, E.; Jing, E.; Schwer, B.; Lombard, D.B.; Grueter, C.A.; Harris, C.; Biddinger, S.; Ilkayeva, O.R.; et al. SIRT3 regulates mitochondrial fatty-acid oxidation by reversible enzyme deacetylation. Nature 2010, 464, 121–125. [Google Scholar] [CrossRef]
- Sundaresan, N.R.; Samant, S.A.; Pillai, V.B.; Rajamohan, S.B.; Gupta, M.P. SIRT3 is a stress-responsive deacetylase in cardiomyocytes that protects cells from stress-mediated cell death by deacetylation of Ku70. Mol. Cell Biol. 2008, 28, 6384–6401. [Google Scholar] [CrossRef] [PubMed]
- Someya, S.; Yamasoba, T.; Weindruch, R.; Prolla, T.A.; Tanokura, M. Caloric restriction suppresses apoptotic cell death in the mammalian cochlea and leads to prevention of presbycusis. Neurobiol. Aging 2007, 28, 1613–1622. [Google Scholar] [CrossRef] [PubMed]
- Someya, S.; Tanokura, M.; Weindruch, R.; Prolla, T.A.; Yamasoba, T. Effects of caloric restriction on age-related hearing loss in rodents and rhesus monkeys. Curr. Aging Sci. 2010, 3, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Fowler, C.G.; Torre, P., III; Kemnitz, J.W. Effects of caloric restriction and aging on the auditory function of rhesus monkeys (Macaca mulatta): The University of Wisconsin study. Hear. Res. 2002, 169, 24–35. [Google Scholar] [CrossRef]
- Ramsey, J.J.; Colman, R.J.; Binkley, N.C.; Christensen, J.D.; Gresl, T.A.; Kemnitz, J.W.; Weindruch, R. Dietary restriction and aging in rhesus monkeys: The University of Wisconsin study. Exp. Gerontol. 2000, 35, 1131–1149. [Google Scholar] [CrossRef]
- Torre, P., III; Mattison, J.A.; Fowler, C.G.; Lane, M.A.; Roth, G.S.; Ingram, D.K. Assessment of auditory function in rhesus monkeys (Macaca mulatta): Effects of age and calorie restriction. Neurobiol. Aging 2004, 25, 945–954. [Google Scholar] [CrossRef] [PubMed]
- Fowler, C.G.; Chiasson, K.B.; Leslie, T.H.; Thomas, D.; Beasley, T.M.; Kemnitz, J.W.; Weindruch, R. Auditory function in rhesus monkeys: Effects of aging and caloric restriction in the Wisconsin monkeys five years later. Hear. Res. 2010, 261, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Rippe, J.M.; Sievenpiper, J.L.; Lê, K.A.; White, J.S.; Clemens, R.; Angelopoulos, T.J. What is the appropriate upper limit for added sugars consumption? Nutr. Rev. 2017, 75, 18–36. [Google Scholar] [CrossRef]
- Xi, B.; Huang, Y.; Reilly, K.H.; Li, S.; Zheng, R.; Barrio-Lopez, M.T.; Martinez-Gonzalez, M.A.; Zhou, D. Sugar-sweetened beverages and risk of hypertension and CVD: A dose-response meta-analysis. Br. J. Nutr. 2015, 113, 709–717. [Google Scholar] [CrossRef]
- Gopinath, B.; Flood, V.M.; McMahon, C.M.; Burlutsky, G.; Brand-Miller, J.; Mitchell, P. Dietary glycemic load is a predictor of age-related hearing loss in older adults. J. Nutr. 2010, 140, 2207–2212. [Google Scholar] [CrossRef]
- Tachibana, N.; Iwaoka, Y.; Hirotsuka, M.; Horio, F.; Kohno, M. Beta-conglycinin lowers very-low-density lipoprotein-triglyceride levels by increasing adiponectin and insulin sensitivity in rats. Biosci. Biotechnol. Biochem. 2010, 74, 1250–1255. [Google Scholar] [CrossRef] [PubMed]
- Tanigawa, T.; Shibata, R.; Kondo, K.; Katahira, N.; Kambara, T.; Inoue, Y.; Nonoyama, H.; Horibe, Y.; Ueda, H.; Murohara, T. Soybean β-Conglycinin Prevents Age-Related Hearing Impairment. PLoS ONE 2015, 10, e0137493. [Google Scholar] [CrossRef] [PubMed]
- Rosen, S.; Olin, P. Hearing loss and coronary heart disease. Bull. N. Y. Acad. Med. 1965, 41, 1052–1068. [Google Scholar] [CrossRef] [PubMed]
- Fusconi, M.; Chistolini, A.; de Virgilio, A.; Greco, A.; Massaro, F.; Turchetta, R.; Benincasa, A.T.; Tombolini, M.; de Vincentiis, M. Sudden sensorineural hearing loss: A vascular cause? Analysis of prothrombotic risk factors in head and neck. Int. J. Audiol. 2012, 51, 800–805. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Harris, W.S.; Chung, M.; Lichtenstein, A.H.; Balk, E.M.; Kupelnick, B.; Jordan, H.S.; Lau, J. n-3 Fatty acids from fish or fish-oil supplements, but not alpha-linolenic acid, benefit cardiovascular disease outcomes in primary- and secondary-prevention studies: A systematic review. Am. J. Clin. Nutr. 2006, 84, 5–17. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, A.; Sun, D.; Rahman, M.; Fernandes, G. Different ratios of eicosapentaenoic and docosahexaenoic omega-3 fatty acids in commercial fish oils differentially alter pro-inflammatory cytokines in peritoneal macrophages from C57BL/6 female mice. J. Nutr. Biochem. 2007, 18, 23–30. [Google Scholar] [CrossRef]
- Gates, G.A.; Cobb, J.L.; D’Agostino, R.B.; Wolf, P.A. The relation of hearing in the elderly to the presence of cardiovascular disease and cardiovascular risk factors. Arch Otolaryngol. Head Neck Surg. 1993, 119, 156–161. [Google Scholar] [CrossRef]
- Gratton, M.A.; Schulte, B.A. Alterations in microvasculature are associated with atrophy of the stria vascularis in quiet-aged gerbils. Hear. Res. 1995, 82, 44–52. [Google Scholar] [CrossRef]
- Durga, J.; Verhoef, P.; Anteunis, L.J.; Schouten, E.; Kok, F.J. Effects of folic acid supplementation on hearing in older adults: A randomized, controlled trial. Ann. Intern. Med. 2007, 146, 1–9. [Google Scholar] [CrossRef]
- Curhan, S.G.; Eavey, R.D.; Wang, M.; Rimm, E.B.; Curhan, G.C. Fish and fatty acid consumption and the risk of hearing loss in women. Am. J. Clin. Nutr. 2014, 100, 1371–1377. [Google Scholar] [CrossRef]
- Péneau, S.; Jeandel, C.; Déjardin, P.; Andreeva, V.A.; Hercberg, S.; Galan, P.; Kesse-Guyot, E. Intake of specific nutrients and foods and hearing level measured 13 years later. Br. J. Nutr. 2013, 109, 2079–2088. [Google Scholar] [CrossRef]
- Church, M.W.; Jen, K.L.; Anumba, J.I.; Jackson, D.A.; Adams, B.R.; Hotra, J.W. Excess omega-3 fatty acid consumption by mothers during pregnancy and lactation caused shorter life span and abnormal ABRs in old adult offspring. Neurotoxicol. Teratol. 2010, 32, 171–181. [Google Scholar] [CrossRef] [PubMed]
- Piolot, A.; Blache, D.; Boulet, L.; Fortin, L.J.; Dubreuil, D.; Marcoux, C.; Davignon, J.; Lussier-Cacan, S. Effect of fish oil on LDL oxidation and plasma homocysteine concentrations in health. J. Lab. Clin. Med. 2003, 141, 41–49. [Google Scholar] [CrossRef]
- Huang, T.; Zheng, J.; Chen, Y.; Yang, B.; Wahlqvist, M.L.; Li, D. High consumption of Ω-3 polyunsaturated fatty acids decrease plasma homocysteine: A meta-analysis of randomized, placebo-controlled trials. Nutrition 2011, 27, 863–867. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Vega, R.; Partearroyo, T.; Vallecillo, N.; Varela-Moreiras, G.; Pajares, M.A.; Varela-Nieto, I. Long-term omega-3 fatty acid supplementation prevents expression changes in cochlear homocysteine metabolism and ameliorates progressive hearing loss in C57BL/6J mice. J. Nutr. Biochem. 2015, 26, 1424–1433. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Sato, K.; Saito, H. An experimental study of auditory dysfunction associated with hyperlipoproteinemia. Arch Otorhinolaryngol. 1986, 243, 242–245. [Google Scholar] [CrossRef]
- Sikora, M.A.; Morizono, T.; Ward, W.D.; Paparella, M.M.; Leslie, K. Diet-induced hyperlipidemia and auditory dysfunction. Acta Otolaryngol. 1986, 102, 372–381. [Google Scholar] [CrossRef]
- Gopinath, B.; Flood, V.M.; Teber, E.; McMahon, C.M.; Mitchell, P. Dietary intake of cholesterol is positively associated and use of cholesterol-lowering medication is negatively associated with prevalent age-related hearing loss. J. Nutr. 2011, 141, 1355–1361. [Google Scholar] [CrossRef]
- Tokgöz, S.A.; Vuralkan, E.; Sonbay, N.D.; Çalişkan, M.; Saka, C.; Beşalti, Ö.İ.A. Protective effects of vitamins E, B and C and L-carnitine in the prevention of cisplatin-induced ototoxicity in rats. J. Laryngol. Otol. 2012, 126, 464–469. [Google Scholar] [CrossRef]
- McFadden, S.L.; Woo, J.M.; Michalak, N.; Ding, D. Dietary vitamin C supplementation reduces noise-induced hearing loss in guinea pigs. Hear. Res. 2005, 202, 200–208. [Google Scholar] [CrossRef]
- Evans, P.; Halliwell, B. Free radicals and hearing. Cause, consequence, and criteria. Ann. N. Y. Acad. Sci. 1999, 884, 19–40. [Google Scholar] [CrossRef]
- Schafer, F.Q.; Wang, H.P.; Kelley, E.E.; Cueno, K.L.; Martin, S.M.; Buettner, G.R. Comparing beta-carotene, vitamin E and nitric oxide as membrane antioxidants. Biol. Chem. 2002, 383, 671–681. [Google Scholar] [CrossRef]
- Le Prell, C.G.; Hughes, L.F.; Miller, J.M. Free radical scavengers vitamins A, C, and E plus magnesium reduce noise trauma. Free Radic. Biol. Med. 2007, 42, 1454–1463. [Google Scholar] [CrossRef]
- Heman-Ackah, S.E.; Juhn, S.K.; Huang, T.C.; Wiedmann, T.S. A combination antioxidant therapy prevents age-related hearing loss in C57BL/6 mice. Otolaryngol. Head Neck Surg. 2010, 143, 429–434. [Google Scholar] [CrossRef]
- Gopinath, B.; Flood, V.M.; McMahon, C.M.; Burlutsky, G.; Spankovich, C.; Hood, L.J.; Mitchell, P. Dietary antioxidant intake is associated with the prevalence but not incidence of age-related hearing loss. J. Nutr. Health Aging. 2011, 15, 896–900. [Google Scholar] [CrossRef]
- Choi, Y.H.; Miller, J.M.; Tucker, K.L.; Hu, H.; Park, S.K. Antioxidant vitamins and magnesium and the risk of hearing loss in the US general population. Am. J. Clin. Nutr. 2014, 99, 148–155. [Google Scholar] [CrossRef]
- Green, K.L.; Swiderski, D.L.; Prieskorn, D.M.; DeRemer, S.J.; Beyer, L.A.; Miller, J.M.; Green, G.E.; Raphael, Y. ACEMg Diet Supplement Modifies Progression of Hereditary Deafness. Sci. Rep. W. 2016, 6. [Google Scholar] [CrossRef]
- Cohen-Salmon, M.; Ott, T.; Michel, V.; Hardelin, J.P.; Perfettini, I.; Eybalin, M.; Wu, T.; Marcus, D.C.; Wangemann, P.; Willecke, K.; et al. Targeted ablation of connexin26 in the inner ear epithelial gap junction network causes hearing impairment and cell death. Curr. Biol. 2002, 12, 1106–1111. [Google Scholar] [CrossRef]
- Thatcher, A.; Le Prell, C.; Miller, J.; Green, G. ACEMg supplementation ameliorates progressive Connexin 26 hearing loss in a child. Int. J. Pediatr. Otorhinolaryngol. 2014, 78, 563–565. [Google Scholar] [CrossRef]
- Kang, J.W.; Choi, H.S.; Kim, K.; Choi, J.Y. Dietary vitamin intake correlates with hearing thresholds in the older population: The Korean National Health and Nutrition Examination Survey. Am. J. Clin. Nutr. 2014, 99, 1407–1413. [Google Scholar] [CrossRef]
- Thomas, D.R. Vitamins in aging, health, and longevity. Clin Interv Aging 2006, 1, 81–91. [Google Scholar] [CrossRef]
- Carpinelli, M.R.; Wise, A.K.; Burt, R.A. Vitamin D-deficient diet rescues hearing loss in Klotho mice. Hear. Res. 2011, 275, 105–109. [Google Scholar] [CrossRef]
- Michikawa, T.; Nishiwaki, Y.; Asakura, K.; Hillebrand, G.; Miyamoto, K.; Ono, M.; Kinjo, Y.; Akiba, S.; Takebayashi, T. Sunlight exposure may be a risk factor of hearing impairment: A community-based study in Japanese older men and women. J. Gerontol. A. Biol. Sci. Med. Sci. 2013, 68, 96–103. [Google Scholar] [CrossRef]
- Hercberg, S.; Galan, P.; Preziosi, P.; Bertrais, S.; Mennen, L.; Malvy, D.; Roussel, A.M.; Favier, A.S.B. The SU.VI.MAX Study: A randomized, placebo-controlled trial of the health effects of antioxidant vitamins and minerals. Arch Intern. Med. 2004, 164, 2335–2342. [Google Scholar] [CrossRef]
- Curhan, S.G.; Stankovic, K.M.; Eavey, R.D.; Wang, M.; Stampfer, M.J.; Curhan, G.C. Carotenoids, vitamin A, vitamin C, vitamin E, and folate and risk of self-reported hearing loss in women. Am. J. Clin. Nutr. 2015, 102, 1167–1175. [Google Scholar] [CrossRef]
- Romand, R.; Dollé, P.; Hashino, E. Retinoid signaling in inner ear development. J. Neurobiol. 2006, 66, 687–704. [Google Scholar] [CrossRef]
- Schmitz, J.; West, K.P., Jr.; Khatry, S.K.; Wu, L.; Leclerq, S.C.; Karna, S.L.; Katz, J.; Sommer, A.; Pillion, J.W. Vitamin A supplementation in preschool children and risk of hearing loss as adolescents and young adults in rural Nepal: Randomised trial cohort follow-up study. BMJ 2012, 344. [Google Scholar] [CrossRef]
- Emmett, S.D.; West, K.P., Jr. Gestational vitamin A deficiency: A novel cause of sensorineural hearing loss in the developing world? Med. Hypotheses. 2014, 82, 6–10. [Google Scholar] [CrossRef]
- Green, R.; Allen, L.H.; Bjørke-Monsen, A.L.; Brito, A.; Guéant, J.L.; Miller, J.W.; Molloy, A.M.; Nexo, E.; Stabler, S.; Toh, B.H.; et al. Vitamin B12 deficiency. Nat. Rev. Dis. Primers. 2017, 3. [Google Scholar] [CrossRef]
- Ubbink, J.B.; Vermaak, W.J.; van der Merwe, A.; Becker, P.J. Vitamin B-12, vitamin B-6, and folate nutritional status in men with hyperhomocysteinemia. Am. J. Clin. Nutr. 1993, 57, 47–53. [Google Scholar] [CrossRef]
- Partearroyo, T.; Vallecillo, N.; Pajares, M.A.; Varela-Moreiras, G.I.V.-N. Cochlear Homocysteine Metabolism at the Crossroad of Nutrition and Sensorineural Hearing Loss. Front. Mol. Neurosci. 2017, 10. [Google Scholar] [CrossRef]
- Park, S.; Johnson, M.A.; Shea-Miller, K.; De Chicchis, A.R.; Allen, R.H.; Stabler, S.P. Age-related hearing loss, methylmalonic acid, and vitamin B12 status in older adults. J. Nutr. Elder. 2006, 25, 105–120. [Google Scholar] [CrossRef]
- Berkiten, G.; Yildirim, G.; Topaloglu, I.; Ugras, H. Vitamin B12 levels in patients with tinnitus and effectiveness of vitamin B12 treatment on hearing threshold and tinnitus. B-ENT. 2013, 9, 111–116. [Google Scholar]
- Kundu, S.M.C.; Tyagi, N.; Sen, U.; Tyagi, A.C.; Tyagi, S.C. Folic acid improves inner ear vascularization in hyperhomocysteinemic mice. Hear. Res. 2012, 284, 42–51. [Google Scholar] [CrossRef]
- Martínez-Vega, R.; Garrido, F.; Partearroyo, T.; Cediel, R.; Zeisel, S.H.; Martínez-Álvarez, C.; Varela-Moreiras, G.; Varela-Nieto, I.; Pajares, M.A. Folic acid deficiency induces premature hearing loss through mechanisms involving cochlear oxidative stress and impairment of homocysteine metabolism. FASEB J. 2015, 29, 418–432. [Google Scholar] [CrossRef]
- Martínez-Vega, R.; Murillo-Cuesta, S.; Partearroyo, T.; Varela-Moreiras, G.; Varela-Nieto, I.; Pajares, M.A. Long-Term Dietary Folate Deficiency Accelerates Progressive Hearing Loss on CBA/Ca Mice. Front. Aging Neurosci. 2016, 8. [Google Scholar] [CrossRef]
- Manche, S.K.; Jangala, M.; Dudekula, D.; Koralla, M.; Akka, J. Polymorphisms in folate metabolism genes are associated with susceptibility to presbycusis. Life Sci. 2018, 196, 77–83. [Google Scholar] [CrossRef]
- Houston, D.K.; Johnson, M.A.; Nozza, R.J.; Gunter, E.W.; Shea, K.J.; Cutler, G.M.; Edmonds, J.T. Age-related hearing loss, vitamin B-12, and folate in elderly women. Am. J. Clin. Nutr. 1999, 69, 564–571. [Google Scholar] [CrossRef]
- Berner, B.; Odum, L.; Parving, A. Age-related hearing impairment and B vitamin status. Acta Otolaryngol. 2000, 120, 633–637. [Google Scholar] [CrossRef]
- Gopinath, B.; Flood, V.M.; Rochtchina, E.; McMahon, C.M.; Mitchell, P. Serum homocysteine and folate concentrations are associated with prevalent age-related hearing loss. J. Nutr. 2010, 140, 1469–1474. [Google Scholar] [CrossRef]
- Shargorodsky, J.; Curhan, S.G.; Eavey, R.; Curhan, G.C. A prospective study of vitamin intake and the risk of hearing loss in men. Otolaryngol. Head Neck Surg. 2010, 142, 231–236. [Google Scholar] [CrossRef]
- Kabagambe, E.K.; Lipworth, L.; Labadie, R.F.; Hood, L.J.; Francis, D.O. Erythrocyte folate, serum vitamin B12, and hearing loss in the 2003-2004 National Health And Nutrition Examination Survey (NHANES). Eur. J. Clin. Nutr. 2018, 72, 720–727. [Google Scholar] [CrossRef]
- Shah, T.; Joshi, K.; Mishra, S.; Otiv, S.; Kumbar, V. Molecular and cellular effects of vitamin B12 forms on human trophoblast cells in presence of excessive folate. Biomed. Pharmacother. 2016, 84, 526–534. [Google Scholar] [CrossRef]
- Brown, K.D.; Maqsood, S.; Huang, J.Y.; Pan, Y.; Harkcom, W.; Li, W.; Sauve, A.; Verdin, E.; Jaffrey, S.R. Activation of SIRT3 by the NAD⁺ precursor nicotinamide riboside protects from noise-induced hearing loss. Cell Metab. 2014, 20, 1059–1068. [Google Scholar] [CrossRef]
- Yu, F.; Hao, S.; Zhao, Y.; Ren, Y.; Yang, J.; Sun, X.; Chen, J. Mild maternal iron deficiency anemia induces DPOAE suppression and cochlear hair cell apoptosis by caspase activation in young guinea pigs. Environ. Toxicol. Pharmacol 2014, 37, 291–299. [Google Scholar] [CrossRef]
- Chung, S.D.; Chen, P.Y.; Lin, H.C.; Hung, S.H. Sudden sensorineural hearing loss associated with iron-deficiency anemia: A population-based study. JAMA Otolaryngol. Head Neck Surg. 2014, 140, 417–422. [Google Scholar] [CrossRef]
- Wang, Y.Y.; Yang, S.H. Improvement in hearing among otherwise normal schoolchildren in iodine-deficient areas of Guizhou, China, following use of iodized salt. Lancet 1985, 2, 518–520. [Google Scholar]
- Azizi, F.; Kalani, H.; Kimiagar, M.; Ghazi, A.; Sarshar, A.; Nafarabadi, M.; Rahbar, N.; Noohi, S.; Mohajer, M.; Yassai, M. Physical, neuromotor and intellectual impairment in non-cretinous schoolchildren with iodine deficiency. Int. J. Vitam. Nutr. Res. 1995, 65, 199–205. [Google Scholar]
- Schieffer, K.M.; Connor, J.R.; Pawelczyk, J.A.; Sekhar, D.L. The Relationship between Iron Deficiency Anemia and Sensorineural Hearing Loss in the Pediatric and Adolescent Population. Am. J. Audiol. 2017, 26, 155–162. [Google Scholar] [CrossRef]
- Lemajić-Komazec, S.; Abenavoli, L. Iron deficiency anemia and hearing loss. Int. J. Pediatr. Otorhinolaryngol. 2017, 01, S655–S876. [Google Scholar] [CrossRef]
- Kamel, T.B.; Deraz, T.E.; Elkabarity, R.H.; Ahmed, R.K. Protein energy malnutrition associates with different types of hearing impairments in toddlers: Anemia increases cochlear dysfunction. Int. J. Pediatr. Otorhinolaryngol. 2016, 85, 27–31. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization (WHO). The Global Prevalence of Anaemia in 2011; World Health Organization: Geneva, Switzerland, 2015. [Google Scholar]
- Schieffer, K.M.; Chuang, C.H.; Connor, J.; Pawelczyk, J.A.; Sekhar, D.L. Association of Iron Deficiency Anemia With Hearing Loss in US Adults. JAMA Otolaryngol. Head Neck Surg. 2017, 143, 350–354. [Google Scholar] [CrossRef] [PubMed]
- Sun, A.H.; Xiao, S.Z.; Li, B.S.; Li, Z.J.; Wang, T.Y.; Zhang, Y.S. Iron deficiency and hearing loss. Experimental study in growing rats. ORL J. Otorhinolaryngol. Relat. Spec. 1987, 49, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Valeix, P.; Preziosi, P.; Rossignol, C.; Farnier, M.A.; Hercberg, S. Relationship between urinary iodine concentration and hearing capacity in children. Eur. J. Clin. Nutr. 1994, 48, 54–59. [Google Scholar] [PubMed]
- Melse-Boonstra, A.; Mackenzie, I. Iodine deficiency, thyroid function and hearing deficit: A review. Nutr. Res. Rev. 2013, 26, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Azizi, F.; Sarshar, A.; Nafarabadi, M.; Ghazi, A.; Kimiagar, M.; Noohi, S.; Rahbar, N.; Bahrami, A.; Kalantari, S. Impairment of neuromotor and cognitive development in iodine-deficient schoolchildren with normal physical growth. Acta Endocrinol. (Copenh) 1993, 129, 501–504. [Google Scholar] [CrossRef]
- Spankovich, C.; Le Prell, C.G. Healthy diets, healthy hearing: National Health and Nutrition Examination Survey, 1999–2002. Int. J. Audiol. 2013, 52, 369–376. [Google Scholar] [CrossRef]
- Bielefeld, E.C.; Tanaka, C.; Chen, G.D.; Henderson, D. Age-related hearing loss: Is it a preventable condition? Hear. Res. 2010, 264, 98–107. [Google Scholar] [CrossRef]
- Seidman, M.; Babu, S.; Tang, W.; Naem, E.; Quirk, W.S. Effects of resveratrol on acoustic trauma. Otolaryngol. Head Neck Surg. 2003, 129, 463–470. [Google Scholar] [CrossRef]
- Hanci, D.; Ulusoy, S.; Acar, M.; Burukoglu, D.; Kutlu, H.M.; Bayar, M.N.; Cingi, C. Potential protective effect of resveratrol on acoustic trauma: Electron microscopy study. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 3469–3475. [Google Scholar]
- Simşek, G.; Tokgoz, S.A.; Vuralkan, E.; Caliskan, M.; Besalti, O.; Akin, I. Protective effects of resveratrol on cisplatin-dependent inner-ear damage in rats. Eur. Arch Otorhinolaryngol. 2013, 270, 1789–1793. [Google Scholar] [CrossRef] [PubMed]
- Duan, M.; Chen, Z.; Qiu, J.; Ulfendahl, M.; Laurell, G.; Borg, E.; Ruan, R. Low-dose, long-term caroverine administration attenuates impulse noise-induced hearing loss in the rat. Acta Otolaryngol. 2006, 126, 1140–1147. [Google Scholar] [CrossRef] [PubMed]
- Coleman, J.K.; Kopke, R.D.; Liu, J.; Ge, X.; Harper, E.A.; Jones, G.E.; Cater, T.L.; Jackson, R.L. Pharmacological rescue of noise induced hearing loss using N-acetylcysteine and acetyl-L-carnitine. Hear. Res. 2007, 226, 104–113. [Google Scholar] [CrossRef] [PubMed]
- García-Alcántara, F.; Murillo-Cuesta, S.; Pulido, S.; Bermúdez-Muñoz, J.M.; Martínez-Vega, R.; Milo, M.; Varela-Nieto, I.; Rivera, T. The expression of oxidative stress response genes is modulated by a combination of resveratrol and N-acetylcysteine to ameliorate ototoxicity in the rat cochlea. Hear. Res. 2017, 358, 10–21. [Google Scholar] [CrossRef] [PubMed]
- De Rivera, C.; Shukitt-Hale, B.; Joseph, J.A.; Mendelson, J.R.W. The effects of antioxidants in the senescent auditory cortex. Neurobiol. Aging. 2006, 27, 1035–1044. [Google Scholar] [CrossRef] [PubMed]
| Animal Model | Effect | Author and Year |
|---|---|---|
| Sprague–Dawley albino rats | Maintenance of the auditory reflex and cellular integrity of the stria vascularis | Mannström et al., 2013 [57] |
| C57BL/6J mice | Cochlear degeneration reduction and Sirt3 induction in the cochlea | Someya et al., 2007 [60] |
| Rhesus monkeys | Lower ABR threshold compared than control | Fowler et al., 2002; Ramsey et al., 2000 [62,63] |
| Rhesus monkeys | No differences with control | Torre et al., 2004 [64] |
| Macronutrient | Study Subjects/Animal Model | Effect | Author and Year |
|---|---|---|---|
| Carbohydrates | Adults (humans) | High glycemic index, glycemic load and overall total carbohydrate levels increase HL risk | Gopinath et al., 2010 [68] |
| Proteins | C57BL/6J wild-type mice | High β-Conglycinin consumption prevent the increase of the ABR threshold | Tanigawa et al., 2015 [70] |
| Lipids | Wistar rats | Animals with high ω-3 consumption suffer from permanent raises in ABR thresholds | Church et al., 2010 [80] |
| C57BL/6J mice | Preventive effect of the ω-3-supplemented diet in the loss of hearing acuity | Martinez-Vega et al., 2015 [83] | |
| Guinea pigs and chinchillas | High dietary saturated and monounsaturated fats and cholesterol intake increase hearing threshold | Saito et al., 1986; Sikora et al., 1986 [84,85] | |
| Adults (humans) | Significant inverse associated between dietary total long chain ω-3 in plasma and the prevalence of HL | Gopinath et al., 2010 [21] | |
| Negative association between regular ingestion of fish and 5-years incidence and progression of HL | |||
| Adults (humans) | Two or more fish servings consumption per week reduced HL | Curhan et al., 2014 [78] | |
| High long-chain ω-3 intake was inversely associated with HL risk | |||
| Adults (humans) | No association between fish consumption (with ω-3) and HL | Péneau et al., 2013 [79] | |
| Adults (humans) | High dietary intake of cholesterol was associated with an increased risk SNHL | Gopinath et al., 2011 [86] | |
| Neither dietary fat intake nor serum lipids showed any association with HL incidence |
| Micronutrient | Animal Model | Effect | Author and Year |
|---|---|---|---|
| Vitamin C | Guinea pigs | Supplementation significantly decreased the permanent ABR threshold shift whereas deficiency had no effects on NIHL | McFadden et al., 2005 [88] |
| Vitamins B1, B2, B6, E, C | Wistar albino rats | Vitamins contributed to decrease or prevent cisplatin-induced ototoxicity | Tokgöz SA, et al., 2012 [87] |
| Vitamin A, C, E and magnesium | Guinea pigs | Synergistic effects of vitamins A, C and E with magnesium in the reduction of noise-induced threshold | Le Prell et al., 2007 [91] |
| Dietary antioxidants | C57BL/6 mice | Synergistic effect of the antioxidants decreasing the threshold shift from baseline at all frequencies compared to control group | Heman-Ackah et al., 2010 [92] |
| β-carotene, vitamins C, E, and magnesium | Mice model of hereditary deafness (Gjb2-CKO and Diap3-Tg) | Dietary supplementation slowed down the progression of HL and improved auditory thresholds in Gjb2-CKO mice | Green et al., 2016 [95] |
| HL increase in Diap3-Tg mice compared to control | |||
| Folic acid | Cbs+/− Mice | FA supplementation in hyperhomocysteinemic mice led to improvement of hearing function | Kundu et al., 2012 [112] |
| Folic acid | C57BL/6J mice | HL detected after two-months on a FA deficient diet | Martinez-Vega el al., 2015 [113] |
| Correlation between HL, hyperhomocysteinemia and histological damage in the cochleae | |||
| Folic acid | CBA/J mice | Initial signs of HL detected after 8-months of vitamin deficiency | Martinez-Vega el al., 2016 [114]. |
| Correlation between HL, hyperhomocysteinemia and histological damage in the cochleae | |||
| Nicotinamide riboside | C57BL/6 mice | Nicotinamide riboside administration, twice daily for 5 days before noise exposure and for 48 h thereafter, prevented NIHL | Brown et al., 2014 [122] |
| Iron | Guinea pigs | Higher ABR thresholds (worse acuity) were detected in females fed with iron deficient diet | Jougleux et al., 2011 [30] |
| Iron-deficient pups suffered from SNHL |
| Micronutrient | Study Subjects | Effect | Author and Year |
|---|---|---|---|
| Carotenoid, retinol, thiamine, riboflavin, niacin and vitamin C | Adults | High dietary intake of vitamin C was associated with a better auditory function | Kang et al., 2014 [98] |
| Retinol, niacin and riboflavin consumption showed minor association with HL | |||
| Vitamin D serum concentration associated with worse auditory function | |||
| Retinol, vitamin B12, β-carotene, folate, vitamins B6, C and E | Adults | High retinol and vitamin B12 intake associated with better auditory function in women | Hercberg et al., 2004 [102] |
| No associations found for β-carotene, folate and vitamins B6, C and E | |||
| Carotenoids, FA, vitamins C, A and E | Women | Inverse correlations between carotenoids (β-carotene and β-cryptoxanthin) and folate intakes and risk of acquired HL | Curhan et al., 2015 [103] |
| Direct correlation between high vitamin C intake (from supplements) and risk of HL | |||
| No significant associations for vitamin A, E, and other carotenoids | |||
| Dietary antioxidants | Adults | High vitamin A and E consumption showed inverse associations with HL prevalence | Gopinath et al., 2011 [93] |
| Dietary antioxidants consumed alone or in combination were unable to predict 5-year incidence of ARHL | |||
| Vitamins and minerals | Adults | Dose-dependent trend between all individual nutrients (except vitamin E) and better speech pure tone average | Choi et al.,2014 [94] |
| Synergistic effect of high intakes of β-carotene and vitamin C with magnesium and better pure tone average at high frequencies | |||
| Folic acid and vitamin B12 | Women | Consistent associations between low vitamin B12 and folate levels and ARHL | Houston et al., 1999 [112] |
| Stronger association with folate intake rather than with vitamin B12 ingestion, according to dietary intake | |||
| Folic acid and vitamin B12 | Elderly | Low serum vitamin levels were significantly associated with HL in the high frequencies | Lasisi et al., 2016 [16] |
| Significant correlations, after adjusting for age, in folate but not in B12 | |||
| Folic acid and vitamin B12 | Adults | Non-significant associations between serum folate, vitamin B12, Hcy and HL | Berner et al., 2000 [113] |
| Folic acid and vitamin B12 | Adults | Low serum folate status increased risk of developing HL | Gopinath et al., 2010 [114] |
| Serum vitamin B12 levels had no association with ARHL | |||
| Neither folate nor vitamin B12 showed predictive potential for 5-year incidence of HL | |||
| Folic acid, carotene, vitamins C and E | Men | No prospective associations between vitamin C, E, β-carotene or folate consumption and HL | Shargorodsky et al., 2010 [119] |
| High folate intakes were associated with reduced risk of HL | |||
| Folic acid | Adults | Adequate folate intake is beneficial for hearing | Kabagambe et al., 2018 [116] |
| High folate intake showed non-significant association with an increased risk of HL | |||
| Iron | Children and adolescents | Increased odds of SNHL in volunteers with iron deficiency anaemia | Schieefer et al., 2017 [119] |
| Iron | Children | Negative correlation between hemoglobin levels and auditory function | Kamel et al., 2016 [121] |
| Iodine | Children | More severe HL in children with mild-to-moderate iodine deficiency compared to those with normal iodine levels | Valeix et al., 1994 [123] |
| Iodine | Children | HL prevalences of 44 to 15% in children with iodine deficiency compared to 2% in children with adequate levels | Azizi et al., 1993 [124] |
| Micronutrient | Study Subjects | Effect | Author and Year |
|---|---|---|---|
| Vitamin A | Preschool children | Vitamin A supplementation reduced risk of HL associated with childhood ear infections | Schmitz et al., 2012 [105] |
| Folic acid | Adults | Folic acid (800 µg) supplementation for 3 years slowed the decline of auditory function only at speech frequencies, but not at high frequencies | Durga et al., 2007 [77] |
| β-carotene, vitamins C, E, and magnesium | Boy with Cx26-related HL | Daily supplementation led to no further progression of HL | Thatcher et al., 2014 [97] |
| Vitamin B12 | Adults | Not possible to demonstrate an improvement on hearing status with vitamin B12 supplementation | Park et al., 2006 [110] |
| Iodine | Adults | Prophylaxis with iodine salt (three years) revealed an improvement of average hearing levels, reaching values similar to controls | Wang and Yang.1985 [125] |
| Iodine | Children | Iodine dietary supplementation with ionised salt led towards a lower auditory threshold | Azizi et al.,1993 [126] |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Puga, A.M.; Pajares, M.A.; Varela-Moreiras, G.; Partearroyo, T. Interplay between Nutrition and Hearing Loss: State of Art. Nutrients 2019, 11, 35. https://doi.org/10.3390/nu11010035
Puga AM, Pajares MA, Varela-Moreiras G, Partearroyo T. Interplay between Nutrition and Hearing Loss: State of Art. Nutrients. 2019; 11(1):35. https://doi.org/10.3390/nu11010035
Chicago/Turabian StylePuga, Ana M., María A. Pajares, Gregorio Varela-Moreiras, and Teresa Partearroyo. 2019. "Interplay between Nutrition and Hearing Loss: State of Art" Nutrients 11, no. 1: 35. https://doi.org/10.3390/nu11010035
APA StylePuga, A. M., Pajares, M. A., Varela-Moreiras, G., & Partearroyo, T. (2019). Interplay between Nutrition and Hearing Loss: State of Art. Nutrients, 11(1), 35. https://doi.org/10.3390/nu11010035







