Non-Alcoholic Fatty Liver Disease in Overweight Children: Role of Fructose Intake and Dietary Pattern
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Laboratory Measurements, Blood Pressure, and Abdominal Ultrasound
2.3. Bacterial Endotoxin
2.4. Glucose Metabolism
2.5. Assessment of Dietary Intake and Leisure Time Activities
2.6. Anthropometric Measurements and Socio-Demographic Data
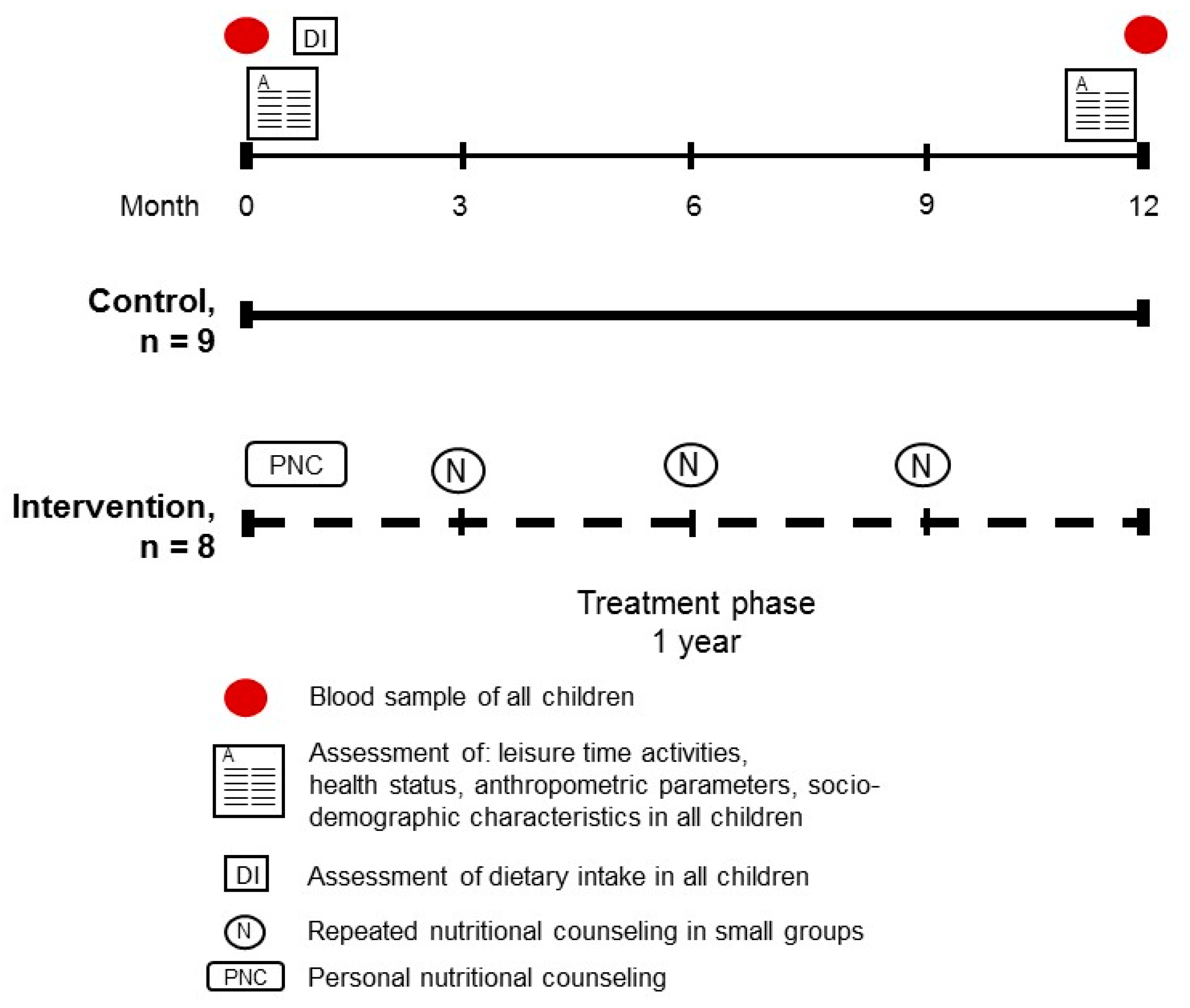
2.7. Dietary Intervention Study
2.8. Statistical Analyses
3. Results
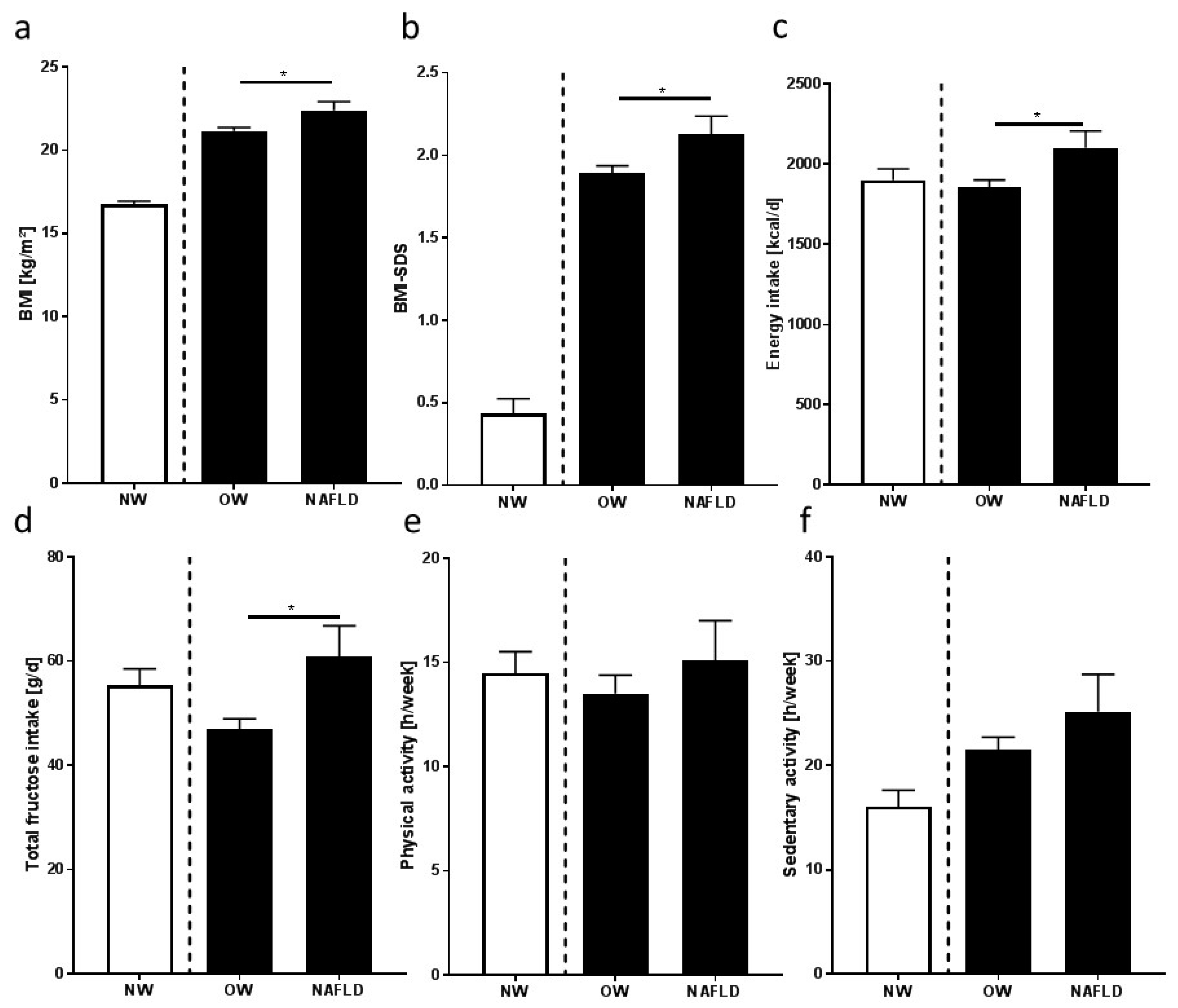
3.1. Characteristics of the Study Participants
3.2. Nutritional Intake, Dietary Pattern, and Leisure Time Activities
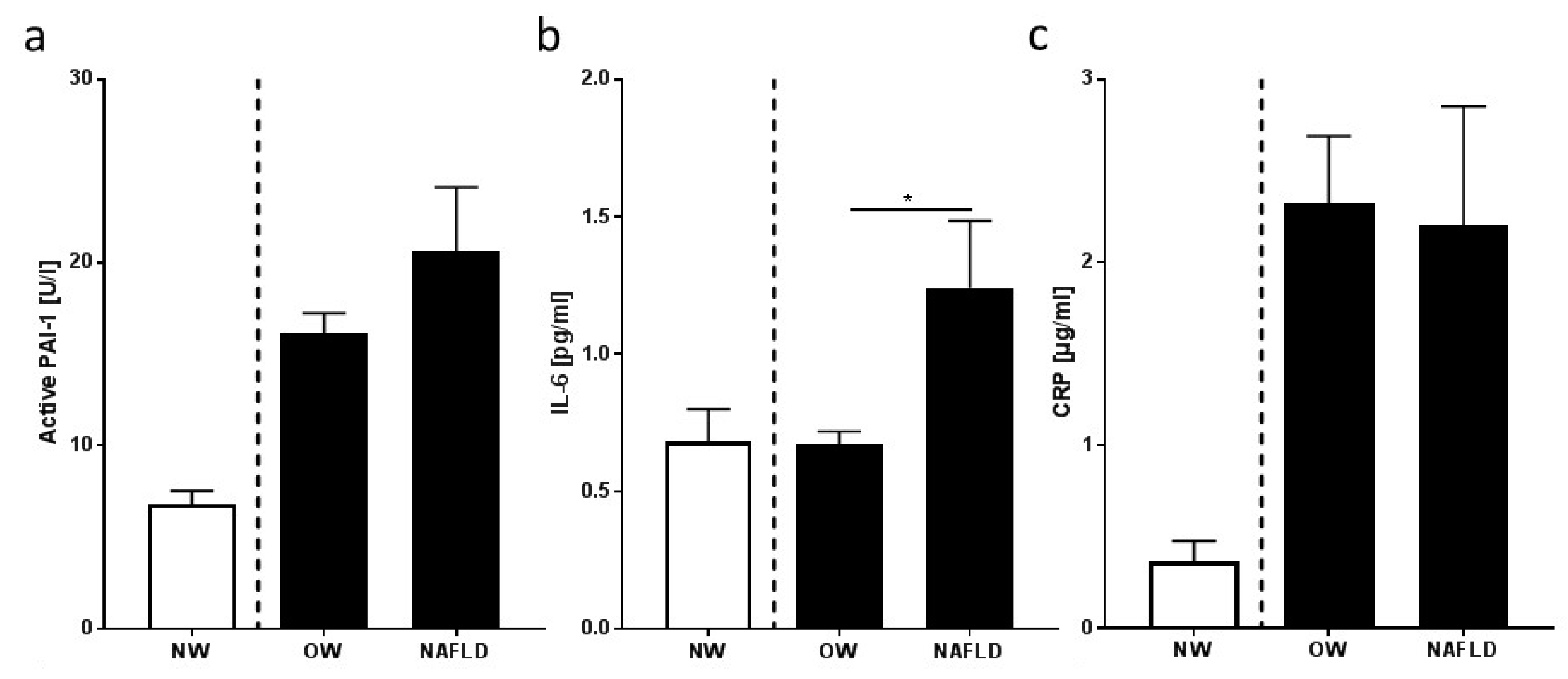
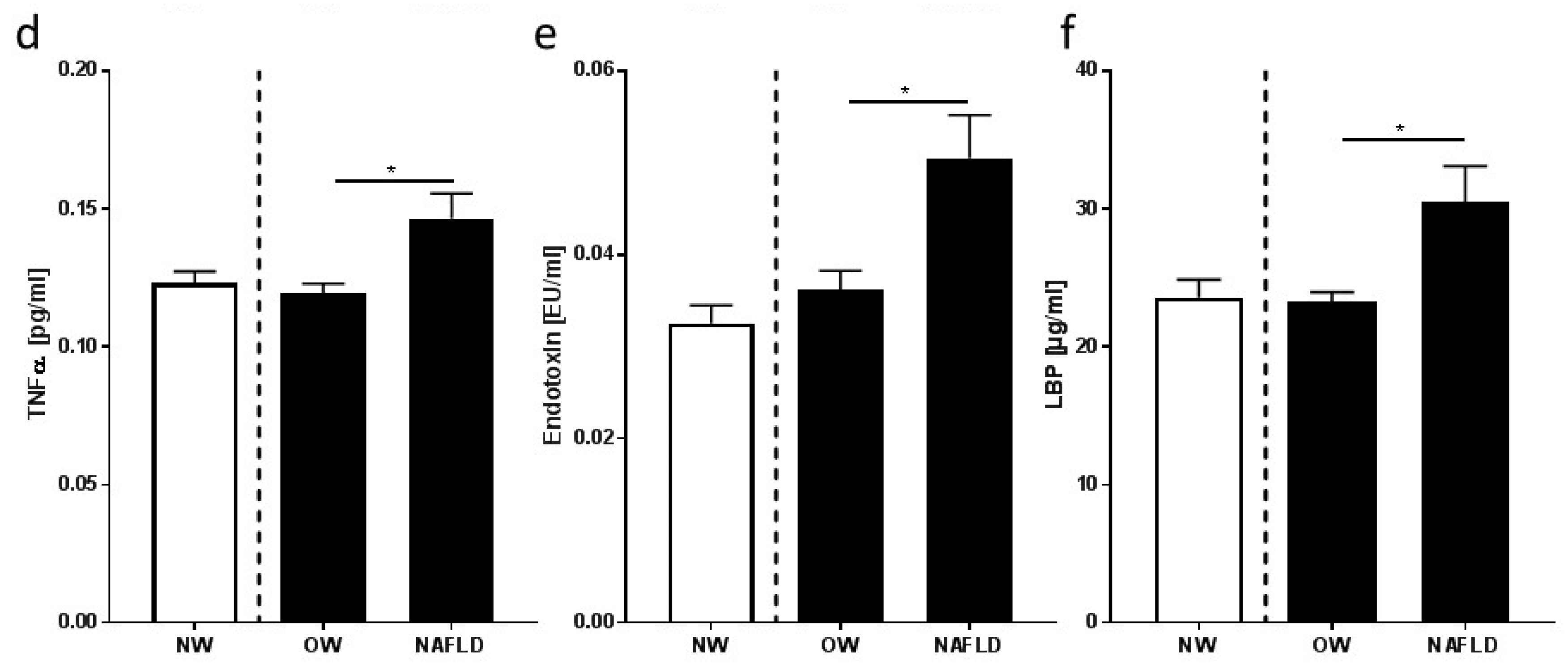
3.3. Markers of Inflammation and Intestinal Permeability
3.4. Feasibility Study: Characteristics of Study Participants, Nutritional Intake, and Metabolic Parameters
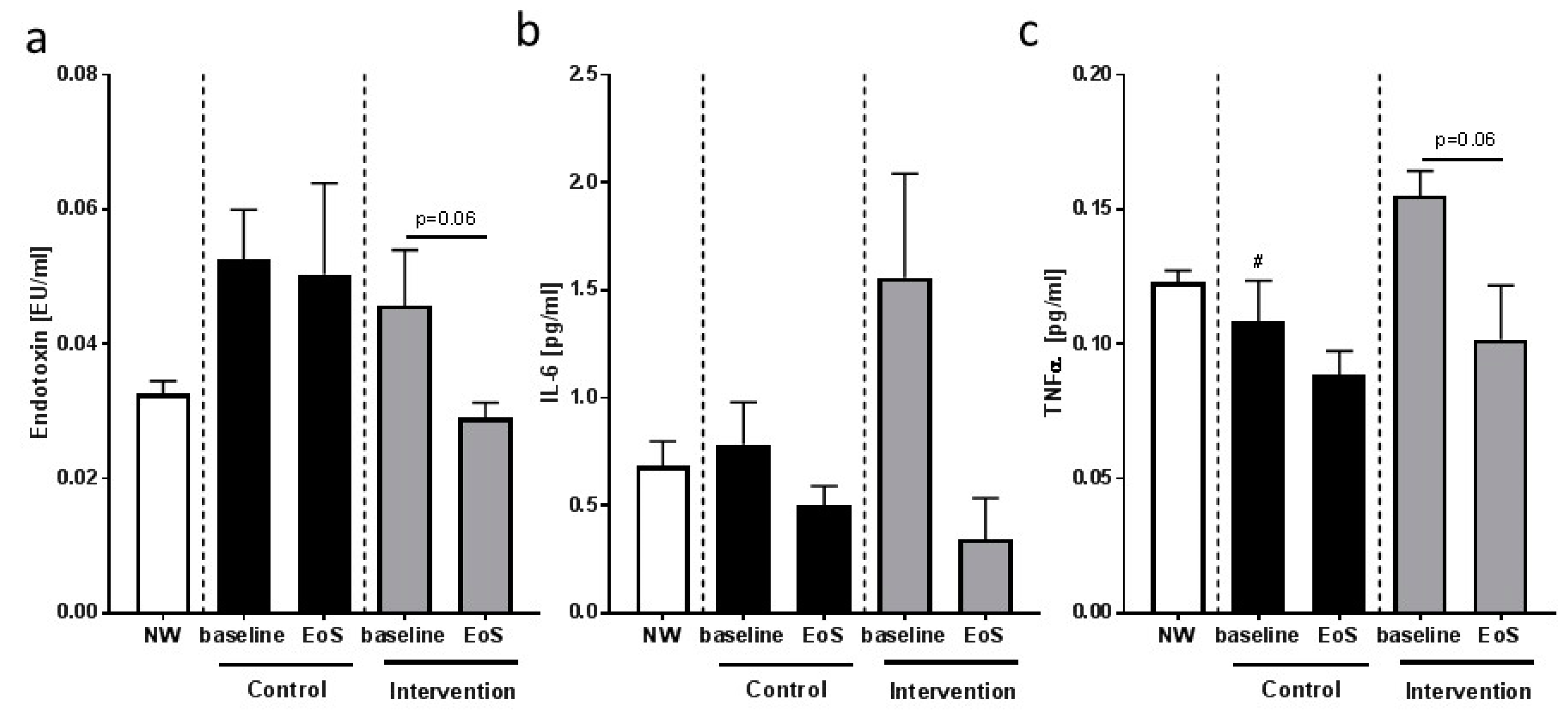
3.5. Feasibility Study: Inflammatory Markers and Indices of Intestinal Permeability
4. Discussion
4.1. Absolute Energy Intake, Nutritional Intake, and Dietary Pattern of Overweight Children with and without NAFLD Differ
4.2. Overweight Children with NAFLD Have Higher Bacterial Endotoxin and LBP Levels in Peripheral Blood Than Overweight Children without NAFLD, Which Were Lowered by Moderate Dietary Counseling
4.3. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Angulo, P.; Lindor, K.D. Non-alcoholic fatty liver disease. J. Gastroenterol. Hepatol. 2002, 17, S186–S190. [Google Scholar] [CrossRef] [PubMed]
- Younossi, Z.; Anstee, Q.M.; Marietti, M.; Hardy, T.; Henry, L.; Eslam, M.; George, J.; Bugianesi, E. Global burden of nafld and nash: Trends, predictions, risk factors and prevention. Nat. Rev. Gastroenterol. Hepatol. 2018, 15, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Alkassabany, Y.M.; Farghaly, A.G.; El-Ghitany, E.M. Prevalence, risk factors, and predictors of nonalcoholic fatty liver disease among schoolchildren: A hospital-based study in alexandria, egypt. Arab. J. Gastroenterol. 2014, 15, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Anderson, E.L.; Howe, L.D.; Jones, H.E.; Higgins, J.P.T.; Lawlor, D.A.; Fraser, A. The prevalence of non-alcoholic fatty liver disease in children and adolescents: A systematic review and meta-analysis. PLoS ONE 2015, 10, e0140908. [Google Scholar] [CrossRef] [PubMed]
- Denzer, C.; Thiere, D.; Muche, R.; Koenig, W.; Mayer, H.; Kratzer, W.; Wabitsch, M. Gender-specific prevalences of fatty liver in obese children and adolescents: Roles of body fat distribution, sex steroids, and insulin resistance. J. Clin. Endocrinol. Metab. 2009, 94, 3872–3881. [Google Scholar] [CrossRef] [PubMed]
- Denzer, C.; Reithofer, E.; Wabitsch, M.; Widhalm, K. The outcome of childhood obesity management depends highly upon patient compliance. Eur. J. Pediatr. 2004, 163, 99–104. [Google Scholar] [CrossRef] [PubMed]
- McManus, K.; Antinoro, L.; Sacks, F. A randomized controlled trial of a moderate-fat, low-energy diet compared with a low fat, low-energy diet for weight loss in overweight adults. Int. J. Obes. Relat. Metab. Disord. 2001, 25, 1503–1511. [Google Scholar] [CrossRef] [PubMed]
- Reinehr, T. Effectiveness of Lifestyle Intervention in Overweight Children. Proc. Nutr. Soc. 2011, 70, 494–505. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, S. Implications of diet on nonalcoholic fatty liver disease. Curr. Opin. Gastroenterol. 2010, 26, 160–164. [Google Scholar] [CrossRef] [PubMed]
- Thuy, S.; Ladurner, R.; Volynets, V.; Wagner, S.; Strahl, S.; Konigsrainer, A.; Maier, K.P.; Bischoff, S.C.; Bergheim, I. Nonalcoholic fatty liver disease in humans is associated with increased plasma endotoxin and plasminogen activator inhibitor 1 concentrations and with fructose intake. J. Nutr. 2008, 138, 1452–1455. [Google Scholar] [CrossRef] [PubMed]
- Toshimitsu, K.; Matsuura, B.; Ohkubo, I.; Niiya, T.; Furukawa, S.; Hiasa, Y.; Kawamura, M.; Ebihara, K.; Onji, M. Dietary habits and nutrient intake in non-alcoholic steatohepatitis. Nutrition 2007, 23, 46–52. [Google Scholar] [CrossRef] [PubMed]
- Volynets, V.; Kuper, M.A.; Strahl, S.; Maier, I.B.; Spruss, A.; Wagnerberger, S.; Konigsrainer, A.; Bischoff, S.C.; Bergheim, I. Nutrition, intestinal permeability, and blood ethanol levels are altered in patients with nonalcoholic fatty liver disease (nafld). Dig. Dis. Sci. 2012, 57, 1932–1941. [Google Scholar] [CrossRef] [PubMed]
- Ackerman, Z.; Oron-Herman, M.; Grozovski, M.; Rosenthal, T.; Pappo, O.; Link, G.; Sela, B.A. Fructose-induced fatty liver disease: Hepatic effects of blood pressure and plasma triglyceride reduction. Hypertension 2005, 45, 1012–1018. [Google Scholar] [CrossRef] [PubMed]
- Bergheim, I.; Weber, S.; Vos, M.; Kramer, S.; Volynets, V.; Kaserouni, S.; McClain, C.J.; Bischoff, S.C. Antibiotics protect against fructose-induced hepatic lipid accumulation in mice: Role of endotoxin. J. Hepatol. 2008, 48, 983–992. [Google Scholar] [CrossRef] [PubMed]
- Kanuri, G.; Spruss, A.; Wagnerberger, S.; Bischoff, S.C.; Bergheim, I. Fructose-induced steatosis in mice: Role of plasminogen activator inhibitor-1, microsomal triglyceride transfer protein and nkt cells. Lab. Investig. 2011, 91, 885–895. [Google Scholar] [CrossRef] [PubMed]
- Spruss, A.; Bergheim, I. Dietary fructose and intestinal barrier: Potential risk factor in the pathogenesis of nonalcoholic fatty liver disease. J. Nutr. Biochem. 2009, 20, 657–662. [Google Scholar] [CrossRef] [PubMed]
- Brandt, A.; Jin, C.J.; Nolte, K.; Sellmann, C.; Engstler, A.J.; Bergheim, I. Short-term intake of a fructose-, fat- and cholesterol-rich diet causes hepatic steatosis in mice: Effect of antibiotic treatment. Nutrients 2017, 9. [Google Scholar] [CrossRef] [PubMed]
- Kanuri, G.; Bergheim, I. In vitro and in vivo models of non-alcoholic fatty liver disease (nafld). Int. J. Mol. Sci. 2013, 14, 11963–11980. [Google Scholar] [CrossRef] [PubMed]
- Sellmann, C.; Priebs, J.; Landmann, M.; Degen, C.; Engstler, A.J.; Jin, C.J.; Garttner, S.; Spruss, A.; Huber, O.; Bergheim, I. Diets rich in fructose, fat or fructose and fat alter intestinal barrier function and lead to the development of nonalcoholic fatty liver disease over time. J. Nutr. Biochem. 2015, 26, 1183–1192. [Google Scholar] [CrossRef] [PubMed]
- Hashemi Kani, A.; Alavian, S.M.; Esmaillzadeh, A.; Adibi, P.; Azadbakht, L. Dietary quality indices and biochemical parameters among patients with non alcoholic fatty liver disease (nafld). Hepat. Mon. 2013, 13, e10943. [Google Scholar] [CrossRef] [PubMed]
- Nier, A.; Engstler, A.J.; Maier, I.B.; Bergheim, I. Markers of intestinal permeability are already altered in early stages of non-alcoholic fatty liver disease: Studies in children. PLoS ONE 2017, 12, e0183282. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Liu, Z.W.; Li, Y.; Gong, C.; Zhang, H.; Song, L.J.; Huang, C.Y.; Li, M. The prevalence of nonalcoholic fatty liver disease and its association with lifestyle/dietary habits among university faculty and staff in chengdu. Biomed. Environ. Sci. 2012, 25, 383–391. [Google Scholar] [CrossRef] [PubMed]
- Engstler, A.J.; Aumiller, T.; Degen, C.; Durr, M.; Weiss, E.; Maier, I.B.; Schattenberg, J.M.; Jin, C.J.; Sellmann, C.; Bergheim, I. Insulin resistance alters hepatic ethanol metabolism: Studies in mice and children with non-alcoholic fatty liver disease. Gut 2016, 65, 1564–1571. [Google Scholar] [CrossRef] [PubMed]
- Marshall, W.A.; Tanner, J.M. Variations in pattern of pubertal changes in girls. Arch. Dis. Child. 1969, 44, 291–303. [Google Scholar] [CrossRef] [PubMed]
- Marshall, W.A.; Tanner, J.M. Variations in the pattern of pubertal changes in boys. Arch. Dis. Child. 1970, 45, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Maier, I.B.; Ozel, Y.; Engstler, A.J.; Puchinger, S.; Wagnerberger, S.; Hulpke-Wette, M.; Bischoff, S.C.; Bergheim, I. Differences in the prevalence of metabolic disorders between prepubertal boys and girls from 5 to 8 years of age. Acta Paediatr. 2014, 103, e154–e160. [Google Scholar] [CrossRef] [PubMed]
- Maier, I.B.; Stricker, L.; Ozel, Y.; Wagnerberger, S.; Bischoff, S.C.; Bergheim, I. A low fructose diet in the treatment of pediatric obesity: A pilot study. Pediatr. Int. 2011, 53, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Maier, I.B.; Ozel, Y.; Wagnerberger, S.; Bischoff, S.C.; Bergheim, I. Dietary pattern and leisure time activity of overweight and normal weight children in germany: Sex-specific differences. Nutr. J. 2013, 12, 14. [Google Scholar] [CrossRef] [PubMed]
- Schofield, W.N. Predicting basal metabolic rate, new standards and review of previous work. Hum. Nutr. Clin. Nutr. 1985, 39 (Suppl. 1), 5–41. [Google Scholar] [PubMed]
- Sichert-Hellert, W.; Kersting, M.; Schoch, G. Underreporting of energy intake in 1 to 18 year old german children and adolescents. Z. Ernahrungswiss. 1998, 37, 242–251. [Google Scholar] [CrossRef] [PubMed]
- DDG. Evidenzbasierte Leitlinie der Deutschen Diabetes-Gesellschaft. Available online: www.deutsche-diabetes-gesellschaft.de (accessed on 20 July 2018).
- Mager, D.R.; Patterson, C.; So, S.; Rogenstein, C.D.; Wykes, L.J.; Roberts, E.A. Dietary and physical activity patterns in children with fatty liver. Eur. J. Clin. Nutr. 2010, 64, 628–635. [Google Scholar] [CrossRef] [PubMed]
- Mosca, A.; Nobili, V.; De Vito, R.; Crudele, A.; Scorletti, E.; Villani, A.; Alisi, A.; Byrne, C.D. Serum uric acid concentrations and fructose consumption are independently associated with nash in children and adolescents. J. Hepatol. 2017, 66, 1031–1036. [Google Scholar] [CrossRef] [PubMed]
- D’Adamo, E.; Impicciatore, M.; Capanna, R.; Loredana Marcovecchio, M.; Masuccio, F.G.; Chiarelli, F.; Mohn, A.A. Liver steatosis in obese prepubertal children: A possible role of insulin resistance. Obesity 2008, 16, 677–683. [Google Scholar] [CrossRef] [PubMed]
- Nath, P.; Singh, S.P. Nonalcoholic fatty liver disease: Time to take the bull by the horns. Eur. J. Hepatol.-Gastroenterol. 2018, 8, 47–51. [Google Scholar] [CrossRef] [PubMed]
- Angin, Y.; Arslan, N.; Kuralay, F. Leptin-to-adiponectin ratio in obese adolescents with nonalcoholic fatty liver disease. Turk. J. Pediatr. 2014, 56, 259–266. [Google Scholar] [PubMed]
- Dhaliwal, J.; Chavhan, G.B.; Lurz, E.; Shalabi, A.; Yuen, N.; Williams, B.; Martincevic, I.; Amirabadi, A.; Wales, P.W.; Lee, W.; et al. Hepatic steatosis is highly prevalent across the paediatric age spectrum, including in pre-school age children. Aliment. Pharmacol. Ther. 2018. [Google Scholar] [CrossRef] [PubMed]
- Papandreou, D.; Karavetian, M.; Karabouta, Z.; Andreou, E. Obese children with metabolic syndrome have 3 times higher risk to have nonalcoholic fatty liver disease compared with those without metabolic syndrome. Int. J. Endocrinol. 2017, 2017, 2671692. [Google Scholar] [CrossRef] [PubMed]
- Prokopowicz, Z.; Malecka-Tendera, E.; Matusik, P. Predictive value of adiposity level, metabolic syndrome, and insulin resistance for the risk of nonalcoholic fatty liver disease diagnosis in obese children. Can. J. Gastroenterol. Hepatol. 2018, 2018, 9465784. [Google Scholar] [CrossRef] [PubMed]
- Alisi, A.; Manco, M.; Devito, R.; Piemonte, F.; Nobili, V. Endotoxin and plasminogen activator inhibitor-1 serum levels associated with nonalcoholic steatohepatitis in children. J. Pediatr. Gastroenterol. Nutr. 2010, 50, 645–649. [Google Scholar] [CrossRef] [PubMed]
- Assuncao, S.N.F.; Sorte, N.; Alves, C.A.D.; Mendes, P.S.A.; Alves, C.R.B.; Silva, L.R. Inflammatory cytokines and non-alcoholic fatty liver disease (nafld) in obese children and adolescents. Nutr. Hosp. 2018, 35, 78–83. [Google Scholar] [CrossRef] [PubMed]
- Estelles, A.; Dalmau, J.; Falco, C.; Berbel, O.; Castello, R.; Espana, F.; Aznar, J. Plasma pai-1 levels in obese children—Effect of weight loss and influence of pai-1 promoter 4g/5g genotype. Thromb. Haemost. 2001, 86, 647–652. [Google Scholar] [PubMed]
- Fitzpatrick, E.; Dew, T.K.; Quaglia, A.; Sherwood, R.A.; Mitry, R.R.; Dhawan, A. Analysis of adipokine concentrations in paediatric non-alcoholic fatty liver disease. Pediatr. Obes. 2012, 7, 471–479. [Google Scholar] [CrossRef] [PubMed]
- Masquio, D.C.; de Piano, A.; Campos, R.M.; Sanches, P.L.; Corgosinho, F.C.; Carnier, J.; Oyama, L.M.; do Nascimento, C.M.; de Mello, M.T.; Tufik, S.; et al. Saturated fatty acid intake can influence increase in plasminogen activator inhibitor-1 in obese adolescents. Horm. Metab. Res. 2014, 46, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.J.; Sellmann, C.; Engstler, A.J.; Ziegenhardt, D.; Bergheim, I. Supplementation of sodium butyrate protects mice from the development of non-alcoholic steatohepatitis (nash). Br. J. Nutr. 2015, 114, 1745–1755. [Google Scholar] [CrossRef] [PubMed]
- Kanuri, G.; Ladurner, R.; Skibovskaya, J.; Spruss, A.; Konigsrainer, A.; Bischoff, S.C.; Bergheim, I. Expression of toll-like receptors 1-5 but not tlr 6-10 is elevated in livers of patients with non-alcoholic fatty liver disease. Liver. Int. 2015, 35, 562–568. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.H.; Choi, D.; Kim, J.Y.; Lee, J.H.; Koo, S.H. Fast food diet-induced non-alcoholic fatty liver disease exerts early protective effect against acetaminophen intoxication in mice. BMC Gastroenterol. 2017, 17, 124. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, J.S.; Le, M.T.; Pan, Z.; Rivard, C.; Love-Osborne, K.; Robbins, K.; Johnson, R.J.; Sokol, R.J.; Sundaram, S.S. Oral fructose absorption in obese children with non-alcoholic fatty liver disease. Pediatr. Obes. 2015, 10, 188–195. [Google Scholar] [CrossRef] [PubMed]
- Abdelmalek, M.F.; Suzuki, A.; Guy, C.; Unalp-Arida, A.; Colvin, R.; Johnson, R.J.; Diehl, A.M.; Clini, N.S. Increased fructose consumption is associated with fibrosis severity in patients with nonalcoholic fatty liver disease. Hepatology 2010, 51, 1961–1971. [Google Scholar] [CrossRef] [PubMed]
- Utz-Melere, M.; Targa-Ferreira, C.; Lessa-Horta, B.; Epifanio, M.; Mouzaki, M.; Mattos, A.A. Non-alcoholic fatty liver disease in children and adolescents: Lifestyle change—A systematic review and meta-analysis. Ann. Hepatol. 2018, 17, 345–354. [Google Scholar] [CrossRef] [PubMed]
- Zelber-Sagi, S.; Nitzan-Kaluski, D.; Goldsmith, R.; Webb, M.; Blendis, L.; Halpern, Z.; Oren, R. Long term nutritional intake and the risk for non-alcoholic fatty liver disease (nafld): A population based study. J. Hepatol. 2007, 47, 711–717. [Google Scholar] [CrossRef] [PubMed]
- Assy, N.; Nasser, G.; Kamayse, I.; Nseir, W.; Beniashvili, Z.; Djibre, A.; Grosovski, M. Soft drink consumption linked with fatty liver in the absence of traditional risk factors. Can. J. Gastroenterol. 2008, 22, 811–816. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, X.; Cirillo, P.; Sautin, Y.; McCall, S.; Bruchette, J.L.; Diehl, A.M.; Johnson, R.J.; Abdelmalek, M.F. Fructose consumption as a risk factor for non-alcoholic fatty liver disease. J. Hepatol. 2008, 48, 993–999. [Google Scholar] [CrossRef] [PubMed]
- Félix, D.R.; Costenaro, F.; Gottschall, C.B.A.; Coral, G.P. Non-alcoholic fatty liver disease (nafld) in obese children- effect of refined carbohydrates in diet. BMC Pediatr. 2016, 16, 187. [Google Scholar] [CrossRef] [PubMed]
- Medrano, M.; Cadenas-Sanchez, C.; Alvarez-Bueno, C.; Cavero-Redondo, I.; Ruiz, J.R.; Ortega, F.B.; Labayen, I. Evidence-based exercise recommendations to reduce hepatic fat content in youth- a systematic review and meta-analysis. Prog. Cardiovasc. Dis. 2018. [Google Scholar] [CrossRef] [PubMed]
- Guercio Nuzio, S.; Di Stasi, M.; Pierri, L.; Troisi, J.; Poeta, M.; Bisogno, A.; Belmonte, F.; Tripodi, M.; Di Salvio, D.; Massa, G.; et al. Multiple gut-liver axis abnormalities in children with obesity with and without hepatic involvement. Pediatr. Obes. 2016. [Google Scholar] [CrossRef]
- Harte, A.L.; da Silva, N.F.; Creely, S.J.; McGee, K.C.; Billyard, T.; Youssef-Elabd, E.M.; Tripathi, G.; Ashour, E.; Abdalla, M.S.; Sharada, H.M.; et al. Elevated endotoxin levels in non-alcoholic fatty liver disease. J. Inflamm. 2010, 7, 15. [Google Scholar] [CrossRef] [PubMed]
- Ouelaa, W.; Jegatheesan, P.; M’Bouyou-Boungou, J.; Vicente, C.; Nakib, S.; Nubret, E.; De Bandt, J.P. Citrulline decreases hepatic endotoxin-induced injury in fructose-induced non-alcoholic liver disease: An ex vivo study in the isolated perfused rat liver. Br. J. Nutr. 2017, 117, 1487–1494. [Google Scholar] [CrossRef] [PubMed]
- Volynets, V.; Machann, J.; Kuper, M.A.; Maier, I.B.; Spruss, A.; Konigsrainer, A.; Bischoff, S.C.; Bergheim, I. A moderate weight reduction through dietary intervention decreases hepatic fat content in patients with non-alcoholic fatty liver disease (nafld): A pilot study. Eur. J. Nutr. 2013, 52, 527–535. [Google Scholar] [CrossRef] [PubMed]
- Murphy, M.J.; Hosking, J.; Metcalf, B.S.; Voss, L.D.; Jeffery, A.N.; Sattar, N.; Williams, R.; Jeffery, J.; Wilkin, T.J. Distribution of adiponectin, leptin, and metabolic correlates of insulin resistance: A longitudinal study in british children; 1: Prepuberty (earlybird 15). Clin. Chem. 2008, 54, 1298–1306. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.K.; Roberts, C.K.; Barnard, R.J. Effect of a short-term diet and exercise intervention on metabolic syndrome in overweight children. Metabolism 2006, 55, 871–878. [Google Scholar] [CrossRef] [PubMed]
| NW | OW | NAFLD | |
|---|---|---|---|
| n | 36 | 73 | 16 |
| Sex (male/female) | 20/16 | 33/40 | 6/10 |
| Ethnicity (Caucasian/Asian) | 27/9 | 54/19 | 8/8 |
| Age (years) | 7.3 ± 0.2 | 7.6 ± 0.1 | 7.8 ± 0.3 |
| Weight (kg) | 27 ± 1 | 36 ± 1 | 40 ± 2 * |
| Height (cm) | 1.26 ± 0.01 | 1.29 ± 0.01 | 1.33 ± 0.02 |
| BMI (kg/m2) | 16.7 ± 0.2 | 21.2 ± 0.2 | 22.4 ± 0.5 * |
| BMI-SD score | 0.43 ± 0.09 | 1.90 ± 0.05 | 2.13 ± 0.11 * |
| Waist circumference (cm) | 59 ± 1 | 72 ± 1 | 77 ± 2 * |
| Leptin (ng/mL) | 2.3 ± 0.3 | 12.2 ± 1.0 | 9.2 ± 1.4 |
| Adiponectin (µg/mL) | 11.4 ± 0.9 | 10.4 ± 0.5 | 11.9 ± 2.2 |
| Leptin/Adiponectin | 0.3 ± 0.03 | 1.46 ± 0.17 | 1.21 ± 0.26 |
| ALT (U/L) | 19 ± 1 | 23 ± 1 | 24 ± 2 |
| AST (U/L) | 33 ± 1 | 31 ± 1 | 35 ± 2 * |
| Systolic blood pressure (mmHg) | 103 ± 1 | 108 ± 1 | 111 ± 3 |
| Diastolic blood pressure (mmHg) | 62 ± 1 | 67 ± 1 | 70 ± 2 |
| Triglycerides (mg/dL) | 57 ± 3 | 81 ± 4 | 75 ± 6 |
| HDL cholesterol (mg/dL) | 57 ± 1 | 52 ± 1 | 48 ± 3 |
| LDL cholesterol (mg/dL) | 100 ± 3 | 117 ± 2 | 105 ± 6 * |
| Total cholesterol (mg/dL) | 170 ± 4 | 185 ± 3 | 169 ± 6 * |
| Uric Acid (mg/dL) | 3.6 ± 0.1 | 4.3 ± 0.1 | 4.1 ± 0.2 |
| Insulin (µIU/mL) | 9 ± 0.4 | 12 ± 0.6 | 16 ± 2.3 |
| Fasting glucose (mg/dL) | 85 ± 1 | 87 ± 1 | 85 ± 2 |
| HOMA-IR | 1.9 ± 0.1 | 2.5 ± 0.1 | 2.9 ± 0.4 |
| Physical activity (h/week) | 15 ± 1 | 14 ± 1 | 15 ± 2 |
| Sedentary activity (h/week) | 16 ± 2 | 22 ± 1 | 25 ± 4 |
| NW | OW | NAFLD | |
|---|---|---|---|
| n | 36 | 73 | 16 |
| Total energy intake (kcal/day) | 1900 ± 70 | 1853 ± 47 | 2101 ± 105 * |
| Total fat intake (g/day) | 78 ± 4 | 77 ± 3 | 86 ± 6 |
| Total protein intake (g/day) | 59 ± 3 | 63 ± 2 | 68 ± 4 |
| Total CHO intake (g/day) | 242 ± 10 | 227 ± 7 | 257 ± 16 |
| Fructose (g/day) a | 55 ± 3 | 47 ± 2 | 61 ± 6 * |
| Glucose (g/day) b | 49 ± 2 | 42 ± 2 | 53 ± 6 * |
| Fiber intake (g/day) | 15 ± 1 | 14 ± 1 | 18 ± 2 |
| NW | OW | NAFLD | |
|---|---|---|---|
| Beverages (kcal/day) | 161 ± 18 | 132 ± 10 | 208 ± 35 * |
| Fruits/dried fruits (kcal/day) | 86 ± 12 | 80 ± 7 | 130 ± 30 |
| Vegetables/legumes (kcal/day) | 27 ± 5 | 30 ± 4 | 24 ± 5 |
| Potatoes/pasta/rice (kcal/day) | 210 ± 33 | 170 ± 14 | 201 ± 36 |
| Bread (kcal/day) | 259 ± 22 | 257 ± 18 | 262 ± 48 |
| Spreads (kcal/day) | 141 ± 23 | 103 ± 10 | 84 ± 18 |
| Bakery goods (kcal/day) | 206 ± 28 | 217 ± 27 | 167 ± 33 |
| Cereals (kcal/day) | 127 ± 35 | 113 ± 12 | 146 ± 5 |
| Meat (kcal/day) | 216 ± 24 | 205 ± 18 | 249 ± 46 |
| Milk and dairy (kcal/day) | 161 ± 16 | 144 ± 11 | 168 ± 37 |
| Cheese and quark (kcal/day) | 92 ± 15 | 104 ± 11 | 77 ± 12 |
| Oils, margarines, and butter (kcal/day) | 94 ± 12 | 103 ± 9 | 136 ± 15 |
| Sweets and sugar (kcal/day) | 154 ± 26 | 178 ± 15 | 145 ± 17 |
| Desserts (kcal/day) | 157 ± 38 | 111 ± 21 | 173 ± 80 |
| Convenience food (kcal/day) | 231 ± 28 | 324 ± 37 | 317 ± 63 |
| Healthy Children | NAFLD Children | ||||
|---|---|---|---|---|---|
| NW (n = 36) | Control (n = 6) | Intervention (n = 7) | |||
| Baseline | After 1 Year | Baseline | After 1 Year | ||
| Sex (male/female) | 20/16 | 3/3 | 4/2 | ||
| Ethnicity (Caucasian/Asian) | 27/9 | 2/4 | 5/1 | ||
| Age (years) | 7.3 ± 0.2 | 8.0 ± 0.3 | 9.0 ± 0.5 * | 7.5 ± 0.4 | 8.7 ± 0.4 * |
| BMI-SD score | 0.43 ± 0.09 | 1.9 ± 0.1 | 1.6 ± 0.2 * | 2.2 ± 0.2 | 2.0 ± 0.3 |
| Waist circumference (cm) | 59 ± 1 | 77 ± 4 | 77 ± 4 | 76 ± 3 | 83 ± 3 * |
| Leptin (ng/mL) | 2.3 ± 0.3 | 9.5 ± 3.0 | 16.6 ± 4.8 | 8.8 ± 2.5 | 17.5 ± 5.7 |
| Adiponectin (µg/mL) | 11.4 ± 0.9 | 7.6 ± 1.4 | 6.9 ± 0.7 | 18.5 ± 4.3 | 8.3 ± 1.9 |
| Leptin/adiponectin | 0.3 ± 0.03 | 1.6 ± 0.6 | 2.5 ± 0.8 | 0.7 ± 0.3 | 2.0 ± 0.3 * |
| LBP (µg/mL) | 23.6 ± 1.3 | 28.3 ± 4.0 | 30.2 ± 3.2 | 28.8 ± 4.5 | 25.6 ± 2.6 |
| Active PAI-1 (U/L) | 6.8 ± 0.8 | 18.2 ± 5.6 | 18.2 ± 8.6 | 17.0 ± 4.0 | 9.6 ± 1.4 |
| ALT (U/L) | 19 ± 1 | 26 ± 4 | 26 ± 3 | 20 ± 1 | 23 ± 3 |
| AST (U/L) | 33 ± 1 | 38 ± 5 | 30 ± 2 | 32 ± 3 | 33 ± 3 |
| Systolic blood pressure (mmHg) | 103 ± 1 | 111 ± 5 | 113 ± 5 | 107 ± 4 | 105 ± 5 |
| Diastolic blood pressure (mmHg) | 62 ± 1 | 69 ± 3 | 70 ± 6 | 69 ± 4 | 65 ± 4 * |
| Triglycerides (mg/dL) | 57 ± 3 | 80 ± 7 | 85 ± 13 | 74 ± 15 | 89 ± 27 |
| HDL cholesterol (mg/dL) | 57 ± 1 | 48 ± 3 | 52 ± 4 | 56 ± 4 | 57 ± 5 |
| LDL cholesterol (mg/dL) | 100 ± 3 | 109 ± 8 | 115 ± 10 | 102 ± 13 | 116 ± 11 * |
| Total cholesterol (mg/dL) | 160 ± 4 | 176 ± 7 | 174 ± 12 | 170 ± 12 | 187 ± 10 * |
| Insulin (µIU/mL) | 9 ± 0.4 | 13 ± 1 | 15 ± 3 | 13 ± 4 | 16 ± 4 |
| Fasting glucose (mg/dL) | 85 ± 1 | 78 ± 4 | 86 ± 3 | 85 ± 2 | 86 ± 3 |
| HOMA-IR | 1.9 ± 0.1 | 2.5 ± 0.3 | 3.3 ± 0.8 | 2.8 ± 0.9 | 3.4 ± 0.9 |
| Physical activity (h/week) | 15 ± 1 | 17 ± 4 | 17 ± 5 | 14 ± 2 | 14 ± 2 |
| Sedentary activity (h/week) | 16 ± 2 | 23 ± 7 | 25 ± 4 | 24 ± 4 | 18 ± 4 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nier, A.; Brandt, A.; Conzelmann, I.B.; Özel, Y.; Bergheim, I. Non-Alcoholic Fatty Liver Disease in Overweight Children: Role of Fructose Intake and Dietary Pattern. Nutrients 2018, 10, 1329. https://doi.org/10.3390/nu10091329
Nier A, Brandt A, Conzelmann IB, Özel Y, Bergheim I. Non-Alcoholic Fatty Liver Disease in Overweight Children: Role of Fructose Intake and Dietary Pattern. Nutrients. 2018; 10(9):1329. https://doi.org/10.3390/nu10091329
Chicago/Turabian StyleNier, Anika, Annette Brandt, Ina Barbara Conzelmann, Yelda Özel, and Ina Bergheim. 2018. "Non-Alcoholic Fatty Liver Disease in Overweight Children: Role of Fructose Intake and Dietary Pattern" Nutrients 10, no. 9: 1329. https://doi.org/10.3390/nu10091329
APA StyleNier, A., Brandt, A., Conzelmann, I. B., Özel, Y., & Bergheim, I. (2018). Non-Alcoholic Fatty Liver Disease in Overweight Children: Role of Fructose Intake and Dietary Pattern. Nutrients, 10(9), 1329. https://doi.org/10.3390/nu10091329





